AB5 Enterotoxin-Mediated Pathogenesis: Perspectives Gleaned from Shiga Toxins
Abstract
1. Introduction
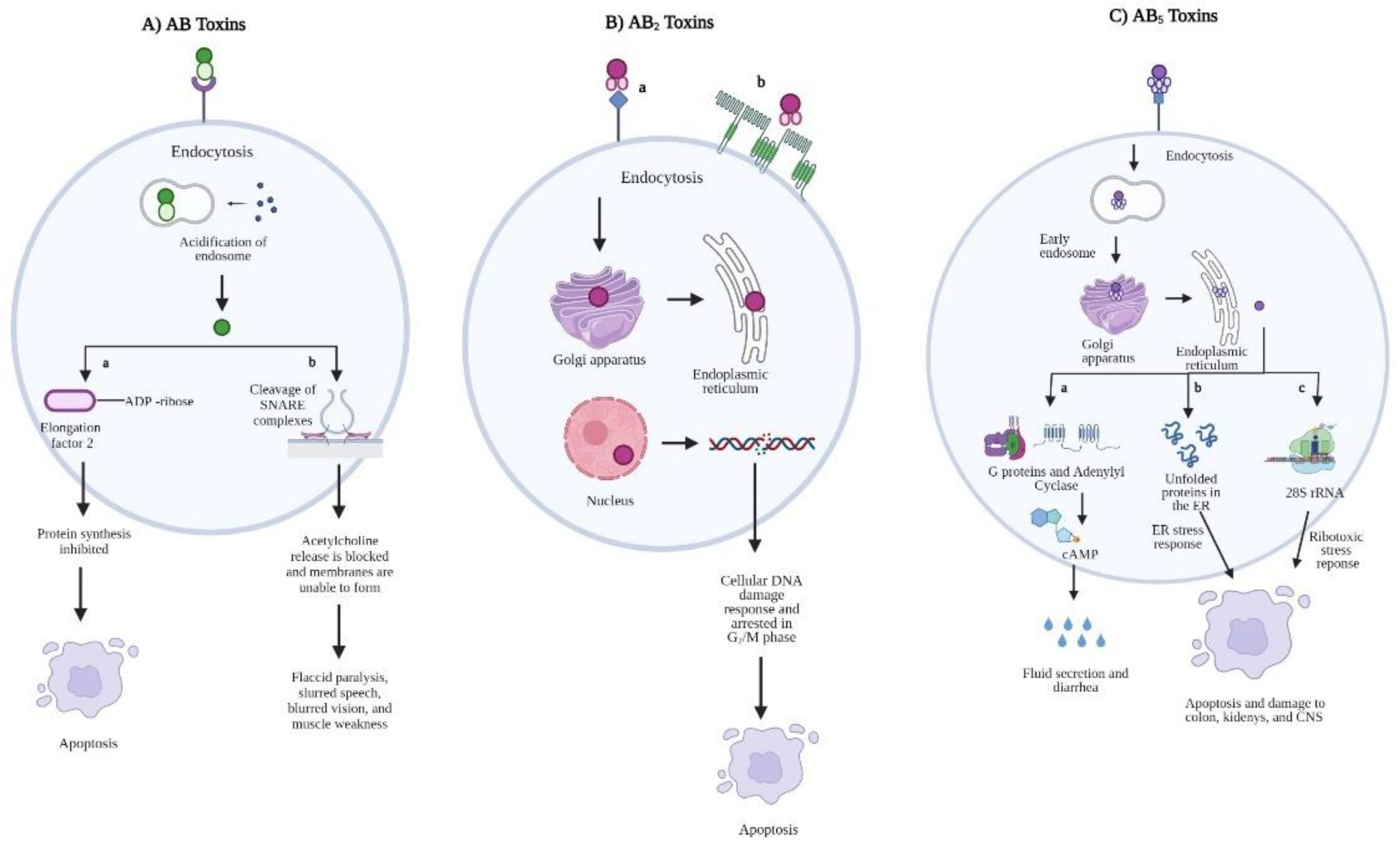
2. AB-Type Toxins
2.1. AB Toxins
2.2. AB2 Toxins
2.3. AB5 Toxins
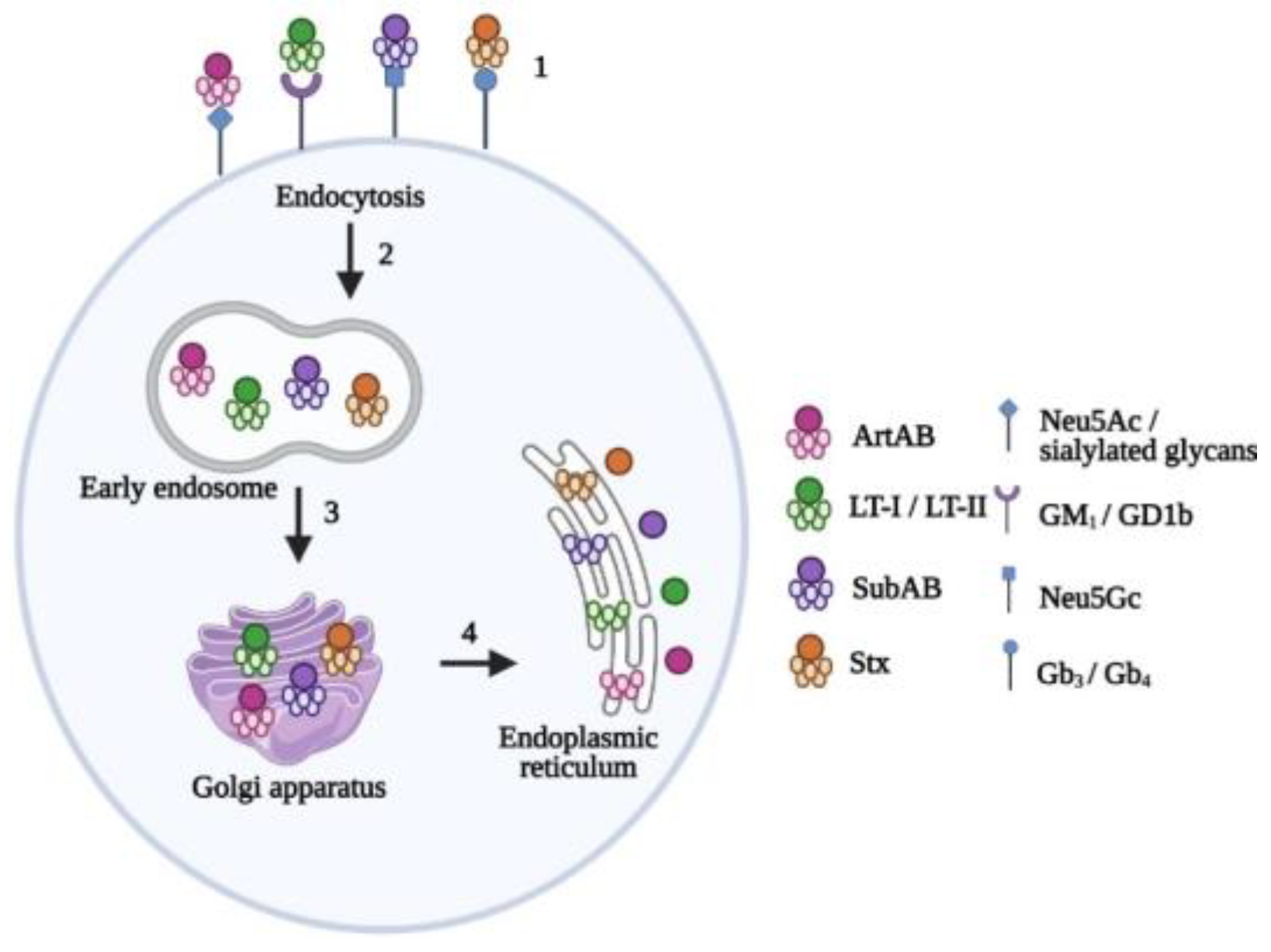
3. AB5 Toxin Families
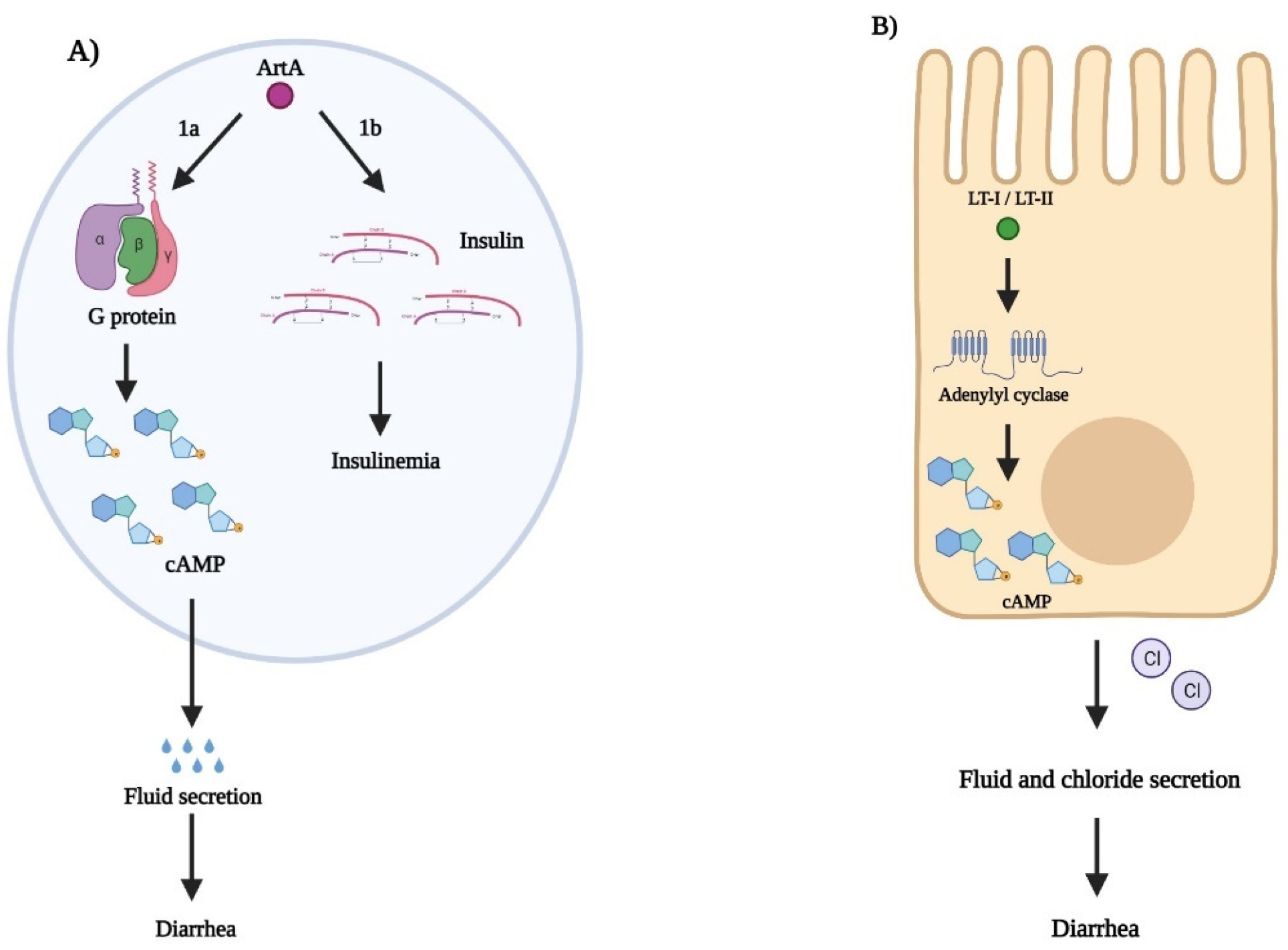
3.1. Pertussis Toxin
3.2. Cholera Toxin
3.3. Subtilase Cytotoxin
3.4. Shiga Toxins
4. Evolution, Lineage, and Stx Variants of E. coli O157:H7
5. Stx Pathogenesis and Disease
6. Stx Detection and STEC Therapies
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Disclaimers
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Burden of Foodborne Illness: Findings. 2018. Available online: https://www.cdc.gov/foodborneburden/2011-foodborne-estimates.html (accessed on 20 October 2020).
- World Health Organization. Food Safety. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/food-safety (accessed on 20 October 2020).
- The World Bank. The Safe Food Imperative: Accelerating Progress in Low- and Middle-Income Countries. 2018. Available online: https://www.worldbank.org/en/topic/agriculture/publication/the-safe-food-imperative-accelerating-progress-in-low-and-middle-income-countries (accessed on 20 October 2020).
- Centers for Disease Control and Prevention. Attribution of Foodborne Illness: Findings. 2018. Available online: https://www.cdc.gov/foodborneburden/attribution/attribution-1998-2008.html (accessed on 20 October 2020).
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Tischendorf, J.; de Avila, R.A.; Safdar, N. Risk of infection following colonization with carbapenem-resistant Enterobactericeae: A systematic review. Am. J. Infect. Control 2016, 44, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Foodborne Diseases Active Surveillance Network (FoodNet). 2020. Available online: https://www.cdc.gov/foodnet/index.html (accessed on 20 October 2020).
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; de Silva, N.R.; Gargouri, N.; et al. World Health Organization Global Estimates and Regional Comparisons of the Burden of Foodborne Disease in 2010. PLoS Med. 2015, 12, e1001923. [Google Scholar] [CrossRef]
- Majowicz, S.E.; Musto, J.; Scallan, E.; Angulo, F.J.; Kirk, M.; O’Brien, S.J.; Jones, T.F.; Fazil, A.; Hoekstra, R.M. The global burden of nontyphoidal Salmonella gastroenteritis. Clin. Infect. Dis. 2010, 50, 882–889. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Yersiniosis- Annual Epidemiological Report 2016 [2014 data]. 2016. Available online: https://www.ecdc.europa.eu/en/publications-data/yersiniosis-annual-epidemiological-report-2016-2014-data (accessed on 30 October 2020).
- Iovleva, A.; Doi, Y. Carbapenem-Resistant Enterobacteriaceae. Clin. Lab. Med. 2017, 37, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. AmpC beta-lactamases. Clin. Microbiol. Rev. 2009, 22, 161–182. [Google Scholar] [CrossRef] [PubMed]
- Guiney, D.; Fierer, J. The Role of the spv Genes in Salmonella Pathogenesis. Front. Microbiol. 2011, 2, 129. [Google Scholar] [CrossRef] [PubMed]
- Maldonado-Contreras, A.; Birtley, J.R.; Boll, E.; Zhao, Y.; Mumy, K.L.; Toscano, J.; Ayehunie, S.; Reinecker, H.C.; Stern, L.J.; McCormick, B.A. Shigella depends on SepA to destabilize the intestinal epithelial integrity via cofilin activation. Gut Microbes 2017, 8, 544–560. [Google Scholar] [CrossRef]
- Mellies, J.L.; Barron, A.M.; Carmona, A.M. Enteropathogenic and enterohemorrhagic Escherichia coli virulence gene regulation. Infect. Immun. 2007, 75, 4199–4210. [Google Scholar] [CrossRef]
- Ruiz, R.C.; Melo, K.C.; Rossato, S.S.; Barbosa, C.M.; Corrêa, L.M.; Elias, W.P.; Piazza, R.M. Atypical enteropathogenic Escherichia coli secretes plasmid encoded toxin. Biomed Res. Int. 2014, 2014, 896235. [Google Scholar] [CrossRef]
- Scaglione, P.; Nemec, K.N.; Burlingame, K.E.; Grabon, A.; Huerta, J.; Navarro-Garcia, F.; Tatulian, S.A.; Teter, K. Structural characteristics of the plasmid-encoded toxin from enteroaggregative Escherichia coli. Biochemistry 2008, 47, 9582–9591. [Google Scholar] [CrossRef] [PubMed]
- Waldor, M.K.; Mekalanos, J.J. Lysogenic Conversion by a Filamentous Phage Encoding Cholera Toxin. Science 1996, 272, 1910–1914. [Google Scholar] [CrossRef] [PubMed]
- Moreno Switt, A.I.; den Bakker, H.C.; Cummings, C.A.; Rodriguez-Rivera, L.D.; Govoni, G.; Raneiri, M.L.; Degoricija, L.; Brown, S.; Hoelzer, K.; Peters, J.E.; et al. Identification and characterization of novel Salmonella mobile elements involved in the dissemination of genes linked to virulence and transmission. PLoS ONE 2012, 7, e41247. [Google Scholar] [CrossRef] [PubMed]
- Hiley, L.; Fang, N.X.; Micalizzi, G.R.; Bates, J. Distribution of Gifsy-3 and of variants of ST64B and Gifsy-1 prophages amongst Salmonella enterica Serovar Typhimurium isolates: Evidence that combinations of prophages promote clonality. PLoS ONE 2014, 9, e86203. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Feltrup, T.M.; Kukreja, R.V.; Patel, K.B.; Cai, S.; Singh, B.R. Evolutionary Features in the Structure and Function of Bacterial Toxins. Toxins 2019, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Dressler, D.; Saberi, F.A.; Barbosa, E.R. Botulinum toxin: Mechanisms of action. Arq. Neuropsiquiatr. 2005, 63, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Masuyer, G.; Stenmark, P. Botulinum and Tetanus Neurotoxins. Annu. Rev. Biochem. 2019, 88, 811–837. [Google Scholar] [CrossRef] [PubMed]
- Parveen, S.; Bishai, W.R.; Murphy, J.R. Corynebacterium diphtheriae: Diphtheria Toxin, the tox Operon, and Its Regulation by Fe2(+) Activation of apo-DtxR. Microbiol. Spectr. 2019, 7, GPP3-0063-2019. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.R. Mechanism of diphtheria toxin catalytic domain delivery to the eukaryotic cell cytosol and the cellular factors that directly participate in the process. Toxins 2011, 3, 294–308. [Google Scholar] [CrossRef]
- Collier, R.J. Diphtheria toxin: Mode of action and structure. Bacteriol. Rev. 1975, 39, 54–85. [Google Scholar] [CrossRef] [PubMed]
- Otsuji, K.; Fukuda, K.; Ogawa, M.; Saito, M. Mutation and Diversity of Diphtheria Toxin in Corynebacterium ulcerans. Emerg. Infect. Dis. 2019, 25, 2122–2123. [Google Scholar] [CrossRef]
- Pollack, M. The role of exotoxin A in pseudomonas disease and immunity. Rev. Infect. Dis. 1983, 5 (Suppl. S5), S979–S984. [Google Scholar] [CrossRef]
- Michalska, M.; Wolf, P. Pseudomonas Exotoxin A: Optimized by evolution for effective killing. Front. Microbiol. 2015, 6, 963. [Google Scholar] [CrossRef]
- Morlon-Guyot, J.; Méré, J.; Bonhoure, A.; Beaumelle, B. Processing of Pseudomonas aeruginosa exotoxin A is dispensable for cell intoxication. Infect. Immun. 2009, 77, 3090–3099. [Google Scholar] [CrossRef]
- Pons, J.B.; Vignard, J.; Mirey, G. Cytolethal Distending Toxin Subunit B: A Review of Structure-Function Relationship. Toxins 2019, 11, 595. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Gharaibeh, R.Z.; Newsome, R.C.; Pope, J.L.; Dougherty, M.W.; Tomkovich, S.; Pons, B.; Mirey, G.; Vignard, J.; Hendrixson, D.R.; et al. Campylobacter jejuni promotes colorectal tumorigenesis through the action of cytolethal distending toxin. Gut 2019, 68, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.X.; Chen, Y.; Fu, R.; Mao, G.Q.; Liu, S.Y.; Shen, T.B. Rab5a Promotes Cytolethal Distending Toxin B-Induced Cytotoxicity and Inflammation. Infect. Immun. 2020, 88, e00132-20. [Google Scholar] [CrossRef] [PubMed]
- Pons, B.J.; Loiseau, N.; Hashim, S.; Tadrist, S.; Mirey, G.; Vignard, J. Functional Study of Haemophilus ducreyi Cytolethal Distending Toxin Subunit B. Toxins 2020, 12, 530. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.J.; Bayles, D.O. The contribution of cytolethal distending toxin to bacterial pathogenesis. Crit. Rev. Microbiol. 2006, 32, 227–248. [Google Scholar] [CrossRef] [PubMed]
- Bharati, K.; Ganguly, N.K. Cholera toxin: A paradigm of a multifunctional protein. Indian J. Med. Res. 2011, 133, 179–187. [Google Scholar]
- Broeck, V.D.; Horvath, C.; de Wolf, M.J.S. Vibrio cholerae: Cholera toxin. Int. J. Biochem. Cell Biol. 2007, 39, 1771–1775. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, J.; Holmgren, J. Cholera toxin structure, gene regulation and pathophysiological and immunological aspects. Cell Mol. Life Sci. 2008, 65, 1347–1360. [Google Scholar] [CrossRef]
- Baldauf, K.J.; Royal, J.M.; Hamorsky, K.T.; Matoba, N. Cholera toxin B: One subunit with many pharmaceutical applications. Toxins 2015, 7, 974–996. [Google Scholar] [CrossRef]
- Kirkeby, S.; Pedersen, A.M.L. Modifications of cholera toxin subunit B binding to human large intestinal epithelium. An immunohistochemical study. Microb. Pathog. 2018, 124, 332–336. [Google Scholar] [CrossRef]
- Scanlon, K.; Skerry, C.; Carbonetti, N. Association of Pertussis Toxin with Severe Pertussis Disease. Toxins 2019, 11, 373. [Google Scholar] [CrossRef] [PubMed]
- Teter, K. Intracellular Trafficking and Translocation of Pertussis Toxin. Toxins 2019, 11, 437. [Google Scholar] [CrossRef] [PubMed]
- Mangmool, S.; Kurose, H. G(i/o) protein-dependent and -independent actions of Pertussis Toxin (PTX). Toxins 2011, 3, 884–899. [Google Scholar] [CrossRef]
- Chen, Q.; Boucher, P.E.; Stibitz, S. Multiple weak interactions between BvgA~P and ptx promoter DNA strongly activate transcription of pertussis toxin genes in Bordetella pertussis. PLoS Pathog. 2020, 16, e1008500. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Deng, L.; Stack, G.; Yu, H.; Chen, X.; Naito-Matsui, Y.; Varki, A.; Galán, J.E. Evolution of host adaptation in the Salmonella typhoid toxin. Nat. Microbiol. 2017, 2, 1592–1599. [Google Scholar] [CrossRef]
- Tamamura, Y.; Tanaka, K.; Uchida, I. Characterization of pertussis-like toxin from Salmonella spp. that catalyzes ADP-ribosylation of G proteins. Sci. Rep. 2017, 7, 2653. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, E.; Morris, B.; Mohammad Mousa, O.; Price, E.; Rodriguez, A.; Cufurovic, L.; Beard, R.S.; Tinker, J.K. Cellular Activity of Salmonella Typhimurium ArtAB Toxin and Its Receptor-Binding Subunit. Toxins 2021, 13, 599. [Google Scholar] [CrossRef]
- Wang, H.; Paton, J.C.; Herdman, B.P.; Rogers, T.J.; Beddoe, T.; Paton, A.W. The B subunit of an AB5 toxin produced by Salmonella enterica serovar Typhi up-regulates chemokines, cytokines, and adhesion molecules in human macrophage, colonic epithelial, and brain microvascular endothelial cell lines. Infect. Immun. 2013, 81, 673–683. [Google Scholar] [CrossRef]
- Herdman, B.P.; Paton, J.C.; Wang, H.; Beddoe, T.; Paton, A.W. Vacuolation Activity and Intracellular Trafficking of ArtB, the Binding Subunit of an AB5 Toxin Produced by Salmonella enterica Serovar Typhi. Infect. Immun. 2017, 85, e00214-17. [Google Scholar] [CrossRef] [PubMed]
- Miura, S.; Tamamura, Y.; Takayasu, M.; Sasaki, M.; Nishimura, N.; Tokugawa, K.; Suwa, I.; Murata, R.; Akiba, M.; Kusumoto, M.; et al. Influence of SOS-inducing agents on the expression of ArtAB toxin gene in Salmonella enterica and Salmonella bongori. Microbiology 2020, 166, 785–793. [Google Scholar] [CrossRef]
- Littler, D.R.; Ang, S.Y.; Moriel, D.G.; Kocan, M.; Kleifeld, O.; Johnson, M.D.; Tran, M.T.; Paton, A.W.; Paton, J.C.; Summers, R.J.; et al. Structure-function analyses of a pertussis-like toxin from pathogenic Escherichia coli reveal a distinct mechanism of inhibition of trimeric G-proteins. J. Biol. Chem. 2017, 292, 15143–15158. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Barbieri, J.T. When Escherichia coli doesn’t fit the mold: A pertussis-like toxin with altered specificity. J. Biol. Chem. 2017, 292, 15159–15160. [Google Scholar] [CrossRef]
- Joffré, E.; Sjöling, Å. The LT1 and LT2 variants of the enterotoxigenic Escherichia coli (ETEC) heat-labile toxin (LT) are associated with major ETEC lineages. Gut Microbes 2016, 7, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Duan, Q.; Zhang, W. Significance of Enterotoxigenic Escherichia coli (ETEC) Heat-Labile Toxin (LT) Enzymatic Subunit Epitopes in LT Enterotoxicity and Immunogenicity. Appl. Environ. Microbiol. 2018, 84, e00849-18. [Google Scholar] [CrossRef] [PubMed]
- Ng, N.M.; Littler, D.R.; Paton, A.W.; Le Nours, J.; Rossjohn, J.; Paton, J.C.; Beddoe, T. EcxAB is a founding member of a new family of metalloprotease AB5 toxins with a hybrid cholera-like B subunit. Structure 2013, 21, 2003–2013. [Google Scholar] [CrossRef]
- Ng, N.; Littler, D.; Le Nours, J.; Paton, A.W.; Paton, J.C.; Rossjohn, J.; Beddoe, T. Cloning, expression, purification and preliminary X-ray diffraction studies of a novel AB5 toxin. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2013, 69 Pt 8, 912–915. [Google Scholar] [CrossRef] [PubMed]
- Paton, A.W.; Srimanote, P.; Talbot, U.M.; Wang, H.; Paton, J.C. A new family of potent AB(5) cytotoxins produced by Shiga toxigenic Escherichia coli. J. Exp. Med. 2004, 200, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Paton, W.A.; Paton, J.C. Escherichia coli Subtilase Cytotoxin. Toxins 2010, 2, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Chong, D.C.; Paton, J.C.; Thorpe, C.M.; Paton, A.W. Clathrin-dependent trafficking of subtilase cytotoxin, a novel AB5 toxin that targets the endoplasmic reticulum chaperone BiP. Cell Microbiol. 2008, 10, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, G.; Morinaga, N.; Yahiro, K.; Komine, R.; Moss, J.; Yoshida, H.; Noda, M. Novel subtilase cytotoxin produced by Shiga-toxigenic Escherichia coli induces apoptosis in vero cells via mitochondrial membrane damage. Infect. Immun. 2009, 77, 2919–2924. [Google Scholar] [CrossRef]
- Irino, K.; Vieira, M.A.; Gomes, T.A.; Guth, B.E.; Naves, Z.V.; Oliveira, M.G.; dos Santos, L.F.; Guirro, M.; Timm, C.D.; Pigatto, C.P.; et al. Subtilase cytotoxin-encoding subAB operon found exclusively among Shiga toxin-producing Escherichia coli strains. J. Clin. Microbiol. 2010, 48, 988–990. [Google Scholar] [CrossRef] [PubMed]
- Palma-Martínez, I.; Guerrero-Mandujano, A.; Ruiz-Ruiz, M.J.; Hernández-Cortez, C.; Molina-López, J.; Bocanegra-García, V.; Castro-Escarpulli, G. Active Shiga-Like Toxin Produced by Some Aeromonas spp., Isolated in Mexico City. Front. Microbiol. 2016, 7, 1522. [Google Scholar] [CrossRef]
- Bergan, J.; Dyve Lingelem, A.B.; Simm, R.; Skotland, T.; Sandvig, K. Shiga toxins. Toxicon 2012, 60, 1085–1107. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.Y.; Ng, T.B. Shiga toxins: From structure and mechanism to applications. Appl. Microbiol. Biotechnol. 2016, 100, 1597–1610. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Strockbine, N.; Omondi, M.; Hise, K.; Fair, M.A.; Mintz, E. Emergence of Shiga toxin 1 genes within Shigella dysenteriae type 4 isolates from travelers returning from the Island of Hispañola. Am. J. Trop. Med. Hyg. 2007, 76, 1163–1165. [Google Scholar] [CrossRef]
- Carter, C.C.; Fierer, J.; Chiu, W.W.; Looney, D.J.; Strain, M.; Mehta, S.R. A Novel Shiga Toxin 1a-Converting Bacteriophage of Shigella sonnei With Close Relationship to Shiga Toxin 2-Converting Pages of Escherichia coli. Open Forum Infect. Dis. 2016, 3, ofw079. [Google Scholar] [CrossRef] [PubMed]
- Paton, C.J.; Paton, A.W. Pathogenesis and diagnosis of Shiga toxin-producing Escherichia coli infections. Clin. Microbiol. Rev. 1998, 11, 450–479. [Google Scholar] [CrossRef]
- Zuverink, M.; Barbieri, J.T. Protein Toxins That Utilize Gangliosides as Host Receptors. Prog. Mol. Biol. Transl. Sci. 2018, 156, 325–354. [Google Scholar] [CrossRef] [PubMed]
- Nowakowska-Gołacka, J.; Sominka, H.; Sowa-Rogozińska, N.; Słomińska-Wojewódzka, M. Toxins Utilize the Endoplasmic Reticulum-Associated Protein Degradation Pathway in Their Intoxication Process. Int. J. Mol. Sci. 2019, 20, 1307. [Google Scholar] [CrossRef] [PubMed]
- Peng Chen, Z.; Morris, J.G., Jr.; Rodriguez, R.L.; Wagle Shukla, A.; Tapia-Núñez, J.; Okun, M.S. Emerging opportunities for serotypes of botulinum neurotoxins. Toxins 2012, 4, 1196–1222. [Google Scholar] [CrossRef]
- Johnson, M.W.; Lior, H. Production of Shiga toxin and a cytolethal distending toxin (CLDT) by serogroups of Shigella spp. FEMS Microbiol. Lett. 1987, 48, 235–238. [Google Scholar] [CrossRef]
- Mendez-Olvera, E.T.; Bustos-Martinez, J.A.; Lopez-Vidal, Y.; Verdugo-Rodriguez, A.; Martinez-Gomez, D. Cytolethal Distending Toxin From Campylobacter jejuni Requires the Cytoskeleton for Toxic Activity. Jundishapur J. Microbiol. 2016, 9, e35591. [Google Scholar] [CrossRef]
- Hickey, T.E.; McVeigh, A.L.; Scott, D.A.; Michielutti, R.E.; Bixby, A.; Carroll, S.A.; Bourgeois, A.L.; Guerry, P. Campylobacter jejuni cytolethal distending toxin mediates release of interleukin-8 from intestinal epithelial cells. Infect. Immun. 2000, 68, 6535–6541. [Google Scholar] [CrossRef]
- Lai, C.K.; Chen, Y.A.; Lin, C.J.; Lin, H.J.; Kao, M.C.; Huang, M.Z.; Lin, Y.H.; Chiang-Ni, C.; Chen, C.J.; Lo, U.G.; et al. Molecular Mechanisms and Potential Clinical Applications of Campylobacter jejuni Cytolethal Distending Toxin. Front. Cell. Infect. Microbiol. 2016, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Jinadasa, R.N.; Bloom, S.E.; Weiss, R.S.; Duhamel, G.E. Cytolethal distending toxin: A conserved bacterial genotoxin that blocks cell cycle progression, leading to apoptosis of a broad range of mammalian cell lineages. Microbiology 2011, 157 Pt 7, 1851–1875. [Google Scholar] [CrossRef] [PubMed]
- Jeon, B.I.K.; Ryu, S. Promoter analysis of cytolethal distending toxin genes (cdtA, B, and C ) and effect of a luxS mutation on CDT production in Campylobacter jejuni. Microbiol. Immunol. 2005, 49, 599–603. [Google Scholar] [CrossRef]
- Beddoe, T.; Paton, A.W.; Le Nours, J.; Rossjohn, J.; Paton, J.C. Structure, biological functions and applications of the AB5 toxins. Trends Biochem. Sci. 2010, 35, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, M.; Tanaka, K.; Nishimori, K.; Makino, S.-i.; Kanno, T.; Ishihara, R.; Hatama, S.; Kitano, R.; Kishima, M.; Sameshima, T.; et al. The artAB genes encode a putative ADP-ribosyltransferase toxin homologue associated with Salmonella enterica serovar Typhimurium DT104. Microbiology 2005, 151, 3089–3096. [Google Scholar] [CrossRef]
- Cheng, A.R.; Wiedmann, M. The ADP-Ribosylating Toxins of Salmonella. Toxins 2019, 11, 416. [Google Scholar] [CrossRef] [PubMed]
- Uchida, I.; Ishihara, R.; Tanaka, K.; Hata, E.; Makino, S.I.; Kanno, T.; Hatama, S.; Kishima, M.; Akiba, M.; Watanabe, A.; et al. Salmonella enterica serotype Typhimurium DT104 ArtA-dependent modification of pertussis toxin-sensitive G proteins in the presence of [32P]NAD. Microbiology 2009, 155 Pt 11, 3710–3718. [Google Scholar] [CrossRef]
- Russo, A.T.; Johnson, J.R. Medical and economic impact of extraintestinal infections due to Escherichia coli: Focus on an increasingly important endemic problem. Microbes Infect. 2003, 5, 449–456. [Google Scholar] [CrossRef]
- Qadri, F.; Svennerholm, A.M.; Faruque, A.S.; Sack, R.B. Enterotoxigenic Escherichia coli in developing countries: Epidemiology, microbiology, clinical features, treatment, and prevention. Clin. Microbiol. Rev. 2005, 18, 465–483. [Google Scholar] [CrossRef] [PubMed]
- Duan, Q.; Xia, P.; Nandre, R.; Zhang, W.; Zhu, G. Review of Newly Identified Functions Associated With the Heat-Labile Toxin of Enterotoxigenic Escherichia coli. Front. Cell. Infect. Microbiol. 2019, 9, 292. [Google Scholar] [CrossRef]
- Viswanathan, V.K.; Hodges, K.; Hecht, G. Enteric infection meets intestinal function: How bacterial pathogens cause diarrhoea. Nat. Rev. Microbiol. 2009, 7, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, L.; Ali, Z.B.; Nygren, E.; Wang, Z.; Karlsson, S.; Zhu, B.; Quiding-Jarbrink, M.; Sjoling, A. Alkaline pH Is a signal for optimal production and secretion of the heat labile toxin, LT in enterotoxigenic Escherichia coli (ETEC). PLoS ONE 2013, 8, e74069. [Google Scholar] [CrossRef] [PubMed]
- Karasawa, T.; Ito, H.; Tsukamoto, T.; Yamasaki, S.; Kurazono, H.; Faruque, S.M.; Nair, G.B.; Nishibuchi, M.; Takeda, Y. Cloning and characterization of genes encoding homologues of the B subunit of cholera toxin and the Escherichia coli heat-labile enterotoxin from clinical isolates of Citrobacter freundii and E. coli. Infect. Immun. 2002, 70, 7153–7155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Paton, A.W.; Woodrow, M.C.; Doyle, R.M.; Lanser, J.A.; Paton, J.C. Molecular characterization of a Shiga toxigenic Escherichia coli O113:H21 strain lacking eae responsible for a cluster of cases of hemolytic-uremic syndrome. J. Clin. Microbiol. 1999, 37, 3357–3361. [Google Scholar] [CrossRef]
- Cergole-Novella, M.C.; Nishimura, L.S.; Dos Santos, L.F.; Irino, K.; Vaz, T.M.; Bergamini, A.M.; Guth, B.E. Distribution of virulence profiles related to new toxins and putative adhesins in Shiga toxin-producing Escherichia coli isolated from diverse sources in Brazil. FEMS Microbiol. Lett. 2007, 274, 329–334. [Google Scholar] [CrossRef]
- Karama, M.; Johnson, R.P.; Holtslander, R.; McEwen, S.A.; Gyles, C.L. Prevalence and characterization of verotoxin-producing Escherichia coli (VTEC) in cattle from an Ontario abattoir. Can. J. Vet. Res. 2008, 72, 297–302. [Google Scholar]
- Heinisch, L.; Zoric, K.; Krause, M.; Schmidt, H. Transcription of the Subtilase Cytotoxin Gene subAB 1 in Shiga Toxin-Producing Escherichia coli Is Dependent on hfq and hns. Appl. Environ. Microbiol. 2019, 85, e01281-19. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Paton, J.C.; Paton, A.W. Pathologic changes in mice induced by subtilase cytotoxin, a potent new Escherichia coli AB5 toxin that targets the endoplasmic reticulum. J. Infect. Dis. 2007, 196, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Velandia, C.V.; Mariel Sanso, A.; Krüger, A.; Suárez, L.V.; Lucchesi, P.M.; Parma, A.E. Occurrence of subtilase cytotoxin and relation with other virulence factors in verocytotoxigenic Escherichia coli isolated from food and cattle in Argentina. Braz. J. Microbiol. 2011, 42, 711–715. [Google Scholar] [CrossRef]
- Paton, W.A.; Paton, J.C. Multiplex PCR for direct detection of Shiga toxigenic Escherichia coli strains producing the novel subtilase cytotoxin. J. Clin. Microbiol. 2005, 43, 2944–2947. [Google Scholar] [CrossRef] [PubMed]
- Shiga, K. Ueber den Erreger der Dysenterie in Japan. Cent. Bakteriol. Parasiteukunde Infektionskrank 1898, 23, 599–600. [Google Scholar]
- Trofa, A.F.; Ueno-Olsen, H.; Oiwa, R.; Yoshikawa, M. Dr. Kiyoshi Shiga: Discoverer of the Dysentery Bacillus. Clin. Infect. Dis. 1999, 29, 1303–1306. [Google Scholar] [CrossRef] [PubMed]
- Strauch, E.; Lurz, R.; Beutin, L. Characterization of a Shiga toxin-encoding temperate bacteriophage of Shigella sonnei. Infect. Immun. 2001, 69, 7588–7595. [Google Scholar] [CrossRef]
- Gray, M.D.; Lampel, K.A.; Strockbine, N.A.; Fernandez, R.E.; Melton-Celsa, A.R.; Maurelli, A.T. Clinical isolates of Shiga toxin 1a-producing Shigella flexneri with an epidemiological link to recent travel to Hispañiola. Emerg. Infect. Dis. 2014, 20, 1669–1677. [Google Scholar] [CrossRef] [PubMed]
- Fogolari, M.; Mavian, C.; Angeletti, S.; Salemi, M.; Lampel, K.A.; Maurelli, A.T. Distribution and characterization of Shiga toxin converting temperate phages carried by Shigella flexneri in Hispaniola. Infect. Genet. Evol. 2018, 65, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Unkmeir, A.; Schmidt, H. Structural analysis of phage-borne stx genes and their flanking sequences in shiga toxin-producing Escherichia coli and Shigella dysenteriae type 1 strains. Infect. Immun. 2000, 68, 4856–4864. [Google Scholar] [CrossRef] [PubMed]
- McDonough, A.M.; Butterton, R.J. Spontaneous tandem amplification and deletion of the Shiga toxin operon in Shigella dysenteriae 1. Mol. Microbiol. 1999, 34, 1058–1069. [Google Scholar] [CrossRef]
- Sandvig, K.; Garred, Ø.; Prydz, K.; Kozlov, J.V.; Hansen, S.H.; van Deurs, B. Retrograde transport of endocytosed Shiga toxin to the endoplasmic reticulum. Nature 1992, 358, 510–512. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-Y.; Lee, M.-S.; Cherla, R.P.; Tesh, V.L. Shiga toxin 1 induces apoptosis through the endoplasmic reticulum stress response in human monocytic cells. Cell. Microbiol. 2008, 10, 770–780. [Google Scholar] [CrossRef]
- Melton-Celsa, A.R. Shiga Toxin (Stx) Classification, Structure, and Function. Microbiol. Spectr. 2014, 2, EHEC-0024-2013. [Google Scholar] [CrossRef]
- Endo, Y.; Tsurugi, K.; Yutsudo, T.; Takeda, Y.; Ogasawara, T.; Igarashi, K. Site of action of a Vero toxin (VT2) from Escherichia coli O157:H7 and of Shiga toxin on eukaryotic ribosomes. Eur. J. Biochem. 1988, 171, 45–50. [Google Scholar] [CrossRef]
- Colpoys, W.E.; Cochran, B.H.; Carducci, T.M.; Thorpe, C.M. Shiga toxins activate translational regulation pathways in intestinal epithelial cells. Cell Signal. 2005, 17, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Jandhyala, M.D.; Thorpe, C.M.; Magun, B. Ricin and Shiga toxins: Effects on host cell signal transduction. Curr. Top. Microbiol. Immunol. 2012, 357, 41–65. [Google Scholar] [CrossRef] [PubMed]
- Riley, L.W.; Remis, R.S.; Helgerson, S.D.; McGee, H.B.; Wells, J.G.; Davis, B.R.; Hebert, R.J.; Olcott, E.S.; Johnson, L.M.; Hargrett, N.T.; et al. Hemorrhagic Colitis Associated with a Rare Escherichia coli Serotype. N. Engl. J. Med. 1983, 308, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.G.; Davis, B.R.; Wachsmuth, I.K.; Riley, L.W.; Remis, R.S.; Sokolow, R.; Morris, G.K. Laboratory investigation of hemorrhagic colitis outbreaks associated with a rare Escherichia coli serotype. J. Clin. Microbiol. 1983, 18, 512–520. [Google Scholar] [CrossRef]
- Proulx, F.; Seidman, E.G.; Karpman, D. Pathogenesis of Shiga toxin-associated hemolytic uremic syndrome. Pediatr. Res. 2001, 50, 163–171. [Google Scholar] [CrossRef] [PubMed]
- King, A.J.; Sundaram, S.; Cendoroglo, M.; Acheson, D.W.K.; Keusch, G.T. Shiga Toxin Induces Superoxide Production in Polymorphonuclear Cells with Subsequent Impairment of Phagocytosis and Responsiveness to Phorbol Esters. J. Infect. Dis. 1999, 179, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, R.A.; Sperandio, V. Shiga toxin in enterohemorrhagic E.coli: Regulation and novel anti-virulence strategies. Front. Cell. Infect. Microbiol. 2012, 2, 81. [Google Scholar] [CrossRef]
- Wlodarska, M.; Willing, B.; Keeney, K.M.; Menendez, A.; Bergstrom, K.S.; Gill, N.; Russell, S.L.; Vallance, B.A.; Finlay, B.B. Antibiotic treatment alters the colonic mucus layer and predisposes the host to exacerbated Citrobacter rodentium-induced colitis. Infect. Immun. 2011, 79, 1536–1545. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Yin, X.; Yu, H.; Feng, Y.; Ying, X.; Gong, J.; Gyles, C.L. Alteration of the Microbiota and Virulence Gene Expression in E. coli O157:H7 in Pig Ligated Intestine with and without AE Lesions. PLoS ONE 2015, 10, e0130272. [Google Scholar] [CrossRef]
- De Sablet, T.; Chassard, C.; Bernalier-Donadille, A.; Vareille, M.; Gobert, A.P.; Martin, C. Human microbiota-secreted factors inhibit shiga toxin synthesis by enterohemorrhagic Escherichia coli O157:H7. Infect. Immun. 2009, 77, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Cameron, E.A.; Curtis, M.M.; Kumar, A.; Dunny, G.M.; Sperandio, V. Microbiota and Pathogen Proteases Modulate Type III Secretion Activity in Enterohemorrhagic Escherichia coli. mBio 2018, 9, e02204-18. [Google Scholar] [CrossRef]
- Pacheco, A.R.; Curtis, M.M.; Ritchie, J.M.; Munera, D.; Waldor, M.K.; Moreira, C.G.; Sperandio, V. Fucose sensing regulates bacterial intestinal colonization. Nature 2012, 492, 113–117. [Google Scholar] [CrossRef]
- Njoroge, J.W.; Nguyen, Y.; Curtis, M.M.; Moreira, C.G.; Sperandio, V. Virulence meets metabolism: Cra and KdpE gene regulation in enterohemorrhagic Escherichia coli. mBio 2012, 3, e00280-12. [Google Scholar] [CrossRef]
- Cordonnier, C.; Le Bihan, G.; Emond-Rheault, J.G.; Garrivier, A.; Harel, J.; Jubelin, G. Vitamin B12 Uptake by the Gut Commensal Bacteria Bacteroides thetaiotaomicron Limits the Production of Shiga Toxin by Enterohemorrhagic Escherichia coli. Toxins 2016, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Pifer, R.; Sperandio, V. The Interplay between the Microbiota and Enterohemorrhagic Escherichia coli. Microbiol. Spectr. 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Curtis, M.M.; Hu, Z.; Klimko, C.; Narayanan, S.; Deberardinis, R.; Sperandio, V. The gut commensal Bacteroides thetaiotaomicron exacerbates enteric infection through modification of the metabolic landscape. Cell Host Microbe 2014, 16, 759–769. [Google Scholar] [CrossRef]
- Nakanishi, N.; Tashiro, K.; Kuhara, S.; Hayashi, T.; Sugimoto, N.; Tobe, T. Regulation of virulence by butyrate sensing in enterohaemorrhagic Escherichia coli. Microbiology 2009, 155 Pt 2, 521–530. [Google Scholar] [CrossRef]
- Zumbrun, S.D.; Melton-Celsa, A.R.; Smith, M.A.; Gilbreath, J.J.; Merrell, D.S.; O’Brien, A.D. Dietary choice affects Shiga toxin-producing Escherichia coli (STEC) O157:H7 colonization and disease. Proc. Natl. Acad. Sci. USA 2013, 110, E2126–E2133. [Google Scholar] [CrossRef]
- Tobe, T.; Nakanishi, N.; Sugimoto, N. Activation of motility by sensing short-chain fatty acids via two steps in a flagellar gene regulatory cascade in enterohemorrhagic Escherichia coli. Infect. Immun. 2011, 79, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Nawrocki, M.E.; Mosso, H.M.; Dudley, E.G. A Toxic Environment: A Growing Understanding of How Microbial Communities Affect Escherichia coli O157:H7 Shiga Toxin Expression. Appl. Environ. Microbiol. 2020, 86, e00509-20. [Google Scholar] [CrossRef] [PubMed]
- Toshima, H.; Yoshimura, A.; Arikawa, K.; Hidaka, A.; Ogasawara, J.; Hase, A.; Masaki, H.; Nishikawa, Y. Enhancement of Shiga Toxin Production in Enterohemorrhagic Escherichia coli Serotype O157:H7 by DNase Colicins. Appl. Environ. Microbiol. 2007, 73, 7582–7588. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mosso, H.M.; Xiaoli, L.; Banerjee, K.; Hoffmann, M.; Yao, K.; Dudley, E.G.; DiRita, V.J. A Putative Microcin Amplifies Shiga Toxin 2a Production of Escherichia coli O157:H7. J. Bacteriol. 2019, 202, e00353-19. [Google Scholar] [CrossRef]
- Tovaglieri, A.; Sontheimer-Phelps, A.; Geirnaert, A.; Prantil-Baun, R.; Camacho, D.M.; Chou, D.B.; Jalili-Firoozinezhad, S.; de Wouters, T.; Kasendra, M.; Super, M.; et al. Species-specific enhancement of enterohemorrhagic E. coli pathogenesis mediated by microbiome metabolites. Microbiome 2019, 7, 43. [Google Scholar] [CrossRef]
- Croxen, M.A.; Law, R.J.; Scholz, R.; Keeney, K.M.; Wlodarska, M.; Finlay, B.B. Recent advances in understanding enteric pathogenic Escherichia coli. Clin. Microbiol. Rev. 2013, 26, 822–880. [Google Scholar] [CrossRef] [PubMed]
- Whittam, T.S.; Wolfe, M.L.; Wachsmuth, I.K.; Orskov, F.; Orskov, I.; Wilson, R.A. Clonal relationships among Escherichia coli strains that cause hemorrhagic colitis and infantile diarrhea. Infect. Immun. 1993, 61, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Lacher, D.W.; Steinsland, H.; Blank, T.E.; Donnenberg, M.S.; Whittam, T.S. Molecular evolution of typical enteropathogenic Escherichia coli: Clonal analysis by multilocus sequence typing and virulence gene allelic profiling. J. Bacteriol. 2007, 189, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Blanco, M.; Blanco, J.E.; Mora, A.; Dahbi, G.; Alonso, M.P.; González, E.A.; Bernárdez, M.I.; Blanco, J. Serotypes, virulence genes, and intimin types of Shiga toxin (verotoxin)-producing Escherichia coli isolates from cattle in Spain and identification of a new intimin variant gene (eae-xi). J. Clin. Microbiol. 2004, 42, 645–651. [Google Scholar] [CrossRef]
- Levine, M.M. Escherichia coli that cause diarrhea: Enterotoxigenic, enteropathogenic, enteroinvasive, enterohemorrhagic, and enteroadherent. J. Infect. Dis. 1987, 155, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Lan, R.; Alles, M.C.; Donohoe, K.; Martinez, M.B.; Reeves, P.R. Molecular evolutionary relationships of enteroinvasive Escherichia coli and Shigella spp. Infect. Immun. 2004, 72, 5080–5088. [Google Scholar] [CrossRef]
- Lan, R.; Reeves, P.R. Escherichia coli in disguise: Molecular origins of Shigella. Microbes Infect. 2002, 4, 1125–1132. [Google Scholar] [CrossRef]
- Johnson, J.T.; Nolan, L.K. Pathogenomics of the virulence plasmids of Escherichia coli. Microbiol. Mol. Biol. Rev. 2009, 73, 750–774. [Google Scholar] [CrossRef]
- Cheasty, T.; Rowe, B. Antigenic relationships between the enteroinvasive Escherichia coli O antigens O28ac, O112ac, O124, O136, O143, O144, O152, and O164 and Shigella O antigens. J. Clin. Microbiol. 1983, 17, 681–684. [Google Scholar] [CrossRef]
- Nataro, J.P.; Kaper, J.B.; Robins-Browne, R.; Prado, V.; Vial, P.; Levine, M.M. Patterns of adherence of diarrheagenic Escherichia coli to HEp-2 cells. Pediatr. Infect. Dis. J. 1987, 6, 829–831. [Google Scholar] [CrossRef] [PubMed]
- Grad, Y.H.; Lipsitch, M.; Feldgarden, M.; Arachchi, H.M.; Cerqueira, G.C.; Fitzgerald, M.; Godfrey, P.; Haas, B.J.; Murphy, C.I.; Russ, C. Genomic epidemiology of the Escherichia coli O104:H4 outbreaks in Europe 2011. Proc. Natl. Acad. Sci. USA 2012, 109, 3065–3070. [Google Scholar] [CrossRef] [PubMed]
- Isidean, S.D.; Riddle, M.S.; Savarino, S.J.; Porter, C.K. A systematic review of ETEC epidemiology focusing on colonization factor and toxin expression. Vaccine 2011, 29, 6167–6178. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.M.; Chaudhuri, R.R.; Jiang, Z.D.; DuPont, H.; Gyles, C.; Penn, C.W.; Pallen, M.J.; Henderson, I.R. Phylogenetic comparisons reveal multiple acquisitions of the toxin genes by enterotoxigenic Escherichia coli strains of different evolutionary lineages. J. Clin. Microbiol. 2006, 44, 4528–4536. [Google Scholar] [CrossRef] [PubMed]
- Scaletsky, C.I.; Silva, M.L.; Trabulsi, L.R. Distinctive patterns of adherence of enteropathogenic Escherichia coli to HeLa cells. Infect. Immun. 1984, 45, 534–536. [Google Scholar] [CrossRef] [PubMed]
- Bouguénec, L.C.; Servin, A.L. Diffusely adherent Escherichia coli strains expressing Afa/Dr adhesins (Afa/Dr DAEC): Hitherto unrecognized pathogens. FEMS Microbiol. Lett. 2006, 256, 185–194. [Google Scholar] [CrossRef]
- Clermont, O.; Bonacorsi, S.; Bingen, E. Rapid and simple determination of the Escherichia coli phylogenetic group. Appl. Environ. Microbiol. 2000, 66, 4555–4558. [Google Scholar] [CrossRef]
- Gordon, D.M.; Clermont, O.; Tolley, H.; Denamur, E. Assigning Escherichia coli strains to phylogenetic groups: Multi-locus sequence typing versus the PCR triplex method. Environ. Microbiol. 2008, 10, 2484–2496. [Google Scholar] [CrossRef] [PubMed]
- Clermont, O.; Christenson, J.K.; Denamur, E.; Gordon, D.M. The Clermont Escherichia coli phylo-typing method revisited: Improvement of specificity and detection of new phylo-groups. Environ. Microbiol. Rep. 2013, 5, 58–65. [Google Scholar] [CrossRef]
- Clermont, O.; Dixit, O.V.A.; Vangchhia, B.; Condamine, B.; Dion, S.; Bridier-Nahmias, A.; Denamur, E.; Gordon, D. Characterization and rapid identification of phylogroup G in Escherichia coli, a lineage with high virulence and antibiotic resistance potential. Environ. Microbiol. 2019, 21, 3107–3117. [Google Scholar] [CrossRef]
- Berthe, T.; Ratajczak, M.; Clermont, O.; Denamur, E.; Petit, F. Evidence for coexistence of distinct Escherichia coli populations in various aquatic environments and their survival in estuary water. Appl. Environ. Microbiol. 2013, 79, 4684–4693. [Google Scholar] [CrossRef]
- Moissenet, D.; Salauze, B.; Clermont, O.; Bingen, E.; Arlet, G.; Denamur, E.; Mérens, A.; Mitanchez, D.; Vu-Thien, H. Meningitis caused by Escherichia coli producing TEM-52 extended-spectrum beta-lactamase within an extensive outbreak in a neonatal ward: Epidemiological investigation and characterization of the strain. J. Clin. Microbiol. 2010, 48, 2459–2463. [Google Scholar] [CrossRef] [PubMed]
- Stoppe, N.C.; Silva, J.S.; Carlos, C.; Sato, M.I.Z.; Saraiva, A.M.; Ottoboni, L.M.M.; Torres, T.T. Worldwide Phylogenetic Group Patterns of Escherichia coli from Commensal Human and Wastewater Treatment Plant Isolates. Front. Microbiol. 2017, 8, 2512. [Google Scholar] [CrossRef]
- Al-Mayahie, S.M.; Al-Khafajy, A.M.A.; Dosh, N.A.; Al-Rekabi, A.R.; Al-Atabie, A.G. Phylogenetic Grouping of Dominant Fecal Escherichia coli Isolates fromHealthy Males and Females in Al-Kut/Wasit Province/Iraq. J. Bacteriol. Parasitol. 2014, 6, 1000215. [Google Scholar] [CrossRef]
- Massot, M.; Daubié, A.S.; Clermont, O.; Jauréguy, F.; Couffignal, C.; Dahbi, G.; Mora, A.; Blanco, J.; Branger, C.; Mentré, F.; et al. Phylogenetic, virulence and antibiotic resistance characteristics of commensal strain populations of Escherichia coli from community subjects in the Paris area in 2010 and evolution over 30 years. Microbiology 2016, 162, 642–650. [Google Scholar] [CrossRef]
- Karmali, M.A.; Mascarenhas, M.; Shen, S.; Ziebell, K.; Johnson, S.; Reid-Smith, R.; Isaac-Renton, J.; Clark, C.; Rahn, K.; Kaper, J.B. Association of genomic O island 122 of Escherichia coli EDL 933 with verocytotoxin-producing Escherichia coli seropathotypes that are linked to epidemic and/or serious disease. J. Clin. Microbiol. 2003, 41, 4930–4940. [Google Scholar] [CrossRef] [PubMed]
- Toma, C.; Martínez Espinosa, E.; Song, T.; Miliwebsky, E.; Chinen, I.; Iyoda, S.; Iwanaga, M.; Rivas, M. Distribution of putative adhesins in different seropathotypes of Shiga toxin-producing Escherichia coli. J. Clin. Microbiol. 2004, 42, 4937–4946. [Google Scholar] [CrossRef]
- Feng, P.; Lampel, K.A.; Karch, H.; Whittam, T.S. Genotypic and Phenotypic Changes in the Emergence of Escherichia coli O157:H7. J. Infect. Dis. 1998, 177, 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Bilge, S.S.; Vary, J.C., Jr.; Dowell, S.F.; Tarr, P.I. Role of the Escherichia coli O157:H7 O side chain in adherence and analysis of an rfb locus. Infect. Immun. 1996, 64, 4795–4801. [Google Scholar] [CrossRef] [PubMed]
- Leopold, S.R.; Magrini, V.; Holt, N.J.; Shaikh, N.; Mardis, E.R.; Cagno, J.; Ogura, Y.; Iguchi, A.; Hayashi, T.; Mellmann, A.; et al. A precise reconstruction of the emergence and constrained radiations of Escherichia coli O157 portrayed by backbone concatenomic analysis. Proc. Natl. Acad. Sci. USA 2009, 106, 8713–8718. [Google Scholar] [CrossRef]
- Kyle, J.L.; Cummings, C.A.; Parker, C.T.; Quiñones, B.; Vatta, P.; Newton, E.; Huynh, S.; Swimley, M.; Degoricija, L.; Barker, M.; et al. Escherichia coli serotype O55:H7 diversity supports parallel acquisition of bacteriophage at Shiga toxin phage insertion sites during evolution of the O157:H7 lineage. J. Bacteriol. 2012, 194, 1885–1896. [Google Scholar] [CrossRef]
- Asadulghani, M.; Ogura, Y.; Ooka, T.; Itoh, T.; Sawaguchi, A.; Iguchi, A.; Nakayama, K.; Hayashi, T. The defective prophage pool of Escherichia coli O157: Prophage-prophage interactions potentiate horizontal transfer of virulence determinants. PLoS Pathog. 2009, 5, e1000408. [Google Scholar] [CrossRef] [PubMed]
- Manning, S.D.; Motiwala, A.S.; Springman, A.C.; Qi, W.; Lacher, D.W.; Ouellette, L.M.; Mladonicky, J.M.; Somsel, P.; Rudrik, J.T.; Dietrich, S.E.; et al. Variation in virulence among clades of Escherichia coli O157:H7 associated with disease outbreaks. Proc. Natl. Acad. Sci. USA 2008, 105, 4868–4873. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Nietfeldt, J.; Benson, A.K. Octamer-based genome scanning distinguishes a unique subpopulation of Escherichia coli O157:H7 strains in cattle. Proc. Natl. Acad. Sci. USA 1999, 96, 13288–13293. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Kovar, J.; Kim, J.; Nietfeldt, J.; Smith, D.R.; Moxley, R.A.; Olson, M.E.; Fey, P.D.; Benson, A.K. Identification of common subpopulations of non-sorbitol-fermenting, beta-glucuronidase-negative Escherichia coli O157:H7 from bovine production environments and human clinical samples. Appl. Environ. Microbiol. 2004, 70, 6846–6854. [Google Scholar] [CrossRef]
- Dallman, T.J.; Ashton, P.M.; Byrne, L.; Perry, N.T.; Petrovska, L.; Ellis, R.; Allison, L.; Hanson, M.; Holmes, A.; Gunn, G.J.; et al. Applying phylogenomics to understand the emergence of Shiga-toxin-producing Escherichia coli O157:H7 strains causing severe human disease in the UK. Microb. Genom. 2015, 1, e000029. [Google Scholar] [CrossRef] [PubMed]
- Iyoda, S.; Manning, S.D.; Seto, K.; Kimata, K.; Isobe, J.; Etoh, Y.; Ichihara, S.; Migita, Y.; Ogata, K.; Honda, M.; et al. Phylogenetic Clades 6 and 8 of Enterohemorrhagic Escherichia coli O157:H7 With Particular stx Subtypes are More Frequently Found in Isolates From Hemolytic Uremic Syndrome Patients Than From Asymptomatic Carriers. Open Forum Infect. Dis. 2014, 1, ofu061. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, D.L.; Jackson, M.P.; Samuel, J.E.; Holmes, R.K.; O’Brien, A.D. Cloning and sequencing of a Shiga-like toxin type II variant from Escherichia coli strain responsible for edema disease of swine. J. Bacteriol. 1988, 170, 4223–4230. [Google Scholar] [CrossRef] [PubMed]
- Scheutz, F.; Teel, L.D.; Beutin, L.; Piérard, D.; Buvens, G.; Karch, H.; Mellmann, A.; Caprioli, A.; Tozzoli, R.; Morabito, S.; et al. Multicenter evaluation of a sequence-based protocol for subtyping Shiga toxins and standardizing Stx nomenclature. J. Clin. Microbiol. 2012, 50, 2951–2963. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, A.O.; Lively, T.A.; Chen, M.E.; Rothman, S.W.; Formal, S.B. Escherichia coli O157:H7 strains associated with haemorrhagic colitis in the United States produce a Shigella dysenteriae 1 (SHIGA) like cytotoxin. Lancet 1983, 1 Pt 1, 702. [Google Scholar] [CrossRef]
- Strockbine, N.A.; Jackson, M.P.; Sung, L.M.; Holmes, R.K.; O’Brien, A.D. Cloning and sequencing of the genes for Shiga toxin from Shigella dysenteriae type 1. J. Bacteriol. 1988, 170, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Basu, D.; Li, X.P.; Kahn, J.N.; May, K.L.; Kahn, P.C.; Tumer, N.E. The A1 Subunit of Shiga Toxin 2 Has Higher Affinity for Ribosomes and Higher Catalytic Activity than the A1 Subunit of Shiga Toxin 1. Infect. Immun. 2016, 84, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Boerlin, P.; McEwen, S.A.; Boerlin-Petzold, F.; Wilson, J.B.; Johnson, R.P.; Gyles, C.L. Associations between virulence factors of Shiga toxin-producing Escherichia coli and disease in humans. J. Clin. Microbiol. 1999, 37, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Tesh, V.L.; Burris, J.A.; Owens, J.W.; Gordon, V.M.; Wadolkowski, E.A.; O’Brien, A.D.; Samuel, J.E. Comparison of the relative toxicities of Shiga-like toxins type I and type II for mice. Infect. Immun. 1993, 61, 3392–3402. [Google Scholar] [CrossRef] [PubMed]
- Persson, S.; Olsen, K.E.; Ethelberg, S.; Scheutz, F. Subtyping method for Escherichia coli shiga toxin (verocytotoxin) 2 variants and correlations to clinical manifestations. J. Clin. Microbiol. 2007, 45, 2020–2024. [Google Scholar] [CrossRef] [PubMed]
- Ethelberg, S.; Olsen, K.E.; Scheutz, F.; Jensen, C.; Schiellerup, P.; Enberg, J.; Petersen, A.M.; Olesen, B.; Gerner-Smidt, P.; Mølbak, K. Virulence factors for hemolytic uremic syndrome, Denmark. Emerg. Infect. Dis. 2004, 10, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, R.A.; Darnell, S.C.; O’Brien, A.D. Activation of Shiga-like toxins by mouse and human intestinal mucus correlates with virulence of enterohemorrhagic Escherichia coli O91:H21 isolates in orally infected, streptomycin-treated mice. Infect. Immun. 1996, 64, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Kokai-Kun, F.J.; Melton-Celsa, A.R.; O’Brien, A.D. Elastase in intestinal mucus enhances the cytotoxicity of Shiga toxin type 2d. J. Biol. Chem. 2000, 275, 3713–3721. [Google Scholar] [CrossRef]
- Yang, X.; Bai, X.; Zhang, J.; Sun, H.; Fu, S.; Fan, R.; He, X.; Scheutz, F.; Matussek, A.; Xiong, Y. Escherichia coli strains producing a novel Shiga toxin 2 subtype circulate in China. Int. J. Med. Microbiol. 2020, 310, 151377. [Google Scholar] [CrossRef]
- MacLeod, L.D.; Gyles, C.L.; Wilcock, B.P. Reproduction of edema disease of swine with purified Shiga-like toxin-II variant. Vet. Pathol. 1991, 28, 66–73. [Google Scholar] [CrossRef]
- Feng, C.P.; Reddy, S. Prevalences of Shiga toxin subtypes and selected other virulence factors among Shiga-toxigenic Escherichia coli strains isolated from fresh produce. Appl. Environ. Microbiol. 2013, 79, 6917–6923. [Google Scholar] [CrossRef]
- Farooq, S.; Hussain, I.; Mir, M.A.; Bhat, M.A.; Wani, S.A. Isolation of atypical enteropathogenic Escherichia coli and Shiga toxin 1 and 2f-producing Escherichia coli from avian species in India. Lett. Appl. Microbiol. 2009, 48, 692–697. [Google Scholar] [CrossRef]
- Friesema, I.; van der Zwaluw, K.; Schuurman, T.; Kooistra-Smid, M.; Franz, E.; van Duynhoven, Y.; van Pelt, W. Emergence of Escherichia coli encoding Shiga toxin 2f in human Shiga toxin-producing E. coli (STEC) infections in the Netherlands, January 2008 to December 2011. Eurosurveillance 2014, 19, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Rauw, D.K.; Jacobs, S.; Piérard, D. Twenty-seven years of screening for Shiga toxin-producing Escherichia coli in a university hospital. Brussels, Belgium, 1987–2014. PLoS ONE 2018, 13, e0199968. [Google Scholar] [CrossRef]
- Bai, X.; Fu, S.; Zhang, J.; Fan, R.; Xu, Y.; Sun, H.; He, X.; Xu, J.; Xiong, Y. Identification and pathogenomic analysis of an Escherichia coli strain producing a novel Shiga toxin 2 subtype. Sci. Rep. 2018, 8, 6756. [Google Scholar] [CrossRef]
- Lacher, D.W.; Gangiredla, J.; Patel, I.; Elkins, C.A.; Feng, P.C.H. Use of the Escherichia coli Identification Microarray for Characterizing the Health Risks of Shiga Toxin–Producing Escherichia coli Isolated from Foods. J. Food Prot. 2016, 79, 1656–1662. [Google Scholar] [CrossRef]
- Hughes, A.C.; Zhang, Y.; Bai, X.; Xiong, Y.; Wang, Y.; Yang, X.; Xu, Q.; He, X. Structural and Functional Characterization of Stx2k, a New Subtype of Shiga Toxin 2. Microorganisms 2019, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Bai, X.; Zhao, A.; Lan, R.; Du, H.; Wang, T.; Shi, C.; Yuan, X.; Bai, X.; Ji, S.; et al. Characterization of Shiga toxin-producing Escherichia coli isolated from healthy pigs in China. BMC Microbiol. 2014, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Majowicz, S.E.; Scallan, E.; Jones-Bitton, A.; Sargeant, J.M.; Stapleton, J.; Angulo, F.J.; Yeung, D.H.; Kirk, M.D. Global incidence of human Shiga toxin-producing Escherichia coli infections and deaths: A systematic review and knowledge synthesis. Foodborne Pathog. Dis. 2014, 11, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Repetto, H.A. Epidemic hemolytic-uremic syndrome in children. Kidney Int. 1997, 52, 1708–1719. [Google Scholar] [CrossRef] [PubMed]
- Lynn, R.M.; O’Brien, S.J.; Taylor, C.M.; Adak, G.K.; Chart, H.; Cheasty, T.; Coia, J.E.; Gillespie, I.A.; Locking, M.E.; Reilly, W.J. Childhood hemolytic uremic syndrome, United Kingdom and Ireland. Emerg. Infect. Dis. 2005, 11, 590–596. [Google Scholar] [CrossRef]
- Acheson, D.W.; Moore, R.; De Breucker, S.; Lincicome, L.; Jacewicz, M.; Skutelsky, E.; Keusch, G.T. Translocation of Shiga toxin across polarized intestinal cells in tissue culture. Infect. Immun. 1996, 64, 3294–3300. [Google Scholar] [CrossRef]
- Békássy, Z.D.; Calderon Toledo, C.; Leoj, G.; Kristoffersson, A.; Leopold, S.R.; Perez, M.T.; Karpman, D. Intestinal damage in enterohemorrhagic Escherichia coli infection. Pediatr. Nephrol. 2011, 26, 2059–2071. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Tesh, V.L. Roles of Shiga Toxins in Immunopathology. Toxins 2019, 11, 212. [Google Scholar] [CrossRef] [PubMed]
- Heyderman, S.R.; Soriani, M.; Hirst, T.R. Is immune cell activation the missing link in the pathogenesis of post-diarrhoeal HUS? Trends Microbiol. 2001, 9, 262–266. [Google Scholar] [CrossRef]
- Van de Kar, N.C.; Monnens, L.A.; Karmali, M.A.; van Hinsbergh, V.W. Tumor necrosis factor and interleukin-1 induce expression of the verocytotoxin receptor globotriaosylceramide on human endothelial cells: Implications for the pathogenesis of the hemolytic uremic syndrome. Blood 1992, 80, 2755–2764. [Google Scholar] [CrossRef] [PubMed]
- Louise, B.C.; Obrig, T.G. Shiga toxin-associated hemolytic-uremic syndrome: Combined cytotoxic effects of Shiga toxin, interleukin-1 beta, and tumor necrosis factor alpha on human vascular endothelial cells in vitro. Infect. Immun. 1991, 59, 4173–4179. [Google Scholar] [CrossRef]
- Betz, J.; Dorn, I.; Kouzel, I.U.; Bauwens, A.; Meisen, I.; Kemper, B.; Bielaszewska, M.; Mormann, M.; Weymann, L.; Sibrowski, W. Shiga toxin of enterohaemorrhagic Escherichia coli directly injures developing human erythrocytes. Cell Microbiol. 2016, 18, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Te Loo, D.; Hinsbergh, V.; Heuvel, L.; Monnens, L.A.H. Detection of verocytotoxin bound to circulating polymorphonuclear leukocytes of patients with hemolytic uremic syndrome. J. Am. Soc. Nephrol. 2001, 12, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Brigotti, M.; Carnicelli, D.; Arfilli, V.; Tamassia, N.; Borsetti, F.; Fabbri, E.; Tazzari, P.L.; Ricci, F.; Pagliaro, P.; Spisni, E.; et al. Identification of TLR4 as the receptor that recognizes Shiga toxins in human neutrophils. J. Immunol. 2013, 191, 4748–4758. [Google Scholar] [CrossRef] [PubMed]
- Geelen, J.M.; van der Velden, T.J.; van den Heuvel, L.P.; Monnens, L.A. Interactions of Shiga-like toxin with human peripheral blood monocytes. Pediatr. Nephrol. 2007, 22, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.A.; Polanowska-Grabowska, R.K.; Fujii, J.; Obrig, T.; Gear, A.R. Shiga toxin binds to activated platelets. J. Thromb. Haemost. 2004, 2, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Legros, N.; Pohlentz, G.; Steil, D.; Müthing, J. Shiga toxin-glycosphingolipid interaction: Status quo of research with focus on primary human brain and kidney endothelial cells. Int. J. Med. Microbiol. 2018, 308, 1073–1084. [Google Scholar] [CrossRef]
- Ståhl, A.L.; Sartz, L.; Nelsson, A.; Békássy, Z.D.; Karpman, D. Shiga toxin and lipopolysaccharide induce platelet-leukocyte aggregates and tissue factor release, a thrombotic mechanism in hemolytic uremic syndrome. PLoS ONE 2009, 4, e6990. [Google Scholar] [CrossRef]
- Ståhl, A.L.; Arvidsson, I.; Johansson, K.E.; Chromek, M.; Rebetz, J.; Loos, S.; Kristoffersson, A.C.; Békássy, Z.D.; Mörgelin, M.; Karpman, D. A novel mechanism of bacterial toxin transfer within host blood cell-derived microvesicles. PLoS Pathog. 2015, 11, e1004619. [Google Scholar] [CrossRef] [PubMed]
- Boyd, B.; Lingwood, C. Verotoxin receptor glycolipid in human renal tissue. Nephron 1989, 51, 207–210. [Google Scholar] [CrossRef]
- Richardson, S.E.; Karmali, M.A.; Becker, L.E.; Smith, C.R. The histopathology of the hemolytic uremic syndrome associated with verocytotoxin-producing Escherichia coli infections. Hum. Pathol. 1988, 19, 1102–1108. [Google Scholar] [CrossRef]
- Coia, J.E. Clinical, microbiological and epidemiological aspects of Escherichia coli O157 infection. FEMS Immunol. Med. Microbiol. 1998, 20, 1–9. [Google Scholar] [CrossRef]
- Villysson, A.; Tontanahal, A.; Karpman, D. Microvesicle Involvement in Shiga Toxin-Associated Infection. Toxins 2017, 9, 376. [Google Scholar] [CrossRef] [PubMed]
- Moake, J.L. Haemolytic-uraemic syndrome: Basic science. Lancet 1994, 343, 393–397. [Google Scholar] [CrossRef]
- Huang, J.; Motto, D.G.; Bundle, D.R.; Sadler, J.E. Shiga toxin B subunits induce VWF secretion by human endothelial cells and thrombotic microangiopathy in ADAMTS13-deficient mice. Blood 2010, 116, 3653–3659. [Google Scholar] [CrossRef]
- Takeda, T.; Dohi, S.; Igarashi, T.; Yamanaka, T.; Yoshiya, K.; Kobayashi, N. Impairment by verotoxin of tubular function contributes to the renal damage seen in haemolytic uraemic syndrome. J. Infect. 1993, 27, 339–341. [Google Scholar] [CrossRef]
- Walters, M.D.; Matthei, I.U.; Kay, R.; Dillon, M.J.; Barratt, T.M. The polymorphonuclear leucocyte count in childhood haemolytic uraemic syndrome. Pediatr. Nephrol. 1989, 3, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, M.M.; Shah, V.; Trompeter, R.S.; Dillon, M.J.; Barratt, T.M. Interleukin-8 and polymorphoneutrophil leucocyte activation in hemolytic uremic syndrome of childhood. Kidney Int. 1992, 42, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Harrison, L.M.; Gaines, D.W.; Babu, U.S.; Balan, K.V.; Reimschuessel, R.; Do, A.B.; Pereira, M.R.; Bigley, E.C., 3rd; Ferguson, M.; Mehta, A. Diet-induced obesity precipitates kidney dysfunction and alters inflammatory mediators in mice treated with Shiga Toxin 2. Microb. Pathog. 2018, 123, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Tarr, I.P.; Gordon, C.A.; Chandler, W.L. Shiga-toxin-producing Escherichia coli and haemolytic uraemic syndrome. Lancet 2005, 365, 1073–1086. [Google Scholar] [CrossRef]
- Parsons, B.D.; Zelyas, N.; Berenger, B.M.; Chui, L. Detection, Characterization, and Typing of Shiga Toxin-Producing Escherichia coli. Front. Microbiol. 2016, 7, 478. [Google Scholar] [CrossRef]
- Ostroff, S.M.; Tarr, P.I.; Neill, M.A.; Lewis, J.H.; Hargrett-Bean, N.; Kobayashi, J.M. Toxin Genotypes and Plasmid Profiles as Determinants of Systemic Sequelae in Escherichia coli O157:H7 Infections. J. Infect. Dis. 1989, 160, 994–998. [Google Scholar] [CrossRef]
- Friedrich, A.W.; Bielaszewska, M.; Zhang, W.-L.; Pulz, M.; Kuczius, T.; Ammon, A.; Karch, H. Escherichia coli Harboring Shiga Toxin 2 Gene Variants: Frequency and Association with Clinical Symptoms. J. Infect. Dis. 2002, 185, 74–84. [Google Scholar] [CrossRef]
- Byrne, L.; Adams, N.; Jenkins, C. Association between Shiga Toxin-Producing Escherichia coli O157:H7 stx Gene Subtype and Disease Severity, England, 2009–2019. Emerg. Infect. Dis. 2020, 26, 2394–2400. [Google Scholar] [CrossRef]
- Orth, D.; Grif, K.; Khan, A.B.; Naim, A.; Dierich, M.P.; Würzner, R. The Shiga toxin genotype rather than the amount of Shiga toxin or the cytotoxicity of Shiga toxin in vitro correlates with the appearance of the hemolytic uremic syndrome. Diagn. Microbiol. Infect. Dis. 2007, 59, 235–242. [Google Scholar] [CrossRef]
- Matthews, L.; Reeve, R.; Gally, D.L.; Low, J.C.; Woolhouse, M.E.; McAteer, S.P.; Locking, M.E.; Chase-Topping, M.E.; Haydon, D.T.; Allison, L.J.; et al. Predicting the public health benefit of vaccinating cattle against Escherichia coli O157. Proc. Natl. Acad. Sci. USA 2013, 110, 16265–16270. [Google Scholar] [CrossRef]
- Xiong, Y.; Wang, P.; Lan, R.; Ye, C.; Wang, H.; Ren, J.; Jing, H.; Wang, Y.; Zhou, Z.; Bai, X.; et al. A novel Escherichia coli O157:H7 clone causing a major hemolytic uremic syndrome outbreak in China. PLoS ONE 2012, 7, e36144. [Google Scholar] [CrossRef] [PubMed]
- Frank, C.; Werber, D.; Cramer, J.P.; Askar, M.; Faber, M.; an der Heiden, M.; Bernard, H.; Fruth, A.; Prager, R.; Spode, A.; et al. Epidemic profile of Shiga-toxin-producing Escherichia coli O104:H4 outbreak in Germany. N. Engl. J. Med. 2011, 365, 1771–1780. [Google Scholar] [CrossRef] [PubMed]
- Verhaegen, B.; De Reu, K.; Heyndrickx, M.; De Zutter, L. Comparison of Six Chromogenic Agar Media for the Isolation of a Broad Variety of Non-O157 Shigatoxin-Producing Escherichia coli (STEC) Serogroups. Int. J. Environ. Res. Public Health 2015, 12, 6965–6978. [Google Scholar] [CrossRef] [PubMed]
- Quiros, P.; Martinez-Castillo, A.; Muniesa, M. Improving detection of Shiga toxin-producing Escherichia coli by molecular methods by reducing the interference of free Shiga toxin-encoding bacteriophages. Appl. Environ. Microbiol. 2015, 81, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.; Bai, X.; Fu, S.; Xu, Y.; Sun, H.; Wang, H.; Xiong, Y. Tellurite resistance profiles and performance of different chromogenic agars for detection of non-O157 Shiga toxin-producing Escherichia coli. Int. J. Food Microbiol. 2018, 266, 295–300. [Google Scholar] [CrossRef]
- Fan, R.; Shao, K.; Yang, X.; Bai, X.; Fu, S.; Sun, H.; Xu, Y.; Wang, H.; Li, Q.; Hu, B.; et al. High prevalence of non-O157 Shiga toxin-producing Escherichia coli in beef cattle detected by combining four selective agars. BMC Microbiol. 2019, 19, 213. [Google Scholar] [CrossRef]
- Gould, L.H.; Bopp, C.; Strockbine, N.; Atkinson, R.; Baselski, V.; Body, B.; Carey, R.; Crandall, C.; Hurd, S.; Kaplan, R.; et al. Recommendations for diagnosis of shiga toxin--producing Escherichia coli infections by clinical laboratories. MMWR Recomm. Rep. 2009, 58, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Boone, J.T.; Campbell, D.E.; Dandro, A.S.; Chen, L.; Herbein, J.F. A Rapid Immunoassay for Detection of Shiga Toxin-Producing Escherichia coli Directly from Human Fecal Samples and Its Performance in Detection of Toxin Subtypes. J. Clin. Microbiol. 2016, 54, 3056–3063. [Google Scholar] [CrossRef]
- Ake, J.A.; Jelacic, S.; Ciol, M.A.; Watkins, S.L.; Murray, K.F.; Christie, D.L.; Klein, E.J.; Tarr, P.I. Relative nephroprotection during Escherichia coli O157:H7 infections: Association with intravenous volume expansion. Pediatrics 2005, 115, e673–e680. [Google Scholar] [CrossRef] [PubMed]
- Canpolat, N. Hemolytic uremic syndrome. Turk. Pediatr. Ars. 2015, 50, 73–82. [Google Scholar] [CrossRef]
- Joseph, A.; Cointe, A.; Mariani Kurkdjian, P.; Rafat, C.; Hertig, A. Shiga Toxin-Associated Hemolytic Uremic Syndrome: A Narrative Review. Toxins 2020, 12, 67. [Google Scholar] [CrossRef] [PubMed]
- Gerber, A.; Karch, H.; Allerberger, F.; Verweyen, H.M.; Zimmerhackl, L.B. Clinical course and the role of shiga toxin-producing Escherichia coli infection in the hemolytic-uremic syndrome in pediatric patients, 1997–2000, in Germany and Austria: A prospective study. J. Infect. Dis. 2002, 186, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Mathew, O.R.; Nayer, A.; Asif, A. The endothelium as the common denominator in malignant hypertension and thrombotic microangiopathy. J. Am. Soc. Hypertens 2016, 10, 352–359. [Google Scholar] [CrossRef]
- Wen, S.X.; Teel, L.D.; Judge, N.A.; O’Brien, A.D. A plant-based oral vaccine to protect against systemic intoxication by Shiga toxin type 2. Proc. Natl. Acad. Sci. USA 2006, 103, 7082–7087. [Google Scholar] [CrossRef]
- Hamabata, T.; Sato, T.; Takita, E.; Matsui, T.; Imaoka, T.; Nakanishi, N.; Nakayama, K.; Tsukahara, T.; Sawada, K. Shiga toxin 2eB-transgenic lettuce vaccine is effective in protecting weaned piglets from edema disease caused by Shiga toxin-producing Escherichia coli infection. Anim. Sci. J. 2019, 90, 1460–1467. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sun, Y.; Feng, S.; Zhu, L.; Guo, X.; Qi, C. Towards an attenuated enterohemorrhagic Escherichia coli O157:H7 vaccine characterized by a deleted ler gene and containing apathogenic Shiga toxins. Vaccine 2009, 27, 5929–5935. [Google Scholar] [CrossRef] [PubMed]
- Toledo, C.C.; Arvidsson, I.; Karpman, D. Cross-reactive protection against enterohemorrhagic Escherichia coli infection by enteropathogenic E. coli in a mouse model. Infect. Immun. 2011, 79, 2224–2233. [Google Scholar] [CrossRef]
- Gu, J.; Ning, Y.; Wang, H.; Xiao, D.; Tang, B.; Luo, P.; Cheng, Y.; Jiang, M.; Li, N.; Zou, Q. Vaccination of attenuated EIS-producing Salmonella induces protective immunity against enterohemorrhagic Escherichia coli in mice. Vaccine 2011, 29, 7395–7403. [Google Scholar] [CrossRef] [PubMed]
- Sreerohini, S.; Balakrishna, K.; Parida, M. Oral immunization of mice with Lactococcus lactis expressing Shiga toxin truncate confers enhanced protection against Shiga toxins of Escherichia coli O157:H7 and Shigella dysenteriae. Apmis 2019, 127, 671–680. [Google Scholar] [CrossRef]
- Ahmed, B.; Loos, M.; Vanrompay, D.; Cox, E. Oral immunization with Lactococcus lactis-expressing EspB induces protective immune responses against Escherichia coli O157:H7 in a murine model of colonization. Vaccine 2014, 32, 3909–3916. [Google Scholar] [CrossRef]
- Marcato, P.; Mulvey, G.; Read, R.J.; Vander Helm, K.; Nation, P.N.; Armstrong, G.D. Immunoprophylactic potential of cloned Shiga toxin 2 B subunit. J. Infect. Dis. 2001, 183, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Kerner, K.; Bridger, P.S.; Köpf, G.; Fröhlich, J.; Barth, S.; Willems, H.; Bauerfeind, R.; Baljer, G.; Menge, C. Evaluation of biological safety in vitro and immunogenicity in vivo of recombinant Escherichia coli Shiga toxoids as candidate vaccines in cattle. Vet. Res. 2015, 46, 38. [Google Scholar] [CrossRef]
- Schmidt, N.; Barth, S.A.; Frahm, J.; Meyer, U.; Dänicke, S.; Geue, L.; Menge, C. Decreased STEC shedding by cattle following passive and active vaccination based on recombinant Escherichia coli Shiga toxoids. Vet. Res. 2018, 49, 28. [Google Scholar] [CrossRef]
- Gao, X.; Cai, K.; Shi, J.; Liu, H.; Hou, X.; Tu, W.; Xiao, L.; Wang, Q.; Wang, H. Immunogenicity of a novel Stx2B-Stx1B fusion protein in a mice model of Enterohemorrhagic Escherichia coli O157:H7 infection. Vaccine 2009, 27, 2070–2076. [Google Scholar] [CrossRef] [PubMed]
- Stearns-Kurosawa, D.J.; Collins, V.; Freeman, S.; Debord, D.; Nishikawa, K.; Oh, S.Y.; Leibowitz, C.S.; Kurosawa, S. Rescue from lethal Shiga toxin 2-induced renal failure with a cell-permeable peptide. Pediatr. Nephrol. 2011, 26, 2031–2039. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.W.; Henderson, T.D.; Patfield, S.; Stanker, L.H.; He, X. Mouse in vivo neutralization of Escherichia coli Shiga toxin 2 with monoclonal antibodies. Toxins 2013, 5, 1845–1858. [Google Scholar] [CrossRef]
- Howard-Varona, C.; Vik, D.R.; Solonenko, N.E.; Li, Y.F.; Gazitua, M.C.; Chittick, L.; Samiec, J.K.; Jensen, A.E.; Anderson, P.; Howard-Varona, A. Fighting Fire with Fire: Phage Potential for the Treatment of E. coli O157 Infection. Antibiotics 2018, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Necel, A.; Bloch, S.; Nejman-Faleńczyk, B.; Grabski, M.; Topka, G.; Dydecka, A.; Kosznik-Kwaśnicka, K.; Grabowski, Ł.; Jurczak-Kurek, A.; Wołkowicz, T.; et al. Characterization of a bacteriophage, vB_Eco4M-7, that effectively infects many Escherichia coli O157 strains. Sci. Rep. 2020, 10, 3743. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, X.; Wang, S.; Guan, L.; Li, X.; Hu, D.; Gao, D.; Song, J.; Chen, H.; Qian, P. A Novel Tail-Associated O91-Specific Polysaccharide Depolymerase from a Podophage Reveals Lytic Efficacy of Shiga Toxin-Producing Escherichia coli. Appl. Environ. Microbiol. 2020, 86, e00145-20. [Google Scholar] [CrossRef] [PubMed]
- Dini, C.; Bolla, P.A.; de Urraza, P.J. Treatment of in vitro enterohemorrhagic Escherichia coli infection using phage and probiotics. J. Appl. Microbiol. 2016, 121, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Yeşilyurt, N.; ılmaz, B.; Ağagündüz, D.; Capasso, R. Involvement of Probiotics and Postbiotics in the Immune System Modulation. Biologics 2021, 1, 89–110. [Google Scholar] [CrossRef]
- Sargeant, J.M.; Amezcua, M.R.; Rajic, A.; Waddell, L. Pre-harvest Interventions to Reduce the Shedding of E. coli O157 in the Faeces of Weaned Domestic Ruminants: A Systematic Review. Zoonoses Public Health 2007, 54, 260–277. [Google Scholar] [CrossRef] [PubMed]
- Callaway, T.R.; Carr, M.A.; Edrington, T.S.; Anderson, R.C.; Nisbet, D.J. Diet, Escherichia coli O157:H7, and cattle: A review after 10 years. Curr. Issues Mol. Biol. 2009, 11, 67–79. [Google Scholar] [PubMed]
- Peterson, R.E.; Klopfenstein, T.J.; Erickson, G.E.; Folmer, J.; Hinkley, S.; Moxley, R.A.; Smith, D.R. Effect of Lactobacillus acidophilus Strain NP51 on Escherichia coli O157:H7 Fecal Shedding and Finishing Performance in Beef Feedlot Cattle. J. Food Prot. 2007, 70, 287–291. [Google Scholar] [CrossRef]
- Tsai, T.-Y.; Cheng, P.-C.; Pan, T.-M. Immunomodulating Activity of Lactobacillus paracasei subsp. paracasei NTU 101 in Enterohemorrhagic Escherichia coli O157H7-Infected Mice. J. Agric. Food Chem. 2010, 58, 11265–11272. [Google Scholar] [CrossRef]
- Etienne-Mesmin, L.; Livrelli, V.; Privat, M.; Denis, S.; Cardot, J.-M.; Alric, M.; Blanquet-Diot, S. Effect of a New Probiotic Saccharomyces cerevisiae Strain on Survival of Escherichia coli O157:H7 in a Dynamic Gastrointestinal Model. Appl. Environ. Microbiol. 2011, 77, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Thévenot, J.; Cordonnier, C.; Rougeron, A.; Le Goff, O.; Nguyen, H.T.T.; Denis, S.; Alric, M.; Livrelli, V.; Blanquet-Diot, S. Enterohemorrhagic Escherichia coli infection has donor-dependent effect on human gut microbiota and may be antagonized by probiotic yeast during interaction with Peyer’s patches. Appl. Microbiol. Biotechnol. 2015, 99, 9097–9110. [Google Scholar] [CrossRef]
- Thévenot, J.; Etienne-Mesmin, L.; Denis, S.; Chalancon, S.; Alric, M.; Livrelli, V.; Blanquet-Diot, S. Enterohemorrhagic Escherichia coli O157:H7 Survival in an In Vitro Model of the Human Large Intestine and Interactions with Probiotic Yeasts and Resident Microbiota. Appl. Environ. Microbiol. 2013, 79, 1058–1064. [Google Scholar] [CrossRef]
- Mogna, L.; Del Piano, M.; Deidda, F.; Nicola, S.; Soattini, L.; Debiaggi, R.; Sforza, F.; Strozzi, G.; Mogna, G. Assessment of the in vitro inhibitory activity of specific probiotic bacteria against different Escherichia coli strains. J. Clin. Gastroenterol. 2012, 46, S29–S32. [Google Scholar] [CrossRef] [PubMed]
- Arena, M.P.; Russo, P.; Capozzi, V.; López, P.; Fiocco, D.; Spano, G. Probiotic abilities of riboflavin-overproducing Lactobacillus strains: A novel promising application of probiotics. Appl. Microbiol. Biotechnol. 2014, 98, 7569–7581. [Google Scholar] [CrossRef]
- Zhang, H.; Gao, J.; He, X.; Gong, Z.; Wan, Y.; Hu, T.; Li, Y.; Cao, H. The postbiotic HM0539 from Lactobacillus rhamnosus GG prevents intestinal infection by enterohemorrhagic E. coli O157:H7 in mice. Nan Fang Yi Ke Da Xue Xue Bao 2020, 40, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Medellin-Peña, M.J.; Wang, H.; Johnson, R.; Anand, S.; Griffiths, M.W. Probiotics affect virulence-related gene expression in Escherichia coli O157:H7. Appl. Environ. Microbiol. 2007, 73, 4259–4267. [Google Scholar] [CrossRef] [PubMed]
- Carey, C.M.; Kostrzynska, M.; Ojha, S.; Thompson, S. The effect of probiotics and organic acids on Shiga-toxin 2 gene expression in enterohemorrhagic Escherichia coli O157:H7. J. Microbiol. Methods 2008, 73, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Oussalah, M.; Caillet, S.; Lacroix, M. Mechanism of action of Spanish oregano, Chinese cinnamon, and savory essential oils against cell membranes and walls of Escherichia coli O157:H7 and Listeria monocytogenes. J. Food Prot. 2006, 69, 1046–1055. [Google Scholar] [CrossRef]
- Voravuthikunchai, P.S.; Suwalak, S.; Mitranan, W. Ellagitannin from Quercus infectoria eradicates intestinal colonization and prevents renal injuries in mice infected with Escherichia coli O157:H7. J. Med. Microbiol. 2012, 61, 1366–1372. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pellarín, M.G.; Albrecht, C.; Rojas, M.J.; Aguilar, J.J.; Konigheim, B.S.; Paraje, M.G.; Albesa, I.; Eraso, A.J. Inhibition of cytotoxicity of Shiga toxin of Escherichia coli O157:H7 on vero cells by Prosopis alba Griseb (Fabaceae) and Ziziphus mistol Griseb (Rhamnaceae) extracts. J. Food Prot. 2013, 76, 1733–1739. [Google Scholar] [CrossRef]
- Sewlikar, S.; D’Souza, D.H. Antimicrobial Effects of Quillaja saponaria Extract Against Escherichia coli O157:H7 and the Emerging Non-O157 Shiga Toxin-Producing E. coli. J. Food Sci. 2017, 82, 1171–1177. [Google Scholar] [CrossRef]
- Sheng, L.; Olsen, S.A.; Hu, J.; Yue, W.; Means, W.J.; Zhu, M.J. Inhibitory effects of grape seed extract on growth, quorum sensing, and virulence factors of CDC "top-six" non-O157 Shiga toxin producing E. coli. Int. J. Food Microbiol. 2016, 229, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Hmoteh, J.; Syed Musthafa, K.; Pomwised, R.; Voravuthikunchai, S.P. Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7. Molecules 2016, 21, 692. [Google Scholar] [CrossRef] [PubMed]
- Fullerton, M.; Khatiwada, J.; Johnson, J.U.; Davis, S.; Williams, L.L. Determination of antimicrobial activity of sorrel (Hibiscus sabdariffa) on Escherichia coli O157:H7 isolated from food, veterinary, and clinical samples. J. Med. Food 2011, 14, 950–956. [Google Scholar] [CrossRef]
- Ardissino, G.; Tel, F.; Possenti, I.; Testa, S.; Consonni, D.; Paglialonga, F.; Salardi, S.; Borsa-Ghiringhelli, N.; Salice, P.; Tedeschi, S.; et al. Early Volume Expansion and Outcomes of Hemolytic Uremic Syndrome. Pediatrics 2016, 137, e20152153. [Google Scholar] [CrossRef]
- Freedman, S.B.; Xie, J.; Neufeld, M.S.; Hamilton, W.L.; Hartling, L.; Tarr, P.I.; for the Alberta Provincial Pediatric Enteric Infection Team (APPETITE); Nettel-Aguirre, A.; Chuck, A.; Lee, B. Shiga Toxin-Producing Escherichia coli Infection, Antibiotics, and Risk of Developing Hemolytic Uremic Syndrome: A Meta-analysis. Clin. Infect. Dis. 2016, 62, 1251–1258. [Google Scholar] [CrossRef]
- Tarr, G.A.M.; Oltean, H.N.; Phipps, A.I.; Rabinowitz, P.; Tarr, P.I. Strength of the association between antibiotic use and hemolytic uremic syndrome following Escherichia coli O157:H7 infection varies with case definition. Int. J. Med. Microbiol. 2018, 308, 921–926. [Google Scholar] [CrossRef]
- Geerdes-Fenge, H.F.; Lobermann, M.; Nurnberg, M.; Fritzsche, C.; Koball, S.; Henschel, J.; Hohn, R.; Schober, H.C.; Mitzner, S.; Podbielski, A. Ciprofloxacin reduces the risk of hemolytic uremic syndrome in patients with Escherichia coli O104:H4-associated diarrhea. Infection 2013, 41, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Bielaszewska, M.; Idelevich, E.A.; Zhang, W.; Bauwens, A.; Schaumburg, F.; Mellmann, A.; Peters, G.; Karch, H. Effects of antibiotics on Shiga toxin 2 production and bacteriophage induction by epidemic Escherichia coli O104:H4 strain. Antimicrob. Agents Chemother. 2012, 56, 3277–3282. [Google Scholar] [CrossRef]
- Nitschke, M.; Sayk, F.; Härtel, C.; Roseland, R.T.; Hauswaldt, S.; Steinhoff, J.; Fellermann, K.; Derad, I.; Wellhöner, P.; Büning, J.; et al. Association between azithromycin therapy and duration of bacterial shedding among patients with Shiga toxin-producing enteroaggregative Escherichia coli O104:H4. JAMA 2012, 307, 1046–1052. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Ida, O.; Kimoto, K.; Takatorige, T.; Nakanishi, N.; Tatara, K. Effect of early fosfomycin treatment on prevention of hemolytic uremic syndrome accompanying Escherichia coli O157:H7 infection. Clin. Nephrol. 1999, 52, 357–362. [Google Scholar]
- Kurioka, T.; Yunou, Y.; Harada, H.; Kita, E. Efficacy of antibiotic therapy for infection with Shiga-like toxin-producing Escherichia coli O157:H7 in mice with protein-calorie malnutrition. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 561–571. [Google Scholar] [CrossRef]
- Fadlallah, S.M.; Rahal, E.A.; Sabra, A.; Kissoyan, K.A.; Matar, G.M. Effect of rifampicin and gentamicin on Shiga toxin 2 expression level and the SOS response in Escherichia coli O104:H4. Foodborne Pathog. Dis. 2015, 12, 47–55. [Google Scholar] [CrossRef] [PubMed]
- McGannon, M.C.; Fuller, C.A.; Weiss, A.A. Different classes of antibiotics differentially influence shiga toxin production. Antimicrob. Agents Chemother. 2010, 54, 3790–3798. [Google Scholar] [CrossRef] [PubMed]
- Mir, A.R.; Kudva, I.T. Antibiotic-resistant Shiga toxin-producing Escherichia coli: An overview of prevalence and intervention strategies. Zoonoses Public Health 2019, 66, 1–13. [Google Scholar] [CrossRef] [PubMed]

| Toxin Type | Bacterium | References |
|---|---|---|
| AB: | ||
| Botulinum toxin | Clostridium botulinum | [22,23] |
| Clostridium baratii | ||
| Clostridium butyricum | ||
| Clostridium argentinense | ||
| Diphtheria toxin | Corynebacterium diphtheriae | [24,25,26,27] |
| Pseudomonas exotoxin A | Pseudomonas aeruginosa | [28,29,30] |
| AB2: | ||
| Cytolethal distending toxin (CTD) | Several Gram-negatives | [31,32,33,34,35] |
| Campylobacter jejuni | ||
| Escherichia coli | ||
| Shigella spp. | ||
| Salmonella enterica Typhi | ||
| Haemophilius ducreyi | ||
| Aggregatibacter actinomycetemcomitans | ||
| Helicobacter spp. | ||
| AB5: | ||
| Cholera toxin (CT) | Vibrio cholerae | [36,37,38,39,40] |
| Pertussis toxin (Ptx) | Bordetella pertussis | [41,42,43,44] |
| ADP-ribosylating toxin (ArtAB) | Salmonella enterica | [45,46,47,48,49,50] |
| Typhimurium | ||
| Salmonella enterica Typhi | ||
| EcPlt | Extra-intestinal E. coli | [51,52] |
| Heat-labile toxin (LT-I and LT-II) | Enterotoxigenic E. coli | [53,54] |
| EcxAB | Clinical strains of E. coli | [55,56] |
| CfxAB | Citrobacter freundii | [55,56] |
| Subtilase (SubAB) | Shiga toxin-producing Escherichia coli (STEC) | [57,58,59,60,61] |
| Shiga toxins (Stx1 and Stx2) | STEC | [62,63,64,65,66,67] |
| Shigella dysenteriae types I and 4 | ||
| Shigella sonnei | ||
| Shigella flexneri | ||
| C. freundii |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biernbaum, E.N.; Kudva, I.T. AB5 Enterotoxin-Mediated Pathogenesis: Perspectives Gleaned from Shiga Toxins. Toxins 2022, 14, 62. https://doi.org/10.3390/toxins14010062
Biernbaum EN, Kudva IT. AB5 Enterotoxin-Mediated Pathogenesis: Perspectives Gleaned from Shiga Toxins. Toxins. 2022; 14(1):62. https://doi.org/10.3390/toxins14010062
Chicago/Turabian StyleBiernbaum, Erika N., and Indira T. Kudva. 2022. "AB5 Enterotoxin-Mediated Pathogenesis: Perspectives Gleaned from Shiga Toxins" Toxins 14, no. 1: 62. https://doi.org/10.3390/toxins14010062
APA StyleBiernbaum, E. N., & Kudva, I. T. (2022). AB5 Enterotoxin-Mediated Pathogenesis: Perspectives Gleaned from Shiga Toxins. Toxins, 14(1), 62. https://doi.org/10.3390/toxins14010062




