Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats. Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK
Abstract
:1. Introduction
2. Results
2.1. Metataxonomic Study of Fungal Communities in Oat Fields
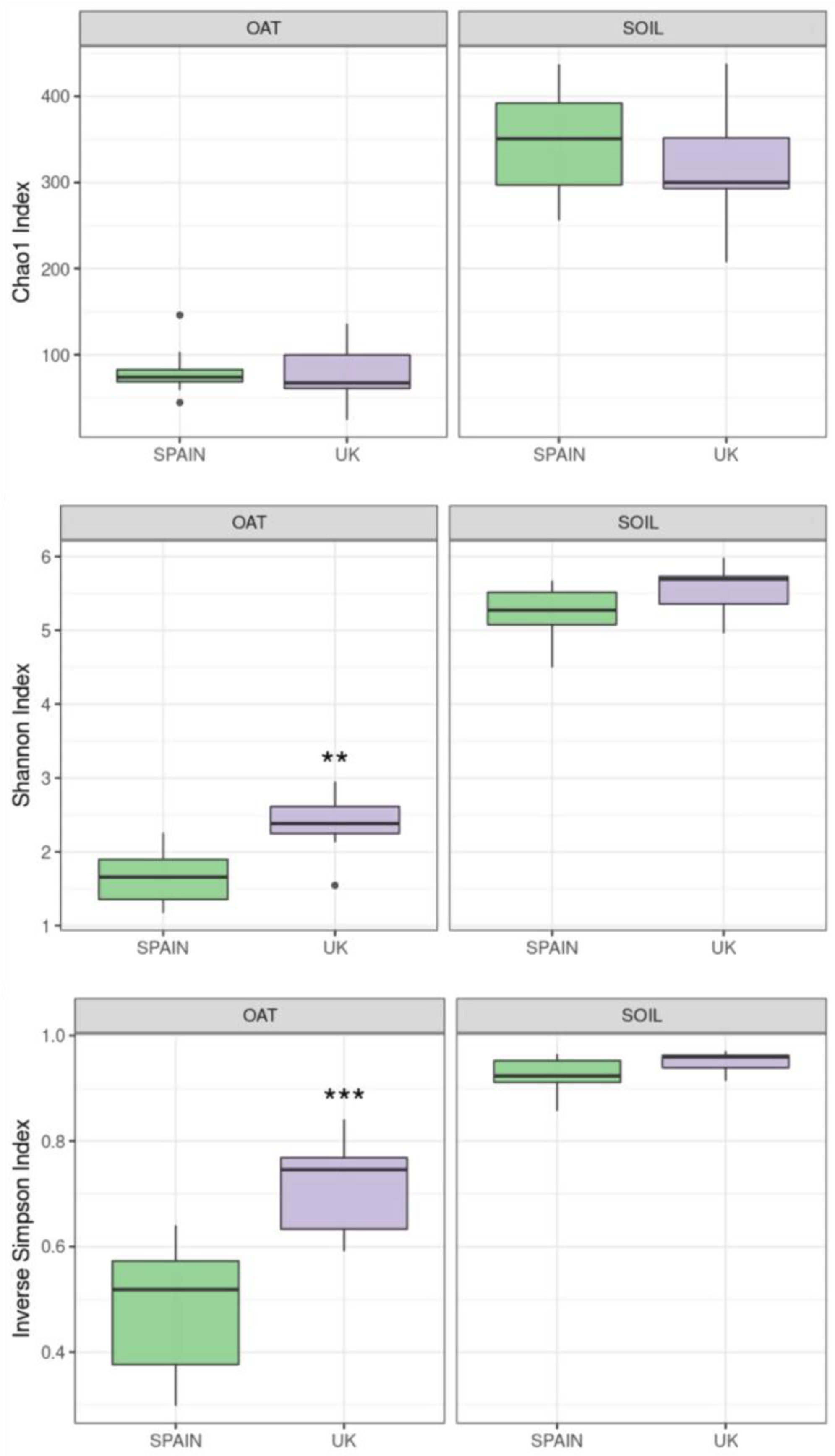
2.1.1. Diversity Analysis
2.1.2. Taxonomical Assignments at the Genus Level
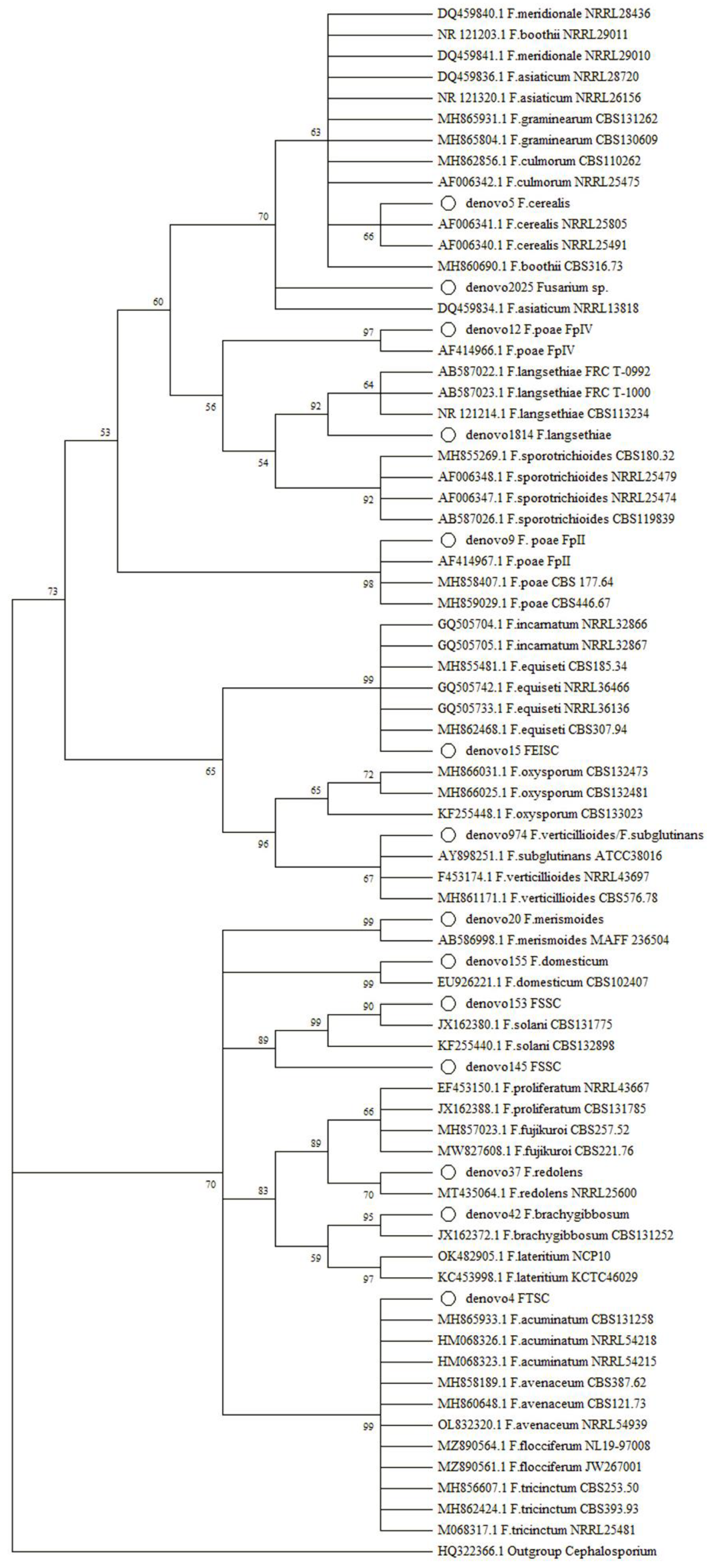
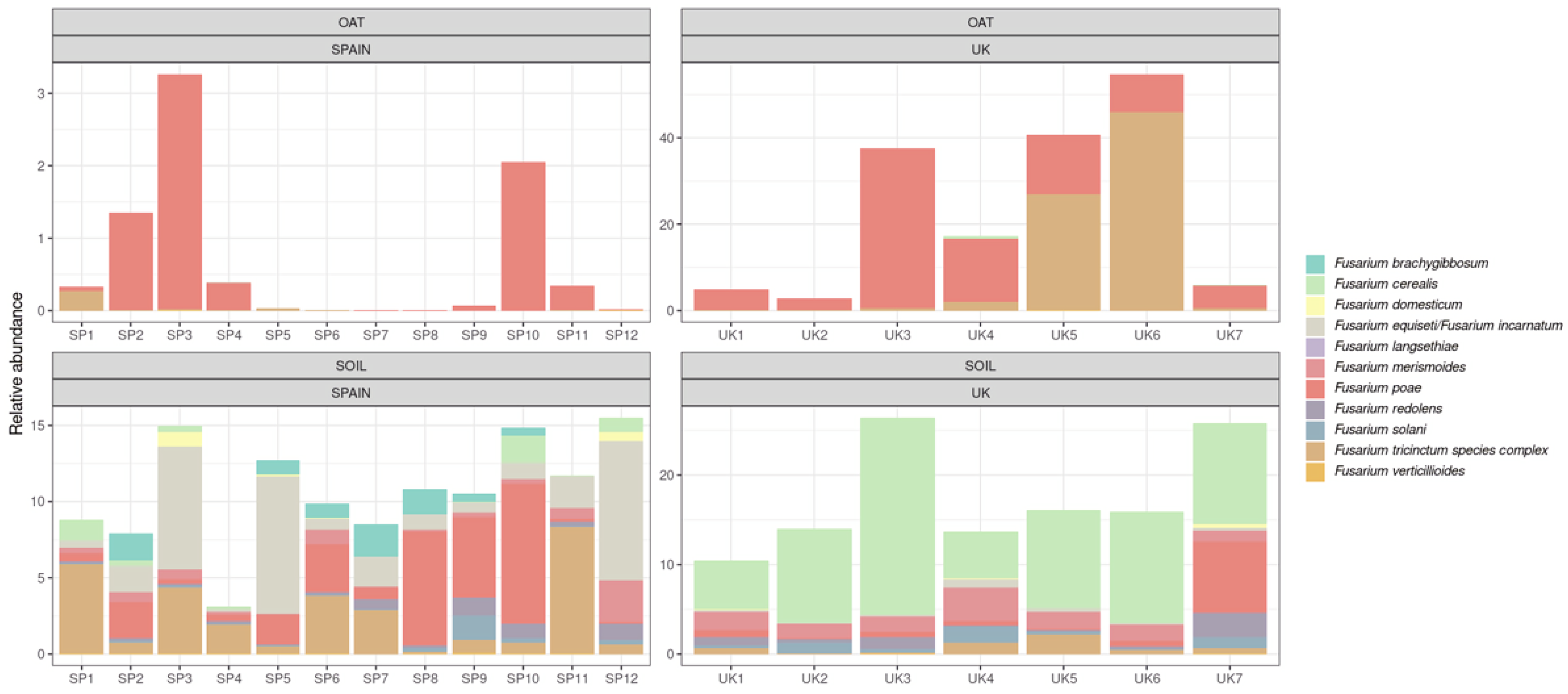
2.1.3. Study on the Occurrence of Fusarium spp.
2.1.4. Study on the Occurrence of Other Potential Trichothecene-Producing Species
2.2. Contamination of Oat Samples with Mycotoxins
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Sample Collection
5.2. DNA Isolation and Metataxonomic Analysis
5.3. Phylogenetic Analysis
5.4. Fungal Metabolite Determination in Oat Samples
5.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferrigo, D.; Raiola, A.; Causin, R. Fusarium toxins in cereals: Occurrence, legislation, factors promoting the appearance and their management. Molecules 2016, 21, 627. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation No 1881/2006 setting maximum levels for certain contaminants in foodstuffs. OJEU 2006, 364, 5–24. [Google Scholar]
- European Commission. Commission Regulation No 1126/2007 amending Regulation No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. OJEU 2007, 255, 14–17. [Google Scholar]
- Agricultural Production-Crops. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Agricultural_production_-_crops#Cereals (accessed on 22 May 2022).
- Foroud, N.A.; Baines, D.; Gagkaeva, T.Y.; Thakor, N.; Badea, A.; Steiner, B.; Bürstmayr, M.; Bürstmayr, H. Trichothecene in cereal grains—An update. Toxins 2019, 11, 634. [Google Scholar] [CrossRef]
- Mielniczuk, E.; Skwarylo-Bednarz, B. Fusarium head blight, mycotoxins and strategies for their reduction. Agronomy 2020, 10, 509. [Google Scholar] [CrossRef]
- Mahato, D.K.; Pandhi, S.; Kamle, M.; Gupta, A.; Sharma, B.; Panda, B.K.; Srivastava, S.; Kumar, M.; Selvakumar, R.; Pandey, A.K.; et al. Trichothecenes in food and feed: Occurrence, impact on human health and their detection and management strategies. Toxicon 2022, 208, 62–77. [Google Scholar] [CrossRef]
- Gil-Serna, J.; Vázquez, C.; Patiño, B. Mycotoxins | Toxicology. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–7. [Google Scholar] [CrossRef]
- European Commission. Commission Recommendation on the presence of T-2 and HT-2 toxin in cereals and cereal products (2013/165/EU). OJEU 2013, 91, 12–15. [Google Scholar]
- Luo, S.; Du, H.; Kebede, H.; Liu, Y.; Xing, F. Contamination status of major mycotoxins in agricultural product and foodstuff in Europe. Food Cont. 2021, 127, 108120. [Google Scholar] [CrossRef]
- Van der Fels-Klerx, H.; Stratakou, I. T-2 toxin and HT-2 toxin in grain and grain- based commodities in Europe: Occurrence, factors affecting occurrence, co- occurrence and toxicological effects. World Mycotoxin J. 2010, 3, 349–367. [Google Scholar] [CrossRef]
- De Colli, L.; De Ruyck, K.; Abdallah, M.F.; Finnan, J.; Mullins, E.; Kildea, S.; Spink, J.; Elliot, C.; Danaher, M. Natural co-occurrence of multiple mycotoxins in unprocessed oats grown in Ireland with various production systems. Toxins 2021, 13, 188. [Google Scholar] [CrossRef]
- Kis, M.; Vulic, A.; Kudumija, N.; Sarkanj, B.; Tkalec, V.J.; Aladic, K.; Skrivanko, M.; Furmeg, S.; Pleadin, J. A two-year occurrence of Fusarium T-2 and HT-2 toxin in Croatian cereals relative of the regional weather. Toxins 2021, 13, 39. [Google Scholar] [CrossRef]
- Kolawole, O.; De Ruyck, K.; Greer, B.; Meneely, J.; Doohan, F.; Danaher, M.; Elliott, C. Agronomic factors influencing the scale of Fusarium mycotoxin contamination of oats. J. Fungi 2021, 7, 965. [Google Scholar] [CrossRef]
- Edwards, S.G.; Barrier-Guillot, B.; Clasen, P.E.; Hietaniemi, V.; Petterson, H. Emerging issues of HT-2 and T-2 toxins in European cereal production. World Mycotoxin J. 2009, 2, 173–179. [Google Scholar] [CrossRef]
- Edwards, S.G.; Imathiu, S.M.; Ray, R.V.; Back, M.; Hare, M.C. Molecular studies to identify the Fusarium species responsible for HT-2 and T-2 mycotoxins in UK oats. Int. J. Food Microbiol. 2012, 156, 168–175. [Google Scholar] [CrossRef]
- Imathiu, S.M.; Ray, R.V.; Back, M.I.; Hare, M.C.; Edwards, S.G. A survey investigating the infection of Fusarium langsethiae and production of HT-2 and T-2 mycotoxins in UK oat fields. J. Phytopathol. 2013, 161, 553–561. [Google Scholar] [CrossRef]
- Fredlund, E.; Gidlund, A.; Petterson, H.; Olsen, M.; Börjesson, T. Real-time PCR detection of Fusarium species in Swedish oats and correlation to T-2 and HT-2 toxin content. World Mycotoxin J. 2010, 3, 77–88. [Google Scholar] [CrossRef]
- Fredlund, E.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Lindblad, M. Deoxynivalenol and other selected Fusarium toxins in Swedish oats—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 276–283. [Google Scholar] [CrossRef]
- Hofgaard, I.S.; Aamot, H.U.; Torp, T.; Jestoi, M.; Lattanzio, V.M.T.; Klemsdal, S.S.; Waalwijk, C.; Van der Lee, T.; Brodal, G. Associations between Fusarium species and mycotoxins in oats and spring wheat from farmers’ fields in Norway over a six-year period. World Mycotoxin J. 2016, 9, 365–378. [Google Scholar] [CrossRef]
- Schöneberg, T.; Jenny, E.; Wettstein, F.E.; Bucheli, T.D.; Mascher, F.; Bertossa, M.; Musa, T.; Seifert, K.; Gräfenhan, T.; Keller, B.; et al. Occurrence of Fusarium species and mycotoxins in Swiss oats—Impact of cropping factors. Eur. J. Agron. 2018, 92, 123–132. [Google Scholar] [CrossRef]
- Hietaniemi, V.; Rämö, S.; Yli-Mattila, T.; Jestoi, M.; Peltonen, S.; Kartio, M.; Sieviläinen, E.; Koivisto, T.; Parikka, P. Updated survey of Fusarium species and toxins in Finnish cereal grains. Food Addit. Contam. A 2016, 33, 831–848. [Google Scholar] [CrossRef]
- Medina, A.; Magan, N. Comparisons of water activity and temperature impacts on growth of Fusarium langsethiae strains from northern Europe on oat-based media. Int. J. Food Microbiol. 2010, 142, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Kokkonen, M.; Medina, A.; Magan, N. Comparative study of water and temperature relations of growth and T-2/HT-2 toxin production by strains of Fusarium sporotrichioides and Fusarium langsethiae. World Mycotoxin J. 2012, 5, 365–372. [Google Scholar] [CrossRef]
- Verheecke-Vaessen, C.; García-Cela, E.; López-Prieto, A.; Osk-Jonsdottir, I.; Medina, A.; Magan, N. Water and temperature relations of Fusarium langsethiae strains and modelling of growth and T-2 and HT-2 mycotoxin production on oat-based matrices. Int. J. Food Microbiol. 2021, 348, 109203. [Google Scholar] [CrossRef]
- Nazari, L.; Pattori, E.; Terzi, V.; Morcia, C.; Rossi, V. Influence of temperature on infection, growth, and mycotoxin production by Fusarium langsethiae and F. sporotrichioides in durum wheat. Food Microbiol. 2014, 39, 19–26. [Google Scholar] [CrossRef]
- Lindblad, M.; Börjesson, T.; Hietaniemi, V.; Elen, O. Statistical analysis of agronomical factors and weather conditions influencing deoxynivalenol levels in oats in Scandinavia. Food Addit. Contam. Part A 2012, 29, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Yli-Mattila, T.; Rämö, S.; Hietaniemi, V.; Hussien, T.; Carlobos-López, A.L.; Cumagun, C.J.R. Molecular quantification and genetic diversity of toxigenic Fusarium species in Northern Europe as compared to those in Southern Europe. Microorganisms 2013, 1, 162–174. [Google Scholar] [CrossRef]
- Juan, C.; Ritieni, A.; Mañes, J. Ocurrence of Fusarium mycotoxins in Italian cereal and cereal products from organic farming. Food Chem. 2013, 141, 1747–1755. [Google Scholar] [CrossRef]
- Parikka, P.; Hakala, K.; Tiilikkala, K. Expected shifts in Fusarium species’ composition on cereal grain in Northern Europe due to climatic change. Food Addit. Contam. A 2012, 29, 1543–1555. [Google Scholar] [CrossRef]
- Perrone, G.; Ferrara, M.; Medina, A.; Pascale, M.; Magan, N. Toxigenic fungi and mycotoxins in a climate change scenario: Ecology, genomics, distribution, prediction and prevention of the risk. Microorganisms 2020, 8, 1496. [Google Scholar] [CrossRef]
- Medina, A.; Akbar, A.; Baazeem, A.; Rodríguez, A.; Magan, N. Climate change, food security and mycotoxins: Do we know enough? Fungal Biol. Rev. 2017, 31, 143–154. [Google Scholar] [CrossRef]
- Infantino, A.; Pucci, N.; Conca, G.; Santori, A. First report of Fusarium langsethiae on durum wheat kernels in Italy. Plant Dis. 2007, 91, 1362. [Google Scholar] [CrossRef] [PubMed]
- Infantino, A.; Santori, A.; Aureli, G.; Belocchi, A.; De Felice, S.; Tizzani, L.; Lattanzio, V.M.T.; Haidukowski, M.; Pascale, M. Occurrence of Fusarium langsethiae strains isolated from Durum Wheat in Italy. J. Phytophatol. 2015, 163, 612–619. [Google Scholar] [CrossRef]
- Morcia, C.; Tumino, G.; Ghizzoni, R.; Badeck, F.W.; Lattanzio, V.M.T.; Pascale, M.; Terzi, V. Occurrence of Fusarium langsethiae and T-2 and HT-2 toxins in Italian malting barley. Toxins 2016, 8, 247. [Google Scholar] [CrossRef]
- Gil-Serna, J.; Mateo, E.M.; González-Jaén, M.T.; Jiménez, M.; Patiño, B. Contamination of barley seeds with Fusarium species and their toxins in Spain: An integrated approach. Food Addit. Contam. A 2013, 30, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Arias-Martín, M.; Haidukowski, M.; Farinós, G.; Patiño, B. Role of Sesamia nonagrioides and Ostrinia nubilalis as vectors of Fusarium spp. and contribution of corn borer-resistant Bt maize to mycotoxin reduction. Toxins 2021, 13, 780. [Google Scholar] [CrossRef] [PubMed]
- Tarazona, A.; Gómez, J.V.; Mateo, F.; Jiménez, M.; Mateo, E.M. Potential health risk associated with mycotoxins in oat grains consumed in Spain. Toxins 2021, 13, 421. [Google Scholar] [CrossRef] [PubMed]
- Imathiu, S.M.; Edwards, S.G.; Ray, R.V.; Back, M.A. Fusarium langsethiae—A HT-2 and T-2 toxins producer that needs more attention. J. Phytopathol. 2013, 161, 1–10. [Google Scholar] [CrossRef]
- McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From simple to complex mycotoxins. Toxins 2011, 3, 802–814. [Google Scholar] [CrossRef]
- Proctor, R.H.; McCormick, S.P.; Kim, H.S.; Cardoza, R.E.; Stanley, A.M.; Lindo, L.; Kelly, A.; Brown, D.W.; Lee, T.; Vaughan, M.M.; et al. Evolution of structural diversity of trichothecenes, a family of toxins produced by plant pathogenic and entomopathogenic fungi. PLoS Pathog. 2018, 14, e1006946. [Google Scholar] [CrossRef]
- Sota, T.; Kagata, H.; Ando, Y.; Utsumi, S.; Osono, T. Metagenomic approach yields insights into fungal diversity and functioning. In Species Diversity and Community Structure; Sota, T., Kagata, H., Ando, Y., Utsumi, S., Osono, T., Eds.; Springer: Tokyo, Japan, 2013; pp. 1–23. [Google Scholar]
- Nilson, R.H.; Anslan, S.; Bahram, M.; Wurzbacher, C.; Baldrian, P.; Tedersoo, L. Mycobiome diversity: High-throughput sequencing and identification of fungi. Nat. Rev. Microbiol. 2019, 17, 95–109. [Google Scholar] [CrossRef]
- Global Oats Market By Type (Steel Cut Oats, Rolled Oats, Oatmeal, Oat Flour, Others), Application (Bakery Products, Animal Feed, Food Ingredients, Health Care, Cosmetic Products, Others) and Regional Analysis (North America, Europe, Asia Pacific, Latin America, and Middle East & Africa) and—Global Industry Analysis, Size, Share, Growth, Trends, and Forecast (2022–2027). Available online: https://www.marketdataforecast.com/market-reports/oats-market (accessed on 22 June 2022).
- Pereira, V.L.; Fernandes, J.O.; Cunha, S.C. Mycotoxins in cereals and related foodstuffs: A review on occurrence and recent methods of analysis. Trends Food Sci. Technol. 2014, 36, 96–136. [Google Scholar] [CrossRef]
- Edwards, S. Fusarium mycotoxin content of UK organic and conventional oats. Food Addit. Contam. Part A 2009, 26, 1063–1069. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Baillie, H.; Patel, S.; Edwards, S.G. Occurrence and fate of Fusarium mycotoxins during commercial processing of oats in the UK. Food Addit. Contam. 2013, 24, 1374–1385. [Google Scholar] [CrossRef] [PubMed]
- Divon, H.H.; Razzaghian, J.; Udnes-Aamot, H.; Klemsdal, S.S. Fusarium langsethiae (Torp and Nirenberg), investigation of alternative infection routes in oats. Eur. J. Plant Pathol. 2012, 132, 147–161. [Google Scholar] [CrossRef]
- Piombo, E.; Abdelfattah, A.; Droby, S.; Wisniewski, M.; Spadaro, D.; Schema, L. Metagenomics approaches for the detection and surveillance of emerging and recurrent plant pathogens. Microorganisms 2021, 9, 188. [Google Scholar] [CrossRef]
- Hofgaard, I.S.; Seehusen, T.; Aamot, H.U.; Riley, H.; Razzaghian, J.; Le, V.H.; Hjelkrem, A.G.R.; Dill-Macky, R.; Brodal, G. Inoculum potential of Fusarium spp. relates to tillage and straw management in Norwegian fields of spring oats. Front. Microbiol. 2016, 7, 556. [Google Scholar] [CrossRef]
- Torp, M.; Nirenberg, H.I. Fusarium langsethiae sp. nov. on cereals in Europe. Int. J. Food Microbiol. 2004, 95, 247–256. [Google Scholar] [CrossRef]
- García-Díaz, M.; Gil-Serna, J.; Vázquez, C.; Botia, M.N.; Patiño, B. A comprehensive study on the occurrence of mycotoxins and their producing fungi during the maize production cycle in Spain. Microorganisms 2020, 8, 141. [Google Scholar] [CrossRef]
- Somma, S.; Ricci, C.A.; Ferracane, L.; Ritieni, A.; Logrieco, A.; Moretti, A. Trichothecene and beauvericin mycotoxin production and genetic variability in Fusarium poae isolated from wheat kernels from northern Italy. Food Addit. Contam. Part A 2010, 27, 729–737. [Google Scholar] [CrossRef]
- Steinglein, S.A. Fusarium poae: A pathogen that needs more attention. J. Plant Pathol. 2009, 91, 25–36. [Google Scholar]
- Vanheule, A.; De Boevre, M.; Moretti, A.; Scauflaire, J.; Munaut, F.; de Saeger, S.; Bekaert, B.; Haesaert, G.; Waalwijk, C.; van der Lee, T.; et al. Genetic divergence and chemotype diversity in the Fusarium head blight pathogen Fusarium poae. Toxins 2017, 9, 255. [Google Scholar] [CrossRef] [PubMed]
- Kuchynková, H.; Kalinová, J.P. Influence of variety and growing conditions on Fusarium occurrence, mycotoxicological quality, and yield parameters of hulled oats. Cereal Res. Commun. 2021, 49, 577–585. [Google Scholar] [CrossRef]
- Xue, A.G.; Chen, Y.; Seifert, K.; Guo, W.; Blackwell, B.A.; Harris, L.J.; Overy, D.P. Prevalence of Fusarium species causing head blight of spring wheat, barley and oat in Ontario during 2001–2017. Can. J. Plant Pathol. 2019, 41, 392–402. [Google Scholar] [CrossRef]
- Islam, M.N.; Tabassum, M.; Banik, M.; Daayf, F.; Fernando, W.G.D.; Harris, L.J.; Sura, S.; Wang, X. Naturally occurring Fusarium species and mycotoxins in oat grains from Manitoba, Canada. Toxins 2021, 13, 670. [Google Scholar] [CrossRef]
- Munkvold, G.P. Fusarium species and their associated mycotoxins. In Mycotoxigenic Fungi: Methods and Protocols; Moretti, A., Susca, A., Eds.; Springer: New York, NY, USA, 2017; pp. 51–106. [Google Scholar]
- Yli-Matila, T.; Mach, R.L.; Alekhina, I.A.; Bulat, S.A.; Koskinen, S.; Kullnig-Gradinger, C.M.; Kubicek, C.P.; Klemsdal, S.S. Phylogenetic relationship of Fusarium langsethiae to Fusarium poae and Fusarium sporotrichioides as inferred by IGS, ITS, β-tubulin sequences and UP-PCR hybridization analysis. Int. J. Food. Microbiol. 2004, 95, 267–285. [Google Scholar] [CrossRef]
- Boutigny, A.L.; Gautier, A.; Basler, R.; Dauthieux, F.; Leite, S.; Valade, R.; Aguayo, J.; Ioos, R.; Laval, V. Metabarcoding targeting the EF1 alpha region to assess Fusarium diversity on cereals. PLoS ONE 2019, 14, e0207988. [Google Scholar] [CrossRef]
- Palacios, S.A.; Del Canto, A.; Erazo, J.; Torres, A.M. Fusarium cerealis causing Fusarium head blight of durum wheat and its associated mycotoxins. Int. J. Food Microbiol. 2021, 346, 109161. [Google Scholar] [CrossRef]
- Vidal, A.; Marín, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Determination of aflatoxins, deoxynivalenol, ochratoxin A and zearalenone in wheat and oat based bran supplements sold in the Spanish market. Food Chem. Toxicol. 2013, 53, 133–138. [Google Scholar] [CrossRef]
- Palumbo, R.; Crisci, A.; Venancio, A.; Cortiñas-Abrahantes, J.; Dorne, J.L.; Battilani, P.; Toscano, P. Occurrence and co-occurrence of mycotoxins in cereal-based feed and food. Microorganisms 2020, 8, 74. [Google Scholar] [CrossRef]
- Polisenska, I.; Jirsa, O.; Vaculova, K.; Pospíchalová, M.; Wawroszova, S.; Frydrych, J. Fusarium mycotoxins in two hulless oat and barley cultivars used for food purposes. Foods 2020, 9, 1037. [Google Scholar] [CrossRef]
- Farhadi, A.; Fakhri, Y.; Kachuei, R.; Vasseghian, Y.; Huseyn, E.; Khaneghah, A.M. Prevalence and concentration of fumonisins in cereal-based foods: A global systematic review and meta-analysis study. Environ. Sci. Pollut. Res. 2021, 28, 10998–21008. [Google Scholar] [CrossRef]
- Karlsson, I.; Persson, P.; Friberg, H. Fusarium head blight from a microbiome perspective. Front. Microbiol. 2021, 12, 628373. [Google Scholar] [CrossRef]
- Terzi, V.; Tumino, G.; Stanca, A.M.; Morcia, C. Reducing the incidence of cereal head infection and mycotoxins in small grain cereal species. J. Cereal Sci. 2014, 59, 284–293. [Google Scholar] [CrossRef]
- Venkatesh, N.; Keller, N.P. Mycotoxins in conversation with bacteria and fungi. Front. Microb. 2019, 10, 403. [Google Scholar] [CrossRef]
- Cobo-Díaz, J.F.; Legrand, F.; Le Floch, G.; Picot, A. Influence of maize residues in shaping soil microbiota and Fusarium spp. Communities. Microb. Ecol. 2022, 83, 702–713. [Google Scholar] [CrossRef]
- Xiong, W.; Li, R.; Ren, Y.; Liu, C.; Zhao, Q.; Wu, H.; Jousset, A.; Shen, Q. Distint roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Biol. Biochem. 2017, 107, 198–207. [Google Scholar] [CrossRef]
- Avila, C.F.; Moreira, G.M.; Nicolli, C.P.; Gomes, L.B.; Abreu, L.M.; Pfenning, L.H.; Haidukowski, M.; Moretti, A.; Logriego, A.; Del Ponte, E.M. Fusarium incarnatum-equiseti species complex associated with Brazilian rice: Phylogeny, morphology and toxigenic potential. Int. J. Food Microbiol. 2019, 306, 108267. [Google Scholar] [CrossRef]
- Villani, A.; Proctor, R.H.; Kim, H.S.; Brown, D.W.; Logrieco, A.F.; Amatulli, M.T.; Moretti, A.; Susca, A. Variation in secondary metabolite production potential in the Fusarium incarnatum-equiseti species complex revealed by comparative analysis of 13 genomes. BMC Genom. 2019, 20, 314. [Google Scholar] [CrossRef]
- Villani, A.; Moretti, A.; De Saeger, S.; Han, Z.; Di Mavungu, J.D.; Soares, C.M.G.; Proctor, R.H.; Venancio, A.; Lima, N.; Paciolla, C.; et al. A polyphasic approach for characterization of a collection of cereal isolates of the Fusarium incarnatum-equiseti species complex. Int. J. Food Microbiol. 2016, 234, 24–35. [Google Scholar] [CrossRef]
- Lu, Y.; Qiu, J.; Wang, S.; Xu, J.; Ma, G.; Shi, J.; Bao, Z. Species diversity and toxigenic potential of Fusarium incarnatum-equiseti species complex isolates from rice and soybean in China. Plant Dis. 2021, 105, 2628–2636. [Google Scholar] [CrossRef]
- Fallahi, M.; Saremi, H.; Javan-Nikkhah, M.; Somma, S.; Haidukowski, M.; Logrieco, A.F.; Moretti, A. Isolation, molecular identification and mycotoxin profile of Fusarium species isolated from maize kernels in Iran. Toxins 2019, 11, 297. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Plant Health. Pest categorization of Fusarium brachygibbosum. EFSA J. 2021, 19, 6887. [Google Scholar]
- Rabaaoui, A.; Dall’Asta, C.; Righetti, L.; Susca, A.; Logrieco, A.F.; Namsi, A.; Gdoura, R.; Werbrouck, S.P.O.; Moretti, A.; Masiello, M. Phylogeny and mycotoxin profile of pathogenic Fusarium species isolated from sudden decline syndrome and leaf wilt symptomns on date palms (Phoenix dactylifera) in Tunisia. Toxins 2021, 13, 463. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Burns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Method and Application; Innis, M.A., Gelfald, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Magoc, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Li, W.; Fu, L.; Niu, B.; Wu, S.; Wooley, J. Ultrafast clustering algorithms for metagenomic sequence analysis. Brief. Bioinform. 2012, 13, 656–668. [Google Scholar] [CrossRef]
- Koljalg, U.; Nilsson, R.H.; Abarenkov, K.; Tedersoo, L.; Taylor, A.F.; Bahram, M.; Bates, S.T.; Bruns, T.D.; Bengtsson-Palme, J.; Callaghan, T.M.; et al. Towards a unified paradigm for sequence-based identification of fungi. Mol. Ecol. 2013, 22, 5271–5277. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Isidro-Sánchez, J.; D’Arcy, K.; Verheecke-Vaessen, C.; Kahla, A.; Bekele, W.; Doohan, F.; Magan, N.; Medina, A. Genome-wide association mapping of Fusarium langsethiae infection and mycotoxin accumulation in oat (Avena sativa L.). Plant Genome 2020, 13, e20023. [Google Scholar] [CrossRef]
- Malachová, A.; Sulyok, M.; Beltrán, E.; Berthiller, F.; Krska, R. Optimization and validation of a quantitative liquid chromatography–tandem mass spectrometric method covering 295 bacterial and fungal metabolites including all regulated mycotoxins in four model food matrices. J. Chromatogr. A 2014, 1362, 145–156. [Google Scholar] [CrossRef]



| Sample | Origin | T-2 | HT-2 | 3-ADON | DON | NIV | DAS | NEO | BEA | 15-AS |
|---|---|---|---|---|---|---|---|---|---|---|
| SP1 | Spain | ND | ND | ND | 61.21 | ND | ND | ND | ND | 6.25 |
| SP2 | Spain | 2123.58 | 7.5 | 423.45 | 73.1 | ND | ND | ND | ND | 4.07 |
| SP3 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP4 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP5 | Spain | ND | ND | ND | 14.77 | ND | ND | ND | ND | ND |
| SP6 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP7 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP8 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP9 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP10 | Spain | ND | ND | ND | 15.37 | ND | ND | ND | ND | ND |
| SP11 | Spain | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| SP12 | Spain | ND | ND | ND | 10.79 | ND | ND | ND | ND | ND |
| UK1 | UK | ND | ND | ND | ND | 2276.19 | 15.29 | ND | 98.95 | 86.60 |
| UK2 | UK | ND | ND | ND | 63.42 | ND | ND | ND | 10.43 | ND |
| UK3 | UK | ND | ND | <LOQ | ND | 424.65 | 0.69 | ND | 1266.00 | 54.75 |
| UK4 | UK | 1400.23 | ND | ND | ND | ND | ND | ND | 44.03 | ND |
| UK5 | UK | ND | ND | <LOQ | ND | 689.26 | ND | ND | 2.44 | ND |
| UK6 | UK | ND | ND | <LOQ | ND | ND | ND | 5.29 | ND | ND |
| UK7 | UK | 391.84 | ND | ND | ND | 233.27 | ND | ND | 456.30 | 5.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gil-Serna, J.; Patiño, B.; Verheecke-Vaessen, C.; Vázquez, C.; Medina, Á. Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats. Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK. Toxins 2022, 14, 592. https://doi.org/10.3390/toxins14090592
Gil-Serna J, Patiño B, Verheecke-Vaessen C, Vázquez C, Medina Á. Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats. Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK. Toxins. 2022; 14(9):592. https://doi.org/10.3390/toxins14090592
Chicago/Turabian StyleGil-Serna, Jéssica, Belén Patiño, Carol Verheecke-Vaessen, Covadonga Vázquez, and Ángel Medina. 2022. "Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats. Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK" Toxins 14, no. 9: 592. https://doi.org/10.3390/toxins14090592
APA StyleGil-Serna, J., Patiño, B., Verheecke-Vaessen, C., Vázquez, C., & Medina, Á. (2022). Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats. Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK. Toxins, 14(9), 592. https://doi.org/10.3390/toxins14090592








