Differences in Toxic Response Induced by Three Variants of the Diarrheic Shellfish Poisoning Phycotoxins in Human Intestinal Epithelial Caco-2 Cells
Abstract
1. Introduction
2. Results
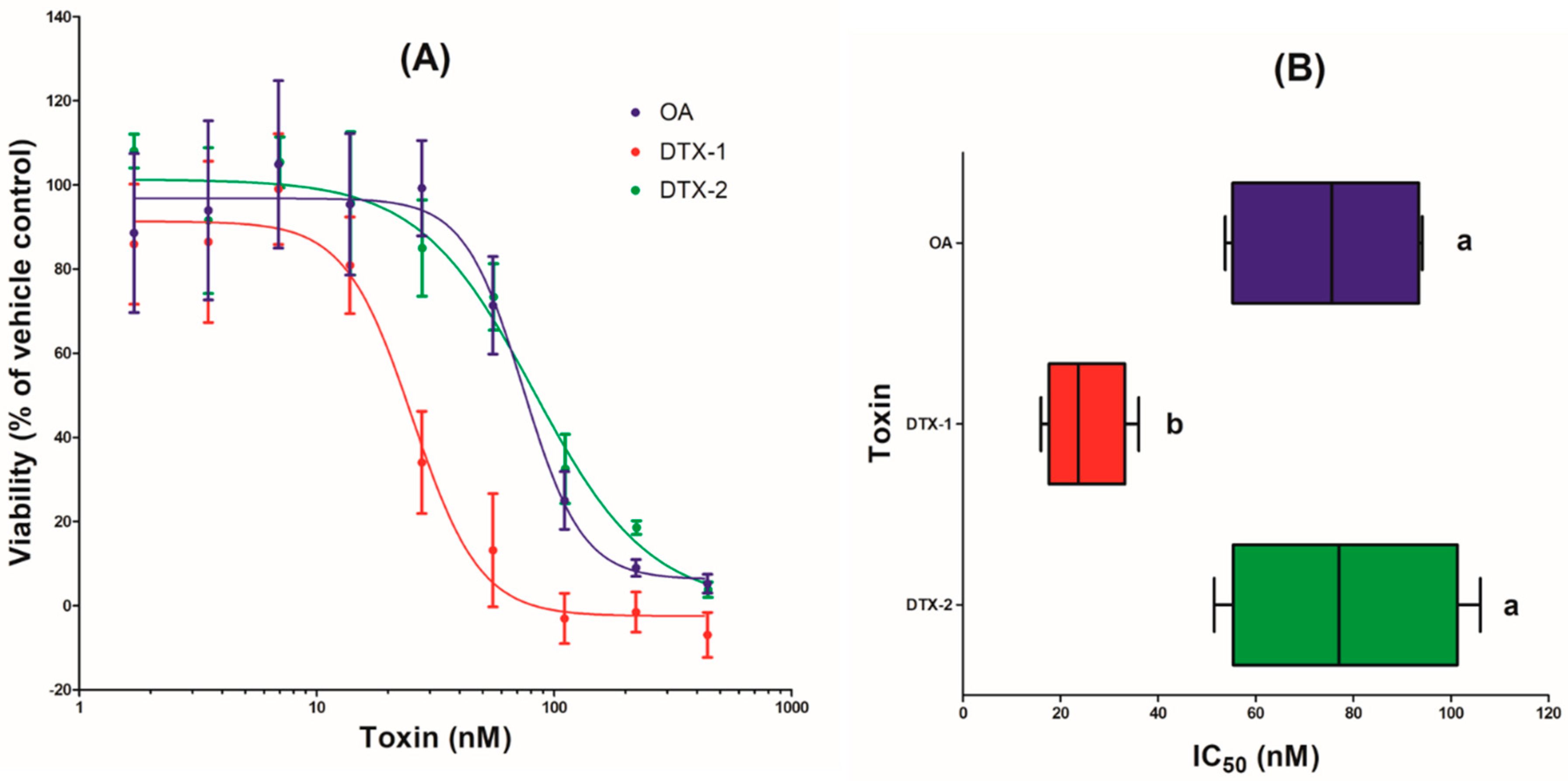
2.1. Cytotoxicity
2.2. Transcriptomic Profile
2.3. OA-, DTX-1- and DTX-2-Induced Gene Expression
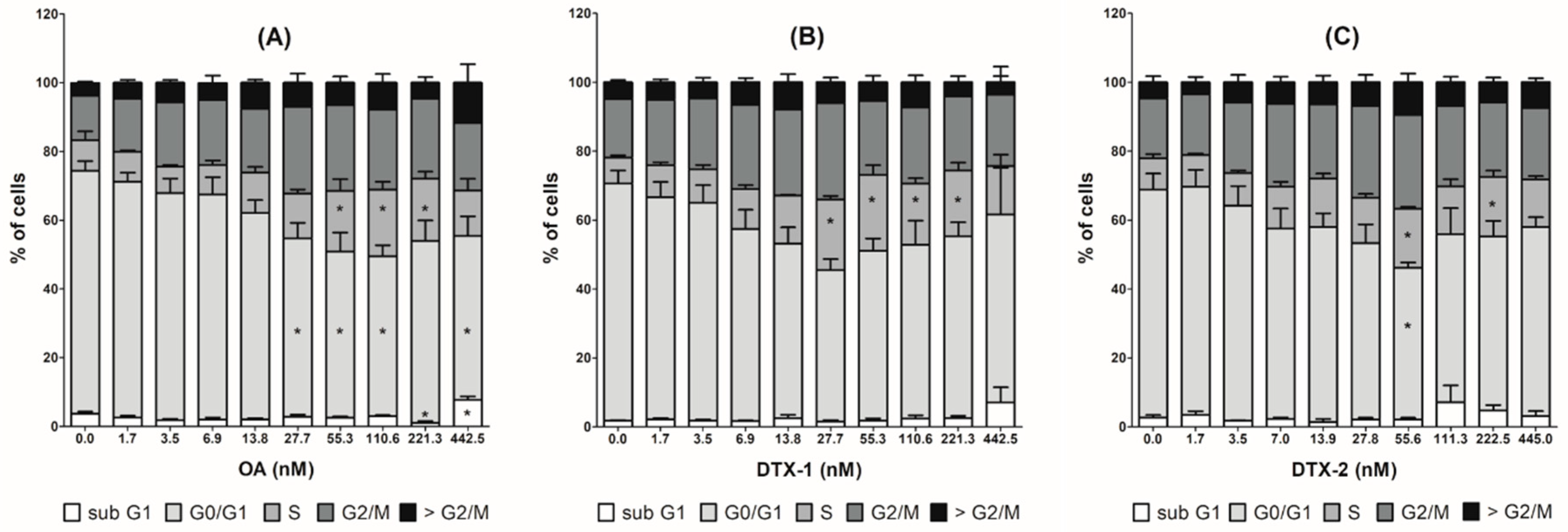
2.4. Cell Cycle Arrest and MAP Kinase Pathway Quantified by High Content Analysis
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Cell Culture and Toxin Exposure
4.3. Cytotoxicity Evaluation by Neutral Red Uptake Assay
4.4. Microarray Experiments and Data Analysis
4.5. RT-qPCR
4.6. HCA of Multiparametric Toxicity Endpoints
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Farabegoli, F.; Blanco, L.; Rodriguez, L.P.; Vieites, J.M.; Cabado, A.G. Phycotoxins in Marine Shellfish: Origin, Occurrence and Effects on Humans. Mar. Drugs 2018, 16, 188. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, S.F.; Carrera, C.; Vilarino, N.; Louzao, M.C.; Santamarina, G.; Cantalapiedra, A.G.; Botana, M.L. Acute cardiotoxicity evaluation of the marine biotoxins OA, DTX-1 and YTX. Toxins 2015, 7, 1030–1047. [Google Scholar] [CrossRef] [PubMed]
- Valdiglesias, V.; Prego-Faraldo, M.V.; Paśaro, E.; Meńdez, J.; Laffon, B. Okadaic Acid: More than a diarrheic toxin. Mar. Drugs 2013, 11, 4328–4343. [Google Scholar] [CrossRef] [PubMed]
- Reguera, B.; Riobó, P.; Rodríguez, F.; Díaz, P.A.; Pizarro, G.; Paz, B.; Franco, J.M.; Blanco, J. Dinophysis toxins: Causative organisms, distribution and fate in shellfish. Mar. Drugs 2014, 12, 394–461. [Google Scholar] [CrossRef]
- Reguera, B.; Velo-Suárez, L.; Raine, R.; Park, M.G. Harmful Dinophysis species: A review. Harmful Algae 2012, 14, 87–106. [Google Scholar] [CrossRef]
- Takai, A.; Murata, M.; Isobe, M.; Mieskes, G.; Yasumoto, T. Inhibitory effect of okadaic acid derivatives on protein phosphatases. A study on structure-affinity relationship. Biochem. J. 1992, 284, 539–544. [Google Scholar] [CrossRef]
- Bialojan, C.; Takai, A. Inhibitory effect of a marine-sponge toxin, okadaic acid, on protein phosphatases. Specificity and kinetics. Biochem. J. 1988, 256, 283–290. [Google Scholar] [CrossRef]
- Louzao, M.C.; Vieytes, M.R.; Botana, L.M. Effect of okadaic acid on glucose regulation. Mini Rev. Med. Chem. 2005, 5, 207–215. [Google Scholar] [CrossRef]
- Vale, C.; Botana, L.M. Marine toxins and the cytoskeleton: Okadaic acid and dinophysistoxins. FEBS J. 2008, 275, 6060–6066. [Google Scholar] [CrossRef]
- Larsen, K.; Petersen, D.; Wilkins, A.L.; Samdal, I.A.; Sandvik, M.; Rundberget, T.; Goldstone, D.; Arcus, V.; Hovgaard, P.; Rise, F.; et al. Clarification of the C-35 stereochemistries of dinophysistoxin-1 and dinophysistoxin-2 and its consequences for binding to protein phosphatase. Chem. Res. Toxicol. 2007, 20, 868–875. [Google Scholar] [CrossRef]
- Huhn, J.; Jeffrey, P.D.; Larsen, K.; Rundberget, T.; Rise, F.; Cox, N.R.; Arcus, V.; Shi, Y.; Miles, C.O. A structural basis for the reduced toxicity of dinophysistoxin-2. Chem. Res. Toxicol. 2009, 22, 1782–1786. [Google Scholar] [CrossRef] [PubMed]
- Rivas, M.; Garcia, C.; Liberona, J.L.; Lagos, N. Biochemical characterization and inhibitory effects of dinophysistoxin-1, okadaic acid and microcystine 1-r on protein phosphatase 2a purified from the mussel Mytilus chilensis. Biol. Res. 2000, 33, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Twiner, M.J.; Doucette, G.J.; Pang, Y.; Fang, C.; Forsyth, C.J.; Miles, C.O. Structure-Activity Relationship Studies Using Natural and Synthetic Okadaic Acid/Dinophysistoxin Toxins. Mar. Drugs 2016, 14, 207. [Google Scholar] [CrossRef] [PubMed]
- Rubiolo, J.A.; Lopez-Alonso, H.; Vega, F.V.; Vieytes, M.R.; Botana, L.M. Comparative study of toxicological and cell cycle effects of okadaic acid and dinophysistoxin-2 in primary rat hepatocytes. Life Sci. 2012, 90, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Ferron, P.J.; Hogeveen, K.; Fessard, V.; Le Hégarat, L. Comparative analysis of the cytotoxic effects of okadaic acid-group toxins on human intestinal cell lines. Mar. Drugs 2014, 12, 4616–4634. [Google Scholar] [CrossRef] [PubMed]
- Solino, L.; Sureda, F.X.; Diogene, J. Evaluation of okadaic acid, dinophysistoxin-1 and dinophysistoxin-2 toxicity on Neuro-2a, NG108-15 and MCF-7 cell lines. Toxicol. Vitro 2015, 29, 59–62. [Google Scholar] [CrossRef]
- Abal, P.; Louzao, M.C.; Cifuentes, J.M.; Vilarino, N.; Rodriguez, I.; Alfonso, A.; Mercedes, R.V.; Botana, L.M. Characterization of the dinophysistoxin-2 acute oral toxicity in mice to define the Toxicity Equivalency Factor. Food Chem. Toxicol. 2017, 102, 166–175. [Google Scholar] [CrossRef]
- Abal, P.; Louzao, M.C.; Suzuki, T.; Watanabe, R.; Vilarino, N.; Carrera, C.; Botana, A.M.; Mercedes, R.V.; Botana, L.M. Toxic Action Reevaluation of Okadaic Acid, Dinophysistoxin-1 and Dinophysistoxin-2: Toxicity Equivalency Factors Based on the Oral Toxicity Study. Cell. Physiol. Biochem. 2018, 49, 743–757. [Google Scholar] [CrossRef]
- Suzuki, H.; Okada, Y. Comparative toxicity of dinophysistoxin-1 and okadaic acid in mice. J. Vet. Med. Sci. 2018, 80, 616–619. [Google Scholar] [CrossRef]
- Munday, R. Is protein phosphatase inhibition responsible for the toxic effects of Okadaic acid in animals? Toxins 2013, 5, 267–285. [Google Scholar] [CrossRef]
- Serandour, A.L.; Ledreux, A.; Morin, B.; Derick, S.; Augier, E.; Lanceleur, R.; Hamlaoui, S.; Moukha, S.; Furger, C.; Biré, R.; et al. Collaborative study for the detection of toxic compounds in shellfish extracts using cell-based assays. Part I: Screening strategy and pre-validation study with lipophilic marine toxins. Anal. Bioanal. Chem. 2012, 403, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, M.; Toledo, H.; Lagos, N. Okadaic acid toxin at sublethal dose produced cell proliferation in gastric and colon epithelial cell lines. Mar. Drugs 2013, 11, 4751–4760. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, D.A.; Louzao, M.C.; Fraga, M.; Vilarino, N.; Vieytes, M.R.; Botana, L.M. Experimental basis for the high oral toxicity of dinophysistoxin 1: A comparative study of DSP. Toxins 2014, 6, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Bodero, M.; Hoogenboom, R.; Bovee, T.F.H.; Portier, L.; de Haan, L.; Peijnenburg, A.; Hendriksen, P.J.M. Whole genome mRNA transcriptomics analysis reveals different modes of action of the diarrheic shellfish poisons okadaic acid and dinophysis toxin-1 versus azaspiracid-1 in Caco-2 cells. Toxicol. Vitro 2018, 46, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Oteri, G.; Stammati, A.; Zampaglioni, F.; Zucco, F. Evaluation of the use of two human cell lines for okadaic acid and DTX-1 determination by cytotoxicity assays and damage characterization. Nat. Toxins 1998, 6, 197–209. [Google Scholar] [CrossRef]
- Ferron, P.J.; Hogeveen, K.; De Sousa, G.; Rahmani, R.; Dubreil, E.; Fessard, V.; Le Hégarat, L. Modulation of CYP3A4 activity alters the cytotoxicity of lipophilic phycotoxins in human hepatic HepaRG cells. Toxicol. Vitro 2016, 33, 136–146. [Google Scholar] [CrossRef]
- Jiménez-Cárcamo, D.; García, C.; Contreras, H.R. Toxins of Okadaic Acid-Group Increase Malignant Properties in Cells of Colon Cancer. Toxins 2020, 12, 179. [Google Scholar] [CrossRef]
- Le Hegarat, L.; Roudot, A.C.; Fessard, V. Benchmark dose analyses of gammaH2AX and pH3 endpoints for quantitative comparison of in vitro genotoxicity potential of lipophilic phycotoxins. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2020, 852, 503169. [Google Scholar] [CrossRef]
- Woetmann, A.; Nielsen, M.; Christensen, S.T.; Brockdorff, J.; Kaltoft, K.; Engel, A.M.; Skov, S.; Brender, C.; Geisler, C.; Svejgaard, C.; et al. Inhibition of protein phosphatase 2A induces serine/threonine phosphorylation, subcellular redistribution, and functional inhibition of STAT3. Proc. Natl. Acad. Sci. USA 1999, 96, 10620–10625. [Google Scholar] [CrossRef]
- Saydam, G.; Aydin, H.H.; Sahin, F.; Selvi, N.; Oktem, G.; Terzioglu, E.; Buyukkececi, F.; Omay, S.B. Involvement of protein phosphatase 2A in interferon-alpha-2b-induced apoptosis in K562 human chronic myelogenous leukaemia cells. Leuk. Res. 2003, 27, 709–717. [Google Scholar] [CrossRef]
- Nicolas, C.S.; Amici, M.; Bortolotto, Z.A.; Doherty, A.; Csaba, Z.; Fafouri, A.; Dournaud, P.; Gressens, P.; Collingridge, G.L.; Peineau, S. The role of JAK-STAT signaling within the CNS. JAKSTAT 2013, 2, e22925. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.J.; Schwartz, D.M.; Villarino, A.V.; Gadina, M.; McInnes, I.B.; Laurence, A. The JAK-STAT pathway: Impact on human disease and therapeutic intervention. Annu. Rev. Med. 2015, 66, 311–328. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.; Clemente-Blanco, A. Cell Cycle and DNA Repair Regulation in the Damage Response: Protein Phosphatases Take Over the Reins. Int. J. Mol. Sci. 2020, 21, 446. [Google Scholar] [CrossRef] [PubMed]
- Von Kriegsheim, A.; Pitt, A.; Grindlay, G.J.; Kolch, W.; Dhillon, A.S. Regulation of the Raf-MEK-ERK pathway by protein phosphatase 5. Nat. Cell Biol. 2006, 8, 1011–1101. [Google Scholar] [CrossRef] [PubMed]
- Osaki, L.H.; Gama, P. MAPKs and signal transduction in the control of gastrointestinal epithelial cell proliferation and differentiation. Int. J. Mol. Sci. 2013, 14, 10143–10161. [Google Scholar] [CrossRef]
- Tang, X.; Liu, H.; Yang, S.; Li, Z.; Zhong, J.; Fang, R. Epidermal Growth Factor and Intestinal Barrier Function. Mediat. Inflamm. 2016, 2016, 1927348. [Google Scholar] [CrossRef]
- Bryce, S.M.; Bernacki, D.T.; Bemis, J.C.; Dertinger, S.D. Genotoxic mode of action predictions from a multiplexed flow cytometric assay and a machine learning approach. Environ. Mol. Mutagen. 2016, 57, 171–189. [Google Scholar] [CrossRef]
- Traore, A.; Baudrimont, I.; Ambaliou, S.; Dano, S.D.; Creppy, E.E. DNA breaks and cell cycle arrest induced by okadaic acid in Caco-2 cells, a human colonic epithelial cell line. Arch. Toxicol. 2001, 75, 110–117. [Google Scholar] [CrossRef]
- Rubiolo, J.A.; López-Alonso, H.; Vega, F.V.; Vieytes, M.R.; Botana, L.M. Okadaic acid and dinophysis toxin 2 have differential toxicological effects in hepatic cell lines inducing cell cycle arrest, at G0/G1 or G2/M with aberrant mitosis depending on the cell line. Arch. Toxicol. 2011, 85, 1541–1550. [Google Scholar] [CrossRef]
- Huguet, A.; Lanceleur, R.; Quenault, H.; Le Hegarat, L.; Fessard, V. Identification of key pathways involved in the toxic response of the cyanobacterial toxin cylindrospermopsin in human hepatic HepaRG cells. Toxicol. Vitro 2019, 58, 69–77. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Huguet, A.; Hatton, A.; Villot, R.; Quenault, H.; Blanchard, Y.; Fessard, V. Modulation of chromatin remodelling induced by the freshwater cyanotoxin cylindrospermopsin in human intestinal caco-2 cells. PLoS ONE 2014, 9, e99121. [Google Scholar] [CrossRef] [PubMed]
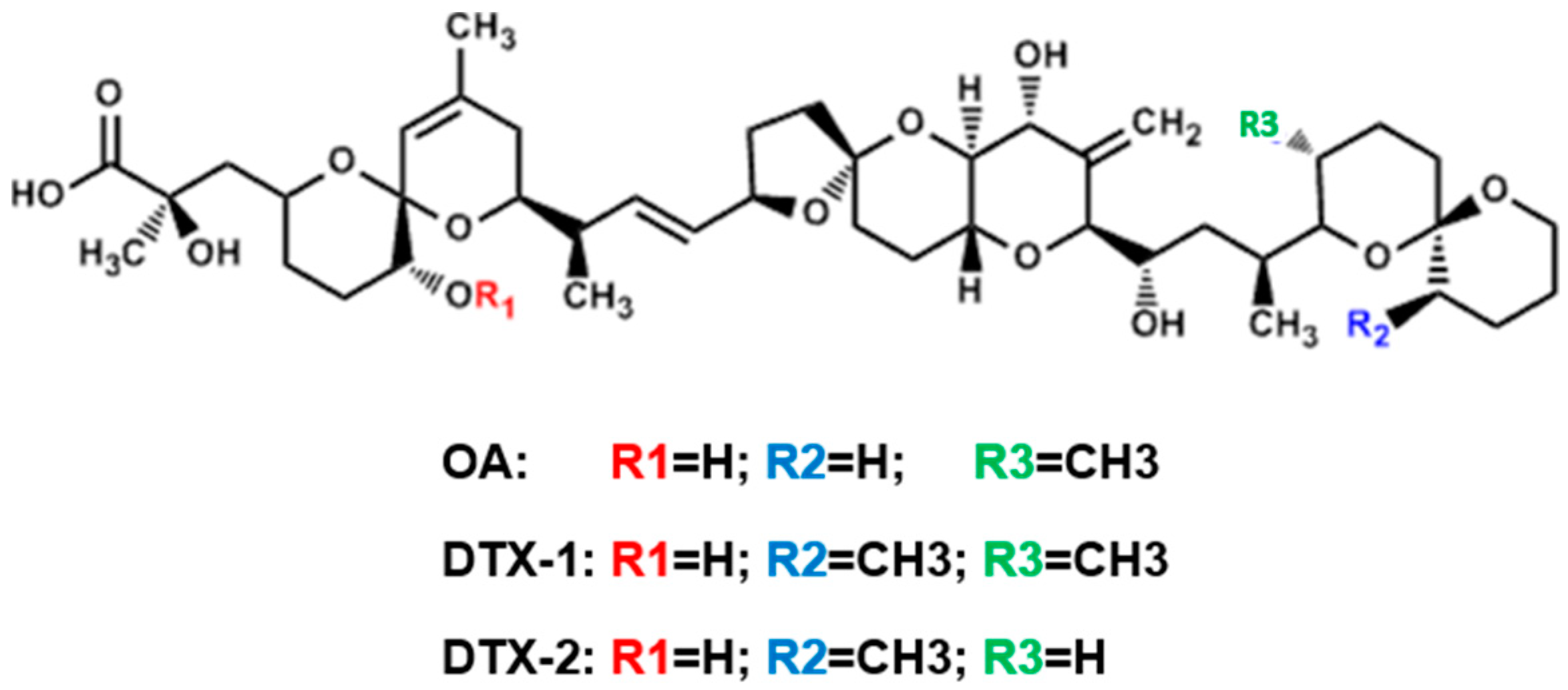
| Gene | Control | OA1 3.8 nM | OA 27.7 nM | OA 55.3 nM | DTX-1 3.5 nM | DTX-1 6.9 nM | DTX-1 13.8 nM | DTX-2 13.9 nM | DTX-2 27.8 nM | DTX-2 55.6 nM |
|---|---|---|---|---|---|---|---|---|---|---|
| ccnd1 | 0.18 ± 0.01 | 0.25 ± 0.03 | 0.35 ± 0.01 b | 0.31 ± 0.01 | 0.19 ± 0.01 | 0.24 ± 0.01 | 0.27 ± 0.01 | 0.20 ± 0.01 | 0.26 ± 0.01 | 0.26 ± 0.01 |
| ccnd2 | 0.28 ± 0.02 | 0.32 ± 0.01 | 0.38 ± 0.08 | 0.09 ± 0.01 b | 0.26 ± 0.04 | 0.29 ± 0.03 | 0.26 ± 0.02 | 0.30 ± 0.02 | 0.27 ± 0.02 | 0.15 ± 0.05 |
| rela | 0.99 ± 0.17 | 0.96 ± 0.05 | 0.96 ± 0.16 | 2.60 ± 0.26 c | 0.83 ± 0.13 | 0.86 ± 0.09 | 1.28 ± 0.17 | 0.86 ± 0.11 | 1.02 ± 0.13 | 1.44 ± 0.25 |
| nras | 0.48 ± 0.05 | 0.48 ± 0.02 | 0.54 ± 0.07 | 0.86 ± 0.05 c | 0.39 ± 0.04 | 0.45 ± 0.03 | 0.51 ± 0.05 | 0.45 ± 0.04 | 0.44 ± 0.04 | 0.60 ± 0.08 |
| map2k1 | 0.64 ± 0.12 | 0.46 ± 0.03 | 0.54 ± 0.12 | 0.93 ± 0.20 | 0.69 ± 0.07 | 0.59 ± 0.04 | 0.54 ± 0.02 | 0.48 ± 0.06 | 0.63 ± 0.09 | 1.13 ± 0.23 a |
| mapk8 | 0.39 ± 0.02 | 0.34 ± 0.04 | 0.35 ± 0.04 | 0.52 ± 0.03 | 0.39 ± 0.03 | 0.41 ± 0.03 | 0.38 ± 0.05 | 0.33 ± 0.03 | 0.40 ± 0.05 | 0.48 ± 0.01 |
| cdkn1b | 0.34 ± 0.04 | 0.30 ± 0.03 | 0.32 ± 0.05 | 0.55 ± 0.07 a | 0.38 ± 0.02 | 0.38 ± 0.04 | 0.44 ± 0.01 | 0.31 ± 0.03 | 0.42 ± 0.08 | 0.60 ± 0.06 b |
| hras | 0.38 ± 0.03 | 0.47 ± 0.04 | 0.53 ± 0.04 | 0.62 ± 0.03 b | 0.39 ± 0.06 | 0.41 ± 0.05 | 0.52 ± 0.03 | 0.37 ± 0.02 | 0.46 ± 0.05 | 0.52 ± 0.05 |
| rfc1 | 0.79 ± 0.16 | 0.66 ± 0.03 | 0.48 ± 0.06 a | 0.33 ± 0.01 c | 0.68 ± 0.05 | 0.58 ± 0.05 | 0.45 ± 0.03 b | 0.55 ± 0.06 | 0.45 ± 0.02 b | 0.45 ± 0.04 b |
| rfc4 | 0.63 ± 0.05 | 0.53 ± 0.04 | 0.46 ± 0.05 | 0.20 ± 0.02 c | 0.56 ± 0.05 | 0.59 ± 0.07 | 0.35 ± 0.03 b | 0.51 ± 0.04 | 0.44 ± 0.05 | 0.37 ± 0.07 b |
| rpa1 | 0.58 ± 0.05 | 0.53 ± 0.07 | 0.50 ± 0.02 | 0.23 ± 0.01 c | 0.49 ± 0.04 | 0.54 ± 0.02 | 0.41 ± 0.01 a | 0.52 ± 0.06 | 0.43 ± 0.04 | 0.38 ± 0.05 a |
| rpa3 | 0.50 ± 0.03 | 0.49 ± 0.03 | 0.46 ± 0.05 | 0.25 ± 0.03 b | 0.55 ± 0.04 | 0.51 ± 0.02 | 0.41 ± 0.01 | 0.62 ± 0.05 | 0.53 ± 0.09 | 0.38 ± 0.06 |
| OA | Control | 1.7 nM | 3.5 nM | 6.9 nM | 13.8 nM | 27.7 nM | 55.3 nM | 110.6 nM | 221.3 nM | 442.5 nM |
| Cell count | 100 ± 0.0 | 94.2 ± 5.1 | 97.8 ± 6.0 | 86.6 ± 4.2 a | 73.2 ± 8.4 a | 48.8 ± 11.2 c | 22.8 ± 6.9 c | 16.3 ± 3.5 c | 11.2 ± 4.0 c | 6.8 ± 1.6 c |
| Phospho-H3 | 2.0 ± 0.0 | 2.2 ± 0.2 | 2.2 ± 0.5 | 2.2 ± 0.5 | 3.2 ± 0.9 | 8.5 ± 1.8 | 13.2 ± 3.1 | 12.0 ± 3.6 | 14.5 ± 5.5 | 19.0 ± 6.3 b |
| EdU | 5.7 ± 0.2 | 14.0 ± 1.2 b | 13.2 ± 2.7 a | 13.7 ± 2.2 b | 9.7 ± 2.4 | 7.2 ± 1.8 | 0.2 ± 0.2 | 0.5 ± 0.3 | 0.5 ± 0.5 | 0.0 ± 0.0 |
| Phospho-MEK2 | 5.0 ± 0.0 | 5.2 ± 1.1 | 5.5 ± 1.6 | 5.0 ± 1.4 | 8.0 ± 2.7 | 6.0 ± 2.0 | 12.5 ± 3.3 | 20.0 ± 4.6 | 29.0 ± 8.3 b | 31.2 ± 9.2 b |
| DTX1 | Control | 1.7 nM | 3.5 nM | 6.9 nM | 13.8 nM | 27.7 nM | 55.3 nM | 110.6 nM | 221.3 nM | 442.5 nM |
| Cell count | 100 ± 0.0 | 106.2 ± 3.2 | 90.9 ± 5.9 | 75.5 ± 7.5 a | 46.5 ± 9.9 c | 23.0 ± 6.6 c | 19.2 ± 5.8 c | 21.1 ± 5.2 c | 15.4 ± 5.4 c | 7.8 ± 2.5 c |
| Phospho-H3 | 2.0 ± 0.0 | 2.7 ± 0.3 | 3.8 ± 0.9 | 4.8 ± 1.6 | 8.3 ± 2.4 | 9.4 ± 4.2 | 8.6 ± 2.7 | 13.1 ± 6.5 | 21.5 ± 13.5 | 16.3 ± 9.2 |
| EdU | 5.7 ± 0.2 | 9.2 ± 0.7 | 5.0 ± 2.7 | 9.4 ± 6.2 | 2.0 ± 1.4 | 0.2 ± 0.1 | 0.1 ± 0.1 | 0.2 ± 0.1 | 0.2 ± 0.2 | 0.0 ± 0.0 |
| Phospho-MEK2 | 5.0 ± 0.0 | 9.8 ± 2.4 | 5.7 ± 1.2 | 9.0 ± 2.9 | 13.9 ± 4.6 | 16.8 ± 2.7 | 13.2 ± 4.1 | 18.3 ± 3.9 | 22.1 ± 10.7 | 42.4 ± 14.0 b |
| DTX2 | Control | 1.7 nM | 3.5 nM | 7 nM | 13.9 nM | 27.8 nM | 55.6 nM | 111.3 nM | 222.5 nM | 445 nM |
| Cell count | 100 ± 0.0 | 88.9 ± 7.1 | 84.3 ± 6.8 | 81.4 ± 5.0 b | 66.3 ± 9.1 b | 42.3 ± 8.9 c | 24.2 ± 6.9 c | 18.2 ± 4.7 c | 22.7 ± 6.4 c | 16.5 ± 4.2 c |
| Phospho-H3 | 2.0 ± 0.0 | 3.0 ± 0.4 | 2.5 ± 0.1 | 2.5 ± 0.5 | 6.1 ± 3.1 | 7.8 ± 3.1 | 10.0 ± 3.3 | 13.0 ± 2.9 | 22.1 ± 10.4 a | 15.3 ± 7.2 |
| EdU | 5.7 ± 0.2 | 4.1 ± 2.0 | 2.7 ± 1.5 | 9.1 ± 5.1 | 7.7 ± 5.9 | 0.7 ± 0.6 | 4.5 ± 4.1 | 3.5 ± 3.1 | 0.1 ± 0.1 | 0.1 ± 0.1 |
| Phospho-MEK2 | 5.0 ± 0.0 | 4.3 ± 0.8 | 4.6 ± 1.3 | 5.2 ± 0.4 | 5.8 ± 2.0 | 7.7 ± 2.1 | 12.3 ± 3.9 | 14.1 ± 3.1 | 13.8 ± 4.3 | 19.2 ± 5.3 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huguet, A.; Drapeau, O.; Rousselet, F.; Quenault, H.; Fessard, V. Differences in Toxic Response Induced by Three Variants of the Diarrheic Shellfish Poisoning Phycotoxins in Human Intestinal Epithelial Caco-2 Cells. Toxins 2020, 12, 783. https://doi.org/10.3390/toxins12120783
Huguet A, Drapeau O, Rousselet F, Quenault H, Fessard V. Differences in Toxic Response Induced by Three Variants of the Diarrheic Shellfish Poisoning Phycotoxins in Human Intestinal Epithelial Caco-2 Cells. Toxins. 2020; 12(12):783. https://doi.org/10.3390/toxins12120783
Chicago/Turabian StyleHuguet, Antoine, Olivia Drapeau, Fanny Rousselet, Hélène Quenault, and Valérie Fessard. 2020. "Differences in Toxic Response Induced by Three Variants of the Diarrheic Shellfish Poisoning Phycotoxins in Human Intestinal Epithelial Caco-2 Cells" Toxins 12, no. 12: 783. https://doi.org/10.3390/toxins12120783
APA StyleHuguet, A., Drapeau, O., Rousselet, F., Quenault, H., & Fessard, V. (2020). Differences in Toxic Response Induced by Three Variants of the Diarrheic Shellfish Poisoning Phycotoxins in Human Intestinal Epithelial Caco-2 Cells. Toxins, 12(12), 783. https://doi.org/10.3390/toxins12120783





