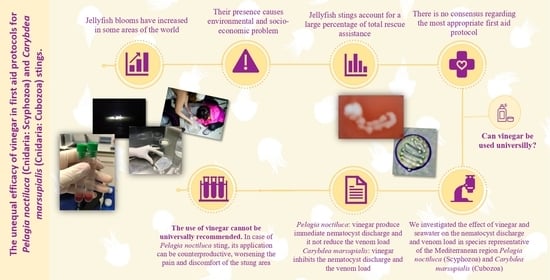
Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings—Implications for First Aid Protocols
Abstract
:1. Introduction
2. Results
2.1. Pelagia noctiluca
2.1.1. Test 1. Rinse Solution Screening
2.1.2. Test 2. Evaluation of the Inhibitory Effect
2.2. Carybdea marsupialis
2.2.1. Test 1. Rinse Solution Screening
2.2.2. Test 2. Evaluation of the Inhibitory Effect
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Collection and Preparation of Jellyfish
5.2. Rinse Solutions and pH Measurements
5.3. Test 1. Nematocyst Discharge—Rinse Solution Screening
- 0: no discharge was observed;
- +: low discharge of nematocysts;
- ++: medium discharge of nematocysts;
- +++: high discharge of nematocysts.
- Activator solution: nematocysts were activated after incubation in the solution;
- Neutral solution: nematocysts were not activated after incubation in the solution.
5.4. Test 2—Evaluation of the Inhibitory Effect
5.4.1. Chemical Stimulation of Nematocyst Discharge
- Neutral solution: nematocysts were not activated after incubation in the first solution but were activated by the subsequent chemical stimulation with ethanol solution;
- Inhibitor solution: nematocysts were not activated after incubation in the first solution or by the subsequent chemical stimulation with ethanol solution.
5.4.2. Natural Stimulation of Nematocyst Discharge
- Neutral solution: tentacle was incubated in microwells for 5 min to carry out the stinging process after incubation.
- Activator solution: solution was applied in spray format on the top of the pig intestine to avoid nematocyst discharge into the microwells during the incubation period.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TSA | Tentacle Solution Assay |
| TSBAA | Tentacle Skin Blood Agarose Assay |
| PBS | Phosphate Buffered Saline |
| SRBCs | Sheep Red Blood Cells |
References
- Daly, M.; Brugler, M.R.; Cartwright, P.; Collins, A.G.; Dawson, M.N.; France, S.C.; McFadden, C.S.; Opresko, D.M.; Rodriguez, E.; Romano, S.; et al. The phylum Cnidaria: A review of phylogenetic patterns and diversity 300 years after Linnaeus. In Linnaeus Tercentenary: Progress in Invertebrate Taxonomy; Zhang, Z.-Q.S., Shear, W.A., Eds.; Magnolia Press: Auckland, New Zealand, 1668; pp. 127–182. [Google Scholar]
- Zhang, Z.-Q. Animal biodiversity: An update of classification and diversity in 2013. Zootaxa 2013, 3703, 5–11. [Google Scholar] [CrossRef] [Green Version]
- Mariscal, R.N. Nematocysts. In Coelenterate Biology; Muscatine, L., Lenhoff, H.M., Eds.; Academic Press: New York, NY, USA, 1974; pp. 129–178. [Google Scholar]
- Beckmann, A.; Özbek, S. The nematocyst: A molecular map of the cnidarian stinging organelle. Int. J. Dev. Biol. 2012, 56, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Watson, G.M.; Wood, R.L. Colloquium on terminology. In The Biology of Nematocysts; Hessinger, D.A., Lenhoff, H.M., Eds.; Academic Press: San Diego, CA, USA, 1988; pp. 21–23. [Google Scholar]
- Östman, C. A guideline to nematocyst nomenclature and classification, and some notes on the systematic value of nematocysts. Sci. Mar. 2000, 64, 31–46. [Google Scholar] [CrossRef]
- Nüchter, T.; Benoit, M.; Engel, U.; Özbek, S.; Holstein, T.W. Nanosecond-scale kinetics of nematocyst discharge. Curr. Biol. 2006, 16, R316–R318. [Google Scholar] [CrossRef] [Green Version]
- Brotz, L.; Cheung, W.W.; Kleisner, K.; Pakhomov, E.; Pauly, D. Increasing jellyfish populations: Trends in large marine ecosystems. In Jellyfish Blooms IV; Purcell, J., Mianzan, H., Frost, J.R., Eds.; Sprinter: Dordrecht, The Netherlands, 2012; pp. 3–20. [Google Scholar]
- Richardson, A.J.; Bakun, A.; Hays, G.C.; Gibbons, M.J. The jellyfish joyride: Causes, consequences and management responses to a more gelatinous future. Trends Ecol. Evol. 2009, 24, 312–322. [Google Scholar] [CrossRef]
- Boulware, D.R. A randomized, controlled field trial for the prevention of jellyfish stings with a topical sting inhibitor. J. Travel Med. 2006, 13, 166–171. [Google Scholar] [CrossRef] [Green Version]
- Organización Mundial del Turismo (OMT). Panorama del Turismo Internacional; OMT: Madrid, Spain, 2019. [Google Scholar] [CrossRef]
- Ariza, E.; Jiménez, J.A.; Sardá, R. A critical assessment of beach management on the Catalan coast. Ocean Coast. Manag. 2008, 51, 141–160. [Google Scholar] [CrossRef]
- Gershwin, L.A.; De Nardi, M.; Winkel, K.D.; Fenner, P.J. Marine stingers: Review of an under-recognized global coastal management issue. Coast. Manag. 2010, 38, 22–41. [Google Scholar] [CrossRef]
- Graham, W.M.; Gelcich, S.; Robinson, K.L.; Duarte, C.M.; Brotz, L.; Purcell, J.E.; Madin, L.P.; Mianzan, H.; Sutherland, K.R.; Uye, S.; et al. Linking human well-being and jellyfish: Ecosystem services, impacts, and societal responses. Front. Ecol. Environ. 2014, 12, 515–523. [Google Scholar] [CrossRef] [Green Version]
- Purcell, J.E.; Uye, S.; Lo, W. Anthropogenic causes of jellyfish blooms and their direct consequences for humans: A review. Mar. Ecol. Prog. Ser. 2007, 350, 153–174. [Google Scholar] [CrossRef]
- Remigante, A.; Costa, R.; Morabito, R.; La Spada, G.; Marino, A.; Dossena, S. Impact of scyphozoan venoms on human health and current first aid options for stings. Toxins 2018, 10, 133. [Google Scholar] [CrossRef] [Green Version]
- Bordehore, C.; Alonso, C.; Sánchez-Fernández, L.; Canepa, A.; Acevedo, M.; Nogué, S.; Fuentes, V.L. Lifeguard assistance at Spanish Mediterranean beaches: Jellyfish prevail and proposals for improving risk management. Ocean. Coast. Manag. 2016, 131, 45–52. [Google Scholar] [CrossRef] [Green Version]
- Marambio, M.; ICM-CSIC-Institute of Marine Sciences, Barcelona, Spain. Personal communication, 2020.
- Marambio, M.; Canepa, A.; Lòpez, L.; Gauci, A.A.; Gueroun, S.K.M.; Zampardi, S.; Boero, F.; Kéfi-Daly Yahia, O.; Daly Yahia, M.N.; Fuentes, V.; et al. Unfolding Jellyfish Bloom Dynamics along the Mediterranean Basin by Transnational Citizen Science Initiatives. Diversity 2021, 13, 274. [Google Scholar] [CrossRef]
- Mariottini, G.L.; Giacco, E.; Pane, L. The Mauve Stinger Pelagia noctiluca (Forsskål, 1775). Distribution, ecology, toxicity and epidemiology of stings. A review. Mar. Drugs 2008, 6, 496–513. [Google Scholar] [CrossRef]
- Mariottini, G.L.; Pane, L. Mediterranean jellyfish venoms: A review on scyphomedusae. Mar. Drugs 2010, 8, 1122–1152. [Google Scholar] [CrossRef] [Green Version]
- De Donno, A.; Idolo, A.; Bagordo, F.; Grassi, T.; Leomanni, A.; Serio, F.; Guido, M.; Canitano, M.; Zampardi, S.; Boero, F.; et al. Impact of stinging jellyfish proliferations along south Italian coasts: Human health hazards, treatment and social costs. Int. J. Environ. Res. Public Health 2014, 1, 2488–2503. [Google Scholar] [CrossRef]
- Marambio, M.; López, L.; Fuentes, V.L.; Gili, J.M.; Canepa, A. More than 10 years dealing with jellyfish blooms: The jellyfish observation network in the NW Mediterranean. In Proceedings of the 6th International Jellyfish Bloom Symposium, Cape Town, South Africa, 6 November 2019. [Google Scholar]
- Hall, A.H. I Pelagia Noctiluca Jellyfish: Can Lesions and Symptoms be Prevented or Ameliorated? J. Mar. Biol. Aquacult. 2018, 4, 48–52. [Google Scholar] [CrossRef]
- Montgomery, L.; Seys, J.; Mees, J. To pee, or not to pee: A review on envenomation and treatment in European jellyfish species. Mar. Drugs 2016, 14, 127. [Google Scholar] [CrossRef] [Green Version]
- Ballesteros, A.; Östman, C.; Santín, A.; Marambio, M.; Narda, M.; Gili, J.M. Cnidome and morphological features of Pelagia noctiluca (Cnidaria: Scyphozoa) throughout the different life cycle stage. Front. Mar. Sci. 2021, in press. [Google Scholar] [CrossRef]
- Marambio, M.; Ballesteros, A.; López, L.; Fuentes, V.L.; Gili, J.M. Guía de identificación de medusas y otros organismos gelatinosos. Span. Natl. Res. Counc. 2021, 1–44. [Google Scholar] [CrossRef]
- Bordehore, C.; Fuentes, V.; Atienza, D.; Barberá, C.; Fernandez-Jover, D.; Roig, M.; Acevedo-Dudley, M.J.; Canepa, A.J.; Gili, J. Detection of an unusual presence of the cubozoan Carybdea marsupialis at shallow beaches located near Denia, Spain (south-western Mediterranean). Mar. Biodivers. Rec. 2011, 4, E69. [Google Scholar] [CrossRef] [Green Version]
- Milla, L.; Segura-Puertas, L.; Celis, L.; Heimer de la Cotera, E. Dermatitis por contacto con Carybdea marsupialis (cnidaria cubozoa). Dermatol. Rev. Mex. 2000, 44, 167–170. [Google Scholar]
- Peca, G.; Rafanelli, S.; Galassi, G.; Bartolo, P.D.; Bertini, S.; Alberani, M.; Beccari, G. Contact reactions to the jellyfish Carybdea marsupialis: Observation of 40 cases. Contact Dermat. 1997, 36, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Di Camillo, C.; Bo, M.; Puce, S.; Tazioli, S.; Bavestrello, G. The cnidome of Carybdea marsupialis (Cnidaria: Cubomedusae) from the Adriatic Sea. J. Mar. Biol. Assoc. UK 2006, 86, 705–709. [Google Scholar] [CrossRef]
- Killi, N.; Mariottini, G.L. Cnidarian Jellyfish: Ecological aspects, nematocyst isolation, and treatment methods of Sting. In Marine Organisms as Model Systems in Biology and Medicine; Kloc, M., Kubiak, J., Eds.; Springer: Cham, Switzerland, 2018; pp. 477–513. [Google Scholar]
- Cegolon, L.; Heymann, W.C.; Lange, J.H.; Mastrangelo, G. Jellyfish stings and their management: A review. Mar. drugs 2013, 11, 523–550. [Google Scholar] [CrossRef] [Green Version]
- Doyle, T.K.; Headlam, J.L.; Wilcox, C.L.; MacLoughlin, E.; Yanagihara, A.A. Evaluation of Cyanea capillata sting management protocols using ex vivo and in vitro envenomation models. Toxins 2017, 9, 215. [Google Scholar] [CrossRef] [Green Version]
- Birsa, L.M.; Verity, P.G.; Lee, R.F. Evaluation of the effects of various chemicals on discharge of and pain caused by jellyfish nematocysts. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2010, 151, 426–430. [Google Scholar] [CrossRef]
- Pyo, M.J.; Lee, H.; Bae, S.K.; Heo, Y.; Choudhary, I.; Yoon, W.D.; Kang, C.; Kim, E. Modulation of jellyfish nematocyst discharges and management of human skin stings in Nemopilema nomurai and Carybdea mora. Toxicon 2016, 109, 26–32. [Google Scholar] [CrossRef]
- Hartwick, R.; Callanan, V.; Williamson, J. Disarming the box jellyfish: Nematocyst inhibition in Chironex fleckeri. Med. J. Aust. 1980, 1, 5–20. [Google Scholar] [CrossRef]
- Fenner, P.J.; Williamson, J. Experiments with the nematocysts of Carybdea rastoni (“Jimble”). Med. J. Aust. 1987, 147, 258–259. [Google Scholar] [CrossRef]
- Yanagihara, A.A.; Wilcox, C.; King, R.; Hurwitz, K.; Castelfranco, A.M. Experimental assays to assess the efficacy of vinegar and other topical first-aid approaches on cubozoan (Alatina alata) tentacle firing and venom toxicity. Toxins 2016, 8, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yanagihara, A.A.; Wilcox, C.L. Cubozoan sting-site seawater rinse, scraping, and ice can increase venom load: Upending current first aid recommendations. Toxins 2017, 9, 105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fenner, P.J.; Fitzpatrick, P.F. Experiments with the nematocysts of Cyanea capillata. Med. J. Aust. 1986, 145, 174. [Google Scholar] [CrossRef] [PubMed]
- Morabito, R.; Marino, A.; Dossena, S.; La Spada, G. Nematocyst discharge in Pelagia noctiluca (Cnidaria, Scyphozoa) oral arms can be affected by lidocaine, ethanol, ammonia and acetic acid. Toxicon 2014, 83, 52–58. [Google Scholar] [CrossRef]
- Salleo, A.; La Spada, G.; Falzea, G.; Denaro, M.G. Discharging effect of anions and inhibitory effect of divalent cations on isolated nematocysts of Pelagia noctiluca. Mol. Physiol. 1984, 5, 25–33. [Google Scholar]
- Colin, S.P.; Costello, J.H. Functional characteristics of nematocysts found on the scyphomedusa Cyanea capillata. J. Exp. Mar. Biol. Ecol. 2007, 351, 114–120. [Google Scholar] [CrossRef] [Green Version]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. 2017. Available online: https://www.R-project.org/ (accessed on 8 March 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. Available online: https://ggplot2.tidyverse.org/ (accessed on 8 March 2020).


| Pelagia noctiluca | Carybdea marsupialis | ||||||
|---|---|---|---|---|---|---|---|
| Rinse Solution | pH | n | Discharge 1 | Effect 2 | n | Discharge 1 | Effect 2 |
| Seawater (control) | 7.80 | 6 | 0 | Neutral | 3 | 0 | Neutral |
| Vinegar | 2.60 | 6 | ++ | Activator | 3 | 0 | Neutral |
| Pelagia noctiluca | Carybdea marsupialis | |||||
|---|---|---|---|---|---|---|
| Rinse Solution | n | Hemolytic Area (%) | Effect | n | Hemolytic Area (%) | Effect |
| Seawater (control) | 5 | 23.80 ± 6.90 | Not inhibitor | 10 | 32.49 ± 19.33 | Not inhibitor |
| Vinegar | 5 | 21.64 ± 11.98 ns | Not inhibitor | 10 | 0.73 ± 2.31 *** | Inhibitor |
| Carybdea marsupialis | |||
|---|---|---|---|
| Rinse Solution | n | Discharge 1 | Effect 2 |
| Ethanol | 3 | +++ | Activator |
| Seawater (control) + ethanol | 3 | +++ | Neutral |
| Vinegar + ethanol | 3 | 0 | Inhibitor |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballesteros, A.; Marambio, M.; Fuentes, V.; Narda, M.; Santín, A.; Gili, J.-M. Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings—Implications for First Aid Protocols. Toxins 2021, 13, 509. https://doi.org/10.3390/toxins13080509
Ballesteros A, Marambio M, Fuentes V, Narda M, Santín A, Gili J-M. Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings—Implications for First Aid Protocols. Toxins. 2021; 13(8):509. https://doi.org/10.3390/toxins13080509
Chicago/Turabian StyleBallesteros, Ainara, Macarena Marambio, Verónica Fuentes, Mridvika Narda, Andreu Santín, and Josep-Maria Gili. 2021. "Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings—Implications for First Aid Protocols" Toxins 13, no. 8: 509. https://doi.org/10.3390/toxins13080509
APA StyleBallesteros, A., Marambio, M., Fuentes, V., Narda, M., Santín, A., & Gili, J.-M. (2021). Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings—Implications for First Aid Protocols. Toxins, 13(8), 509. https://doi.org/10.3390/toxins13080509