Mycotoxins in Pistachios (Pistacia vera L.): Methods for Determination, Occurrence, Decontamination
Abstract
1. Introduction
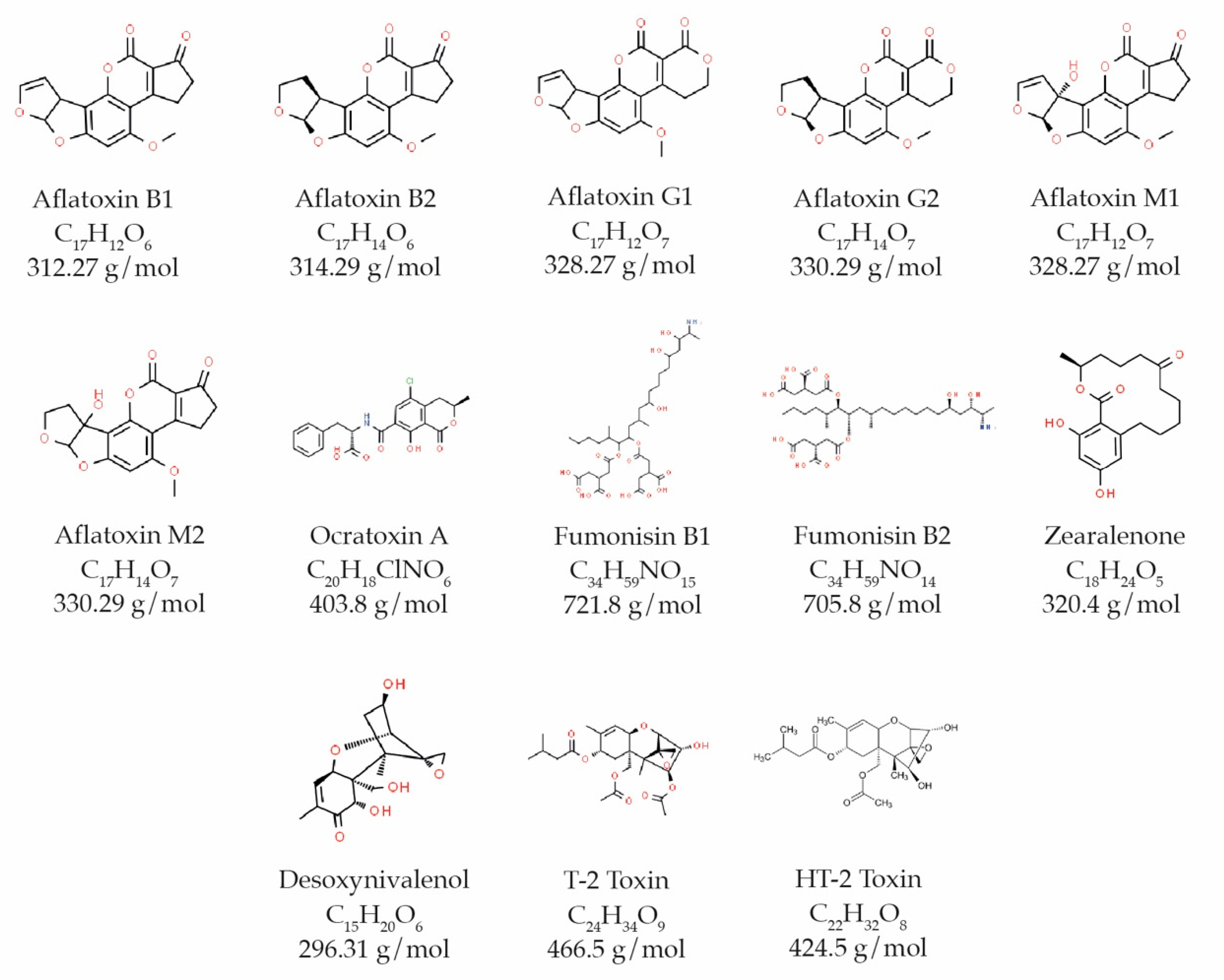
2. Aflatoxins
2.1. Physical and Chemical Characteristics
2.2. Toxicokinetics
2.3. Toxicity
2.3.1. Acute Toxicity
2.3.2. Chronic Toxicity
3. Ochratoxin A (OTA)
4. Fumonisins (FB1 and FB2)
5. Zearalenone (ZEA)
6. Trichothecenes
6.1. Desoxynivalenol (DON)
6.2. HT-2 Toxin and T-2 Toxin
7. Emerging mycotoxins
8. Analytical Methods for the Determination of Mycotoxins
8.1. Detection and Quantification
8.1.1. Chromatographic Techniques
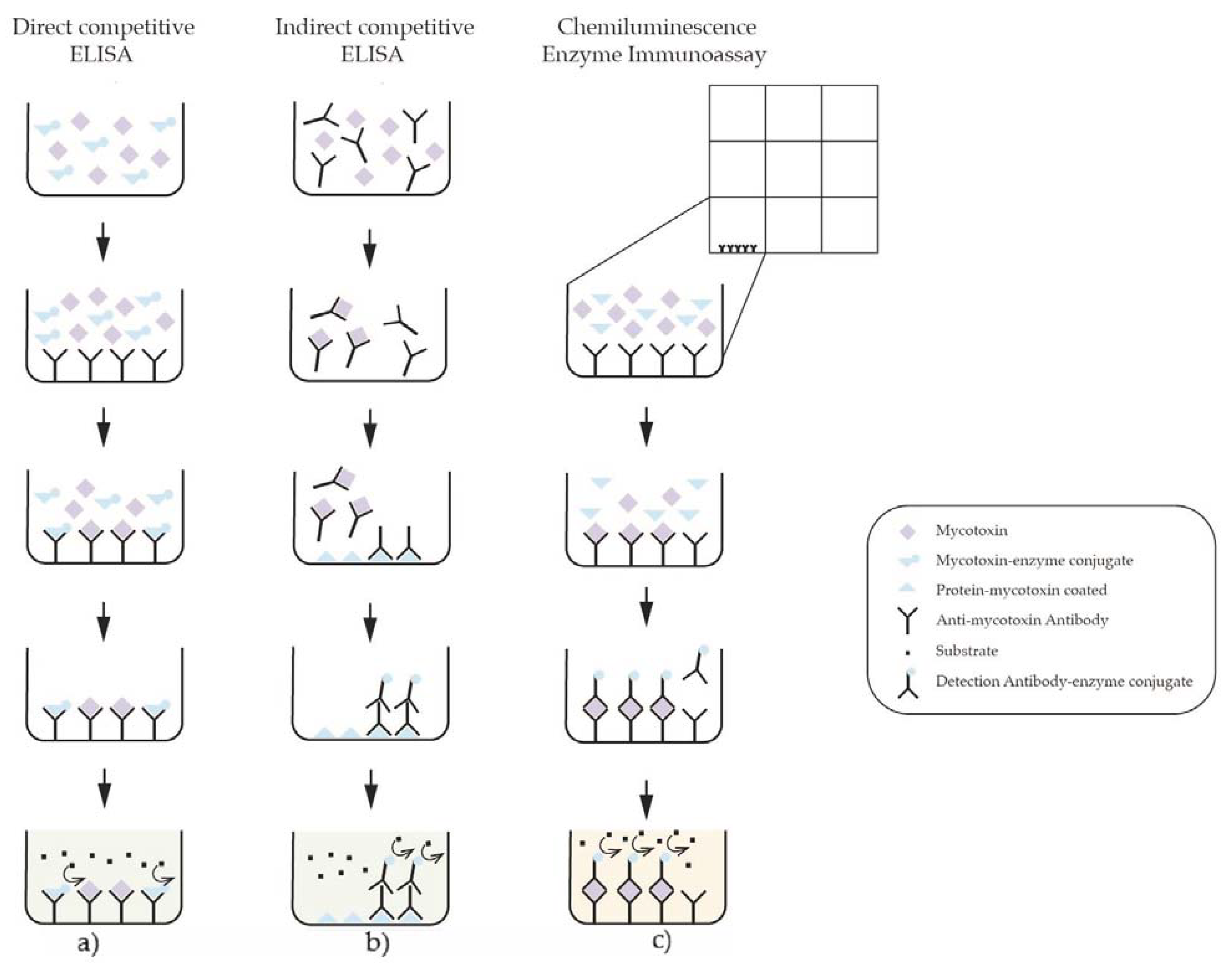
8.1.2. Immunoassays
8.1.3. Biosensors
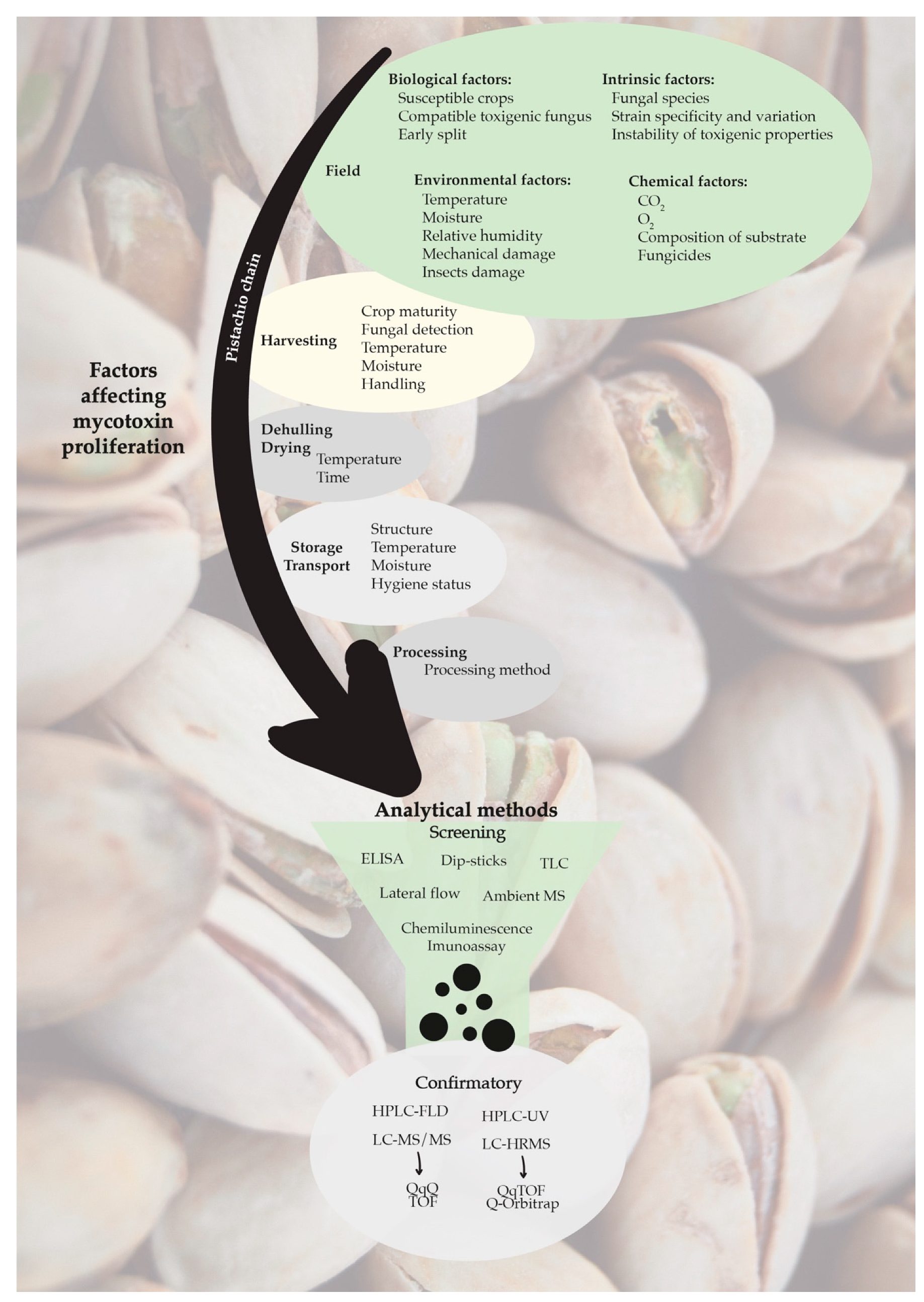
9. Occurrence of Mycotoxins in Pistachios
10. Biomonitoring
11. Prevention and Control
12. Decontamination
12.1. Physical Decontamination
12.2. Chemical Decontamination
12.3. Biological Decontamination
13. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean Diet, its Components, and Cardiovascular Disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef]
- Shahbandeh, M. Consumption of Pistachios Worldwide from 2011/2012 to 2019/2021. Available online: https://www.statista.com/statistics/932999/pistachio-consumption-global/ (accessed on 30 August 2021).
- Shahbandeh, M. Per Capita Consumption of Pistachios (Shelled) in the United States from 2000 to 2020. Available online: https://www.statista.com/statistics/964218/per-capita-consumption-of-pistachios-in-the-us/ (accessed on 30 August 2021).
- INE. Consumo Humano de Frutos per Capita (kg/hab.) por Espécie Frutícola. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_indicadores&indOcorrCod=0000166&selTab=tab0&xlang=pt (accessed on 11 May 2021).
- PortFIR (Plataforma Portuguesa de Informação Alimentar). Pistachio Nutrional Composition. Available online: http://portfir.insa.pt/foodcomp/food?21237 (accessed on 11 May 2021).
- Kashaninejad, M.; Tabil, L.G. Pistachio (Pistacia vera L.). In Postharvest Postharvest Biology and Technology of Tropical and Subtropical Fruits; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 4, pp. 218–247. ISBN 9780857090904. [Google Scholar]
- Dreher, M.L. Pistachio nuts: Composition and potential health benefits. Nutr. Rev. 2012, 70, 234–240. [Google Scholar] [CrossRef]
- FAO STAT. Pistachio World Production. Available online: http://www.fao.org/faostat/en/?#data/QC (accessed on 3 March 2021).
- Bui-Klimke, T.R.; Guclu, H.; Kensler, T.W.; Yuan, J.M.; Wu, F. Aflatoxin regulations and global pistachio trade: Insights from social network analysis. PLoS ONE 2014, 9, e92149. [Google Scholar] [CrossRef]
- Mendes, M.M.; Santos, M.; Soares, A. A segurança alimentar dos frutos secos e secados colocados no mercado, face aos resultados do Plano Nacional de Colheita de Amostras da ASAE. ASAE-Riscos e Aliment. 2016, 11, 5–9. [Google Scholar]
- Mycotoxins, C.A.S.T. Mycotoxins: Risks in Plant, Animal, and Human Systems Council for Agricultural Science and Technology; Council of Agricultural Science and Technology: Ames, IA, USA, 2003; ISBN 1887383220. [Google Scholar]
- Sataque Ono, E.Y.; Hirooka, E.Y.; Rossi, C.N.; Ono, M.A. Mycotoxins in Seeds and Nuts. In Nuts and Seeds in Health and Disease Prevention; Elsevier: San Diego, CA, USA, 2011; pp. 121–127. ISBN 9780123756886. [Google Scholar]
- Aiko, V.; Mehta, A. Occurrence, detection and detoxification of mycotoxins. J. Biosci. 2015, 40, 943–954. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, G.; Wu, D.; Liu, J.; Li, X.; Luo, P.; Hu, N.; Wang, H.; Wu, Y. Recent advances on toxicity and determination methods of mycotoxins in foodstuffs. Trends Food Sci. Technol. 2020, 96, 233–252. [Google Scholar] [CrossRef]
- Silva, A.S.; Brites, C.; Pouca, A.V.; Barbosa, J.; Freitas, A. UHPLC-ToF-MS method for determination of multi-mycotoxins in maize: Development and validation. Curr. Res. Food Sci. 2019, 1, 1–7. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Morales, H.; Soares, C.; Calado, T.; Vila-Chã, A.S.; Pereira, M.; Venâncio, A. A Review of Mycotoxins in Food and Feed Products in Portugal and Estimation of Probable Daily Intakes. Crit. Rev. Food Sci. Nutr. 2016, 56, 249–265. [Google Scholar] [CrossRef] [PubMed]
- Magnussen, A.; Parsi, M.A. Aflatoxins, hepatocellular carcinoma and public health. World J. Gastroenterol. 2013, 19, 1508–1512. [Google Scholar] [CrossRef] [PubMed]
- Šegvić Klarić, M. Adverse Effects of Combined Mycotoxins. Arch. Ind. Hyg. Toxicol. 2012, 63, 519–530. [Google Scholar] [CrossRef]
- Smith, M.-C.; Madec, S.; Coton, E.; Hymery, N. Natural Co-Occurrence of Mycotoxins in Foods and Feeds and Their in vitro Combined Toxicological Effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef]
- Williams, J.H.; Phillips, T.D.; Jolly, P.E.; Stiles, J.K.; Jolly, C.M.; Aggarwal, D. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr. 2004, 80, 1106–1122. [Google Scholar] [CrossRef]
- FAO/WHO Codex Alimentarius Comission. Codex Alimentarius; Food and Agriculture Organization of the United Nations: Rome, Italy, 1995. [Google Scholar]
- EFSA. Opinion of the scientific panel on contaminants in the food chain on a request from the comission related to the potential increase of consumer health risk by a possible increase of the existing maximum levels for aflatoxins in almonds hazelnuts and deriv. EFSA J. 2007, 446, 1–127. [Google Scholar]
- Cheraghali, A.M.; Yazdanpanah, H.; Doraki, N.; Abouhossain, G.; Hassibi, M.; Ali-abadi, S.; Aliakbarpoor, M.; Amirahmadi, M.; Askarian, A.; Fallah, N.; et al. Incidence of aflatoxins in Iran pistachio nuts. Food Chem. Toxicol. 2007, 45, 812–816. [Google Scholar] [CrossRef]
- Varga, E.; Glauner, T.; Berthiller, F.; Krska, R.; Schuhmacher, R.; Sulyok, M. Development and validation of a (semi-)quantitative UHPLC-MS/MS method for the determination of 191 mycotoxins and other fungal metabolites in almonds, hazelnuts, peanuts and pistachios. Anal. Bioanal. Chem. 2013, 405, 5087–5104. [Google Scholar] [CrossRef]
- Siegel, J.P.; Kuenen, L.P.S.B. Variable Developmental Rate and Survival of Navel Orangeworm (Lepidoptera: Pyralidae) on Pistachio. J. Econ. Entomol. 2011, 104, 532–539. [Google Scholar] [CrossRef]
- Doster, M.A. Aspergillus Molds and Aflatoxins in Pistachio Nuts in California. Phytopathology 1994, 84, 583. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A Global Concern for Food Safety, Human Health and Their Management. Front. Microbiol. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Kumar, V.V. Aflatoxins: Properties, Toxicity and Detoxification. Nutr. Food Sci. Int. J. 2018, 6, 555696. [Google Scholar] [CrossRef]
- Fletcher, M.T.; Blaney, B.J. Mycotoxins. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–7. ISBN 9780081005965. [Google Scholar]
- Nazhand, A.; Durazzo, A.; Lucarini, M.; Souto, E.B.; Santini, A. Characteristics, occurrence, detection and detoxification of aflatoxins in foods and feeds. Foods 2020, 9, 644. [Google Scholar] [CrossRef]
- Benkerroum, N. Aflatoxins: Producing-molds, structure, health issues and incidence in southeast asian and sub-saharan african countries. Int. J. Environ. Res. Public Health 2020, 17, 1215. [Google Scholar] [CrossRef]
- IARC. Aflatoxins IARC Monographs. Int. Agency Res. Cancer 2012, 100F, 225–248. [Google Scholar]
- Rushing, B.R.; Selim, M.I. Aflatoxin B1: A review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem. Toxicol. 2019, 124, 81–100. [Google Scholar] [CrossRef]
- Wacoo, A.P.; Wendiro, D.; Vuzi, P.C.; Hawumba, J.F. Methods for Detection of Aflatoxins in Agricultural Food Crops. J. Appl. Chem. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Kew, M.C. Aflatoxins as a Cause of Hepatocellular Carcinoma. J. Gastrointestin. Liver Dis. 2013, 22, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.; Nebbia, C.S.; et al. Risk assessment of aflatoxins in food. EFSA J. 2020, 18, e06040. [Google Scholar] [CrossRef] [PubMed]
- Alshannaq, A.; Yu, J.H. Occurrence, toxicity, and analysis of major mycotoxins in food. Int. J. Environ. Res. Public Health 2017, 14, 632. [Google Scholar] [CrossRef]
- Bhat, R.; Rai, R.V.; Karim, A.A. Mycotoxins in Food and Feed: Present Status and Future Concerns. Compr. Rev. Food Sci. Food Saf. 2010, 9, 57–81. [Google Scholar] [CrossRef]
- Ostry, V.; Malir, F.; Toman, J.; Grosse, Y. Mycotoxins as human carcinogens—the IARC Monographs classification. Mycotoxin Res. 2017, 33, 65–73. [Google Scholar] [CrossRef]
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R.; Diseases, L.; Angeles, L.; Angeles, L.; Angeles, L.; et al. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef]
- Schrenk, D.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.; Nebbia, C.S.; Nielsen, E.; et al. Risk assessment of ochratoxin A in food. EFSA J. 2020, 18, e06113. [Google Scholar] [CrossRef]
- Duarte, S.C.; Pena, A.; Lino, C.M. A review on ochratoxin A occurrence and effects of processing of cereal and cereal derived food products. Food Microbiol. 2010, 27, 187–198. [Google Scholar] [CrossRef]
- Ringot, D.; Chango, A.; Schneider, Y.J.; Larondelle, Y. Toxicokinetics and toxicodynamics of ochratoxin A, an update. Chem. Biol. Interact. 2006, 159, 18–46. [Google Scholar] [CrossRef]
- De Ruyck, K.; de Boevre, M.; Huybrechts, I.; de Saeger, S. Dietary mycotoxins, co-exposure, and carcinogenesis in humans: Short review. Mutat. Res.-Rev. Mutat. Res. 2015, 766, 32–41. [Google Scholar] [CrossRef]
- IARC. Chemical and physical characteristics of the principal mycotoxins. In IARC Scientific Publications; IARC: Lyon, France, 2012; Volume 2, pp. 31–38. [Google Scholar]
- Al-Jaal, B.A.; Jaganjac, M.; Barcaru, A.; Horvatovich, P.; Latiff, A. Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018. Food Chem. Toxicol. 2019, 129, 211–228. [Google Scholar] [CrossRef]
- Bansal, J.; Pantazopoulos, P.; Tam, J.; Cavlovic, P.; Kwong, K.; Turcotte, A.M.; Lau, B.P.Y.; Scott, P.M. Surveys of rice sold in canada for aflatoxins, ochratoxin a and fumonisins. Food Addit. Contam.-Part A Chem. Anal. Control. Expo. Risk Assess. 2011, 28, 767–774. [Google Scholar] [CrossRef]
- FAO/WHO. Safety Evaluation of Certain Contaminants in Food: Prepared by the Eighty-Third Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA); World Health Organization and Food and Agriculture Organization of the United Nations: Geneva, Switzerland, 2018; Volume 74, ISBN 9789241660747. [Google Scholar]
- Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Risks to human and animal health related to the presence of deoxynivalenol and its acetylated and modified forms in food and feed. EFSA J. 2017, 15, e04718. [Google Scholar] [CrossRef]
- Vidal, A.; Marín, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Determination of aflatoxins, deoxynivalenol, ochratoxin A and zearalenone in wheat and oat based bran supplements sold in the Spanish market. Food Chem. Toxicol. 2013, 53, 133–138. [Google Scholar] [CrossRef]
- Knutsen, H.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Appropriateness to set a group health based guidance value for T2 and HT2 toxin and its modified forms. EFSA J. 2017, 15, 1–53. [Google Scholar] [CrossRef]
- Arcella, D.; Gergelova, P.; Innocenti, M.L.; Steinkellner, H. Human and animal dietary exposure to T-2 and HT-2 toxin. EFSA J. 2017, 15, e04972. [Google Scholar] [CrossRef]
- Vaclavikova, M.; Macmahon, S.; Zhang, K.; Begley, T.H. Application of single immunoaffinity clean-up for simultaneous determination of regulated mycotoxins in cereals and nuts. Talanta 2013, 117, 345–351. [Google Scholar] [CrossRef]
- Gruber-Dorninger, C.; Novak, B.; Nagl, V.; Berthiller, F. Emerging Mycotoxins: Beyond Traditionally Determined Food Contaminants. J. Agric. Food Chem. 2017, 65, 7052–7070. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Analysis and Detection of Major Mycotoxins in Foods. Foods 2020, 9, 518. [Google Scholar] [CrossRef]
- Cole, R.J. Toxin from Fusarium moniliforme: Effects on Plants and Animals. Science 1973, 179, 1324–1326. [Google Scholar] [CrossRef]
- Jestoi, M. Emerging Fusarium -Mycotoxins Fusaproliferin, Beauvericin, Enniatins, And Moniliformin—A Review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef]
- Fraeyman, S.; Croubels, S.; Devreese, M.; Antonissen, G. Emerging Fusarium and Alternaria Mycotoxins: Occurrence, Toxicity and Toxicokinetics. Toxins 2017, 9, 228. [Google Scholar] [CrossRef]
- Liao, C.; Wong, J.W.; Zhang, K.; Yang, P.; Wittenberg, J.B.; Trucksess, M.W.; Hayward, D.G.; Lee, N.S.; Chang, J.S. Multi-mycotoxin Analysis of Finished Grain and Nut Products Using Ultrahigh-Performance Liquid Chromatography and Positive Electrospray Ionization—Quadrupole Orbital Ion Trap High-Resolution Mass Spectrometry. J. Agric. food Chem. 2015, 63, 8314–8332. [Google Scholar] [CrossRef]
- Battilani, P.; Costa, L.G.; Dossena, A.; Gullino, M.L.; Marchelli, R.; Galaverna, G.; Pietri, A.; Dall’Asta, C.; Giorni, P.; Spadaro, D.; et al. Scientific information on mycotoxins and natural plant toxicants. Agronnova 2008, 1, 1–467. [Google Scholar] [CrossRef]
- Cunha, S.C.; Sá, S.V.M.; Fernandes, J.O. Multiple mycotoxin analysis in nut products: Occurrence and risk characterization. Food Chem. Toxicol. 2018, 114, 260–269. [Google Scholar] [CrossRef]
- Mbundi, L.; Gallar-Ayala, H.; Khan, M.R.; Barber, J.L.; Losada, S.; Busquets, R. Advances in the Analysis of Challenging Food Contaminants: Nanoparticles, Bisphenols, Mycotoxins, and Brominated Flame Retardants. In Advances in Molecular Toxicology; Fishbein, J.C., Heilman, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 8, pp. 35–105. [Google Scholar]
- Sinha, K.K. Testing methods for aflatoxins in foods. Food Nutr. Bull. 1999, 20, 458–464. [Google Scholar] [CrossRef]
- Vargas Medina, D.A.; Bassolli Borsatto, J.V.; Maciel, E.V.S.; Lanças, F.M. Current role of modern chromatography and mass spectrometry in the analysis of mycotoxins in food. TrAC Trends Anal. Chem. 2021, 135, 116156. [Google Scholar] [CrossRef]
- Spanjer, M.C.; Rensen, P.M.; Scholten, J.M. LC–MS/MS multi-method for mycotoxins after single extraction, with validation data for peanut, pistachio, wheat, maize, cornflakes, raisins and figs. Food Addit. Contam.-Part A Chem. Anal. Control. Expo. Risk Assess. 2008, 25, 472–489. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, Y.; Saito, K.; Hanioka, N.; Narimatsu, S.; Kataoka, H. Determination of aflatoxins in food samples by automated on-line in-tube solid-phase microextraction coupled with liquid chromatography-mass spectrometry. J. Chromatogr. A 2009, 1216, 4416–4422. [Google Scholar] [CrossRef]
- Fernane, F.; Sanchis, V.; Marín, S.; Ramos, A.J. First Report on Mould and Mycotoxin Contamination of Pistachios Sampled in Algeria. Mycopathologia 2010, 170, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Ulca, P.; Evcimen, M.K.; Senyuva, H.Z. Surveys of aflatoxin B1 contamination of retail Turkish foods and of products intended for export between 2007 and 2009. Food Addit. Contam. Part B Surveill. 2010, 3, 120–125. [Google Scholar] [CrossRef]
- Shadbad, S.; Reza, M.; Masoud, A.; Ali, T.; Faranak, G.; Mahboob, N. Determination of aflatoxins in nuts of Tabriz confectionaries by ELISA and HPLC methods. Adv. Pharm. Bull. 2012, 2, 123–126. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; Huertas-Pérez, J.F.; Gámiz-Gracia, L.; García-Campaña, A.M. A new approach in sample treatment combined with UHPLC-MS/MS for the determination of multiclass mycotoxins in edible nuts and seeds. Talanta 2013, 115, 61–67. [Google Scholar] [CrossRef]
- El Tawila, M.M.; Neamatallah, A.; Serdar, S.A. Incidence of aflatoxins in commercial nuts in the holy city of Mekkah. Food Control 2013, 29, 121–124. [Google Scholar] [CrossRef]
- Ostadrahimi, A.; Ashrafnejad, F.; Kazemi, A.; Sargheini, N.; Mahdavi, R.; Farshchian, M.; Mahluji, S. Aflatoxin in raw and salt-roasted nuts (pistachios, peanuts and walnuts) sold in markets of Tabriz, Iran. Jundishapur J. Microbiol. 2014, 7, 1–4. [Google Scholar] [CrossRef]
- Diella, G.; Caggiano, G.; Ferrieri, F.; Ventrella, A.; Palma, M.; Napoli, C.; Rutigliano, S.; Lopuzzo, M.; Lovero, G.; Montagna, M.T. Aflatoxin contamination in nuts marketed in Italy: Preliminary results. Ann. di Ig. 2018, 30, 401–409. [Google Scholar] [CrossRef]
- Alcántara-Durán, J.; Moreno-González, D.; García-Reyes, J.F.; Molina-Díaz, A. Use of a modified QuEChERS method for the determination of mycotoxin residues in edible nuts by nano flow liquid chromatography high resolution mass spectrometry. Food Chem. 2019, 279, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Alsharif, A.M.A.; Choo, Y.M.; Tan, G.H. Detection of five mycotoxins in different food matrices in the malaysian market by using validated liquid chromatography electrospray ionization triple quadrupole mass spectrometry. Toxins 2019, 11, 196. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Ruiz, J.L.; Romero-González, R.; Martínez Vidal, J.L.; Garrido Frenich, A. Determination of mycotoxins in nuts by ultra high-performance liquid chromatography-tandem mass spectrometry: Looking for a representative matrix. J. Food Compos. Anal. 2019, 82, 103228. [Google Scholar] [CrossRef]
- Narváez, A.; Rodríguez-Carrasco, Y.; Castaldo, L.; Izzo, L.; Graziani, G.; Ritieni, A. Occurrence and exposure assessment of mycotoxins in ready-to-eat tree nut products through ultra-high performance liquid chromatography coupled with high resolution q-orbitrap mass spectrometry. Metabolites 2020, 10, 344. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.Z.; Richard, J.L.; Binder, J. A review of rapid methods for the analysis of mycotoxins. Mycopathologia 2006, 161, 261–273. [Google Scholar] [CrossRef] [PubMed]
- FAO. Training in mycotoxins analysis. In Manuals of Food Quality Control; Food and Agriculture Organization of the United Nations: Rome, Italy, 1990; p. 128. ISBN 9251029474. [Google Scholar]
- Katz, S.E.; Siewierski, M. Drug residue analysis using immunoaffinity chromatography. J. Chromatogr. A 1992, 624, 403–409. [Google Scholar] [CrossRef]
- Laranjeiro, C.; Lino, C.; Pena, A. Ocorrência de Zearalenona em Águas de Superfície em Portugal Ocorrência de Zearalenona em Águas de Superfície em Portugal; Faculty of Pharmacy: Coimbra, Portugal, 2015. [Google Scholar]
- Pereira, V.L.; Fernandes, J.O.; Cunha, S.C. Mycotoxins in cereals and related foodstuffs: A review on occurrence and recent methods of analysis. Trends Food Sci. Technol. 2014, 36, 96–136. [Google Scholar] [CrossRef]
- Perestrelo, R.; Silva, P.; Porto-Figueira, P.; Pereira, J.A.M.; Silva, C.; Medina, S.; Câmara, J.S. QuEChERS-Fundamentals, relevant improvements, applications and future trends. Anal. Chim. Acta 2019, 1070, 1–28. [Google Scholar] [CrossRef]
- Desmarchelier, A.; Tessiot, S.; Bessaire, T.; Racault, L.; Fiorese, E.; Urbani, A.; Chan, W.-C.; Cheng, P.; Mottier, P. Combining the quick, easy, cheap, effective, rugged and safe approach and clean-up by immunoaffinity column for the analysis of 15 mycotoxins by isotope dilution liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2014, 1337, 75–84. [Google Scholar] [CrossRef]
- Bessaire, T.; Mujahid, C.; Mottier, P.; Desmarchelier, A. Multiple Mycotoxins Determination in Food by LC-MS/MS: An International Collaborative Study. Toxins 2019, 11, 658. [Google Scholar] [CrossRef]
- Wang, Y. Occurrence and co-occurrence of mycotoxins in nuts and dried fruits from China. Food Control 2018, 88, 181–189. [Google Scholar] [CrossRef]
- Rejczak, T.; Tuzimski, T. A review of recent developments and trends in the QuEChERS sample preparation approach. Open Chem. 2015, 13, 980–1010. [Google Scholar] [CrossRef]
- Romero-González, R.; Garrido Frenich, A.; Martínez Vidal, J.L.; Prestes, O.D.; Grio, S.L. Simultaneous determination of pesticides, biopesticides and mycotoxins in organic products applying a quick, easy, cheap, effective, rugged and safe extraction procedure and ultra-high performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 1477–1485. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-M.; Wu, Y.-L.; Lu, Y.-B. Simultaneous determination of carbamate insecticides and mycotoxins in cereals by reversed phase liquid chromatography tandem mass spectrometry using a quick, easy, cheap, effective, rugged and safe extraction procedure. J. Chromatogr. B 2013, 915–916, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Lacina, O.; Zachariasova, M.; Urbanova, J.; Vaclavikova, M.; Cajka, T.; Hajslova, J. Critical assessment of extraction methods for the simultaneous determination of pesticide residues and mycotoxins in fruits, cereals, spices and oil seeds employing ultra-high performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 2012, 1262, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Banerjee, K. A Review: Sample Preparation and Chromatographic Technologies for Detection of Aflatoxins in Foods. Toxins 2020, 12, 539. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.; Walker, J. Principles and Techniques of Biochemistry and Molecular Biology, 7th ed.; Wilson, K., Walker, J., Eds.; Cambridge University Press: Cambridge, UK, 2010; ISBN 9788578110796. [Google Scholar]
- Malik, A.K.; Blasco, C.; Picó, Y. Liquid chromatography-mass spectrometry in food safety. J. Chromatogr. A 2010, 1217, 4018–4040. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wong, J.W.; Krynitsky, A.J.; Trucksess, M.W. Determining Mycotoxins in Baby Foods and Animal Feeds Using Stable Isotope Dilution and Liquid Chromatography Tandem Mass Spectrometry. J. Agric. Food Chem. 2014, 62, 8935–8943. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Dall’Asta, C.; Schuhmacher, R.; Lemmens, M.; Adam, G.; Krska, R. Masked Mycotoxins: Determination of a Deoxynivalenol Glucoside in Artificially and Naturally Contaminated Wheat by Liquid Chromatography−Tandem Mass Spectrometry. J. Agric. Food Chem. 2005, 53, 3421–3425. [Google Scholar] [CrossRef]
- Zhai, A. Determination of Mycotoxins in Peanuts With Enhanced Matrix Removal—Lipid by LC/MS/MS. Agil. Technol. Appl. Note 2016, 5991–8962, 1–8. [Google Scholar]
- Chu, F.S. Immunoassays for analysis of mycotoxins. J. Food Prot. 1984, 47, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.W.; Bramhmbhatt, H.; Szabo-Vezse, M.; Poma, A.; Coker, R.; Piletsky, S.A. Analytical methods for determination of mycotoxins: An update (2009–2014). Anal. Chim. Acta 2015, 901, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Leszczyńska, J.; MasŁowska, J.; Owczarek, A.; Kucharska, U. Determination of aflatoxins in food products by the ELISA method. Czech. J. Food Sci. 2018, 19, 8–12. [Google Scholar] [CrossRef]
- Mehan, V.K.; Gowda, C.L.L. (Eds.) Aflatoxin Contamination Problems in Groundnut in Asia; International Crops Research Institute for the Semi-Arid Tropics: Patancheru, India, 1997; ISBN 929066360X. [Google Scholar]
- Ono, E.Y.S.; Bordini, J.G.; Omori, A.M.; Hirozawa, M.T.; Ono, M.A. Métodos imunológicos para a detecção de fungos toxigênicos e micotoxinas em grãos, alimentos e rações. In Biotecnologia Aplicada à Agro&Indústria; Editora Blucher: São Paulo, Brazil, 2017; Volume 4, pp. 895–936. ISBN 9788521211150. [Google Scholar]
- Lee, N.A.; Wang, S.; Allan, R.D.; Kennedy, I.R. A Rapid Aflatoxin B 1 ELISA: Development and Validation with Reduced Matrix Effects for Peanuts, Corn, Pistachio, and Soybeans. J. Agric. Food Chem. 2004, 52, 2746–2755. [Google Scholar] [CrossRef] [PubMed]
- Bensassi, F.; Rhouma, A.; Ghrab, M.; Bacha, H.; Rabeh Hajlaoui, M. Evaluation of cultivar susceptibility and storage periods towards aflatoxin B1 contamination on pistachio nuts. Mycotoxin Res. 2010, 26, 199–203. [Google Scholar] [CrossRef]
- Sapsford, K.E.; Taitt, C.R.; Fertig, S.; Moore, M.H.; Lassman, M.E.; Maragos, C.M.; Shriver-Lake, L.C. Indirect competitive immunoassay for detection of aflatoxin B1 in corn and nut products using the array biosensor. Biosens. Bioelectron. 2006, 21, 2298–2305. [Google Scholar] [CrossRef]
- Kong, D.; Xie, Z.; Liu, L.; Song, S.; Kuang, H.; Cui, G.; Xu, C. Development of indirect competitive ELISA and lateral-flow immunochromatographic assay strip for the detection of sterigmatocystin in cereal products. Food Agric. Immunol. 2017, 28, 260–273. [Google Scholar] [CrossRef]
- Azri, F.; Sukor, R.; Selamat, J.; Abu Bakar, F.; Yusof, N.; Hajian, R. Electrochemical Immunosensor for Detection of Aflatoxin B1 Based on Indirect Competitive ELISA. Toxins 2018, 10, 196. [Google Scholar] [CrossRef]
- Azer, M.; Cooper, C. Determination of aflatoxins in foods using HPLC and a commercial ELISA system. J. Food Prot. 1991, 54, 291–294. [Google Scholar] [CrossRef]
- Xu, W.; Han, X.; Li, F.; Zhang, L. Natural Occurrence of Alternaria Toxins in the 2015 Wheat from Anhui Province, China. Toxins 2016, 8, 308. [Google Scholar] [CrossRef]
- Rahman, H.U.; Yue, X.; Yu, Q.; Xie, H.; Zhang, W.; Zhang, Q.; Li, P. Specific antigen-based and emerging detection technologies of mycotoxins. J. Sci. Food Agric. 2019, 99, 4869–4877. [Google Scholar] [CrossRef]
- Yu, S.; Yu, F.; Liu, L.; Zhang, H.; Zhang, Z.; Qu, L.; Wu, Y. Which one of the two common reporter systems is more suitable for chemiluminescent enzyme immunoassay: Alkaline phosphatase or horseradish peroxidase? Lumin. Biol. Chem. Lumin. 2016, 31, 888–892. [Google Scholar] [CrossRef]
- Freitas, A.; Barros, S.; Brites, C.; Barbosa, J.; Silva, A.S. Validation of a Biochip Chemiluminescent Immunoassay for Multi-Mycotoxins Screening in Maize (Zea mays L.). Food Anal. Methods 2019, 12, 2675–2684. [Google Scholar] [CrossRef]
- Kaminiaris, M.D.; Mavrikou, S.; Georgiadou, M.; Paivana, G.; Tsitsigiannis, D.I.; Kintzios, S. An Impedance Based Electrochemical Immunosensor for Aflatoxin B1 Monitoring in Pistachio Matrices. Chemosensors 2020, 8, 121. [Google Scholar] [CrossRef]
- Orina, I.; Manley, M.; Williams, P.J. Non-destructive techniques for the detection of fungal infection in cereal grains. Food Res. Int. 2017, 100, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Paghaleh, S.J.; Askari, H.R.; Marashi, S.M.B.; Rahimi, M.; Bahrampour, A.R. A method for the measurement of in line pistachio aflatoxin concentration based on the laser induced fluorescence spectroscopy. J. Lumin. 2015, 161, 135–141. [Google Scholar] [CrossRef]
- Wu, Q.; Xu, H. Application of multiplexing fiber optic laser induced fluorescence spectroscopy for detection of aflatoxin B1 contaminated pistachio kernels. Food Chem. 2019, 290, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Valasi, L.; Georgiadou, M.; Tarantilis, P.A.; Yanniotis, S.; Pappas, C.S. Rapid screening on aflatoxins’ presence in Pistachia vera nuts using diffuse reflectance infrared Fourier transform spectroscopy and chemometrics. J. Food Sci. Technol. 2021, 58, 356–365. [Google Scholar] [CrossRef]
- JECFA. Draft code of practice for the prevention and reduction of aflatoxin contamination in tree nuts comments at step 6. In Microseismic Monitoring and Geomechanical Modelling of CO2 Storage in Subsurface Reservoirs; Springer: Cham, Switzerland, 2005; pp. 1–8. ISBN 9783642253874. [Google Scholar]
- Baazeem, A.; Garcia-Cela, E.; Medina, A.; Magan, N. Interacting Abiotic Factors Affect Growth and Aflatoxin B1 Production Profiles of Aspergillus flavus Strains on Pistachio-Based Matrices and Pistachio Nuts. Front. Microbiol. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Miraglia, M.; Marvin, H.J.P.; Kleter, G.A.; Battilani, P.; Brera, C.; Coni, E.; Cubadda, F.; Croci, L.; de Santis, B.; Dekkers, S.; et al. Climate change and food safety: An emerging issue with special focus on Europe. Food Chem. Toxicol. 2009, 47, 1009–1021. [Google Scholar] [CrossRef]
- Coronel, M.B.; Marín, S.; Cano-Sancho, G.; Ramos, A.J.; Sanchis, V. Exposure assessment to ochratoxin A in Catalonia (Spain) based on the consumption of cereals, nuts, coffee, wine, and beer. Food Addit. Contam. Part A 2012, 29, 979–993. [Google Scholar] [CrossRef]
- Set, E.; Erkmen, O. The aflatoxin contamination of ground red pepper and pistachio nuts sold in Turkey. Food Chem. Toxicol. 2010, 48, 2532–2537. [Google Scholar] [CrossRef]
- Kulahi, A.; Kabak, B. A preliminary assessment of dietary exposure of ochratoxin A in Central Anatolia Region, Turkey. Mycotoxin Res. 2020, 36, 327–337. [Google Scholar] [CrossRef]
- Probst, C.; Njapau, H.; Cotty, P.J. Outbreak of an acute aflatoxicosis in Kenya in 2004: Identification of the causal agent. Appl. Environ. Microbiol. 2007, 73, 2762–2764. [Google Scholar] [CrossRef]
- Tan, K. Aflatoxin and Its Toxic Tragedies in Kenya. J. Young Investig. 2020, 38, 10–12. [Google Scholar] [CrossRef]
- JECFA. Evaluation of Certain Food Additives and Contaminants: Sixty-Eighth Report of the Joint FAO/WHO Expert Committee on Food Additives; Springer US: Geneva, Switzerland, 2007. [Google Scholar]
- Fernane, F.; Cano-Sancho, G.; Sanchis, V.; Marin, S.; Ramos, A.J. Aflatoxins and ochratoxin A in pistachios sampled in Spain: Occurrence and presence of mycotoxigenic fungi. Food Addit. Contam. Part B Surveill. 2010, 3, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Juan, C.; Mojemmi, B.; Moltó, J.C.; Bouklouze, A.; Cherrah, Y.; Idrissi, L.; Aouad, R.E.; Mañes, J. Incidence of ochratoxin A in rice and dried fruits from Rabat and Salé area, Morocco. Food Addit. Contam. 2007, 24, 285–291. [Google Scholar] [CrossRef]
- Tolosa, J.; Font, G.; Mañes, J.; Ferrer, E. Nuts and dried fruits: Natural occurrence of emerging Fusarium mycotoxins. Food Control 2013, 33, 215–220. [Google Scholar] [CrossRef]
- Martins, C.; Vidal, A.; de Boevre, M.; de Saeger, S.; Nunes, C.; Torres, D.; Goios, A.; Lopes, C.; Assunção, R.; Alvito, P. Exposure assessment of Portuguese population to multiple mycotoxins: The human biomonitoring approach. Int. J. Hyg. Environ. Health 2019, 222, 913–925. [Google Scholar] [CrossRef] [PubMed]
- JECFA. Evaluation of Certain Contaminants in Food: Eighty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives; WHO/FAO: Geneva, Switzerland, 2017; ISBN 9789241210027. [Google Scholar]
- Yin, Y.; Yan, L.; Jiang, J.; Ma, Z. Biological control of aflatoxin contamination of crops. J. Zhejiang Univ. Sci. B 2008, 9, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Doster, M.A.; Cotty, P.J.; Michailides, T.J. Evaluation of the Atoxigenic Aspergillus flavus Strain AF36 in Pistachio Orchards. Plant Dis. 2014, 98, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Moral, J.; Garcia-Lopez, M.T.; Camiletti, B.X.; Jaime, R.; Michailides, T.J.; Bandyopadhyay, R.; Ortega-Beltran, A. Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products. Agronomy 2020, 10, 491. [Google Scholar] [CrossRef]
- Michailides, T.J.; Puckett, R.D.; Moral, J.; García-Lopez, M.T.; Felts, D.; Doster, L.; Jaime-Garcia, R. Factors Affecting the Efficacy of AF36 Improvement of the Biocontrol Agent and Monitoring Commercial Applications. Calif. Pist. Res. Board 2018, 4, 1–3. [Google Scholar]
- JECFA. Safety Evaluation of Certain Contaminants in food: Prepared by the Eighty-Third Meeting of the Joint FAO/WHO Expert Committee on Food Additives; WHO Food Additives Series; No. 74, FAO JECFA Monographs 19 bis: Geneva, Switzerland, 2018; ISBN 9789241660747. [Google Scholar]
- Marín, S.; Ramos, A.J.; Sanchis, V. Modelling Aspergillus flavus growth and aflatoxins production in pistachio nuts. Food Microbiol. 2012, 32, 378–388. [Google Scholar] [CrossRef]
- Aldars-García, L.; Ramos, A.J.; Sanchis, V.; Marín, S. An attempt to model the probability of growth and aflatoxin B1 production of Aspergillus flavus under non-isothermal conditions in pistachio nuts. Food Microbiol. 2015, 51, 117–129. [Google Scholar] [CrossRef]
- Kaminiaris, M.D.; Camardo Leggieri, M.; Tsitsigiannis, D.I.; Battilani, P. AFLA-PISTACHIO: Development of a Mechanistic Model to Predict the Aflatoxin Contamination of Pistachio Nuts. Toxins 2020, 12, 445. [Google Scholar] [CrossRef]
- Marín, S.; Hodžić, I.; Ramos, A.J.; Sanchis, V. Predicting the growth/no-growth boundary and ochratoxin A production by Aspergillus carbonarius in pistachio nuts. Food Microbiol. 2008, 25, 683–689. [Google Scholar] [CrossRef]
- Richard-Forget, F.; Atanasova, V.; Chéreau, S. Using metabolomics to guide strategies to tackle the issue of the contamination of food and feed with mycotoxins: A review of the literature with specific focus on Fusarium mycotoxins. Food Control 2021, 121, 107610. [Google Scholar] [CrossRef]
- Park, D.L. Effect of processing on Aflatoxin. In Mycotoxins and Food Safety; DeVries, J.W., Trucksess, M.W., Jackson, L.S., Eds.; Springer: Boston, MA, USA, 2002; pp. 173–179. [Google Scholar]
- Yazdanpanah, H.; Mohammadi, T.; Abouhossain, G.; Cheraghali, A.M. Effect of roasting on degradation of Aflatoxins in contaminated pistachio nuts. Food Chem. Toxicol. 2005, 43, 1135–1139. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, I.; Orfi, M.; Shamma, M. Effect of gamma radiation on the inactivation of aflatoxin B1 in food and feed crops. Brazilian J. Microbiol. 2008, 39, 787–791. [Google Scholar] [CrossRef]
- Akbas, M.Y.; Ozdemir, M. Effect of different ozone treatments on aflatoxin degradation and physicochemical properties of pistachios. J. Sci. Food Agric. 2006, 86, 2099–2104. [Google Scholar] [CrossRef]
- Velazhahan, R.; Vijayanandraj, S.; Vijayasamundeeswari, A.; Paranidharan, V.; Samiyappan, R.; Iwamoto, T.; Friebe, B.; Muthukrishnan, S. Detoxification of aflatoxins by seed extracts of the medicinal plant, Trachyspermum ammi (L.) Sprague ex Turrill-Structural analysis and biological toxicity of degradation product of aflatoxin G1. Food Control 2010, 21, 719–725. [Google Scholar] [CrossRef]
- Vijayanandraj, S.; Brinda, R.; Kannan, K.; Adhithya, R.; Vinothini, S.; Senthil, K.; Chinta, R.R.; Paranidharan, V.; Velazhahan, R. Detoxification of aflatoxin B1 by an aqueous extract from leaves of Adhatoda vasica Nees. Microbiol. Res. 2014, 169, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Iram, W.; Anjum, T.; Iqbal, M.; Ghaffar, A.; Abbas, M. Mass spectrometric identification and toxicity assessment of degraded products of aflatoxin B1 and B2 by Corymbia citriodora aqueous extracts. Sci. Rep. 2015, 5, 14672. [Google Scholar] [CrossRef]
- Iram, W.; Anjum, T.; Iqbal, M.; Ghaffar, A.; Abbas, M.; Khan, A.M. Structural Analysis and Biological Toxicity of Aflatoxins B1 and B2 Degradation Products Following Detoxification by Ocimum basilicum and Cassia fistula Aqueous Extracts. Front. Microbiol. 2016, 7, 1–18. [Google Scholar] [CrossRef]
- Ansari, F.; Khodaiyan, F.; Rezaei, K.; Rahmani, A. Modelling of aflatoxin G1 reduction by kefir grain using response surface methodology. J. Environ. Heal. Sci. Eng. 2015, 13, 40. [Google Scholar] [CrossRef][Green Version]
- Farzaneh, M.; Shi, Z.-Q.; Ghassempour, A.; Sedaghat, N.; Ahmadzadeh, M.; Mirabolfathy, M.; Javan-Nikkhah, M. Aflatoxin B1 degradation by Bacillus subtilis UTBSP1 isolated from pistachio nuts of Iran. Food Control 2012, 23, 100–106. [Google Scholar] [CrossRef]
- Rahaie, S.; Emam-Djomeh, Z.; Razavi, S.H.; Mazaheri, M. Immobilized Saccharomyces cerevisiae as a potential aflatoxin decontaminating agent in pistachio nuts. Braz. J. Microbiol. 2010, 41, 82–90. [Google Scholar] [CrossRef]
- Rastegar, H.; Shoeibi, S.; Yazdanpanah, H.; Amirahmadi, M.; Khaneghah, A.M.; Campagnollo, F.B.; Sant’Ana, A.S. Removal of aflatoxin B1 by roasting with lemon juice and/or citric acid in contaminated pistachio nuts. Food Control 2017, 71, 279–284. [Google Scholar] [CrossRef]
- Ismail, A.; Gonçalves, B.L.; de Neeff, D.V.; Ponzilacqua, B.; Coppa, C.F.S.C.; Hintzsche, H.; Sajid, M.; Cruz, A.G.; Corassin, C.H.; Oliveira, C.A.F. Aflatoxin in foodstuffs: Occurrence and recent advances in decontamination. Food Res. Int. 2018, 113, 74–85. [Google Scholar] [CrossRef]
- Mousavi Khaneghah, A.; Hashemi Moosavi, M.; Oliveira, C.A.F.; Vanin, F.; Sant’Ana, A.S. Electron beam irradiation to reduce the mycotoxin and microbial contaminations of cereal-based products: An overview. Food Chem. Toxicol. 2020, 143, 111557. [Google Scholar] [CrossRef]
- Janić Hajnal, E.; Vukić, M.; Pezo, L.; Orčić, D.; Puač, N.; Škoro, N.; Milidrag, A.; Šoronja Simović, D. Effect of Atmospheric Cold Plasma Treatments on Reduction of Alternaria Toxins Content in Wheat Flour. Toxins 2019, 11, 704. [Google Scholar] [CrossRef]
- Bosch, L.; Pfohl, K.; Avramidis, G.; Wieneke, S.; Viöl, W.; Karlovsky, P. Plasma-Based Degradation of Mycotoxins Produced by Fusarium, Aspergillus and Alternaria Species. Toxins 2017, 9, 97. [Google Scholar] [CrossRef]
- Aron Maftei, N.; Ramos-Villarroel, A.Y.; Nicolau, A.I.; Martín-Belloso, O.; Soliva-Fortuny, R. Pulsed light inactivation of naturally occurring moulds on wheat grain. J. Sci. Food Agric. 2014, 94, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Adebo, O.A.; Molelekoa, T.; Makhuvele, R.; Adebiyi, J.A.; Oyedeji, A.B.; Gbashi, S.; Adefisoye, M.A.; Ogundele, O.M.; Njobeh, P.B. A review on novel non-thermal food processing techniques for mycotoxin reduction. Int. J. Food Sci. Technol. 2021, 56, 13–27. [Google Scholar] [CrossRef]
- Sipos, P.; Peles, F.; Brassó, D.L.; Béri, B.; Pusztahelyi, T.; Pócsi, I.; Győri, Z. Physical and Chemical Methods for Reduction in Aflatoxin Content of Feed and Food. Toxins 2021, 13, 204. [Google Scholar] [CrossRef] [PubMed]
- Udomkun, P.; Wiredu, A.N.; Nagle, M.; Müller, J.; Vanlauwe, B.; Bandyopadhyay, R. Innovative technologies to manage aflatoxins in foods and feeds and the profitability of application—A review. Food Control 2017, 76, 127–138. [Google Scholar] [CrossRef]
- Wu, Y.; Cheng, J.-H.; Sun, D.-W. Blocking and degradation of aflatoxins by cold plasma treatments: Applications and mechanisms. Trends Food Sci. Technol. 2021, 109, 647–661. [Google Scholar] [CrossRef]
- Yousefi, M.; Mohammadi, M.A.; Khajavi, M.Z.; Ehsani, A.; Scholtz, V. Application of Novel Non-Thermal Physical Technologies to Degrade Mycotoxins. J. Fungi 2021, 7, 395. [Google Scholar] [CrossRef] [PubMed]
- Tasouji, M.A. Inactivation of pistachio contaminant aspergillus flavus by atmospheric pressure capacitive coupled plasma (Ap-Ccp). J. Microbiol. Biotechnol. Food Sci. 2018, 8, 668–671. [Google Scholar] [CrossRef]
- Sen, Y.; Onal-Ulusoy, B.; Mutlu, M. Detoxification of hazelnuts by different cold plasmas and gamma irradiation treatments. Innov. Food Sci. Emerg. Technol. 2019, 54, 252–259. [Google Scholar] [CrossRef]
- Adibian, M. Aflatoxins in Pistachio, Detection and Prevention. J. Nov. Appl. Sci. 2016, 5, 27–33. [Google Scholar]
- Hadavi, E.; Feizi, H.; Gheibi, B. Aflatoxin-Contaminated Nut Separation by Applied Machinery and Processing Stages in Fresh Pistachio Processing Plant. Front. Microbiol. 2017, 8, 1–6. [Google Scholar] [CrossRef]
- Georgiadou, M.; Dimou, A.; Yanniotis, S. Aflatoxin contamination in pistachio nuts: A farm to storage study. Food Control 2012, 26, 580–586. [Google Scholar] [CrossRef]
- Doster, M.A.; Michailides, T.J. Relationship Between Shell Discoloration of Pistachio Nuts and Incidence of Fungal Decay and Insect Infestation. Plant. Dis. 1999, 83, 259–264. [Google Scholar] [CrossRef]
- Shakerardekani, A.; Karim, R.; Mirdamadiha, F. The Effect of Sorting on Aflatoxin Reduction of Pistachio Nuts. SSRN Electron. J. 2012, 10, 459–461. [Google Scholar] [CrossRef]
- McClure, W.F.; Farsaie, A. Dual-Wavelength Fiber Optic Photometer Measures Fluorescence of Aflatoxin Contaminated Pistachio Nuts. Trans. ASAE 1980, 23, 0204–0207. [Google Scholar] [CrossRef]
- Özlüoymak, Ö.; Güzel, E. Aflatoksinli ve Aflatoksinsiz Antep Fıstıkları Arasındaki Renk ve Kinetik Parametre Farklılıklarının Yapay Görme Sistemi Kullanılarak Belirlenmesi. Tekirdağ Ziraat Fakültesi Derg. 2020, 18, 157–168. [Google Scholar] [CrossRef]
- Yanniotis, S.; Proshlyakov, A.; Revithi, A.; Georgiadou, M.; Blahovec, J. X-ray imaging for fungal necrotic spot detection in pistachio nuts. Procedia Food Sci. 2011, 1, 379–384. [Google Scholar] [CrossRef][Green Version]
- Laokuldilok, N.; Thakeow, P.; Kopermsub, P.; Utama-ang, N. Optimisation of microencapsulation of turmeric extract for masking flavour. Food Chem. 2016, 194, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Christman, L.M.; Dean, L.L.; Bueno Almeida, C.; Weissburg, J.R. Acceptability of Peanut Skins as a Natural Antioxidant in Flavored Coated Peanuts. J. Food Sci. 2018, 83, 2571–2577. [Google Scholar] [CrossRef] [PubMed]
| Type of Sample | Analytes | Clean-Up Methods | Procedure of Extraction | Detector | Conditions | Analytical Column | Internal Standard | LOD (μg/kg) | LOQ (μg/kg) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Pistachio | AFB1; AFB2; AFG1; AFG2 | IAC | Sample quantity: 125 g Sample extraction: 475 mL MeOH/H2O/Hexane (63:16:21 v/v/v); filtration, dilution with water; IAC:10 mL PBS; 75 mL filtrate; wash 15 mL H2O, vacuum; elution with 0.5 mL MeOH | HPLC—FLD with PCD | Mobile phase: H2O/MeOH/ACN (42:29:17, v/v/v) Flow-rate: 1 mL/min Temperature column: Injection volume: 100 μL λexcitation: 365 nm λemission:450 mm | C18 250 mm × 4.6 mm 5 μm | - | 0.1–0.4 | - | [24] |
| Peanuts, pistachio, wheat, maize, cornflakes, raisins and figs | AFB1, AFB2, AFG1, AFG2, OTA, DON, FB1, FB2, T2, HT2, ZEA, CIT, etc | - | Sample quantity: 25 g Sample extration: 100 mL ACN/H2O (80:20 v/v), shaken 2 h, diluted 1 mL extract with 3 mL H2O, filtration | LC-MS/MS | Mobile phase:(A) H2O with 0.1% FA (B) ACN with 0.1%FA Gradient program: 90% A at 0 min, 30% A at 12 min, 10% A at 17.5 min, 90% at 21 min (t = 25 min) Flow-rate: 0.3 mL/min Ionization: ESI source in the positive mode Temperature column: 30 °C Injection volume: 20 μL Ionization: ESI source in the positive mode Capillary voltage: 2.5 kV Collision gas pressure: 0.8 bar Vaporizer temperature: 450 °C Sheath gas pressure: Auxiliary gas flow: 600 L/h | Alltima C18 150 mm × 3.2 mm 5 μm | - | 0.5–200 | 1–200 | [66] |
| Dried fruits (peanuts, walnut, cashews, pistachio, almond, pecan walnut), cereals, dehydrated fruits and spices | AFB1, AFB2, AFG1, AFG2 | SPME | Sample quantity: 0.5 g Sample extraction: 1 mL MeOH:H2O (80:20 v/v), centrifugation, filtration of supernatant and added to in-tube SPME | HPLC—MS | Mobile phase: MeOH/ACN (60:40, v/v):5 mM ammonium formate (45:55) Gradient program: After 8 min, washed with MeOH/ACN (60/40, v/v) for 2 min and returned to the initial conditions in 2 min Flow-rate: 1 mL/min Temperature column: 40 °C Injection volume: 10 μL Ionization: ESI source in the positive mode Capillary voltage: 2.5 kV Collision gas pressure: Vaporizer temperature: 350 °C Sheath gas pressure: 30 psi Auxiliary gas flow: 13 L/min | Zorbax Eclipse XD8-C8 150 mm × 4.6 mm 5 μm | AFM1 | 0.02 | 0.05 | [67] |
| Pistachios | AFB1, AFB2, AFG1, AFG2, OTA | IAC | Sample quantity: 5 g for AFs and 10 g for OTA Sample extraction: 30 mL ACN/H2O (60:40 v/v), belnded 10 min, 2 mL extract diluted with 48 mL PBS; Easi-extart AF IAC for AFs and Ochraprep IAC for OTA | HPLC—FLD with PCD | Mobile phase: ACN/H2O/acetic acid (51:47:2, v/v/v) Flow-rate: 1 mL/min Temperature column: 40 °C Injection volume: 100 μL λexcitation: 333 nm λemission:443 mm | Spherisorb ODS2 150 mm × 4.6 mm 5 μm | - | 0.2 | - | [68] |
| Pistachios, walnuts, cashews, almonds, peanuts, seeds, etc. | AFB1 | IAC | Sample quantity: 50 g Sample extraction:100 mL H2O + 4 g NaCl, 150 mL MeOH, filtration, 5 mL filtrate + 25 mL PBS; IAC:10 mL PBS, 30 mL filtrate, wash 15 mL H2O, elution 0.5 mL MeOH, 1 mL H2O; filtration if solution not clear. | HPLC—FLD with PCD | Mobile phase: H2O/MeOH/ACN (42:29:17, v/v/v) Flow-rate: 1 mL/min Temperature column: 40 °C Injection volume: λexcitation: 362 nm λemission:456 mm | Luna C18 25 cm × 4.6 mm, 5 μm | - | 0.2 | 0.6 | [69] |
| Almonds, walnuts, sunflower seeds, sesame seeds, peanuts, pistachios, hazelnuts and cashews | AFB1, AFB2, AFG1, AFG2, AFM1, AFM2 | IAC | Sample quantity: 10 g Sample extraction: 33% MeOH, filtration, 500 μL filtrate + 500 μL 33% MeOH | ELISA | Euroclon kit Absorbance at 450 nm | - | - | - | - | [70] |
| Sample quantity: 10 g Sample extraction: 1 g NaCl + 40 mL MeOH/H2O (80:20 v/v) + 20 mL n-hexane, blended for 3 min, eliminate n-hexane phase, filtration; 7 mL filtrate + 43 mL PBS; IAC: 10 mL PBS, 50 mL filtrate, wash 20 mL H2O, dried with air, elution 2 mL MeOH | HPLC- FLD with PCD | Mobile phase:ACN/MeOH/H2O (17:29:54, v/v/v) Flow-rate: 1 mL/min Temperature column: Injection volume: 20 mL λexcitation: 365 nm λemission:435 nm | Hichrom ODS 250 mm × 4.6 mm 5 mm | - | 0.05–0.42 | 0.19–1.4 | ||||
| Dried fruits (peanuts, almonds, walnuts, pistachios, hazelnuts) and seeds (sunflower, pumpkin, pine nuts) | AFBI, AFB2 AFG1, AFG2, OTA, FB1, FB2, T-2, HT-2, STE, CIT, DON, ZEN | QuEChERS | Sample quantity: 2 g Sample extration: 8 mL H2O + 10 mL ACN: 5% FA; 4 g MgSO4 + 1 g NaCl + 1 g sodium citrate + 0.5 g disodium hydrogen citrate sesquihydrate, centrifugation; DLLME for AFs: 2 mL supernatant: evaporation and 1 mL MeOH/H2O (50:50), 4 mL H2O, 0.21 g NaCl; injection 950 μL ACN + 620 μL chloroform | UHPLC—MS/MS | Mobile phase:(A) H2O with 0.3% FA and 5 mM ammonium formate, (B) MeOH with 0.3% FA and 5 mM ammonium formate Gradient program: 0 min:5% B; 1 min:50% B; 2 min:72% B; 4 min:80% B; and 6 min:90% B, finally back to 5 B in 0.2 min and maintained for 1.8 min for column equilibration Flow-rate: 0.4 mL/min Temperature column: 35 °C Injection volume: 5 μL Ionization: ESI source in the positive mode Capillary voltage: 5 kV Collision gas pressure: 30 psi Vaporizer temperature: 500 °C Sheath gas pressure: 50 psi Auxiliary gas flow: | Zorbax Eclipse Plus RRHD 50 mm × 2.1 mm 1.8 um | - | 0.17–9.68 | 0.57–32.6 | [71] |
| Walnuts, pistachios, hazelnuts, cashews, almonds | AFB1, AFB2, AFG1, AFG2 | IAC | Sample quantity: 25 g Sample extraction: 5 g NaCl + 125 mL MeOH/H2O (60:40 v/v), blended for 1min, filtration; 20 mL filtrate + 20 mL H2O; IAC: 10 mL filtrate diluted, wash 10 mL H2O, elution 1 mL MeOH | HPLC—FLD with PCD | Mobile phase: H2O/ACN/MeOH (6:3:1, v/v/v) Flow-rate: 1 mL/min Temperature column: Injection volume: 20 μL λexcitation: 360 nm λemission:440 mm | Spherisorb ODS C18 150 mm × 4.5 mm 5 μm | - | 0.273–0.536 | 0.9–1.8 | [72] |
| Cereals and nuts (almond, peanut, pistachio) | AFBI, AFB2 AFG1, AFG2, OTA, OTB, T-2, HT-2, STE, CIT, DON, ZEN, etc. | - | Sample quantity: 1 g Sample extraction: 5 mL ACN/H2O (85:15 v/v), shaking for 30 min in higher speed with pulsation, centrifugation, 500 μL extract + 20 μL ISs + 480 μL 20 mM ammonium formate, vortex and filtration. | UHPLC—MS | Mobile phase:(A) H2O with 0.3% FAand 5 mM ammonium formate, (B) MeOH with 0.3% FA and 5mM ammonium formate Gradient program: 100% A at 0 min, increase to 100% B at 8 min, until 12 min, then, return to 100% A in 8.5 min, equilibration for 5.5 min (t = 18 min) Flow-rate: 0.3 mL/min Temperature column: 35 °C Injection volume: 5 μL Ionization: ESI source in the positive mode Capillary voltage: 4 kV Collision gas pressure: Vaporizer temperature: 350 °C Sheath gas pressure: Auxiliary gas flow: 15 L/min | Hypersil GOLD aQ 100 × 2,1 mm - | Isotope labeled 13C | - | - | [60] |
| Pistachios, peanuts and walnuts (raw and roasted with salt) | AFB1, AFB2, AFG1, AFG2 | - | Sample quantity: 10 g Sample extraction: 50 mL 33% MeOH, filtration, dilution 1:2 with 33% MeOH | ELISA | Clone total AF ELISA test kit, Absorbance at 450 nm | - | - | - | - | [73] |
| Almond, hazelnuts, peanuts, pistachio, walnuts, brazil nuts, chestnuts and apricot | AFB1, AFB2, AFG1, AFG2 | IAC | Sample quantity: 25 g Sample extraction: 5 g NaCl + 125 mL MeOH/H2O (60:40 v/v), blended with hight speed 1 min, sediment, filtration of supernatant; 20 mL filtrate + 20 mL PBS; IAC:20 mL diluted filtrate, wash MeOH/H2O (25:75 v/v), elution 2 mL MeOH + 3 mL H2O | HPLC—FLD with PCD | Mobile phase:H2O/MeOH/ACN (64:23:13, v/v/v) Isocratic program Flow-rate: 1 mL/min Injection volume: 100 μL λexcitation: 364 nm λemission: 440 mm | C18 150 mm × 4.6 mm 5 μm | - | - | 0.4–1.3 | [74] |
| Peanuts, almonds and pistachios | AFBI, AFB2, AFG1, AFG2, OTA, FB1, FB2, T-2, HT-2, STE, CIT, DON, ZEN, etc. | QuEChERS | Sample quantity: 5 g Sample extraction:10 mL H2O, 10 mL ACN:FA 0.1%; 4 g MgSO4 + 1 g NaCl + 1 g sodium citrate + 0.5 g disodium hydrogen citrate sesquihydrate, centrifugation; d-SPE with EMR-lipid: activation with 5ml H2O + 5 mL extrat, centrifugation, 5 mL supernatant + 0.4 g NaCl + 1.6 g MgSO4, centrifugation | HPLC-MS | Mobile phase: (A) H2O with 0.1% FA, (B) ACN with 0.1% AF Gradient program: 0–5 min 4% B, 5–20 min 100% B, 20–24 min 100% B, 24–28 min 2% B and this latest rate was maintained for 10 min (t = 38 min) Flow-rate: 200 nL/min Ionization: ESI source in the positive mode Temperature column: 25 °C Injection volume: 100 nL Ionization: ESI source in the positive mode Capillary voltage: 2.2 kV Collision gas pressure: Vaporizer temperature: 250 °C Sheath gas pressure: Auxiliary gas flow: | Easy-Spray PepMap C18 nano 150 mm × 75 μm 3 μm | - | - | 0.05–5 | [75] |
| Raw peanuts and roasted pistachios | AFB1; AFB2; AFG1; AFG2; OTA | QuEChERS | Sample quantity: 2.5 g Sample extration: 10 mL ACN + 10 mL H2O with 0.2% FA, rotation for 30 min; 4 g MgSO4 + 1 g NaCl + 1 g sodium citrate + 0.5 g disodium hydrogen citrate sesquihydrate, centrifugation, follow by 2 extraction with 20 mL hexane; d-SPE: supernatant + 150 mg C18 + 900 mg MgSO4, centrifugation, wash 2 × with 5 mL ACN | LC—MS/MS | Mobile phase:(A) H2O Gradient program: Flow-rate: 0.2 mL/min Temperature column: 30 °C Injection volume: 4 μL Ionization: ESI source in the positive mode Capillary voltage: Collision gas pressure: 25 psi Vaporizer temperature: 250 °C Sheath gas pressure: Auxiliary gas flow: 14 L/min | ODS C18 150 mm × 2.1 mm 5 μm | - | 0.05–0.10 | 0.08–0.30 | [76] |
| Almonds, hazelnuts, peanuts, pistachios, walnuts | AFB1, AFB2, AFG1, AFG2; ZEA | QuEChERS | Sample quantity: 2 g Sample extraction: 10 mL ACN/H2O (80:20 v/v), rotation for 20 min, 4 g Na2SO4 + 1 g NaCl, centrifugation; d-SPE: 3 mL supernant + 100 mg C18, centrifugation | UHPLC—MS/MS | Mobile phase: (A) H2O with 0.1% FA, (B) ACN with 0.1% FA Gradient program: 25% A increased to 100% in 3.75 min, reduction to 25% A in 6 min (t = 7.5 min) Flow-rate: 0.2 mL/min Ionization: ESI source in the negative mode Temperature column: 25 °C Injection volume: 5 μL Ionization: ESI source in the positive mode for AFs Capillary voltage: 3.5 kV Collision gas pressure: 45 psi Vaporizer temperature: 400 °C Sheath gas pressure: Auxiliary gas flow: 11 L/min | C18, 100 mm × 2.1 mm, 1.8 μm | - | - | 0.5–1.0 | [77] |
| Almonds, hazelnuts and pistachios | AFBI, AFB2 AFG1, AFG2, OTA, OTB, T-2, HT-2, STE, CIT, DON, ZEN, etc. | QuEChERS | Sample quantity: 1 g Sample extraction: 5 mL H2O, 5 mL ACN with 0,1% FA; 0.5 g NaCl + 2 g MgSO4, centrifugation; d-SPE: 1.5 mL supernatant + 50 mg C18, centrifugation | UHPLC—MS | Mobile phase: (A) H2O with 0.1% FA, (B)MeOH with 0.1% FA Gradient program: 0% B for 1 min, 95% B for 1.5 min, 75% B for 2.5 min, decrease to 60% in 1 min, back to 0% B in 0.5 min and held for 1.5 min (t = 8 min) Flow-rate: 0,4 mL/min Ionization: ESI source in the posiive and negative mode Temperature column: 30 °C Injection volume: 5 μL Ionization: ESI source in the positive and negative mode Capillary voltage: ± 4 kV Collision gas pressure: Vaporizer temperature: 290 °C Sheath gas pressure: 35 psi Auxiliary gas flow: | Luna Omega Polar C18, 50 mm × 2.1 mm, 1.6 μm | - | - | 0.2–0.78 | [78] |
| Reference | Country | Number Samples | Mycotoxin | Nº Positive Samples | % Positive Samples | Average Concentration (μg/kg) | Min-Max (μg/kg) |
|---|---|---|---|---|---|---|---|
| [24] | Iran | 10,068 | AFB1 | 3699 | 37 | 5.9 | - |
| AFs | 2852 | 28 | 7.3 | - | |||
| [68] | Algeria | 31 | AFs | 2 | 6 | - | 0.4–0.7 |
| OTA | 1 | 3 | 170 | - | |||
| [121] | Spain | 70 | OTA | 2 | 3 | 0.228 | 0.134–0.321 |
| [70] | Iran | 32 | AFB1 | 17 | 53 | - | 9.5–43.8 |
| AFB2 | - | 0.9–9.4 | |||||
| AFG1 | - | n.d.–19.7 | |||||
| AFG2 | - | n.d.–7.1 | |||||
| [122] | Spain | 70 | AFs | 14 | 20 | 8.9 | n.d.–108 |
| [72] | Saudi Arabia | 53 | AFS | 18 | 34 | 16.6 | - |
| 9 | AFB1 | 9 | - | 1.9–411 | |||
| AFB2 | - | n.d.–10.7 | |||||
| AFG1 | - | n.d.–4.6 | |||||
| AFG2 | - | n.d.–0.8 | |||||
| [25] | Austria | 8 | AFs | 0 | 0 | - | - |
| H-T2 | 0 | - | - | - | |||
| OTA | 1 | 13 | <LOQ | - | |||
| T2 | 0 | - | - | - | |||
| ZEA | 0 | - | - | - | |||
| [60] | USA | 10 | AFB1 | 2 | 20 | - | 0.5–1.2 |
| AFB2 | 1 | 10 | 0.9 | - | |||
| AFG1 | 1 | 10 | 0.5 | - | |||
| AFG2 | 0 | - | 0.0 | - | |||
| DON | 0 | - | - | - | |||
| FB1 | 0 | - | - | - | |||
| FB2 | 0 | - | - | - | |||
| OTA | 3 | 30 | 1.4 | 1.0–6.6 | |||
| T2 | 0 | - | - | - | |||
| ZEA | 0 | - | - | - | |||
| [74] | Italy | 8 | AFB1 | 4 | 50 | 31.9 (median) | 8.2–354.5 |
| AFs | 50 | 33.9 (median) | 8.8–387.3 | ||||
| [76] | Malaysia | 10 | AFB1 | 4 | 40 | 7.10 | 5.30–10.15 |
| AFB2 | 3 | 30 | 2.18 | 1.46–3.47 | |||
| AFG1 | 4 | 40 | 2.45 | 1.90–3.31 | |||
| AFG2 | 2 | 20 | 0.86 | 0.81–0.90 | |||
| OTA | 0 | - | - | - | |||
| [123] | Turkey | 50 | OTA | 2 | 4 | 0.527 | 0.198–0.850 |
| Method | Treatment | Assay Conditions | Reduction AFB1 | Reference |
|---|---|---|---|---|
| Physical | Heat/Roasting | 150 °C for 30 min | 63% | [143] |
| Gamma radiation | 10 kGy | 68% | [144] | |
| Chemical | Ozonation | 0.9 mg/L for 420 min | 23% | [145] |
| Seed extract Trachyspermum ammi | 37 °C for 24 h | 91% | [146] | |
| Leaf extract Adhatoda vasica | 37 °C for 24 h | 96% | [147] | |
| Leaf extract Corymbia citriodora | 30 °C for 72 h | 95% | [148] | |
| Leaf extract Ocimum basilicum | 30 °C for 72 h | 90% | [149] | |
| Biological | Kefir-grains | 30 °C for 6 h | 97% | [150] |
| Bacillus subtilis UTBSP1 | 35 °C for 5 days | 95% | [151] | |
| Saccharomyces cerevisiae | - | 40–70% | [152] | |
| Others | Heat + Acidification | 15 mL lemon juice 6 g citric acid 120 °C for 1 h | 49% | [153] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares Mateus, A.R.; Barros, S.; Pena, A.; Sanches Silva, A. Mycotoxins in Pistachios (Pistacia vera L.): Methods for Determination, Occurrence, Decontamination. Toxins 2021, 13, 682. https://doi.org/10.3390/toxins13100682
Soares Mateus AR, Barros S, Pena A, Sanches Silva A. Mycotoxins in Pistachios (Pistacia vera L.): Methods for Determination, Occurrence, Decontamination. Toxins. 2021; 13(10):682. https://doi.org/10.3390/toxins13100682
Chicago/Turabian StyleSoares Mateus, Ana Rita, Sílvia Barros, Angelina Pena, and Ana Sanches Silva. 2021. "Mycotoxins in Pistachios (Pistacia vera L.): Methods for Determination, Occurrence, Decontamination" Toxins 13, no. 10: 682. https://doi.org/10.3390/toxins13100682
APA StyleSoares Mateus, A. R., Barros, S., Pena, A., & Sanches Silva, A. (2021). Mycotoxins in Pistachios (Pistacia vera L.): Methods for Determination, Occurrence, Decontamination. Toxins, 13(10), 682. https://doi.org/10.3390/toxins13100682








