Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review
Abstract
1. Introduction
2. Major Source of Zearalenone
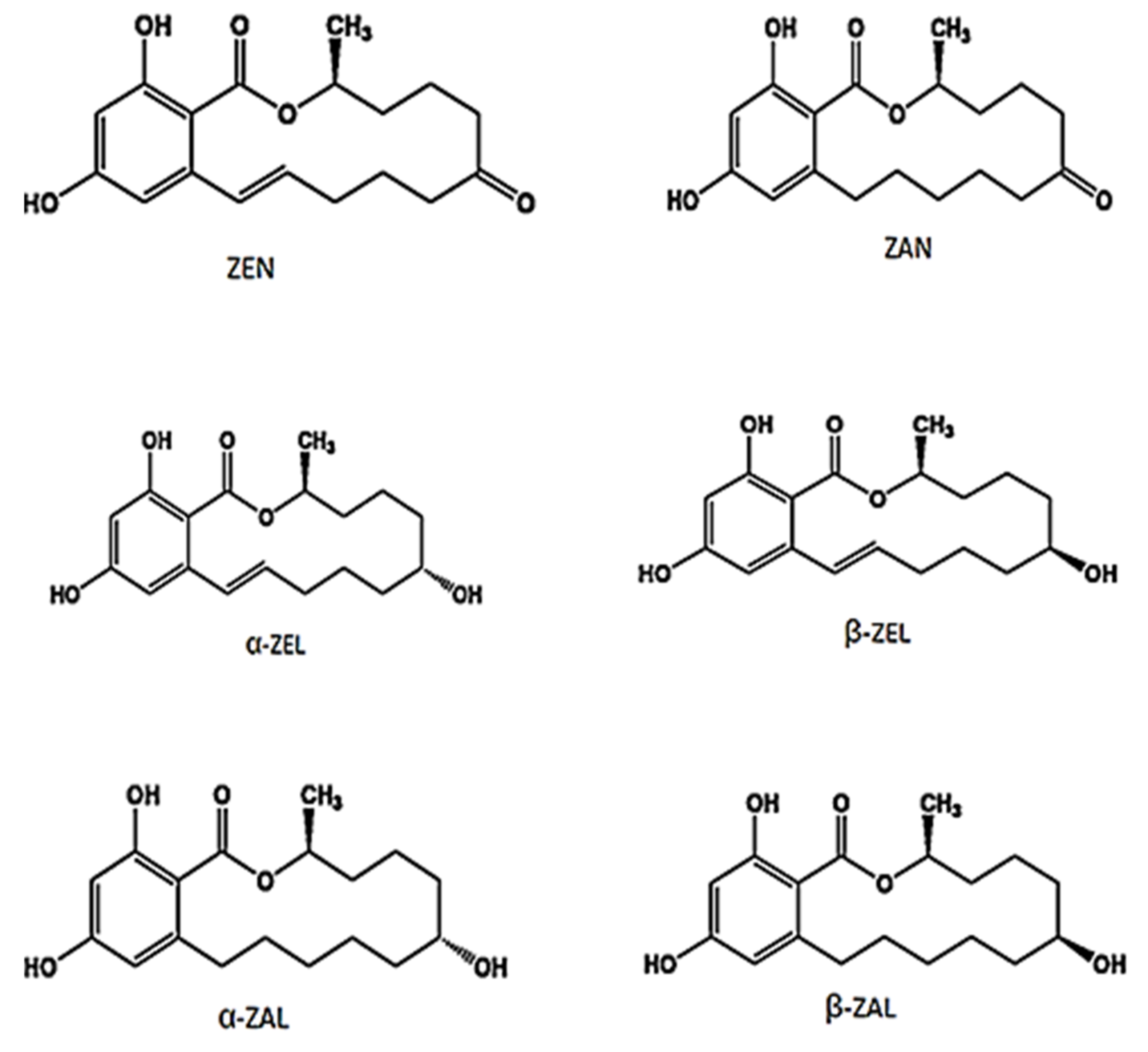
3. Chemistry and Biosynthesis of Zearalenone
4. Genes Responsible for Zearalenone Production
5. Occurrence in Food and Feed
6. Effects on Agricultural Food and Feed
| Food/Feed Matrix | Country | Range (μg/kg) | Detection Technique | Reference |
|---|---|---|---|---|
| Food | ||||
| Wheat | China | 10.1–3049 | LC–MS/MS | [56] |
| Malt | Botswana | 102–2213 | TLC and HPLC | [57] |
| Amaranthus | Argentina | 420–1980 | TLC | [58] |
| Cheese snacks | Iran | 1471 | HPLC-FD | [59] |
| Wheat | China | 5–1400 | HPLC | [60] |
| Corn byproducts | Germany | 369–1362 | GC-MS | [61] |
| Wheat | Romania | 327–1135 | LC-MS/MS | [62] |
| Corn | Thailand | 923 | HPLC-GC-MS | [63] |
| Corn flour | Iran | 889 | HPLC-FD | [59] |
| Corn | Germany | 48–860 | GC-MS | [61] |
| Maize | Tanzania | 729 | UHPLC/TOFMS | [64] |
| Corn | Brazil | 36.8–719 | LC-MS/MS | [65] |
| Corn | Brazil | 46.7–719 | HPLC | [66] |
| Barley | Brazil | 300–630 | LC-MS/MS | [67] |
| Durum wheat | Tunisia | 3–560 | HPLC | [68] |
| Corn | Philippines | 59–505 | HPLC-GC-MS | [63] |
| Maize | Italy | 453 | HPLC | [69] |
| Sorghum | USA | 443 | GC-MS | [70] |
| Wort | Botswana | 26–285 μg/L | TLC and HPLC | [57] |
| Wheat | Finland | 1.9–234 | LC-MS/MS | [71] |
| Wheat | Italy | 7–231 | HPLC-MS/MS | [72] |
| Maize | Iran | 100–212 | TLC | [73] |
| Soya meal | Germany | 51–211 | GC-MS | [61] |
| Beer | Botswana | 20–201 μg/L | TLC and HPLC | [57] |
| Barley foods | South Korea | 3.4–120 | ELISA and LC | [74] |
| Wheat | Germany | 17–104 | LC-MS | [75] |
| Wheat | Hungary | 50–98 | ELISA | [76] |
| Wheat | Kenya | 1–96 | ELISA | [77] |
| Corn foods | South Korea | 3.6–84 | ELISA and LC | [74] |
| Maize | Argentina | 0–83 | TLC | [78] |
| Oat | Finland | 76.9 | LC-MS/MS | [71] |
| Wheat bran | Germany | 3–67 | HPLC-FD | [79] |
| Barley | Czech Republic | 59.4 | HPLC-MS | [80] |
| Corn | Brazil | 55 | TLC | [81] |
| Maize | Italy | 53 | HPLC | [82] |
| Rice | South Korea | 21.7–47 | RP-HPLC-FLD | [83] |
| Paddy rice | Turkey | 42.9 | HPLC-PDA/HPLC-FLD | [20] |
| Barley malt | German federal states | 1.41–42.4 | LC-MS/MS | [84] |
| Barley | Lithuania | 10–41.4 | LC-MS/MS | [85] |
| Corn flour | UK | 6.5–40.8 | HPLC-FD | [86] |
| Maize | Botswana | 40 | HPLC | [87] |
| Wheat | Brazil | 40 | TLC | [81] |
| Corn flour | Germany | 2–40 | HPLC-FD | [79] |
| Maize | Poland | 18–39 | HPLC | [88] |
| Wheat | Syria | 4–34 | HPLC-MS/MS | [72] |
| Barley | Croatia | 32 | ELISA | [89] |
| Sorghum | Ethiopia | 19–32 | HPLC | [90] |
| Maize | Turkey | 28 | HPLC-PDA/HPLC-FLD | [20] |
| Breakfast cereal | Spain | 25 | HPLC | [91] |
| Oat | Germany | 21 | GC-MS | [61] |
| Wheat, corn, oat bran, oat products | Germany | 2–18 | HPLC-FD | [79] |
| Banana | India | 17 | HPLC | [92] |
| Red pepper | Germany | 2–17 | HPLC-FD | [79] |
| Maize | Morocco | 13.5–16.5 | HPLC | [93] |
| Chilli oil/chilli powder/chilli sauce | UK | 5–4/4.5–15.4/7.1 | HPLC-FD | [86] |
| Wheat | Germany | 15 | GC-MS | [61] |
| Gluten-free food | Germany | 2–14 | HPLC-FD | [79] |
| Barley | Finland | 13.7 | LC-MS/MS | [71] |
| Corn | Indonesia | 11–12 | HPLC-GC-MS | [94] |
| Milk | Egypt | 1.0–11.9 | HPLC–FLD | [21] |
| Curry powder | UK | 1.2–10.8 | HPLC-FD | [86] |
| Semolina | Germany | 2–9 | HPLC-FD | [79] |
| Garlic pickle | UK | 3–8 | HPLC-FD | [86] |
| Fennel | UK | 7 | HPLC-FD | [86] |
| Bean | Germany | 7 | HPLC-FD | [79] |
| Corn | Argentina | 3–7 | HPLC | [95] |
| Corn flakes | Qatar | 3.8–6.81 | HPLC | [96] |
| Coriander | UK | 3.6–6.7 | HPLC-FD | [86] |
| Canned foods | UK | 6.1 | HPLC-FD | [86] |
| Hazelnut | Germany | 6 | HPLC-FD | [79] |
| Corn | South Korea | 3.4–5.8 | ELISA and LC | [74] |
| Wheat | Sweden | 5 | HPLC/ESI-MS/MS | [97] |
| Pumpkin kernel | Germany | 4 | HPLC-FD | [79] |
| Sunflower seed | Germany | 2–4 | HPLC-FD | [79] |
| Wheat germ/corn flakes | Germany | 3/2–3 | HPLC-FD | [79] |
| Wheat flour | Turkey | 2.66 | HPLC-PDA/HPLC-FLD | [20] |
| Wheat | Qatar | 0.21–2.1 | HPLC | [96] |
| Potato products | Germany | 2 | HPLC-FD | [79] |
| Chicken meat | Pakistan | 0.85–1.83 | HPLC | [22] |
| Rice | Qatar | 0.18–1.4 | HPLC | [96] |
| Wheat | Turkey | 1.34 | HPLC-PDA/HPLC-FLD | [20] |
| Feed | ||||
| Complete feed | China | 10–3261.2 | HPLC-FD | [98] |
| Cow feeding stuffs | Argentina | 1200–3060 | HPLC | [99] |
| Pig complete feed (powder) | China | 10–835.4 | HPLC | [100] |
| Corn and poultry feed | Indonesia | 5.5–526 | ELISA and HPLC | [101] |
| Duck complete feed | China | 10–357.9 | HPLC | [100] |
| Cattle, poultry and swine feed | Poland | 0.07–349 | HPLC-MS/MS | [102] |
| Pig complete feed (pellets) | China | 10–329 | HPLC | [100] |
| Broiler feeds | Thailand | 2.22–263.51 | HPLC | [103] |
| Complete feed for pigs | Norway | 1.5–217.2 | HPLC-FLD | [104] |
| Swine feed | Hungary | 18–192 | ELISA | [105] |
| Sheep compound feed | Spain | 50–104.40 | UPLC–MS/MS and UPLC–QTOF–MS | [106] |
| Cattle compound feed | Spain | 88.2 | LC-MS | [107] |
| Poultry feed mixture | Slovakia | 3–86 | RP-HPLC-FLD | [108] |
| Fish feed | Europe | 67.9 | HPLC | [109] |
| Chicken feed | China | 61.59 | UPLC-MS/MS | [110] |
| Swine compound feed | Spain | 50 | UPLC–MS/MS and UPLC–QTOF–MS | [106] |
| Chicken feed | Botswana | 40 | HPLC | [87] |
| Pig feed | South Korea | 31.70 | HPLC | [13] |
| Poultry feed | South Korea | 0.24–26.80 | HPLC | [13] |
| Pig feed/cattle feed/rabbit feed | China | 18.78/14.43/10.46 | UPLC-MS/MS | [110] |
| Starter feed | India | 5.13–6.73 | HPLC and TLC | [111] |
| Compound feeds | South Africa | 0.56–1.85 | ELISA | [112] |
7. Effects of Environmental Factors on Zearalenone Production
8. Mechanism of Toxicity and Health Effects of Zearalenone
9. Effects of Processing on Zearalenone
10. Detection Techniques
11. Masked Mycotoxins as a Major Concern in Detection
| Origin | Conjugated Zearalenone | Matrix | Reference |
|---|---|---|---|
| Fungal conjugates | Zearalenone 4-sulfate | Fusarium graminearum, Rhizopus arrhizus | [191,210] |
| Zearalenone 4-glucoside | Rhizopus sp. | [211] | |
| Plant conjugates | α-/β-Zearalenol 4-glucoside | Maize cells | [212] |
| Zearalenone 4-glucoside | Maize cells, wheat | [75,196] | |
| Zearalenone dihexoside, α-Zearalenol dihexoside, β-Zearalenol dihexoside, Zearalenone malonylhexoside, α-Zearalenol malonylhexoside, β-Zearalenol malonylhexoside, Zearalenone pentosylhexoside, α-Zearalenol pentosylhexoside, β-Zearalenol pentosylhexoside | Arabidopsis thaliana | [197] | |
| Palmitoyl zearalenone | Fusarium-infected banana | [92] | |
| Mammalian conjugates | Zearalenone 3-glucuronide | Urine | [213] |
| Zearalenone 4-sulfate | Urine | [214,215] |
12. Degradation Kinetics
13. Management and Control Strategies
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rogowska, A.; Pomastowski, P.; Sagandykova, G.; Buszewski, B. Zearalenone and its metabolites: Effect on human health, metabolism and neutralisation methods. Toxicon 2019, 162, 46–56. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbiol. 2017, 7, 2170. [Google Scholar] [CrossRef]
- Sellamani, M.; Kalagatur, N.K.; Siddaiah, C.; Mudili, V.; Krishna, K.; Natarajan, G.; Rao Putcha, V.L. Antifungal and zearalenone inhibitory activity of Pediococcus pentosaceus isolated from dairy products on Fusarium graminearum. Front. Microbiol. 2016, 7, 890. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xie, Y. Review on microbial degradation of zearalenone and aflatoxins. Grain Oil Sci. Technol. 2020, 3, 117–125. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, D.; Sun, D.; Cui, S. Zearalenone affects reproductive functions of male offspring via transgenerational cytotoxicity on spermatogonia in mouse. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 234, 108766. [Google Scholar] [CrossRef] [PubMed]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef] [PubMed]
- Al-Jaal, B.A.; Jaganjac, M.; Barcaru, A.; Horvatovich, P.; Latiff, A. Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018. Food Chem. Toxicol. 2019, 129, 211–228. [Google Scholar] [CrossRef]
- Caglayan, M.O.; Şahin, S.; Üstündağ, Z. Detection strategies of Zearalenone for food safety: A review. Crit. Rev. Anal. Chem. 2020. [Google Scholar] [CrossRef]
- Yang, J.Y.; Wang, G.X.; Liu, J.L.; Fan, J.J.; Cui, S. Toxic effects of zearalenone and its derivatives α-zearalenol on male reproductive system in mice. Reprod. Toxicol. 2007, 24, 381–387. [Google Scholar] [CrossRef]
- Rai, A.; Das, M.; Tripathi, A. Occurrence and toxicity of a fusarium mycotoxin, zearalenone. Crit. Rev. Food Sci. Nutr. 2019, 60, 2710–2729. [Google Scholar] [CrossRef]
- Taranu, I.; Braicu, C.; Marin, D.E.; Pistol, G.C.; Motiu, M.; Balacescu, L.; Neagoe, I.B.; Burlacu, R. Exposure to zearalenone mycotoxin alters in vitro porcine intestinal epithelial cells by differential gene expression. Toxicol. Lett. 2015, 232, 310–325. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Molto, J.C.; Manes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Kim, W.; Park, J.-H.; Kim, D.; Kim, C.-R.; Chung, S.; Lee, C. The occurrence of zearalenone in South Korean feedstuffs between 2009 and 2016. Toxins 2017, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Fruhauf, S.; Novak, B.; Nagl, V.; Hackl, M.; Hartinger, D.; Rainer, V.; Labudová, S.; Adam, G.; Aleschko, M.; Moll, W.-D. Biotransformation of the mycotoxin zearalenone to its metabolites hydrolyzed zearalenone (HZEN) and decarboxylated hydrolyzed zearalenone (DHZEN) diminishes its estrogenicity in vitro and in vivo. Toxins 2019, 11, 481. [Google Scholar] [CrossRef] [PubMed]
- Ayed, Y.; Ayed-Boussema, I.; Ouanes, Z.; Bacha, H. In vitro and in vivo induction of chromosome aberrations by alpha-and beta-zearalenols: Comparison with zearalenone. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2011, 726, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Afshar, P.; Shokrzadeh, M.; Raeisi, S.N.; Ghorbani-HasanSaraei, A.; Nasiraii, L.R. Aflatoxins biodetoxification strategies based on probiotic bacteria. Toxicon 2020, 178, 50–58. [Google Scholar] [CrossRef]
- Haque, M.A.; Wang, Y.; Shen, Z.; Li, X.; Saleemi, M.K.; He, C. Mycotoxin contamination and control strategy in human, domestic animal and poultry: A review. Microb. Pathog. 2020, 142, 104095. [Google Scholar] [CrossRef]
- Pereyra, M.L.G.; Di Giacomo, A.L.; Lara, A.L.; Martínez, M.P.; Cavaglieri, L. Aflatoxin-degrading Bacillus sp. strains degrade zearalenone and produce proteases, amylases and cellulases of agro-industrial interest. Toxicon 2020, 180, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Santos Pereira, C.; Cunha, S.C.; Fernandes, J.O. Prevalent mycotoxins in animal feed: Occurrence and analytical methods. Toxins 2019, 11, 290. [Google Scholar] [CrossRef] [PubMed]
- Golge, O.; Kabak, B. Occurrence of deoxynivalenol and zearalenone in cereals and cereal products from Turkey. Food Control 2020, 110, 106982. [Google Scholar] [CrossRef]
- Abdallah, M.F.; Girgin, G.; Baydar, T. Mycotoxin detection in maize, commercial feed, and raw dairy milk samples from Assiut City, Egypt. Vet. Sci. 2019, 6, 57. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.Z.; Nisar, S.; Asi, M.R.; Jinap, S. Natural incidence of aflatoxins, ochratoxin A and zearalenone in chicken meat and eggs. Food Control 2014, 43, 98–103. [Google Scholar] [CrossRef]
- Su, Y.; Sun, Y.; Ju, D.; Chang, S.; Shi, B.; Shan, A. The detoxification effect of vitamin C on zearalenone toxicity in piglets. Ecotoxicol. Environ. Saf. 2018, 158, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.W.; Butchko, R.A.E.; Baker, S.E.; Proctor, R.H. Phylogenomic and functional domain analysis of polyketide synthases in Fusarium. Fungal Biol. 2012, 116, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-E.; Son, H.; Lee, Y.-W. Biosynthetic mechanism and regulation of zearalenone in Fusarium graminearum. JSM Mycotoxins 2018, 68, 1–6. [Google Scholar] [CrossRef]
- Warth, B.; Sulyok, M.; Berthiller, F.; Schuhmacher, R.; Krska, R. New insights into the human metabolism of the Fusarium mycotoxins deoxynivalenol and zearalenone. Toxicol. Lett. 2013, 220, 88–94. [Google Scholar] [CrossRef]
- Kuiper-Goodman, T.; Scott, P.M.; Watanabe, H. Risk assessment of the mycotoxin zearalenone. Regul. Toxicol. Pharmacol. 1987, 7, 253–306. [Google Scholar] [CrossRef]
- Biehl, M.L.; Prelusky, D.B.; Koritz, G.D.; Hartin, K.E.; Buck, W.B.; Trenholm, H.L. Biliary excretion and enterohepatic cycling of zearalenone in immature pigs. Toxicol. Appl. Pharmacol. 1993, 121, 152–159. [Google Scholar] [CrossRef]
- Kennedy, D.G.; Hewitt, S.A.; McEvoy, J.D.G.; Currie, J.W.; Cannavan, A.; Blanchflower, W.J.; Elliot, C.T. Zeranol is formed from Fusarium spp. toxins in cattle in vivo. Food Addit. Contam. 1998, 15, 393–400. [Google Scholar] [CrossRef]
- Miles, C.O.; Erasmuson, A.F.; Wilkins, A.L.; Towers, N.R.; Smith, B.L.; Garthwaite, I.; Scahill, B.G.; Hansen, R.P. Ovine metabolism of zearalenone to α-zearalanol (zeranol). J. Agric. Food Chem. 1996, 44, 3244–3250. [Google Scholar] [CrossRef]
- Migdalof, B.H.; Dugger, H.A.; Heider, J.G.; Coombs, R.A.; Terry, M.K. Biotransformation of zeranol: Disposition and metabolism in the female rat, rabbit, dog, monkey and man. Xenobiotica 1983, 13, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Shier, W.T.; Shier, A.C.; Xie, W.; Mirocha, C.J. Structure-activity relationships for human estrogenic activity in zearalenone mycotoxins. Toxicon 2001, 39, 1435–1438. [Google Scholar] [CrossRef]
- Richardson, K.E.; Hagler, W.M.; Mirocha, C.J. Production of zearalenone. alpha.-and. beta.-zearalenol, and. alpha.-and. beta.-zearalanol by Fusarium spp. in rice culture. J. Agric. Food Chem. 1985, 33, 862–866. [Google Scholar] [CrossRef]
- Celius, T.; Haugen, T.B.; Grotmol, T.; Walther, B.T. A sensitive zonagenetic assay for rapid in vitro assessment of estrogenic potency of xenobiotics and mycotoxins. Environ. Health Perspect. 1999, 107, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Le Guevel, R.; Pakdel, F. Assessment of oestrogenic potency of chemicals used as growth promoter by in-vitro methods. Hum. Reprod. 2001, 16, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, G.S.; Alexander, J. Fusarium Toxins in Cereals: A Risk Assessment; Nordic Council of Ministers: Copenhagen, Denmark, 1998; pp. 7–58. [Google Scholar]
- Miksicek, R.J. Interaction of naturally occurring nonsteroidal estrogens with expressed recombinant human estrogen receptor. J. Steroid Biochem. Mol. Biol. 1994, 49, 153–160. [Google Scholar] [CrossRef]
- Fitzpatrick, D.W.; Picken, C.A.; Murphy, L.C.; Buhr, M. Measurement of the relative binding affinity of zearalenone, alpha-zearalenol and beta-zearalenol for uterine and oviduct estrogen receptors in swine, rats and chickens: An indicator of estrogenic potencies. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 1989, 94, 691–694. [Google Scholar] [CrossRef]
- Gaffoor, I.; Trail, F. Characterization of two polyketide synthase genes involved in zearalenone biosynthesis in Gibberella zeae. Appl. Environ. Microbiol. 2006, 72, 1793–1799. [Google Scholar] [CrossRef]
- Huffman, J.; Gerber, R.; Du, L. Recent advancements in the biosynthetic mechanisms for polyketide-derived mycotoxins. Biopolymers 2010, 93, 764–776. [Google Scholar] [CrossRef]
- Lysøe, E.; Klemsdal, S.S.; Bone, K.R.; Frandsen, R.J.N.; Johansen, T.; Thrane, U.; Giese, H. The PKS4 gene of Fusarium graminearum is essential for zearalenone production. Appl. Environ. Microbiol. 2006, 72, 3924–3932. [Google Scholar] [CrossRef]
- Meng, K.; Wang, Y.; Yang, P.; Luo, H.; Bai, Y.; Shi, P.; Yuan, T.; Ma, R.; Yao, B. Rapid detection and quantification of zearalenone-producing Fusarium species by targeting the zearalenone synthase gene PKS4. Food Control 2010, 21, 207–211. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Bottalico, A.; Visconti, A.; Logrieco, A.; Solfrizzo, M.; Mirocha, C.J. Occurrence of zearalenols (diastereomeric mixture) in corn stalk rot and their production by associated Fusarium species. Appl. Environ. Microbiol. 1985, 49, 547–551. [Google Scholar] [CrossRef] [PubMed]
- D’mello, J.P.F.; Placinta, C.M.; Macdonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- Romero, A.; Ares, I.; Ramos, E.; Castellano, V.; Martínez, M.; Martínez-Larrañaga, M.-R.; Anadón, A.; Martínez, M.-A. Mycotoxins modify the barrier function of Caco-2 cells through differential gene expression of specific claudin isoforms: Protective effect of illite mineral clay. Toxicology 2016, 353, 21–33. [Google Scholar] [CrossRef]
- Reddy, K.E.; Song, J.; Lee, H.-J.; Kim, M.; Kim, D.-W.; Jung, H.J.; Kim, B.; Lee, Y.; Yu, D.; Kim, D.-W. Effects of high levels of deoxynivalenol and zearalenone on growth performance, and hematological and immunological parameters in pigs. Toxins 2018, 10, 114. [Google Scholar] [CrossRef]
- Li, N.; Liu, X.-L.; Zhang, F.-L.; Tian, Y.; Zhu, M.; Meng, L.-Y.; Dyce, P.W.; Shen, W.; Li, L. Whole-transcriptome analysis of the toxic effects of zearalenone exposure on ceRNA networks in porcine granulosa cells. Environ. Pollut. 2020, 261, 114007. [Google Scholar] [CrossRef]
- Eskola, M.; Altieri, A.; Galobart, J. Overview of the activities of the European Food Safety Authority on mycotoxins in food and feed. World Mycotoxin J. 2018, 11, 277–289. [Google Scholar] [CrossRef]
- European Commission. Commission Recommendation of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding (2006/576/EC). Off. J. Eur. Union 2006, 229, 7–9. [Google Scholar]
- Jia, R.; Liu, W.; Zhao, L.; Cao, L.; Shen, Z. Low doses of individual and combined deoxynivalenol and zearalenone in naturally moldy diets impair intestinal functions via inducing inflammation and disrupting epithelial barrier in the intestine of piglets. Toxicol. Lett. 2020, 333, 159–169. [Google Scholar] [CrossRef]
- Lewczuk, B.; Przybylska-Gornowicz, B.; Gajęcka, M.; Targońska, K.; Ziółkowska, N.; Prusik, M.; Gajęcki, M. Histological structure of duodenum in gilts receiving low doses of zearalenone and deoxynivalenol in feed. Exp. Toxicol. Pathol. 2016, 68, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Pistol, G.C.; Gras, M.A.; Marin, D.E.; Israel-Roming, F.; Stancu, M.; Taranu, I. Natural feed contaminant zearalenone decreases the expressions of important pro-and anti-inflammatory mediators and mitogen-activated protein kinase/NF-κB signalling molecules in pigs. Br. J. Nutr. 2014, 111, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Przybylska-Gornowicz, B.; Tarasiuk, M.; Lewczuk, B.; Prusik, M.; Ziółkowska, N.; Zielonka, Ł.; Gajęcki, M.; Gajęcka, M. The effects of low doses of two Fusarium toxins, zearalenone and deoxynivalenol, on the pig jejunum. A light and electron microscopic study. Toxins 2015, 7, 4684–4705. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Panel on Contaminants in the Food Chain. Scientific Opinion on the risks for public health related to the presence of zearalenone in food. EFSA J. 2011, 9, 2197. [Google Scholar] [CrossRef]
- Ji, F.; Xu, J.; Liu, X.; Yin, X.; Shi, J. Natural occurrence of deoxynivalenol and zearalenone in wheat from Jiangsu province, China. Food Chem. 2014, 157, 393–397. [Google Scholar] [CrossRef]
- Nkwe, D.O.; Taylor, J.E.; Siame, B.A. Fungi, aflatoxins, fumonisin B l and zearalenone contaminating sorghum-based traditional malt, wort and beer in Botswana. Mycopathologia 2005, 160, 177–186. [Google Scholar] [CrossRef]
- Bresler, G.; Vaamonde, G.; Brizzio, S. Natural occurrence of zearalenone and toxicogenic fungi in amaranth grain. Int. J. Food Microbiol. 1991, 13, 75–80. [Google Scholar] [CrossRef]
- Reza Oveisi, M.; Hajimahmoodi, M.; Memarian, S.; Sadeghi, N.; Shoeibi, S. Determination of zearalenone in corn flour and a cheese snack product using high-performance liquid chromatography with fluorescence detection. Food Addit. Contam. 2005, 22, 443–448. [Google Scholar] [CrossRef]
- Li, F.-Q.; Li, Y.-W.; Luo, X.-Y.; Yoshizawa, T. Fusarium toxins in wheat from an area in Henan Province, PR China, with a previous human red mould intoxication episode. Food Addit. Contam. 2002, 19, 163–167. [Google Scholar] [CrossRef]
- Schollenberger, M.; Müller, H.-M.; Rüfle, M.; Suchy, S.; Plank, S.; Drochner, W. Natural occurrence of 16 Fusarium toxins in grains and feedstuffs of plant origin from Germany. Mycopathologia 2006, 161, 43–52. [Google Scholar] [CrossRef]
- Stanciu, O.; Juan, C.; Miere, D.; Loghin, F.; Mañes, J. Occurrence and co-occurrence of Fusarium mycotoxins in wheat grains and wheat flour from Romania. Food Control 2017, 73, 147–155. [Google Scholar] [CrossRef]
- Yamashita, A.; Yoshizawa, T.; Aiura, Y.; Sanchez, P.C.; Dizon, E.I.; Arim, R.H.; Sardjono. Fusarium mycotoxins (fumonisins, nivalenol, and zearalenone) and aflatoxins in corn from Southeast Asia. Biosci. Biotechnol. Biochem. 1995, 59, 1804–1807. [Google Scholar] [CrossRef] [PubMed]
- Kamala, A.; Ortiz, J.; Kimanya, M.; Haesaert, G.; Donoso, S.; Tiisekwa, B.; De Meulenaer, B. Multiple mycotoxin co-occurrence in maize grown in three agro-ecological zones of Tanzania. Food Control 2015, 54, 208–215. [Google Scholar] [CrossRef]
- Vargas, E.A.; Preis, R.A.; Castro, L.; Silva, C.M.G. Co-occurrence of aflatoxins B 1, B 2, G 1, G 2, zearalenone and fumonisin B 1 in Brazilian corn. Food Addit. Contam. 2001, 18, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.M.G.; Vargas, E.A. A survey of zearalenone in corn using Romer Mycosep™ 224 column and high performance liquid chromatography. Food Addit. Contam. 2001, 18, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, K.C.; Rocha, L.; Savi, G.D.; Carnielli-Queiroz, L.; Almeida, F.; Minella, E.; Corrêa, B. Occurrence of deoxynivalenol and zearalenone in brewing barley grains from Brazil. Mycotoxin Res. 2018, 34, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Zaied, C.; Zouaoui, N.; Bacha, H.; Abid, S. Natural occurrence of zearalenone in Tunisian wheat grains. Food Control 2012, 25, 773–777. [Google Scholar] [CrossRef]
- Pietri, A.; Bertuzzi, T.; Pallaroni, L.; Piva, G. Occurrence of mycotoxins and ergosterol in maize harvested over 5 years in Northern Italy. Food Addit. Contam. 2004, 21, 479–487. [Google Scholar] [CrossRef]
- Hagler, W.M., Jr.; Bowman, D.T.; Babadoost, M.; Haney, C.A.; Swanson, S.P. Aflatoxin, Zearalenone, and Deoxynivalenol in North Carolina Grain Sorghum 1. Crop Sci. 1987, 27, 1273–1278. [Google Scholar] [CrossRef]
- Nathanail, A.V.; Syvähuoko, J.; Malachová, A.; Jestoi, M.; Varga, E.; Michlmayr, H.; Adam, G.; Sieviläinen, E.; Berthiller, F.; Peltonen, K. Simultaneous determination of major type A and B trichothecenes, zearalenone and certain modified metabolites in Finnish cereal grains with a novel liquid chromatography-tandem mass spectrometric method. Anal. Bioanal. Chem. 2015, 407, 4745–4755. [Google Scholar] [CrossRef]
- Alkadri, D.; Rubert, J.; Prodi, A.; Pisi, A.; Mañes, J.; Soler, C. Natural co-occurrence of mycotoxins in wheat grains from Italy and Syria. Food Chem. 2014, 157, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Hadiani, M.R.; Yazdanpanah, H.; Ghazi-Khansari, M.; Cheraghali, A.M.; Goodarzi, M. Survey of the natural occurrence of zearalenone in maize from northern Iran by thin-layer chromatography densitometry. Food Addit. Contam. 2003, 20, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Kim, E.K.; Shon, D.H.; Kim, Y.B. Natural co-occurrence of aflatoxin B1, fumonisin B1 and ochratoxin A in barley and corn foods from Korea. Food Addit. Contam. 2002, 19, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Schneweis, I.; Meyer, K.; Engelhardt, G.; Bauer, J. Occurrence of zearalenone-4-β-D-glucopyranoside in wheat. J. Agric. Food Chem. 2002, 50, 1736–1738. [Google Scholar] [CrossRef] [PubMed]
- Tima, H.; Brückner, A.; Mohácsi-Farkas, C.; Kiskó, G. Fusarium mycotoxins in cereals harvested from Hungarian fields. Food Addit. Contam. Part B 2016, 9, 127–131. [Google Scholar] [CrossRef]
- Muthomi, J.W.; Ndung’u, J.K.; Gathumbi, J.K.; Mutitu, E.W.; Wagacha, J.M. The occurrence of Fusarium species and mycotoxins in Kenyan wheat. Crop Prot. 2008, 27, 1215–1219. [Google Scholar] [CrossRef]
- Garrido, C.; Pezzani, C.H.; Pacin, A. Mycotoxins occurrence in Argentina’s maize (Zea mays L.), from 1999 to 2010. Food Control 2012, 25, 660–665. [Google Scholar] [CrossRef]
- Schollenberger, M.; Müller, H.-M.; Rüfle, M.; Suchy, S.; Planck, S.; Drochner, W. Survey of Fusarium toxins in foodstuffs of plant origin marketed in Germany. Int. J. Food Microbiol. 2005, 97, 317–326. [Google Scholar] [CrossRef]
- Běláková, S.; Benešová, K.; Čáslavský, J.; Svoboda, Z.; Mikulíková, R. The occurrence of the selected fusarium mycotoxins in czech malting barley. Food Control 2014, 37, 93–98. [Google Scholar] [CrossRef]
- Almeida-Ferreira, G.C.; Barbosa-Tessmann, I.P.; Sega, R.; Machinski Junior, M. Occurrence of zearalenone in wheat-and corn-based products commercialized in the State of Paraná, Brazil. Braz. J. Microbiol. 2013, 44, 371–375. [Google Scholar] [CrossRef]
- Camardo Leggieri, M.; Bertuzzi, T.; Pietri, A.; Battilani, P. Mycotoxin occurrence in maize produced in Northern Italy over the years 2009–2011: Focus on the role of crop related factors. Phytopathol. Mediterr. 2015, 54, 212–221. [Google Scholar] [CrossRef]
- Park, J.W.; Choi, S.-Y.; Hwang, H.-J.; Kim, Y.-B. Fungal mycoflora and mycotoxins in Korean polished rice destined for humans. Int. J. Food Microbiol. 2005, 103, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Habler, K.; Rychlik, M. Multi-mycotoxin stable isotope dilution LC-MS/MS method for Fusarium toxins in cereals. Anal. Bioanal. Chem. 2016, 408, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Mankevičienė, A.; Butkutė, B.; Gaurilčikienė, I.; Dabkevičius, Z.; Supronienė, S. Risk assessment of Fusarium mycotoxins in Lithuanian small cereal grains. Food Control 2011, 22, 970–976. [Google Scholar] [CrossRef]
- Patel, S.; Hazel, C.M.; Winterton, A.G.M.; Mortby, E. Survey of ethnic foods for mycotoxins. Food Addit. Contam. 1996, 13, 833–841. [Google Scholar] [CrossRef]
- Siame, B.A.; Mpuchane, S.F.; Gashe, B.A.; Allotey, J.; Teffera, G. Occurrence of aflatoxins, fumonisin B1, and zearalenone in foods and feeds in Botswana. J. Food Prot. 1998, 61, 1670–1673. [Google Scholar] [CrossRef]
- Czembor, E.; Stępień, Ł.; Waśkiewicz, A. Effect of environmental factors on Fusarium species and associated mycotoxins in maize grain grown in Poland. PLoS ONE 2015, 10, e0133644. [Google Scholar] [CrossRef]
- Pleadin, J.; Vahčić, N.; Perši, N.; Ševelj, D.; Markov, K.; Frece, J. Fusarium mycotoxins’ occurrence in cereals harvested from Croatian fields. Food Control 2013, 32, 49–54. [Google Scholar] [CrossRef]
- Ayalew, A.; Fehrmann, H.; Lepschy, J.; Beck, R.; Abate, D. Natural occurrence of mycotoxins in staple cereals from Ethiopia. Mycopathologia 2006, 162, 57–63. [Google Scholar] [CrossRef]
- Ibáñez-Vea, M.; Martínez, R.; González-Peñas, E.; Lizarraga, E.; de Cerain, A.L. Co-occurrence of aflatoxins, ochratoxin A and zearalenone in breakfast cereals from Spanish market. Food Control 2011, 22, 1949–1955. [Google Scholar] [CrossRef]
- Chakrabarti, D.K.; Ghosal, S. Occurrence of free and conjugated 12, 13-epoxytrichothecenes and zearalenone in banana fruits infected with Fusarium moniliforme. Appl. Environ. Microbiol. 1986, 51, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Brera, C.; Elakhdari, S.; Catano, C.; Debegnach, F.; Angelini, S.; De Santis, B.; Faid, M.; Benlemlih, M.; Minardi, V. Natural occurrence of mycotoxins in cereals and spices commercialized in Morocco. Food Control 2006, 17, 868–874. [Google Scholar] [CrossRef]
- Ali, N.; Sardjono; Yamashita, A.; Yoshizawa, T. Natural co-occurrence of aflatoxins and Fusarium mycotoxins (fumonisins, deoxynivalenol, nivalenol and zearalenone) in corn from Indonesia. Food Addit. Contam. 1998, 15, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, H.H.L.; Martinez, E.J.; Pacin, A.M.; Resnik, S.L.; Sydenham, E.W. Natural co-occurrence of fumonisins, deoxynivalenol, zearalenone and aflatoxins in field trial corn in Argentina. Food Addit. Contam. 1999, 16, 565–569. [Google Scholar] [CrossRef]
- Abdulkadar, A.H.W.; Al-Ali, A.A.; Al-Kildi, A.M.; Al-Jedah, J.H. Mycotoxins in food products available in Qatar. Food Control 2004, 15, 543–548. [Google Scholar] [CrossRef]
- Lindblad, M.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Fredlund, E. Deoxynivalenol and other selected Fusarium toxins in Swedish wheat—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 284–291. [Google Scholar] [CrossRef]
- Liu, J.; Sun, L.; Zhang, J.; Guo, J.; Chen, L.; Qi, D.; Zhang, N. Aflatoxin B1, zearalenone and deoxynivalenol in feed ingredients and complete feed from central China. Food Addit. Contam. Part B 2016, 9, 91–97. [Google Scholar] [CrossRef]
- Cavaglieri, L.R.; Gonzalez Pereyra, L.M.; Pereyra, C.M.; Magnoli, C.E.; Chulze, S.N.; Dalcero, A.M. Fungal and mycotoxin contamination of cow feeding stuffs in Argentina. In Proceedings of the International Conference. Fifth Framework Program. European Union Myco-Globe Project, Accra, Ghana, 13–16 September 2005; European Commission: Brussels, Belgium, 2005; pp. 13–16. [Google Scholar]
- Wu, L.; Li, J.; Li, Y.; Li, T.; He, Q.; Tang, Y.; Liu, H.; Su, Y.; Yin, Y.; Liao, P. Aflatoxin B 1, zearalenone and deoxynivalenol in feed ingredients and complete feed from different Province in China. J. Anim. Sci. Biotechnol. 2016, 7, 63. [Google Scholar] [CrossRef]
- Nuryono, N.; Noviandi, C.T.; Böhm, J.; Razzazi-Fazeli, E. A limited survey of zearalenone in Indonesian maize-based food and feed by ELISA and high performance liquid chromatography. Food Control 2005, 16, 65–71. [Google Scholar] [CrossRef]
- Kosicki, R.; Błajet-Kosicka, A.; Grajewski, J.; Twarużek, M. Multiannual mycotoxin survey in feed materials and feedingstuffs. Anim. Feed Sci. Technol. 2016, 215, 165–180. [Google Scholar] [CrossRef]
- Kongkapan, J.; Poapolathep, S.; Isariyodom, S.; Kumagai, S.; Poapolathep, A. Simultaneous detection of multiple mycotoxins in broiler feeds using a liquid chromatography tandem-mass spectrometry. J. Vet. Med. Sci. 2015, 78, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Janić Hajnal, E.; Kos, J.; Krulj, J.; Krstović, S.; Jajić, I.; Pezo, L.; Šarić, B.; Nedeljković, N. Aflatoxins contamination of maize in Serbia: The impact of weather conditions in 2015. Food Addit. Contam. Part A 2017, 34, 1999–2010. [Google Scholar] [CrossRef] [PubMed]
- Tima, H.; Rácz, A.; Guld, Z.; Mohácsi-Farkas, C.; Kiskó, G. Deoxynivalenol, zearalenone and T-2 in grain based swine feed in Hungary. Food Addit. Contam. Part B 2016, 9, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Romera, D.; Mateo, E.M.; Mateo-Castro, R.; Gómez, J.V.; Gimeno-Adelantado, J.V.; Jiménez, M. Determination of multiple mycotoxins in feedstuffs by combined use of UPLC–MS/MS and UPLC–QTOF–MS. Food Chem. 2018, 267, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Kim, H.J. Development of an immunoaffinity chromatography and LC-MS/MS method for the determination of 6 zearalenones in animal feed. PLoS ONE 2018, 13, e0193584. [Google Scholar] [CrossRef] [PubMed]
- Labuda, R.; Parich, A.; Berthiller, F.; Tančinová, D. Incidence of trichothecenes and zearalenone in poultry feed mixtures from Slovakia. Int. J. Food Microbiol. 2005, 105, 19–25. [Google Scholar] [CrossRef]
- Pietsch, C.; Kersten, S.; Burkhardt-Holm, P.; Valenta, H.; Dänicke, S. Occurrence of deoxynivalenol and zearalenone in commercial fish feed: An initial study. Toxins 2013, 5, 184–192. [Google Scholar] [CrossRef]
- Hu, X.; Hu, R.; Zhang, Z.; Li, P.; Zhang, Q.; Wang, M. Development of a multiple immunoaffinity column for simultaneous determination of multiple mycotoxins in feeds using UPLC–MS/MS. Anal. Bioanal. Chem. 2016, 408, 6027–6036. [Google Scholar] [CrossRef]
- Koteswara Rao, V.; Girisham, S.; Madhusudhan Reddy, S. Prevalence of toxigenic Penicillium species associated with poultry house in Telangana, India. Arch. Environ. Occup. Health 2016, 71, 353–361. [Google Scholar] [CrossRef]
- Gizachew, D.; Szonyi, B.; Tegegne, A.; Hanson, J.; Grace, D. Aflatoxin contamination of milk and dairy feeds in the Greater Addis Ababa milk shed, Ethiopia. Food Control 2016, 59, 773–779. [Google Scholar] [CrossRef]
- Chilaka, C.A.; De Boevre, M.; Atanda, O.O.; De Saeger, S. The status of Fusarium mycotoxins in sub-Saharan Africa: A review of emerging trends and post-harvest mitigation strategies towards food control. Toxins 2017, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Mathlouthi, M. Water content, water activity, water structure and the stability of foodstuffs. Food Control 2001, 12, 409–417. [Google Scholar] [CrossRef]
- Barbosa-Cánovas, G.V.; Fontana, A.J., Jr.; Schmidt, S.J.; Labuza, T.P. Water Activity in Foods: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2020. [Google Scholar]
- Stanciu, O.; Juan, C.; Berrada, H.; Miere, D.; Loghin, F.; Mañes, J. Study on trichothecene and zearalenone presence in romanian wheat relative to weather conditions. Toxins 2019, 11, 163. [Google Scholar] [CrossRef] [PubMed]
- Mannaa, M.; Kim, K.D. Influence of temperature and water activity on deleterious fungi and mycotoxin production during grain storage. Mycobiology 2017, 45, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Pleadin, J.; Frece, J.; Lešić, T.; Zadravec, M.; Vahčić, N.; Malenica Staver, M.; Markov, K. Deoxynivalenol and zearalenone in unprocessed cereals and soybean from different cultivation regions in Croatia. Food Addit. Contam. Part B 2017, 10, 268–274. [Google Scholar] [CrossRef]
- Habschied, K.; Šarkanj, B.; Klapec, T.; Krstanović, V. Distribution of zearalenone in malted barley fractions dependent on Fusarium graminearum growing conditions. Food Chem. 2011, 129, 329–332. [Google Scholar] [CrossRef]
- Wu, L.; Qiu, L.; Zhang, H.; Sun, J.; Hu, X.; Wang, B. Optimization for the Production of Deoxynivalenoland Zearalenone by Fusarium graminearum Using Response Surface Methodology. Toxins 2017, 9, 57. [Google Scholar] [CrossRef]
- Hamed, M.A.; Abdel Ghany, T.M.; Elhussieny, N.I.; Nabih, M.A. Exploration of fungal infection in agricultural grains, aflatoxin and zearalenone synthesis under pH stress. Int. J. Curr. Microbiol. App. Sci. 2016, 5, 1007–1017. [Google Scholar] [CrossRef]
- Velluti, A.; Sanchis, V.; Ramos, A.J.; Turon, C.; Marin, S. Impact of essential oils on growth rate, zearalenone and deoxynivalenol production by Fusarium graminearum under different temperature and water activity conditions in maize grain. J. Appl. Microbiol. 2004, 96, 716–724. [Google Scholar] [CrossRef]
- Martins, M.L.; Martins, H.M. Influence of water activity, temperature and incubation time on the simultaneous production of deoxynivalenol and zearalenone in corn (Zea mays) by Fusarium graminearum. Food Chem. 2002, 79, 315–318. [Google Scholar] [CrossRef]
- Peter Mshelia, L.; Selamat, J.; Iskandar Putra Samsudin, N.; Rafii, M.Y.; Abdul Mutalib, N.-A.; Nordin, N.; Berthiller, F. Effect of Temperature, Water Activity and Carbon Dioxide on Fungal Growth and Mycotoxin Production of Acclimatised Isolates of Fusarium verticillioides and F. graminearum. Toxins 2020, 12, 478. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Kunsági-Máté, S.; Bálint, M.; Hetényi, C.; Gerner, Z.; Lemli, B. Interaction of mycotoxin zearalenone with human serum albumin. J. Photochem. Photobiol. B Biol. 2017, 170, 16–24. [Google Scholar] [CrossRef]
- Grenier, B.; Applegate, T.J. Modulation of intestinal functions following mycotoxin ingestion: Meta-analysis of published experiments in animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef] [PubMed]
- Cheat, S.; Pinton, P.; Cossalter, A.-M.; Cognie, J.; Vilariño, M.; Callu, P.; Raymond-Letron, I.; Oswald, I.P.; Kolf-Clauw, M. The mycotoxins deoxynivalenol and nivalenol show in vivo synergism on jejunum enterocytes apoptosis. Food Chem. Toxicol. 2016, 87, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Mally, A.; Solfrizzo, M.; Degen, G.H. Biomonitoring of the mycotoxin Zearalenone: Current state-of-the art and application to human exposure assessment. Arch. Toxicol. 2016, 90, 1281–1292. [Google Scholar] [CrossRef] [PubMed]
- Kollarczik, B.; Gareis, M.; Hanelt, M. In vitro transformation of the Fusarium mycotoxins deoxynivalenol and zearalenone by the normal gut microflora of pigs. Nat. Toxins 1994, 2, 105–110. [Google Scholar] [CrossRef]
- Braicu, C.; Selicean, S.; Cojocneanu-Petric, R.; Lajos, R.; Balacescu, O.; Taranu, I.; Marin, D.E.; Motiu, M.; Jurj, A.; Achimas-Cadariu, P. Evaluation of cellular and molecular impact of zearalenone and Escherichia coli co-exposure on IPEC-1 cells using microarray technology. BMC Genom. 2016, 17, 576. [Google Scholar] [CrossRef] [PubMed]
- Obremski, K. Changes in Th1 and Th2 cytokine concentrations in ileal Peyer’s patches in gilts exposed to zearalenone. Pol. J. Vet. Sci. 2014, 17, 53–59. [Google Scholar] [CrossRef]
- Cheng, Q.; Jiang, S.; Huang, L.; Ge, J.; Wang, Y.; Yang, W. Zearalenone induced oxidative stress in the jejunum in postweaning gilts through modulation of the Keap1–Nrf2 signaling pathway and relevant genes. J. Anim. Sci. 2019, 97, 1722–1733. [Google Scholar] [CrossRef]
- Gerez, J.R.; Pinton, P.; Callu, P.; Grosjean, F.; Oswald, I.P.; Bracarense, A.P.F.L. Deoxynivalenol alone or in combination with nivalenol and zearalenone induce systemic histological changes in pigs. Exp. Toxicol. Pathol. 2015, 67, 89–98. [Google Scholar] [CrossRef]
- Abbès, S.; Ouanes, Z.; Salah-Abbès, J.B.; Abdel-Wahhab, M.A.; Oueslati, R.; Bacha, H. Preventive role of aluminosilicate clay against induction of micronuclei and chromosome aberrations in bone-marrow cells of Balb/c mice treated with Zearalenone. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2007, 631, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Fleck, S.C.; Hildebrand, A.A.; Müller, E.; Pfeiffer, E.; Metzler, M. Genotoxicity and inactivation of catechol metabolites of the mycotoxin zearalenone. Mycotoxin Res. 2012, 28, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Abid-Essefi, S.; Baudrimont, I.; Hassen, W.; Ouanes, Z.; Mobio, T.A.; Anane, R.; Creppy, E.E.; Bacha, H. DNA fragmentation, apoptosis and cell cycle arrest induced by zearalenone in cultured DOK, Vero and Caco-2 cells: Prevention by Vitamin E. Toxicology 2003, 192, 237–248. [Google Scholar] [CrossRef]
- Ouanes, Z.; Ayed-Boussema, I.; Baati, T.; Creppy, E.E.; Bacha, H. Zearalenone induces chromosome aberrations in mouse bone marrow: Preventive effect of 17β-estradiol, progesterone and Vitamin E. Mutat. Res. 2005, 565, 139–149. [Google Scholar] [CrossRef]
- Salah-Abbès, J.B.; Abbès, S.; Houas, Z.; Abdel-Wahhab, M.A.; Oueslati, R. Zearalenone induces immunotoxicity in mice: Possible protective effects of radish extract (Raphanus sativus). J. Pharm. Pharmacol. 2008, 60, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Čonková, E.; Laciaková, A.; Pástorová, B.; Seidel, H.; Kováč, G. The effect of zearalenone on some enzymatic parameters in rabbits. Toxicol. Lett. 2001, 121, 145–149. [Google Scholar] [CrossRef]
- Islam, M.R.; Kim, J.W.; Roh, Y.-S.; Kim, J.-H.; Han, K.M.; Kwon, H.-J.; Lim, C.W.; Kim, B. Evaluation of immunomodulatory effects of zearalenone in mice. J. Immunotoxicol. 2017, 14, 125–136. [Google Scholar] [CrossRef]
- Zhou, M.; Yang, L.; Chen, Y.; Sun, T.; Wang, N.; Chen, X.; Yang, Z.; Ge, J.; Jiang, S. Comparative study of stress response, growth and development of uteri in post-weaning gilts challenged with zearalenone and estradiol benzoate. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1885–1894. [Google Scholar] [CrossRef]
- Meurens, F.; Summerfield, A.; Nauwynck, H.; Saif, L.; Gerdts, V. The pig: A model for human infectious diseases. Trends Microbiol. 2012, 20, 50–57. [Google Scholar] [CrossRef]
- Kowalska, K.; Habrowska-Górczyńska, D.E.; Piastowska-Ciesielska, A.W. Zearalenone as an endocrine disruptor in humans. Environ. Toxicol. Pharmacol. 2016, 48, 141–149. [Google Scholar] [CrossRef]
- Metzler, M.; Pfeiffer, E.; Hildebrand, A. Zearalenone and its metabolites as endocrine disrupting chemicals. World Mycotoxin J. 2010, 3, 385–401. [Google Scholar] [CrossRef]
- Zhu, L.; Yuan, H.; Guo, C.; Lu, Y.; Deng, S.; Yang, Y.; Wei, Q.; Wen, L.; He, Z. Zearalenone induces apoptosis and necrosis in porcine granulosa cells via a caspase-3-and caspase-9-dependent mitochondrial signaling pathway. J. Cell. Physiol. 2012, 227, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.-Q.; Sun, X.-F.; Wu, R.-Y.; Cheng, S.-F.; Zhang, G.-L.; Zhai, Q.-Y.; Liu, X.-L.; Zhao, Y.; Shen, W.; Li, L. Zearalenone exposure elevated the expression of tumorigenesis genes in mouse ovarian granulosa cells. Toxicol. Appl. Pharmacol. 2018, 356, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Assumaidaee, A.A.M.; Ali, N.M.; Ahmed, S.W. Zearalenone Mycotoxicosis: Pathophysiology and Immunotoxicity. Iraqi J. Vet. Med. 2020, 44, 29–38. [Google Scholar] [CrossRef]
- Massart, F.; Meucci, V.; Saggese, G.; Soldani, G. High growth rate of girls with precocious puberty exposed to estrogenic mycotoxins. J. Pediatr. 2008, 152, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Xue, K.S.; Sun, X.; Williams, P.L.; Wang, J.-S.; Tang, L. Toxicogenomic responses to zearalenone in Caenorhabditis elegans reveal possible molecular mechanisms of reproductive toxicity. Food Chem. Toxicol. 2018, 122, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Yousef, M.S.; Takagi, M.; Talukder, A.K.; Marey, M.A.; Kowsar, R.; Abdel-Razek, A.-R.K.; Shimizu, T.; Fink-Gremmels, J.; Miyamoto, A. Zearalenone (ZEN) disrupts the anti-inflammatory response of bovine oviductal epithelial cells to sperm in vitro. Reprod. Toxicol. 2017, 74, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Massart, F.; Micillo, F.; Rivezzi, G.; Perrone, L.; Baggiani, A.; Miccoli, M.; Meucci, V. Zearalenone screening of human breast milk from the Naples area. Toxicol. Environ. Chem. 2016, 98, 128–136. [Google Scholar] [CrossRef]
- Minervini, F.; Dell’Aquila, M.E. Zearalenone and reproductive function in farm animals. Int. J. Mol. Sci. 2008, 9, 2570–2584. [Google Scholar] [CrossRef]
- Mauro, T.; Hao, L.; Pop, L.C.; Buckley, B.; Schneider, S.H.; Bandera, E.V.; Shapses, S.A. Circulating zearalenone and its metabolites differ in women due to body mass index and food intake. Food Chem. Toxicol. 2018, 116, 227–232. [Google Scholar] [CrossRef]
- Hueza, I.M.; Raspantini, P.C.F.; Raspantini, L.E.R.; Latorre, A.O.; Gornjak, S.L. Zearalenone, an estrogenic mycotoxin, is an immunotoxic compound. Toxins 2014, 6, 1080–1095. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Nakajima, M.; Yokoi, T. Cytochrome P450-mediated metabolism of estrogens and its regulation in human. Cancer Lett. 2005, 227, 115–124. [Google Scholar] [CrossRef]
- Pfeiffer, E.; Hildebrand, A.; Damm, G.; Rapp, A.; Cramer, B.; Humpf, H.U.; Metzler, M. Aromatic hydroxylation is a major metabolic pathway of the mycotoxin zearalenone in vitro. Mol. Nutr. Food Res. 2009, 53, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, Z.; Sahni, S.; Lok, H.; Davies, M.J.; Wink, D.A.; Richardson, D.R. Regulation and control of nitric oxide (NO) in macrophages: Protecting the “professional killer cell” from its own cytotoxic arsenal via MRP1 and GSTP1. Biochim. Biophys. Acta. Gen. Subj. 2017, 1861, 995–999. [Google Scholar] [CrossRef]
- Billat, P.-A.; Roger, E.; Faure, S.; Lagarce, F. Models for drug absorption from the small intestine: Where are we and where are we going? Drug Discov. Today 2017, 22, 761–775. [Google Scholar] [CrossRef] [PubMed]
- Salah-Abbès, J.B.; Abbès, S.; Ouanes, Z.; Houas, Z.; Abdel-Wahhab, M.A.; Bacha, H.; Oueslati, R. Tunisian radish extract (Raphanus sativus) enhances the antioxidant status and protects against oxidative stress induced by zearalenone in Balb/c mice. J. Appl. Toxicol. 2008, 28, 6–14. [Google Scholar] [CrossRef]
- Marin, M.L.; Murtha, J.; Dong, W.; Pestka, J.J. Effects of mycotoxins on cytokine production and proliferation in EL-4 thymoma cells. J. Toxicol. Environ. Health 1996, 48, 379–396. [Google Scholar] [CrossRef] [PubMed]
- Pleadin, J.; Babić, J.; Vulić, A.; Kudumija, N.; Aladić, K.; Kiš, M.; Jaki Tkalec, V.; Škrivanko, M.; Lolić, M.; Šubarić, D. The effect of thermal processing on the reduction of deoxynivalenol and zearalenone cereal content. Croat. J. Food Sci. Technol. 2019, 11, 44–51. [Google Scholar] [CrossRef]
- Luo, X.; Qi, L.; Liu, Y.; Wang, R.; Yang, D.; Li, K.; Wang, L.; Li, Y.; Zhang, Y.; Chen, Z. Effects of electron beam irradiation on zearalenone and ochratoxin a in naturally contaminated corn and corn quality parameters. Toxins 2017, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Zhai, Y.; Qi, L.; Pan, L.; Wang, J.; Xing, J.; Wang, R.; Wang, L.; Zhang, Q.; Yang, K. Influences of Electron Beam Irradiation on the Physical and Chemical Properties of Zearalenone-and Ochratoxin A-Contaminated Corn and In Vivo Toxicity Assessment. Foods 2020, 9, 376. [Google Scholar] [CrossRef]
- Calado, T.; Abrunhosa, L.; Cabo Verde, S.; Alté, L.; Venâncio, A.; Fernández-Cruz, M.L. Effect of Gamma-Radiation on Zearalenone—Degradation, Cytotoxicity and Estrogenicity. Foods 2020, 9, 1687. [Google Scholar] [CrossRef]
- Hojnik, N.; Modic, M.; Tavčar-Kalcher, G.; Babič, J.; Walsh, J.L.; Cvelbar, U. Mycotoxin Decontamination Efficacy of Atmospheric Pressure Air Plasma. Toxins 2019, 11, 219. [Google Scholar] [CrossRef] [PubMed]
- Drzymala, S.S.; Weiz, S.; Heinze, J.; Marten, S.; Prinz, C.; Zimathies, A.; Garbe, L.-A.; Koch, M. Automated solid-phase extraction coupled online with HPLC-FLD for the quantification of zearalenone in edible oil. Anal. Bioanal. Chem. 2015, 407, 3489–3497. [Google Scholar] [CrossRef] [PubMed]
- Ok, H.E.; Choi, S.-W.; Kim, M.; Chun, H.S. HPLC and UPLC methods for the determination of zearalenone in noodles, cereal snacks and infant formula. Food Chem. 2014, 163, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Senyuva, H.Z.; Gilbert, J.; Türköz, G.; Leeman, D.; Donnelly, C. Analysis of Deoxynivalenol, Zearalenone, T-2, and HT-2 Toxins in Animal Feed by LC/MS/MS–A Critical Comparison of Immunoaffinity Column Cleanup with No Cleanup. J. AOAC Int. 2012, 95, 1701–1708. [Google Scholar] [CrossRef]
- Vendl, O.; Berthiller, F.; Crews, C.; Krska, R. Simultaneous determination of deoxynivalenol, zearalenone, and their major masked metabolites in cereal-based food by LC–MS–MS. Anal. Bioanal. Chem. 2009, 395, 1347–1354. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Moltó, J.C.; Berrada, H.; Mañes, J. A survey of trichothecenes, zearalenone and patulin in milled grain-based products using GC–MS/MS. Food Chem. 2014, 146, 212–219. [Google Scholar] [CrossRef]
- Wang, G.; Chen, W.; Hu, J.; Fan, B.; Shi, J.; Xu, J. Preparative isolation and purification of zearalenone from rice culture by combined use of macroporous resin column and high-speed counter-current chromatography. J. Chromatogr. B 2019, 1110, 43–50. [Google Scholar] [CrossRef]
- Anfossi, L.; Di Nardo, F.; Cavalera, S.; Giovannoli, C.; Spano, G.; Speranskaya, E.S.; Goryacheva, I.Y.; Baggiani, C. A lateral flow immunoassay for straightforward determination of fumonisin mycotoxins based on the quenching of the fluorescence of CdSe/ZnS quantum dots by gold and silver nanoparticles. Microchim. Acta 2018, 185, 94. [Google Scholar] [CrossRef]
- Yang, Y.; Li, W.; Shen, P.; Liu, R.; Li, Y.; Xu, J.; Zheng, Q.; Zhang, Y.; Li, J.; Zheng, T. Aptamer fluorescence signal recovery screening for multiplex mycotoxins in cereal samples based on photonic crystal microsphere suspension array. Sens. Actuators B Chem. 2017, 248, 351–358. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, X.; Wen, K.; Li, C.; Mujtaba Mari, G.; Jiang, H.; Shi, W.; Shen, J.; Wang, Z. Multiplex lateral flow immunoassays based on amorphous carbon nanoparticles for detecting three Fusarium mycotoxins in maize. J. Agric. Food Chem. 2017, 65, 8063–8071. [Google Scholar] [CrossRef] [PubMed]
- Guan, D.; Li, P.; Zhang, Q.; Zhang, W.; Zhang, D.; Jiang, J. An ultra-sensitive monoclonal antibody-based competitive enzyme immunoassay for aflatoxin M1 in milk and infant milk products. Food Chem. 2011, 125, 1359–1364. [Google Scholar] [CrossRef]
- Li, X.; Li, P.; Zhang, Q.; Li, R.; Zhang, W.; Zhang, Z.; Ding, X.; Tang, X. Multi-component immunochromatographic assay for simultaneous detection of aflatoxin B1, ochratoxin A and zearalenone in agro-food. Biosens. Bioelectron. 2013, 49, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, N.; Ning, B.; Liu, M.; Lv, Z.; Sun, Z.; Peng, Y.; Chen, C.; Li, J.; Gao, Z. Simultaneous and rapid detection of six different mycotoxins using an immunochip. Biosens. Bioelectron. 2012, 34, 44–50. [Google Scholar] [CrossRef]
- Zhang, J.; Xia, Y.-K.; Chen, M.; Wu, D.-Z.; Cai, S.-X.; Liu, M.-M.; He, W.-H.; Chen, J.-H. A fluorescent aptasensor based on DNA-scaffolded silver nanoclusters coupling with Zn (II)-ion signal-enhancement for simultaneous detection of OTA and AFB1. Sens. Actuators B Chem. 2016, 235, 79–85. [Google Scholar] [CrossRef]
- Li, C.; Mi, T.; Oliveri Conti, G.; Yu, Q.; Wen, K.; Shen, J.; Ferrante, M.; Wang, Z. Development of a screening fluorescence polarization immunoassay for the simultaneous detection of fumonisins B1 and B2 in maize. J. Agric. Food Chem. 2015, 63, 4940–4946. [Google Scholar] [CrossRef]
- Li, C.; Wen, K.; Mi, T.; Zhang, X.; Zhang, H.; Zhang, S.; Shen, J.; Wang, Z. A universal multi-wavelength fluorescence polarization immunoassay for multiplexed detection of mycotoxins in maize. Biosens. Bioelectron. 2016, 79, 258–265. [Google Scholar] [CrossRef]
- Kolosova, A.Y.; De Saeger, S.; Sibanda, L.; Verheijen, R.; Van Peteghem, C. Development of a colloidal gold-based lateral-flow immunoassay for the rapid simultaneous detection of zearalenone and deoxynivalenol. Anal. Bioanal. Chem. 2007, 389, 2103–2107. [Google Scholar] [CrossRef]
- Lattanzio, V.M.; Nivarlet, N. Multiplex dipstick immunoassay for semiquantitative determination of fusarium mycotoxins in oat. In Oat; Humana Press: New York, NY, USA, 2017; pp. 137–142. [Google Scholar]
- Wang, Y.; Ning, B.; Peng, Y.; Bai, J.; Liu, M.; Fan, X.; Sun, Z.; Lv, Z.; Zhou, C.; Gao, Z. Application of suspension array for simultaneous detection of four different mycotoxins in corn and peanut. Biosens. Bioelectron. 2013, 41, 391–396. [Google Scholar] [CrossRef]
- Liu, G.; Han, Z.; Nie, D.; Yang, J.; Zhao, Z.; Zhang, J.; Li, H.; Liao, Y.; Song, S.; De Saeger, S. Rapid and sensitive quantitation of zearalenone in food and feed by lateral flow immunoassay. Food Control 2012, 27, 200–205. [Google Scholar] [CrossRef]
- Jin, Y.; Chen, Q.; Luo, S.; He, L.; Fan, R.; Zhang, S.; Yang, C.; Chen, Y. Dual near-infrared fluorescence-based lateral flow immunosensor for the detection of zearalenone and deoxynivalenol in maize. Food Chem. 2020, 336, 127718. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, J.; Yi, C.; Jiang, L.; Wu, J.; Chen, X.; Shen, X.; Sun, Y.; Lei, H. A smartphone-based quantitative detection device integrated with latex microsphere immunochromatography for on-site detection of zearalenone in cereals and feed. Sens. Actuators B Chem. 2019, 290, 170–179. [Google Scholar] [CrossRef]
- Ren, X.; Zhang, Q.; Wu, W.; Yan, T.; Tang, X.; Zhang, W.; Yu, L.; Li, P. Anti-idiotypic nanobody-phage display-mediated real-time immuno-PCR for sensitive, simultaneous and quantitative detection of total aflatoxins and zearalenone in grains. Food Chem. 2019, 297, 124912. [Google Scholar] [CrossRef] [PubMed]
- Caglayan, M.O.; Üstündağ, Z. Detection of zearalenone in an aptamer assay using attenuated internal reflection ellipsometry and it’s cereal sample applications. Food Chem. Toxicol. 2020, 136, 111081. [Google Scholar] [CrossRef] [PubMed]
- Rychlik, M.; Humpf, H.-U.; Marko, D.; Dänicke, S.; Mally, A.; Berthiller, F.; Klaffke, H.; Lorenz, N. Proposal of a comprehensive definition of modified and other forms of mycotoxins including “masked” mycotoxins. Mycotoxin Res. 2014, 30, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef]
- Plasencia, J.; Mirocha, C.J. Isolation and characterization of zearalenone sulfate produced by Fusarium spp. Appl. Environ. Microbiol. 1991, 57, 146–150. [Google Scholar] [CrossRef]
- Poppenberger, B.; Berthiller, F.; Lucyshyn, D.; Sieberer, T.; Schuhmacher, R.; Krska, R.; Kuchler, K.; Glössl, J.; Luschnig, C.; Adam, G. Detoxification of the Fusarium mycotoxin deoxynivalenol by a UDP-glucosyltransferase from Arabidopsis thaliana. J. Biol. Chem. 2003, 278, 47905–47914. [Google Scholar] [CrossRef]
- Berthiller, F.; Schuhmacher, R.; Adam, G.; Krska, R. Formation, determination and significance of masked and other conjugated mycotoxins. Anal. Bioanal. Chem. 2009, 395, 1243–1252. [Google Scholar] [CrossRef]
- Coleman, J.; Blake-Kalff, M.; Davies, E. Detoxification of xenobiotics by plants: Chemical modification and vacuolar compartmentation. Trends Plant Sci. 1997, 2, 144–151. [Google Scholar] [CrossRef]
- He, J.; Zhou, T.; Young, J.C.; Boland, G.J.; Scott, P.M. Chemical and biological transformations for detoxification of trichothecene mycotoxins in human and animal food chains: A review. Trends Food Sci. Technol. 2010, 21, 67–76. [Google Scholar] [CrossRef]
- Engelhardt, G.; Zill, G.; Wohner, B.; Wallnöfer, P.R. Transformation of the Fusarium mycotoxin zearalenone in maize cell suspension cultures. Naturwissenschaften 1988, 75, 309–310. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Werner, U.; Sulyok, M.; Krska, R.; Hauser, M.-T.; Schuhmacher, R. Liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) determination of phase II metabolites of the mycotoxin zearalenone in the model plant Arabidopsis thaliana. Food Addit. Contam. 2006, 23, 1194–1200. [Google Scholar] [CrossRef] [PubMed]
- Vendl, O.; Crews, C.; MacDonald, S.; Krska, R.; Berthiller, F. Occurrence of free and conjugated Fusarium mycotoxins in cereal-based food. Food Addit. Contam. 2010, 27, 1148–1152. [Google Scholar] [CrossRef]
- Huang, L.C.; Zheng, N.; Zheng, B.Q.; Wen, F.; Cheng, J.B.; Han, R.W.; Xu, X.M.; Li, S.L.; Wang, J.Q. Simultaneous determination of aflatoxin M1, ochratoxin A, zearalenone and α-zearalenol in milk by UHPLC–MS/MS. Food Chem. 2014, 146, 242–249. [Google Scholar] [CrossRef]
- Han, Z.; Ren, Y.; Zhou, H.; Luan, L.; Cai, Z.; Wu, Y. A rapid method for simultaneous determination of zearalenone, α-zearalenol, β-zearalenol, zearalanone, α-zearalanol and β-zearalanol in traditional Chinese medicines by ultra-high-performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2011, 879, 411–420. [Google Scholar] [CrossRef]
- Belhassen, H.; Jiménez-Díaz, I.; Ghali, R.; Ghorbel, H.; Molina-Molina, J.M.; Olea, N.; Hedili, A. Validation of a UHPLC–MS/MS method for quantification of zearalenone, α-zearalenol, β-zearalenol, α-zearalanol, β-zearalanol and zearalanone in human urine. J. Chromatogr. B 2014, 962, 68–74. [Google Scholar] [CrossRef]
- Grabley, S.; Gareis, M.; Böckers, W.; Thiem, J. Glycosylation of mycotoxins. Synthesis 1992, 1992, 1078–1080. [Google Scholar] [CrossRef]
- Berthiller, F.; Krska, R.; Domig, K.J.; Kneifel, W.; Juge, N.; Schuhmacher, R.; Adam, G. Hydrolytic fate of deoxynivalenol-3-glucoside during digestion. Toxicol. Lett. 2011, 206, 264–267. [Google Scholar] [CrossRef]
- Mahato, D.K.; Lee, K.E.; Kamle, M.; Devi, S.; Dewangan, K.; Kumar, P.; Kang, S.G. Aflatoxins in food and feed: An overview on prevalence, detection and control strategies. Front. Microbiol. 2019, 10, 2266. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Sharma, B.; Borah, R.; Haque, S.; Mahmud, M.M.C.; Shah, A.K.; Rawal, D.; Bora, H.; Bui, S. Ochratoxins in food and feed: Occurrence and its impact on human health and management strategies. Toxicon 2020, 187, 151–162. [Google Scholar] [CrossRef]
- Kamle, M.; Mahato, D.K.; Devi, S.; Lee, K.E.; Kang, S.G.; Kumar, P. Fumonisins: Impact on agriculture, food, and human health and their management strategies. Toxins 2019, 11, 328. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, W.; Wu, H.; Zhang, W.; Mu, W. Identification of a Potent Enzyme for the Detoxification of Zearalenone. J. Agric. Food Chem. 2019, 68, 376–383. [Google Scholar] [CrossRef]
- Beloglazova, N.V.; De Boevre, M.; Goryacheva, I.Y.; Werbrouck, S.; Guo, Y.; De Saeger, S. Immunochemical approach for zearalenone-4-glucoside determination. Talanta 2013, 106, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Marín, S.; Sanchis, V.; De Saeger, S.; De Boevre, M. Hydrolysers of modified mycotoxins in maize: α-Amylase and cellulase induce an underestimation of the total aflatoxin content. Food Chem. 2018, 248, 86–92. [Google Scholar] [CrossRef] [PubMed]
- El-Sharkaway, S.H.; Selim, M.I.; Afifi, M.S.; Halaweish, F.T. Microbial transformation of zearalenone to a zearalenone sulfate. Appl. Environ. Microbiol. 1991, 57, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Kamimura, H. Conversion of zearalenone to zearalenone glycoside by Rhizopus sp. Appl. Environ. Microbiol. 1986, 52, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Boutigny, A.-L.; Richard-Forget, F.; Barreau, C. Natural mechanisms for cereal resistance to the accumulation of Fusarium trichothecenes. Eur. J. Plant Pathol. 2008, 121, 411–423. [Google Scholar] [CrossRef]
- EFSA. Opinion of the scientific panel on contaminants in the food chain on a request from the commission related to zearalenone as undesirable substance in animal feed. EFSA J. 2004, 89, 1–35. [Google Scholar] [CrossRef]
- Mirocha, C.J.; Pathre, S.V.; Robison, T.S. Comparative metabolism of zearalenone and transmission into bovine milk. Food Cosmet. Toxicol. 1981, 19, 25–30. [Google Scholar] [CrossRef]
- Olsen, M.; Mirocha, C.J.; Abbas, H.K.; Johansson, B. Metabolism of high concentrations of dietary zearalenone by young male turkey poults. Poult. Sci. 1986, 65, 1905–1910. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Dong, M.; Yang, Q.; Apaliya, M.T.; Li, J.; Zhang, X. Biodegradation of zearalenone by Saccharomyces cerevisiae: Possible involvement of ZEN responsive proteins of the yeast. J. Proteom. 2016, 143, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Numanoglu, E.; Yener, S.; Gökmen, V.; Uygun, U.; Koksel, H. Modelling thermal degradation of zearalenone in maize bread during baking. Food Addit. Contam. Part A 2013, 30, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Sun, C.; Xu, J.; Liu, D.; Han, Y.; Wu, S.; Luo, X. Detoxification of zearalenone from corn oil by adsorption of functionalized GO systems. Appl. Surf. Sci. 2018, 430, 198–207. [Google Scholar] [CrossRef]
- Wang, M.; Yin, L.; Hu, H.; Selvaraj, J.N.; Zhou, Y.; Zhang, G. Expression, functional analysis and mutation of a novel neutral zearalenone-degrading enzyme. Int. J. Biol. Macromol. 2018, 118, 1284–1292. [Google Scholar] [CrossRef]
- Wang, G.; Xi, Y.; Lian, C.; Sun, Z.; Zheng, S. Simultaneous detoxification of polar aflatoxin B1 and weak polar zearalenone from simulated gastrointestinal tract by zwitterionic montmorillonites. J. Hazard. Mater. 2019, 364, 227–237. [Google Scholar] [CrossRef]
- Olopade, B.K.; Oranusi, S.U.; Nwinyi, O.C.; Njobeh, P.B.; Lawal, I.A. Modification of montmorillonite clay with Cymbopogon citratus for the decontamination of zearalenone in millet. AIMS Agric. Food 2019, 4, 643–657. [Google Scholar] [CrossRef]
- Sun, Z.; Song, A.; Wang, B.; Wang, G.; Zheng, S. Adsorption behaviors of aflatoxin B1 and zearalenone by organo-rectorite modified with quaternary ammonium salts. J. Mol. Liq. 2018, 264, 645–651. [Google Scholar] [CrossRef]
- Marković, M.; Daković, A.; Rottinghaus, G.E.; Kragović, M.; Petković, A.; Krajišnik, D.; Milić, J.; Mercurio, M.; de Gennaro, B. Adsorption of the mycotoxin zearalenone by clinoptilolite and phillipsite zeolites treated with cetylpyridinium surfactant. Colloids Surf. B Biointerfaces 2017, 151, 324–332. [Google Scholar] [CrossRef]
- Sprynskyy, M.; Gadzała-Kopciuch, R.; Nowak, K.; Buszewski, B. Removal of zearalenone toxin from synthetics gastric and body fluids using talc and diatomite: A batch kinetic study. Colloids Surf. B Biointerfaces 2012, 94, 7–14. [Google Scholar] [CrossRef]
- Kalagatur, N.K.; Karthick, K.; Allen, J.A.; Nirmal Ghosh, O.S.; Chandranayaka, S.; Gupta, V.K.; Krishna, K.; Mudili, V. Application of activated carbon derived from seed shells of Jatropha curcas for decontamination of zearalenone mycotoxin. Front. Pharmacol. 2017, 8, 760. [Google Scholar] [CrossRef] [PubMed]
- Spasojević, M.P.; Dakovic, A.; Rottinghaus, G.E.; Mihajlović, A.S.R.; Marković, M.A.; Krajišnik, D.R. Zearalenone and ochratoxin A: Adsorption by kaolin modified with surfactant. Metall. Mater. Eng. 2019, 25, 39–45. [Google Scholar] [CrossRef]
- Yang, K.; Li, K.; Pan, L.; Luo, X.; Xing, J.; Wang, J.; Wang, L.; Wang, R.; Zhai, Y.; Chen, Z. Effect of Ozone and Electron Beam Irradiation on Degradation of Zearalenone and Ochratoxin A. Toxins 2020, 12, 138. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Li, Y.; Luo, X.; Wang, R.; Zheng, R.; Wang, L.; Li, Y.; Yang, D.; Fang, W.; Chen, Z. Detoxification of zearalenone and ochratoxin A by ozone and quality evaluation of ozonised corn. Food Addit. Contam. Part A 2016, 33, 1700–1710. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Y.; Ji, J.; Wu, H.; Pi, F.; Zhang, Y.; Sun, X. Chemical and toxicological alterations of zearalenone under ozone treatment. Food Addit. Contam. Part A 2019, 36, 163–174. [Google Scholar] [CrossRef]
- Alexandre, A.P.S.; Castanha, N.; Costa, N.S.; Santos, A.S.; Badiale-Furlong, E.; Augusto, P.E.D.; Calori-Domingues, M.A. Ozone technology to reduce zearalenone contamination in whole maize flour: Degradation kinetics and impact on quality. J. Sci. Food Agric. 2019, 99, 6814–6821. [Google Scholar] [CrossRef]
- Wang, N.; Wu, W.; Pan, J.; Long, M. Detoxification strategies for zearalenone using microorganisms: A review. Microorganisms 2019, 7, 208. [Google Scholar] [CrossRef]
- Chlebicz, A.; Śliżewska, K. In vitro detoxification of aflatoxin B 1, deoxynivalenol, fumonisins, T-2 toxin and zearalenone by probiotic bacteria from genus Lactobacillus and Saccharomyces cerevisiae yeast. Probiotics Antimicrob. Proteins 2020, 12, 289–301. [Google Scholar] [CrossRef]
- Sangsila, A.; Faucet-Marquis, V.; Pfohl-Leszkowicz, A.; Itsaranuwat, P. Detoxification of zearalenone by Lactobacillus pentosus strains. Food Control 2016, 62, 187–192. [Google Scholar] [CrossRef]
- Zhao, L.; Jin, H.; Lan, J.; Zhang, R.; Ren, H.; Zhang, X.; Yu, G. Detoxification of zearalenone by three strains of Lactobacillus plantarum from fermented food in vitro. Food Control 2015, 54, 158–164. [Google Scholar] [CrossRef]
- Yu, Y.; Qiu, L.; Wu, H.; Tang, Y.; Yu, Y.; Li, X.; Liu, D. Degradation of zearalenone by the extracellular extracts of Acinetobacter sp. SM04 liquid cultures. Biodegradation 2011, 22, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Tinyiro, S.E.; Wokadala, C.; Xu, D.; Yao, W. Adsorption and degradation of zearalenone by Bacillus strains. Folia Microbiol. 2011, 56, 321. [Google Scholar] [CrossRef] [PubMed]
- Harkai, P.; Szabó, I.; Cserháti, M.; Krifaton, C.; Risa, A.; Radó, J.; Balázs, A.; Berta, K.; Kriszt, B. Biodegradation of aflatoxin-B1 and zearalenone by Streptomyces sp. collection. Int. Biodeterior. Biodegrad. 2016, 108, 48–56. [Google Scholar] [CrossRef]
- Liu, C.; Chang, J.; Wang, P.; Yin, Q.; Huang, W.; Dang, X.; Lu, F.; Gao, T. Zearalenone biodegradation by the combination of probiotics with cell-free extracts of Aspergillus oryzae and its mycotoxin-alleviating effect on pig production performance. Toxins 2019, 11, 552. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, H.; Zhu, Z.; Ji, F.; Yin, X.; Hong, Q.; Shi, J. Isolation and characterization of Bacillus amyloliquefaciens ZDS-1: Exploring the degradation of Zearalenone by Bacillus spp. Food Control 2016, 68, 244–250. [Google Scholar] [CrossRef]
- Cho, K.J.; Kang, J.S.; Cho, W.T.; Lee, C.H.; Ha, J.K.; Song, K.B. In vitro degradation of zearalenone by Bacillus subtilis. Biotechnol. Lett. 2010, 32, 1921–1924. [Google Scholar] [CrossRef]
- Guo, Y.; Zhou, J.; Tang, Y.; Ma, Q.; Zhang, J.; Ji, C.; Zhao, L. Characterization and Genome Analysis of a Zearalenone-Degrading Bacillus velezensis Strain ANSB01E. Curr. Microbiol. 2020, 77, 273–278. [Google Scholar] [CrossRef]
- Xu, L.; Sun, X.; Wan, X.; Li, H.; Yan, F.; Han, R.; Li, H.; Li, Z.; Tian, Y.; Liu, X. Identification of a Bacillus amyloliquefaciens H6 Thioesterase Involved in Zearalenone Detoxification by Transcriptomic Analysis. J. Agric. Food Chem. 2020, 68, 10071–10080. [Google Scholar] [CrossRef]
- Sun, X.; He, X.; siyu Xue, K.; Li, Y.; Xu, D.; Qian, H. Biological detoxification of zearalenone by Aspergillus niger strain FS10. Food Chem. Toxicol. 2014, 72, 76–82. [Google Scholar] [CrossRef]
- Kriszt, R.; Krifaton, C.; Szoboszlay, S.; Cserháti, M.; Kriszt, B.; Kukolya, J.; Czéh, Á.; Fehér-Tóth, S.; Török, L.; Szőke, Z. A new zearalenone biodegradation strategy using non-pathogenic Rhodococcus pyridinivorans K408 strain. PLoS ONE 2012, 7, e43608. [Google Scholar] [CrossRef]
- Chang, X.; Liu, H.; Sun, J.; Wang, J.; Zhao, C.; Zhang, W.; Zhang, J.; Sun, C. Zearalenone Removal from Corn Oil by an Enzymatic Strategy. Toxins 2020, 12, 117. [Google Scholar] [CrossRef] [PubMed]
- Kosawang, C.; Karlsson, M.; Vélëz, H.; Rasmussen, P.H.; Collinge, D.B.; Jensen, B.; Jensen, D.F. Zearalenone detoxification by zearalenone hydrolase is important for the antagonistic ability of Clonostachys rosea against mycotoxigenic Fusarium graminearum. Fungal Biol. 2014, 118, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Garcia, S.O.; Feltrin, A.C.P.; Garda-Buffon, J. Zearalenone reduction by commercial peroxidase enzyme and peroxidases from soybean bran and rice bran. Food Addit. Contam. Part A 2018, 35, 1819–1831. [Google Scholar] [CrossRef]
- Azam, M.S.; Yu, D.; Liu, N.; Wu, A. Degrading Ochratoxin A and Zearalenone Mycotoxins Using a Multifunctional Recombinant Enzyme. Toxins 2019, 11, 301. [Google Scholar] [CrossRef] [PubMed]
- Janssen, E.M.; Mourits, M.C.M.; van der Fels-Klerx, H.J.; Lansink, A.G.J.M.O. Factors underlying Dutch farmers’ intentions to adapt their agronomic management to reduce Fusarium species infection in wheat. PLoS ONE 2020, 15, e0237460. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.F.; Lilly, M.; Rheeder, J.P.; Burger, H.-M.; Shephard, G.S.; Gelderblom, W.C.A. Technological and community-based methods to reduce mycotoxin exposure. Food Control 2017, 73, 101–109. [Google Scholar] [CrossRef]
- Adebiyi, J.A.; Kayitesi, E.; Adebo, O.A.; Changwa, R.; Njobeh, P.B. Food fermentation and mycotoxin detoxification: An African perspective. Food Control 2019, 106, 106731. [Google Scholar] [CrossRef]
- Janssen, E.M.; Liu, C.; Van der Fels-Klerx12, H.J. Fusarium infection and trichothecenes in barley and its comparison with wheat. World Mycotoxin J. 2018, 11, 33–46. [Google Scholar] [CrossRef]
- Hofer, K.; Barmeier, G.; Schmidhalter, U.; Habler, K.; Rychlik, M.; Hückelhoven, R.; Hess, M. Effect of nitrogen fertilization on Fusarium head blight in spring barley. Crop Prot. 2016, 88, 18–27. [Google Scholar] [CrossRef]
- Schöneberg, T.; Martin, C.; Wettstein, F.E.; Bucheli, T.D.; Mascher, F.; Bertossa, M.; Musa, T.; Keller, B.; Vogelgsang, S. Fusarium and mycotoxin spectra in Swiss barley are affected by various cropping techniques. Food Addit. Contam. Part A 2016, 33, 1608–1619. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.F.; Ameye, M.; De Saeger, S.; Audenaert, K.; Haesaert, G. Biological control of mycotoxigenic fungi and their toxins: An update for the pre-harvest approach. In Mycotoxins-Impact and Management Strategies; IntechOpen: London, UK, 2018. [Google Scholar]
- Abdallah, M.F.; De Boevre, M.; Landschoot, S.; De Saeger, S.; Haesaert, G.; Audenaert, K. Fungal endophytes control Fusarium graminearum and reduce trichothecenes and zearalenone in maize. Toxins 2018, 10, 493. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Aldred, D. Post-harvest control strategies: Minimizing mycotoxins in the food chain. Int. J. Food Microbiol. 2007, 119, 131–139. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahato, D.K.; Devi, S.; Pandhi, S.; Sharma, B.; Maurya, K.K.; Mishra, S.; Dhawan, K.; Selvakumar, R.; Kamle, M.; Mishra, A.K.; et al. Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review. Toxins 2021, 13, 92. https://doi.org/10.3390/toxins13020092
Mahato DK, Devi S, Pandhi S, Sharma B, Maurya KK, Mishra S, Dhawan K, Selvakumar R, Kamle M, Mishra AK, et al. Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review. Toxins. 2021; 13(2):92. https://doi.org/10.3390/toxins13020092
Chicago/Turabian StyleMahato, Dipendra Kumar, Sheetal Devi, Shikha Pandhi, Bharti Sharma, Kamlesh Kumar Maurya, Sadhna Mishra, Kajal Dhawan, Raman Selvakumar, Madhu Kamle, Awdhesh Kumar Mishra, and et al. 2021. "Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review" Toxins 13, no. 2: 92. https://doi.org/10.3390/toxins13020092
APA StyleMahato, D. K., Devi, S., Pandhi, S., Sharma, B., Maurya, K. K., Mishra, S., Dhawan, K., Selvakumar, R., Kamle, M., Mishra, A. K., & Kumar, P. (2021). Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review. Toxins, 13(2), 92. https://doi.org/10.3390/toxins13020092









