Proteasome as a Molecular Target of Microcystin-LR
Abstract
:1. Introduction
2. Results and Discussion
2.1. MC-LR Selectively Inhibited Proteasome TL Activity

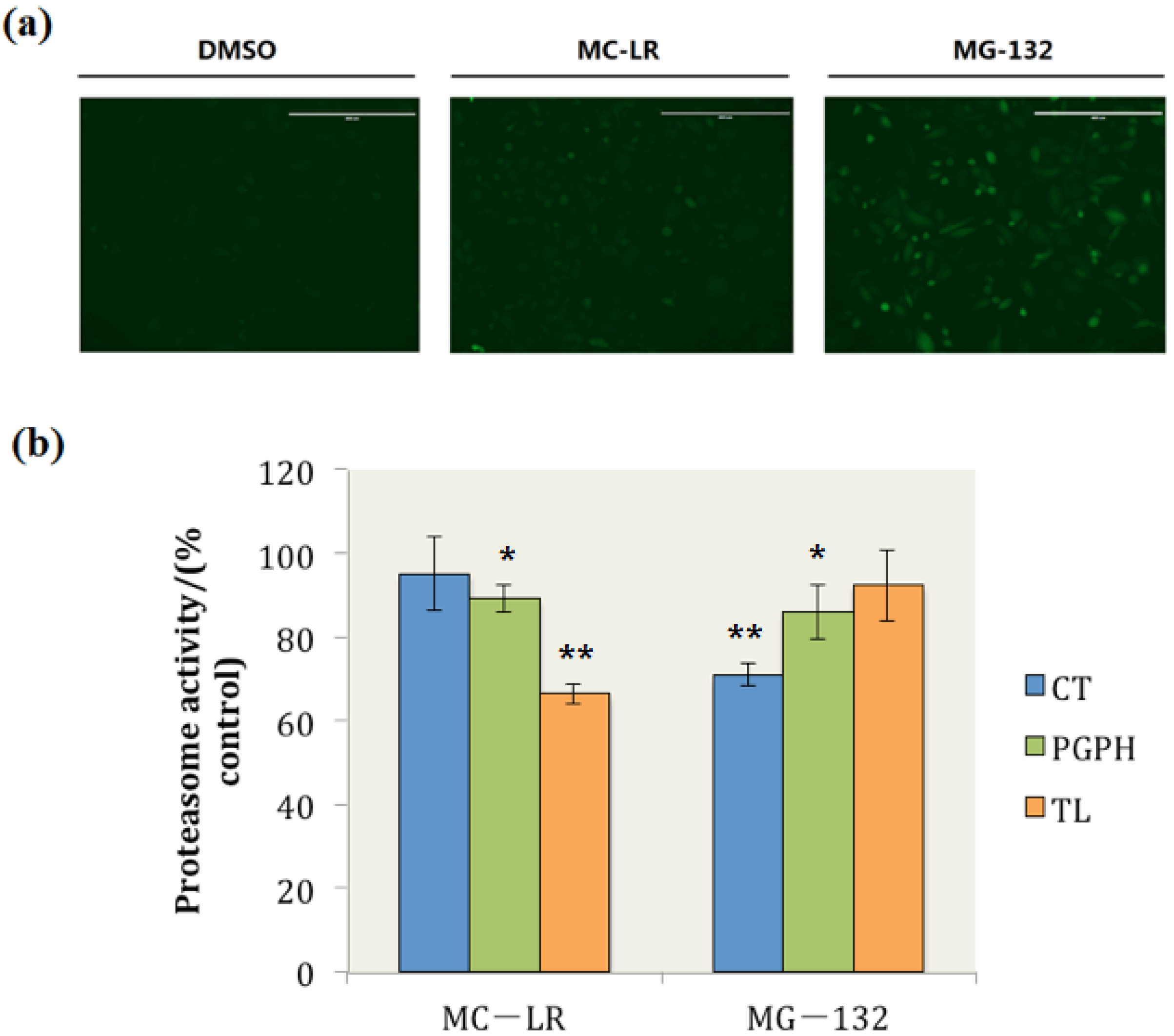
2.2. MC-LR Exposure Retarded the Degradation of Green Fluorescent Protein (GFP) by Proteasome
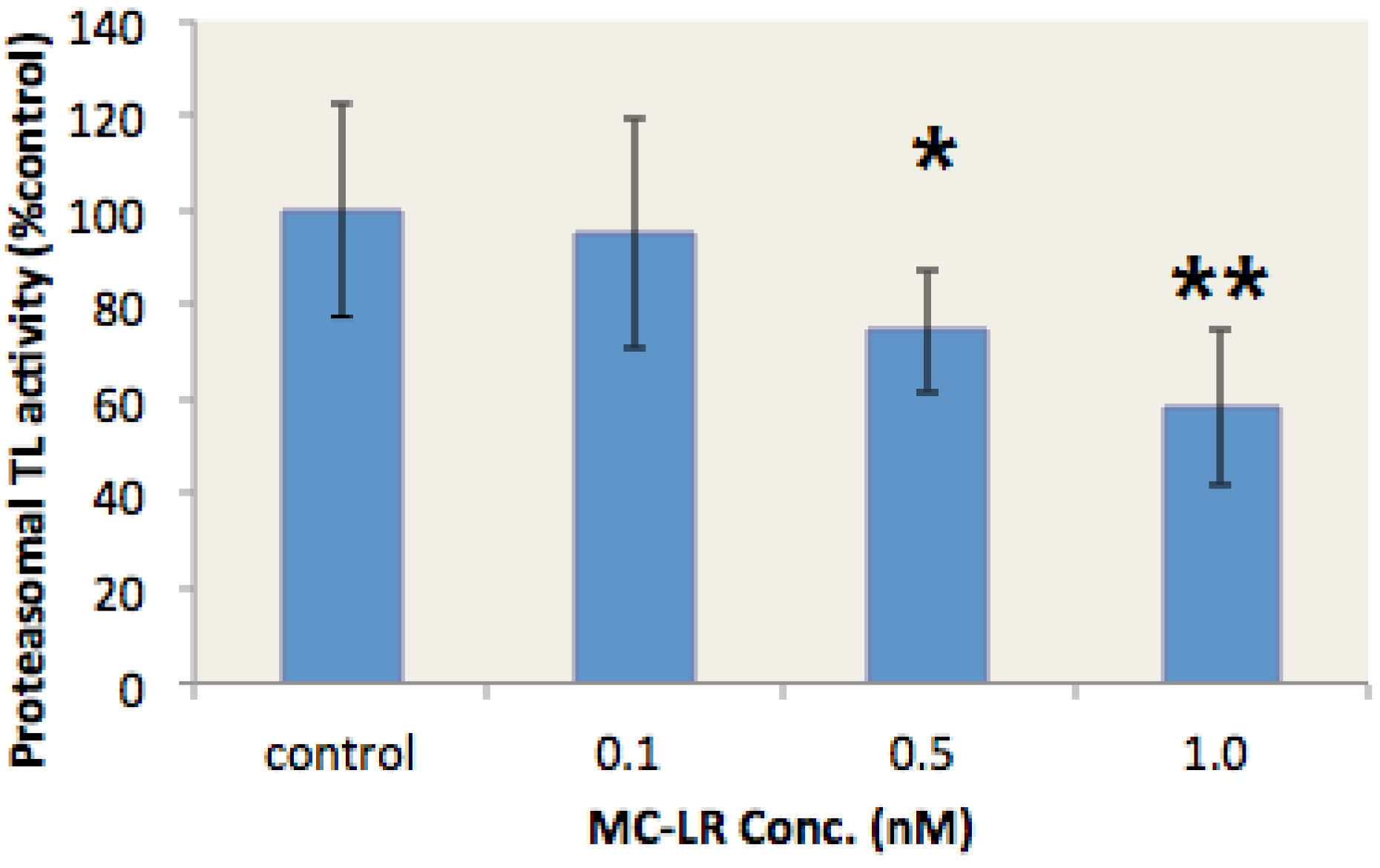
2.3. MC-LR Inhibited the Proteasome TL Activity in the Liver of Gobiocypris rarus
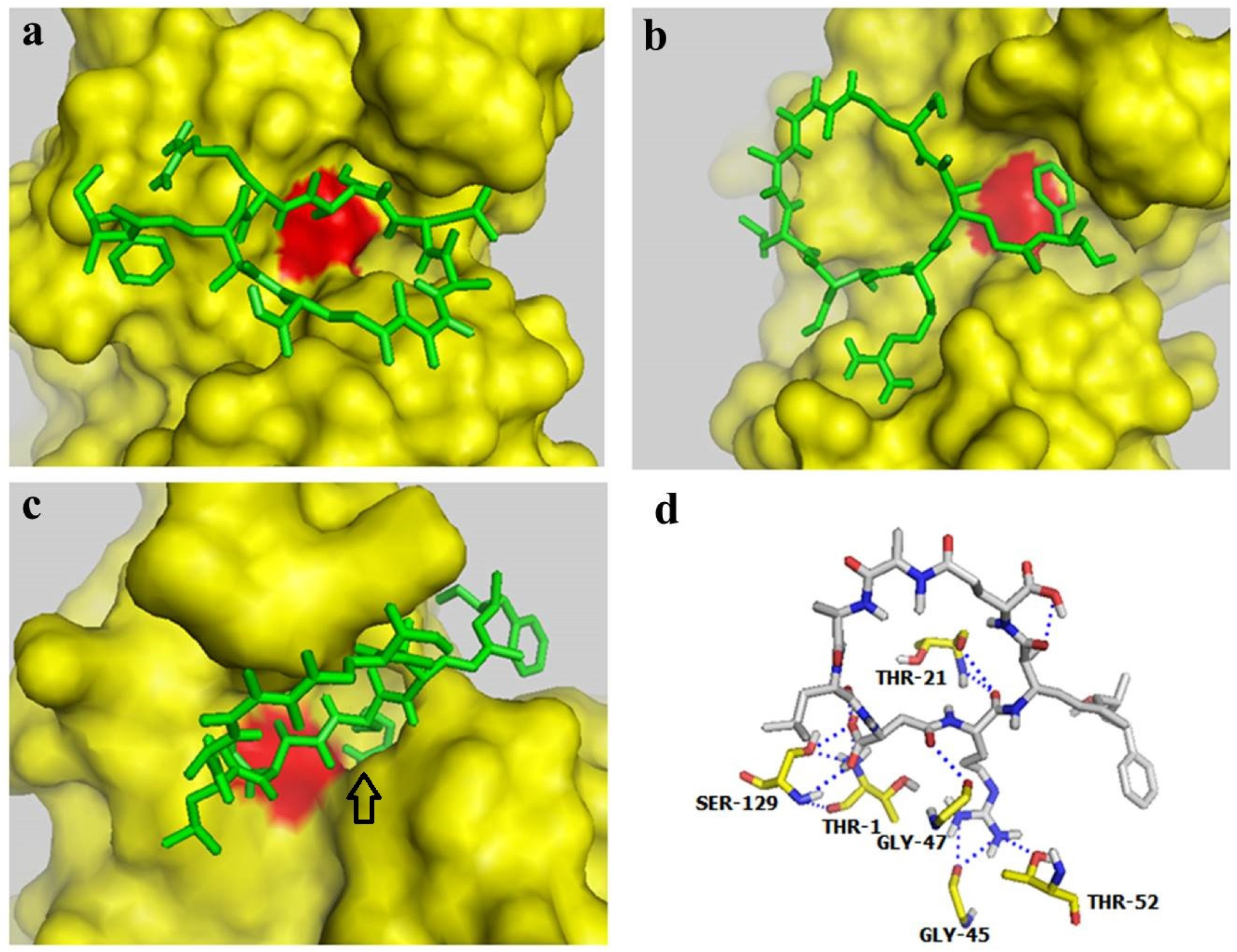
2.4. Docking Studies Indicated that MC-LR could Block the Active Site of the Proteasome β2 Subunit but not the Active Site of β1 and β5 Subunits
3. Materials and Methods
3.1. Materials
3.2. MC-LR Inhibition of Purified 20S Proteasome Activity
3.3. MC-LR Exposure to PC-3 Cells with UPS Reporter
3.4. MC-LR Inhibition of the Proteasome Activities in G. rarus
3.5. In silico Modeling of MC-LR Binding to Proteasome Subunits
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Figueiredo, D.R.; Azeiteiro, U.M.; Esteves, S.M.; Goncalves, F.J.; Pereira, M.J. Microcystin-producing blooms—A serious global public health issue. Ecotoxicol. Environ. Saf. 2004, 59, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.; Bakker, G.J. Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 7–60. [Google Scholar] [CrossRef] [PubMed]
- Dawson, R.M. The toxicology of microcystins. Toxicon 1998, 36, 953–962. [Google Scholar] [CrossRef]
- Zhou, Y.; Yuan, J.; Wu, J.; Han, X. The toxic effects of microcystin-LR on rat spermatogonia in vitro. Toxicol. Lett. 2012, 212, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Srivastava, A.; Oh, H.M.; Ahn, C.Y.; Choi, G.G.; Asthana, R.K. Recent trends in development of biosensors for detection of microcystin. Toxicon 2012, 60, 878–894. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, K. The toxicology of microcystin-LR: Occurrence, toxicokinetics, toxicodynamics, diagnosis and treatment. Vet. Hum. Toxicol. 2001, 43, 294–297. [Google Scholar] [PubMed]
- Milutinovic, A.; Sedmak, B.; Horvat-Znidarsic, I.; Suput, D. Renal injuries induced by chronic intoxication with microcystins. Cell. Mol. Biol. Lett. 2002, 7, 139–141. [Google Scholar] [PubMed]
- Huynh-Delerme, C.; Edery, M.; Huet, H.; Puiseux-Dao, S.; Bernard, C.; Fontaine, J.J.; Crespeau, F.; de Luze, A. Microcystin-LR and embryo-larval development of medaka fish, Oryzias latipes. I. Effects on the digestive tract and associated systems. Toxicon 2005, 46, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Trinchet, I.; Djediat, C.; Huet, H.; Dao, S.P.; Edery, M. Pathological modifications following sub-chronic exposure of medaka fish (Oryzias latipes) to microcystin-LR. Reprod. Toxicol. 2011, 32, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ying, F.; Chen, Y.; Han, X. Microcystin (-LR) affects hormones level of male mice by damaging hypothalamic-pituitary system. Toxicon 2012, 59, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Rymuszka, A.; Adaszek, L. Cytotoxic effects and changes in cytokine gene expression induced by microcystin-containing extract in fish immune cells—An in vitro and in vivo study. Fish Shellfish Immunol. 2013, 34, 1524–1532. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.E.; Lein, P.J.; Puschner, B. Microcystins alter chemotactic behavior in Caenorhabditis elegans by selectively targeting the AWA sensory neuron. Toxins 2014, 6, 1813–1836. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.; Vasconcelos, V. Molecular mechanisms of microcystin toxicity in animal cells. Int. J. Mol. Sci. 2010, 11, 268–287. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Da, L. Insights into the selective binding and toxic mechanism of microcystin to catalase. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2014, 121, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Kubo, T.; Kaya, K.; Hosoya, K. Comprehensive study of proteins that interact with microcystin-LR. Anal. Bioanal. Chem. 2012, 402, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Cui, J.; Liang, Y.; Xin, X.; Owen, Y.D.; Chen, C.; Shen, P. Identification of human liver mitochondrial aldehyde dehydrogenase as a potential target for microcystin-LR. Toxicology 2006, 220, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, A.; Harmala-Brasken, A.S.; Hellman, J.; Meriluoto, J.; Eriksson, J.E. Identification of ATP-synthase as a novel intracellular target for microcystin-LR. Chem. Biol. Interact. 2003, 142, 223–237. [Google Scholar] [CrossRef]
- Ciechanover, A. The ubiquitin-proteasome pathway: On protein death and cell life. EMBO J. 1998, 17, 7151–7160. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Finley, D. Regulation of proteasome activity in health and disease. Biochim. Biophys. Acta 2014, 1843, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Coux, O.; Tanaka, K.; Goldberg, A.L. Structure and functions of the 20S and 26S proteasomes. Annu. Rev. Biochem. 1996, 65, 801–847. [Google Scholar] [CrossRef] [PubMed]
- Heinemeyer, W.; Fischer, M.; Krimmer, T.; Stachon, U.; Wolf, D.H. The active sites of the eukaryotic 20S proteasome and their involvement in subunit precursor processing. J. Biol. Chem. 1997, 272, 25200–25209. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo-Pereira, M.E.; Berg, K.A.; Wilk, S. A new inhibitor of the chymotrypsin-like activity of the multicatalytic proteinase complex (20S proteasome) induces accumulation of ubiquitin-protein conjugates in a neuronal cell. J. Neurochem. 1994, 63, 1578–1581. [Google Scholar] [CrossRef] [PubMed]
- Hempel, D.; Wojtukiewicz, M.Z.; Kozlowski, L.; Romatowski, J.; Ostrowska, H. Increased plasma proteasome chymotrypsin-like activity in patients with advanced solid tumors. Tumour Biol. 2011, 32, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Parlati, F.; Lee, S.J.; Aujay, M.; Suzuki, E.; Levitsky, K.; Lorens, J.B.; Micklem, D.R.; Ruurs, P.; Sylvain, C.; Lu, Y.; et al. Carfilzomib can induce tumor cell death through selective inhibition of the chymotrypsin-like activity of the proteasome. Blood 2009, 114, 3439–3447. [Google Scholar] [CrossRef] [PubMed]
- Saling, P.M. Involvement of trypsin-like activity in binding of mouse spermatozoa to zonae pellucidae. Proc. Natl. Acad. Sci. USA 1981, 78, 6231–6235. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, E.; Pasten, C.; Kong, M.; Morales, P. Proteasomal activity in mammalian spermatozoa. Mol. Reprod. Dev. 2004, 69, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Mirabella, A.C.; Pletnev, A.A.; Downey, S.L.; Florea, B.I.; Shabaneh, T.B.; Britton, M.; Verdoes, M.; Filippov, D.V.; Overkleeft, H.S.; Kisselev, A.F. Specific cell-permeable inhibitor of proteasome trypsin-like sites selectively sensitizes myeloma cells to bortezomib and carfilzomib. Chem. Biol. 2011, 18, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Moreno, I.M.; Maraver, J.; Aguete, E.C.; Leao, M.; Gago-Martinez, A.; Camean, A.M. Decomposition of microcystin-LR, microcystin-RR, and microcystin-YR in water samples submitted to in vitro dissolution tests. J. Agric. Food Chem. 2004, 52, 5933–5938. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.Y.; Liang, X.R.; Hu, Y.F.; Shi, G.Q. The construction of a cellular model to monitor intracellular proteasome activity. Adv. Mater. Res. 2012, 518–523, 5586–5589. [Google Scholar] [CrossRef]
- Gilon, T.; Chomsky, O.; Kulka, R.G. Degradation signals for ubiquitin system proteolysis in Saccharomyces cerevisiae. EMBO J. 1998, 17, 2759–2766. [Google Scholar] [CrossRef] [PubMed]
- Bence, N.F.; Sampat, R.M.; Kopito, R.R. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 2001, 292, 1552–1555. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Hoeger, S.J.; Stemmer, K.; Feurstein, D.J.; Knobeloch, D.; Nussler, A.; Dietrich, D.R. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010, 245, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.L.; Kwon, E.M.; Ostrander, E.A.; Montgomery, R.B.; Lin, D.W.; Vessella, R.; Stanford, J.L.; Mostaghel, E.A. Expression of SLCO transport genes in castration-resistant prostate cancer and impact of genetic variation in SLCO1B3 and SLCO2B1 on prostate cancer outcomes. Cancer Epidemiol. Biomarkers Prev. 2011, 20, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Xu, Y.; Liang, Y.; Liao, T.; Wang, J. Vitellogenin in rare minnow (Gobiocypris rarus): Identification and induction by waterborne diethylstilbestrol. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2004, 137, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wang, Z.; Gong, S. Comparative sensitivity in Chinese rare minnow (Gobiocypris rarus) and Japanese Medaka (Oryzias latipes) exposed to ethinylestradiol. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng. 2007, 42, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Sun, Q.; Yang, H.; Dou, Q.; Deng, Q.; Wang, H.; Zhong, G. Molecular modeling for the interaction between proteasome beta 5 subunit and organotin compounds. Sci. China Chem. 2010, 53, 2387–2393. [Google Scholar] [CrossRef]
- Smith, D.M.; Daniel, K.G.; Wang, Z.; Guida, W.C.; Chan, T.H.; Dou, Q.P. Docking studies and model development of tea polyphenol proteasome inhibitors: Applications to rational drug design. Proteins 2004, 54, 58–70. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Chen, D.; Zhai, G.; Chen, M.S.; Cui, Q.C.; Zhou, Q.; He, B.; Dou, Q.P.; Jiang, G. The proteasome is a molecular target of environmental toxic organotins. Environ. Health Perspect. 2009, 117, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.J.; Dean, R.T. Assessment of proteasome activity in cell lysates and tissue homogenates using peptide substrates. Int. J. Biochem. Cell. Biol. 2003, 35, 716–727. [Google Scholar] [CrossRef]
- Furet, P.; Imbach, P.; Furst, P.; Lang, M.; Noorani, M.; Zimmermann, J.; Garcia-Echeverria, C. Modeling of the binding mode of a non-covalent inhibitor of the 20S proteasome. Application to structure-based analogue design. Bioorg. Med. Chem. Lett. 2001, 11, 1321–1324. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Z.; Zhang, L.; Shi, G. Proteasome as a Molecular Target of Microcystin-LR. Toxins 2015, 7, 2221-2231. https://doi.org/10.3390/toxins7062221
Zhu Z, Zhang L, Shi G. Proteasome as a Molecular Target of Microcystin-LR. Toxins. 2015; 7(6):2221-2231. https://doi.org/10.3390/toxins7062221
Chicago/Turabian StyleZhu, Zhu, Li Zhang, and Guoqing Shi. 2015. "Proteasome as a Molecular Target of Microcystin-LR" Toxins 7, no. 6: 2221-2231. https://doi.org/10.3390/toxins7062221
APA StyleZhu, Z., Zhang, L., & Shi, G. (2015). Proteasome as a Molecular Target of Microcystin-LR. Toxins, 7(6), 2221-2231. https://doi.org/10.3390/toxins7062221





