The Effects of Low Doses of Two Fusarium Toxins, Zearalenone and Deoxynivalenol, on the Pig Jejunum. A Light and Electron Microscopic Study
Abstract
:1. Introduction
2. Results and Discussion
2.1. Light Microscopic Study
2.1.1. Architecture of the Mucosa
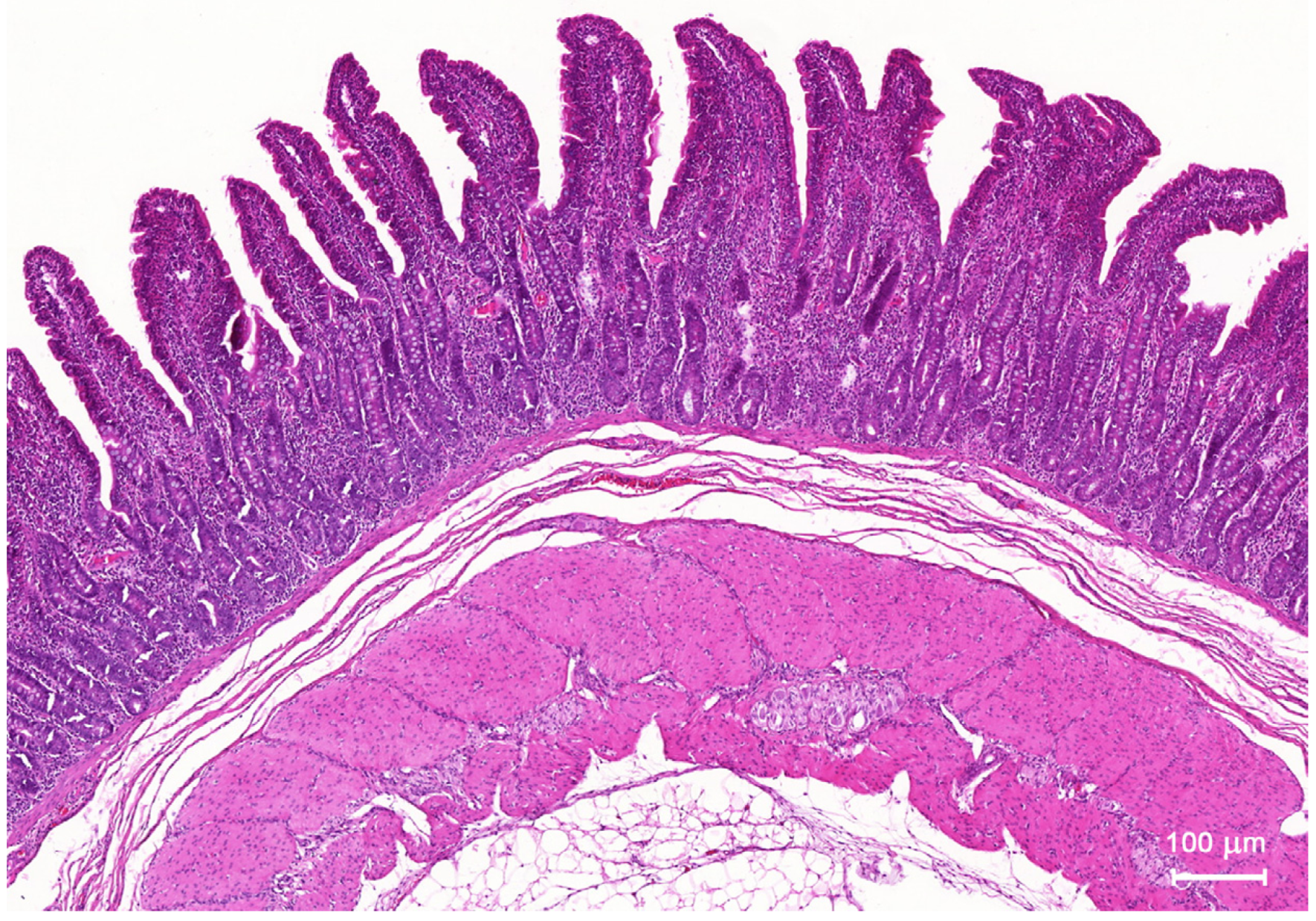
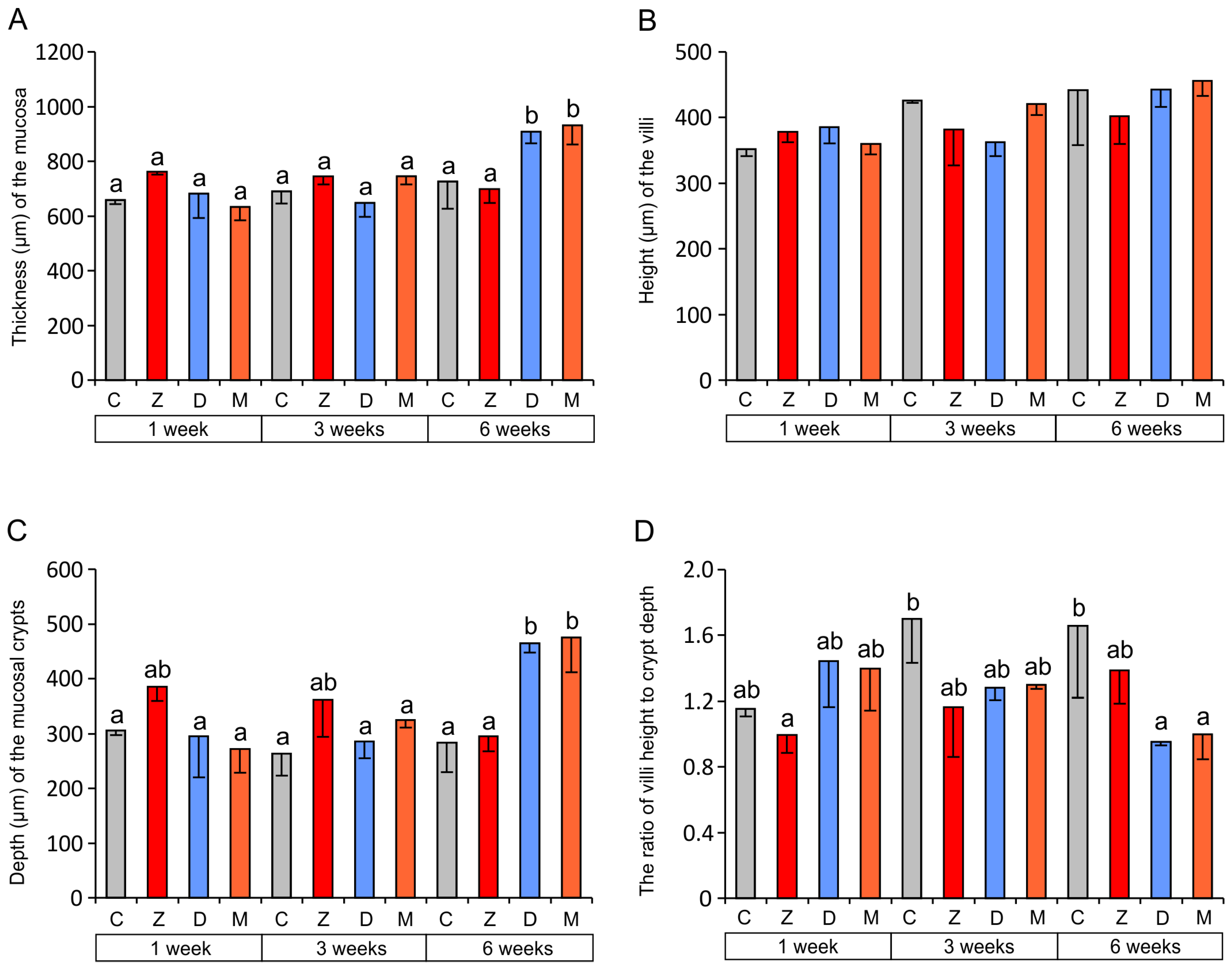
2.1.2. Goblet Cells

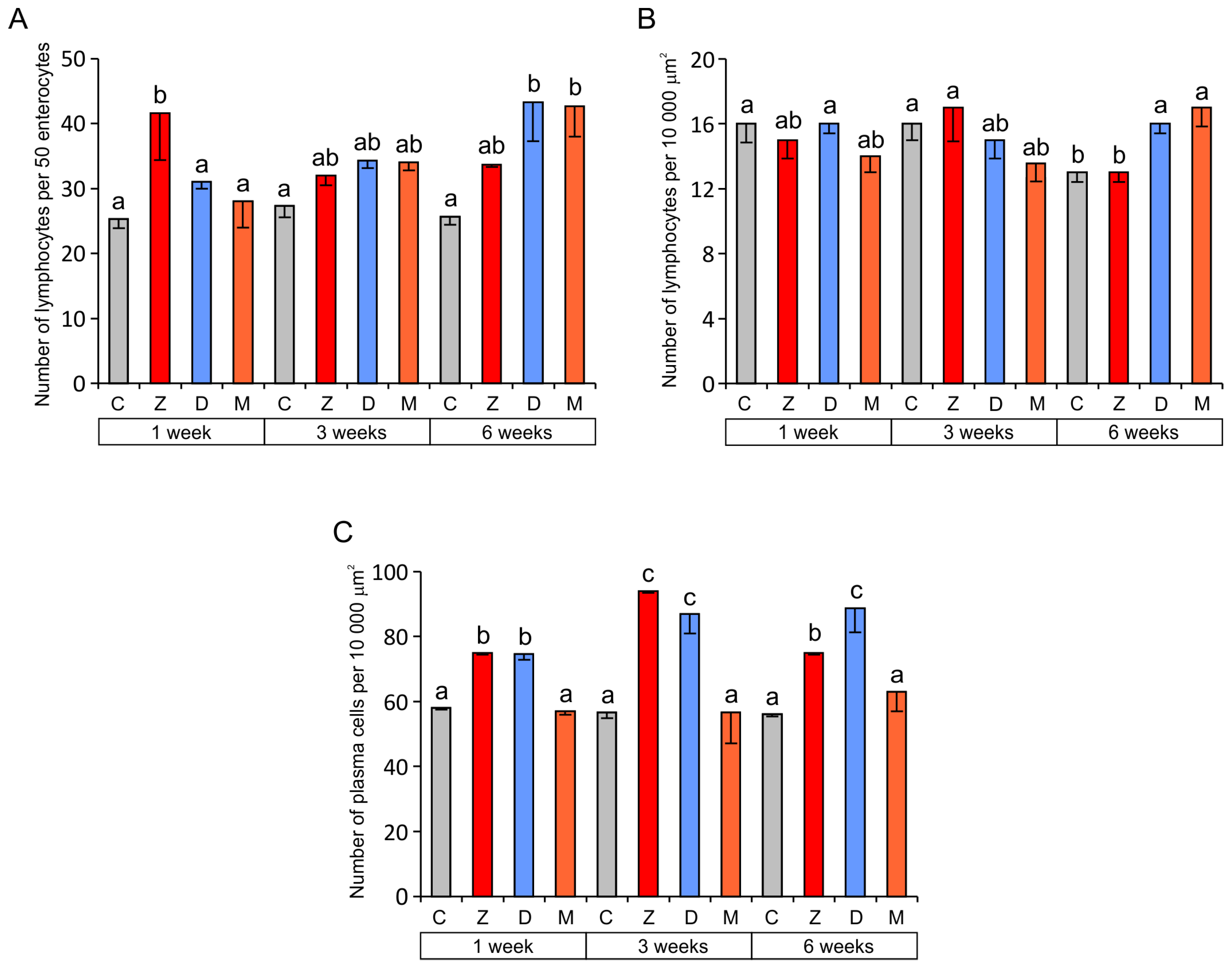
2.1.3. Cells of the Immune System
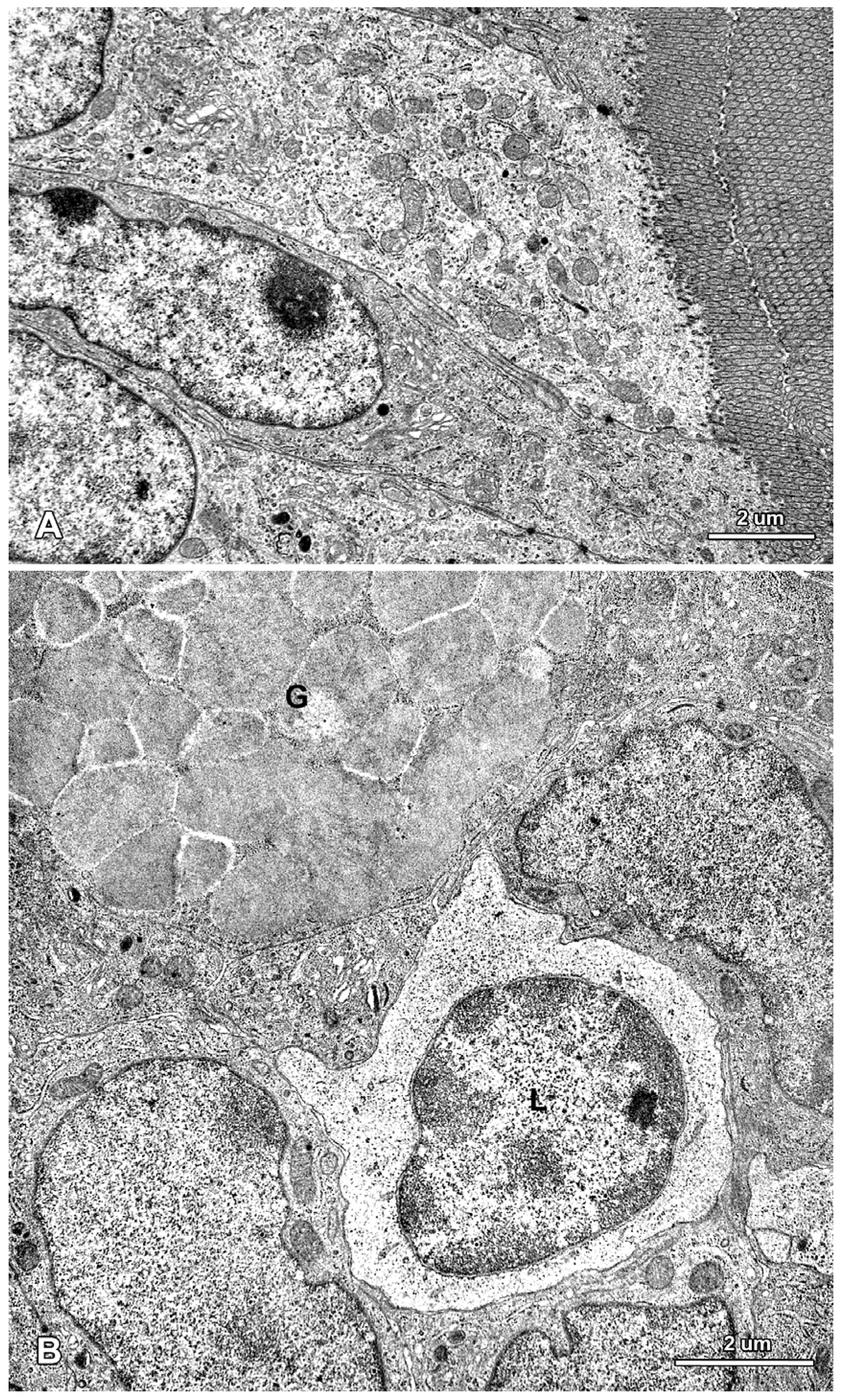
2.2. Ultrastructure of the Mucosa

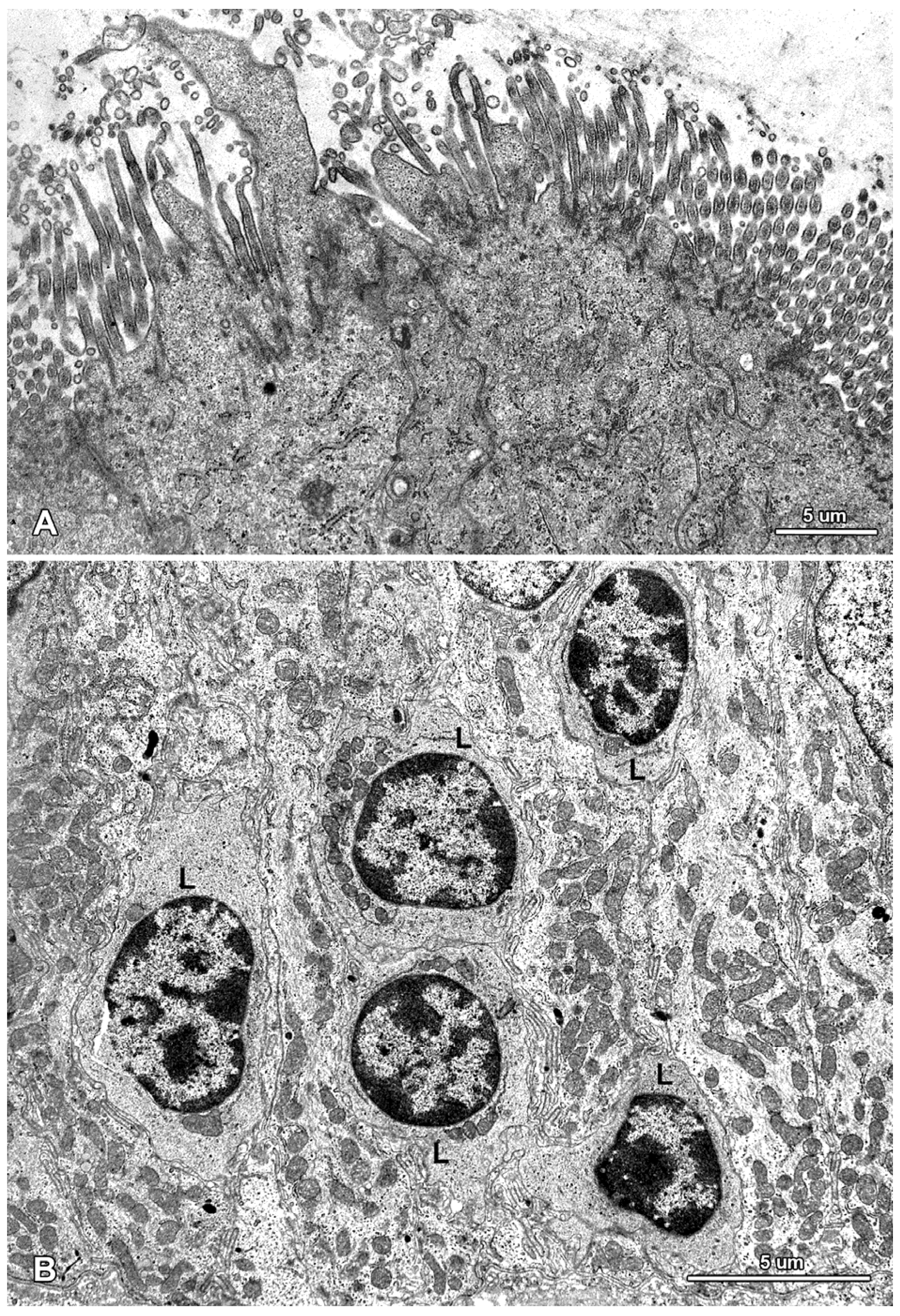
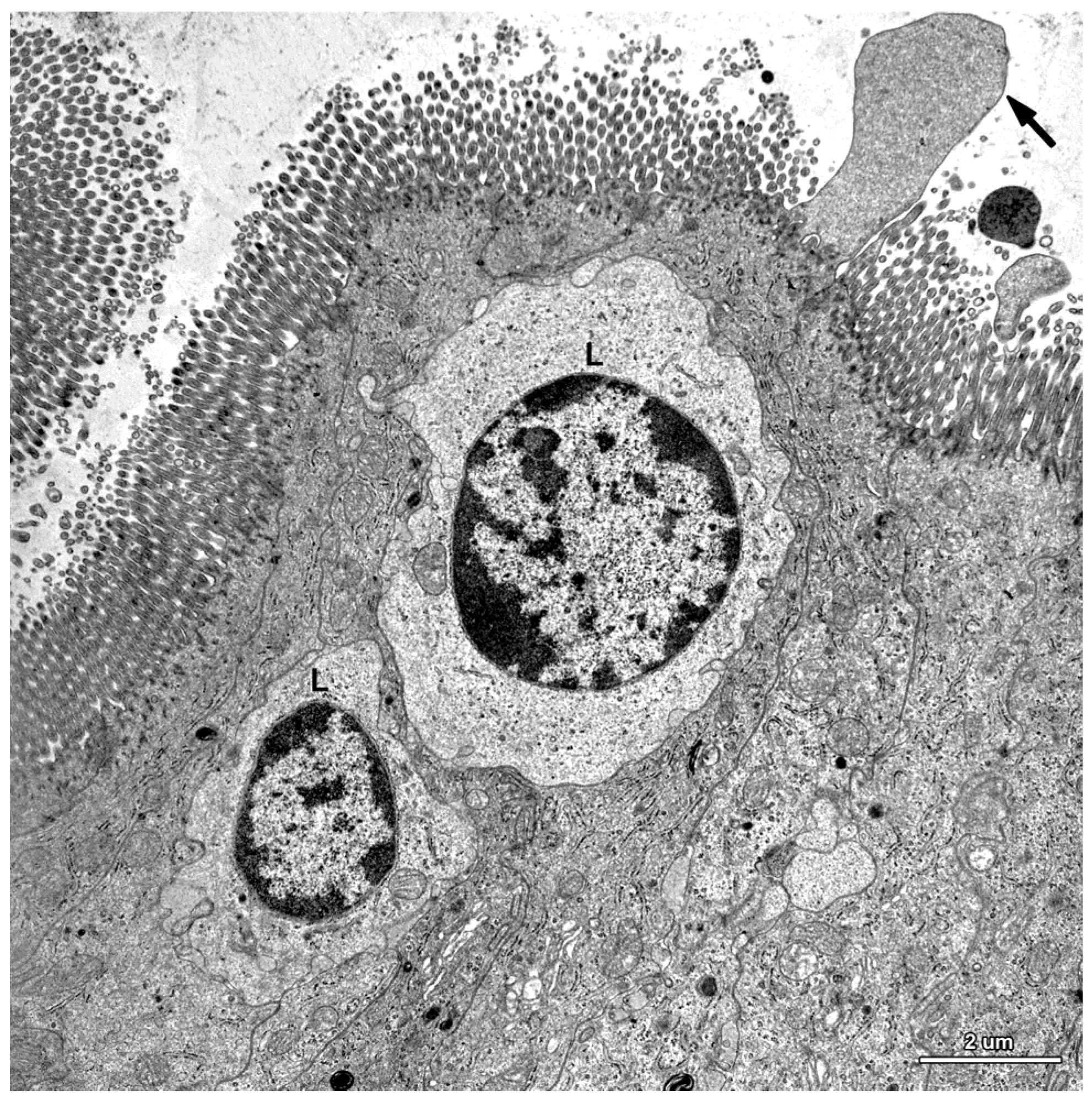
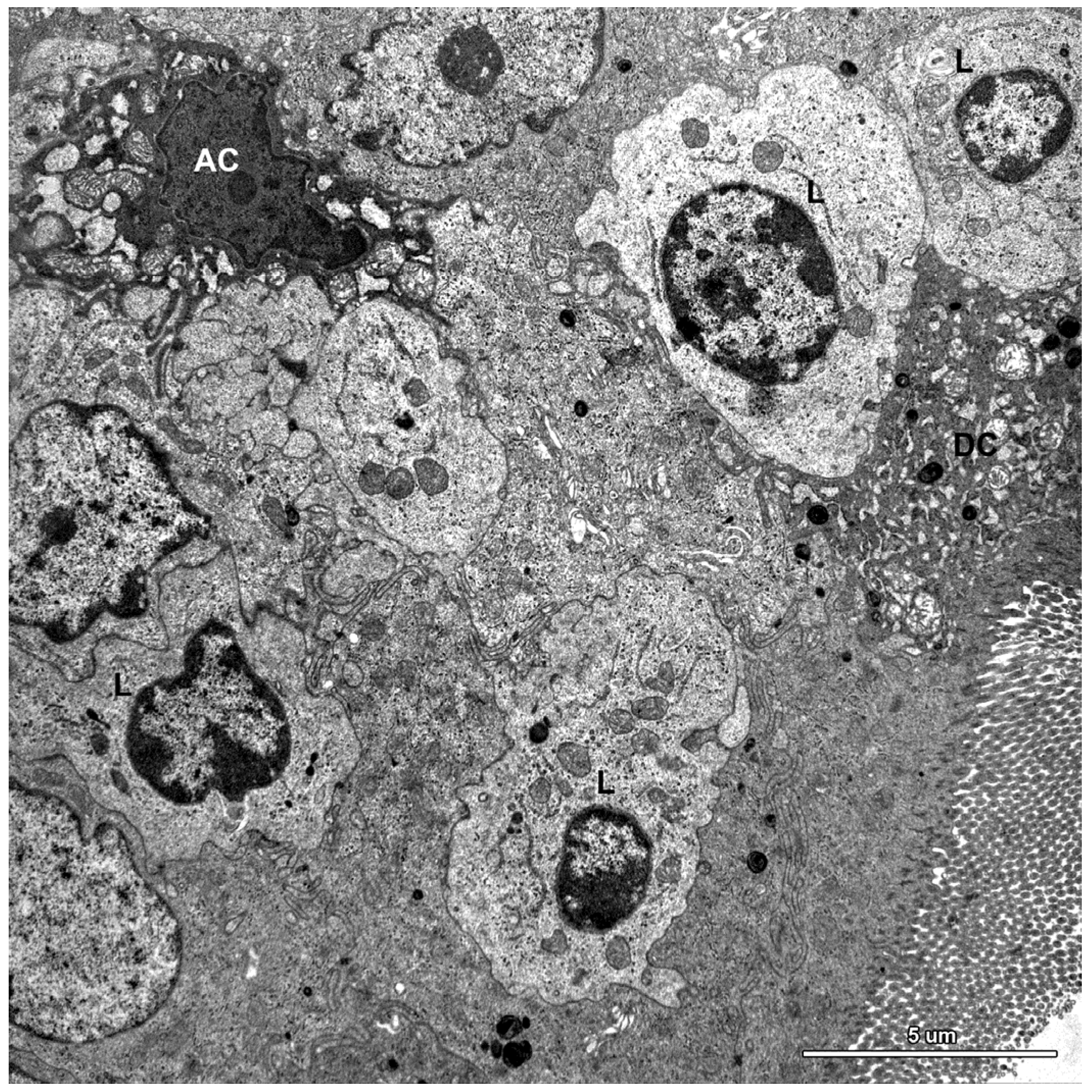

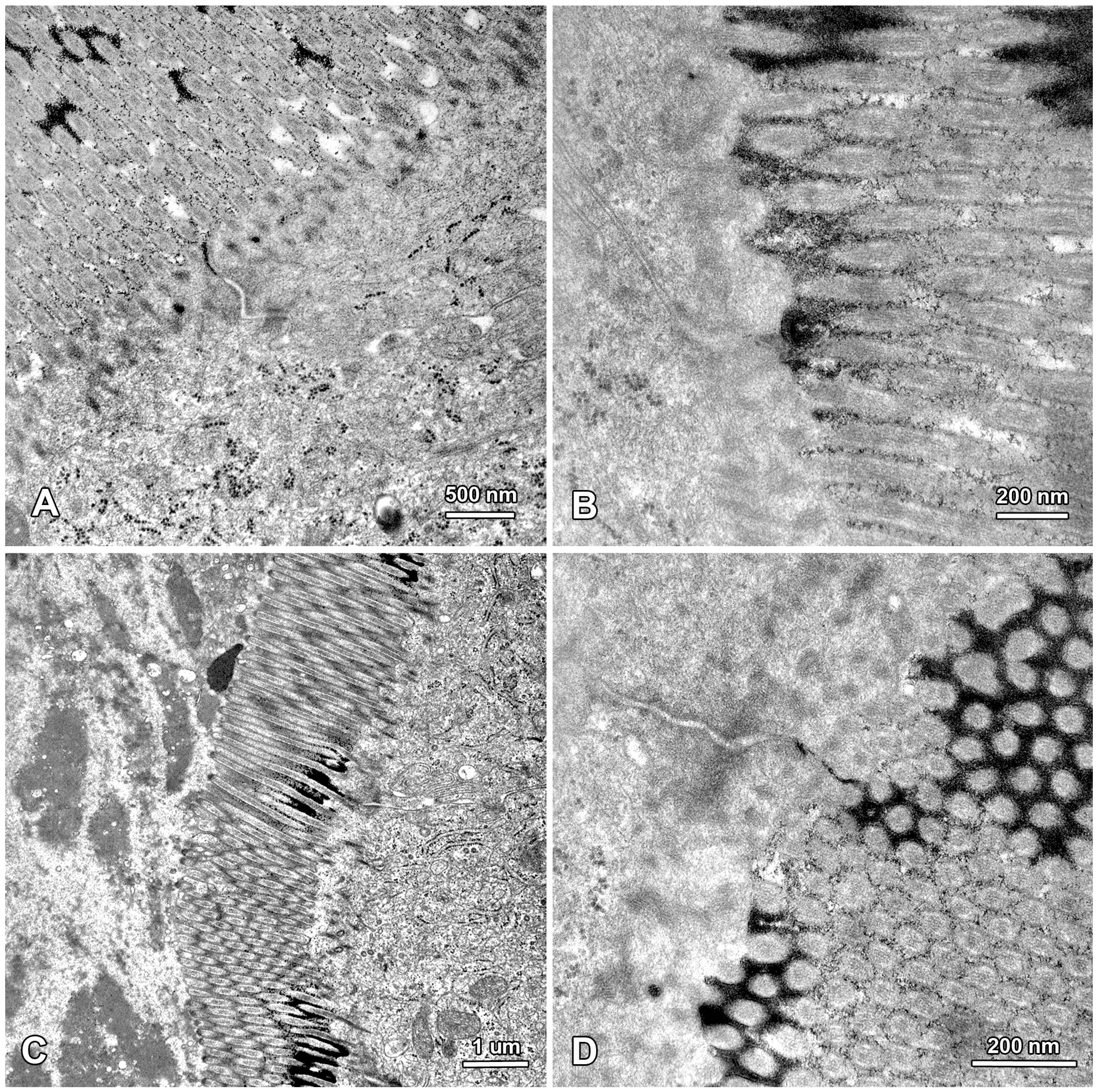
2.3. Functional Analysis of Epithelial Cell Junctions Using Lanthanum Ions
2.4. Summary
3. Experimental Section
3.1. Animals and Experimental Design
| Ingredients | % | Composition | g/kg |
|---|---|---|---|
| Wheat middlings | 60.2 | Starch | 449.0 |
| Barley middlings | 20.0 | Crude protein | 204.7 |
| Post-extraction soya meal | 16.0 | Crude fiber | 49.3 |
| Chalk | 0.5 | Fat | 32.3 |
| Multipremix PWT/3E | 3.0 | Ash | 82.2 |
| Zitrosan | 0.3 | - | - |
3.2. Histological Examinations
3.3. Ultrastructural Examinations
3.4. Lanthanum Procedure
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Döll, S.; Dänicke, S. The Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) in animal feeding. Prev. Vet. Med. 2011, 102, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Bracarense, A.P.; Lucioli, J.; Grenier, B.; Pacheco, G.D.; Moll, W.D.; Schatzmayr, G.; Oswald, I.P. Chronic ingestion of deoxynivalenol and fumonisin, alone or in interaction, induces morphological and immunological changes in the intestine of piglets. Br. J. Nutr. 2012, 107, 1776–1786. [Google Scholar] [CrossRef] [PubMed]
- Kolf-Clauw, M.; Castellote, J.; Joly, B.; Bourges-Abella, N.; Raymond-Letron, I.; Pinton, P.; Oswald, I.P. Development of a pig jejunal explant culture for studying the gastrointestinal toxicity of the mycotoxin deoxynivalenol: Histopathological analysis. Toxicol. In Vitro 2009, 23, 1580–1584. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Applegate, T.J. Modulation of intestinal functions following mycotoxin ingestion: Meta-analysis of published experiments in animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef] [PubMed]
- Swindle, M.M.; Smith, A.C.; Hepburn, B.J. Swine as models in experimental surgery. J. Invest. Surg. 1988, 1, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Cavret, S.; Lecoeur, S. Fusariotoxin transfer in animal. Food Chem. Toxicol. 2006, 44, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Malekinejad, H.; Maas-Bakker, R.F.; Fink-Gremmels, J. Bioactivation of zearalenone by porcine hepatic biotransformation. Vet. Res. 2005, 36, 799–810. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Brüssow, K.P.; Valenta, H.; Ueberschär, K.H.; Tiemann, U.; Schollenberger, M. On the effects of graded levels of Fusarium toxin contaminated wheat in diets for gilts on feed intake, growth performance and metabolism of deoxynivalenol and zearalenone. Mol. Nutr. Food Res. 2005, 49, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Tulayakul, P.; Li, J.Y.; Dong, K.S.; Manabe, N.; Kumagai, S. Metabolic conversion of zearalenone to α-zearalenol by goat tissues. J. Vet. Med. Sci. 2010, 72, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M.; Pettersson, H.; Sandholm, K.; Visconti, A.; Kiessling, K.H. Metabolism of zearalenone by sow intestinal mucosa in vitro. Food Chem. Toxicol. 1987, 25, 681–683. [Google Scholar] [CrossRef]
- Pinton, P.; Oswald, I.P. Effect of deoxynivalenol and other type B trichothecenes on the intestine: A review. Toxins 2014, 6, 1615–1643. [Google Scholar] [CrossRef] [PubMed]
- Basso, K.; Gomes, F.; Bracarense, A.P.L. Deoxynivanelol and Fumonisin, Alone or in Combination, Induce Changes on Intestinal Junction Complexes and in E-Cadherin Expression. Toxins 2013, 5, 2341–2352. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Nougayrède, J.P.; del Rio, J.C.; Moreno, C.; Marin, D.E.; Ferrier, L.; Bracarense, A.P.; Kolf-Clauw, M.; Oswald, I.P. The food contaminant deoxynivalenol, decreases intestinal barrier permeability and reduces claudin expression. Toxicol. Appl. Pharm. 2009, 237, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Goyarts, T.; Döll, S.; Grove, N.; Spolders, M.; Flachowsky, G. Effects of the Fusarium toxin deoxynivalenol on tissue protein synthesis in pigs. Toxicol. Lett. 2006, 165, 297–311. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Zhou, H.R.; Pestka, J.J. Targets and intracellular signaling mechanisms for deoxynivalenol-induced ribosomal RNA cleavage. Toxicol. Sci. 2012, 127, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Deoxynivalenol: Toxicity, mechanisms and animal health risks. Anim. Feed Sci. Tech. 2007, 137, 283–298. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol-induced proinflammatory gene expression: Mechanisms and pathological sequelae. Toxins 2010, 2, 1300–1317. [Google Scholar] [CrossRef] [PubMed]
- Flannery, B.M.; Clark, E.S.; Pestka, J.J. Anorexia induction by the trichothecene deoxynivalenol (vomitoxin) is mediated by the release of the gut satiety hormone peptide YY. Toxicol. Sci. 2012, 30, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Lucioli, J.; Pinton, P.; Callu, P.; Laffitte, J.; Grosjean, F.; Kolf-Clauw, M.; Oswald, I.P.; Bracarense, A.P.F.R.L. The food contaminant deoxynivalenol activates the mitogen activated protein kinases in the intestine: Interest of ex vivo models as an alternative to in vivo experiments. Toxicon 2013, 66, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Canady, R.; Coker, R.; Rgan, S.; Krska, R.; Kuiper-Goodman, T.; Olsen, M.; Pestka, J.; Resnik, S.; Schlatter, J. Deoxynivalenol. In Safety Evaluation of Certain Mycotoxins in Food; World Heath Organization: Geneva, Switzerland, 2001; pp. 419–555. [Google Scholar]
- Gajęcki, M. Zearalenone-undesirable substances in feed. Pol. J. Vet. Sci. 2002, 5, 117–122. [Google Scholar] [PubMed]
- Schoevers, E.J.; Santos, R.R.; Colenbrander, B.; Fink-Gremmels, J.; Roelen, B.A. Transgenerational toxicity of zearalenone in pigs. Reprod. Toxicol. 2012, 34, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Marin, D.E.; Taranu, I.; Burlacu, R.; Manda, G.; Motiu, M.; Neagoe, I.; Dragomir, C.; Stancu, M.; Calin, L. Effects of zearalenone and its derivatives on porcine immune response. Toxicol. In Vitro 2011, 25, 1981–1988. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on the risks for public health related to the presence of zearalenone in food. EFSA J. 2011. [Google Scholar] [CrossRef]
- Eriksen, G.S.; Pennington, J.; Schlatter, J.; Alexander, J.; Thuvander, A. Zearalenone. In Safety Evaluation of Certain Food Additives and Contaminants; World Heath Organization: Geneva, Switzerland, 2000; pp. 393–482. [Google Scholar]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed—Focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [PubMed]
- Avantaggiato, G.; Havenaar, R.; Visconti, A. Evaluation of the intestinal absorption of deoxynivalenol and nivalenol by an in vitro gastrointestinal model, and the binding efficacy of activated carbon and other adsorbent materials. Food Chem. Toxicol. 2004, 42, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, S. Intestinal absorption and excretion of aflatoxin in rats. Toxicol. Appl. Pharmacol. 1989, 97, 88–97. [Google Scholar] [CrossRef]
- Dänicke, S.; Valenta, S.; Döll, S. On the toxicokinetics and the metabolism of deoxynivalenol (DON) in the pig. Arch. Anim. Nutr. 2004, 58, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Swiech, E.; Buraczewska, L.; Ueberschär, K.H. Kinetics and metabolism of zearalenone in young female pigs. J. Anim. Physiol. Anim. Nutr. 2005, 89, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef] [PubMed]
- Waśkiewicz, A.; Beszterda, M.; Kostecki, M.; Zielonka, Ł.; Goliński, P.; Gajęcki, M. Deoxynivalenol in Gastrointestinal Tract of Immature Gilts under per os Toxin Application. Toxins 2014, 6, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, Ł.; Waśkiewicz, A.; Beszterda, M.; Kostecki, M.; Dąbrowski, M.; Obremski, K.; Goliński, P.; Gajęcki, M. Zearalenone in the Intestinal Tissues of Immature Gilts Exposed per os to Mycotoxins. Toxins 2015, 7, 3210–3223. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M.; Fantini, J. Some food-associated mycotoxins as potential risk factors in humans predisposed to chronic intestinal inflammatory diseases. Toxicon 2010, 56, 282–294. [Google Scholar] [CrossRef] [PubMed]
- World Heath Organization (WHO). Deoxynivalenol. In Evaluation of Certain Mycotoxins in Food; WHO: Geneva, Switzerland, 2002; pp. 35–42. [Google Scholar]
- The Commission of the European Communities. Deoxynivalenol, Zearalenone, Ochratoxin A, T-2 and HT-2 and Fumonisins in Products Intended for Animal Feeding. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006H0576&from=EN (accessed on 15 April 2015).
- Moeser, A.J.; Kim, I.B.; van Heugten, E.; Kempen, T.A.T.G. The nutritional value of degermed, dehulled corn for pigs and its impact on the gastrointestinal tract and nutrient excretion. J. Anim. Sci. 2002, 80, 2629–2638. [Google Scholar] [PubMed]
- Mills, S.E. Histology for Pathologists, 3rd ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 603–626. [Google Scholar]
- Vijaya, L.B.; Sesikeran, B.; Udaykumar, P.; Kalyanasundaram, S.; Raghunath, M. Effects of vitamin restriction and supplementation on rat intestinal epithelial cell apoptosis. Free Radic. Biol. Med. 2005, 15, 1614–1624. [Google Scholar]
- Gerez, J.R.; Pinton, P.; Callu, P.; Grosjean, F.; Oswald, I.P.; Bracarense, A.P. Deoxynivalenol alone or in combination with nivalenol and zearalenone induce systemic histological changes in pigs. Exp. Toxicol. Pathol. 2015, 67, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Tsybulskyy, D.; Lucioli, J.; Laffitte, J.; Callu, P.; Lyazhri, F.; Grosjean, F.; Bracarense, A.P.; Kolf-Clauw, M.; Oswald, I.P. Toxicity of deoxynivalenol and its acetylated derivatives on the intestine: Differential effects on morphology, barrier function, tight junction proteins, and mitogen-activated protein kinases. Toxicol. Sci. 2012, 130, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Obremski, K.; Gajęcka, M.; Zielonka, Ł.; Jakimiuk, E.; Gajęcki, M. Morphology and ultrastructure of small intestine mucosa in gilts with zearalenone mycotoxicosis. Pol. J. Vet. Sci. 2005, 8, 301–307. [Google Scholar] [PubMed]
- Obremski, K.; Zielonka, Ł.; Gajęcka, M.; Jakimiuk, E.; Bakuła, T.; Baranowski, M.; Gajęcki, M. Histological estimation of the small intestine wall after administration of feed containing deoxynivalenol, T-2 toxin and zearalenone in the pig. Pol. J. Vet. Sci. 2008, 11, 339–345. [Google Scholar] [PubMed]
- Awad, W.A.; Böhm, J.; Razzazi-Fazeli, E.; Zentek, J. Effects of feeding deoxynivalenol contaminated wheat on growth performance, organ weights and histological parameters of the intestine of broiler chickens. J. Anim. Physiol. Anim. Nutr. 2006, 90, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Girish, C.K.; Smith, T.K. Effects of feeding blends of grains naturally contaminated with Fusarium mycotoxins on small intestinal morphology of turkeys. Poult. Sci. 2008, 87, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Girgis, G.N.; Barta, J.R.; Brash, M.; Smith, T.K. Morphologic changes in the intestine of broiler breeder pullets fed diets naturally contaminated with Fusarium mycotoxins with or without coccidial challenge. Avian Dis. 2010, 54, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Bodiga, V.L.; Boindala, S.; Putcha, U.; Subramaniam, K.; Manchala, R. Chronic low intake of protein or vitamins increases the intestinal epithelial cell apoptosis in Wistar/NIN rats. Nutrition 2005, 21, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.; Braber, S.; Akbari, P.; Garssen, J.; Fink-Gremmels, J. Deoxynivalenol impairs weight gain and affects markers of gut health after low-dose, short-term exposure of growing pigs. Toxins 2015, 7, 2071–2095. [Google Scholar] [CrossRef] [PubMed]
- Törnwall, J.; Carey, A.B.; Fox, R.I.; Fox, H.S. Estrogen in autoimmunity: Expression of estrogen receptors in thymic and autoimmune T cells. J. Gend. Specif. Med. 1999, 2, 33–40. [Google Scholar] [PubMed]
- Obremski, K.; Poniatowska-Broniek, G. Zearalenone induces apoptosis and inhibits proliferation in porcine ileal Peyer’s patch lymphocytes. Pol. J. Vet. Sci. 2015, 18, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Cano, P.M.; Seeboth, J.; Meurens, F.; Cognie, J.; Abrami, R.I.; Oswald, P.; Guzylack-Piriou, L. Deoxynivalenol as a new factor in the persistence of intestinal inflammatory diseases: An emerging hypothesis through possible modulation of Th17-mediated response. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, V.; Croubels, S.; Martel, A.; Verbrugghe, E.; Goossens, J.; van Deun, K.; Boyen, F.; Thompson, A.; Shearer, N.; de Backer, P.; et al. The mycotoxin deoxynivalenol potentiates intestinal inflammation by Salmonella typhimurium in porcine ileal loops. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Nossol, C.; Diesing, A.K.; Kahlert, S.; Kersten, S.; Kluess, J.; Ponsuksili, S.; Hartig, R.; Wimmers, K.; Dänicke, S.; Rothkötter, H.J. Deoxynivalenol affects the composition of the basement membrane proteins and influences en route the migration of CD16(+) cells into the intestinal epithelium. Mycotoxin Res. 2013, 29, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, Ł.; Jakimiuk, E.; Obremski, L.; Gajęcka, M.; Dąbrowski, M.; Gajęcki, M. Evaluation of the proliferative activity of immunocompetent cells in the jejunal and iliac lymph nodes of prepubertal female wild boars diagnosed with mixed mycotoxicosis. Bull. Vet. Inst. Pulawy 2015, 59, 197–203. [Google Scholar] [CrossRef]
- Obremski, K. The effect of in vivo exposure to zearalenone on cytokine secretion by Th1 and Th2 lymphocytes in porcine Peyer’s patches after in vitro stimulation with LPS. Pol. J. Vet. Sci. 2014, 17, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Obremski, K. Changes in Th1 and Th2 cytokine concentrations in ileal Peyer’s patches in gilts exposed to zearalenone. Pol. J. Vet. Sci. 2014, 17, 53–59. [Google Scholar] [CrossRef] [PubMed]
- De Souza, L.C.M.; Retamal, C.A.; Rocha, G.M.; Lopez, M.L. Morphological evidence for a permeability barrier in the testis and spermatic duct of Gymnotus carapo (Teleostei: Gymnotidae). Mol. Reprod. Dev. 2015. [Google Scholar] [CrossRef]
- Park, C.J.; Ha, C.M.; Lee, J.E.; Gye, M.C. Claudin 11 inter-Sertoli tight junctions in the testis of the Korean soft-shelled turtle (Pelodiscus maackii). Biol. Reprod. 2015. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Lee, S.E.; Choi, K.J.; Choi, E.H.; Lee, S.H. Acute modulations in stratum corneum permeability barrier function affect claudin expression and epidermal tight junction function via changes of epidermal calcium gradient. Yonsei Med. J. 2013, 54, 523–528. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zwierzchowski, W.; Gajęcki, M.; Obremski, K.; Zielonka, Ł.; Baranowski, M. The occurrence of zearalenone and its derivatives in standard and therapeutic M. feeds for companion animals. Pol. J. Vet. Sci. 2004, 7, 289–293. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybylska-Gornowicz, B.; Tarasiuk, M.; Lewczuk, B.; Prusik, M.; Ziółkowska, N.; Zielonka, Ł.; Gajęcki, M.; Gajęcka, M. The Effects of Low Doses of Two Fusarium Toxins, Zearalenone and Deoxynivalenol, on the Pig Jejunum. A Light and Electron Microscopic Study. Toxins 2015, 7, 4684-4705. https://doi.org/10.3390/toxins7114684
Przybylska-Gornowicz B, Tarasiuk M, Lewczuk B, Prusik M, Ziółkowska N, Zielonka Ł, Gajęcki M, Gajęcka M. The Effects of Low Doses of Two Fusarium Toxins, Zearalenone and Deoxynivalenol, on the Pig Jejunum. A Light and Electron Microscopic Study. Toxins. 2015; 7(11):4684-4705. https://doi.org/10.3390/toxins7114684
Chicago/Turabian StylePrzybylska-Gornowicz, Barbara, Michał Tarasiuk, Bogdan Lewczuk, Magdalena Prusik, Natalia Ziółkowska, Łukasz Zielonka, Maciej Gajęcki, and Magdalena Gajęcka. 2015. "The Effects of Low Doses of Two Fusarium Toxins, Zearalenone and Deoxynivalenol, on the Pig Jejunum. A Light and Electron Microscopic Study" Toxins 7, no. 11: 4684-4705. https://doi.org/10.3390/toxins7114684
APA StylePrzybylska-Gornowicz, B., Tarasiuk, M., Lewczuk, B., Prusik, M., Ziółkowska, N., Zielonka, Ł., Gajęcki, M., & Gajęcka, M. (2015). The Effects of Low Doses of Two Fusarium Toxins, Zearalenone and Deoxynivalenol, on the Pig Jejunum. A Light and Electron Microscopic Study. Toxins, 7(11), 4684-4705. https://doi.org/10.3390/toxins7114684







