Bio-Preservative Potential of Microorganisms Isolated from Red Grape against Food Contaminant Fungi
Abstract
1. Introduction
2. Results and Discussion
2.1. Isolation and Identification of LAB Strains
2.2. Antifungal Activity Assays
2.3. Identification and Quantification of Compounds Present in the CFS
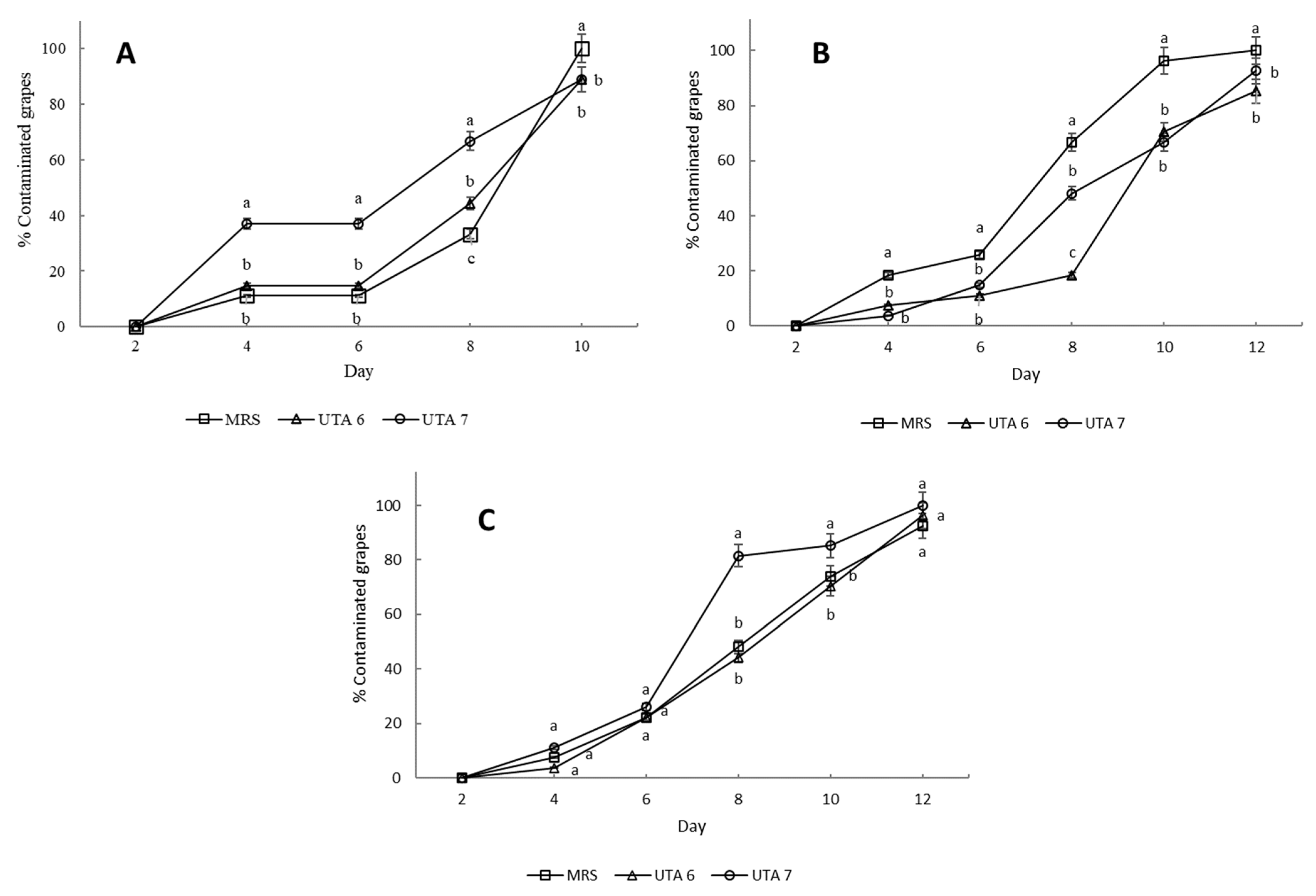
2.4. Leuconostoc fallax DSM 10614 and DSM 10615 as a Grape Bio-Preservative
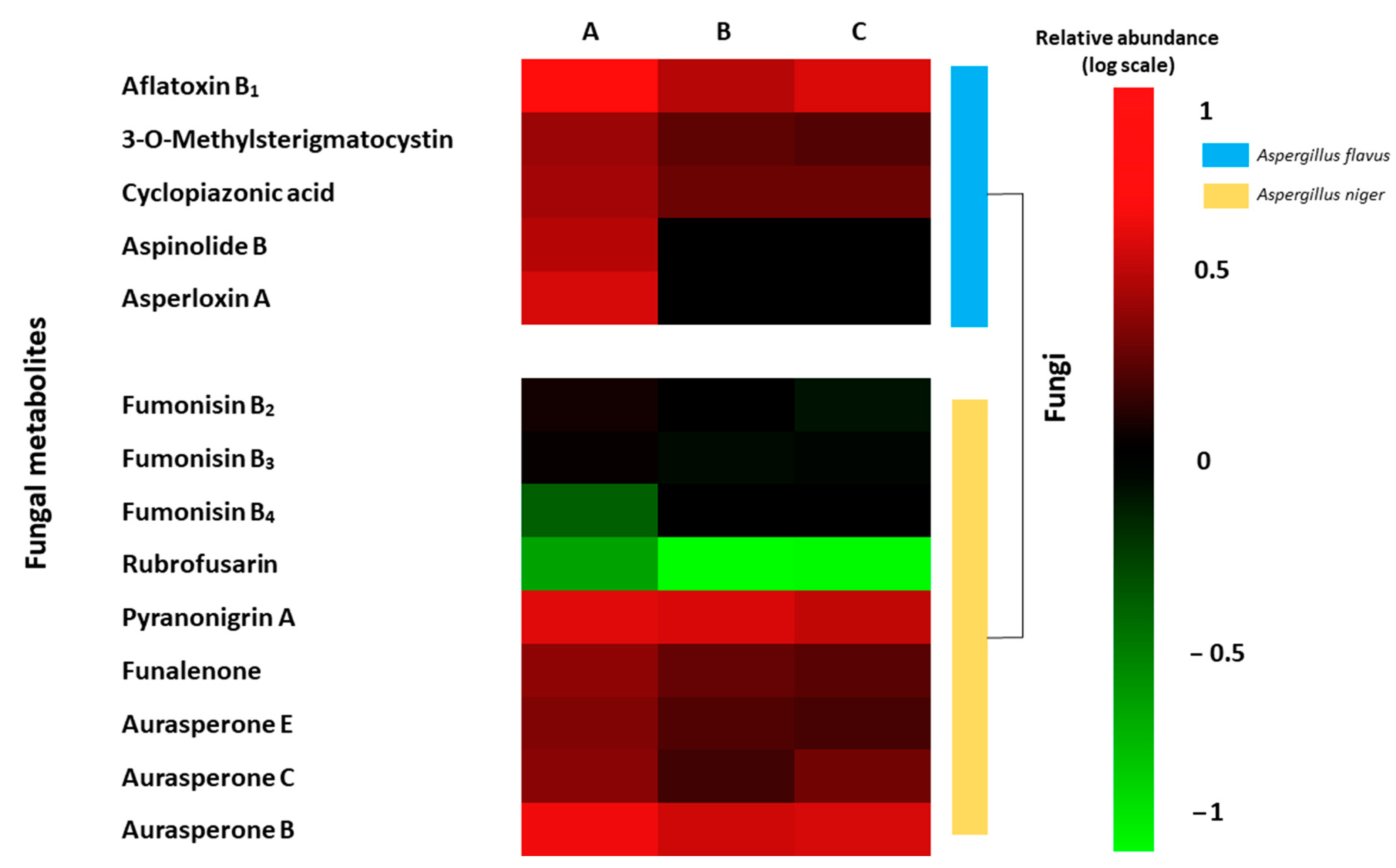
2.5. Metabolite Profiling of Fungal Growth on Grape
3. Conclusions
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Microorganisms Isolation
4.3. Use of a MALDI-TOF MS System and 16S rRNA Gene Sequencing for Bacterial Identification
4.4. Preparation of the CFS
4.5. Antifungal Activity Assays by Qualitative Method
4.6. Antifungal Activity Assays by Quantitative Method
4.7. Determination of Organic Acids and Phenolic Compounds in the CFS
4.8. Determination of Volatile Organic Compounds (VOC) in the CFS
4.9. Protection of Red Grapes from Fungal Spoilage by CFS
4.10. Metabolite Profiling of Fungal Growth on Grape
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leyva Salas, M.; Mounier, J.; Valence, F.; Coton, M.; Thierry, A.; Coton, E. Antifungal Microbial Agents for Food Biopreservation—A Review. Microorganisms 2017, 5, 37. [Google Scholar] [CrossRef]
- Barrios-Roblero, C.; Rosas-Quijano, R.; Salvador-Figueroa, M.; Gálvez-López, D.; Vázquez-Ovando, A. Antifungal Lactic Acid Bacteria Isolated from Fermented Beverages with Activity against Colletotrichum gloeosporioides. Food Biosci. 2019, 29, 47–54. [Google Scholar] [CrossRef]
- Lan, W.T.; Chen, Y.S.; Wu, H.C.; Yanagida, F. Bio-Protective Potential of Lactic Acid Bacteria Isolated from Fermented Wax Gourd. Folia Microbiol. 2012, 57, 99–105. [Google Scholar] [CrossRef]
- Ngolong Ngea, G.L.; Yang, Q.; Tchabo, W.; Castoria, R.; Zhang, X.; Zhang, H. Leuconostoc Mesenteroides subsp. Mesenteroides LB7 Isolated from Apple Surface Inhibits P. expansum in Vitro and Reduces Patulin in Fruit Juices. Int. J. Food Microbiol. 2021, 339, 109025. [Google Scholar]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The Microbial Ecology of Wine Grape Berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Malir, F.; Toman, J.; Grosse, J. Mycotoxins as human carcinogens—The IARC Monographs classification. Mycotoxin Res. 2017, 33, 65–73. [Google Scholar] [CrossRef]
- Nunes, C.A. Biological Control of Postharvest Diseases of Fruit. Eur. J. Plant Pathol. 2012, 133, 181–196. [Google Scholar] [CrossRef]
- Calado, T.; Venâncio, A.; Abrunhosa, L. Irradiation for Mold and Mycotoxin Control: A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1049–1061. [Google Scholar] [CrossRef]
- Hou, L.; Kou, X.; Li, R.; Wang, S. Thermal inactivation of fungi in chestnuts by hot air assisted radio frequency treatments. Food Control 2018, 93, 297–304. [Google Scholar] [CrossRef]
- Basak, S.; Guha, P. A review on antifungal activity and mode of action of essential oils and their delivery as nano-sized oil droplets in food system. J. Food Sci. Technol. 2018, 55, 4701–4710. [Google Scholar] [CrossRef]
- Yi, Y.J.; Li, Y.S.; Xia, B.; Li, W.P.; Pang, L.; Tong, Z.D. Optimization of Medium Composition and Culture Conditions for Antifungal Activity of a Tomato Endophytic Bacterium. Biol. Control 2015, 82, 69–75. [Google Scholar] [CrossRef]
- Luz, C.; Dopazo, V.; Quiles, J.M.; Romano, R.; Mañes, J.; Meca, G. Biopreservation of Tomatoes Using Fermented Media by Lactic Acid Bacteria. LWT Food Sci. Technol. 2020, 130, 109618. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, M.; Wang, J.; Lv, D.; Ma, Y.; Zhou, B.; Wang, B. Biocontrol Effects of Brevibacillus laterosporus AMCC100017 on Potato Common Scab and Its Impact on Rhizosphere Bacterial Communities. Biol. Control 2017, 106, 89–98. [Google Scholar] [CrossRef]
- Vinodkumar, S.; Indumathi, T.; Nakkeeran, S. Trichoderma Asperellum (NVTA2) as a Potential Antagonist for the Management of Stem Rot in Carnation under Protected Cultivation. Biol. Control 2017, 113, 58–64. [Google Scholar] [CrossRef]
- Dukare, A.; Paul, S. Biological Control of Fusarium Wilt and Growth Promotion in Pigeon Pea (Cajanus Cajan) by Antagonistic Rhizobacteria, Displaying Multiple Modes of Pathogen Inhibition. Rhizosphere 2021, 17, 100278. [Google Scholar] [CrossRef]
- Carr, F.J.; Chill, D.; Maida, N. The Lactic Acid Bacteria: A Literature Survey. Crit. Rev. Microbiol. 2002, 28, 281–370. [Google Scholar] [CrossRef]
- Li, K.; Zhang, W.; Kwok, L.Y.; Menghe, B. Screening of Lactobacillus plantarum with Broad-Spectrum Antifungal Activity and Its Application in Preservation of Golden-Red Apples. Czech J. Food Sci. 2020, 38, 315–322. [Google Scholar] [CrossRef]
- Salman, M.; Tariq, A.; Ijaz, A.; Naheed, S.; Hashem, A.; Abd-Allah, E.F.; Soliman, M.H.; Javed, M.R. In Vitro Antimicrobial and Antioxidant Activities of Lactobacillus coryniformis BCH-4 Bioactive Compounds and Determination of Their Bioprotective Effects on Nutritional Components of Maize (Zea Mays L.). Molecules 2020, 25, 4685. [Google Scholar] [CrossRef]
- Francesca, N.; Chiurazzi, M.; Romano, R.; Aponte, M.; Settanni, L.; Moschetti, G. Indigenous yeast communities in the environment of “Rovello bianco” grape variety and their use in commercial white wine fermentation. World J. Microbiol. Biotechnol. 2010, 26, 337–351. [Google Scholar] [CrossRef]
- Rice, T.; Sahin, A.W.; Lynch, K.M.; Arendt, E.K.; Coffey, A. Isolation, characterisation and exploitation of lactic acid bacteria capable of efficient conversion of sugars to mannitol. Int. J. Food Microbiol. 2020, 321, 108546. [Google Scholar] [CrossRef]
- Barrangou, R.; Yoon, S.S.; Breidt, F.; Fleming, H.P.; Klaenhammer, T.R. Identification and Characterization of Leuconostoc fallax Strains Isolated from an Industrial Sauerkraut. Ferment. Appl. Environ. Microbiol. 2002, 68, 2877–2884. [Google Scholar] [CrossRef]
- Trias, R.; Badosa, E.; Montesinos, E.; Bañeras, L. Bioprotective Leuconostoc strains against Listeria monocytogenes in fresh fruits and vegetables. Int. J. Food Microbiol. 2008, 127, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Le Lay, C.; Coton, E.; Le Blay, G.; Chobert, J.M.; Haertlé, T.; Choiset, Y.; Van Long, N.N.; Meslet-Cladière, L.; Mounier, J. Identification and Quantification of Antifungal Compounds Produced by Lactic Acid Bacteria and Propionibacteria. Int. J. Food Microbiol. 2016, 239, 79–85. [Google Scholar] [CrossRef]
- Kharchoufi, S.; Parafati, L.; Licciardello, F.; Muratore, G.; Hamdi, M.; Cirvilleri, G.; Restuccia, C. Edible Coatings Incorporating Pomegranate Peel Extract and Biocontrol Yeast to Reduce Penicillium digitatum Postharvest Decay of Oranges. Food Microbiol. 2018, 74, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Zamani-Zadeh, M.; Soleimanian-Zad, S.; Sheikh-Zeinoddin, M. Biocontrol of Gray Mold Disease on Strawberry Fruit by Integration of Lactobacillus plantarum A7 with Ajwain and Cinnamon Essential Oils. J. Food Sci. 2013, 78, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Izzo, L.; Luz, C.; Ritieni, A.; Quiles Beses, J.; Mañes, J.; Meca, G. Inhibitory Effect of Sweet Whey Fermented by Lactobacillus plantarum Strains against Fungal Growth: A Potential Application as an Antifungal Agent. J. Food Sci. 2020, 85, 3920–3926. [Google Scholar] [CrossRef]
- Martí-Quijal, F.J.; Príncep, A.; Tornos, A.; Luz, C.; Meca, G.; Tedeschi, P.; Ruiz, M.J.; Barba, F.J.; Mañes, J. Isolation, Identification and Investigation of Fermentative Bacteria from Sea Bass (Dicentrarchus Labrax): Evaluation of Antifungal Activity of Fermented Fish Meat and by-Products Broths. Foods 2020, 9, 576. [Google Scholar] [CrossRef]
- Lipinska-Zubrycka, L.; Klewicki, R.; Sojka, M.; Bonikowski, R.; Milczarek, A.; Klewicka, E. Anticandidal Activity of Lactobacillus spp. in the Presence of Galactosyl Polyols. Microbiol. Res. 2020, 240, 126540. [Google Scholar] [CrossRef]
- Nischitha, R.; Shivanna, M.B. Antimicrobial Activity and Metabolite Profiling of Endophytic Fungi in Digitaria bicornis (Lam) Roem. and Schult. and Paspalidium flavidum (Retz.) A. Camus. 3 Biotech 2021, 11, 53. [Google Scholar] [CrossRef]
- Gerez, C.L.; Torino, M.I.; Rollán, G.; Font de Valdez, G. Prevention of Bread Mould Spoilage by Using Lactic Acid Bacteria with Antifungal Properties. Food Control 2009, 20, 144–148. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Shi, X.; Wang, B.; Li, M.; Wang, Q.; Zhang, S. Antifungal Effect of Volatile Organic Compounds from Bacillus velezensis CT32 against Verticillium dahliae and Fusarium oxysporum. Processes 2020, 8, 1674. [Google Scholar] [CrossRef]
- Wang, Z.; Zhong, T.; Chen, K.; Du, M.; Chen, G.; Chen, X.; Wang, K.; Zalán, Z.; Takács, K.; Kan, J. Antifungal Activity of Volatile Organic Compounds Produced by Pseudomonas fluorescens ZX and Potential Biocontrol of Blue Mold Decay on Postharvest Citrus. Food Control 2021, 120, 107499. [Google Scholar] [CrossRef]
- Lappa, I.K.; Mparampouti, S.; Lanza, B.; Panagou, E.Z. Control of Aspergillus carbonarius in Grape Berries by Lactobacillus plantarum: A Phenotypic and Gene Transcription Study. Int. J. Food Microbiol. 2018, 275, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Kasfi, K.; Taheri, P.; Jafarpour, B.; Tarighi, S. Characterization of Antagonistic Microorganisms against Aspergillus spp. from Grapevine Leaf and Berry Surfaces. J. Plant Pathol. 2018, 100, 179–190. [Google Scholar] [CrossRef]
- Císarová, M.; Hleba, L.; Medo, J.; Tančinová, D.; Mašková, Z.; Čuboň, J.; Kováčik, A.; Foltinová, D.; Božik, M.; Klouček, P. The in vitro and in Situ Effect of Selected Essential Oils in Vapour Phase against Bread Spoilage Toxicogenic Aspergilli. Food Control 2020, 110, 107007. [Google Scholar] [CrossRef]
- Nazareth, T.M.; Luz, C.; Torrijos, R.; Quiles, J.M.; Luciano, F.B.; Mañes, J.; Meca, G. Potential Application of Lactic Acid Bacteria to Reduce Aflatoxin B1 and Fumonisin B1 Occurrence on Corn Kernels and Corn Ears. Toxins 2019, 12, 21. [Google Scholar] [CrossRef]
- Guimarães, A.; Santiago, A.; Teixeira, J.A.; Venâncio, A.; Abrunhosa, L. Antiaflatoxigenic effect of organic acids produced by Lactobacillus plantarum. Int. J. Food Microbiol. 2018, 264, 31–38. [Google Scholar] [CrossRef]
- Erkmen, O. Practice 4—Pure culture techniques. In Laboratory Practices in Microbiology; Erkmen, O., Ed.; Academic Press: Cambridge, MA, USA, 2021; pp. 41–50. [Google Scholar]
- Maier, T.; Klepel, S.; Renner, U.; Kostrzewa, M. Fast and Reliable MALDI-TOF MS-Based Microorganism Identification. Nat. Methods 2006, 3, 4. [Google Scholar] [CrossRef]
- Chenoll, E.; Moreno, I.; Sánchez, M.; Garcia-Grau, I.; Silvia, A.; González-Monfort, M.; Genovés, S.; Vilella, F.; Seco-Durban, C.; Simón, C.; et al. Selection of new probiotics for endometrial health. Front. Cell. Infect. Microbiol. 2019, 9, 114. [Google Scholar] [CrossRef]
- Khosravi, F.; Rastakhiz, N.; Iranmanesh, B.; Olia, S. Determination of organic acids in fruit juices by UPLC. Int. J. Biol. Sci. 2015, 9, 41–44. [Google Scholar] [CrossRef]
- Brosnan, B.; Coffey, A.; Arendt, E.K.; Furey, A. The QuEChERS Approach in a Novel Application for the Identification of Antifungal Compounds Produced by Lactic Acid Bacteria Cultures. Talanta 2014, 129, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Rodriguez, L.; Romano, R.; Mañes, J.; Meca, G. A Natural Strategy to Improve the Shelf Life of the Loaf Bread against Toxigenic Fungi: The Employment of Fermented Whey Powder. Int. J. Dairy Technol. 2020, 73, 88–97. [Google Scholar] [CrossRef]
- Xu, X.; Wu, B.; Zhao, W.; Lao, F.; Chen, F.; Liao, X.; Wu, J. Shifts in Autochthonous Microbial Diversity and Volatile Metabolites during the Fermentation of Chili Pepper (Capsicum Frutescens L.). Food Chem. 2021, 335, 127512. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.M.; De Melo Nazareth, T.; Luz, C.; Luciano, F.B.; Mañes, J.; Meca, G. Development of an Antifungal and Antimycotoxigenic Device Containing Allyl Isothiocyanate for Silo Fumigation. Toxins 2019, 11, 137. [Google Scholar] [CrossRef] [PubMed]



| Fungi | Sample | ||||||||||||||
| MRS | UTA1 | UTA2 | UTA3 | UTA4 | UTA5 | UTA6 | UTA7 | UTA8 | UTA9 | UTA10 | UTA11 | UTA12 | UTA13 | UTA14 | |
| P. expansum CECT 2278 | - | + | ++ | + | ++ | + | + | + | ++ | + | + | + | + | + | + |
| P. digitatum CECT 2954 | - | - | - | - | + | - | + | - | - | + | - | + | + | + | - |
| P. commune CECT 20767 | - | + | ++ | + | + | + | + | + | + | + | + | + | + | + | + |
| A. flavus ITEM 8111 | - | - | + | + | + | + | + | + | + | + | + | + | + | + | + |
| A. carbonarius ISPA 5010 | - | - | - | + | + | - | + | - | - | + | + | - | - | - | - |
| A. niger CECT 2088 | - | + | + | + | - | + | + | - | + | - | + | + | - | + | + |
| Al. alternata ITEM 8121 | - | - | + | + | + | + | + | + | + | + | + | + | + | + | + |
| F. graminearum ITEM 126 | - | + | + | + | + | + | + | + | ++ | + | + | + | + | + | + |
| F. proliferatum ITEM 12072 | - | - | + | + | + | + | + | + | + | + | + | + | + | + | + |
| F. verticillioides ITEM 12043 | - | - | + | + | + | + | + | + | + | + | + | + | + | + | + |
| B. cinerea CECT 20973 | - | - | + | + | + | + | + | + | + | + | + | + | + | + | + |
| Fungi | Sample | ||||||||||||||
| UTA15 | UTA16 | UTA17 | UTA18 | UTA19 | UTA20 | UB5 | UB6 | UB7 | UB8 | UBC1 | UTC6 | UT5 | VBC9 | ||
| P. expansum CECT 2278 | + | + | ++ | + | + | + | - | - | + | + | - | - | - | + | |
| P. digitatum CECT 2954 | - | - | - | - | + | + | - | - | - | - | - | - | - | - | |
| P. commune CECT 20767 | ++ | + | + | + | ++ | + | + | + | + | + | + | + | + | + | |
| A. flavus ITEM 8111 | + | + | + | + | + | + | - | - | + | + | - | - | - | - | |
| A. carbonarius ISPA 5010 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| A. niger CECT 2088 | + | + | - | + | + | + | + | + | + | + | + | + | + | + | |
| Al. alternata ITEM 8121 | + | + | + | + | + | + | - | - | + | + | - | - | - | + | |
| F. graminearum ITEM 126 | + | + | + | + | + | + | - | - | + | + | + | - | - | + | |
| F. proliferatum ITEM 12072 | + | + | + | + | + | + | - | - | + | + | - | - | - | + | |
| F. verticillioides ITEM 12043 | + | + | + | + | + | + | - | - | + | + | + | + | + | + | |
| B. cinerea CECT 20973 | + | + | + | + | + | + | - | - | + | + | - | - | - | + | |
| Fungi | Samples | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UTA2 | UTA3 | UTA4 | UTA5 | UTA6 | UTA7 | UTA8 | ||||||||
| MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | |
| P. expansum CECT 2278 | 13 | 13 | 13 | 13 | 13 | 13 | 13 | 13 | 13 | 25 | 25 | 25 | 13 | 25 |
| P. digitatum CECT 2954 | 100 | >100 | 50 | >100 | 25 | >100 | 25 | >100 | 50 | >100 | 100 | >100 | 13 | >100 |
| P. commune ECT 20767 | 25 | 50 | 25 | 50 | 25 | 25 | 25 | 50 | 25 | 50 | 25 | 50 | 25 | 50 |
| A. flavus ITEM 8111 | 50 | 50 | 50 | 100 | 50 | 50 | 25 | 50 | 25 | 50 | 25 | 50 | 50 | 50 |
| A. carbonarius ISPA 5010 | 50 | >100 | 50 | >100 | 50 | >100 | 50 | >100 | 50 | >100 | 50 | >100 | 50 | >100 |
| A. niger CECT 2088 | 100 | 100 | 100 | 100 | nd | nd | 100 | 100 | 25 | 100 | 25 | 100 | 50 | >100 |
| Al. alternata ITEM 8121 | 13 | 25 | 13 | 25 | 13 | 13 | 13 | 50 | 6 | 25 | 13 | 25 | 13 | 25 |
| F. graminearum ITEM 126 | 13 | 25 | 13 | 25 | 13 | 50 | 13 | 13 | 6 | 6 | 13 | 13 | 6 | 25 |
| F. proliferatum ITEM 12072 | 13 | 25 | 13 | 25 | 13 | 25 | 13 | 50 | 13 | 13 | 13 | 50 | 13 | 50 |
| F. verticillioides ITEM 12043 | 6 | 13 | 6 | 13 | 3 | 13 | 3 | 13 | 6 | 13 | 6 | 25 | 6 | 25 |
| B. cinerea CECT 20973 | 50 | 100 | 50 | >100 | 50 | 50 | 50 | 100 | 25 | 50 | 25 | 50 | 50 | 100 |
| Fungi | UTA9 | UTA10 | UTA11 | UTA12 | UTA13 | UTA14 | UTA15 | |||||||
| P. expansum CECT 2278 | 13 | 50 | 25 | 50 | 25 | >100 | 13 | 13 | 25 | 25 | 13 | 25 | 13 | 50 |
| P. digitatum CECT 2954 | 25 | >100 | 25 | >100 | 25 | >100 | 25 | >100 | 100 | >100 | 25 | >100 | 50 | >100 |
| P. commune ECT 20767 | 25 | 50 | 25 | 50 | 25 | 50 | 25 | 50 | 50 | 50 | 25 | 50 | 25 | 50 |
| A. flavus ITEM 8111 | 25 | 50 | 50 | 50 | 50 | 100 | 50 | 50 | 50 | 100 | 50 | 50 | 50 | 50 |
| A. carbonarius ISPA 5010 | 50 | 100 | 50 | 100 | 50 | >100 | 50 | 100 | 50 | 100 | 50 | >100 | 50 | 100 |
| A. niger CECT 2088 | nd | nd | 50 | >100 | 50 | >100 | nd | nd | 100 | >100 | 100 | 100 | 100 | >100 |
| Al. alternata ITEM 8121 | 13 | 25 | 13 | 25 | 13 | 50 | 13 | 25 | 13 | 25 | 13 | 25 | 13 | 25 |
| F. graminearum ITEM 126 | 6 | 50 | 6 | 50 | 6 | 50 | 13 | 25 | 13 | 25 | 6 | 50 | 6 | 50 |
| F. proliferatum ITEM 12072 | 13 | 50 | 13 | 50 | 13 | 50 | 13 | 50 | 13 | 50 | 13 | 50 | 6 | 25 |
| F. verticillioides ITEM 12043 | 3 | 13 | 3 | 6 | 3 | 6 | 13 | 13 | 3 | 25 | 3 | 25 | 3 | 13 |
| B. cinerea CECT 20973 | 50 | 50 | 50 | 100 | 50 | 100 | 50 | 50 | 50 | >100 | 50 | >100 | 50 | 100 |
| Fungi | UTA16 | UTA17 | UTA18 | UTA19 | UTA20 | UB7 | UB8 | |||||||
| P. expansum CECT 2278 | 13 | 25 | 13 | 50 | 13 | 50 | 13 | 50 | 13 | 50 | 13 | 25 | 13 | 22 |
| P. digitatum CECT 2954 | 50 | >100 | 50 | >100 | 100 | >100 | 100 | >100 | 50 | >100 | 50 | >100 | 25 | >100 |
| P. commune ECT 20767 | 25 | 25 | 25 | 50 | 50 | 50 | 50 | >100 | 50 | 50 | 50 | 50 | 25 | 50 |
| A. flavus ITEM 8111 | 50 | 100 | 50 | 50 | 25 | 100 | 25 | 100 | 25 | 100 | 50 | >100 | 50 | 100 |
| A. carbonarius ISPA 5010 | 50 | 100 | 50 | 100 | 50 | >100 | 50 | 100 | 50 | 100 | 100 | 100 | 100 | 100 |
| A. niger CECT 2088 | nd | nd | nd | nd | 50 | 100 | 100 | >100 | 100 | >100 | 100 | >100 | 100 | >100 |
| Al. alternata ITEM 8121 | 6 | 25 | 6 | 25 | 6 | 50 | 13 | 25 | 25 | 50 | 13 | 25 | 13 | 25 |
| F. graminearum ITEM 126 | 13 | 25 | 13 | 50 | 13 | 50 | 13 | 100 | 6 | 100 | 6 | 13 | 25 | 25 |
| F. proliferatum ITEM 12072 | 13 | 25 | 13 | 25 | 13 | 25 | 6 | 25 | 13 | 25 | 13 | 25 | 13 | 25 |
| F. verticillioides ITEM 12043 | 3 | 13 | 3 | 13 | 3 | 6 | 3 | 6 | 3 | 13 | 6 | 13 | 6 | 6 |
| B. cinerea CECT 20973 | 50 | 50 | 50 | >100 | 50 | >100 | 50 | >100 | 100 | >100 | 50 | >100 | 50 | 100 |
| (a) Oraganic acids | UTA1 | UTA2 | UTA3 | UTA4 | UTA5 | UTA6 | UTA7 | UTA8 | UTA9 | UTA10 |
| Lactic acid | 1.5 ± 0.6 cf | 3.5 ± 0.2 ae | 3.0 ± 0.3 ae | 3.7 ± 1.1 ae | 4.3 ± 0.7 a | 3.9 ± 0.7 ad | 4.2 ± 0.2 ab | 4.3 ± 0.2 a | 3.5 ± 0.2 ae | 3.9 ± 0.8 ac |
| Acetic acid | 1.3 ± 0.5 a | 0.8 ± 0.3 a | 1.2 ± 0.4 a | 1.7 ± 0.6 a | 1.4 ± 0.8 a | 1.3 ± 0.1 a | 0.8 ± 0.1 a | 1.1 ± 0.4 a | 1.3 ± 0.6 a | 1.4 ± 0.8 a |
| UTA11 | UTA12 | UTA13 | UTA14 | UTA15 | UTA16 | UTA17 | UTA18 | UTA19 | ||
| Lactic acid | 3.5 ± 0.5 ae | 3.4 ± 0.4 ae | 3.1 ± 0.2 ae | 2.3 ± 0.6 af | 4.1 ± 1.7 ab | 3.9 ± 0.9 ad | 3.5 ± 0.5 ae | 2.2 ± 1.0 af | 3.3 ± 0.4 ae | |
| Acetic acid | 1.1 ± 0.2a | 1.4 ± 0.3 a | 1.7 ± 0.4 a | 1.0 ± 0.4 a | 1.5 ± 0.7 a | 1.5 ± 0.3 a | 2.0 ± 0.3 a | 0.3 ± 0.6 a | 1.5 ± 0.3 a | |
| UTA20 | UB5 | UB6 | UB7 | UB8 | UBC1 | UTC6 | UT5 | VBC9 | ||
| Lactic acid | 3.2 ± 0.4 abcde | 1.8 ± 0.4 bf | nd | 3.1 ± 0.5 ae | 3.0 ± 0.8 ae | 1.3 ± 0.5 ef | 2.9 ± 0.2 ae | 1.4 ± 0.3 df | 2.9 ± 1.6 abe | |
| Acetic acid | 1.5 ± 0.3 a | 1.7 ± 0.4 a | 2.2 ± 0.6 a | 2.1 ± 0.4 a | 1.8 ± 0.6 a | 1.4 ± 0.3 a | 1.3 ± 0.5 a | 1.4 ± 0.3 a | 1.3 ± 0.1 a | |
| (b) Phenolic compounds | UTA1 | UTA2 | UTA3 | UTA4 | UTA5 | UTA6 | UTA7 | UTA8 | UTA9 | UTA10 |
| Phenyllactic acid | 1.3 ± 0.1 ad | 1.0 ± 0.1 dg | 1.4 ± 0.2 ab | 1.2 ± 0.1 ad | 1.2 ± 0.1 ad | 1.4 ± 0.1 ab | 0.8 ± 0.1 fh | 1.0 ± 0.1 cf | 1.4 ± 0.1 a | 1.3 ± 0.1 ac |
| UTA11 | UTA12 | UTA13 | UTA14 | UTA15 | UTA16 | UTA17 | UTA18 | UTA19 | ||
| 0.8 ± 0.1 fh | 0.7 ± 0.1 gh | 0.9 ± 0.1 fh | 0.9 ± 0.1 eg | 1.2 ± 0.1 ad | 0.8 ± 0.0 fh | 0.6 ± 0.1 h | 0.8 ± 0.1 fh | 1.2 ± 0.1 be | ||
| UTA20 | UB5 | UB6 | UB7 | UB8 | UBC1 | UTC6 | UT5 | VBC9 | ||
| 0.9 ± 0.1 fh | nd | nd | 0.8 ± 0.1 fh | 0.8 ± 0.1 fh | nd | 0.8 ± 0.1 gh | nd | 0.6 ± 0.1 h |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dopazo, V.; Luz, C.; Mañes, J.; Quiles, J.M.; Carbonell, R.; Calpe, J.; Meca, G. Bio-Preservative Potential of Microorganisms Isolated from Red Grape against Food Contaminant Fungi. Toxins 2021, 13, 412. https://doi.org/10.3390/toxins13060412
Dopazo V, Luz C, Mañes J, Quiles JM, Carbonell R, Calpe J, Meca G. Bio-Preservative Potential of Microorganisms Isolated from Red Grape against Food Contaminant Fungi. Toxins. 2021; 13(6):412. https://doi.org/10.3390/toxins13060412
Chicago/Turabian StyleDopazo, Victor, Carlos Luz, Jordi Mañes, Juan Manuel Quiles, Raquel Carbonell, Jorge Calpe, and Giuseppe Meca. 2021. "Bio-Preservative Potential of Microorganisms Isolated from Red Grape against Food Contaminant Fungi" Toxins 13, no. 6: 412. https://doi.org/10.3390/toxins13060412
APA StyleDopazo, V., Luz, C., Mañes, J., Quiles, J. M., Carbonell, R., Calpe, J., & Meca, G. (2021). Bio-Preservative Potential of Microorganisms Isolated from Red Grape against Food Contaminant Fungi. Toxins, 13(6), 412. https://doi.org/10.3390/toxins13060412






