Medium Cut-Off Dialysis Membrane and Dietary Fiber Effects on Inflammation and Protein-Bound Uremic Toxins: A Systematic Review and Protocol for an Interventional Study
Abstract
1. Introduction
2. Results
2.1. Performance of Medium Cut-Off Dialyzers in the Elimination of Middle Molecules and Protein-Bound Uremic Toxins Compared to Hemodiafiltration or High-Flux Hemodialysis
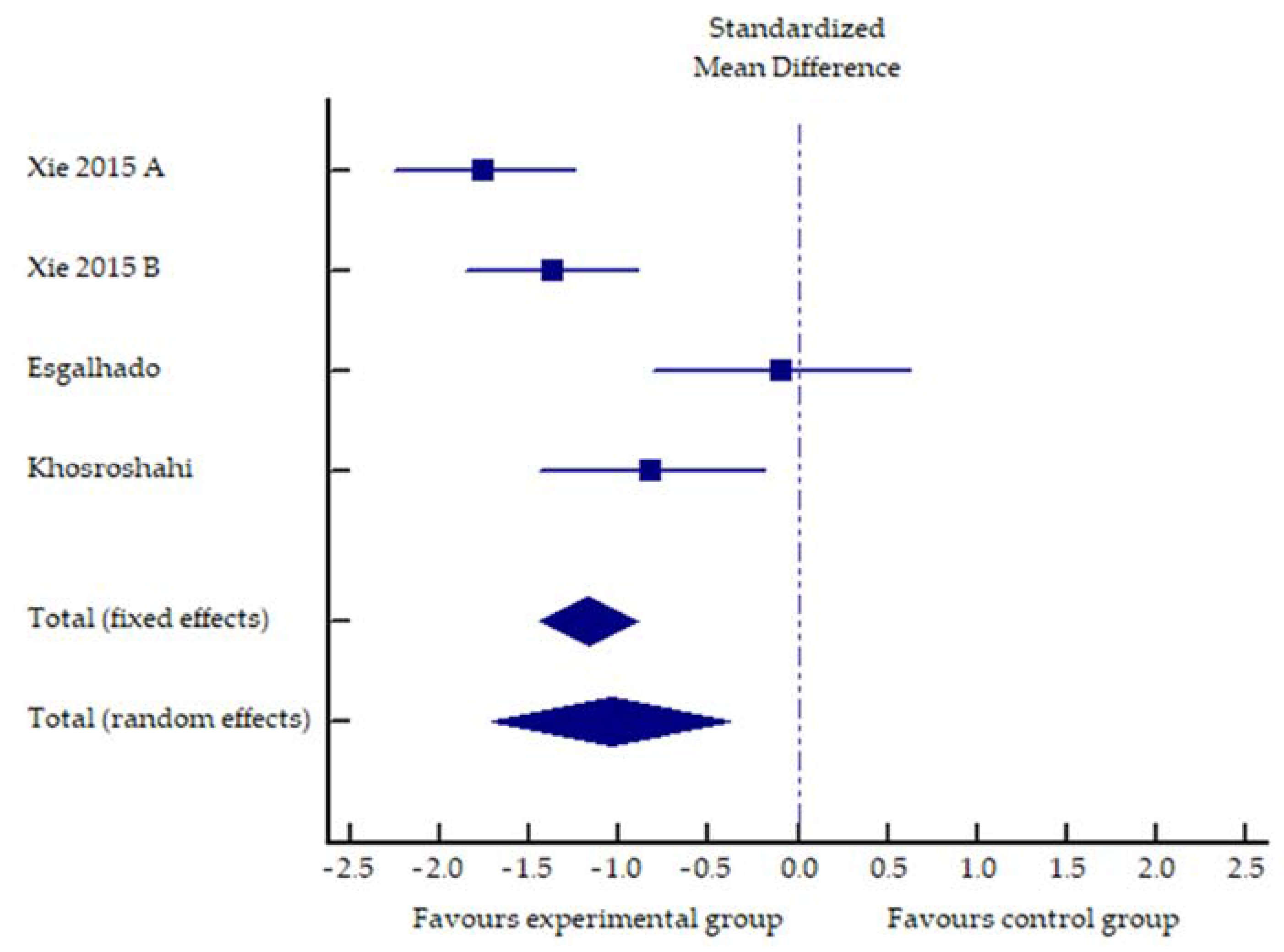
2.2. Impact of Increased Dietary Fiber Supplementation on Serum Concentration of PCS, IS and Inflammatory Markers
2.3. Quality Assessment
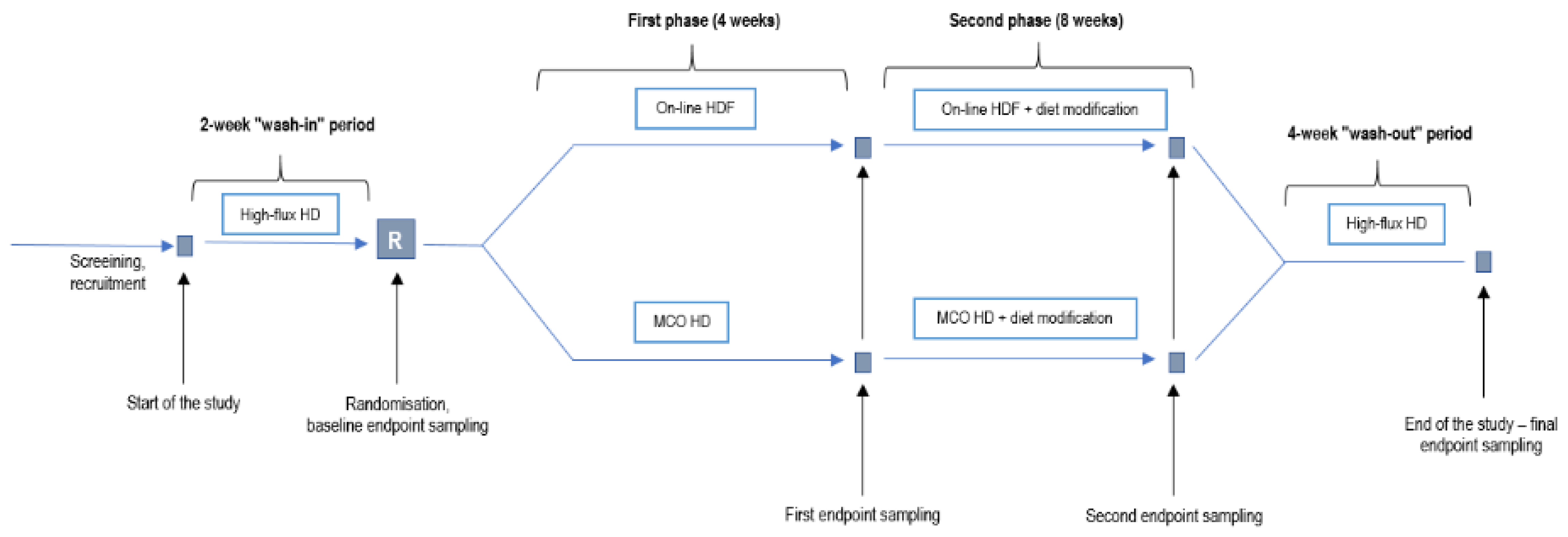
3. Discussion and an Interventional Trial Proposal
- Experimental group: 4 weeks of dialysis with medium cut-off (Theranova) membrane (first phase), and dialysis for 8 weeks with the same membrane and increased fiber and sodium propionate intake (second phase);
- Control group: 4 weeks of dialysis with a high-flux membrane using online hemodiafiltration (first phase) and 8 weeks of high-flux membrane hemodiafiltration and increased fiber and sodium propionate intake (second phase).
Sample Size
4. Conclusions
5. Materials and Methods
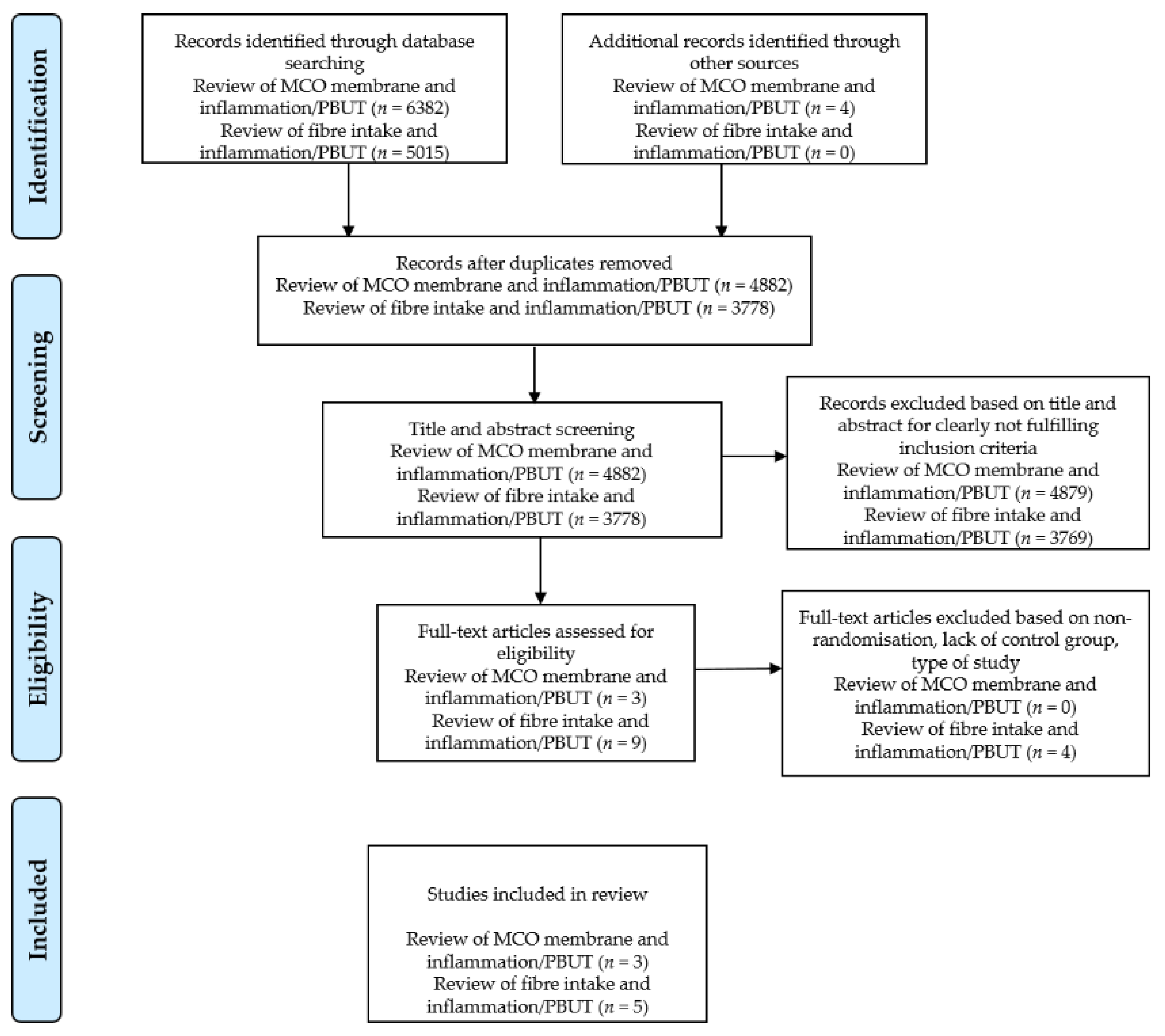
5.1. Literature Search Strategy
5.2. Inclusion and Exclusion Criteria
5.3. Quality Assessment
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vanholder, R.; De Smet, R.; Glorieux, G.; Argilés, A.; Baurmeister, U.; Brunet, P.; Clark, W.; Cohen, G.; De Deyn, P.P.; Deppisch, R.; et al. Review on uremic toxins: Classification, concentration, and interindividual variability. Kidney Int. 2003, 63, 1934–1943. [Google Scholar] [CrossRef]
- Vanholder, R.; Pletinck, A.; Schepers, E.; Glorieux, G. Biochemical and Clinical Impact of Organic Uremic Retention Solutes: A Comprehensive Update. Toxins 2018, 10, 33. [Google Scholar] [CrossRef]
- Graboski, A.L.; Redinbo, M.R. Gut-Derived Protein-Bound Uremic Toxins. Toxins 2020, 12, 590. [Google Scholar] [CrossRef]
- Eloot, S.; Van Biesen, W.; Glorieux, G.; Neirynck, N.; Dhondt, A.; Vanholder, R. Does the adequacy parameter Kt/V(urea) reflect uremic toxin concentrations in hemodialysis patients? PLoS ONE 2013, 8, e76838. [Google Scholar] [CrossRef]
- Maduell, F.; Moreso, F.; Pons, M.; Ramos, R.; Mora-Macià, J.; Carreras, J.; Soler, J.; Torres, F.; Campistol, J.M.; Martinez-Castelao, A.; et al. High-efficiency postdilution online hemodiafiltration reduces all-cause mortality in hemodialysis patients. J. Am. Soc. Nephrol. 2013, 24, 487–497. [Google Scholar] [CrossRef]
- Peters, S.A.E.; Bots, M.L.; Canaud, B.; Davenport, A.; Grooteman, M.P.C.; Kircelli, F.; Locatelli, F.; Maduell, F.; Morena, M.; Nubé, M.J.; et al. Haemodiafiltration and mortality in end-stage kidney disease patients: A pooled individual participant data analysis from four randomized controlled trials. Nephrol. Dial. Transplant. 2016, 31, 978–984. [Google Scholar] [CrossRef]
- Mostovaya, I.M.; Blankestijn, P.J.; Bots, M.L.; Covic, A.; Davenport, A.; Grooteman, M.P.C.; Hegbrant, J.; Locatelli, F.; Vanholder, R.; Nubé, M.J. Clinical Evidence on Hemodiafiltration: A Systematic Review and a Meta-analysis. Semin. Dial. 2014, 27, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Y.; Ninomiya, T.; Al-Kahwa, A.; Perkovic, V.; Gallagher, M.P.; Hawley, C.; Jardine, M.J. Effect of hemodiafiltration or hemofiltration compared with hemodialysis on mortality and cardiovascular disease in chronic kidney failure: A systematic review and meta-analysis of randomized trials. Am. J. Kidney Dis. 2014, 63, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C.; La Manna, G. Expanded Hemodialysis: A New Therapy for a New Class of Membranes. Contrib. Nephrol. 2017, 190, 124–133. [Google Scholar] [PubMed]
- Ronco, C. The Rise of Expanded Hemodialysis. Blood Purif. 2017, 44, I–VIII. [Google Scholar] [CrossRef] [PubMed]
- Zweigart, C.; Boschetti-de-Fierro, A.; Hulko, M.; Nilsson, L.G.; Beck, W.; Storr, M.; Krause, B. Medium cut-off membranes—closer to the natural kidney removal function. Int. J. Artif. Organs 2017, 40, 328–334. [Google Scholar] [CrossRef] [PubMed]
- De Smet, R.; David, F.; Sandra, P.; Van Kaer, J.; Lesaffer, G.; Dhondt, A.; Lameire, N.; Vanholder, R. A sensitive HPLC method for the quantification of free and total p-cresol in patients with chronic renal failure. Clin. Chim. Acta 1998, 278, 1–21. [Google Scholar] [CrossRef]
- Evenepoel, P.; Glorieux, G.; Meijers, B. P-Cresol Sulfate and Indoxyl Sulfate: Some Clouds Are Gathering in the Uremic Toxin Sky. Kidney Int. 2017, 92, 1323–1324. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Johnson, D.W.; Xu, H.; Carrero, J.J.; Pascoe, E.; French, C.; Campbell, K.L. Dietary protein-fiber ratio associates with circulating levels of indoxyl sulfate and p-cresyl sulfate in chronic kidney disease patients. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Campbell, K.; Johnson, D.; Stanton, T.; Pascoe, E.; Hawley, C.; Dimeski, G.; McWhinney, B.; Ungerer, J.; Isbel, N. Uraemic toxins and cardiovascular disease across the chronic kidney disease spectrum: An observational study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1035–1042. [Google Scholar] [CrossRef]
- Wu, I.W.; Hsu, K.H.; Lee, C.C.; Sun, C.Y.; Hsu, H.J.; Tsai, C.J.; Tzen, C.Y.; Wang, Y.C.; Lin, C.Y.; Wu, M.S. P-cresyl sulphate and indoxyl sulphate predict progression of chronic kidney disease. Nephrol. Dial. Transplant. 2011, 26, 938–947. [Google Scholar] [CrossRef]
- Vanholder, R.; Massy, Z.; Argiles, A.; Spasovski, G.; Verbeke, F.; Lameire, N.; Beige, J.; Brunet, P.; Cohen, G.; De Deyn, P.P.; et al. Chronic kidney disease as cause of cardiovascular morbidity and mortality. Nephrol. Dial. Transplant. 2005, 20, 1048–1056. [Google Scholar] [CrossRef]
- Khosroshahi, H.T.; Abedi, B.; Ghojazadeh, M.; Samadi, A.; Jouyban, A. Effects of fermentable high fiber diet supplementation on gut derived and conventional nitrogenous product in patients on maintenance hemodialysis: A randomized controlled trial. Nutr. Metab. 2019, 16, 1–8. [Google Scholar] [CrossRef]
- Esgalhado, M.; Kemp, J.A.; Azevedo, R.; Paiva, B.R.; Stockler-Pinto, M.B.; Dolenga, C.J.; Borges, N.A.; Nakao, L.S.; Mafra, D. Could resistant starch supplementation improve inflammatory and oxidative stress biomarkers and uremic toxins levels in hemodialysis patients? A pilot randomized controlled trial. Food Funct. 2018, 9, 6508–6516. [Google Scholar] [CrossRef]
- Sirich, T.L.; Plummer, N.S.; Gardner, C.D.; Hostetter, T.H.; Meyer, T.W. Effect of increasing dietary fiber on plasma levels of colon-derived solutes in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2014, 9, 1603–1610. [Google Scholar] [CrossRef]
- Tayebi Khosroshahi, H.; Vaziri, N.D.; Abedi, B.; Asl, B.H.; Ghojazadeh, M.; Jing, W.; Vatankhah, A.M. Effect of high amylose resistant starch (HAM-RS2) supplementation on biomarkers of inflammation and oxidative stress in hemodialysis patients: A randomized clinical trial. Hemodial. Int. 2018, 22, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.M.; Ge, Y.Y.; Huang, X.; Zhang, Y.Q.; Li, J.X. Effects of fermentable dietary fiber supplementation on oxidative and inflammatory status in hemodialysis patients. Int. J. Clin. Exp. Med. 2015, 8, 1363–1369. [Google Scholar] [PubMed]
- Montemurno, E.; Cosola, C.; Dalfino, G.; Daidone, G.; De Angelis, M.; Gobbetti, M.; Gesualdo, L. What would you like to eat, Mr CKD Microbiota? A Mediterranean Diet, please! Kidney Blood Press. Res. 2014, 39, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Y.; Hsu, H.H.; Wu, M.S. P-Cresol sulfate and indoxyl sulfate induce similar cellular inflammatory gene expressions in cultured proximal renal tubular cells. Nephrol. Dial. Transplant. 2013, 28, 70–78. [Google Scholar] [CrossRef]
- Lekawanvijit, S.; Adrahtas, A.; Kelly, D.J.; Kompa, A.R.; Wang, B.H.; Krum, H. Does indoxyl sulfate, a uraemic toxin, have direct effects on cardiac fibroblasts and myocytes? Eur. Heart J. 2010, 31, 1771–1779. [Google Scholar] [CrossRef]
- Shimizu, H.; Bolati, D.; Adijiang, A.; Muteliefu, G.; Enomoto, A.; Nishijima, F.; Dateki, M.; Niwa, T. NF-κb plays an important role in indoxyl sulfate-induced cellular senescence, fibrotic gene expression, and inhibition of proliferation in proximal tubular cells. Am. J. Physiol. Cell Physiol. 2011, 301, 1201–1213. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Miyamoto, Y.; Honda, D.; Tanaka, H.; Wu, Q.; Endo, M.; Noguchi, T.; Kadowaki, D.; Ishima, Y.; Kotani, S.; et al. P-Cresyl sulfate causes renal tubular cell damage by inducing oxidative stress by activation of NADPH oxidase. Kidney Int. 2013, 83, 582–592. [Google Scholar] [CrossRef]
- Rossi, M.; Campbell, K.L.; Johnson, D.W.; Stanton, T.; Vesey, D.A.; Coombes, J.S.; Weston, K.S.; Hawley, C.M.; McWhinney, B.C.; Ungerer, J.P.J.; et al. Protein-bound uremic toxins, inflammation and oxidative stress: A cross-sectional study in stage 3-4 chronic kidney disease. Arch. Med. Res. 2014, 45, 309–317. [Google Scholar] [CrossRef]
- Vanholder, R.; Schepers, E.; Pletinck, A.; Nagler, E.V.; Glorieux, G. The uremic toxicity of indoxyl sulfate and p-cresyl sulfate: A systematic review. J. Am. Soc. Nephrol. 2014, 25, 1897–1907. [Google Scholar] [CrossRef]
- Nataatmadja, M.; Cho, Y.; Campbell, K.; Johnson, D.W. The Roles of Indoxyl Sulphate and p-Cresyl Sulphate in Patients with Chronic Kidney Disease: A Review of Therapeutic Options. In Chronic Kidney Disease-from Pathophysiology to Clinical Improvements; IntechOpen: London, UK, 2018. [Google Scholar]
- Hung, S.C.; Kuo, K.L.; Wu, C.C.; Tarng, D.C. Indoxyl sulfate: A novel cardiovascular risk factor in chronic kidney disease. J. Am. Heart Assoc. 2017, 6, 1–9. [Google Scholar] [CrossRef]
- Tan, X.; Cao, X.; Zou, J.; Shen, B.; Zhang, X.; Liu, Z.; Lv, W.; Teng, J.; Ding, X. Indoxyl sulfate, a valuable biomarker in chronic kidney disease and dialysis. Hemodial. Int. 2017, 21, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Barreto, F.C.; Barreto, D.V.; Liabeuf, S.; Meert, N.; Glorieux, G.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A.; on behalf of the European Uremic Toxin Work Group (EUTox). Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1551–1558. [Google Scholar] [CrossRef] [PubMed]
- Kamiński, T.W.; Pawlak, K.; Karbowska, M.; Myśliwiec, M.; Pawlak, D. Indoxyl sulfate—The uremic toxin linking hemostatic system disturbances with the prevalence of cardiovascular disease in patients with chronic kidney disease. BMC Nephrol. 2017, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-C.; Tomino, Y.; Lu, K.-C. Impacts of Indoxyl Sulfate and p-Cresol Sulfate on Chronic Kidney Disease and Mitigating Effects of AST-120. Toxins 2018, 10, 367. [Google Scholar] [CrossRef] [PubMed]
- Weiner, D.E.; Falzon, L.; Skoufos, L.; Bernardo, A.; Beck, W.; Xiao, M.; Tran, H. Efficacy and safety of expanded hemodialysis with the theranova 400 dialyzer: A randomized controlled trial. Clin. J. Am. Soc. Nephrol. 2020, 15, 1310–1319. [Google Scholar] [CrossRef] [PubMed]
- Zickler, D.; Schindler, R.; Willy, K.; Martus, P.; Pawlak, M.; Storr, M.; Hulko, M.; Boehler, T.; Glomb, M.A.; Liehr, K.; et al. Medium Cut-Off (MCO) membranes reduce inflammation in chronic dialysis patients—A randomized controlled clinical trial. PLoS ONE 2017, 12, e0169024. [Google Scholar]
- Belmouaz, M.; Bauwens, M.; Hauet, T.; Bossard, V.; Jamet, P.; Joly, F.; Chikhi, E.; Joffrion, S.; Gand, E.; Bridoux, F. Comparison of the removal of uraemic toxins with medium cut-off and high-flux dialysers: A randomized clinical trial. Nephrol. Dial. Transplant. 2020, 35, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; Regolisti, G.; Brusasco, I.; Cabassi, A.; Morabito, S.; Fiaccadori, E. Alterations of intestinal barrier and microbiota in chronic kidney disease. Nephrol. Dial. Transplant. 2015, 30, 924–933. [Google Scholar] [CrossRef]
- Kuribayashi, T. Elimination half-lives of interleukin-6 and cytokine-induced neutrophil chemoattractant-1 synthesized in response to inflammatory stimulation in rats. Lab. Anim. Res. 2018, 34, 80. [Google Scholar] [CrossRef]
- Fukagawa, M.; Yumita, S.; Akizawa, T.; Uchida, E.; Tsukamoto, Y.; Iwasaki, M.; Koshikawa, S. Cinacalcet (KRN1493) effectively decreases the serum intact PTH level with favorable control of the serum phosphorus and calcium levels in Japanese dialysis patients. Nephrol. Dial. Transplant. 2008, 23, 328–335. [Google Scholar] [CrossRef]
- Wirtz, D.C.; Heller, K.D.; Miltner, O.; Zilkens, K.W.; Wolff, J.M. Interleukin-6: A potential inflammatory marker after total joint replacement. Int. Orthop. 2000, 24, 194–196. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.S. The role and clinical significance of high-sensitivity C-reactive protein in cardiovascular disease. Korean Circ. J. 2012, 42, 151–153. [Google Scholar] [CrossRef] [PubMed]
- Pepys, M.B.; Hirschfield, G.M. C-reactive protein: A critical update. J. Clin. Investig. 2003, 111, 1805–1812. [Google Scholar] [CrossRef]
- Lesaffer, G.; De Smet, R.; D’heuvaert, T.; Belpaire, F.M.; Lameire, N.; Vanholder, R. Comparative kinetics of the uremic toxin p-cresol versus creatinine in rats with and without renal failure. Kidney Int. 2003, 64, 1365–1373. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meijers, B.K.I.; De Loor, H.; Bammens, B.; Verbeke, K.; Vanrenterghem, Y.; Evenepoel, P. P-Cresyl Sulfate and Indoxyl Sulfate in Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1932–1938. [Google Scholar] [CrossRef]
- Rocchetti, M.T.; Cosola, C.; di Bari, I.; Magnani, S.; Galleggiante, V.; Scandiffio, L.; Dalfino, G.; Netti, G.S.; Atti, M.; Corciulo, R.; et al. Efficacy of Divinylbenzenic Resin in Removing Indoxyl Sulfate and P-cresol Sulfate in Hemodialysis Patients: Results from an In Vitro Study and an In Vivo Pilot Trial (xuanro4-Nature 3.2). Toxins 2020, 12, 170. [Google Scholar] [CrossRef] [PubMed]
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Ž.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar]
- Marzocco, S.; Fazeli, G.; Di Micco, L.; Autore, G.; Adesso, S.; Dal Piaz, F.; Heidland, A.; Di Iorio, B. Supplementation of Short-Chain Fatty Acid, Sodium Propionate, in Patients on Maintenance Hemodialysis: Beneficial Effects on Inflammatory Parameters and Gut-Derived Uremic Toxins, A Pilot Study (PLAN Study). J. Clin. Med. 2018, 7, 315. [Google Scholar] [CrossRef]
- Vinolo, M.A.R.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of inflammation by short chain fatty acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, 332–336. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro Scale for Rating Quality of Randomized Controlled Trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef] [PubMed]
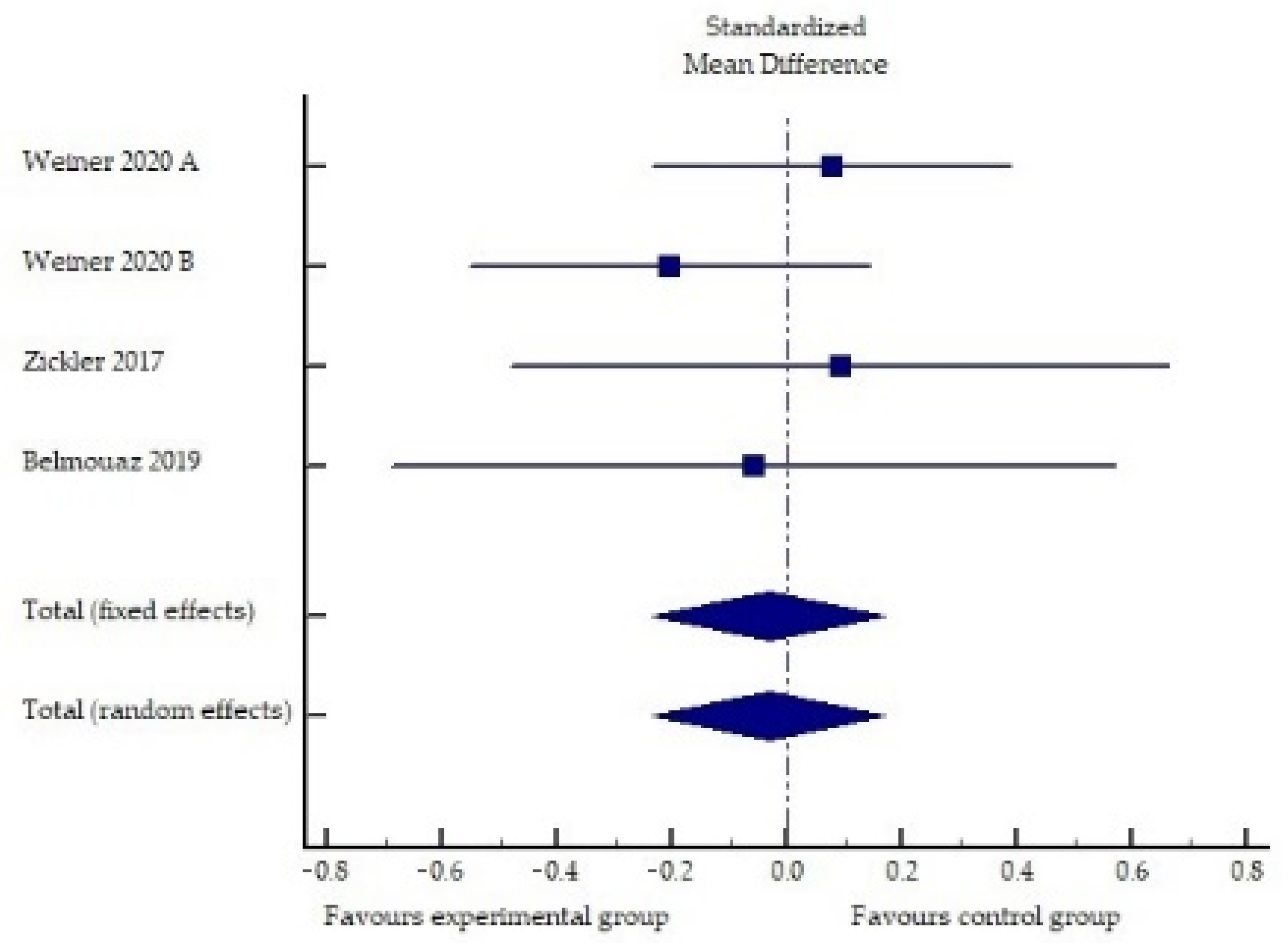
| Study | Sample Size | Exp Intervention | Con Intervention | Duration | Endpoint | Results |
|---|---|---|---|---|---|---|
| Weiner, 2020 [36] | Exp (n = 86) Con (n = 86) | MCO membrane | High-flux HD | 24 weeks | IL-6 | Exp: 15% ↓ |
| Con: 50.6% ↑ | ||||||
| p Exp vs. Con n.s. | ||||||
| TNF-α | Exp: 48.9% ↓ | |||||
| Con: 34.7% ↓ | ||||||
| p Exp vs. Con n.s. | ||||||
| Zickler, 2017 † [37] | Exp (n = 23) Con (n = 25) | MCO membrane | High-flux HD | 4 weeks of each dialysis modality + 8 weeks of extension phase | IL-6 | Exp: 33.3% ↓ ** |
| Con: 43.9% ↓ * | ||||||
| p Exp vs. Con n.s. | ||||||
| TNF-α | Exp: 14.5% ↓ ** | |||||
| Con: 5.1% ↓ | ||||||
| p Exp vs. Con n.s. | ||||||
| CRP | Exp: 39.2% ↓ | |||||
| Con: 28.4% ↓ | ||||||
| p Exp vs. Con n.s. | ||||||
| Belmouaz, 2020 [38] | Exp (n = 20) Con (n = 20) | MCO membrane | High-flux HD | 3 months of each dialysis modality | IL-6 | Exp: 6.3% ↑ |
| Con: 12.8% ↑ | ||||||
| p Exp vs. Con n.s. | ||||||
| TNF-α | Exp: 19.4% ↓ | |||||
| Con: 16.4% ↓ | ||||||
| p Exp vs. Con n.s. | ||||||
| CRP | Exp: missing data 1 | |||||
| Con: missing data 1 | ||||||
| p Exp vs. Con n.s. |
| Study | Sample Size | Exp Intervention | Con Intervention | Duration | Endpoint | Results |
|---|---|---|---|---|---|---|
| Khosroshahi, 2019 [18] | Exp (n = 23) Con (n = 21) | 20–25 g of HAM-RS2 20 g week 1–4 25 g week 5–8 | 20–25 g of waxy corn starch 20 g week 1–4 25 g week 5–8 | 8 weeks | Exp: 31.5% ↓ * | |
| PC | Con: 0.7% ↑ | |||||
| p Exp vs. Con 0.992 | ||||||
| Exp: 2.96% ↑ | ||||||
| IS | Con: 11.7% ↑ | |||||
| p Exp vs. Con 0.606 | ||||||
| Exp: 110.6% ↑ | ||||||
| Con: 25.05% ↑ | ||||||
| hs-CRP | p Exp vs. Con 0.866 | |||||
| Esgalhado, 2018 [19] | Exp (n = 15) Con (n = 16) | 16 g HAM-RS2 | 20 g manioc flour | 4 weeks | Exp: 18.15% ↓ ** | |
| IS | Con: 9.28% ↑ | |||||
| p Exp vs. Con 0.008 | ||||||
| Exp: 6.83% ↑ | ||||||
| Con: 1.53% ↓ | ||||||
| PCS | p Exp vs. Con 0.77 | |||||
| Exp: 11.86% ↓ ** | ||||||
| Con: 3.28% ↓ | ||||||
| IL-6 | p Exp vs. Con 0.06 | |||||
| Exp: 20.0 ↓ | ||||||
| Con: 12.77 ↓ | ||||||
| hs-CRP | p Exp vs. Con 0.16 | |||||
| Sirich, 2014 [20] | Exp (n = 20) Con (n = 20) | 15 g HAM-RS2 week 1, 30 g HAM-RS2 week 2–6 | 15 g waxy corn starch week 1, 30 g waxy corn starch week 2–6 | 6 weeks | Exp: 30.56% ↓ * | |
| free IS | Con: 0% | |||||
| p Exp vs. Con 0.02 | ||||||
| Exp: 17.14% ↓ | ||||||
| Con: 3.12% ↓ | ||||||
| total IS | p Exp vs. Con 0.04 | |||||
| Exp: 22.22% ↓ | ||||||
| Con: 4.55% ↑ | ||||||
| free PCS | p Exp vs. Con 0.05 | |||||
| Exp: 12.12 % ↓ | ||||||
| Con: 3.12% ↓ | ||||||
| total PCS | p Exp vs. Con 0.63 | |||||
| Exp: 10% ↑ | ||||||
| Con: 33.33 % ↑ | ||||||
| CRP | p Exp vs. Con 0.11 | |||||
| Xie, 2015 [22] | ExpA (n = 41) ExpB (n = 39) Con (n = 44) | ExpA: 10 g water soluble fiber ExpB: 20 g water soluble fiber | placebo starch | 6 weeks | ExpA: 32.63% ↓ * | |
| ExpB: 27.22% ↓ * | ||||||
| IL-6 | Con: 6.19% ↑ | |||||
| p ExpA vs. Con < 0.05 | ||||||
| p ExpB vs. Con < 0.05 | ||||||
| ExpA: 76.58% ↓ * | ||||||
| ExpB: 62.25% ↓ * | ||||||
| Con: 10.05% ↑ | ||||||
| IL-8 | p ExpA vs. Con < 0.05 | |||||
| p ExpB vs. Con < 0.05 | ||||||
| ExpA: 24.06% ↓ * | ||||||
| ExpB: 15.2% ↓ * | ||||||
| Con: 0.77% ↑ | ||||||
| TNF-α | p ExpA vs. Con < 0.05 | |||||
| p ExpB vs. Con < 0.05 | ||||||
| ExpA: 55.14% ↓ * | ||||||
| ExpB: 52.04% ↓ * | ||||||
| Con: 1.06% ↑ | ||||||
| p ExpA vs. Con < 0.05 | ||||||
| hs-CRP | p ExpB vs. Con < 0.05 | |||||
| Khosroshahi, 2018 [21] | Exp (n = 22) Con (n = 22) | 20–25 g of HAM-RS2 20 g week 1–4 25 g week 5–8 | 20–25 g regular wheat-flour 20 g week 1–4 25 g week 5–8 | 8 weeks | Exp: 25.0% ↓ * | |
| IL-6 | Con: 21.9% ↑ | |||||
| p Exp vs. Con < 0.01 | ||||||
| Exp: 6.49% ↓ | ||||||
| Con: 20.0% ↑ | ||||||
| IL-1β | p Exp vs. Con. n.s. | |||||
| Exp: 18.23% ↓ * | ||||||
| Con: 43.14% ↑ *** | ||||||
| p Exp vs. Con 0.01 | ||||||
| TNF-α | ||||||
| Exp: 21.6% ↑ | ||||||
| Con: 1.27% ↑ | ||||||
| hs-CRP | p Exp vs. Con n.s. |
| Study | Criterion 1 | Criterion 2 | Criterion 3 | Criterion 4 | Criterion 5 | Criterion 6 | Criterion 7 | Criterion 8 | Criterion 9 | Criterion 10 | Criterion 11 | Score |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weiner 2020 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 6 |
| Zickler 2017 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 8 |
| Belmouaz 2020 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 5 |
| Khosroshahi 2019 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Esgalhado 2018 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 7 |
| Sirich 2014 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 5 |
| Xie 2015 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 7 |
| Khosroshahi 2018 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herič, T.; Vivoda, T.; Bogataj, Š.; Pajek, J. Medium Cut-Off Dialysis Membrane and Dietary Fiber Effects on Inflammation and Protein-Bound Uremic Toxins: A Systematic Review and Protocol for an Interventional Study. Toxins 2021, 13, 244. https://doi.org/10.3390/toxins13040244
Herič T, Vivoda T, Bogataj Š, Pajek J. Medium Cut-Off Dialysis Membrane and Dietary Fiber Effects on Inflammation and Protein-Bound Uremic Toxins: A Systematic Review and Protocol for an Interventional Study. Toxins. 2021; 13(4):244. https://doi.org/10.3390/toxins13040244
Chicago/Turabian StyleHerič, Tjaša, Tjaša Vivoda, Špela Bogataj, and Jernej Pajek. 2021. "Medium Cut-Off Dialysis Membrane and Dietary Fiber Effects on Inflammation and Protein-Bound Uremic Toxins: A Systematic Review and Protocol for an Interventional Study" Toxins 13, no. 4: 244. https://doi.org/10.3390/toxins13040244
APA StyleHerič, T., Vivoda, T., Bogataj, Š., & Pajek, J. (2021). Medium Cut-Off Dialysis Membrane and Dietary Fiber Effects on Inflammation and Protein-Bound Uremic Toxins: A Systematic Review and Protocol for an Interventional Study. Toxins, 13(4), 244. https://doi.org/10.3390/toxins13040244







