Evaluation of Anti-PT Antibody Response after Pertussis Vaccination and Infection: The Importance of Both Quantity and Quality
Abstract
1. Introduction
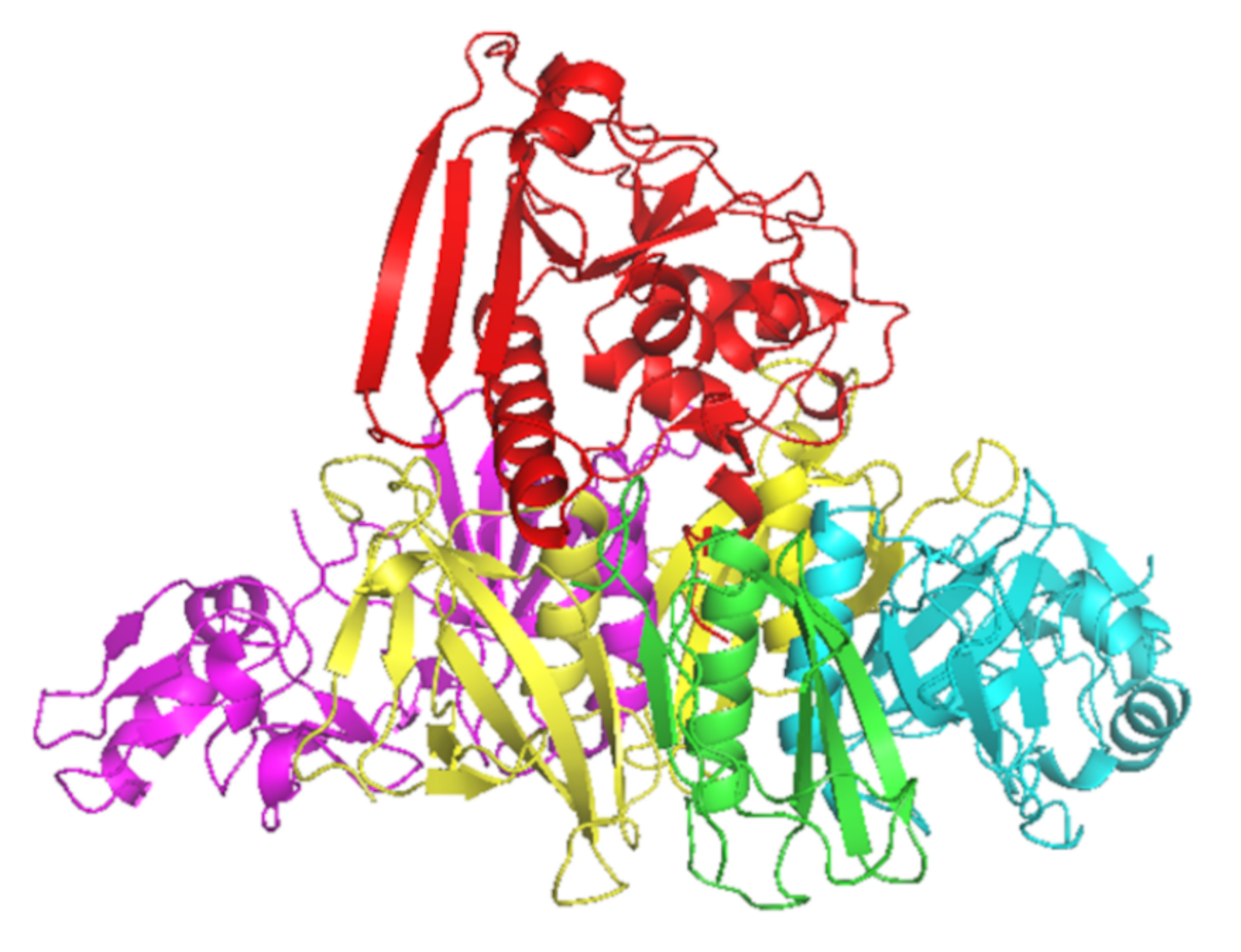
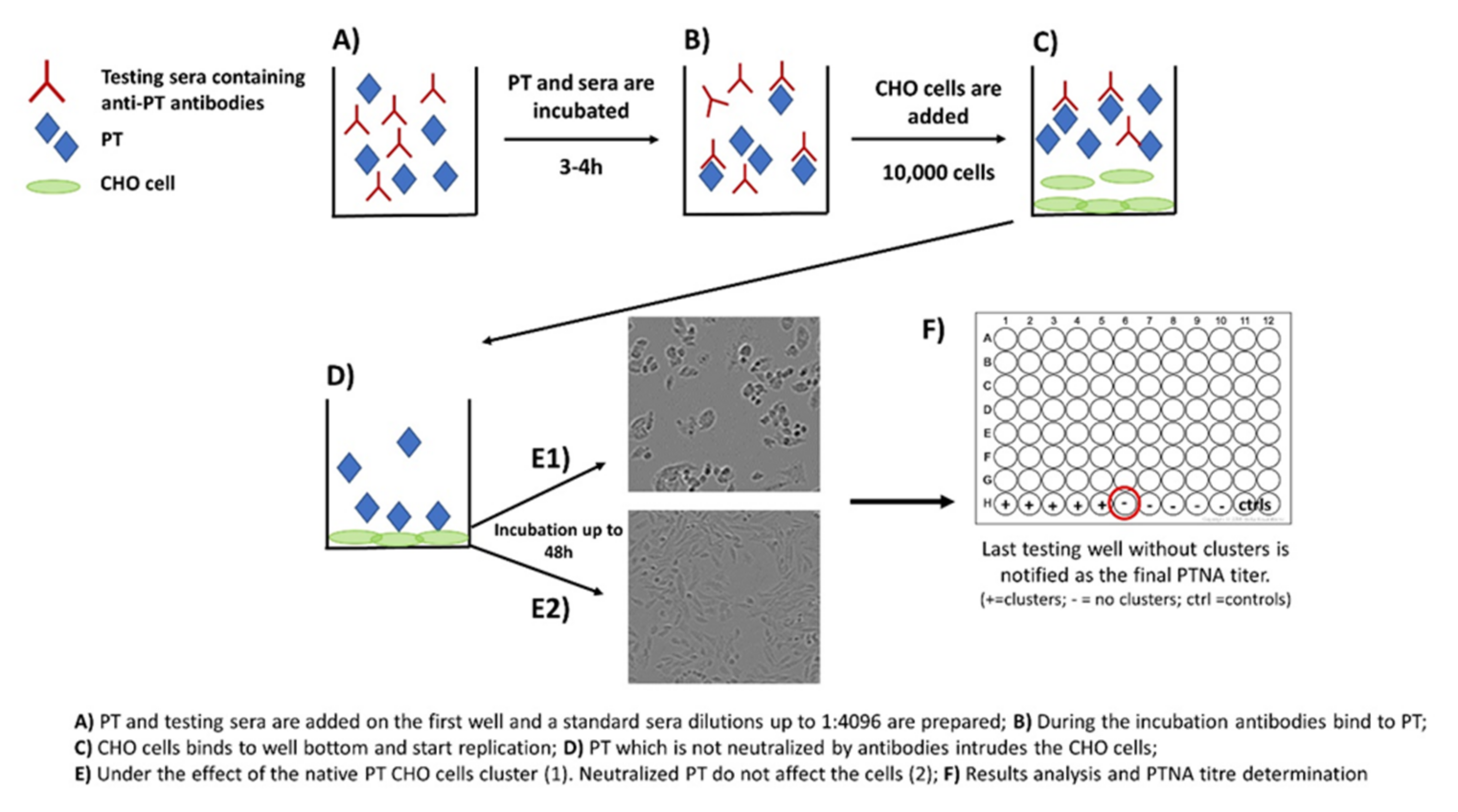
2. The Role of Neutralizing Antibodies to Pertussis Toxin (PTNAs)
3. B Memory Cells as Carriers of Improved Antibodies
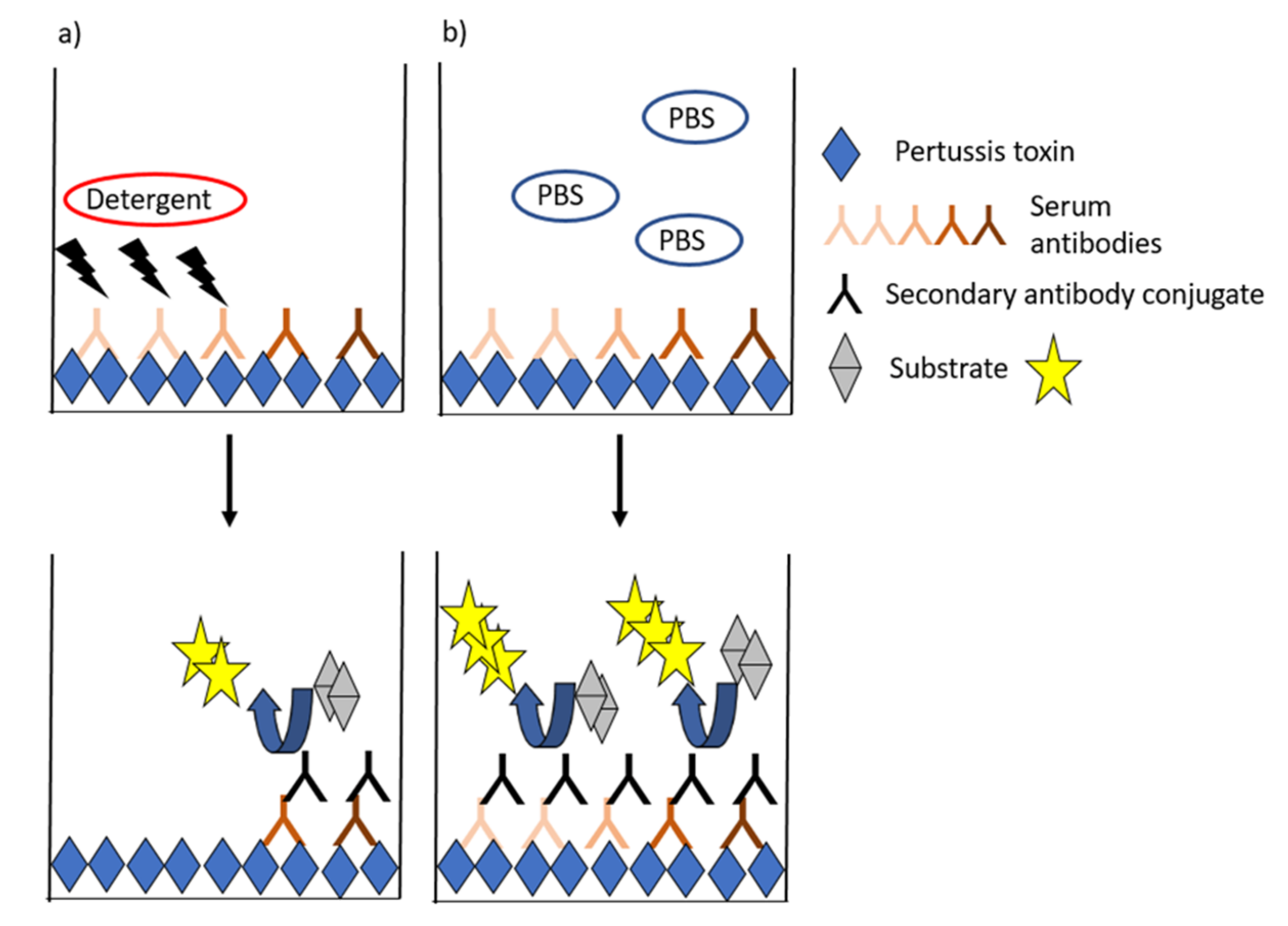
4. Avidity as a Marker of Effective Antibodies to PT
5. PT Specific Epitopes after Infection and Vaccination
6. Final Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pittman, M. Pertussis toxin: The cause of the harmful effects and prolonged immunity of whooping cough. A hypothesis. Rev. Infect. Dis. 1979, 1, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Locht, C. Molecular aspects of Bordetella pertussis pathogenesis. Int. Microbiol. 1999, 2, 137–144. [Google Scholar] [PubMed]
- Carbonetti, N.H. Pertussis toxin and adenylate cyclase toxin: Key virulence factors of Bordetella pertussis and cell biology tools. Future Microbiol. 2010, 5, 455–469. [Google Scholar] [CrossRef]
- Nicosia, A.; Perugini, M.; Franzini, C.; Casagli, M.C.; Borri, M.G.; Antoni, G.; Neri, P.; Ratti, G.; Rappuoli, R. Cloning and sequencing of the pertussis toxin genes: Operon structure and gene duplication. Proc. Natl. Acad. Sci. USA 1986, 83, 4631–4635. [Google Scholar] [CrossRef]
- Sato, H.; Ito, A.; Chiba, J.; Sato, Y. Monoclonal antibody against pertussis toxin: Effect on toxin activity and pertussis infections. Infect. Immun. 1984, 46, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Locht, C.; Keith, J.M. Pertussis toxin gene: Nucleotide sequence and genetic organization. Science 1986, 232, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Podda, A.; Nencioni, L.; De Magistris, M.T.; Di Tommaso, A.; Bossù, P.; Nuti, S.; Pileri, P.; Peppoloni, S.; Bugnoli, M.; Ruggiero, P.; et al. Metabolic, humoral, and cellular responses in adult volunteers immunized with the genetically inactivated pertussis toxin mutant PT-9K/129G. J. Exp. Med. 1990, 172, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Podda, A.; Carapella De Luca, E.; Titone, L.; Casadei, A.M.; Cascio, A.; Bartalini, M.; Volpini, G.; Peppoloni, S.; Marsili, I.; Nencioni, L.; et al. Immunogenicity of an acellular pertussis vaccine composed of genetically inactivated pertussis toxin combined with filamentous hemagglutinin and pertactin in infants and children. J. Pediatr. 1993, 123, 81–84. [Google Scholar] [CrossRef]
- Rappuoli, R. The vaccine containing recombinant pertussis toxin induces early and long-lasting protection. Biologicals 1999, 27, 99–102. [Google Scholar] [CrossRef]
- Cherry, J.D.; Gornbein, J.; Heininger, U.; Stehr, K. A search for serologic correlates of immunity to Bordetella pertussis cough illnesses. Vaccine 1998, 16, 1901–1906. [Google Scholar] [CrossRef]
- Storsaeter, J.; Hallander, H.O.; Gustafsson, L.; Olin, P. Levels of anti-pertussis antibodies related to protection after household exposure to Bordetella pertussis. Vaccine 1998, 16, 1907–1916. [Google Scholar] [CrossRef]
- Storsaeter, J.; Hallander, H.O.; Gustafsson, L.; Olin, P. Low levels of antipertussis antibodies plus lack of history of pertussis correlate with susceptibility after household exposure to Bordetella pertussis. Vaccine 2003, 21, 3542–3549. [Google Scholar] [CrossRef]
- Schure, R.M.; Hendrikx, L.H.; de Rond, L.G.; Oztürk, K.; Sanders, E.A.; Berbers, G.A.; Buisman, A.M. Differential T- and B-cell responses to pertussis in acellular vaccine-primed versus whole-cell vaccine-primed children 2 years after preschool acellular booster vaccination. Clin. Vaccine Immunol. 2013, 20, 1388–1395. [Google Scholar] [CrossRef]
- Carollo, M.; Pandolfi, E.; Tozzi, A.E.; Buisman, A.M.; Mascart, F.; Ausiello, C.M. Humoral and B-cell memory responses in children five years after pertussis acellular vaccine priming. Vaccine 2014, 3, 2093–2099. [Google Scholar] [CrossRef]
- Plotkin, S.A. Complex correlates of protection after vaccination. Clin. Infect. Dis. 2013, 56, 1458–1465. [Google Scholar] [CrossRef] [PubMed]
- Edelman, K.; He, Q.; Makinen, J.; Sahlberg, A.; Haanpera, M.; Schuerman, L.; Wolter, J.; Mertsola, J. Immunity to pertussis 5 years after booster immunization during adolescence. Clin. Infect. Dis. 2007, 44, 1271–1277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dalby, T.; Petersen, J.W.; Harboe, Z.B.; Krogfelt, K.A. Antibody responses to pertussis toxin display different kinetics after clinical Bordetella pertussis infection than after vaccination with an acellular pertussis vaccine. J. Med. Microbiol. 2010, 59, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Hallander, H.O.; Ljungman, M.; Storsaeter, J.; Gustafsson, L. Kinetics and sensitivity of ELISA IgG pertussis antitoxin after infection and vaccination with Bordetella pertussis in young children. APMIS 2009, 117, 797–807. [Google Scholar] [CrossRef]
- Dalby, T.; Andersen, P.H.; Hoffmann, S. Epidemiology of pertussis in Denmark, 1995 to 2013. Eurosurveillance 2016, 21. [Google Scholar] [CrossRef]
- Bhavsar, A.; Mertsola, J.; Poulsen, A.; Silfverdal, S.A. Pertussis in infants in Nordic countries. Acta Paediatr. 2021. [Google Scholar] [CrossRef]
- Barkoff, A.M.; Grondahl-Yli-Hannuksela, K.; He, Q. Seroprevalence studies of pertussis: What have we learned from different immunized populations. Pathog. Dis. 2015, 73. [Google Scholar] [CrossRef]
- Minh, N.N.; He, Q.; Ramalho, A.; Kaufhold, A.; Viljanen, M.K.; Arvilommi, H.; Mertsola, J. Acellular vaccines containing reduced quantities of pertussis antigens as a booster in adolescents. Pediatrics 1999, 104, e70. [Google Scholar] [CrossRef]
- Blanchard Rohner, G.; Chatzis, O.; Chinwangso, P.; Rohr, M.; Grillet, S.; Salomon, C.; Lemaître, B.; Boonrak, P.; Lawpoolsri, S.; Clutterbuck, E.; et al. Boosting Teenagers with Acellular Pertussis Vaccines Containing Recombinant or Chemically Inactivated Pertussis Toxin: A Randomized Clinical Trial. Clin. Infect. Dis. 2019, 68, 1213–1222. [Google Scholar] [CrossRef]
- Locht, C.; Mielcarek, N. New pertussis vaccination approaches: En route to protect newborns? FEMS Immunol. Med. Microbiol. 2012, 66, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Van der Lee, S.; van Rooijen, D.M.; de Zeeuw-Brouwer, M.L.; Bogaard, M.J.M.; van Gageldonk, P.G.M.; Marinovic, A.B.; Sanders, E.A.M.; Berbers, G.A.M.; Buisman, A.-M. Robust Humoral and Cellular Immune Responses to Pertussis in Adults after a First Acellular Booster Vaccination. Front. Immunol. 2018, 9, 681–690. [Google Scholar] [CrossRef]
- Hovingh, E.S.; Kuipers, B.; Bonacic Marinovic, A.A.; Jan Hamstra, H.; Hijdra, D.; Mughini Gras, L.; Van Twillert, I.; Jongerius, I.; Van Els, C.A.C.M.; Pinelli, E. Detection of opsonizing antibodies directed against a recently circulating Bordetella pertussis strain in paired plasma samples from symptomatic and recovered pertussis patients. Sci. Rep. 2018, 8, 12039. [Google Scholar] [CrossRef]
- Hewlett, E.L.; Sauer, K.T.; Myers, G.A.; Cowell, J.L.; Guerrant, R.L. Induction of a novel morphological response in Chinese hamster ovary cells by pertussis toxin. Infect. Immun. 1983, 40, 1198–1203. [Google Scholar] [CrossRef] [PubMed]
- Gillenius, P.; Jäätmaa, E.; Askelöf, P.; Granström, M.; Tiru, M. The standardization of an assay for pertussis toxin and antitoxin in microplate culture of Chinese hamster ovary cells. J. Biol Stand. 1985, 13, 61–66. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Chen, Z.; Liu, X.; Peng, X.; He, Q. Determination of serum neutralizing antibodies reveals important difference in quality of antibodies against pertussis toxin in children after infection. Vaccine 2021, 39, 1826–1830. [Google Scholar] [CrossRef]
- Barkoff, A.M.; Grondahl-Yli-Hannuksela, K.; Vuononvirta, J.; Mertsola, J.; Kallonen, T.; He, Q. Differences in avidity of IgG antibodies to pertussis toxin after acellular pertussis booster vaccination and natural infection. Vaccine 2012, 30, 6897–6902. [Google Scholar] [CrossRef]
- Knuutila, A.; Dalby, T.; Barkoff, A.M.; Jorgensen, C.S.; Fuursted, K.; Mertsola, J.; Markey, K.; He, Q. Differences in epitope-specific antibodies to pertussis toxin after infection and acellular vaccinations. Clin. Transl. Immunol. 2020, 9, e1161. [Google Scholar] [CrossRef]
- Van Twillert, I.; Han, W.G.; van Els, C.A. Waning and aging of cellular immunity to Bordetella pertussis. Pathog. Dis. 2015, 73, ftv071. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Siegrist, C.A.; Aspinall, R. B-cell responses to vaccination at the extremes of age. Nat. Rev. Immunol. 2009, 9, 185–194. [Google Scholar] [CrossRef]
- Edara, V.V.; Hudson, W.H.; Xie, X.; Ahmed, R.; Suthar, M.S. Neutralizing Antibodies Against SARS-CoV-2 Variants After Infection and Vaccination. JAMA 2021, 325, 1896–1898. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.; Zhang, A.J.; Hung, I.F.; Xu, T.; Ip, W.C.; Wong, R.T.; Ng, J.C.K.; Chan, J.F.W.; Chan, K.-H.; Yuen, K.-Y. High titer and avidity of nonneutralizing antibodies against influenza vaccine antigen are associated with severe influenza. Clin. Vaccine Immunol. 2012, 19, 1012–1018. [Google Scholar] [CrossRef]
- Jalkanen, P.; Pasternack, A.; Maljanen, S.; Melén, K.; Kolehmainen, P.; Huttunen, M.; Lundberg, R.; Tripathi, L.; Kna, H.; Ritvos, M.A.; et al. A combination of N and S antigens with IgA and IgG measurement strengthens the accuracy of SARS-CoV-2 serodiagnostics. J. Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Palmer, S.R.; Balfour, A.H.; Jephcott, A.E. Immunisation of adults during an outbreak of diphtheria. Br. Med. J. Clin. Res. Ed. 1983, 286, 624–626. [Google Scholar] [CrossRef]
- Lagergard, T.; Trollfors, B.; Claesson, B.A.; Karlberg, J.; Taranger, J. Determination of neutralizing antibodies and specific immunoglobulin isotype levels in infants after vaccination against diphtheria. Eur. J. Clin. Microbiol. Infect. Dis. 1992, 11, 341–345. [Google Scholar] [CrossRef]
- Storsaeter, J.; Olin, P. Relative efficacy of two acellular pertussis vaccines during three years of passive surveillance. Vaccine 1992, 10, 142–144. [Google Scholar] [CrossRef]
- Granström, M.; Granström, G. Serological correlates in whooping cough. Vaccine 1993, 11, 445–448. [Google Scholar] [CrossRef]
- Markey, K.; Asokanathan, C.; Feavers, I. Assays for Determining Pertussis Toxin Activity in Acellular Pertussis Vaccines. Toxins 2019, 11, 417. [Google Scholar] [CrossRef]
- Isbrucker, R.; Daas, A.; Wagner, L.; Costanzo, A. Transferability study of CHO cell clustering assays for monitoring of pertussis toxin activity in acellular pertussis vaccines. Pharmeur. Bio Sci. Notes 2016, 2015, 97–114. [Google Scholar]
- Markey, K.; Douglas-Bardsley, A.; Hockley, J.; Le Tallec, D.; Costanzo, A. Calibration of pertussis toxin BRP batch 1 in a standardised CHO cell-based clustering assay. Pharmeur. Bio Sci. Notes 2018, 2018, 112–123. [Google Scholar]
- Sato, H.; Sato, Y.; Ito, A.; Ohishi, I. Effect of monoclonal antibody to pertussis toxin on toxin activity. Infect. Immun. 1987, 55, 909–915. [Google Scholar] [CrossRef]
- Granstrom, M.; Granstrom, G.; Gillenius, P.; Askelof, P. Neutralizing antibodies to pertussis toxin in whooping cough. J. Infect. Dis. 1985, 151, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Trollfors, B.; Krantz, I.; Sigurs, N.; Taranger, J.; Zackrisson, G.; Roberson, R. Toxin-neutralizing antibodies in patients with pertussis, as determined by an assay using Chinese hamster ovary cells. J. Infect. Dis. 1988, 158, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, R.M.; Claesson, B.A.; Selstam, U.; Fagerlund, E.; Granstrom, M.; Blondeau, C.; Hoffenbach, A. Safety and immunogenicity of a combined diphtheria-tetanus-acellular pertussis-inactivated polio vaccine-Haemophilus influenzae type b vaccine administered at 2–4-6–13 or 3–5-12 months of age. Pediatr. Infect. Dis. J. 1998, 17, 1026–1033. [Google Scholar] [CrossRef]
- Carlsson, R.M.; Claesson, B.A.; Fagerlund, E.; Knutsson, N.; Lundin, C. Antibody persistence in five-year-old children who received a pentavalent combination vaccine in infancy. Pediatr. Infect. Dis. J. 2002, 21, 535–541. [Google Scholar] [CrossRef]
- Sirivichayakul, C.; Chanthavanich, P.; Limkittikul, K.; Siegrist, C.A.; Wijagkanalan, W.; Chinwangso, P.; Petre, J.; Thai, P.H.; Chauhan, M.; Viviani, S. Safety and immunogenicity of a combined Tetanus, Diphtheria, recombinant acellular Pertussis vaccine (TdaP) in healthy Thai adults. Hum. Vaccin. Immunother. 2017, 13, 136–143. [Google Scholar] [CrossRef]
- Pichichero, M.E.; Francis, A.B.; Marsocci, S.M.; Green, J.L.; Disney, F.A.; Meschievitz, C. Safety and immunogenicity of an acellular pertussis vaccine booster in 15- to 20-month-old children previously immunized with acellular or whole-cell pertussis vaccine as infants. Pediatrics 1993, 91, 756–760. [Google Scholar]
- Pichichero, M.E.; Deloria, M.A.; Rennels, M.B.; Anderson, E.L.; Edwards, K.M.; Decker, M.D.; Englund, J.A.; Steinhoff, M.C.; Deforest, A.; Meade, B.D. A safety and immunogenicity comparison of 12 acellular pertussis vaccines and one whole-cell pertussis vaccine given as a fourth dose in 15- to 20-month-old children. Pediatrics 1997, 100, 772–788. [Google Scholar] [CrossRef]
- Giuliano, M.; Mastrantonio, P.; Giammanco, A.; Piscitelli, A.; Salmaso, S.; Wassilak, S.G. Antibody responses and persistence in the two years after immunization with two acellular vaccines and one whole-cell vaccine against pertussis. J. Pediatr. 1998, 132, 983–988. [Google Scholar] [CrossRef]
- Rutter, D.A.; Ashworth, L.A.; Day, A.; Funnell, S.; Lovell, F.; Robinson, A. Trial of a new acellular pertussis vaccine in healthy adult volunteers. Vaccine 1988, 6, 29–32. [Google Scholar] [CrossRef]
- Greco, D.; Salmaso, S.; Mastrantonio, P.; Giuliano, M.; Tozzi, A.E.; Anemona, A.; Ciofi degli Atti, M.L.; Giammanco, A.; Panei, P.; Blackwelder, W.C.; et al. A controlled trial of two acellular vaccines and one whole-cell vaccine against pertussis. Progetto Pertosse Working Group. N. Engl. J. Med. 1996, 334, 341–348. [Google Scholar] [CrossRef]
- Meade, B.D.; Lynn, F.; Reed, G.F.; Mink, C.M.; Romani, T.A.; Deforest, A.; Deloria, M.A. Relationships between functional assays and enzyme immunoassays as measurements of responses to acellular and whole-cell pertussis vaccines. Pediatrics 1995, 96, 595–600. [Google Scholar]
- Millen, S.H.; Bernstein, D.I.; Connelly, B.; Ward, J.I.; Chang, S.J.; Weiss, A.A. Antibody-mediated neutralization of pertussis toxin-induced mitogenicity of human peripheral blood mononuclear cells. Infect. Immun. 2004, 72, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Isacson, J.; Trollfors, B.; Lagergrd, T.; Taranger, J. Comparison of a toxin neutralization assay and ELISA for determination of pertussis toxin antibodies. Serodiagn. Immunother. Infect. Dis. 1997, 8, 163–167. [Google Scholar] [CrossRef]
- Sato, Y.; Sato, H.; Kodama, H.; Uchimura, M.; Miwa, N.; Kobayashi, T.; Yamamoto, E.; Fujita, I.; Kumamoto, T. An improved ELISA system for the measurement of IgG antibodies against pertussis toxin (PT) and filamentous hemagglutinin (FHA) in human sera. Dev. Biol. Stand. 1991, 73, 167–174. [Google Scholar]
- Dalby, T.; Sørensen, C.; Petersen, J.W.; Krogfelt, K.A. Pertussis serology: Assessment of IgG anti-PT ELISA for replacement of the CHO cell assay. APMIS 2010, 118, 968–972. [Google Scholar] [CrossRef]
- Moriuchi, T.; Otsuka, N.; Hiramatsu, Y.; Shibayama, K.; Kamachi, K. A high seroprevalence of antibodies to pertussis toxin among Japanese adults: Qualitative and quantitative analyses. PLoS ONE 2017, 12, e0181181. [Google Scholar] [CrossRef]
- Weiss, A.A.; Mobberley, P.S.; Fernandez, R.C.; Mink, C.M. Characterization of human bactericidal antibodies to Bordetella pertussis. Infect. Immun. 1999, 67, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Aase, A.; Herstad, T.K.; Merino, S.; Brandsdal, K.T.; Berdal, B.P.; Aleksandersen, E.M.; Aaberge, I.S. Opsonophagocytic activity and other serological indications of Bordetella pertussis infection in military recruits in Norway. Clin. Vaccine Immunol. 2007, 14, 855–862. [Google Scholar] [CrossRef]
- Jahnmatz, M.; Kesa, G.; Netterlid, E.; Buisman, A.M.; Thorstensson, R.; Ahlborg, N. Optimization of a human IgG B-cell ELISpot assay for the analysis of vaccine-induced B-cell responses. J. Immunol. Methods. 2013, 391, 50–59. [Google Scholar] [CrossRef]
- Marcellini, V.; Piano Mortari, E.; Fedele, G.; Gesualdo, F.; Pandolfi, E.; Midulla, F.; Leone, P.; Stefanelli, P.; Tozzi, A.E.; Carsetti, R.; et al. Protection against Pertussis in Humans Correlates to Elevated Serum Antibodies and Memory B Cells. Front. Immunol. 2017, 8, 1158. [Google Scholar] [CrossRef]
- Aloulou, M.; Fazilleau, N. Regulation of B cell responses by distinct populations of CD4 T cells. Biomed. J. 2019, 42, 243–251. [Google Scholar] [CrossRef]
- Van Twillert, I.; van Gaans-van den Brink, J.A.M.; Poelen, M.C.M.; Helm, K.; Kuipers, B.; Schipper, M.; Boog, C.J.P.; Verheij, T.J.M.; Versteegh, F.G.A.; van Els, C.A.C.M. Age related differences in dynamics of specific memory B cell populations after clinical pertussis infection. PLoS ONE 2014, 9, e85227. [Google Scholar] [CrossRef]
- Buisman, A.M.; de Rond, C.G.; Oztürk, K.; Ten Hulscher, H.I.; van Binnendijk, R.S. Long-term presence of memory B-cells specific for different vaccine components. Vaccine 2009, 28, 179–186. [Google Scholar] [CrossRef]
- Hendrikx, L.H.; Oztürk, K.; de Rond, L.G.; Veenhoven, R.H.; Sanders, E.A.; Berbers, G.A.; Buisman, A.-M. Identifying long-term memory B-cells in vaccinated children despite waning antibody levels specific for Bordetella pertussis proteins. Vaccine 2011, 29, 1431–1437. [Google Scholar] [CrossRef]
- Hendrikx, L.H.; Felderhof, M.K.; Ozturk, K.; de Rond, L.G.; van Houten, M.A.; Sanders, E.A.; Berbers, G.A.M.; Busiman, A.-M. Enhanced memory B-cell immune responses after a second acellular pertussis booster vaccination in children 9 years of age. Vaccine 2011, 30, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Warfel, J.M.; Edwards, K.M. Pertussis vaccines and the challenge of inducing durable immunity. Curr. Opin. Immunol. 2015, 35, 48–54. [Google Scholar] [CrossRef]
- Giammanco, A.; Taormina, S.; Chiarini, A.; Dardanoni, G.; Stefanelli, P.; Salmaso, S.; Mastrantonio, P. Analogous IgG subclass response to pertussis toxin in vaccinated children, healthy or affected by whooping cough. Vaccine 2003, 21, 1924–1931. [Google Scholar] [CrossRef]
- Diavatopoulos, D.A.; Edwards, K.M. What Is Wrong with Pertussis Vaccine Immunity? Why Immunological Memory to Pertussis Is Failing. Cold Spring Harb. Perspect. Biol. 2017, 9, a029553. [Google Scholar] [CrossRef]
- Jahnmatz, M.; Amu, S.; Ljungman, M.; Wehlin, L.; Chiodi, F.; Mielcarek, N.; Locht, C.; Thorstensson, R. B-cell responses after intranasal vaccination with the novel attenuated Bordetella pertussis vaccine strain BPZE1 in a randomized phase I clinical trial. Vaccine 2014, 32, 3350–3356. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Apostolovic, D.; Jahnmatz, M.; Liang, F.; Ols, S.; Tecleab, T.; Wu, C.; van Hage, M.; Solovay, K.; Rubin, K.; et al. Live attenuated pertussis vaccine BPZE1 induces a broad antibody response in humans. J. Clin. Invest. 2020, 130, 2332–2346. [Google Scholar] [CrossRef]
- Solans, L.; Locht, C. The Role of Mucosal Immunity in Pertussis. Front. Immunol. 2019, 9, 3068. [Google Scholar] [CrossRef] [PubMed]
- Denoel, P.A.; Goldblatt, D.; de Vleeschauwer, I.; Jacquet, J.M.; Pichichero, M.E.; Poolman, J.T. Quality of the Haemophilus influenzae type b (Hib) antibody response induced by diphtheria-tetanus-acellular pertussis/Hib combination vaccines. Clin. Vaccine Immunol. 2007, 14, 1362–1369. [Google Scholar] [CrossRef]
- Ahmed, R.; Gray, D. Immunological memory and protective immunity: Understanding their relation. Science 1996, 272, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Goldblatt, D.; Vaz, A.R.; Miller, E. Antibody avidity as a surrogate marker of successful priming by Haemophilus influenzae type b conjugate vaccines following infant immunization. J. Infect. Dis. 1998, 177, 1112–1115. [Google Scholar] [CrossRef]
- Goodnow, C.C.; Vinuesa, C.G.; Randall, K.L.; Mackay, F.; Brink, R. Control systems and decision making for antibody production. Nat. Immunol. 2010, 11, 681–688. [Google Scholar] [CrossRef]
- Almanzar, G.; Ottensmeier, B.; Liese, J.; Prelog, M. Assessment of IgG avidity against pertussis toxin and filamentous hemagglutinin via an adapted enzyme-linked immunosorbent assay (ELISA) using ammonium thiocyanate. J. Immunol Methods. 2013, 387, 36–42. [Google Scholar] [CrossRef]
- Liesenfeld, O.; Montoya, J.G.; Kinney, S.; Press, C.; Remington, J.S. Effect of testing for IgG avidity in the diagnosis of Toxoplasma gondii infection in pregnant women: Experience in a US reference laboratory. J. Infect. Dis. 2001, 183, 1248–1253. [Google Scholar] [CrossRef]
- McHeyzer-Williams, M.G.; McLean, M.J.; Lalor, P.A.; Nossal, G.J. Antigen-driven B cell differentiation in vivo. J. Exp. Med. 1993, 178, 295–307. [Google Scholar] [CrossRef]
- Schallert, N.; Pihlgren, M.; Kovarik, J.; Roduit, C.; Tougne, C.; Bozzotti, P.; Giudice, G.D.; Siegrist, C.-A.; Lambert, P.-H. Generation of adult-like antibody avidity profiles after early-life immunization with protein vaccines. Eur. J. Immunol. 2002, 32, 752–760. [Google Scholar] [CrossRef]
- Berek, C.; Berger, A.; Apel, M. Maturation of the immune response in germinal centers. Cell 1991, 67, 1121–1129. [Google Scholar] [CrossRef]
- Kepler, T.B.; Perelson, A.S. Somatic hypermutation in B cells: An optimal control treatment. J. Theor. Biol. 1993, 164, 37–64. [Google Scholar] [CrossRef]
- French, D.L.; Laskov, R.; Scharff, M.D. The role of somatic hypermutation in the generation of antibody diversity. Science 1989, 244, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Eisen, H.N. Determination of Antibody Affinity for Haptens and Antigens by Means of Fluorescence Quenching. Methods Med. Res. 1964, 10, 115–121. [Google Scholar]
- Dorner, T.; Radbruch, A. Antibodies and B cell memory in viral immunity. Immunity 2007, 27, 384–392. [Google Scholar] [CrossRef]
- Prelog, M.; Almanzar, G.; Rieber, N.; Ottensmeier, B.; Zlamy, M.; Liese, J. Differences of IgG antibody avidity after an acellular pertussis (aP) booster in adolescents after a whole cell (wcP) or aP primary vaccination. Vaccine 2013, 31, 387–393. [Google Scholar] [CrossRef]
- Dimitrov, J.D.; Lacroix-Desmazes, S.; Kaveri, S.V. Important parameters for evaluation of antibody avidity by immunosorbent assay. Anal. Biochem. 2011, 418, 149–151. [Google Scholar] [CrossRef]
- Pullen, G.R.; Fitzgerald, M.G.; Hosking, C.S. Antibody avidity determination by ELISA using thiocyanate elution. J. Immunol Methods. 1986, 86, 83–87. [Google Scholar] [CrossRef]
- Cabore, R.N.; Maertens, K.; Dobly, A.; Leuridan, E.; Van Damme, P.; Huygen, K. Influence of maternal vaccination against diphtheria, tetanus, and pertussis on the avidity of infant antibody responses to a pertussis containing vaccine in Belgium. Virulence 2017, 8, 1245–1254. [Google Scholar] [CrossRef]
- Abu Raya, B.; Bamberger, E.; Almog, M.; Peri, R.; Srugo, I.; Kessel, A. Immunization of pregnant women against pertussis: The effect of timing on antibody avidity. Vaccine 2015, 33, 1948–1952. [Google Scholar] [CrossRef]
- Fumimoto, R.; Otsuka, N.; Sunagawa, T.; Tanaka-Taya, K.; Kamiya, H.; Kamachi, K. Age-related differences in antibody avidities to pertussis toxin and filamentous hemagglutinin in a healthy Japanese population. Vaccine 2019, 37, 2463–2469. [Google Scholar] [CrossRef]
- Hendrikx, L.H.; Berbers, G.A.; Veenhoven, R.H.; Sanders, E.A.; Buisman, A.M. IgG responses after booster vaccination with different pertussis vaccines in Dutch children 4 years of age: Effect of vaccine antigen content. Vaccine 2009, 27, 6530–6536. [Google Scholar] [CrossRef]
- Ibrahim, N.M.; El-Kady, E.M.; Eissa, S.A.; Wahby, A.F. Assessment of antibody level and avidity against Bordetella pertussis in a cohort of Egyptian individuals aged 1–18 years. J. Adv. Res. 2016, 7, 105–111. [Google Scholar] [CrossRef]
- Maertens, K.; Hoang, T.H.T.; Cabore, R.N.; Leuridan, E. Avidity of maternal pertussis antibodies after vaccination during pregnancy. Vaccine 2015, 33, 5489. [Google Scholar] [CrossRef]
- Mosley, Y.C.; Radder, J.E.; Berndt, A.; HogenEsch, H. Genome-Wide Association Mapping of the Antibody Response to Diphtheria, Tetanus and Acellular Pertussis Vaccine in Mice. J. Infect. Dis. 2017, 215, 466–474. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abu-Raya, B.; Giles, M.L.; Kollmann, T.R.; Sadarangani, M. Profiling avidity of antibodies elicited by vaccination using enzyme-linked immunosorbent assay-based elution—Insights into a novel experimental and analytical approach. Vaccine 2020, 38, 5389–5392. [Google Scholar] [CrossRef] [PubMed]
- Abu-Raya, B.; Giles, M.L.; Kollmann, T.R.; Sadarangani, M. The Effect of Timing of Tetanus-Diphtheria-Acellular Pertussis Vaccine Administration in Pregnancy on the Avidity of Pertussis Antibodies. Front. Immunol. 2019, 10, 2423. [Google Scholar] [CrossRef]
- Cherry, J.D. Pertussis: Challenges today and for the future. PLoS Pathog. 2013, 9, e1003418. [Google Scholar] [CrossRef]
- Ibsen, P.H. The effect of formaldehyde, hydrogen peroxide and genetic detoxification of pertussis toxin on epitope recognition by murine monoclonal antibodies. Vaccine 1996, 14, 359–368. [Google Scholar] [CrossRef]
- Nencioni, L.; Volpini, G.; Peppoloni, S.; Bugnoli, M.; De Magistris, T.; Marsili, I.; Rappuoli, R. Properties of pertussis toxin mutant PT-9K/129G after formaldehyde treatment. Infect. Immun. 1991, 59, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.; Kim, B.G.; Nam, K.T.; Hong, S.H.; Ahn, D.H.; Choi, G.S.; Kim, H.; Hong, J.-T.; Ahn, B.-Y. Characterization of the carbohydrate binding and ADP-ribosyltransferase activities of chemically detoxified pertussis toxins. Vaccine 2013, 31, 2988–2993. [Google Scholar] [CrossRef]
- Yuen, C.T.; Asokanathan, C.; Cook, S.; Lin, N.; Xing, D. Effect of different detoxification procedures on the residual pertussis toxin activities in vaccines. Vaccine 2016, 34, 2129–2134. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.N.; Chang, C.; Yoder, S.M.; Rock, M.T.; Maynard, J.A. Antibodies recognizing protective pertussis toxin epitopes are preferentially elicited by natural infection versus acellular immunization. Clin. Vaccine Immunol. 2011, 18, 954–962. [Google Scholar] [CrossRef]
- Sato, H.; Sato, Y.; Ohishi, I. Comparison of pertussis toxin (PT)-neutralizing activities and mouse-protective activities of anti-PT mouse monoclonal antibodies. Infect. Immun. 1991, 59, 3832–3835. [Google Scholar] [CrossRef]
- Sato, H.; Sato, Y. Protective activities in mice of monoclonal antibodies against pertussis toxin. Infect. Immun. 1990, 58, 3369–3374. [Google Scholar] [CrossRef]
- Nguyen, A.W.; Wagner, E.K.; Laber, J.R.; Goodfield, L.L.; Smallridge, W.E.; Harvill, E.T.; Papin, J.F.; Wolf, R.F.; Padlan, E.A.; Bristol, A.; et al. A cocktail of humanized anti-pertussis toxin antibodies limits disease in murine and baboon models of whooping cough. Sci. Transl. Med. 2015, 7, 316ra195. [Google Scholar] [CrossRef]
- Wagner, E.K.; Wang, X.; Bui, A.; Maynard, J.A. Synergistic Neutralization of Pertussis Toxin by a Bispecific Antibody In Vitro and In Vivo. Clin. Vaccine Immunol. 2016, 23, 851–862. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.W.; DiVenere, A.M.; Papin, J.F.; Connelly, S.; Kaleko, M.; Maynard, J.A. Neutralization of pertussis toxin by a single antibody prevents clinical pertussis in neonatal baboons. Sci. Adv. 2020, 6, eaay9258. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.N.; Maynard, J.A. Characterization of a key neutralizing epitope on pertussis toxin recognized by monoclonal antibody 1B7. Biochemistry 2009, 48, 11982–11993. [Google Scholar] [CrossRef]
- Kowalsky, C.A.; Faber, M.S.; Nath, A.; Dann, H.E.; Kelly, V.W.; Liu, L.; Shanker, P.; Wagner, E.K.; Maynard, J.A.; Chan, C.; et al. Rapid fine conformational epitope mapping using comprehensive mutagenesis and deep sequencing. J. Biol. Chem. 2015, 290, 26457–26470. [Google Scholar] [CrossRef]
- Acquaye-Seedah, E.; Huang, Y.; Sutherland, J.N.; DiVenere, A.M.; Maynard, J.A. Humanised monoclonal antibodies neutralise pertussis toxin by receptor blockade and reduced retrograde trafficking. Cell Microbiol. 2018, 20, e12948. [Google Scholar] [CrossRef]
- Acquaye-Seedah, E.; Reczek, E.E.; Russell, H.H.; DiVenere, A.M.; Sandman, S.O.; Collins, J.H.; Stein, C.A.; Whitehead, T.A.; Maynard, J.A. Characterization of Individual Human Antibodies That Bind Pertussis Toxin Stimulated by Acellular Immunization. Infect. Immun. 2018, 86, 4. [Google Scholar] [CrossRef]
- Vaughan, K.; Seymour, E.; Peters, B.; Sette, A. Substantial gaps in knowledge of Bordetella pertussis antibody and T cell Cross Mark epitopes relevant for natural immunity and vaccine efficacy. Hum. Immunol. 2014, 75, 440–451. [Google Scholar] [CrossRef][Green Version]
- Seubert, A.; D’Oro, U.; Scarselli, M.; Pizza, M. Genetically detoxified pertussis toxin (PT-9K/129G): Implications for immunization and vaccines. Expert Rev. Vaccines 2014, 13, 1191–1204. [Google Scholar] [CrossRef]
- Surendran, N.; Pichichero, M. Genetically detoxified pertussis toxin induces superior antigen specific CD4 T cell responses compared to chemically detoxified pertussis toxin. Hum. Vaccine Immunother. 2019, 15, 1167–1170. [Google Scholar] [CrossRef]
- Pitisuttithum, P.; Chokephaibulkit, K.; Sirivichayakul, C.; Sricharoenchai, S.; Dhitavat, J.; Pitisuthitham, A.; Phingsamart, W.; Boonnak, K.; Lapphra, K.; Sabmee, Y.; et al. Antibody persistence after vaccination of adolescents with monovalent and combined acellular pertussis vaccines containing genetically inactivated pertussis toxin: A phase 2/3 randomised, controlled, non-inferiority trial. Lancet Infect. Dis. 2018, 18, 1260–1268. [Google Scholar] [CrossRef]
- Pizza, M.; Covacci, A.; Bartoloni, A.; Perugini, M.; Nencioni, L.; De Magistris, M.T.; Villa, L.; Nucci, D.; Manetti, R.; Bugnoli, M.; et al. Mutants of pertussis toxin suitable for vaccine development. Science 1989, 246, 497–500. [Google Scholar] [CrossRef]
- Davies, V.; Vaughan, K.; Damle, R.; Peters, B.; Sette, A. Classification of the universe of immune epitope literature: Representation and knowledge gaps. PLoS ONE 2009, 4, e6948. [Google Scholar] [CrossRef]
- Van Gent, M.; Bart, M.J.; van der Heide, H.G.; Heuvelman, K.J.; Mooi, F.R. Small mutations in Bordetella pertussis are associated with selective sweeps. PLoS ONE 2012, 7, e46407. [Google Scholar] [CrossRef]
- Octavia, S.; Maharjan, R.P.; Sintchenko, V.; Stevenson, G.; Reeves, P.R.; Gilbert, G.L.; Lan, R. Insight into evolution of Bordetella pertussis from comparative genomic analysis: Evidence of vaccine-driven selection. Mol. Biol. Evol. 2011, 2, 707–715. [Google Scholar] [CrossRef]
- Maharjan, R.P.; Gu, C.; Reeves, P.R.; Sintchenko, V.; Gilbert, G.L.; Lan, R. Genome-wide analysis of single nucleotide polymorphisms in Bordetella pertussis using comparative genomic sequencing. Res. Microbiol. 2008, 159, 602–608. [Google Scholar] [CrossRef]
- Bart, M.J.; van Gent, M.; van der Heide, H.G.; Boekhorst, J.; Hermans, P.; Parkhill, J.; Mooi, F.R. Comparative genomics of prevaccination and modern Bordetella pertussis strains. BMC Genom. 2010, 11, 627. [Google Scholar] [CrossRef] [PubMed]
- Jahnmatz, M.; Richert, L.; Al-Tawil, N.; Storsaeter, J.; Colin, C.; Bauduin, C.; Thalen, M.; Solovay, K.; Rubin, K.; Melcarek, N.; et al. Safety and immunogenicity of the live attenuated intranasal pertussis vaccine BPZE1: A phase 1b, double-blind, randomised, placebo-controlled dose-escalation study. Lancet Infect. Dis. 2020, 20, 1290–1301. [Google Scholar] [CrossRef]
- Raeven, R.H.M.; Rockx-Brouwer, D.; Kanojia, G.; van der Maas, L.; Bindels, T.H.E.; ten Have, R.; van Riet, E.; Metz, B.; Kersten, G.F.A. Intranasal immunization with outer membrane vesicle pertussis vaccine confers broad protection through mucosal IgA and Th17 responses. Sci. Rep. 2020, 10, 7396. [Google Scholar] [CrossRef] [PubMed]
- Diavatopoulos, D.A.; Mills, K.H.; Kester, K.E.; Kampmann, B.; Silerova, M.; Heininger, U.; van Dongen, J.J.M.; van der Most, R.; Huijinen, M.A.; Siena, E.; et al. PERISCOPE: Road towards effective control of pertussis. Lancet Infect. Dis. 2019, 19, e179–e186. [Google Scholar] [CrossRef]
| Model. | Description | Advantages | Requirements |
|---|---|---|---|
| AI | The absorbance of detergent treated sample/Absorbance of the control sample | Simple | Only duplicate wells to be tested |
| AI thresholds | Avidity is considered strong if it exceeds a certain level, or weak if it does not | Simple to categorize strong and weak binding | Clinically or experientially set relevant thresholds to determine strong and weak binding |
| Fractional avidity | The percentage from overall avidity within a concentration range of detergent | The proportion of strong binding can be more accurately evaluated | Dilution series of detergent |
| IC50 | Antibody or detergent concentration corresponding to 50% avidity | Less reliant on initial antibody or single detergent levels | Dilution series of sample or detergent |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barkoff, A.-M.; Knuutila, A.; Mertsola, J.; He, Q. Evaluation of Anti-PT Antibody Response after Pertussis Vaccination and Infection: The Importance of Both Quantity and Quality. Toxins 2021, 13, 508. https://doi.org/10.3390/toxins13080508
Barkoff A-M, Knuutila A, Mertsola J, He Q. Evaluation of Anti-PT Antibody Response after Pertussis Vaccination and Infection: The Importance of Both Quantity and Quality. Toxins. 2021; 13(8):508. https://doi.org/10.3390/toxins13080508
Chicago/Turabian StyleBarkoff, Alex-Mikael, Aapo Knuutila, Jussi Mertsola, and Qiushui He. 2021. "Evaluation of Anti-PT Antibody Response after Pertussis Vaccination and Infection: The Importance of Both Quantity and Quality" Toxins 13, no. 8: 508. https://doi.org/10.3390/toxins13080508
APA StyleBarkoff, A.-M., Knuutila, A., Mertsola, J., & He, Q. (2021). Evaluation of Anti-PT Antibody Response after Pertussis Vaccination and Infection: The Importance of Both Quantity and Quality. Toxins, 13(8), 508. https://doi.org/10.3390/toxins13080508






