Spread of Jacobaea vulgaris and Occurrence of Pyrrolizidine Alkaloids in Regionally Produced Honeys from Northern Germany: Inter- and Intra-Site Variations and Risk Assessment for Special Consumer Groups
Abstract
1. Introduction
2. Results
2.1. Performance of the Method
2.2. Occurrence of PA/PANO
2.3. PA/PANO-Pattern and Botanical Origin
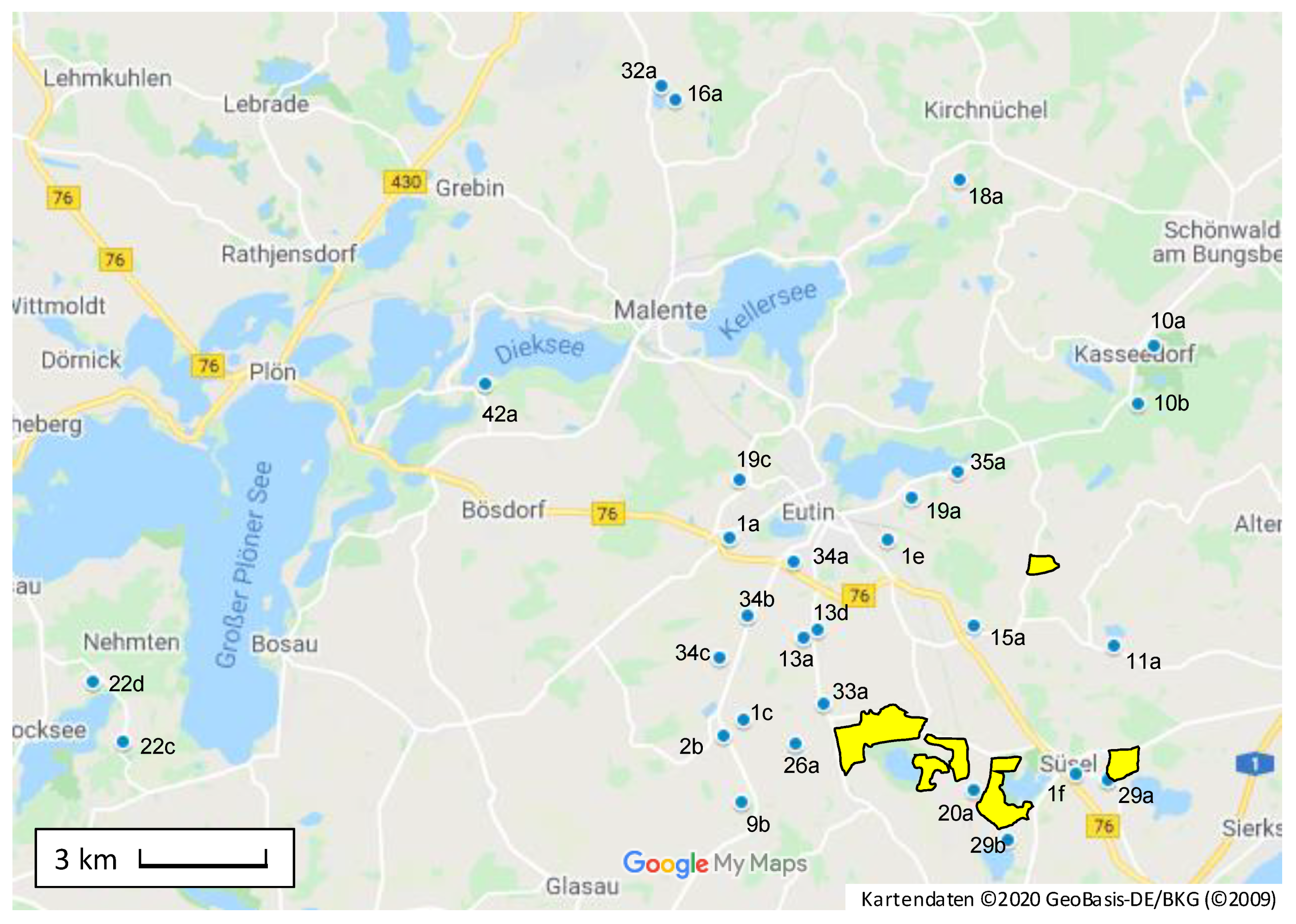
2.4. Inter- and Intra Site Variations
2.5. Analysis of Pollen of Selected Samples
2.6. Risk Assessment
3. Discussion
4. Materials and Methods
4.1. Samples
4.2. Chemicals
4.3. Sample Pretreatment
4.4. LC–MS/MS Measurement and Quantification
4.5. Method Validation
4.6. Pollen Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stegelmaier, B. Pyrrolizidine Alkaloids. In Clinical Veterinary Toxicology; Mosby Inc.: St. Louis, MO, USA, 2004. [Google Scholar]
- Hartmann, T.; Witte, L. Chemistry, biology and chemoecology of the pyrrolizidine alkaloids. In Alkaloids: Chemical and Biological Perspectives; Pelletier, S.W., Ed.; Elsevier: Amsterdam, The Netherlands, 1995; Volume 9, pp. 155–233. [Google Scholar]
- Smith, L.W.; Culvenor, C.C.J. Plant sources of hepatotoxic pyrrolizidine alkaloids. J. Nat. Prod. 1981, 44, 129–152. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements. EFSA J. 2017, 15. [Google Scholar] [CrossRef]
- EFSA. Opinion of the Panel on contaminants in the food chain related to pyrrolizidine alkaloids as undesirable substances in animal feed. EFSA J. 2007, 5, 447. [Google Scholar] [CrossRef]
- Mattocks, A.R. Chemistry and Toxicology of Pyrrolizidine Alkaloids; Academic Press: Cambridge, MA, USA, 1986; p. 393. [Google Scholar]
- Fu, P.P.; Xia, Q.; Lin, G.; Chou, M.W. Pyrrolizidine Alkaloids—Genotoxicity, Metabolism Enzymes, Metabolic Activation, and Mechanisms. Drug Metab. Rev. 2004, 36, 1–55. [Google Scholar] [CrossRef]
- Stegelmeier, B.; Colegate, S.; Brown, A. Dehydropyrrolizidine Alkaloid Toxicity, Cytotoxicity, and Carcinogenicity. Toxins 2016, 8, 356. [Google Scholar] [CrossRef]
- Edgar, J.A.; Molyneux, R.J.; Colegate, S.M. Pyrrolizidine alkaloids: Potential role in the etiology of cancers, pulmonary hypertension, congenital anomalies, and liver disease. Chem. Res. Toxicol. 2015, 28, 4–20. [Google Scholar] [CrossRef]
- Council of the European Union. Council Regulation (EEC) No. 315/93 laying down Community procedures for contaminants in food. Off. J. Eur. Communities 1993, L37, 1. [Google Scholar]
- EFSA. Scientific opinion on pyrrolizidine alkaloids in food and feed. EFSA J. 2011, 9, 2406. [Google Scholar] [CrossRef]
- Selmar, D.; Wittke, C.; Beck-von Wolffersdorff, I.; Klier, B.; Lewerenz, L.; Kleinwächter, M.; Nowak, M. Transfer of pyrrolizidine alkaloids between living plants: A disregarded source of contaminations. Environ. Pollut. 2019, 248, 456–461. [Google Scholar] [CrossRef]
- BfR. Opinion No. 017/2019: Pyrrolizidine Alkaloid Levels in Dried and Deep-Frozen Spices and Herbs Too High; BfR (Federal Institute for Risk Assessment): Berlin, Germany, 2019.
- Kaltner, F.; Rychlik, M.; Gareis, M.; Gottschalk, C. Occurrence and Risk Assessment of Pyrrolizidine Alkaloids in Spices and Culinary Herbs from Various Geographical Origins. Toxins 2020, 12, 155. [Google Scholar] [CrossRef]
- EFSA. Occurrence of Pyrrolizidine Alkaloids in food. EFSA J. 2015, 35, 118–133. [Google Scholar] [CrossRef]
- Deinzer, M.; Thomson, P.; Burgett, D.; Isaacson, D. Pyrrolizidine alkaloids: Their occurrence in honey from tansy ragwort (Senecio jacobaea L.). Science 1977, 195, 497–499. [Google Scholar] [CrossRef] [PubMed]
- Culvenor, C.C.; Edgar, J.A.; Smith, L.W. Pyrrolizidine alkaloids in honey from Echium plantagineum L. J. Agric. Food Chem. 1981, 29, 958–960. [Google Scholar] [CrossRef] [PubMed]
- Crews, C.; Startin, J.R.; Clarke, P.A. Determination of pyrrolizidine alkaloids in honey from selected sites by solid phase extraction and HPLC-MS. Food Addit. Contam. 1997, 14, 419–428. [Google Scholar] [CrossRef]
- Beales, K.A.; Betteridge, K.; Colegate, S.M.; Edgar, J.A. Solid-phase extraction and LC-MS analysis of pyrrolizidine alkaloids in honeys. J. Agric. Food Chem. 2004, 52, 6664–6672. [Google Scholar] [CrossRef]
- Dübecke, A.; Beckh, G.; Lüllmann, C. Pyrrolizidine alkaloids in honey and bee pollen. Food Addit. Contam. Part A 2011, 28, 348–358. [Google Scholar] [CrossRef]
- Kempf, M.; Wittig, M.; Reinhard, A.; von der Ohe, K.; Blacquière, T.; Raezke, K.P.; Michel, R.; Schreier, P.; Beuerle, T. Pyrrolizidine alkaloids in honey: Comparison of analytical methods. Food Addit. Contam. Part A 2011, 28, 332–347. [Google Scholar] [CrossRef]
- Orantes-Bermejo, F.J.; Bonvehí, J.S.; Gómez-Pajuelo, A.; Megías, M.; Torres, C. Pyrrolizidine alkaloids: Their occurrence in Spanish honey collected from purple viper’s bugloss (Echium spp.). Food Addit. Contam. Part A 2013, 30, 1799–1806. [Google Scholar] [CrossRef]
- Kast, C.; Dübecke, A.; Kilchenmann, V.; Bieri, K.; Böhlen, M.; Zoller, O.; Beckh, G.; Lüllmann, C. Analysis of Swiss honeys for pyrrolizidine alkaloids. J. Apic. Res. 2014, 53, 75–83. [Google Scholar] [CrossRef]
- Bodi, D.; Ronczka, S.; Gottschalk, C.; Behr, N.; Skibba, A.; Wagner, M.; Lahrssen-Wiederholt, M.; Preiss-Weigert, A.; These, A. Determination of pyrrolizidine alkaloids in tea, herbal drugs and honey. Food Addit. Contam. Part A 2014, 31, 1886–1895. [Google Scholar] [CrossRef]
- Lorena, L.; Roberta, M.; Alessandra, R.; Clara, M.; Francesca, C. Evaluation of Some Pyrrolizidine Alkaloids in Honey Samples from the Veneto Region (Italy) by LC-MS/MS. Food Anal. Methods 2016, 9, 1825–1836. [Google Scholar] [CrossRef]
- Neumann, H.; Huckauf, A. Tansy ragwort (Senecio jacobaea): A source of pyrrolizidine alkaloids in summer honey? J. Verbrauch. Lebensm. 2016, 11, 105–115. [Google Scholar] [CrossRef]
- Letsyo, E.; Jerz, G.; Winterhalter, P.; Dübecke, A.; von der Ohe, W.; von der Ohe, K.; Beuerle, T. Pyrrolizidine alkaloids in floral honeys of tropical Ghana: A health risk assessment. Food Addit. Contam. Part B Surveill. 2017, 10, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, E.; Sieradzki, Z.; Kwiatek, K. Determination of Pyrrolizidine Alkaloids in Honey with Sensitive Gas Chromatography-Mass Spectrometry Method. Food Anal. Methods 2018, 11, 1345–1355. [Google Scholar] [CrossRef]
- Carpinelli de Jesus, M.; Hungerford, N.L.; Carter, S.J.; Anuj, S.R.; Blanchfield, J.T.; De Voss, J.J.; Fletcher, M.T. Pyrrolizidine alkaloids of Blue Heliotrope (Heliotropium amplexicaule) and their presence in Australian honey. J. Agric. Food Chem. 2019, 67, 7995–8006. [Google Scholar] [CrossRef]
- Betteridge, K.; Cao, Y.; Colegate, S.M. Improved method for extraction and LC-MS analysis of pyrrolizidine alkaloids and their N-oxides in honey: Application to Echium vulgare honeys. J. Agric. Food Chem. 2005, 53, 1894–1902. [Google Scholar] [CrossRef]
- Kempf, M.; Beuerle, T.; Bühringer, M.; Denner, M.; Trost, D.; Von Der Ohe, K.; Bhavanam, V.B.R.; Schreier, P. Pyrrolizidine alkaloids in honey: Risk analysis by gas chromatography-mass spectrometry. Mol. Nutr. Food Res. 2008, 52, 1193–1200. [Google Scholar] [CrossRef]
- Griffin, C.T.; Danaher, M.; Elliott, C.T.; Kennedy, D.G.; Furey, A. Detection of pyrrolizidine alkaloids in commercial honey using liquid chromatography–ion trap mass spectrometry. Food Chem. 2013, 136, 1577–1583. [Google Scholar] [CrossRef]
- Martinello, M.; Cristofoli, C.; Gallina, A.; Mutinelli, F. Easy and rapid method for the quantitative determination of pyrrolizidine alkaloids in honey by ultra performance liquid chromatography-mass spectrometry: An evaluation in commercial honey. Food Control 2014, 37, 146–152. [Google Scholar] [CrossRef]
- Griffin, C.T.; Mitrovic, S.M.; Danaher, M.; Furey, A. Development of a fast isocratic LC-MS/MS method for the high-throughput analysis of pyrrolizidine alkaloids in Australian honey. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 214–228. [Google Scholar] [CrossRef]
- Griffin, C.T.; O’Mahony, J.; Danaher, M.; Furey, A. Liquid Chromatography Tandem Mass Spectrometry Detection of Targeted Pyrrolizidine Alkaloids in Honeys Purchased within Ireland. Food Anal. Methods 2015, 8, 18–31. [Google Scholar] [CrossRef]
- Valese, A.C.; Molognoni, L.; de Sá Ploêncio, L.A.; de Lima, F.G.; Gonzaga, L.V.; Górniak, S.L.; Daguer, H.; Barreto, F.; Costa, A.C.O. A fast and simple LC-ESI-MS/MS method for detecting pyrrolizidine alkaloids in honey with full validation and measurement uncertainty. Food Control 2016, 67, 183–191. [Google Scholar] [CrossRef]
- Hungerford, N.L.; Carter, S.J.; Anuj, S.R.; Tan, B.L.; Hnatko, D.; Martin, C.L.; Sharma, E.; Yin, M.; Nguyen, T.T.; Melksham, K.J. Analysis of Pyrrolizidine Alkaloids in Queensland Honey: Using Low Temperature Chromatography to Resolve Stereoisomers and Identify Botanical Sources by UHPLC-MS/MS. Toxins 2019, 11, 726. [Google Scholar] [CrossRef] [PubMed]
- Celano, R.; Piccinelli, A.L.; Campone, L.; Russo, M.; Rastrelli, L. Determination of Selected Pyrrolizidine Alkaloids in Honey by Dispersive Liquid–Liquid Microextraction and Ultrahigh-Performance Liquid Chromatography–Tandem Mass Spectrometry. J. Agric. Food Chem. 2019, 67, 8689–8699. [Google Scholar] [CrossRef] [PubMed]
- BfR. Opinion No. 038/2011: Chemical Analysis and Toxicity of Pyrrolizidine Alkaloids and Assessment of the Health Risks Posed by Their Occurrence in Honey; BfR (Federal Institute for Risk Assessment): Berlin, Germany, 2011.
- EFSA. Opinion of the Scientific Committee on a request from EFSA related to A Harmonised Approach for Risk Assessment of Substances Which are both Genotoxic and Carcinogenic. EFSA J. 2005, 3, 282. [Google Scholar] [CrossRef]
- BfR. Opinion No. 020/2018: Actual Risk Assessment Concerning Contents of 1,2-Unsaturated Pyrrolizidine Alkaloids in Food; BfR (Federal Institute for Risk Assessment): Berlin, Germany, 2018.
- NCI. Bioassay of Lasiocarpine for Possible Carcinogenicity; National Cancer Insitute: Bethesda, MD, USA, 1978; pp. 1–66.
- NTP. Toxicology and Carcinogenesis Studies of Riddelliine (CAS No. 23246-96-0) in F344/N rats and B6C3F1 mice (gavage studies). Natl. Toxicol. Program Tech. Rep. Ser. 2003, 508, 1. [Google Scholar]
- Merz, K.H.; Schrenk, D. Interim Relative Potency Factors for the Toxicological Risk Assessment of Pyrrolizidine Alkaloids in Food and Herbal Medicines. Toxicol. Lett. 2016, 263, 44–57. [Google Scholar] [CrossRef]
- COT. Statement of the Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. Available online: https://cot.food.gov.uk/sites/default/files/cot/cotstatementpa200806.pdf (accessed on 4 July 2020).
- German Federal Agency for Nature Conservation. Databases and Data Collections. FloraWeb. Available online: http://www.floraweb.de/pflanzenarten/pflanzenarten.html (accessed on 4 July 2020).
- Mulder, P.P.J.; Beumer, B.; Oosterink, E.; de Jong, J. Dutch Survey Pyrrolizidine Alkaloids in Animal Forage; Institute of Food Safety: Wageningen, The Netherlands, 2009; p. 45. [Google Scholar]
- Steffens, S.; Gareis, M.; Gottschalk, C. Vorkommen von Pyrrolizidinalkaloiden in regional erzeugtem Honig. In Proceedings of the 56. Arbeitstagung des Arbeitsgebietes Lebensmittelhygiene der Deutschen Veterinärmedizinischen Gesellschaft, Garmisch-Partenkirchen, Germany, 29 September–2 October 2015. [Google Scholar]
- MRI. German National Nutrition Survey II: Food Consumption and Nutrient Intake on the Basis of 24 h Recalls; Max Rubner-Institut: Karlsruhe, Germany, 2008; p. 308. [Google Scholar]
- Banasiak, U.; Heseker, H.; Sieke, C.; Sommerfeld, C.; Vohmann, C. Abschätzung der Aufnahme von Pflanzenschutzmittel-Rückständen in der Nahrung mit neuenVerzehrsmengen für Kinder. Bundesgesundheitsblatt Gesundh. Gesundh. 2005, 48, 84–98. [Google Scholar] [CrossRef]
- Beekman, M.; Sumpter, D.; Seraphides, N.; Ratnieks, F. Comparing foraging behaviour of small and large honey-bee colonies by decoding waggle dances made by foragers. Funct. Ecol. 2004, 18, 829–835. [Google Scholar] [CrossRef]
- Steffan-Dewenter, I.; Kuhn, A. Honeybee foraging in differentially structured landscapes. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2003, 270, 569–575. [Google Scholar] [CrossRef]
- Hagler, J.R.; Mueller, S.; Teuber, L.R.; Machtley, S.A.; Van Deynze, A. Foraging range of honey bees, Apis mellifera, in alfalfa seed production fields. J. Insect Sci. 2011, 11, 144. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, C.; Huckauf, A.; Dübecke, A.; Kaltner, F.; Zimmermann, M.; Rahaus, I.; Beuerle, T. Uncertainties in the determination of pyrrolizidine alkaloid levels in naturally contaminated honeys and comparison of results obtained by different analytical approaches. Food Addit. Contam. Part A 2018, 35, 1366–1383. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Lauter, J.; Hartung, N.M.; These, A.; Hamscher, G.; Wissemann, V. Genetic and chemical diversity of the toxic herb Jacobaea vulgaris Gaertn. (syn. Senecio jacobaea L.) in Northern Germany. Phytochemistry 2020, 172, 112235. [Google Scholar] [CrossRef] [PubMed]
- Mädge, I.; Gehling, M.; Schöne, C.; Winterhalter, P.; These, A. Pyrrolizidine alkaloid profiling of four Boraginaceae species from Northern Germany and implications for the analytical scope proposed for monitoring of maximum levels. Food Addit. Contam. Part A 2020, 1–20. [Google Scholar] [CrossRef]
- Chen, L.; Mulder, P.P.J.; Louisse, J.; Peijnenburg, A.; Wesseling, S.; Rietjens, I.M.C.M. Risk assessment for pyrrolizidine alkaloids detected in (herbal) teas and plant food supplements. Regul. Toxicol. Pharmacol. 2017, 86, 292–302. [Google Scholar] [CrossRef]
- Flade, J.; Beschow, H.; Wensch-Dorendorf, M.; Plescher, A.; Wätjen, W. Occurrence of nine pyrrolizidine alkaloids in Senecio vulgaris L. Depending on developmental stage and season. Plants 2019, 8, 54. [Google Scholar] [CrossRef]
- Chizzola, R.; Bassler, G.; Kriechbaum, M.; Karrer, G. Pyrrolizidine alkaloid production of Jacobaea aquatica under different cutting regimes. J. Agric. Food Chem. 2015, 63, 1293–1299. [Google Scholar] [CrossRef]
- Eller, A.; Chizzola, R. Seasonal variability in pyrrolizidine alkaloids in Senecio inaequidens from the Val Venosta (Northern Italy). Plant Biosyst. 2016, 150, 1306–1312. [Google Scholar] [CrossRef]
- Kaltner, F.; Rychlik, M.; Gareis, M.; Gottschalk, C. Influence of Storage on the Stability of Toxic Pyrrolizidine Alkaloids and Their N-Oxides in Peppermint Tea, Hay, and Honey. J. Agric. Food Chem. 2018, 66, 5221–5228. [Google Scholar] [CrossRef]
- Kast, C.; Kilchenmann, V.; Reinhard, H.; Bieri, K.; Zoller, O. Pyrrolizidine alkaloids: The Botanical Origin of Pollen Collected during the Flowering Period of Echium vulgare and the Stability of Pyrrolizidine Alkaloids in Bee Bread. Molecules 2019, 24, 2214. [Google Scholar] [CrossRef]
- Teuscher, E.; Lindequist, U. Biogene Gifte-Biologie-Chemie-Pharmakologie-Toxikologie, 3rd ed.; Wissenschaftliche Verlagsgesellschaft: Stuttgart, Germany, 2010. [Google Scholar]
- Rosenkranz, P. Eintönige Agrarlandschaften schaden der Honigbiene. Ökologie Landbau 2012, 161, 54–56. [Google Scholar]
- Oplatowska, M.; Elliott, C.T.; Huet, A.C.; McCarthy, M.; Mulder, P.P.J.; Von Holst, C.; Delahaut, P.; Van Egmond, H.P.; Campbell, K. Development and validation of a rapid multiplex ELISA for pyrrolizidine alkaloids and their N-oxides in honey and feed Rapid Detection in Food and Feed. Anal. Bioanal. Chem. 2014, 406, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Justice, European Affairs and Consumer Protection, Schleswig-Holstein, Germany. Lebensmittelrechtlicher Umgang mit Honigen mit Gehalten von über 474 μg/kg PA, Decree of 20.04.2018.
- June 2019 Stakeholder Update on Rapidly Developing Policy on Food Contaminants. Available online: https://www.food.gov.uk/news-alerts/consultations/june-2019-stakeholder-update-on-rapidly-developing-policy-on-food-contaminants (accessed on 4 July 2020).
- DIB. Deutscher Imkerbund (German beekeper association). Available online: https://deutscherimkerbund.de/161-Imkerei_in_Deutschland_Zahlen_Daten_Fakten (accessed on 4 July 2020).
- DIN. Deutsches Institut für Normung (German Institute for Standardization). DIN 32645:2008-11 Chemical Analysis Decision Limit, Detection Limit and Determination Limit Under Repeatability Conditions-Terms, Method, Evaluation; Beuth Verlag GmbH: Berlin, Germany, 2008. [Google Scholar]

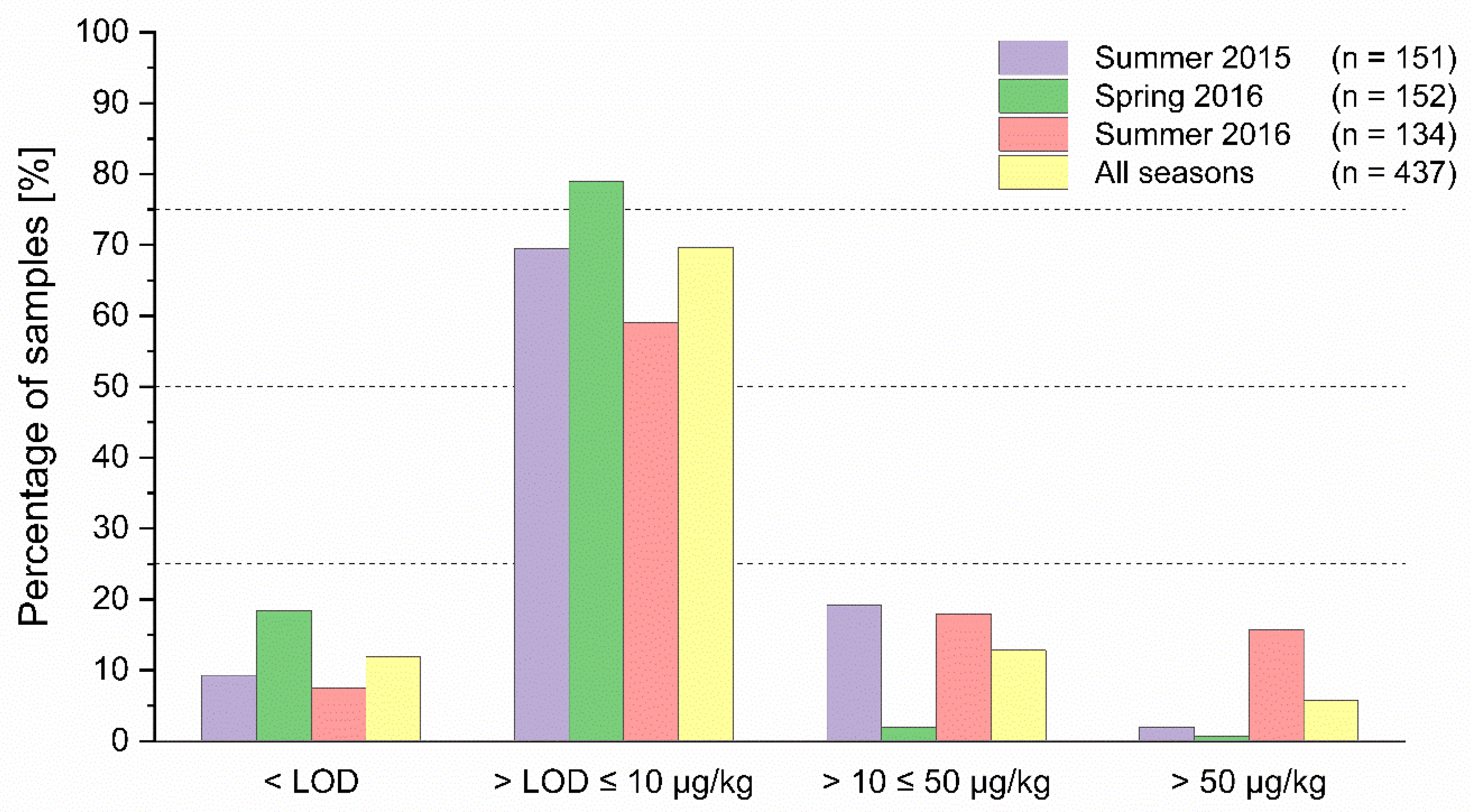
| Season | N | Incidence (%) | PA/PANO Sum Content (µg/kg) | |||||
|---|---|---|---|---|---|---|---|---|
| Bee- Keepers | Sites | Samples | Median | Mean | 95th Percentile | Maximum | ||
| Summer 2015 | 30 | 53 | 151 | 91 | 2.3 | 8.4 | 35.4 | 111 |
| Spring 2016 | 35 | 53 | 152 | 82 | 0.2 | 1.5 | 3.6 | 59.4 |
| Summer 2016 | 29 | 47 | 134 | 93 | 3.9 | 72.6 | 181 | 3313 |
| All | 42 | 73 | 437 | 88 | 1.0 | 25.7 | 59.5 | 3313 |
| Season | Senecio-Type | Borago/Echium/Eupatorium-Type | Mixed Origin |
|---|---|---|---|
| Summer 2015 | 32% | 12% | 56% |
| Spring 2016 | 26% | 45% | 29% |
| Summer 2016 | 44% | 2.0% | 54% |
| Season (n Samples) | PA/PANO (Senecio-Type) (µg/kg) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Incidence (%)/Levels | Ec | EcN | Jb | JbN | Rs | RsN | Sc | ScN | Sp | SpN | Sk | |
| Summer 2015 (n = 151) | incidence | 40 | 7.9 | 60 | 15 | 56 | 5.3 | 70 | 21 | 66 | 25 | 7.9 |
| mean | 0.1 | 0.0 | 1.2 | 0.0 | 0.4 | 0.0 | 2.8 | 0.1 | 3.3 | 0.1 | 0.0 | |
| 95th perc. | 0.6 | 0.3 | 5.5 | 0.2 | 1.8 | 0.1 | 15.0 | 0.5 | 15.3 | 0.5 | 0.1 | |
| max. | 1.9 | 0.3 | 16.2 | 0.3 | 4.6 | 0.4 | 40.4 | 2.0 | 40.7 | 2.6 | 0.7 | |
| Spring 2016 (n = 152) | incidence | 3.3 | 0.7 | 13 | 11 | 18 | 3.3 | 23 | 32 | 22 | 29 | 7.9 |
| mean | 0.0 | 0.0 | 0.1 | 0.0 | 0.1 | 0.0 | 0.4 | 0.1 | 0.5 | 0.1 | 0.0 | |
| 95th perc. | 0.0 | 0.0 | 0.3 | 0.1 | 0.2 | 0.0 | 1.1 | 0.3 | 1.1 | 0.4 | 0.0 | |
| max. | 0.7 | 0.3 | 7.2 | 1.4 | 2.7 | 0.7 | 16.5 | 3.8 | 31.0 | 4.9 | 0.2 | |
| Summer 2016 (n = 134) | incidence | 29 | 21 | 64 | 54 | 49 | 40 | 89 | 73 | 84 | 77 | 12 |
| mean | 0.7 | 2.1 | 10.3 | 4.4 | 2.4 | 1.7 | 10.8 | 12.7 | 14.7 | 12.1 | 0.0 | |
| 95th perc. | 5.0 | 7.4 | 22.1 | 10.6 | 4.0 | 4.9 | 33.1 | 36.3 | 38.3 | 40.5 | 0.2 | |
| max. | 16.4 | 111 | 504 | 325 | 136 | 120 | 482 | 711 | 609 | 630 | 0.3 | |
| Site- ID | Season | PA/PANO Sum Content (µg/kg) (Percentage Senecio-Type) | Mean (µg/kg) | RSD (%) | Δ min-max (µg/kg) | Distance J. vulgaris | ||
|---|---|---|---|---|---|---|---|---|
| Sample A | Sample B | Sample C | ||||||
| 1a | spring 2016 | 22.4 (100%) | 0.9 (100%) | 59.4 (100%) | 27.6 | 107 | 58.5 | >3 km |
| 1a | summer 2016 | 18.8 (100%) | 130 (100%) | 151 (99.9%) | 99.8 | 71 | 132 | >3 km |
| 1c | summer 2016 | 9.5 (100%) | 45.9 (100%) | 135 (100%) | 63.4 | 102 | 125 | <3 km |
| 1e | summer 2016 | 38.8 (100%) | 60.4 (100%) | 20.9 (100%) | 40.0 | 49 | 39.5 | <3 km |
| 1f | summer 2016 | 346 (100%) | 26.5 (100%) | 1.3 (15.1%) | 125 | 154 | 345 | <3 km |
| 2b | summer 2015 | 17.4 (96.1%) | 41.9 (98.3%) | 8.6 (93.0%) | 22.7 | 76 | 33.3 | <3 km |
| 9b | summer 2015 | 30.9 (97.5%) | 30.1 (99.3%) | 4.7 (93.5%) | 21.9 | 68 | 26.2 | <3 km |
| 10a | summer 2015 | 3.3 (93.1%) | 37.0 (98.6%) | 33.3 (99.6%) | 24.6 | 75 | 30.0 | >3 km |
| 10b | summer 2015 | 33.8 (99.6%) | 6.4 (100%) | 0.5 (100%) | 13.6 | 131 | 33.3 | >3 km |
| 11a | summer 2016 | 33.0 (90.2%) | 22.1 (94.2%) | 1.3 (100%) | 18.8 | 86 | 31.7 | <3 km |
| 13a | spring 2016 | 7.2 (100%) | 1.0 (97.4%) | 24.8 (100%) | 11.0 | 112 | 23.8 | <3 km |
| 13d | summer 2016 | 1.1 (100%) | 28.4 (100%) | 5.1 (100%) | 11.5 | 128 | 23.3 | <3 km |
| 15a | summer 2016 | 7.7 (72.9%) | 16.2 (96.5%) | 35.2 (95.3%) | 19.7 | 72 | 27.5 | <3 km |
| 16a | summer 2015 | 3.9 (91.8%) | 79.5 (100%) | n.a. | 41.7 | 128 | 75.6 | >3 km |
| 18a | summer 2015 | 7.1 (100%) | 41.8 (97.6%) | 5.3 (100%) | 18.0 | 114 | 36.5 | >3 km |
| 19a | summer 2016 | 25.1 (100%) | 19.9 (100%) | 0.4 (100%) | 15.2 | 86 | 24.7 | <3 km |
| 19c | summer 2016 | 2.5 (100%) | 34.5 (100%) | 6.1 (100%) | 14.4 | 122 | 32.0 | >3 km |
| 20a | summer 2015 | 24.3 (100%) | 27.8 (99.5%) | 21.6 (100%) | 24.6 | 13 | 6.2 | <3 km |
| 20a | summer 2016 | 12.7 (98.2%) | 47.8 (100%) | 331 (100%) | 131 | 134 | 319 | <3 km |
| 22c | summer 2015 | 0.4 (46.1%) | 47.6 (99.7%) | 3.9 (96.4%) | 17.3 | 152 | 47.2 | >3 km |
| 22d | summer 2015 | 111 (98.7%) | 21.2 (95.3%) | 5.5 (97.3%) | 45.7 | 124 | 105 | >3 km |
| 26a | summer 2015 | 22.5 (99.0%) | 4.4 (92.8%) | 5.7 (94.9%) | 10.9 | 92 | 18.1 | <3 km |
| 29a | summer 2015 | 27.2 (99.2%) | 10.4 (100%) | n.a. | 18.8 | 63 | 16.8 | <3 km |
| 29a | summer 2016 | 64.2 (84.0%) | 101 (95.7%) | 314 (95.4%) | 160 | 84 | 249 | <3 km |
| 29b | summer 2015 | 29.5 (100%) | 58.1 (99.0%) | 32.2 (100%) | 39.9 | 40 | 28.6 | <3 km |
| 29b | summer 2016 | 173 (98.9%) | 446 (99.7%) | 197 (99.3%) | 272 | 56 | 273 | <3 km |
| 32a | spring 2016 | 9.0 (100%) | 39.2 (99.2%) | 0.6 (100%) | 16.3 | 125 | 30.2 | >3 km |
| 33a | summer 2015 | 12.0 (94.0%) | 44.9 (99.5%) | 22.0 (100%) | 26.3 | 64 | 32.9 | <3 km |
| 34a | summer 2016 | 48.3 (100%) | 25.7 (100%) | 20.2 (99.6%) | 31.4 | 47 | 28.1 | >3 km |
| 34b | summer 2016 | 61.9 (100%) | 133 (100%) | 136 (99.8%) | 110 | 38 | 74.2 | >3 km |
| 34c | summer 2015 | 6.5 (94.3%) | 2.4 (73.1%) | 29.3 (98.0%) | 12.7 | 114 | 26.9 | >3 km |
| 34c | summer 2016 | 79.5 (100%) | 81.8 (100%) | 59.7 (100%) | 73.7 | 17 | 22.1 | >3 km |
| 35a | summer 2016 | 3313 (99.9%) | 2510 (99.9%) | n.a. | 2912 | 20 | 803 | <3 km |
| 42a | summer 2016 | 53.0 (98.4%) | 21.2 (95.0%) | 31.5 (99.9%) | 35.2 | 46 | 31.8 | >3 km |
| Season | Site- ID | Sample 1 | PA/PANO | Percentage (%) of Pollen 2 | Distance J. vulgaris | |||
|---|---|---|---|---|---|---|---|---|
| Sum Level (µg/kg) | Senecio-Type (%) | Senecio | Eupatorium | Borago | ||||
| Summer 2015 | 16a | B | 79.5 | 100 | 2.0 | 2.0 | n.d. | >3 km |
| 20a | B | 27.8 | 99.5 | n.d. | n.d. | n.d. | <3 km | |
| 22d | A | 111 | 98.7 | <0.1 | n.d. | n.d. | >3 km | |
| 29b | B | 58.1 | 99.0 | 2.0 | <0.1 | n.d. | <3 km | |
| 33a | B | 44.9 | 99.5 | n.d. | n.d. | n.d. | <3 km | |
| Spring 2016 | 1a | C | 59.4 | 100 | <0.1 | <0.1 | n.d. | >3 km |
| 8a | B | 2.4 | 0.0 | <0.1 | n.d. | n.d. | >3 km | |
| 15a | B | 7.5 | 100 | <0.1 | n.d. | n.d. | <3 km | |
| 21a | A | 5.2 | 93.2 | <0.1 | n.d. | n.d. | >3 km | |
| 32a | B | 39.2 | 99.2 | n.d. | n.d. | 3.0 | >3 km | |
| Summer 2016 | 1f | A | 346 | 100 | <0.1 | n.d. | n.d. | <3 km |
| 22a | A | 6.7 | 100 | <0.1 | n.d. | n.d. | >3 km | |
| 34b | C | 136 | 99.8 | 1.0 | 0.5 | n.d. | >3 km | |
| 29a | C | 314 | 95.4 | 2.0 | 1.0 | n.d. | <3 km | |
| 29b | B | 446 | 99.7 | 3.0 | n.d. | n.d. | <3 km | |
| Season | Intake 1 (g honey/kg b.w./day) | Exposure Scenario 2 | PA/PANO Intake (ng/kg b.w./day) | MOE 3 | Percentage of HBGV 4 |
|---|---|---|---|---|---|
| summer 2015 | mean (0.05) | mean case | 0.42 | 564,000 | 0.4% |
| worst case | 1.8 | 134,000 | 1.8% | ||
| high (0.28) | mean case | 2.4 | 101,000 | 2.4% | |
| worst case | 9.9 | 23,900 | 9.9% | ||
| acute (0.88) | mean case | 7.4 | 32,100 | 7.4% | |
| worst case | 31.2 | 7610 | 31.2% | ||
| spring 2016 | mean (0.05) | mean case | 0.08 | 3,160,000 | 0.1% |
| worst case | 0.18 | 1,317,000 | 0.2% | ||
| high (0.28) | mean case | 0.42 | 564,000 | 0.4% | |
| worst case | 1.0 | 235,000 | 1.0% | ||
| acute (0.88) | mean case | 1.3 | 180,000 | 1.3% | |
| worst case | 3.2 | 74,800 | 3.2% | ||
| summer 2016 | mean (0.05) | mean case | 3.6 | 65,300 | 3.6% |
| worst case | 9.1 | 26,100 | 9.1% | ||
| high (0.28) | mean case | 20.3 | 11,700 | 20.3% | |
| worst case | 50.8 | 4670 | 50.8% | ||
| acute (0.88) | mean case | 63.9 | 3710 | 63.9% | |
| worst case | 160 | 1490 | 160% |
| Season | Intake 1 (g honey/kg b.w./day) | Exposure Scenario 2 | PA/PANO Intake (ng/kg b.w./day) | MOE 3 | Percentage of HBGV 4 |
|---|---|---|---|---|---|
| summer 2015 | mean (0.10) | mean case | 0.84 | 282,000 | 0.8% |
| worst case | 3.5 | 66,900 | 3.5% | ||
| high (0.40) | mean case | 3.4 | 70,500 | 3.4% | |
| worst case | 14.2 | 16,700 | 14.2% | ||
| acute (1.36) | mean case | 11.4 | 20,700 | 11.4% | |
| worst case | 48.1 | 4920 | 48.1% | ||
| spring 2016 | mean (0.10) | mean case | 0.15 | 1,580,000 | 0.2% |
| worst case | 0.36 | 658,000 | 0.4% | ||
| high (0.40) | mean case | 0.60 | 395,000 | 0.6% | |
| worst case | 1.4 | 164,000 | 1.4% | ||
| acute (1.36) | mean case | 2.0 | 116,000 | 2.0% | |
| worst case | 4.9 | 48,400 | 4.9% | ||
| summer 2016 | mean (0.10) | mean case | 7.3 | 32,600 | 7.3% |
| worst case | 18.1 | 13,100 | 18.1% | ||
| high (0.40) | mean case | 29.0 | 8160 | 29.0% | |
| worst case | 72.5 | 3270 | 72.5% | ||
| acute (1.36) | mean case | 98.7 | 2400 | 98.7% | |
| worst case | 247 | 960 | 247% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gottschalk, C.; Kaltner, F.; Zimmermann, M.; Korten, R.; Morris, O.; Schwaiger, K.; Gareis, M. Spread of Jacobaea vulgaris and Occurrence of Pyrrolizidine Alkaloids in Regionally Produced Honeys from Northern Germany: Inter- and Intra-Site Variations and Risk Assessment for Special Consumer Groups. Toxins 2020, 12, 441. https://doi.org/10.3390/toxins12070441
Gottschalk C, Kaltner F, Zimmermann M, Korten R, Morris O, Schwaiger K, Gareis M. Spread of Jacobaea vulgaris and Occurrence of Pyrrolizidine Alkaloids in Regionally Produced Honeys from Northern Germany: Inter- and Intra-Site Variations and Risk Assessment for Special Consumer Groups. Toxins. 2020; 12(7):441. https://doi.org/10.3390/toxins12070441
Chicago/Turabian StyleGottschalk, Christoph, Florian Kaltner, Matthias Zimmermann, Rainer Korten, Oliver Morris, Karin Schwaiger, and Manfred Gareis. 2020. "Spread of Jacobaea vulgaris and Occurrence of Pyrrolizidine Alkaloids in Regionally Produced Honeys from Northern Germany: Inter- and Intra-Site Variations and Risk Assessment for Special Consumer Groups" Toxins 12, no. 7: 441. https://doi.org/10.3390/toxins12070441
APA StyleGottschalk, C., Kaltner, F., Zimmermann, M., Korten, R., Morris, O., Schwaiger, K., & Gareis, M. (2020). Spread of Jacobaea vulgaris and Occurrence of Pyrrolizidine Alkaloids in Regionally Produced Honeys from Northern Germany: Inter- and Intra-Site Variations and Risk Assessment for Special Consumer Groups. Toxins, 12(7), 441. https://doi.org/10.3390/toxins12070441





