Toxicokinetics of Hydrolyzed Fumonisin B1 after Single Oral or Intravenous Bolus to Broiler Chickens Fed a Control or a Fumonisins-Contaminated Diet
Abstract
1. Introduction
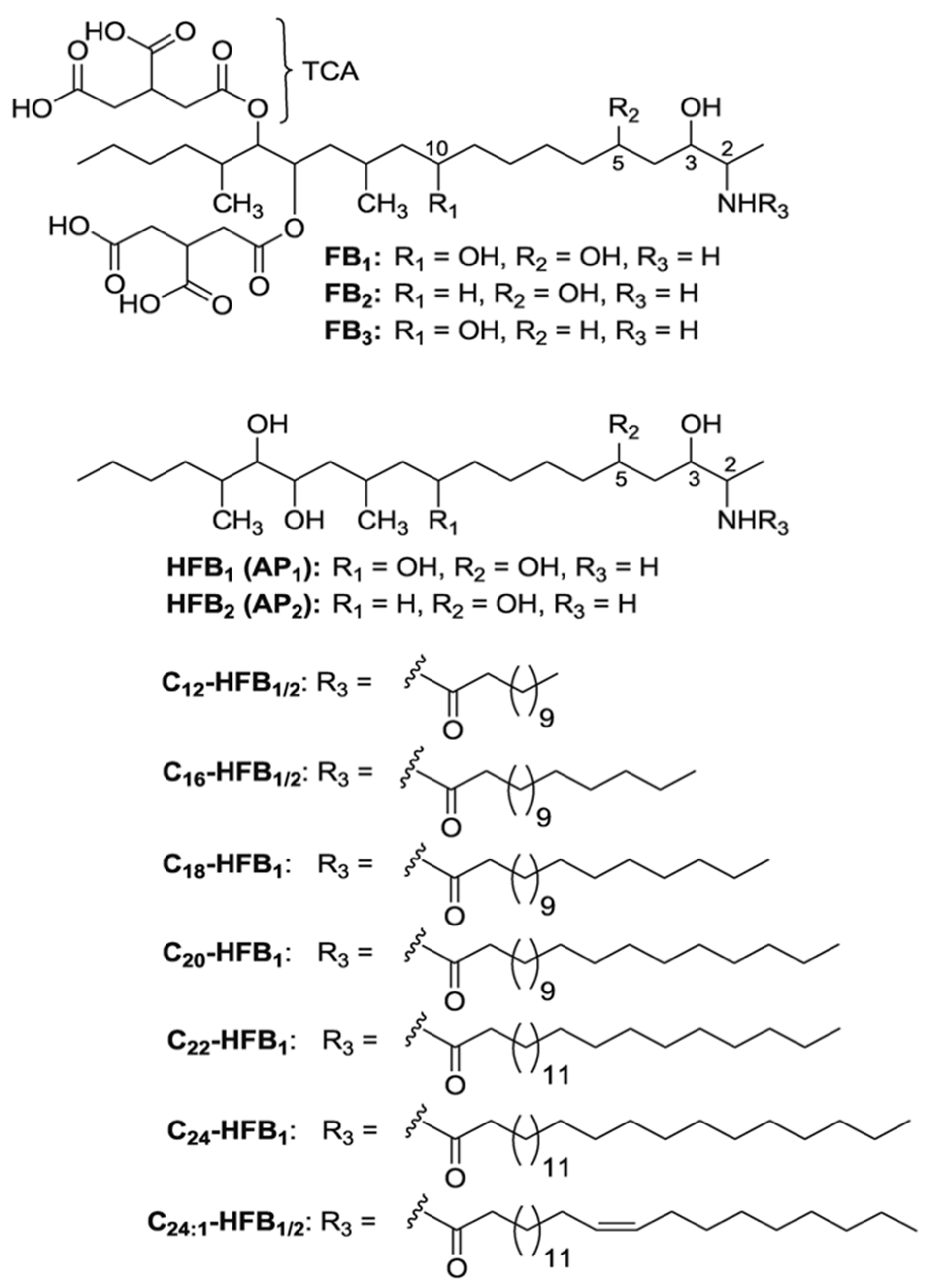
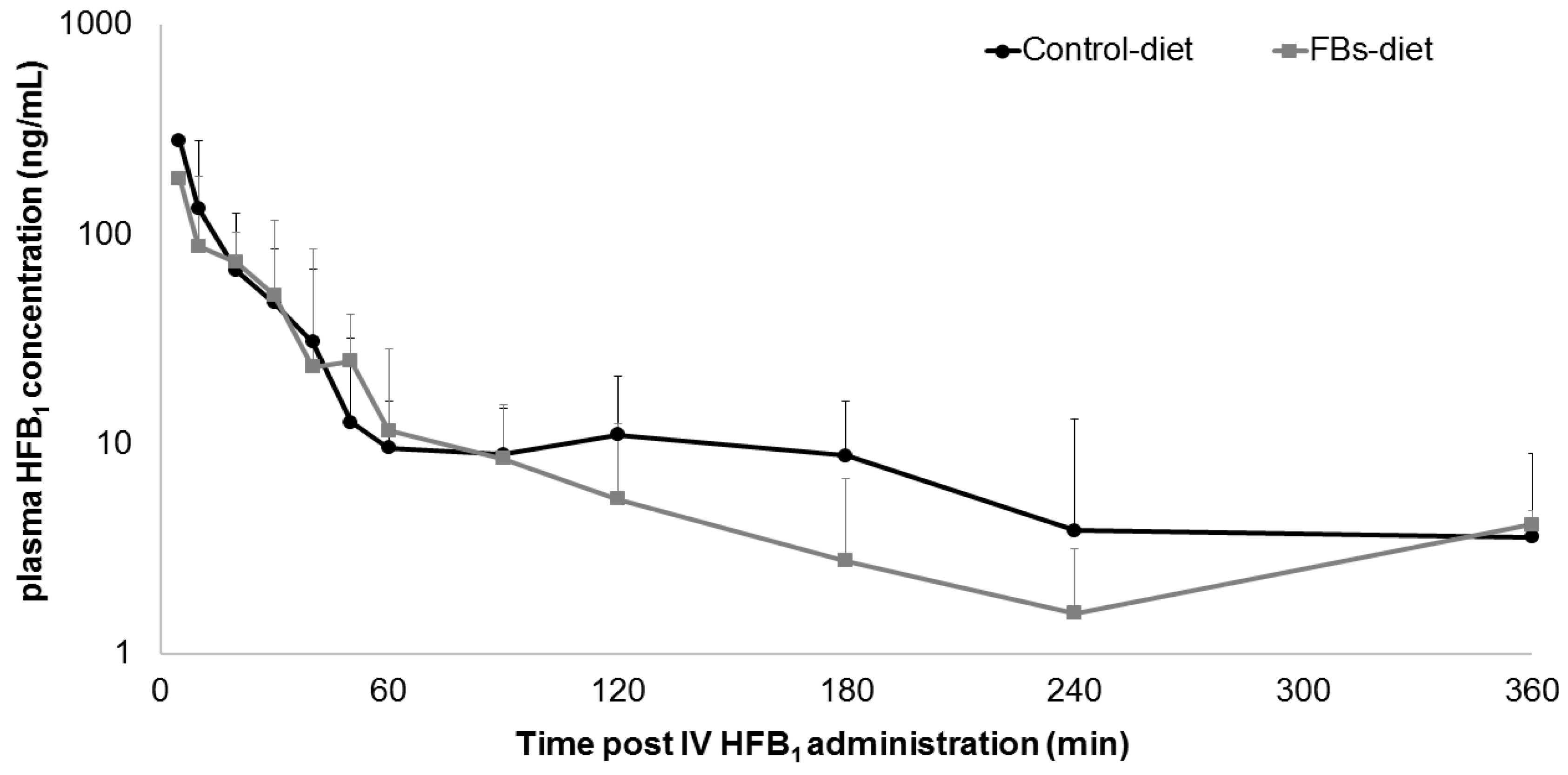
2. Results and Discussion
3. Conclusions
4. Materials and Methods
4.1. HFB1 for Animal Trial
4.2. Feed Preparation and Experimental Diets
4.3. Animal Experiment
4.4. Plasma Fumonisins Analysis: FB1, FB2, FB3, HFB1, pHFB1a, pHFB1b, Phase II Metabolites and N-Acyl Metabolites
4.4.1. Preparation of Standard Solutions
4.4.2. Plasma Sample Pre-Treatment
4.4.3. UPLC-MS/MS Method for Quantification
4.4.4. UPLC-HR-MS Analysis for Identification
4.5. Toxicokinetic Modelling
(dA2/dt) × 1/Vp = Q × (C − C2)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gelderblom, W.C.; Jaskiewicz, K.; Marasas, W.F.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P. Fumonisins-novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1988, 54, 1806–1811. [Google Scholar] [CrossRef] [PubMed]
- Voss, K.; Smith, G.; Haschek, W. Fumonisins: Toxicokinetics, mechanism of action and toxicity. Anim. Feed. Sci. Technol. 2007, 137, 299–325. [Google Scholar] [CrossRef]
- Humpf, H.-U.; Voss, K.A. Effects of thermal food processing on the chemical structure and toxicity of fumonisin mycotoxins. Mol. Nutr. Food Res. 2004, 48, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Stockmann-Juvala, H.; Savolainen, K. A review of the toxic effects and mechanisms of action of fumonisin B1. Hum. Exp. Toxicol. 2008, 27, 799–809. [Google Scholar] [CrossRef]
- Seiferlein, M.; Humpf, H.-U.; Voss, K.A.; Sullards, M.C.; Allegood, J.C.; Wang, E.; Merrill, A.H. Hydrolyzed fumonisins HFB1 and HFB2are acylatedin vitroandin vivoby ceramide synthase to form cytotoxicN-acyl-metabolites. Mol. Nutr. Food Res. 2007, 51, 1120–1130. [Google Scholar] [CrossRef]
- Merrill, A.H.; Sullards, M.C.; Wang, E.; Voss, K.A.; Riley, R.T. Sphingolipid metabolism: Roles in signal transduction and disruption by fumonisins. Environ. Health Perspect. 2001, 109, 283–289. [Google Scholar]
- Riley, R.T.; Enongene, E.; Voss, K.A.; Norred, W.P.; Meredith, F.I.; Sharma, R.P.; Spitsbergen, J.; Williams, D.E.; Carlson, D.B.; Merrill, A.H. Sphingolipid perturbations as mechanisms for fumonisin carcinogenesis. Environ. Health Perspect. 2001, 109, 301–308. [Google Scholar]
- Wang, E.; Norred, W.P.; Bacon, C.W.; Riley, R.T.; Merrill, A.H. Inhibition of sphingolipid biosynthesis by fumonisins. Implications for diseases associated with Fusarium moniliforme. J. Boil. Chem. 1991, 266, 14486–14490. [Google Scholar]
- Benlasher, E.; Geng, X.; Nguyen, N.T.X.; Tardieu, D.; Bailly, J.-D.; Auvergne, A.; Guerre, P. Comparative Effects of Fumonisins on Sphingolipid Metabolism and Toxicity in Ducks and Turkeys. Avian Dis. 2012, 56, 120–127. [Google Scholar] [CrossRef]
- Qiu, M.; Liu, X. Determination of sphinganine, sphingosine and Sa/So ratio in urine of humans exposed to dietary fumonisin B1. Food Addit. Contam. 2001, 18, 263–269. [Google Scholar] [CrossRef]
- Bouhet, S.; Oswald, I.P. The intestine as a possible target for fumonisin toxicity. Mol. Nutr. Food Res. 2007, 51, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Applegate, T. Modulation of Intestinal Functions Following Mycotoxin Ingestion: Meta-Analysis of Published Experiments in Animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef] [PubMed]
- Javed, T.; Bennett, G.A.; Richard, J.L.; Dombrink-Kurtzman, M.A.; Côté, L.M.; Buck, W.B. Mortality in broiler chicks on feed amended withFusarium proliferatum culture material or with purified fumonisin B1 and moniliformin. Mycopathologia 1993, 123, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Kubena, L.; Harvey, R.; Buckley, S.; Bailey, R.; Rottinghaus, G. Effects of long-term feeding of diets containing moniliformin, supplied by Fusarium fujikuroi culture material, and fumonisin, supplied by Fusarium moniliforme culture material, to laying hens. Poult. Sci. 1999, 78, 1499–1505. [Google Scholar] [CrossRef]
- le Doux, D.R.; Brown, T.P.; Weibking, T.S.; Rottinghaus, G. Fumonisin Toxicity in Broiler Chicks. J. Veter Diagn. Investig. 1992, 4, 330–333. [Google Scholar] [CrossRef]
- Tardieu, D.; Bailly, J.D.; Benard, G.; Tran, T.S.; Guerre, P. Toxicity of maize containing known levels of fumonisin B1 during force-feeding of ducks. Poult. Sci. 2004, 83, 1287–1293. [Google Scholar] [CrossRef]
- Tran, S.T.; Auvergne, A.; Benard, G.; Bailly, J.D.; Tardieu, D.; Babile, R.; Guerre, P. Chronic effects of fumonisin B1 on ducks. Poult. Sci. 2005, 84, 22–28. [Google Scholar] [CrossRef]
- Weibking, T.S.; le Doux, D.R.; Bermudez, A.J.; Turk, J.R.; Rottinghaus, G.E.; Wang, E.; Merrill, A.H. Effects of Feeding Fusarium moniliforme Culture Material, Containing Known Levels of Fumonisin B1, on the Young Broiler Chick. Poult. Sci. 1993, 72, 456–466. [Google Scholar] [CrossRef]
- Commission of the European Communities Comission recommendation 2006/576/ec of 17 August 20076 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products. Off. J. Eur. Union 2006, 229, 7–9.
- Antonissen, G.; Martel, A.; Pasmans, F.; Ducatelle, R.; Verbrugghe, E.; Vandenbroucke, V.; Li, S.; Haesebrouck, F.; van Immerseel, F.; Croubels, S. The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases. Toxins 2014, 6, 430–452. [Google Scholar] [CrossRef]
- Antonissen, G.; Croubels, S.; Pasmans, F.; Ducatelle, R.; Eeckhaut, V.; Devreese, M.; Verlinden, M.; Haesebrouck, F.; Eeckhout, M.; de Saeger, S.; et al. Fumonisins affect the intestinal microbial homeostasis in broiler chickens, predisposing to necrotic enteritis. Veter Res. 2015, 46, 98. [Google Scholar] [CrossRef] [PubMed]
- Antonissen, G.; van Immerseel, F.; Pasmans, F.; Ducatelle, R.; Janssens, G.P.J.; de Baere, S.; Mountzouris, K.; Su, S.; Wong, E.A.; de Meulenaer, B.; et al. Mycotoxins Deoxynivalenol and Fumonisins Alter the Extrinsic Component of Intestinal Barrier in Broiler Chickens. J. Agric. Food Chem. 2015, 63, 10846–10855. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Schwartz-Zimmermann, H.E.; Caha, S.; Moll, W.-D.; Schatzmayr, G.; Applegate, T. Dose-Dependent Effects on Sphingoid Bases and Cytokines in Chickens Fed Diets Prepared with Fusarium Verticillioides Culture Material Containing Fumonisins. Toxins 2015, 7, 1253–1272. [Google Scholar] [CrossRef] [PubMed]
- Antonissen, G.; Devreese, M.; de Baere, S.; Martel, A.; van Immerseel, F.; Croubels, S. Impact of Fusarium mycotoxins on hepatic and intestinal mRNA expression of cytochrome P450 enzymes and drug transporters, and on the pharmacokinetics of oral enrofloxacin in broiler chickens. Food Chem. Toxicol. 2017, 101, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Bryden, W. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed. Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Heinl, S.; Hartinger, D.; Thamhesl, M.; Vekiru, E.; Krska, R.; Schatzmayr, G.; Moll, W.-D.; Grabherr, R. Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J. Biotechnol. 2010, 145, 120–129. [Google Scholar] [CrossRef]
- Hartinger, D.; Moll, W.-D. Fumonisin elimination and prospects for detoxification by enzymatic transformation. World Mycotoxin J. 2011, 4, 271–283. [Google Scholar] [CrossRef]
- Grenier, B.; Schwartz-Zimmermann, H.; Gruber-Dorninger, C.; Dohnal, I.; Aleschko, M.; Schatzmayr, G.; Moll, W.-D.; Applegate, T.J. Enzymatic hydrolysis of fumonisins in the gastrointestinal tract of broiler chickens. Poult. Sci. 2017, 96, 4342–4351. [Google Scholar] [CrossRef]
- Shephard, G.S.; Thiel, P.; Sydenham, E.; Vleggaar, R.; Alberts, J. Determination of the mycotoxin fumonisin B1 and identification of its partially hydrolysed metabolites in the faeces of non-human primates. Food Chem. Toxicol. 1994, 32, 23–29. [Google Scholar] [CrossRef]
- Fodor, J.; Balogh, K.; Weber, M.; Mézes, M.; Kametler, L.; Pósa, R.; Mamet, R.; Bauer, J.; Horn, P.; Kovács, F.; et al. Absorption, distribution and elimination of fumonisin B1 metabolites in weaned piglets. Food Addit. Contam. Part A 2007, 25, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Hahn, I.; Nagl, V.; Schwartz-Zimmermann, H.; Varga, E.; Schwarz, C.; Slavik, V.; Reisinger, N.; Malachová, A.; Cirlini, M.; Generotti, S.; et al. Effects of orally administered fumonisin B1 (FB1), partially hydrolysed FB1, hydrolysed FB1 and N-(1-deoxy-D-fructos-1-yl) FB1 on the sphingolipid metabolism in rats. Food Chem. Toxicol. 2015, 76, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Masching, S.; Naehrer, K.; Schwartz-Zimmermann, H.E.; Sărăndan, M.; Schaumberger, S.; Dohnal, I.; Nagl, V.; Schatzmayr, D. Gastrointestinal Degradation of Fumonisin B1 by Carboxylesterase FumD Prevents Fumonisin Induced Alteration of Sphingolipid Metabolism in Turkey and Swine. Toxins 2016, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Harrer, H.; Humpf, H.-U.; Voss, K. In vivo formation of N-acyl-fumonisin B1. Mycotoxin Res. 2014, 31, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Humpf, H.-U.; Schmelz, E.M.; Meredith, F.I.; Vesper, H.; Vales, T.R.; Wang, E.; Menaldino, D.S.; Liotta, D.C.; Merrill, A.H. Acylation of Naturally Occurring and Synthetic 1-Deoxysphinganines by Ceramide Synthase. J. Boil. Chem. 1998, 273, 19060–19064. [Google Scholar] [CrossRef]
- Hopmans, E.C.; Hauck, C.C.; Hendrich, S.; Murphy, P.A. Excretion of Fumonisin B1, Hydrolyzed Fumonisin B1, and the Fumonisin B1−Fructose Adduct in Rats. J. Agric. Food Chem. 1997, 45, 2618–2625. [Google Scholar] [CrossRef]
- Vudathala, D.K.; Prelusky, D.B.; Ayroud, M.; Trenholm, H.L.; Miller, J.D. Pharmacokinetic fate and pathological effects of14C-fumonisin B1 in laying hens. Nat. Toxins 1994, 2, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Harrer, H.; Laviad, E.L.; Humpf, H.-U.; Futerman, A.H. Identification ofN-acyl-fumonisin B1 as new cytotoxic metabolites of fumonisin mycotoxins. Mol. Nutr. Food Res. 2012, 57, 516–522. [Google Scholar] [CrossRef]
- Gu, M.J.; Han, S.E.; Hwang, K.; Mayer, E.; Reisinger, N.; Schatzmayr, D.; Park, B.-C.; Han, S.H.; Yun, C.-H. Hydrolyzed fumonisin B1 induces less inflammatory responses than fumonisin B1 in the co-culture model of porcine intestinal epithelial and immune cells. Toxicol. Lett. 2019, 305, 110–116. [Google Scholar] [CrossRef]
- Voss, K.A.; Bacon, C.W.; Meredith, F.I.; Norred, W.P. Comparative Subchronic Toxicity Studies of Nixtamalized and Water-extracted Fusarium. Food Chem. Toxicol. 1996, 34, 623–632. [Google Scholar] [CrossRef]
- Howard, P.C.C.; Howarda, P.; Coucha, L.H.; Pattonb, R.E.; Eppleyc, R.M.; Doergea, D.R.; Churchwella, M.I.; Marques, M.M.; Okerbergb, C.V. Comparison of the Toxicity of Several Fumonisin Derivatives in a 28-Day Feeding Study with Female B6C3F1 Mice. Toxicol. Appl. Pharmacol. 2002, 185, 153–165. [Google Scholar] [CrossRef]
- Grenier, B.; Bracarense, A.-P.F.; Schwartz-Zimmermann, H.; Trumel, C.; Cossalter, A.-M.; Schatzmayr, G.; Kolf-Clauw, M.; Moll, W.-D.; Oswald, I.P. The low intestinal and hepatic toxicity of hydrolyzed fumonisin B1 correlates with its inability to alter the metabolism of sphingolipids. Biochem. Pharmacol. 2012, 83, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Schertz, H.; Kluess, J.; Frahm, J.; Schatzmayr, D.; Dohnal, I.; Bichl, G.; Schwartz-Zimmermann, H.; Breves, G.; Dänicke, S. Oral and Intravenous Fumonisin Exposure in Pigs—A Single-Dose Treatment Experiment Evaluating Toxicokinetics and Detoxification. Toxins 2018, 10, 150. [Google Scholar] [CrossRef] [PubMed]
- Antonissen, G.; Devreese, M.; van Immerseel, F.; de Baere, S.; Hessenberger, S.; Martel, A.; Croubels, S. Chronic Exposure to Deoxynivalenol Has No Influence on the Oral Bioavailability of Fumonisin B1 in Broiler Chickens. Toxins 2015, 7, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Guerre, P. Fusariotoxins in Avian Species: Toxicokinetics, Metabolism and Persistence in Tissues. Toxins 2015, 7, 2289–2305. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, W.R.; Hopper, J.; Mullin, K.; Hendrich, S.; Murphy, P.A. Excretion of14C-Fumonisin B1,14C-Hydrolyzed Fumonisin B1, and14C-Fumonisin B1-Fructose in Rats. J. Agric. Food Chem. 1999, 47, 4291–4296. [Google Scholar] [CrossRef] [PubMed]
- Monbaliu, S.; van Poucke, C.; Detavernier, C.; Dumoulin, F.; van de Velde, M.; Schoeters, E.; van Dyck, S.; Averkieva, O.; van Peteghem, C.; de Saeger, S. Occurrence of Mycotoxins in Feed as Analyzed by a Multi-Mycotoxin LC-MS/MS Method. J. Agric. Food Chem. 2010, 58, 66–71. [Google Scholar] [CrossRef]
- Leslie, J.F.; Doe, F.J.; Plattner, R.D.; Shackelford, D.D.; Jonz, J. Fumonisin B1 production and vegetative compatibility of strains from Gibberella fujikuroi mating population ?A? (Fusarium moniliforme). Mycopathologia 1992, 117, 37–45. [Google Scholar] [CrossRef]
- de Baere, S.; Croubels, S.; Novak, B.; Bichl, G.; Antonissen, G. Development and Validation of a UPLC-MS/MS and UPLC-HR-MS Method for the Determination of Fumonisin B1 and Its Hydrolysed Metabolites and Fumonisin B2 in Broiler Chicken Plasma. Toxins 2018, 10, 62. [Google Scholar] [CrossRef]

| Θ | Tvθ | CV (%) | ω |
|---|---|---|---|
| Vc (L/kg) | 3.68 | 23.47 | 0.095 |
| Vp (L/kg) | 5.04 | 41.47 | 0.009 |
| VSS (L/kg) | 8.72 | 28.75 | / |
| Cl (L/kg·h) | 16.39 | 12.67 | 0.126 |
| Q (L/kg·h) | 8.34 | 41.37 | 0.391 |
| AUC0-inf (ng·h/mL) | 76.26 | 12.67 | / |
| C0 (ng/mL) | 339.59 | 23.47 | / |
| Ke (1/h) | 4.45 | 26.27 | / |
| MRT (h) | 0.53 | 31.56 | / |
| T1/2α (h) | 0.09 | 33.26 | / |
| T1/2β (h) | 0.69 | 40.41 | / |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antonissen, G.; De Baere, S.; Novak, B.; Schatzmayr, D.; den Hollander, D.; Devreese, M.; Croubels, S. Toxicokinetics of Hydrolyzed Fumonisin B1 after Single Oral or Intravenous Bolus to Broiler Chickens Fed a Control or a Fumonisins-Contaminated Diet. Toxins 2020, 12, 413. https://doi.org/10.3390/toxins12060413
Antonissen G, De Baere S, Novak B, Schatzmayr D, den Hollander D, Devreese M, Croubels S. Toxicokinetics of Hydrolyzed Fumonisin B1 after Single Oral or Intravenous Bolus to Broiler Chickens Fed a Control or a Fumonisins-Contaminated Diet. Toxins. 2020; 12(6):413. https://doi.org/10.3390/toxins12060413
Chicago/Turabian StyleAntonissen, Gunther, Siegrid De Baere, Barbara Novak, Dian Schatzmayr, Danica den Hollander, Mathias Devreese, and Siska Croubels. 2020. "Toxicokinetics of Hydrolyzed Fumonisin B1 after Single Oral or Intravenous Bolus to Broiler Chickens Fed a Control or a Fumonisins-Contaminated Diet" Toxins 12, no. 6: 413. https://doi.org/10.3390/toxins12060413
APA StyleAntonissen, G., De Baere, S., Novak, B., Schatzmayr, D., den Hollander, D., Devreese, M., & Croubels, S. (2020). Toxicokinetics of Hydrolyzed Fumonisin B1 after Single Oral or Intravenous Bolus to Broiler Chickens Fed a Control or a Fumonisins-Contaminated Diet. Toxins, 12(6), 413. https://doi.org/10.3390/toxins12060413







