Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish
Abstract
:1. Introduction
2. Results and Discussion
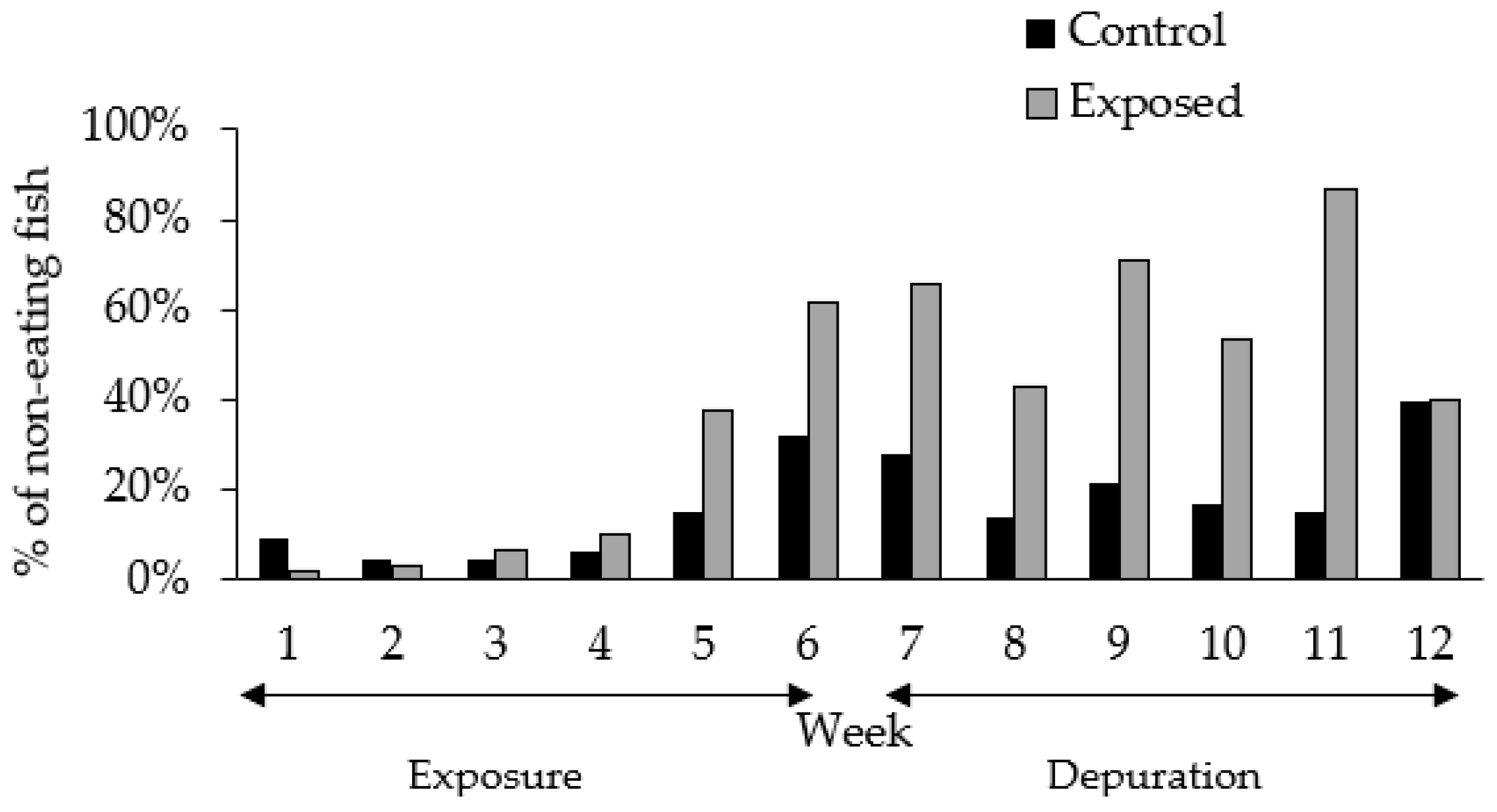
2.1. Experimental Setting and Fish Behavior
2.2. Toxin Levels in the Food and Fish Dosing
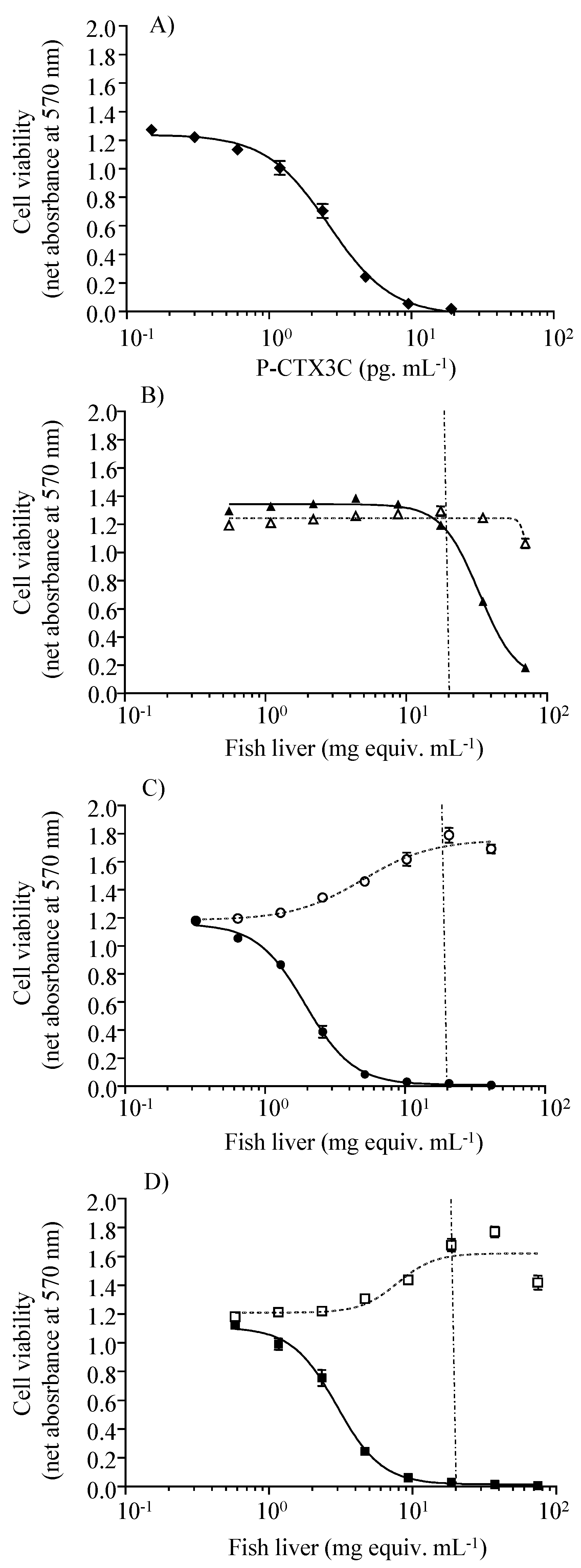
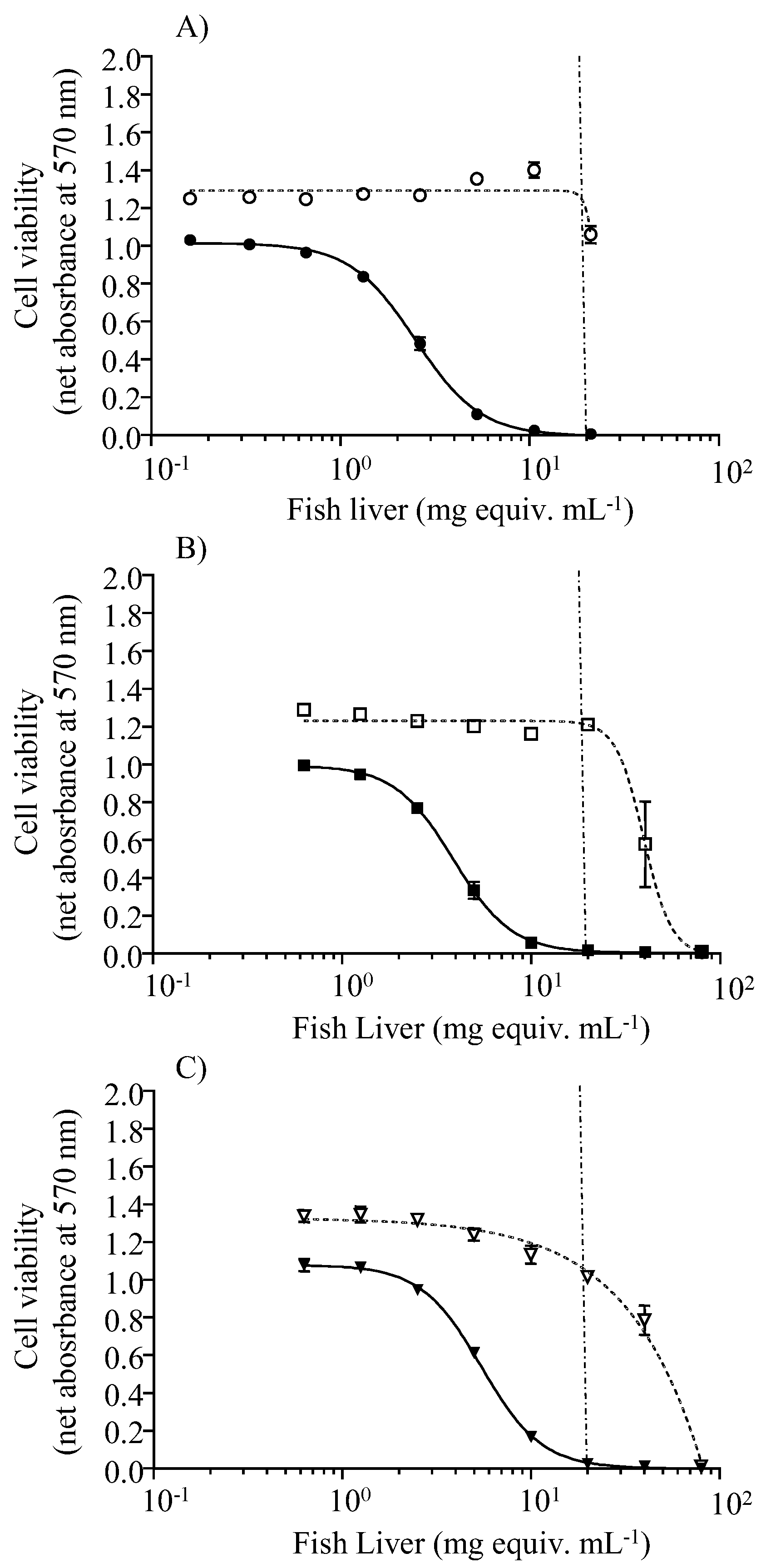
2.3. Toxin Levels in Fish Tissues
3. Conclusions
4. Materials and Methods
4.1. Fish Acclimation and Maintenance
4.2. Preparation of the Experimental Diets
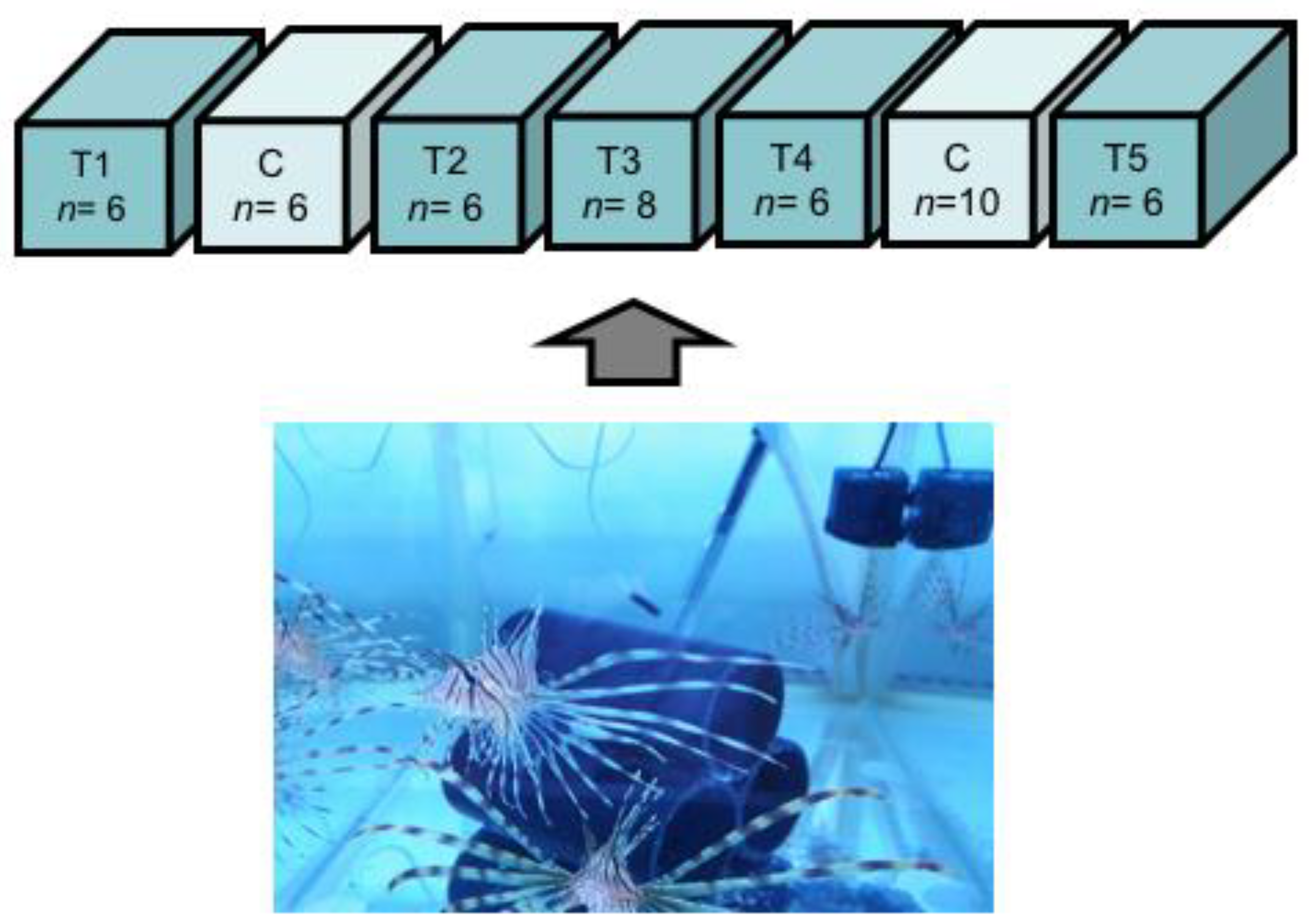
4.3. Experimental Design
Feeding Experiment
4.4. Toxin Determination in Food and Exposed Fish
4.4.1. Sample Extraction
4.4.2. Toxin Analysis
Radioligand-Receptor Binding Assay (r-RBA)
Liquid Chromatography Coupled with Tandem Mass Spectrometry (LC-MS/MS)
Cell Based Assay on Neuroblastoma (CBA-N2a)
4.5. Data Analysis
4.5.1. r-RBA
4.5.2. LC-MS/MS
4.5.3. CBA-N2a
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cuypers, E.; Abdel-Mottaleb, Y.; Kopljar, I.; Rainier, J.D.; Raes, A.L.; Snyders, D.J.; Tytgat, J. Gambierol, a toxin produced by the dinoflagellate Gambierdiscus toxicus, is a potent blocker of voltage-gated potassium channels. Toxicon 2008, 51, 974–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoppenrath, M.; Murray, S.A.; Chomérat, N.; Horiguchi, T. Marine Benthic Dinoflagellates—Unveiling Their Worldwide Biodiversity; Senckenberg-Reihe: Frankfurt, Germany, 2014. [Google Scholar]
- Morohashi, A.; Satake, M.; Nagai, H.; Oshima, Y.; Yasumoto, T. The absolute configuration of gambieric acids A-D, potent antifungal polyethers, isolated from the marine dinoflagellate Gambierdiscus toxicus. Tetrahedron 2000, 56, 8995–9001. [Google Scholar] [CrossRef]
- Murray, J.S.; Selwood, A.I.; Harwood, D.T.; van Ginkel, R.; Puddick, J.; Rhodes, L.L.; Rise, F.; Wilkins, A.L. 44-Methylgambierone, a new gambierone analogue isolated from Gambierdiscus australes. Tetrahedron Lett. 2019, 60, 621–625. [Google Scholar] [CrossRef]
- Soliño, L.; Costa, P.R. Differential toxin profiles of ciguatoxins in marine organisms: Chemistry, fate and global distribution. Toxicon 2018, 150, 1–70. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, R.; Uchida, H.; Suzuki, T.; Matsushima, R.; Nagae, M.; Toyohara, Y.; Satake, M.; Oshima, Y.; Inoue, A.; Yasumoto, T. Gambieroxide, a novel epoxy polyether compound from the dinoflagellate Gambierdiscus toxicus GTP2 strain. Tetrahedron 2013, 69, 10299–10303. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain. Scientific opinion on marine biotoxins in shellfish—Emerging toxins: Ciguatoxin group. EFSA J. 2010, 8, 1–38. [Google Scholar] [CrossRef]
- Friedman, M.A.; Fernandez, M.; Backer, L.C.; Dickey, R.W.; Bernstein, J.; Schrank, K.; Kibler, S.; Stephan, W.; Gribble, M.O.; Bienfang, P.; et al. An updated review of ciguatera fish poisoning: Clinical, epidemiological, environmental, and public health management. Mar. Drugs 2017, 15, 72. [Google Scholar] [CrossRef]
- Parsons, M.L.; Aligizaki, K.; Bottein, M.Y.D.; Fraga, S.; Morton, S.L.; Penna, A.; Rhodes, L. Gambierdiscus and Ostreopsis: Reassessment of the state of knowledge of their taxonomy, geography, ecophysiology, and toxicology. Harmful Algae 2012, 14, 107–129. [Google Scholar] [CrossRef]
- Yasumoto, T.; Seino, N.; Murakami, Y.; Murata, M. Toxins produced by benthic dinoflagellates. Biol. Bull. 1987, 172, 128–131. [Google Scholar] [CrossRef]
- Dechraoui, M.-Y.; Naar, J.; Pauillac, S.; Legrand, A.M. Ciguatoxins and brevetoxins, neurotoxic polyether compounds active on sodium channels. Toxicon 1999, 37, 125–143. [Google Scholar] [CrossRef]
- Kumar-Roiné, S.; Darius, H.T.; Matsui, M.; Fabre, N.; Haddad, M.; Chinain, M.; Pauillac, S.; Laurent, D. A review of traditional remedies of ciguatera fish poisoning in the Pacific. Phytother. Res. 2011, 25, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Lombet, A.; Bidard, J.-N.; Lazdunski, M. Ciguatoxins and brevetoxins share a common receptor site on the neuronal voltage-dependent Na+ channel. FEBS Lett. 1987, 219, 355–359. [Google Scholar] [CrossRef] [Green Version]
- Poli, M.A.; Mende, T.J.; Baden, D.G. Brevetoxins, unique activators of voltage-sensitive sodium channels, bind to specific sites in rat brain synaptosomes. Mol. Pharmacol. 1986, 30, 129–135. [Google Scholar]
- Au, N.P.B.; Kumar, G.; Asthana, P.; Tin, C.; Mak, Y.L.; Chan, L.L.; Lam, P.K.S.; Ma, C.H.E. Ciguatoxin reduces regenerative capacity of axotomized peripheral neurons and delays functional recovery in pre-exposed mice after peripheral nerve injury. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chinain, M.; Gatti, C.M.; Roué, M.; Darius, H.T. Ciguatera poisoning in French Polynesia: Insights into the novel trends of an ancient disease. New Microbes New Infect. 2019, 31, 100565. [Google Scholar] [CrossRef]
- Dickey, R.W.; Plakas, S.M. Ciguatera: A public health perspective. Toxicon 2010, 56, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Davin, W.T.; Kohler, C.C.; Tindall, D.R. Effects of ciguatera toxins on the bluehead. Trans. Am. Fish. Soc. 1986, 115, 908–912. [Google Scholar] [CrossRef]
- Kelly, A.M.; Kohler, C.C.; Tindall, D.R. Are crustaceans linked to the ciguatera food chain? Environ. Biol. Fishes 1992, 33, 275–286. [Google Scholar] [CrossRef]
- Colman, J.R.; Dechraoui, M.Y.B.; Dickey, R.W.; Ramsdell, J.S. Characterization of the developmental toxicity of Caribbean ciguatoxins in finfish embryos. Toxicon 2004, 44, 59–66. [Google Scholar] [CrossRef]
- Edmunds, J.S.G.; McCarthy, R.A.; Ramsdell, J.S. Ciguatoxin reduces larval survivability in finfish. Toxicon 1999, 37, 1827–1832. [Google Scholar] [CrossRef]
- Mak, Y.L.; Li, J.; Liu, C.N.; Cheng, S.H.; Lam, P.K.S.; Cheng, J.; Chan, L.L. Physiological and behavioural impacts of Pacific ciguatoxin-1 (P-CTX-1) on marine medaka (Oryzias melastigma). J. Hazard. Mater. 2017, 321, 782–790. [Google Scholar] [CrossRef]
- Yan, M.; Leung, P.T.Y.; Ip, J.C.H.; Cheng, J.; Wu, J.-J.; Gu, J.R.; Lam, P.K.S. Developmental toxicity and molecular responses of marine medaka (Oryzias melastigma) embryos to ciguatoxin P-CTX-1 exposure. Aquat. Toxicol. 2017, 185, 149–159. [Google Scholar] [CrossRef]
- Yan, M.; Mak, M.Y.L.; Cheng, J.; Li, J.; Gu, J.R.; Leung, P.T.Y.; Lam, P.K.S. Effects of dietary exposure to ciguatoxin P-CTX-1 on the reproductive performance in marine medaka (Oryzias melastigma). Mar. Pollut. Bull. 2020, 152, 110–837. [Google Scholar] [CrossRef] [PubMed]
- Ledreux, A.; Brand, H.; Chinain, M.; Bottein, M.Y.D.; Ramsdell, J.S. Dynamics of ciguatoxins from Gambierdiscus polynesiensis in the benthic herbivore Mugil cephalus: Trophic transfer implications. Harmful Algae 2014, 39, 165–174. [Google Scholar] [CrossRef]
- Bottein, M.Y.D.; Kashinsky, L.; Wang, Z.; Littnan, C.; Ramsdell, J.S. Identification of ciguatoxins in hawaiian monk seals Monachus schauinslandi from the northwestern and main hawaiian Islands. Environ. Sci. Technol. 2011, 45, 5403–5409. [Google Scholar] [CrossRef] [PubMed]
- Clausing, R.J.; Losen, B.; Oberhaensli, F.R.; Darius, H.T.; Sibat, M.; Hess, P.; Swarzenski, P.W.; Chinain, M.; Dechraoui Bottein, M.Y. Experimental evidence of dietary ciguatoxin accumulation in an herbivorous coral reef fish. Aquat. Toxicol. 2018, 200, 257–265. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Mak, Y.L.; Chang, Y.H.; Xiao, C.; Chen, Y.M.; Shen, J.; Wang, Q.; Ruan, Y.; Lam, P.K.S. Uptake and depuration kinetics of Pacific Ciguatoxins in orange-spotted grouper (Epinephelus coioides). Environ. Sci. Technol. 2020, 54, 4475–4483. [Google Scholar] [CrossRef]
- Dechraoui-Bottein, M.-Y.; Tiedeken, J.A.; Persad, R.; Wang, Z.; Granade, H.R.; Dickey, R.W.; Ramsdell, J.S. Use of two detection methods to discriminate ciguatoxins from brevetoxins: Application to great barracuda from Florida Keys. Toxicon 2005, 46, 261–270. [Google Scholar] [CrossRef]
- Sanchez-Henao, A.; García-Álvarez, N.; Sergent, F.S.; Estévez, P.; Gabo-Martínez, A.; Martín, F.; Ramos-Sosa, M.; Fernández, A.; Diogène, J.; Real, F. Presence of CTXs in moray eels and dusky groupers in the marine environment of the Canary Islands. Aquat. Toxicol. 2020, 221, 105427. [Google Scholar] [CrossRef] [PubMed]
- Yogi, K.; Oshiro, N.; Inafuku, Y.; Hirama, M.; Yasumoto, T. Detailed LC-MS/MS analysis of ciguatoxins revealing distinct regional and species characteristics in fish and causative alga from the Pacific. Anal. Chem. 2011, 83, 8886–8891. [Google Scholar] [CrossRef]
- Halstead, B.W. Poisonous and Venomous Marine Animals of the World; The Darwin Press: Princeton, NJ, USA, 1978. [Google Scholar]
- WHO. Report of the Expert Meeting on Ciguatera Poisoning; Food Safety and Quality: Rome, Italy, 2020; Volume 9, pp. 19–23. [Google Scholar] [CrossRef]
- Clausing, R.J.; Chinain, M.; Dechraoui-Bottein, M.Y. Practical sampling guidance for determination of ciguatoxin. In Guide for Designing and Implementing a Plan to Monitor Toxin-Producing Microalgae; Reguera, B., Alonso, R., Moreira, A., Mendez, S., Dechraoui Bottein, M.Y., Eds.; IOC-UNESCO: Paris, France, 2016; pp. 51–63. [Google Scholar]
- Chan, W.H.; Mak, Y.L.; Wu, J.J.; Jin, L.; Sit, W.H.; Lam, J.C.W.; Mitcheson, Y.S.; Chan, L.L.; Lam, P.K.S.; Murphy, M.B. Spatial distribution of ciguateric fish in the Republic of Kiribati. Chemosphere 2011, 84, 117–123. [Google Scholar] [CrossRef]
- Díaz-Asencio, L.; Clausing, R.J.; Vandersea, M.; Chamero-Lago, D.; Gómez-Batiste, M.; Hernández-Albernas, J.I.; Chomérat, N.; Rojas-Abrahantes, G.; Litaker, R.W.; Tester, P.; et al. Ciguatoxin occurrence in food-web components of a cuban coral reef ecosystem: Risk-assessment implications. Toxins 2019, 11, 722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, R.J.; Holmes, M.J. Mini review origin and transfer of toxins involved in Ciguatera. Biochem. Physiol 1993, 106, 615–628. [Google Scholar]
- Chinain, M.; Darius, H.T.; Ung, A.; Tchou Fouc, M.; Revel, T.; Cruchet, P.; Pauillac, S.; Laurent, D. Ciguatera risk management in French Polynesia: The case study of Raivavae Island (Australes Archipelago). Toxicon 2010, 56, 674–690. [Google Scholar] [CrossRef]
- Chinain, M.; Gatti, C.M.; Ung, A.; Cruchet, P.; Revel, T.; Viallon, J.; Sibat, M.; Varney, P.; Laurent, V.; Hess, P.; et al. Evidence for the range expansion of Ciguatera in French Polynesia: A revisit of the 2009 mass-poisoning outbreak in Rapa Island (Australes Archipelago). Toxins 2020, 12, 759. [Google Scholar] [CrossRef]
- Darius, H.T.; Ponton, D.; Revel, T.; Cruchet, P.; Ung, A.; Tchou Fouc, M.; Chinain, M. Ciguatera risk assessment in two toxic sites of French Polynesia using the receptor-binding assay. Toxicon 2007, 50, 612–626. [Google Scholar] [CrossRef]
- Gaboriau, M.; Ponton, D.; Darius, H.T.; Chinain, M. Ciguatera fish toxicity in French Polynesia: Size does not always matter. Toxicon 2014, 84, 41–50. [Google Scholar] [CrossRef]
- Mak, Y.L.; Wai, T.C.; Murphy, M.B.; Chan, W.H.; Wu, J.J.; Lam, J.C.W.; Chan, L.L.; Lam, P.K.S. Pacific ciguatoxins in food web components of coral reef systems in the Republic of Kiribati. Environ. Sci. Technol. 2013, 47, 14070–14079. [Google Scholar] [CrossRef] [PubMed]
- Saunders, P.R.; Taylor, P.B. Venom of the lionfish Pterois volitans. Am. J. Physiol. 1959, 197, 437–440. [Google Scholar] [CrossRef] [Green Version]
- Snyder, D.B.; Burgess, G.H. The Indo-Pacific red lionfish, Pterois volitans (Pisces: Scorpaenidae), new to Bahamian ichthyofauna. Coral Reefs 2007, 26, 175. [Google Scholar] [CrossRef]
- Whitfield, P.E.; Gardner, T.; Vives, S.P.; Gilligan, M.R.; Courtenay, W.R.; Ray, G.C.; Hare, J.A. Biological invasion of the Indo-Pacific lionfish Pterois volitans along the Atlantic coast of North America. Mar. Ecol. Prog. Ser. 2002, 235, 289–297. [Google Scholar] [CrossRef] [Green Version]
- Hardison, D.R.; Holland, W.C.; Darius, H.T.; Chinain, M.; Tester, P.A.; Shea, D.; Bogdanoff, A.K.; Morris, J.A.J.; Quintana, H.A.F.; Loeffler, C.R.; et al. Investigation of ciguatoxins in invasive lionfish from the greater caribbean region: Implications for fishery development. PLoS ONE 2018, 13, e0198358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, J.A.J.; Whitfield, P.E. Biology, ecology, control and management of the invasive Indo-Pacific Lionfish: An updated integrated assessment. In NOAA Technical Memorandum NOS NCCOS, 99; NOAA: Washington, DC, USA, 2009; p. 57. [Google Scholar]
- FDA. Fish and fishery products hazards and controls guidance. Fish. Prod. Hazard. Control. Guide 2011, 4, 1–401. [Google Scholar]
- Robertson, A.; Garcia, A.C.; Flores Quintana, H.A.; Smith, T.B.; Castillo, B.F.; Reale-Munroe, K.; Gulli, J.A.; Olsen, D.A.; Hooe-Rollman, J.I.; Jester, E.L.E.; et al. Invasive lionfish (Pterois volitans): A potential human health threat for ciguatera fish poisoning in tropical waters. Mar. Drugs 2014, 12, 88. [Google Scholar] [CrossRef] [Green Version]
- Wilcox, C.L.; Hixon, M.A. False positive tests for ciguatera may derail efforts to control invasive lionfish. Environ. Biol. Fishes 2014, 98, 961–969. [Google Scholar] [CrossRef]
- Boucaud-Maitre, D.; Vernoux, J.P.; Pelczar, S.; Daudens-Vaysse, E.; Aubert, L.; Boa, S.; Ferracci, S.; Garnier, R. Incidence and clinical characteristics of ciguatera fish poisoning in Guadeloupe (French West Indies) between 2013 and 2016: A retrospective cases-series. Sci. Rep. 2018, 8, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banner, A.; Helfrich, P.; Yoshida, T. Research on ciguatera in the tropical Pacific. Proc. Gulf Caribb. Fish. Inst. 1963, 16, 84–98. [Google Scholar]
- Hahn, S.T.; Capra, M.F.; Walsh, T.P. Ciguatoxin-protein association in skeletal muscle of spanish mackerel (Scomberomorus commersoni). Toxicon 1992, 30, 843–852. [Google Scholar] [CrossRef]
- Chinain, M.; Gatti, C.M.; Martin-Yken, H.; Roué, M.; Darius, H.T. Ciguatera poisoning: An increasing burden for Pacific Islands communities in light of climate change? In Climate Change and Marine and Freshwater Toxins, 2nd ed.; Botana, L.M., Louzao, M.C., Vilariño, N., Eds.; De Gruyter: Berlin, Germany; Boston, MA, USA, 2021; pp. 369–428. [Google Scholar] [CrossRef]
- Ikehara, T.; Kuniyoshi, K.; Oshiro, N.; Yasumoto, T. Biooxidation of ciguatoxins leads to species-specific toxin profiles. Toxins 2017, 9, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yogi, K.; Sakugawa, S.; Oshiro, N.; Ikehara, T.; Sugiyama, K.; Yasumoto, T. Determination of toxins involved in ciguatera fish poisoning in the Pacific by LC/MS. J. AOAC Int. 2014, 97, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J.; Sellin, M.; Poli, M.A.; Norton, R.S.; MacLeod, J.K.; Sheil, M.M. Purification and characterization of ciguatoxins from moray eel (Lycodontis javanicus, Muraenidae). Toxicon 1991, 29, 1115–1127. [Google Scholar] [CrossRef]
- Banner, A.H.; Helfrich, P.; Piyakarnchana, T. Retention of Ciguatera toxin by the red snapper, Lutjanus bohar. Copeia 1966, 2, 297–301. [Google Scholar] [CrossRef]
- Pottier, I.; Vernoux, J.P.; Jones, A.; Lewis, R.J. Analysis of toxin profiles in three different fish species causing ciguatera fish poisoning in Guadeloupe, French West Indies. Food Addit. Contam. 2002, 19, 1034–1042. [Google Scholar] [CrossRef]
- Pottier, I.; Hamilton, B.; Jones, A.; Lewis, R.J.; Vernoux, J.P. Identification of slow and fast-acting toxins in a highly ciguatoxic barracuda (Sphyraena barracuda) by HPLC/MS and radiolabelled ligand binding. Toxicon 2003, 42, 663–672. [Google Scholar] [CrossRef]
- Soliño, L.; Widgy, S.; Pautonnier, A.; Turquet, J.; Loeffler, C.R.; Flores Quintana, H.A.; Diogène, J. Prevalence of ciguatoxins in lionfish (Pterois spp.) from Guadeloupe, Saint Martin, and Saint Barthélmy Islands (Caribbean). Toxicon 2015, 102, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Green, S.J.; Akins, J.L.; Morris, J. Lionfish dissection: Techniques and applications. NOAA Tech. Memo. 2012, 139, 1–24. [Google Scholar]
- Detection of Harmful Algal Toxins Using the Radioligand Receptor Binding Assay: A Manual of Methods; IAEA-TECDOC-1729; IAEA: Vienna, Austria, 2013. [CrossRef]
- Díaz-Asencio, L.; Clausing, R.J.; Rañada, M.L.; Alonso-Hernández, C.M.; Dechraoui Bottein, M.-Y. A radioligand receptor binding assay for ciguatoxin monitoring in environmental samples: Method development and determination of quality control criteria. J. Environ. Radioact. 2018, 192, 289–294. [Google Scholar] [CrossRef]
- Dechraoui Bottein, M.-Y.; Clausing, R.J. Receptor binding assay for the analysis of marine toxins: Detection and mode of action. In Recent Advances in the Analysis of Marine Toxins; Diogène, J., Campàs, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 277–301. [Google Scholar]
- Viallon, J.; Chinain, M.; Darius, H.T. Revisiting the neuroblastoma cell-based assay (CBA-N2a) for the improved detection of marine toxins active on voltage gated sodium channels (VGSCs). Toxins 2020, 12, 281. [Google Scholar] [CrossRef] [PubMed]
| Periods of Experiment | Fish Individual | Days of Exposure/ Depuration | Fish Total Weight (g) | Liver Wet Weight (g) (% Contribution) | Toxin Concentration (ng CTX3C Equiv. g−1) |
|---|---|---|---|---|---|
| 1 | 30 | 20.3 | 0.53 (3%) | <LOD ** | |
| Exposure | 2 | 30 | 16.9 | 0.45 (3%) | 7.08 |
| 3 | 30 | 10.0 | 0.30 (3%) | <LOD ** | |
| 4 | 30 | 5.56 | 0.08 (1.5%) | <LOD ** | |
| 5 | 41 | 21.0 | 0.74 (3.5%) | <LOD ** | |
| 6 | 41 | 14.4 | 0.33 (2%) | <LOD ** | |
| 7 | 41 | 17.6 | 0.39 (2%) | 9.43 | |
| 8 | 41 | 14.4 | 0.40 (3%) | <LOD ** | |
| 9 | 41 | 9.08 | 0.22 (2%) | <LOD ** | |
| Depuration | 10 | 8 | 19.4 | 0.65 (3%) | 9.30 |
| 11 | 8 | 15.0 | 0.61 (4%) | 9.42 | |
| 12 | 29 | 16.7 | 0.55 (3%) | 8.93 | |
| 13 | 29 | 8.72 | 0.02 (0.2%) | <LOD ** | |
| 14 | 29 | 9.81 | 0.11(1.1%) | <LOD ** | |
| 15 | 43 | 21.2 | 0.34 (2%) | 9.77 | |
| 16 | 43 | 10.3 | 0.18 (2%) | <LOD ** |
| Periods of Experiment | Days of Exposure/ Depuration | Pooled Samples (n) | Fish Liver Weight Per Pool (g) | EC50 (mg Equiv. mL−1) | Toxin Concentration (ng CTX3C equiv. g−1) |
|---|---|---|---|---|---|
| Exposure | 30 | 4 | 0.47 | 1.93 | 1.36 |
| 41 | 5 | 0.19 | 2.97 | 0.89 | |
| Control | 42 | 2 | 0.50 | ND * | <LOD ** |
| 8 | 1 | 0.51 | ND * | <LOD ** | |
| Depuration | 8 | 2 | 0.23 | 2.49 | 0.81 |
| 29 | 3 | 0.13 | 3.88 | 0.52 | |
| 43 | 2 | 0.11 | 5.46 | 0.37 |
| Periods of Experiment | Cumulative Number of Experimental Weeks | Days of Exposure/ Depuration | Cumulative Number of Feeding Episodes Toxic/Control Food | n |
|---|---|---|---|---|
| Exposure | 5 | 30 days of exposure | 22/0 | 4 |
| 6 | 41 days of exposure | 29/0 | 5 | |
| Depuration | 7 | 8 days of depuration | 29/6 | 3 |
| 10 | 29 days of depuration | 29/20 | 3 | |
| 12 | 43 days of depuration | 29/30 | 2 |
| Compound | MRM Transitions (m/z) | Precursor Ion Species | Targeted Time (min) | CE (eV) | |
|---|---|---|---|---|---|
| Precursor Ion (Q1) | Product Ion (Q3) | ||||
| CTX3C or CTX3B | 1040.6 | 1005.6 | [M + NH4]+ | 11.2 | 30 |
| 1023.6 | 1005.6 | [M + H]+ | 20 | ||
| 1023.6 | 125.1 | 50 | |||
| M-seco-CTX3C | 1041.6 | 1023.6 | [M + H]+ | 4.7 | 20 |
| 1041.6 | 1005.6 | 30 | |||
| 1041.6 | 125.1 | 50 | |||
| 2-hydroxyCTX3C | 1058.6 | 1005.6 | [M + NH4]+ | 5.0 | 30 |
| 1058.6 | 1023.6 | 20 | |||
| 1058.6 | 125.1 | 50 | |||
| 51-hydroxyCTX3C | 1056.6 | 1021.6 | [M + NH4]+ | 6.3 | 30 |
| 1039.6 | 1021.6 | [M + H]+ | 20 | ||
| 1039.6 | 1003.6 | 20 | |||
| CTX4A or CTX4B | 1078.6 | 1043.6 | [M + NH4]+ | 12.8 | 30 |
| 1061.6 | 1043.6 | [M + H]+ | 20 | ||
| 1061.6 | 125.1 | 50 | |||
| 54-deoxyCTX1B or 52-epi-54- deoxyCTX1B | 1112.6 | 1077.6 | [M + NH4]+ | 6.8 | 20 |
| 1112.6 | 1059.6 | 30 | |||
| 1112.6 | 95.1 | 90 | |||
| CTX1B | 1128.6 | 1093.6 | [M + NH4]+ | 3.2 | 20 |
| 1128.6 | 1075.6 | 30 | |||
| 1128.6 | 95.1 | 90 | |||
| C-CTX1 or C-CTX2 | 1141.4 | 1123.4 | [M + H]+ | 4.4 | 30 |
| 1123.4 | 1105.6 | [M + H–H2O]+ | 30 | ||
| 1123.4 | 1087.6 | 30 | |||
| 2,3-dihydro-2,3- dihydroxyCTX3C | 1074.6 | 1039.6 | [M + NH4]+ | 6.0 | 30 |
| 1057.6 | 1039.6 | [M + H]+ | 20 | ||
| 1057.6 | 125.1 | 50 | |||
| CTX3C analog 1 | 1040.6 | 1005.6 | [M + NH4]+ | 10.0 | 30 |
| 1023.6 | 1005.6 | [M + H]+ | 20 | ||
| 1023.5 | 125.1 | 50 | |||
| CTX3C analog 2 | 1040.6 | 1005.6 | [M + NH4]+ | 8.5 | 30 |
| 1023.6 | 1005.6 | [M + H]+ | 20 | ||
| 1023.5 | 125.1 | 50 | |||
| CTX3C analog 3 | 1040.6 | 1005.6 | [M + NH4]+ | 7.5 | 30 |
| 1023.6 | 1005.6 | [M + H]+ | 20 | ||
| 1023.5 | 125.1 | 50 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leite, I.d.P.; Sdiri, K.; Taylor, A.; Viallon, J.; Gharbia, H.B.; Mafra Júnior, L.L.; Swarzenski, P.; Oberhaensli, F.; Darius, H.T.; Chinain, M.; et al. Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish. Toxins 2021, 13, 564. https://doi.org/10.3390/toxins13080564
Leite IdP, Sdiri K, Taylor A, Viallon J, Gharbia HB, Mafra Júnior LL, Swarzenski P, Oberhaensli F, Darius HT, Chinain M, et al. Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish. Toxins. 2021; 13(8):564. https://doi.org/10.3390/toxins13080564
Chicago/Turabian StyleLeite, Isabel do Prado, Khalil Sdiri, Angus Taylor, Jérôme Viallon, Hela Ben Gharbia, Luiz Laureno Mafra Júnior, Peter Swarzenski, François Oberhaensli, Hélène Taiana Darius, Mireille Chinain, and et al. 2021. "Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish" Toxins 13, no. 8: 564. https://doi.org/10.3390/toxins13080564
APA StyleLeite, I. d. P., Sdiri, K., Taylor, A., Viallon, J., Gharbia, H. B., Mafra Júnior, L. L., Swarzenski, P., Oberhaensli, F., Darius, H. T., Chinain, M., & Bottein, M.-Y. D. (2021). Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish. Toxins, 13(8), 564. https://doi.org/10.3390/toxins13080564









