The Occurrence of Mycotoxins in Raw Materials and Fish Feeds in Europe and the Potential Effects of Deoxynivalenol (DON) on the Health and Growth of Farmed Fish Species—A Review
Abstract
1. Introduction
1.1. Mycotoxin-Producing Fungi
1.2. Classification of Fusarium Mycotoxins: “Traditional”, “Emerging” and “Masked”
2. Results
2.1. Survey of Feed Ingredients and Aquafeeds
2.1.1. Wheat
Wheat as an Ingredient in Aquafeeds
Mycotoxins in Wheat
2.1.2. Corn as an Ingredient in Aquafeeds
Mycotoxins in Corn
2.1.3. Soybean Meal
Soybean Meal as an Ingredient in Aquafeeds
Mycotoxins in Soybean Meal
2.1.4. Probability of Mycotoxin Co-Occurrence in Feedstuffs: The Case of Corn
2.1.5. Aquafeeds
Mycotoxins in Aquafeeds
2.2. Effects of Deoxynivalenol (DON) on Fish Species
2.2.1. Systematic Review
Salmon
Rainbow Trout
Carp
Zebrafish
Tilapia
Catfish
Metabolic Fate of DON
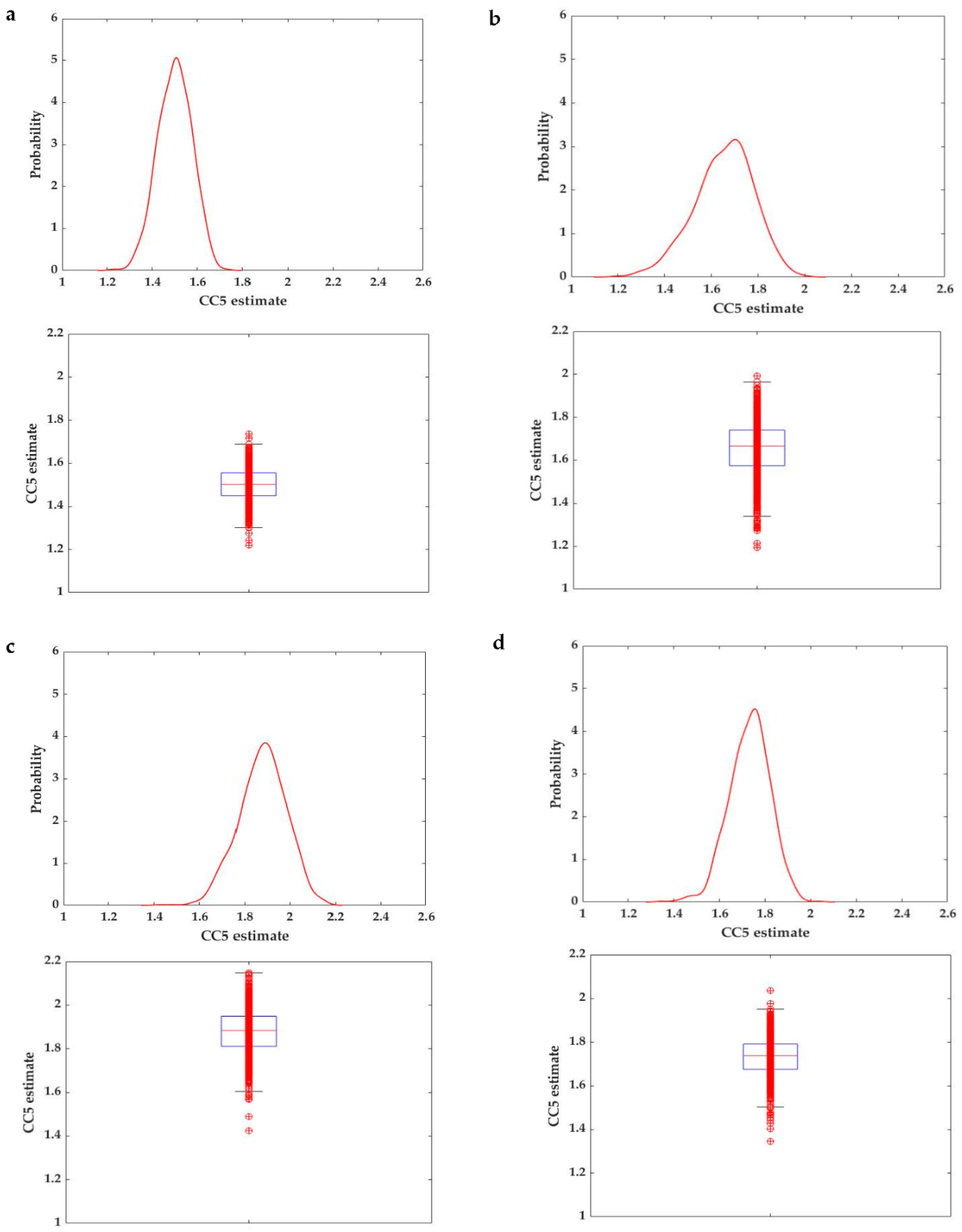
2.2.2. Quantifying the Risk of DON Exposure in Fish
2.2.3. A Meta-Analytical Approach
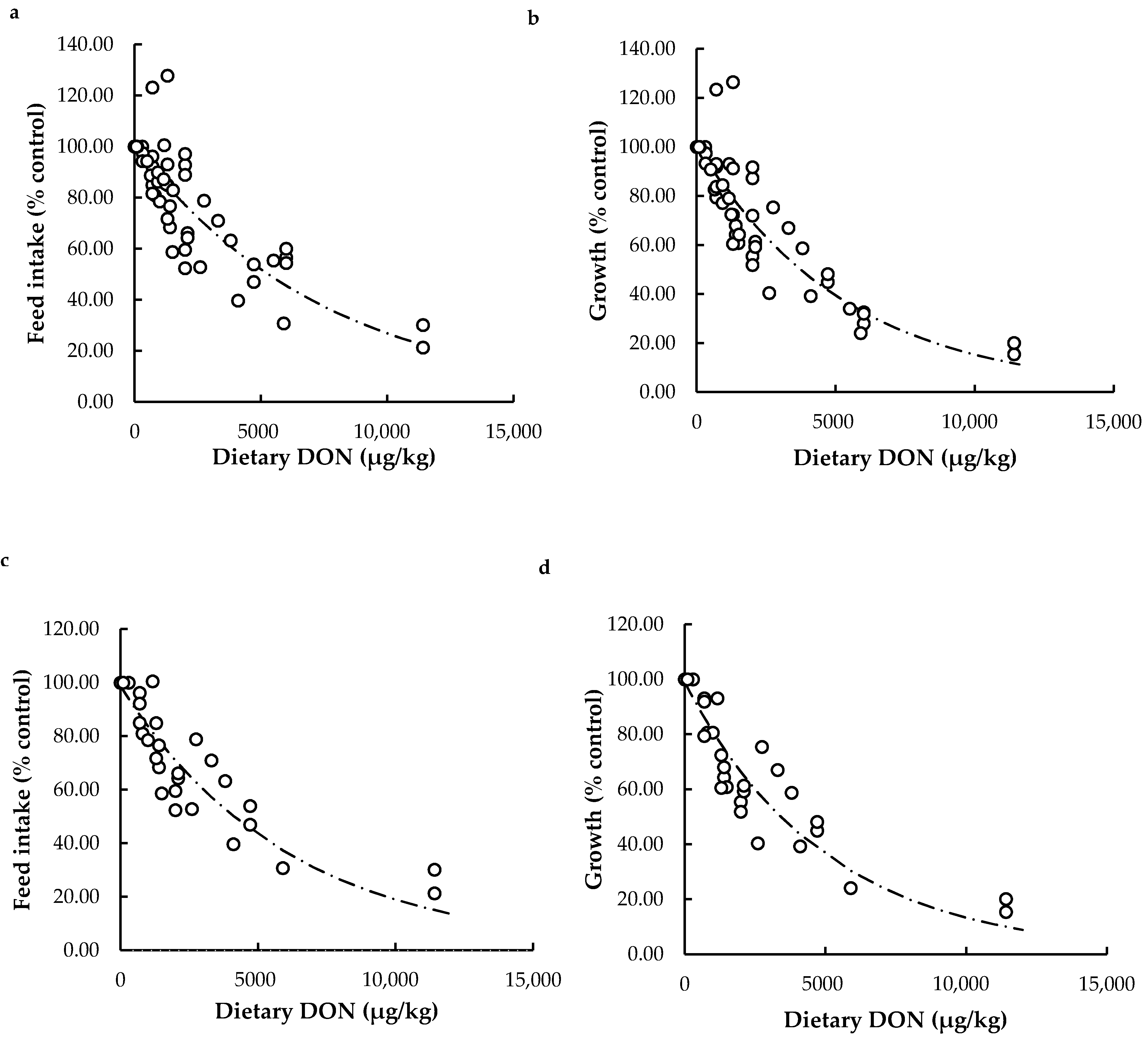
Effects of Dietary DON on Feed Intake and Growth
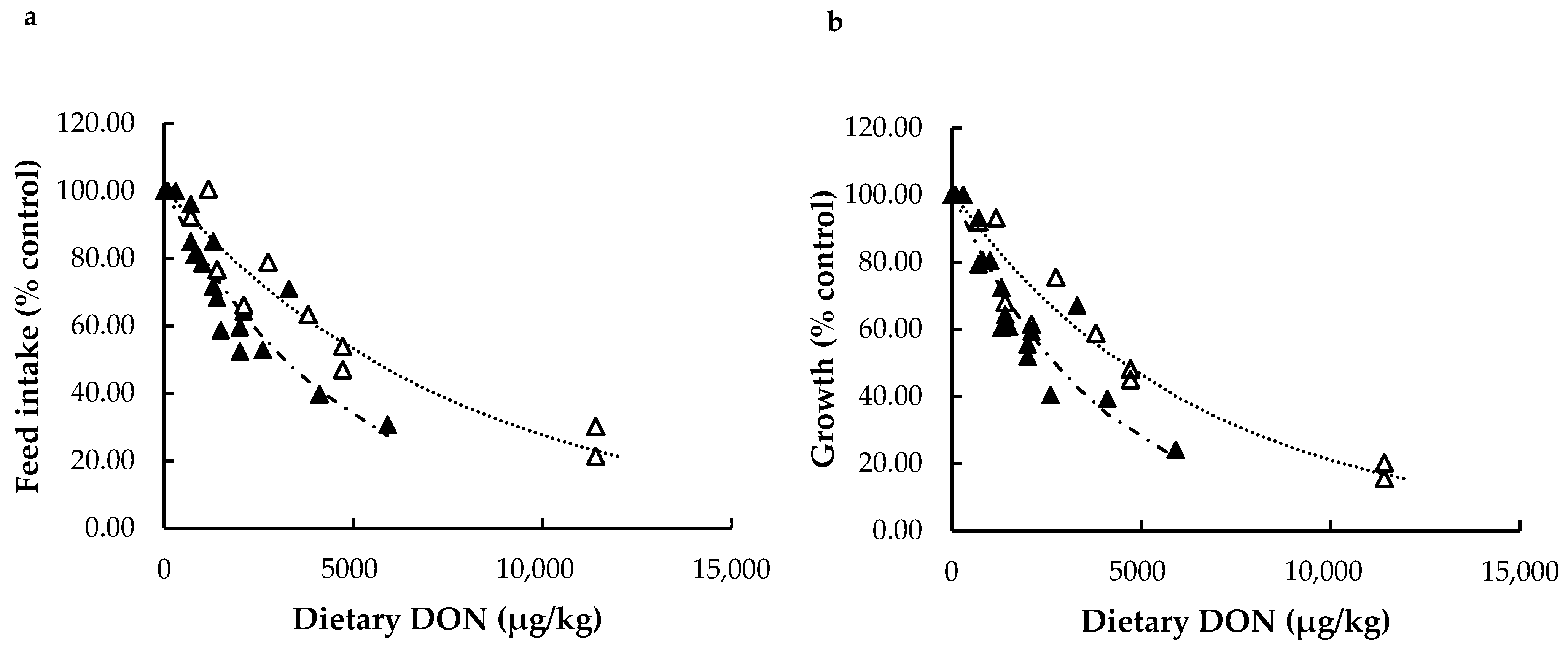
Different Types of DON in Rainbow Trout: Natural vs. Pure
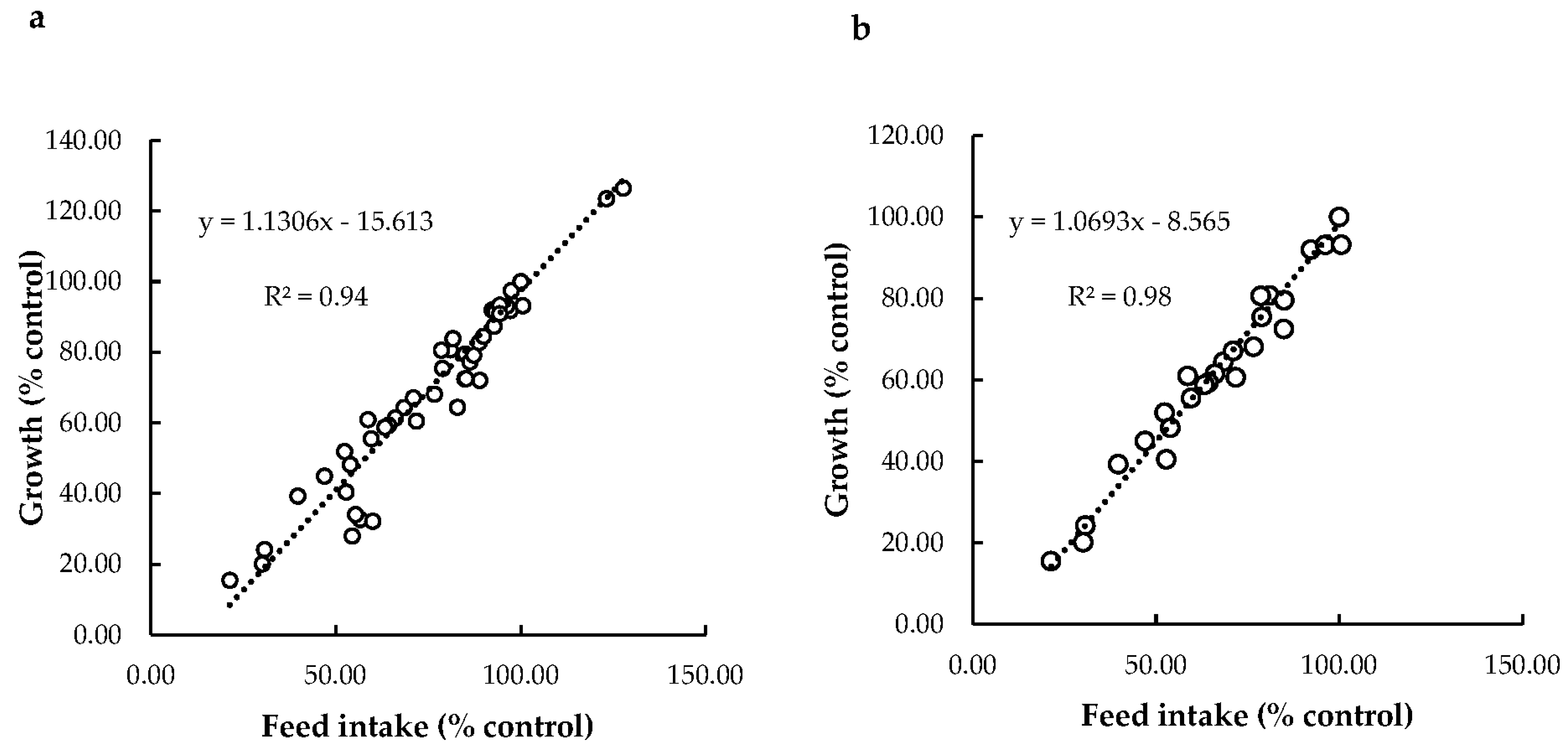
Relationship between Feed Intake and Growth
3. Discussion
4. Materials and Methods
4.1. Survey
4.2. Systematic Review
4.3. Risk Assessment
4.4. Meta-Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018. [Google Scholar]
- Naylor, R.L.; Hardy, R.W.; Bureau, D.P.; Chiu, A.; Elliott, M.; Farrell, A.P.; Forster, I.; Gatlin, D.M.; Goldburg, R.J.; Hua, K.; et al. Feeding aquaculture in an era of finite resources. Proc. Natl. Acad. Sci. USA 2009, 106, 15103–15110. [Google Scholar] [CrossRef]
- Troell, M.; Naylor, R.L.; Metian, M.; Beveridge, M.; Tyedmers, P.H.; Folke, C.; Arrow, K.J.; Barrett, S.; Crépin, A.-S.; Ehrlich, P.R.; et al. Does aquaculture add resilience to the global food system? Proc. Natl. Acad. Sci. USA 2014, 111, 13257–13263. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Feed matters: Satisfying the feed demand of aquaculture. Rev. Fish. Sci. Aquac. 2015, 23, 1–10. [Google Scholar] [CrossRef]
- Alltech. Global Feed Survey. 2019. Available online: https://www.alltech.com/feed-survey (accessed on 20 November 2019).
- Fry, J.P.; Love, D.C.; MacDonald, G.K.; West, P.C.; Engstrom, P.M.; Nachman, K.E.; Lawrence, R.S. Environmental health impacts of feeding crops to farmed fish. Environ. Int. 2016, 91, 201–214. [Google Scholar] [CrossRef]
- Naylor, R.L.; Hardy, R.W.; Buschmann, A.H.; Bush, S.R.; Cao, L.; Klinger, D.H.; Little, D.C.; Lubchenco, J.; Shumway, S.E.; Troell, M. A 20-year retrospective review of global aquaculture. Nature 2021, 591, 551–563. [Google Scholar] [CrossRef]
- Ytrestøyl, T.; Aas, T.S.; Åsgård, T. Utilisation of feed resources in production of Atlantic salmon (Salmo salar) in Norway. Aquaculture 2015, 448, 365–374. [Google Scholar] [CrossRef]
- Turchini, G.M.; Torstensen, B.E.; Ng, W.-K. Fish oil replacement in finfish nutrition. Rev. Aquac. 2009, 1, 10–57. [Google Scholar] [CrossRef]
- Hardy, R.W. Utilization of plant proteins in fish diets: Effects of global demand and supplies of fishmeal. Aquac. Res. 2010, 41, 770–776. [Google Scholar] [CrossRef]
- Francis, G.; Makkar, H.P.S.; Becker, K. Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 2001, 199, 197–227. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Aquaculture feed and food safety. Ann. N. Y. Acad. Sci. 2008, 1140, 50–59. [Google Scholar] [CrossRef]
- Softeland, L.; Kirwan, J.A.; Hori, T.S.; Storseth, T.R.; Sommer, U.; Berntssen, M.H.; Viant, M.R.; Rise, M.L.; Waagbo, R.; Torstensen, B.E.; et al. Toxicological effect of single contaminants and contaminant mixtures associated with plant ingredients in novel salmon feeds. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2014, 73, 157–174. [Google Scholar] [CrossRef] [PubMed]
- Glencross, B. 3—Understanding the nutritional and biological constraints of ingredients to optimize their application in aquaculture feeds. In Aquafeed Formulation; Nates, S.F., Ed.; Academic Press: San Diego, CS, USA, 2016; pp. 33–73. [Google Scholar]
- Kokou, F.; Fountoulaki, E. Aquaculture waste production associated with antinutrient presence in common fish feed plant ingredients. Aquaculture 2018, 495, 295–310. [Google Scholar] [CrossRef]
- De Boevre, M.; Di Mavungu, J.D.; Landschoot, S.; Audenaert, K.; Eeckhout, M.; Maene, P.; Haesaert, G.; De Saeger, S. Natural occurrence of mycotoxins and their masked forms in food and feed products. World Mycotoxin J. 2012, 5, 207–219. [Google Scholar] [CrossRef]
- Pettersson, H.; Fink-Gremmels, J. 11—Mycotoxin contamination of animal feed. In Animal Feed Contamination; Woodhead Publishing: Cambridge, UK, 2012; pp. 233–285. [Google Scholar]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed-focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef]
- Kovalsky, P.; Kos, G.; Nährer, K.; Schwab, C.; Jenkins, T.; Schatzmayr, G.; Sulyok, M.; Krska, R. Co-occurrence of regulated, masked and emerging mycotoxins and secondary metabolites in finished feed and maize-an extensive survey. Toxins 2016, 8, 363. [Google Scholar] [CrossRef]
- Pietsch, C.; Kersten, S.; Burkhardt-Holm, P.; Valenta, H.; Danicke, S. Occurrence of deoxynivalenol and zearalenone in commercial fish feed: An initial study. Toxins 2013, 5, 184–192. [Google Scholar] [CrossRef]
- Greco, M.; Pardo, A.; Pose, G. Mycotoxigenic fungi and natural co-occurrence of mycotoxins in rainbow trout (Oncorhynchus mykiss) feeds. Toxins 2015, 7, 4595–4609. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.A.; Schatzmayr, D.; Hofstetter, U.; Santos, G.A. Occurrence of mycotoxins in aquaculture: Preliminary overview of Asian and European plant ingredients and finished feeds. World Mycotoxin J. 2017, 10, 183–194. [Google Scholar] [CrossRef]
- Gonçalves, R.A.; Schatzmayr, D.; Albalat, A.; Mackenzie, S. Mycotoxins in aquaculture: Feed and food. Rev. Aquac. 2018, 12, 145–175. [Google Scholar] [CrossRef]
- Marijani, E.; Wainaina, J.M.; Charo-Karisa, H.; Nzayisenga, L.; Munguti, J.; Joselin Benoit Gnonlonfin, G.; Kigadye, E.; Okoth, S. Mycoflora and mycotoxins in finished fish feed and feed ingredients from smallholder farms in East Africa. Egypt. J. Aquat. Res. 2017, 43, 169–176. [Google Scholar] [CrossRef]
- Tola, M.; Kebede, B. Occurrence, importance and control of mycotoxins: A review. Cogent Food Agric. 2016, 2, 1191103. [Google Scholar] [CrossRef]
- Sweeney, M.J.; Dobson, A.D. Mycotoxin production by Aspergillus, Fusarium and Penicillium species. Int. J. Food Microbiol. 1998, 43, 141–158. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Magan, N.; Aldred, D. Post-harvest control strategies: Minimizing mycotoxins in the food chain. Int. J. Food Microbiol. 2007, 119, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Mannaa, M.; Kim, K.D. Influence of temperature and water activity on deleterious fungi and mycotoxin production during grain storage. Mycobiology 2017, 45, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Neme, K.; Ibrahim, A.M. Mycotoxin occurrence in grains and the role of postharvest management as a mitigation strategies. A Review. Food Control. 2017, 78, 412–425. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2010, 43, 1902–1914. [Google Scholar] [CrossRef]
- Almeida, I.F.M.; Martins, H.M.L.; Santos, S.M.O.; Freitas, M.S.; da Costa, J.M.G.N.; Almeida Bernardo, F.M.D. Mycobiota and aflatoxin B1 in feed for farmed sea bass (Dicentrarchus labrax). Toxins 2011, 3, 163–171. [Google Scholar] [CrossRef]
- Alkhayyat, F.; Yu, J.-H. Chapter Five—Upstream Regulation of Mycotoxin Biosynthesis. In Advances in Applied Microbiology; Sariaslani, S., Gadd, G.M., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 86, pp. 251–278. [Google Scholar]
- Venkatesh, N.; Keller, N.P. Mycotoxins in conversation with bacteria and fungi. Front. Microbiol. 2019, 10, 403. [Google Scholar] [CrossRef]
- Pietsch, C.; Müller, G.; Mourabit, S.; Carnal, S.; Bandara, K. Occurrence of fungi and fungal toxins in fish feed during storage. Toxins 2020, 12, 171. [Google Scholar] [CrossRef] [PubMed]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant. Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Perincherry, L.; Lalak-Kańczugowska, J.; Stępień, Ł. Fusarium-produced mycotoxins in plant-pathogen interactions. Toxins 2019, 11, 664. [Google Scholar] [CrossRef] [PubMed]
- Nesic, K.; Ivanovic, S.; Nesic, V. Fusarial toxins: Secondary metabolites of Fusarium fungi. In Reviews of Environmental Contamination and Toxicology; Springer International Publishing: Cham, Switzerland, 2014; Volume 228, pp. 101–120. [Google Scholar]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 60, 218–237. [Google Scholar] [CrossRef] [PubMed]
- Nathanail, A.V.; Varga, E.; Meng-Reiterer, J.; Bueschl, C.; Michlmayr, H.; Malachova, A.; Fruhmann, P.; Jestoi, M.; Peltonen, K.; Adam, G.; et al. Metabolism of the Fusarium mycotoxins T-2 toxin and HT-2 toxin in wheat. J. Agric. Food Chem. 2015, 63, 7862–7872. [Google Scholar] [CrossRef] [PubMed]
- Dohnal, V.; Jezkova, A.; Jun, D.; Kuca, K. Metabolic pathways of T-2 toxin. Curr. Drug Metab. 2008, 9, 77–82. [Google Scholar] [PubMed]
- Li, Y.; Wang, Z.; Beier, R.C.; Shen, J.; Smet, D.D.; De Saeger, S.; Zhang, S. T-2 toxin, a trichothecene mycotoxin: Review of toxicity, metabolism, and analytical methods. J. Agric. Food Chem. 2011, 59, 3441–3453. [Google Scholar] [CrossRef]
- Adhikari, M.; Negi, B.; Kaushik, N.; Adhikari, A.; Al-Khedhairy, A.A.; Kaushik, N.K.; Choi, E.H. T-2 mycotoxin: Toxicological effects and decontamination strategies. Oncotarget 2017, 8, 33933–33952. [Google Scholar] [CrossRef]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef]
- Jestoi, M. Emerging fusarium-mycotoxins fusaproliferin, beauvericin, enniatins, and moniliformin: A review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef] [PubMed]
- Gratz, S.W. Do plant-bound masked mycotoxins contribute to toxicity? Toxins 2017, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Nie, D.; Fan, K.; Yang, J.; Guo, W.; Meng, J.; Zhao, Z.; Han, Z. A systematic review of plant-conjugated masked mycotoxins: Occurrence, toxicology, and metabolism. Crit. Rev. Food Sci. Nutr. 2019, 60, 1523–1537. [Google Scholar] [CrossRef] [PubMed]
- Nagl, V.; Schwartz, H.; Krska, R.; Moll, W.-D.; Knasmüller, S.; Ritzmann, M.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in rats. Toxicol. Lett. 2012, 213, 367–373. [Google Scholar] [CrossRef]
- Schwartz-Zimmermann, H.E.; Binder, S.B.; Hametner, C.; Miró-Abella, E.; Schwarz, C.; Michlmayr, H.; Reiterer, N.; Labudova, S.; Adam, G.; Berthiller, F. Metabolism of nivalenol and nivalenol-3-glucoside in rats. Toxicol. Lett. 2019, 306, 43–52. [Google Scholar] [CrossRef]
- Nagl, V.; Woechtl, B.; Schwartz-Zimmermann, H.E.; Hennig-Pauka, I.; Moll, W.-D.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in pigs. Toxicol. Lett. 2014, 229, 190–197. [Google Scholar] [CrossRef]
- Binder, S.B.; Schwartz-Zimmermann, H.E.; Varga, E.; Bichl, G.; Michlmayr, H.; Adam, G.; Berthiller, F. Metabolism of zearalenone and its major modified forms in pigs. Toxins 2017, 9, 56. [Google Scholar] [CrossRef]
- Rychlik, M.; Humpf, H.-U.; Marko, D.; Dänicke, S.; Mally, A.; Berthiller, F.; Klaffke, H.; Lorenz, N. Proposal of a comprehensive definition of modified and other forms of mycotoxins including “masked” mycotoxins. Mycotoxin Res. 2014, 30, 197–205. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 2002/32/EC of the European parliament and of the council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Commun. 2002, 45, L140. [Google Scholar]
- European Commission. Commission recommendation of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding (2006/576/EC). Off. J. Eur. Union 2006, 229, 7–9. [Google Scholar]
- European Commission. Commission recommendation No 2012/154/EU of 15 March 2012 on the monitoring of the presence of ergot alkaloids in feed and food. Off. J. Eur Union 2012, 77, 20–21. [Google Scholar]
- European Commission. Commission recommendation of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereals and cereal products. Off. J. Eur. Union L 2013, 91, 12–15. [Google Scholar]
- Smith, M.C.; Madec, S.; Coton, E.; Hymery, N. Natural co-occurrence of mycotoxins in foods and feeds and their in vitro combined toxicological effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef]
- FAOSTAT. Crops Production. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 30 May 2021).
- Tacon, A.G.; Hasan, M.R.; Metian, M. Demand and Supply of Feed Ingredients for Farmed Fish and Crustaceans: Trends and Prospects; FAO Fisheries and Aquaculture Technical Paper 564; FAO: Rome, Italy, 2011. [Google Scholar]
- Hatfield, J.L.; Dold, C. Agroclimatology and wheat production: Coping with climate change. Front. Plant. Sci. 2018, 9, 9. [Google Scholar] [CrossRef]
- Asseng, S.; Ewert, F.; Martre, P.; Rötter, R.P.; Cammarano, D.; Kimball, B.A.; Ottman, M.J.; Wall, G.W.; White, J.W.; Reynolds, M.P.; et al. Letter: Rising temperatures reduce global wheat production. Nat. Clim. Chang. 2015, 5, 143–147. [Google Scholar] [CrossRef]
- Thielecke, F.; Nugent, A.P. Contaminants in grain-a major risk for whole grain safety? Nutrients 2018, 10, 1213. [Google Scholar] [CrossRef] [PubMed]
- Gatlin Iii, D.M.; Barrows, F.T.; Brown, P.; Dabrowski, K.; Gaylord, T.G.; Hardy, R.W.; Herman, E.; Hu, G.; Krogdahl, Å.; Nelson, R.; et al. Expanding the utilization of sustainable plant products in aquafeeds: A review. Aquac. Res. 2007, 38, 551–579. [Google Scholar] [CrossRef]
- Jouany, J.P. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. Technol. 2007, 137, 342–362. [Google Scholar] [CrossRef]
- Lall, S.P.; Dumas, A.; Davis, D.A. 3—Nutritional requirements of cultured fish: Formulating nutritionally adequate feeds. In Feed and Feeding Practices in Aquaculture; Woodhead Publishing: Oxford, UK, 2015; pp. 53–109. [Google Scholar]
- Curtui, V.; Usleber, E.; Dietrich, R.; Lepschy, J.; Märtlbauer, E. A survey on the occurrence of mycotoxins in wheat and maize from western Romania. Mycopathologia 1998, 143, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Rafai, P.; Bata, A.; Jakab, L.; Vanyi, A. Evaluation of mycotoxin-contaminated cereals for their use in animal feeds in Hungary. Food Addit. Contam. 2000, 17, 799–808. [Google Scholar] [CrossRef]
- Škrbić, B.; Malachova, A.; Živančev, J.; Veprikova, Z.; Hajšlová, J. Fusarium mycotoxins in wheat samples harvested in Serbia: A preliminary survey. Food Control. 2011, 22, 1261–1267. [Google Scholar] [CrossRef]
- Alkadri, D.; Rubert, J.; Prodi, A.; Pisi, A.; Mañes, J.; Soler, C. Natural co-occurrence of mycotoxins in wheat grains from Italy and Syria. Food Chem. 2014, 157, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Monbaliu, S.; Van Poucke, C.; Detavernier, C.l.; Dumoulin, F.; Van De Velde, M.; Schoeters, E.; Van Dyck, S.; Averkieva, O.; Van Peteghem, C.; De Saeger, S. Occurrence of mycotoxins in feed as analyzed by a multi-mycotoxin LC-MS/MS method. J. Agric. Food Chem. 2010, 58, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Van der Fels-Klerx, H.J.; Focker, M.; De Rijk, T.; Liu, C. Mycotoxins in wheat cultivated in the Netherlands: Results from eight years of field surveys. Mycotoxin Res. 2021, 37, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, O.; Juan, C.; Miere, D.; Loghin, F.; Mañes, J. Occurrence and co-occurrence of Fusarium mycotoxins in wheat grains and wheat flour from Romania. Food Control. 2017, 73, 147–155. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Nawaz, S.; Hetmanski, M.T. Mycotoxins in ingredients of animal feeding stuffs: II. Determination of mycotoxins in maize and maize products. Food Addit. Contam. 1998, 15, 30–55. [Google Scholar] [CrossRef]
- Kos, J.; Janić Hajnal, E.; Malachová, A.; Steiner, D.; Stranska, M.; Krska, R.; Poschmaier, B.; Sulyok, M. Mycotoxins in maize harvested in Republic of Serbia in the period 2012–2015. Part 1: Regulated mycotoxins and its derivatives. Food Chem. 2020, 312, 126034. [Google Scholar] [CrossRef]
- Battilani, P.; Toscano, P.; Van der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B1 contamination in maize in Europe increases due to climate change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef]
- Oliva-Teles, A.; Enes, P.; Peres, H. 8—Replacing fishmeal and fish oil in industrial aquafeeds for carnivorous fish. In Feed and Feeding Practices in Aquaculture; Davis, D.A., Ed.; Woodhead Publishing: Oxford, UK, 2015; pp. 203–233. [Google Scholar]
- Nordrum, S.; Bakke-McKellep, A.M.; Krogdahl, A.; Buddington, R.K. Effects of soybean meal and salinity on intestinal transport of nutrients in Atlantic salmon (Salmo salar L.) and rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2000, 125, 317–335. [Google Scholar] [CrossRef]
- Heikkinen, J.; Vielma, J.; Kemiläinen, O.; Tiirola, M.; Eskelinen, P.; Kiuru, T.; Navia-Paldanius, D.; von Wright, A. Effects of soybean meal based diet on growth performance, gut histopathology and intestinal microbiota of juvenile rainbow trout (Oncorhynchus mykiss). Aquaculture 2006, 261, 259–268. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Penn, M.; Thorsen, J.; Refstie, S.; Bakke, A.M. Important antinutrients in plant feedstuffs for aquaculture: An update on recent findings regarding responses in salmonids. Aquac. Res. 2010, 41, 333–344. [Google Scholar] [CrossRef]
- Urán, P.A.; Gonçalves, A.A.; Taverne-Thiele, J.J.; Schrama, J.W.; Verreth, J.A.J.; Rombout, J.H.W.M. Soybean meal induces intestinal inflammation in common carp (Cyprinus carpio L.). Fish Shellfish Immunol. 2008, 25, 751–760. [Google Scholar] [CrossRef]
- Bonvini, E.; Bonaldo, A.; Mandrioli, L.; Sirri, R.; Dondi, F.; Bianco, C.; Fontanillas, R.; Mongile, F.; Gatta, P.P.; Parma, L. Effects of feeding low fishmeal diets with increasing soybean meal levels on growth, gut histology and plasma biochemistry of sea bass. Animal 2018, 12, 923–930. [Google Scholar] [CrossRef]
- El-Saidy, D.M.S.D.; Gaber, M.M.A. Complete replacement of fish meal by soybean meal with dietary L-lysine supplementation for Nile tilapia Oreochromis niloticus (L.) fingerlings. J. World Aquac. Soc. 2002, 33, 297–306. [Google Scholar] [CrossRef]
- Gutleb, A.C.; Caloni, F.; Giraud, F.; Cortinovis, C.; Pizzo, F.; Hoffmann, L.; Bohn, T.; Pasquali, M. Detection of multiple mycotoxin occurrences in soy animal feed by traditional mycological identification combined with molecular species identification. Toxicol. Rep. 2015, 2, 275–279. [Google Scholar] [CrossRef][Green Version]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Lanubile, A.; Maschietto, V.; De Leonardis, S.; Battilani, P.; Paciolla, C.; Marocco, A. Defense responses to mycotoxin-producing fungi Fusarium proliferatum, F. subglutinans, and Aspergillus flavus in kernels of susceptible and resistant maize genotypes. Mol. Plant. Microbe Interact. 2015, 28, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Giorni, P.; Bertuzzi, T.; Battilani, P. Impact of fungi co-occurrence on mycotoxin contamination in maize during the growing season. Front. Microbiol. 2019, 10, 1265. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, R.; Crisci, A.; Venâncio, A.; Cortiñas Abrahantes, J.; Dorne, J.-L.; Battilani, P.; Toscano, P. Occurrence and co-occurrence of mycotoxins in cereal-based feed and food. Microorganisms 2020, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Mulè, G.; Moretti, A.; Bottalico, A. Toxigenic Fusarium species and mycotoxins associated with maize ear rot in Europe. Eur. J. Plant. Pathol. 2002, 108, 597–609. [Google Scholar] [CrossRef]
- Obradovic, A.; Krnjaja, V.; Nikolic, M.; Delibasic, G.; Filipovic, M.; Stankovic, G.; Stanković, S. Impacts of climatic conditions on aflatoxin B1 and fumonisins contamination of maize kernels and their co-occurrence. Biotechnol. Anim. Husb. 2018, 34, 469–480. [Google Scholar] [CrossRef]
- Gonçalves, R.A.; Naehrer, K.; Santos, G.A. Occurrence of mycotoxins in commercial aquafeeds in Asia and Europe: A real risk to aquaculture? Rev. Aquac. 2018, 10, 263–280. [Google Scholar] [CrossRef]
- Berthiller, F.; Krska, R.; Domig, K.J.; Kneifel, W.; Juge, N.; Schuhmacher, R.; Adam, G. Hydrolytic fate of deoxynivalenol-3-glucoside during digestion. Toxicol. Lett. 2011, 206, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.K.; McMillan, E.G.; Castillo, J.B. Effect of feeding blends of Fusarium mycotoxin-contaminated grains containing deoxynivalenol and fusaric acid on growth and feed consumption of immature swine. J. Anim. Sci. 1997, 75, 2184–2191. [Google Scholar] [CrossRef] [PubMed]
- Iordanov, M.S.; Pribnow, D.; Magun, J.L.; Dinh, T.H.; Pearson, J.A.; Chen, S.L.; Magun, B.E. Ribotoxic stress response: Activation of the stress-activated protein kinase JNK1 by inhibitors of the peptidyl transferase reaction and by sequence-specific RNA damage to the alpha-sarcin/ricin loop in the 28S rRNA. Mol. Cell. Biol. 1997, 17, 3373–3381. [Google Scholar] [CrossRef] [PubMed]
- Plotnikov, A.; Zehorai, E.; Procaccia, S.; Seger, R. The MAPK cascades: Signaling components, nuclear roles and mechanisms of nuclear translocation. Biochim. Biophys. Acta BBA Mol. Cell Res. 2011, 1813, 1619–1633. [Google Scholar] [CrossRef]
- Zhou, H.R.; Islam, Z.; Pestka, J.J. Rapid, sequential activation of mitogen-activated protein kinases and transcription factors precedes proinflammatory cytokine mRNA expression in spleens of mice exposed to the trichothecene vomitoxin. Toxicol Sci 2003, 72, 130–142. [Google Scholar] [CrossRef]
- Wu, Q.H.; Wang, X.; Yang, W.; Nussler, A.K.; Xiong, L.Y.; Kuca, K.; Dohnal, V.; Zhang, X.J.; Yuan, Z.H. Oxidative stress-mediated cytotoxicity and metabolism of T-2 toxin and deoxynivalenol in animals and humans: An update. Arch. Toxicol. 2014, 88, 1309–1326. [Google Scholar] [CrossRef]
- Ren, Z.; He, H.; Zuo, Z.; Xu, Z.; Wei, Z.; Deng, J. ROS: Trichothecenes’ handy weapon? Food Chem. Toxicol. 2020, 142, 111438. [Google Scholar] [CrossRef]
- Mayer, E.; Novak, B.; Springler, A.; Schwartz-Zimmermann, H.E.; Nagl, V.; Reisinger, N.; Hessenberger, S.; Schatzmayr, G. Effects of deoxynivalenol (DON) and its microbial biotransformation product deepoxy-deoxynivalenol (DOM-1) on a trout, pig, mouse, and human cell line. Mycotoxin Res. 2017, 33, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Oswald, I.P. Effect of deoxynivalenol and other type b trichothecenes on the intestine: A review. Toxins 2014, 6, 1615–1643. [Google Scholar] [CrossRef] [PubMed]
- Oswald, I.P.; Marin, D.E.; Bouhet, S.; Pinton, P.; Taranu, I.; Accensi, F. Immunotoxicological risk of mycotoxins for domestic animals. Food Addit. Contam. 2005, 22, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef]
- Akbari, P.; Braber, S.; Varasteh, S.; Alizadeh, A.; Garssen, J.; Fink-Gremmels, J. The intestinal barrier as an emerging target in the toxicological assessment of mycotoxins. Arch. Toxicol. 2017, 91, 1007–1029. [Google Scholar] [CrossRef] [PubMed]
- Liew, W.P.; Mohd-Redzwan, S. Mycotoxin: Its impact on gut health and microbiota. Front. Cell. Infect. Microbiol. 2018, 8, 60. [Google Scholar] [CrossRef] [PubMed]
- Anater, A.; Manyes, L.; Meca, G.; Ferrer, E.; Luciano, F.B.; Pimpão, C.T.; Font, G. Mycotoxins and their consequences in aquaculture: A review. Aquaculture 2016, 451, 1–10. [Google Scholar] [CrossRef]
- Oliveira, M.; Vasconcelos, V. Occurrence of mycotoxins in fish feed and its effects: A review. Toxins 2020, 12, 160. [Google Scholar] [CrossRef]
- Bernhoft, A.; Høgåsen, H.R.; Rosenlund, G.; Ivanova, L.; Berntssen, M.H.G.; Alexander, J.; Eriksen, G.S.; Fæste, C.K. Tissue distribution and elimination of deoxynivalenol and ochratoxin A in dietary-exposed Atlantic salmon (Salmo salar). Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 1211–1224. [Google Scholar] [CrossRef]
- Bernhoft, A.; Høgåsen, H.R.; Rosenlund, G.; Moldal, T.; Grove, S.; Berntssen, M.H.G.; Thoresen, S.I.; Alexander, J. Effects of dietary deoxynivalenol or ochratoxin A on performance and selected health indices in Atlantic salmon (Salmo salar). Food Chem. Toxicol. 2018, 121, 374–386. [Google Scholar] [CrossRef]
- Moldal, T.; Bernhoft, A.; Rosenlund, G.; Kaldhusdal, M.; Koppang, E.O. Dietary deoxynivalenol (don) may impair the epithelial barrier and modulate the cytokine signaling in the intestine of Atlantic salmon (Salmo salar). Toxins 2018, 10, 376. [Google Scholar] [CrossRef]
- Woodward, B.; Young, L.G.; Lun, A.K. Vomitoxin in diets for rainbow trout (Salmo gairdneri). Aquaculture 1983, 35, 93–101. [Google Scholar] [CrossRef]
- Hooft, J.M.; Elmor, A.E.H.I.; Encarnação, P.; Bureau, D.P. Rainbow trout (Oncorhynchus mykiss) is extremely sensitive to the feed-borne Fusarium mycotoxin deoxynivalenol (DON). Aquaculture 2011, 311, 224–232. [Google Scholar] [CrossRef]
- Ryerse, I.A.; Hooft, J.M.; Bureau, D.P.; Hayes, M.A.; Lumsden, J.S. Purified deoxynivalenol or feed restriction reduces mortality in rainbow trout, Oncorhynchus mykiss (Walbaum), with experimental bacterial coldwater disease but biologically relevant concentrations of deoxynivalenol do not impair the growth of Flavobacterium psychrophilum. J. Fish. Dis. 2015, 38, 809–819. [Google Scholar] [PubMed]
- Ryerse, I.A.; Hooft, J.M.; Bureau, D.P.; Anthony Hayes, M.; Lumsden, J.S. Diets containing corn naturally contaminated with deoxynivalenol reduces the susceptibility of rainbow trout (Oncorhynchus mykiss) to experimental Flavobacterium psychrophilum infection. Aquac. Res. 2016, 47, 787–796. [Google Scholar] [CrossRef]
- Hooft, J.M.; Bureau, D.P. Evaluation of the efficacy of a commercial feed additive against the adverse effects of feed-borne deoxynivalenol (DON) on the performance of rainbow trout (Oncorhynchus mykiss). Aquaculture 2017, 473, 237–245. [Google Scholar] [CrossRef]
- Hooft, J.M.; Ferreira, C.; Lumsden, J.S.; Sulyok, M.; Krska, R.; Bureau, D.P. The effects of naturally occurring or purified deoxynivalenol (DON) on growth performance, nutrient utilization and histopathology of rainbow trout (Oncorhynchus mykiss). Aquaculture 2019, 505, 319–332. [Google Scholar] [CrossRef]
- Hooft, J.M.; Wu, P.; Powell, C.D.; Lou, Y.; Squires, E.J.; Cant, J.P.; Sulyok, M.; Krska, R.; Bureau, D.P. A comparative investigation of the effects of feed-borne deoxynivalenol (DON) on growth performance, nutrient utilization and metabolism of detoxification in rainbow trout (Oncorhynchus mykiss) and Nile tilapia (Oreochromis niloticus) fed diets containing different levels of digestible carbohydrates. Aquaculture 2019, 505, 306–318. [Google Scholar]
- Gonçalves, R.A.; Menanteau-Ledouble, S.; Schöller, M.; Eder, A.; Schmidt-Posthaus, H.; Mackenzie, S.; El-Matbouli, M. Effects of deoxynivalenol exposure time and contamination levels on rainbow trout. J. World Aquac. Soc. 2019, 50, 137–154. [Google Scholar] [CrossRef]
- Gonçalves, R.A.; Navarro-Guillén, C.; Gilannejad, N.; Dias, J.; Schatzmayr, D.; Bichl, G.; Czabany, T.; Moyano, F.J.; Rema, P.; Yúfera, M.; et al. Impact of deoxynivalenol on rainbow trout: Growth performance, digestibility, key gene expression regulation and metabolism. Aquaculture 2018, 490, 362–372. [Google Scholar] [CrossRef]
- Matejova, I.; Modra, H.; Blahova, J.; Franc, A.; Fictum, P.; Sevcikova, M.; Svobodova, Z. The effect of mycotoxin deoxynivalenol on haematological and biochemical indicators and histopathological changes in rainbow trout (Oncorhynchus mykiss). BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Matejova, I.; Vicenova, M.; Vojtek, L.; Kudlackova, H.; Nedbalcova, K.; Faldyna, M.; Sisperova, E.; Modra, H.; Svobodova, Z. Effect of the mycotoxin deoxynivalenol on the immune responses of rainbow trout (Oncorhynchus mykiss). Vet. Med. 2015, 60, 515–521. [Google Scholar] [CrossRef]
- Šišperová, E.; Modrá, H.; Ziková, A.; Kloas, W.; Blahová, J.; Matejová, I.; Živná, D.; Svobodová, Z. The effect of mycotoxin deoxynivalenol (DON) on the oxidative stress markers in rainbow trout (Oncorhynchus mykiss, Walbaum 1792). J. Appl. Ichthyol. 2015, 31, 855–861. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in Action; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Pietsch, C.; Schulz, C.; Rovira, P.; Kloas, W.; Burkhardt-Holm, P. Organ damage and hepatic lipid accumulation in carp (Cyprinus carpio L.) after feed-borne exposure to the mycotoxin, deoxynivalenol (DON). Toxins 2014, 6, 756–778. [Google Scholar] [CrossRef]
- Pietsch, C.; Michel, C.; Kersten, S.; Valenta, H.; Dänicke, S.; Schulz, C.; Kloas, W.; Burkhardt-Holm, P. In vivo effects of deoxynivalenol (DON) on innate immune responses of carp (Cyprinus carpio L.). Food Chem. Toxicol. 2014, 68, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, C.; Katzenback, B.A.; Garcia-Garcia, E.; Schulz, C.; Belosevic, M.; Burkhardt-Holm, P. Acute and subchronic effects on immune responses of carp (Cyprinus carpio L.) after exposure to deoxynivalenol (DON) in feed. Mycotoxin Res. 2015, 31, 151–164. [Google Scholar] [CrossRef]
- Pietsch, C.; Burkhardt-Holm, P. Feed-borne exposure to deoxynivalenol leads to acute and chronic effects on liver enzymes and histology in carp. World Mycotoxin J. 2015, 8, 619–627. [Google Scholar] [CrossRef]
- Tiemann, U.; Brüssow, K.P.; Küchenmeister, U.; Jonas, L.; Kohlschein, P.; Pöhland, R.; Dänicke, S. Influence of diets with cereal grains contaminated by graded levels of two Fusarium toxins on selected enzymatic and histological parameters of liver in gilts. Food Chem. Toxicol. 2006, 44, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Mechanisms of deoxynivalenol-induced gene expression and apoptosis. Food Addit. Contaminants. Part A Chem. Anal. Control. Expo. Risk Assess. 2008, 25, 1128–1140. [Google Scholar] [CrossRef]
- Pelyhe, C.; Kovesi, B.; Zandoki, E.; Kovacs, B.; Szabo-Fodor, J.; Mezes, M.; Balogh, K. Effect of 4-week feeding of deoxynivalenol- or T-2-toxin-contaminated diet on lipid peroxidation and glutathione redox system in the hepatopancreas of common carp (Cyprinus carpio L.). Mycotoxin Res. 2016, 32, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Kovesi, B.; Pelyhe, C.; Zandoki, E.; Mezes, M.; Balogh, K. Combined effects of aflatoxin B1 and deoxynivalenol on the expression of glutathione redox system regulatory genes in common carp. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1531–1539. [Google Scholar] [CrossRef]
- Huang, C.; Wu, P.; Jiang, W.D.; Liu, Y.; Zeng, Y.Y.; Jiang, J.; Kuang, S.Y.; Tang, L.; Zhang, Y.A.; Zhou, X.Q.; et al. Deoxynivalenol decreased the growth performance and impaired intestinal physical barrier in juvenile grass carp (Ctenopharyngodon idella). Fish. Shellfish Immunol. 2018, 80, 376–391. [Google Scholar] [CrossRef]
- Huang, C.; Feng, L.; Jiang, W.D.; Wu, P.; Liu, Y.; Zeng, Y.Y.; Jiang, J.; Kuang, S.Y.; Tang, L.; Zhou, X.Q. Deoxynivalenol decreased intestinal immune function related to NF-κB and TOR signalling in juvenile grass carp (Ctenopharyngodon idella). Fish. Shellfish Immunol. 2019, 84, 470–484. [Google Scholar] [CrossRef]
- Huang, C.; Feng, L.; Liu, X.A.; Jiang, W.D.; Wu, P.; Liu, Y.; Jiang, J.; Kuang, S.Y.; Tang, L.; Zhou, X.Q. The toxic effects and potential mechanisms of deoxynivalenol on the structural integrity of fish gill: Oxidative damage, apoptosis and tight junctions disruption. Toxicon 2020, 174, 32–42. [Google Scholar] [CrossRef]
- López Nadal, A.; Ikeda-Ohtsubo, W.; Sipkema, D.; Peggs, D.; McGurk, C.; Forlenza, M.; Wiegertjes, G.F.; Brugman, S. Feed, microbiota, and gut immunity: Using the zebrafish model to understand fish health. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Juan-García, A.; Bind, M.-A.; Engert, F. Larval zebrafish as an in vitro model for evaluating toxicological effects of mycotoxins. Ecotoxicol. Environ. Saf. 2020, 202, 110909. [Google Scholar] [CrossRef]
- Sanden, M.; Jørgensen, S.; Hemre, G.I.; Ørnsrud, R.; Sissener, N.H. Zebrafish (Danio rerio) as a model for investigating dietary toxic effects of deoxynivalenol contamination in aquaculture feeds. Food Chem. Toxicol. 2012, 50, 4441–4448. [Google Scholar] [CrossRef]
- Khezri, A.; Herranz-Jusdado, J.G.; Ropstad, E.; Fraser, T.W.K. Mycotoxins induce developmental toxicity and behavioural aberrations in zebrafish larvae. Environ. Pollut. 2018, 242, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Nobrega, R.O.; Banze, J.F.; Batista, R.O.; Fracalossi, D.M. Improving winter production of Nile tilapia: What can be done? Aquac. Rep. 2020, 18, 100453. [Google Scholar] [CrossRef]
- Tola, S.; Bureau, D.P.; Hooft, J.M.; Beamish, F.W.; Sulyok, M.; Krska, R.; Encarnação, P.; Petkam, R. Effects of wheat naturally contaminated with Fusarium mycotoxins on growth performance and selected health indices of red tilapia (Oreochromis niloticus × O. mossambicus). Toxins 2015, 7, 1929–1944. [Google Scholar] [CrossRef]
- Manning, B.B.; Abbas, H.K.; Wise, D.J.; Greenway, T. The effect of feeding diets containing deoxynivalenol contaminated corn on channel catfish (Ictalurus punctatus) challenged with Edwardsiella ictaluri. Aquac. Res. 2014, 45, 1782–1786. [Google Scholar]
- Guan, S.; He, J.; Young, J.C.; Zhu, H.; Li, X.Z.; Ji, C.; Zhou, T. Transformation of trichothecene mycotoxins by microorganisms from fish digesta. Aquaculture 2009, 290, 290–295. [Google Scholar] [CrossRef]
- McCormick, S.P. Microbial detoxification of mycotoxins. J. Chem. Ecol. 2013, 39, 907–918. [Google Scholar] [CrossRef]
- Payros, D.; Alassane-Kpembi, I.; Pierron, A.; Loiseau, N.; Pinton, P.; Oswald, I.P. Toxicology of deoxynivalenol and its acetylated and modified forms. Arch. Toxicol. 2016, 90, 2931–2957. [Google Scholar] [CrossRef]
- Maul, R.; Warth, B.; Kant, J.-S.; Schebb, N.H.; Krska, R.; Koch, M.; Sulyok, M. Investigation of the hepatic glucuronidation pattern of the Fusarium mycotoxin deoxynivalenol in various species. Chem. Res. Toxicol. 2012, 25, 2715–2717. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.; Engrola, S.; Aragão, C.; Mackenzie, S.; Bichl, G.; Czabany, T.; Schatzmayr, D. Fate of [3H]-deoxynivalenol in rainbow trout (Oncorhynchus mykiss) juveniles: Tissue distribution and excretion. J. Aquac. Res. Dev. 2018, 9. [Google Scholar] [CrossRef]
- Nácher-Mestre, J.; Serrano, R.; Beltrán, E.; Pérez-Sánchez, J.; Silva, J.; Karalazos, V.; Hernández, F.; Berntssen, M.H.G. Occurrence and potential transfer of mycotoxins in gilthead sea bream and Atlantic salmon by use of novel alternative feed ingredients. Chemosphere 2015, 128, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, C. Risk assessment for mycotoxin contamination in fish feeds in Europe. Mycotoxin Res. 2019, 36, 41–62. [Google Scholar] [CrossRef]
- Andretta, I.; Kipper da Silva, M.; Hauschild, L.; Lehnen, C.; Remus, A.; Melchior, R. Meta-analysis of individual and combined effects of mycotoxins on growing pigs. Sci. Agric. 2016, 73, 328–331. [Google Scholar] [CrossRef]
- Bullerman, L.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar] [CrossRef]
- Wu, Q.; Lohrey, L.; Cramer, B.; Yuan, Z.; Humpf, H.-U. Impact of physicochemical parameters on the decomposition of deoxynivalenol during extrusion cooking of wheat grits. J. Agric. Food Chem. 2011, 59, 12480–12485. [Google Scholar] [CrossRef]
- Zhou, H.; George, S.; Li, C.; Gurusamy, S.; Sun, X.; Gong, Z.; Qian, H. Combined toxicity of prevalent mycotoxins studied in fish cell line and zebrafish larvae revealed that type of interactions is dose-dependent. Aquat. Toxicol. 2017, 193, 60–71. [Google Scholar] [CrossRef]
- Andretta, I.; Kipper, M.; Lehnen, C.R.; Hauschild, L.; Vale, M.M.; Lovatto, P.A. Meta-analytical study of productive and nutritional interactions of mycotoxins in broilers. Poult. Sci. 2011, 90, 1934–1940. [Google Scholar] [CrossRef]
- Pastorelli, H.; van Milgen, J.; Lovatto, P.; Montagne, L. Meta-analysis of feed intake and growth responses of growing pigs after a sanitary challenge. Animal 2012, 6, 952–961. [Google Scholar] [CrossRef]
- Kipper, M.; Andretta, I.; Ribeiro, A.M.L.; Pires, P.G.d.S.; Franceschina, C.S.; Cardinal, K.M.; Moraes, P.d.O.; Schroeder, B. Assessing the implications of mycotoxins on productive efficiency of broilers and growing pigs. Sci. Agric. 2020, 77. [Google Scholar] [CrossRef]
- Akande, K.E.; Abubakar, M.M.; Adegbola, T.A.; Bogoro, S.E. Nutritional and health implications of mycotoxins in animal feeds: A review. Pak. J. Nutr. 2006, 5, 398–403. [Google Scholar]
- Terciolo, C.; Maresca, M.; Pinton, P.; Oswald, I.P. Review article: Role of satiety hormones in anorexia induction by trichothecene mycotoxins. Food Chem. Toxicol. 2018, 121, 701–714. [Google Scholar] [CrossRef] [PubMed]
- Hosmer, D.W., Jr.; Lemeshow, S.; Sturdivant, R.X. Applied Logistic Regression; John Wiley & Sons: Hoboken, NJ, USA, 2013; Volume 398. [Google Scholar]
- Whitaker, T.B. Sampling foods for mycotoxins. Food Addit. Contam. 2006, 23, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Kövesi, B.; Kulcsár, S.; Zándoki, E.; Szabó-Fodor, J.; Mézes, M.; Balogh, K.; Ancsin, Z.; Pelyhe, C. Short-term effects of deoxynivalenol, T-2 toxin, fumonisin B1 or ochratoxin on lipid peroxidation and glutathione redox system and its regulatory genes in common carp (Cyprinus carpio L.) liver. Fish. Physiol. Biochem. 2020, 46, 1921–1932. [Google Scholar] [CrossRef]
- Andretta, I.; Kipper da Silva, M.; Lehnen, C.; Hauschild, L.; Vale, M.; Lovatto, P.A. Meta-analytical study of productive and nutritional interactions of mycotoxins in growing pigs. Anim. Int. J. Anim. Biosci. 2012, 6, 1476–1482. [Google Scholar] [CrossRef]
- Remus, A.; Hauschild, L.; Andretta, I.; Kipper, M.; Lehnen, C.R.; Sakomura, N.K. A meta-analysis of the feed intake and growth performance of broiler chickens challenged by bacteria. Poult. Sci. 2014, 93, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
| Wheat | Corn | Soybean Meal | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mycotoxin | Occurrence 2 (%) | Mean (µg/kg) | Maximum (µg/kg) | Occurrence 2 (%) | Mean (µg/kg) | Maximum (µg/kg) | Occurrence 2 (%) | Mean (µg/kg) | Maximum (µg/kg) |
| 15-acetyl-deoxynivalenol (15-AcDON) | 4 | 51 | 217 | 20 | 133 | 1667 | 1 | 13 | 13 |
| 3-acetyl-deoxynivalenol (3-AcDON) | 7 | 28 | 101 | 14 | 46 | 406 | . | . | . |
| Aflatoxin B1 (AFB1) | 2 | 2 | 6 | 4 | 12 | 148 | 6 | 1 | 2 |
| Aflatoxin B2 (AFB2) | 3 | 9 | 51 | 4 | 19 | 92 | 2 | 4 | 5 |
| Aflatoxin G1 (AFG1) | 1 | 3 | 14 | 2 | 12 | 67 | 2 | 19 | 51 |
| Aflatoxin G2 (AFG2) | 3 | 3 | 14 | 8 | 7 | 60 | 1 | 2 | 2 |
| Alternariol | 12 | 30 | 247 | 3 | 20 | 110 | 9 | 27 | 109 |
| Beauvericin (BEA) | 1 | 5 | 14 | 5 | 56 | 552 | 4 | 11 | 27 |
| Citreoviridin | 0.1 | 1172 | 1172 | 0.1 | 33 | 33 | . | . | . |
| Citrinin | 1 | 9 | 17 | 0.3 | 10 | 18 | 5 | 84 | 224 |
| Cyclopiazonic acid | 1 | 19 | 44 | 2 | 16 | 73 | 2 | 19 | 30 |
| Deoxynivalenol (DON) | 41 | 470 | 8872 | 47 | 826 | 10,020 | 11 | 85 | 543 |
| Diacetoxyscirpenol (DAS) | 1 | 39 | 81 | 3 | 26 | 187 | . | . | . |
| DON-3-Glucoside (DON3Glc) | 7 | 137 | 1072 | 7 | 202 | 851 | 4 | 59 | 62 |
| Ergocristin(in)e | 0.4 | 95 | 189 | . | . | . | . | . | . |
| Ergocryptin(in)e | 0.5 | 10 | 25 | . | . | . | . | . | . |
| Ergometrin(in)e | 4 | 23 | 361 | 2 | 8 | 34 | 5 | 4 | 9 |
| Ergosin(in)e | 0.2 | 35 | 46 | . | . | . | . | . | . |
| Ergotamin(in)e | 3 | 119 | 1891 | 4 | 7 | 102 | 4 | 4 | 6 |
| Fumonisin B1 (FB1) | 27 | 561 | 9122 | 70 | 2234 | 49,347 | 26 | 371 | 1462 |
| Fumonisin B2 (FB2) | 14 | 59 | 590 | 54 | 262 | 7944 | 19 | 83 | 424 |
| Fumonisin B3 (FB3) | 4 | 67 | 417 | 41 | 189 | 3203 | 6 | 50 | 159 |
| Fusarenon X (FX) | 23 | 91 | 1267 | 10 | 96 | 604 | 12 | 65 | 196 |
| Fusaric acid (FA) | 5 | 54 | 337 | 67 | 266 | 4327 | 42 | 89 | 754 |
| Gliotoxin | 2 | 292 | 811 | 1 | 247 | 879 | . | . | . |
| HT-2 toxin | 4 | 44 | 456 | 9 | 190 | 2643 | 4 | 155 | 561 |
| Lysergol | 3 | 4 | 8 | 2 | 2 | 6 | 8 | 3 | 9 |
| Methylergonovine | 6 | 3 | 11 | 5 | 5 | 30 | 7 | 3 | 14 |
| Moniliformin (MON) | 1 | 14 | 24 | 10 | 171 | 1103 | . | . | . |
| Mycophenolic acid | 2 | 39 | 228 | 4 | 79 | 478 | 1 | 297 | 297 |
| Neosolaniol (NEO) | 6 | 18 | 79 | 8 | 48 | 589 | 8 | 26 | 158 |
| Nivalenol (NIV) | 1 | 275 | 453 | 4 | 661 | 1660 | 3 | 231 | 291 |
| Ochratoxin A (OTA) | 11 | 6 | 45 | 9 | 24 | 648 | 12 | 3 | 7 |
| Ochratoxin B | 5 | 3 | 9 | 6 | 4 | 53 | 6 | 3 | 6 |
| Patulin | 1 | 128 | 183 | 1 | 102 | 183 | 2 | 101 | 106 |
| Penicillic acid | . | . | . | 3 | 297 | 2156 | . | . | . |
| Roquefortine C | 5 | 3 | 26 | 10 | 4 | 71 | 10 | 2 | 5 |
| Sterigmatocystin | 6 | 4 | 21 | 8 | 2 | 5 | 12 | 2 | 4 |
| T-2 toxin | 7 | 46 | 551 | 14 | 81 | 757 | 23 | 49 | 348 |
| Verruculogen | 8 | 15 | 367 | 5 | 65 | 802 | 3 | 10 | 17 |
| Wortmannin | 4 | 39 | 474 | 2 | 124 | 508 | 1 | 25 | 28 |
| Zearalanone | 0.2 | 463 | 606 | 1 | 137 | 555 | . | . | . |
| Zearalenone (ZEN) | 5 | 64 | 738 | 16 | 165 | 1282 | 14 | 81 | 354 |
| “Toxin X” | Category | Frequency 1 | % DON 2 | Odds Ratio (OR) | 95% CI | Wald p-Value 3 | |
|---|---|---|---|---|---|---|---|
| n | % | ||||||
| DON-3-Glucoside (DON3Glc) | present | 53 | 7.3 | 98.1 | 69.6 | 9.6–505.9 | *** |
| absent | 672 | 92.7 | 42.7 | Ref. | |||
| 15-acetyl-deoxynivalenol (15-AcDON) | present | 142 | 19.6 | 96.5 | 51.7 | 20.8–128.2 | *** |
| absent | 583 | 80.41 | 34.7 | Ref. | |||
| Nivalenol (NIV) | present | 30 | 4.1 | 96.7 | 36.0 | 4.9–265.9 | *** |
| absent | 695 | 95.9 | 44.6 | Ref. | |||
| 3-acetyl-deoxynivalenol (3-AcDON) | present | 98 | 13.5 | 91.8 | 17.1 | 8.1–35.8 | *** |
| absent | 627 | 86.5 | 39.7 | Ref. | |||
| Zearalenone (ZEN) | present | 116 | 16.0 | 90.5 | 15.3 | 8.0–29.1 | *** |
| absent | 609 | 84.0 | 38.4 | Ref. | |||
| Sterigmatocystin | present | 57 | 7.9 | 68.4 | 2.7 | 1.5–4.7 | *** |
| absent | 668 | 92.1 | 44.9 | Ref. | |||
| Roquefortine C | present | 72 | 9.9 | 68.1 | 2.7 | 1.6–4.5 | *** |
| absent | 653 | 90.1 | 44.4 | Ref. | |||
| Alternariol | present | 20 | 2.8 | 70.0 | 2.7 | 1.04–7.2 | * |
| absent | 705 | 97.2 | 46.1 | Ref. | |||
| HT-2 Toxin | present | 67 | 9.2 | 64.2 | 2.2 | 1.3–3.7 | ** |
| absent | 658 | 90.8 | 45.0 | Ref. | |||
| T-2 toxin | present | 103 | 14.2 | 62.1 | 2.1 | 1.3–3.2 | *** |
| absent | 622 | 85.8 | 44.2 | Ref. | |||
| Fusarenon X (FX) | present | 70 | 9.7 | 60.0 | 1.8 | 1.1–3.0 | * |
| absent | 655 | 90.3 | 45.3 | Ref. | |||
| Neosolaniol (NEO) | present | 57 | 7.9 | 59.7 | 1.8 | 1.01–3.1 | * |
| absent | 668 | 92.1 | 45.7 | Ref. | |||
| Fumonisin B1 (FB1) | present | 505 | 69.7 | 50.3 | 1.6 | 1.2–2.2 | ** |
| absent | 220 | 30.3 | 38.6 | Ref. | |||
| Fumonisin B3 (FB3) | present | 296 | 40.8 | 53.0 | 1.5 | 1.1–2.1 | ** |
| absent | 429 | 59.2 | 42.4 | Ref. | |||
| Beauvericin (BEA) | present | 36 | 5.0 | 25.0 | 0.36 | 0.17–0.78 | ** |
| absent | 689 | 95.0 | 47.9 | Ref. | |||
| Moniliformin (MON) | present | 70 | 9.7 | 31.4 | 0.48 | 0.29–0.83 | ** |
| absent | 655 | 90.3 | 48.4 | Ref. | |||
| Aflatoxin B2 (AFB2) | present | 29 | 4.0 | 31.0 | 0.49 | 0.22–1.1 | # |
| absent | 696 | 96.0 | 47.4 | Ref. | |||
| Gliotoxin | present | 5 | 0.7 | 80.0 | 4.6 | 0.51–41.3 | NS |
| absent | 720 | 99.3 | 46.5 | Ref. | |||
| Zearalanone | present | 8 | 1.1 | 75.0 | 3.5 | 0.69–17.3 | NS |
| absent | 717 | 98.9 | 46.4 | Ref. | |||
| Lysergol | present | 12 | 1.7 | 66.7 | 2.3 | 0.69–7.7 | NS |
| absent | 713 | 98.3 | 46.4 | Ref. | |||
| Diacetoxyscirpenol (DAS) | present | 25 | 3.5 | 60.0 | 1.7 | 0.77–3.9 | NS |
| absent | 700 | 96.6 | 46.3 | Ref. | |||
| Methylergonovine | present | 35 | 4.8 | 57.1 | 1.6 | 0.78–3.1 | NS |
| absent | 690 | 95.2 | 46.2 | Ref. | |||
| Ochratoxin A (OTA) | present | 62 | 8.6 | 56.5 | 1.5 | 0.91–2.6 | NS |
| absent | 663 | 91.5 | 45.9 | Ref. | |||
| Ergotamin(in)e | present | 30 | 4.1 | 56.7 | 1.5 | 0.73–3.2 | NS |
| absent | 695 | 95.9 | 46.3 | Ref. | |||
| Verruculogen | present | 37 | 5.1 | 54.1 | 1.4 | 0.70–2.6 | NS |
| absent | 688 | 94.9 | 46.4 | Ref. | |||
| Aflatoxin G1 (AFG1) | present | 13 | 1.8 | 53.9 | 1.3 | 0.45–4.0 | NS |
| absent | 712 | 98.2 | 46.6 | Ref. | |||
| Aflatoxin B1 (AFB1) | present | 27 | 3.7 | 51.9 | 1.2 | 0.57–2.7 | NS |
| absent | 698 | 96.3 | 46.6 | Ref. | |||
| Aflatoxin G2 (AFG2) | present | 61 | 8.4 | 50.8 | 1.2 | 0.71–2.0 | NS |
| absent | 664 | 91.6 | 46.4 | Ref. | |||
| Fusaric acid (FA) | present | 485 | 66.9 | 48.0 | 1.2 | 0.86–1.6 | NS |
| absent | 240 | 33.1 | 44.2 | Ref. | |||
| Ochratoxin B | present | 44 | 6.1 | 50.0 | 1.1 | 0.62–2.1 | NS |
| absent | 681 | 93.9 | 46.6 | Ref. | |||
| Ergometrin(in)e | present | 12 | 1.7 | 50.0 | 1.1 | 0.37–3.6 | NS |
| absent | 713 | 98.3 | 46.7 | Ref. | |||
| Citreoviridin | present | 1 | 0.1 | 100.0 | 1.1 | 0.06-∞ | NS 4 |
| absent | 724 | 99.9 | 46.8 | Ref. | |||
| Mycophenolic acid | present | 31 | 4.3 | 48.4 | 1.1 | 0.52–2.2 | NS |
| absent | 694 | 95.7 | 46.7 | Ref. | |||
| Fumonisin B2 (FB2) | present | 394 | 54.3 | 47.0 | 1.0 | 0.76–1.4 | NS |
| absent | 331 | 45.7 | 46.5 | Ref. | |||
| Wortmannin | present | 17 | 2.3 | 47.1 | 1.0 | 0.39–2.7 | NS |
| absent | 708 | 97.7 | 46.8 | Ref. | |||
| Patulin | present | 5 | 0.7 | 20.0 | 0.28 | 0.03–2.5 | NS |
| absent | 720 | 99.3 | 46.7 | Ref. | |||
| Citrinin | present | 2 | 0.3 | 0.0 | 0.47 | 0.0–4.0 | NS 4 |
| absent | 723 | 99.7 | 46.9 | Ref. | |||
| Cyclopiazonic acid | present | 16 | 2.2 | 43.8 | 0.88 | 0.33–2.4 | NS |
| absent | 709 | 97.8 | 46.8 | Ref. | |||
| Penicillic acid | present | 24 | 3.3 | 45.8 | 0.96 | 0.43–2.2 | NS |
| absent | 701 | 96.7 | 46.8 | Ref. | |||
| Mycotoxin | Occurrence (%) | Mean (µg/kg) | Maximum (µg/kg) |
|---|---|---|---|
| 15-acetyl-deoxynivalenol (15-AcDON) | 5 | 82 | 127 |
| Aflatoxin B1 (AFB1) | 5 | 2 | 4 |
| Aflatoxin G2 (AFG2) | 2 | 6 | 6 |
| Alternariol | 14 | 21 | 51 |
| Deoxynivalenol (DON) | 48 | 136 | 469 |
| Diacetoxyscirpenol (DAS) | 2 | 9 | 9 |
| DON-3-Glucoside (DON3Glc) | 18 | 98 | 155 |
| Ergometrin(in)e | 7 | 4 | 5 |
| Ergotamin(in)e | 20 | 38 | 125 |
| Fumonisin B1 (FB1) | 36 | 628 | 4923 |
| Fumonisin B2 (FB2) | 27 | 120 | 778 |
| Fumonisin B3 (FB3) | 11 | 86 | 223 |
| Fusarenon X (FX) | 2 | 28 | 28 |
| Fusaric acid (FA) | 55 | 41 | 265 |
| Gliotoxin | 2 | 92 | 92 |
| HT-2 toxin | 2 | 43 | 43 |
| Lysergol | 9 | 10 | 23 |
| Ochratoxin A (OTA) | 2 | 3 | 3 |
| Penicillic acid | 11 | 41 | 58 |
| Sterigmatocystin | 2 | 1 | 1 |
| T-2 toxin | 2 | 46 | 46 |
| Verruculogen | 9 | 560 | 636 |
| Wortmannin | 2 | 20 | 20 |
| Zearalenone (ZEN) | 2 | 348 | 348 |
| All Fish Species | Control | Challenged |
| Initial body weight (g) | 30.90 ± 6.74 | 27.85 ± 3.96 |
| DON dose (µg/kg) | 70.61 ± 22.03 | 2575.04 ± 383.32 |
| Feed intake (g/fish) | 1.30 ± 0.17 | 0.98 ± 0.08 |
| Growth (g/day) | 1.27 ± 0.19 | 0.86 ± 0.08 |
| Rainbow Trout | Control | Challenged |
| Initial body weight (g) | 29.25 ± 9.95 | 29.41 ± 5.70 |
| DON dose (µg/kg) | 97.40 ± 36.19 | 2994.52 ± 581.40 |
| Feed intake (g/fish) | 1.51 ± 0.30 | 1.05 ± 0.14 |
| Growth (g/day) | 1.54 ± 0.33 | 1.00 ± 0.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koletsi, P.; Schrama, J.W.; Graat, E.A.M.; Wiegertjes, G.F.; Lyons, P.; Pietsch, C. The Occurrence of Mycotoxins in Raw Materials and Fish Feeds in Europe and the Potential Effects of Deoxynivalenol (DON) on the Health and Growth of Farmed Fish Species—A Review. Toxins 2021, 13, 403. https://doi.org/10.3390/toxins13060403
Koletsi P, Schrama JW, Graat EAM, Wiegertjes GF, Lyons P, Pietsch C. The Occurrence of Mycotoxins in Raw Materials and Fish Feeds in Europe and the Potential Effects of Deoxynivalenol (DON) on the Health and Growth of Farmed Fish Species—A Review. Toxins. 2021; 13(6):403. https://doi.org/10.3390/toxins13060403
Chicago/Turabian StyleKoletsi, Paraskevi, Johan W. Schrama, Elisabeth A. M. Graat, Geert F. Wiegertjes, Philip Lyons, and Constanze Pietsch. 2021. "The Occurrence of Mycotoxins in Raw Materials and Fish Feeds in Europe and the Potential Effects of Deoxynivalenol (DON) on the Health and Growth of Farmed Fish Species—A Review" Toxins 13, no. 6: 403. https://doi.org/10.3390/toxins13060403
APA StyleKoletsi, P., Schrama, J. W., Graat, E. A. M., Wiegertjes, G. F., Lyons, P., & Pietsch, C. (2021). The Occurrence of Mycotoxins in Raw Materials and Fish Feeds in Europe and the Potential Effects of Deoxynivalenol (DON) on the Health and Growth of Farmed Fish Species—A Review. Toxins, 13(6), 403. https://doi.org/10.3390/toxins13060403







