Detoxification of the Fumonisin Mycotoxins in Maize: An Enzymatic Approach
Abstract
:1. Introduction
2. Results
2.1. Validation of The Extraction and Chromatographic Quantification Methods
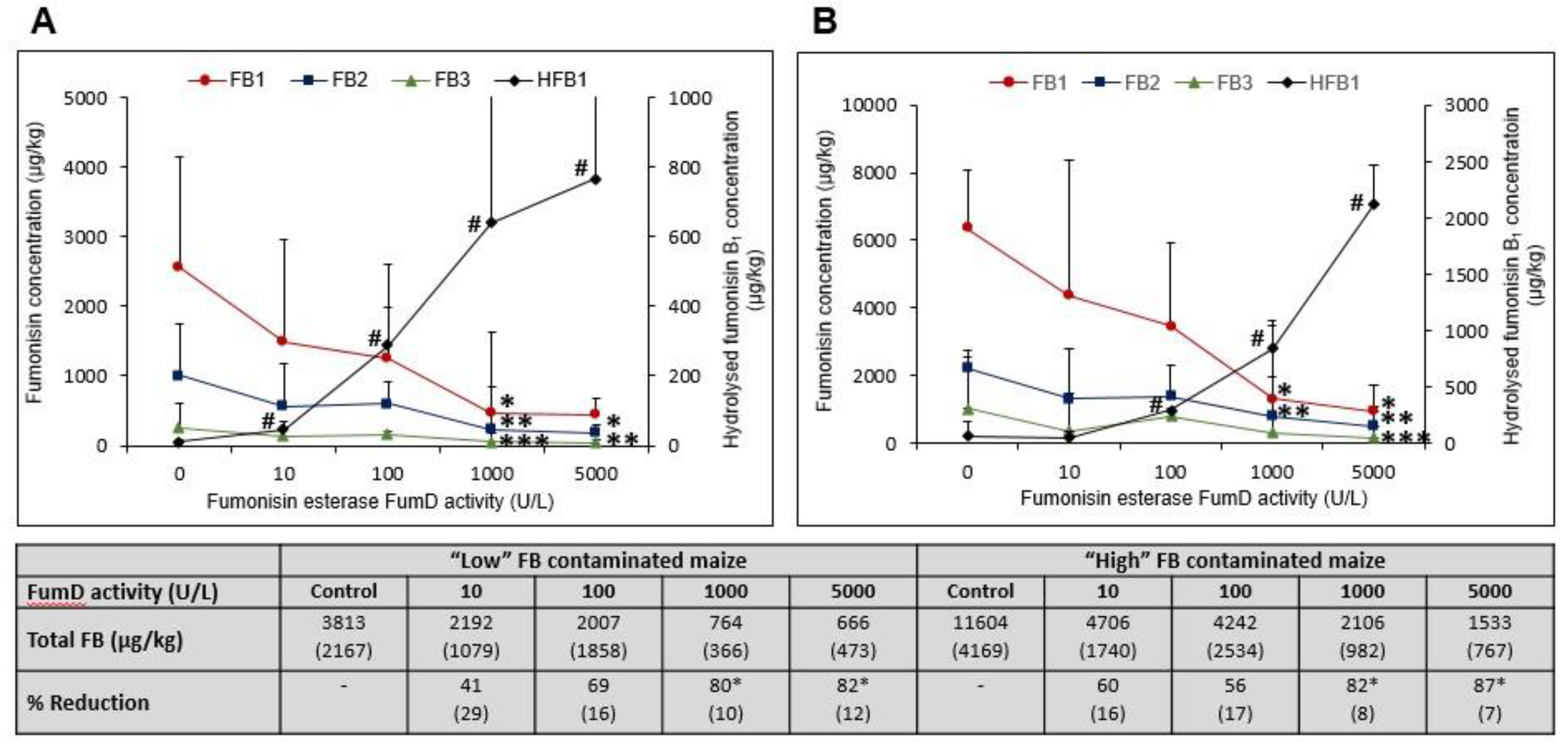
2.2. FB and HFB1 Concentrations in Maize
2.3. Optimal FumD activity and Conversion Ratios (Fixed Time Incubation)
2.3.1. FB1 Hydrolysis and Formation of HFB1 in Residual Maize Kernels as a Function of Fumd Activity
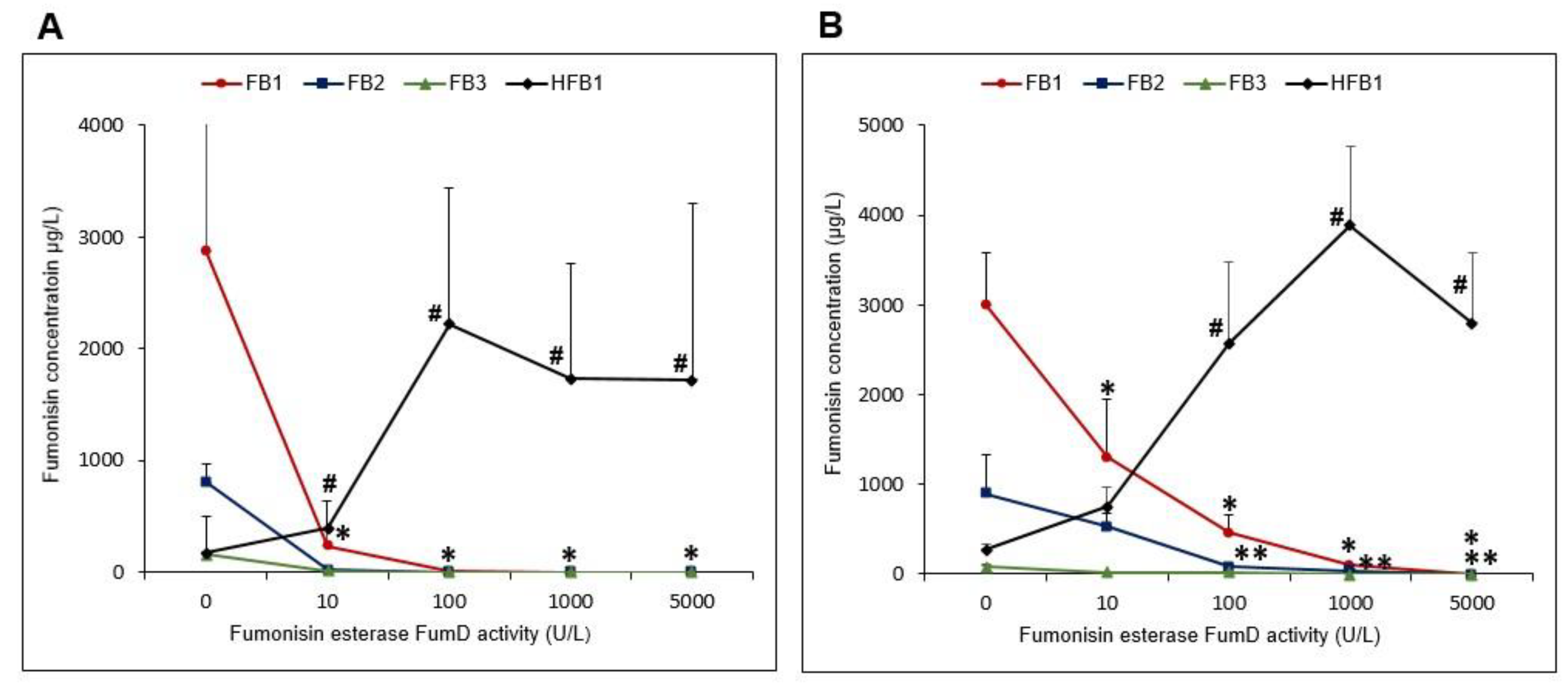
2.3.2. FB1 Hydrolysis and Formation of Hfb1 in the Residual Solutions as a Function of Fumd Activity
2.3.3. FB1 Hydrolysis Relative to the Formation of HFB1
2.3.4. Relative HFB1 Distribution in Residual Maize Kernels and Solutions Following FumD Incubations
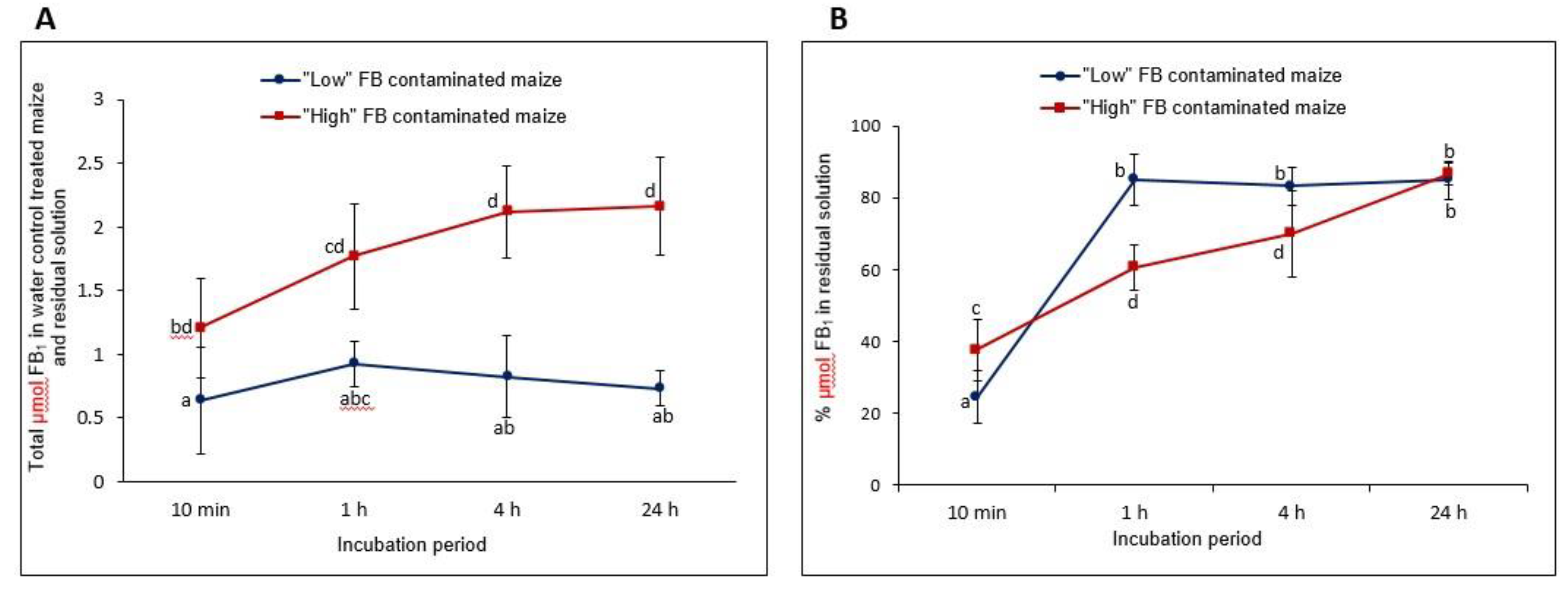
2.4. Comparative Enzyme Kinetics of FB1 Conversion as a Function of Time and FumD Activity
2.4.1. FB1 Leaching into the Aqueous Phase (Water Control Treatment)
2.4.2. Comparative Enzyme Kinetics of FB1 Hydrolysis
2.4.3. Comparative of FB1 Hydrolysis to HFB1 Formation Ratios
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemicals
5.2. Fumonisin Standard Solutions
5.3. Maize Sample Collection
5.4. FumD Enzyme Preparation
5.5. The FumD FB Reduction Method
5.6. FumD Incubation Protocols
5.7. Analyses of FB and HFB1 Concentrations in Maize and Residual Solutions
5.7.1. Extraction Methods
5.7.2. Chromatographic Quantification of FB and HFB1
5.7.3. Validation of the Extraction and FB Quantification Methods
5.8. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alberts, J.F.; van Zyl, W.H.; Gelderblom, W.C.A. Biologically based methods for control of fumonisin-producing Fusarium species and reduction of the fumonisins. Front. Microbiol. 2016, 7, 548. [Google Scholar] [CrossRef] [PubMed]
- Yates, I.E.; Meredith, F.; Smart, W.; Bacon, C.W.; Jaworski, A.J. Trichoderma viride suppresses fumonisin B1 production by Fusarium moniliforme. J. Food Prot. 1999, 62, 1326–1332. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.W.; Yates, I.E.; Hinton, D.M.; Meredith, F. Biological control of Fusarium moniliforme in maize. Environ. Health Perspect. 2001, 109, 325–332. [Google Scholar]
- Duvick, J. Prospects for reducing fumonisin contamination of maize through genetic modification. Environ. Health Perspect. 2001, 109, 337–342. [Google Scholar]
- Duvick, J.; Maddox, J.; Gilliam, J. Composition and methods for fumonisin detoxification. Patent No US6538177, 25 March 2003. [Google Scholar]
- Cleveland, T.E.; Dowd, P.F.; Desjardins, A.E.; Bhatnagar, D.; Cotty, P.J. United States Department of Agriculture-Agricultural Research Service research on pre-harvest prevention of mycotoxins and mycotoxigenic fungi in US crops. Pest. Manag. Sci. 2003, 59, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Cavaglieri, L.; Passone, A.; Etcheverry, M. Screening procedures for selecting rhizobacteria with biocontrol effects upon Fusarium verticillioides growth and fumonisin B1 production. Res. Microbiol. 2004, 155, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Samapundo, S.; De Meulenaer, B.; Osei-Nimoh, D.; Lamboni, Y.; Debevere, J.; Devlieghere, F. Can phenolic compounds be used for the protection of corn from fungal invasion and mycotoxin contamination during storage? Food Microbiol. 2007, 24, 465–473. [Google Scholar] [CrossRef]
- Dalie, D.K.; Deschamps, A.M.; Atanasova-Penichon, V.; Richard-Forget, F. Potential of Pediococcus pentosaceus (L006) isolated from maize leaf to suppress fumonisin-producing fungal growth. J. Food Prot. 2010, 73, 1129–1137. [Google Scholar] [CrossRef]
- Bacon, C.W.; Hinton, D.M. In planta reduction of maize seedling stalk lesions by the bacterial endophyte Bacillus mojavensis. Can. J. Microbiol. 2011, 57, 485–492. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Xue, K.S.; Lin, S.; Marroquin-Cardona, A.; Brown, K.A.; Elmore, S.E.; Tang, L.; Romoser, A.; Gelderblom, W.C.A.; Wang, J.; et al. Calcium montmorillonite clay reduces AFB1 and FB1 biomarkers in rats exposed to single and co-exposures of aflatoxin and fumonisin. J. Appl. Toxicol. 2014, 34, 795–804. [Google Scholar] [CrossRef]
- Alberts, J.F.; Lilly, M.; Rheeder, J.P.; Burger, H.M.; Shephard, G.S.; Gelderblom, W.C.A. Technological and community-based methods to reduce mycotoxin exposure. Food Control. 2017, 73, 101–109. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Cawood, M.E.; Snyman, S.D.; Vleggaar, R.; Marasas, W.F.O. Structure-activity relationships of fumonisins in short-term carcinogenesis and cytotoxicity assays. Food Chem. Toxicol. 1993, 31, 407–414. [Google Scholar] [CrossRef]
- Heinl, S.; Hartinger, D.; Thamhesl, M.; Kunz-Vekiru, E.; Krska, R.; Schatzmayr, G.; Moll, W.; Grabherr, R. Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J. Biotechnol. 2010, 145, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Duvick, J.; Rood, T.; Maddox, J.; Gilliam, J. Detoxification of mycotoxins in planta as a strategy for improving grain quality and disease resistance: Identification of fumonisin-degrading microbes from maize. In Molecular Genetics of Host-Specific Toxins in Plant Disease, Developments in Plant Pathology; Kohmoto, K., Yoder, O.C., Eds.; Springer International Publishing AG: Basel, Switzerland, 1998; Volume 13, pp. 369–381. [Google Scholar]
- Duvick, J.; Rood, T.; Wang, X. Fumonisin detoxification enzymes. Patent No US5716820, 10 February 1998. [Google Scholar]
- Blackwell, B.A.; Gilliam, J.T.; Savard, M.E.; Miller, D.; Duvick, J.P. Oxidative deamination of hydrolysed fumonisin B1 (AP1) by cultures of Exophiala spinifera. Nat. Toxins 1999, 7, 31–38. [Google Scholar] [CrossRef]
- Hartinger, D.; Schwartz, H.; Hametner, C.; Schatzmayr, G.; Haltrich, D.; Moll, W.D. Enzyme characteristics of aminotransferase FumI of Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1. Appl. Microbiol. Biotechnol. 2011, 91, 757–768. [Google Scholar] [CrossRef]
- Moll, D.; Hartinger, D.; Grießler, K.; Binder, E.M.; Schatzmayr, G. Method for the production of an additive for the enzymatic decomposition of mycotoxins, additive, and use thereof. Patent No US8703460B2, 22 April 2014. [Google Scholar]
- Oswald, I.P.; Grenier, B.; Schatzmayr, G.; Moll, W. Enzymatic detoxification of mycotoxins: Hydrolysis of fumonisin B1 strongly reduced the toxicity for piglets. In World Nutrition Forum, Nutri Economics: Balancing Global Nutrition and Productivity; Binder, E.M., Ed.; Anytime Publishing: Leicestershire, UK, 2012; pp. 263–271. [Google Scholar]
- BIOMIN. Available online: https://www.BIOMIN.net/en/home/ (accessed on 12 July 2019).
- Masching, S.; Naehrer, K.; Schwartz-Zimmermann, H.E.; Sãrãndan, M.; Schaumberger, S.; Dohnal, I.; Nagl, V.; Schatzmayr, D. Gastrointestinal degradation of fumonisin B1 by carboxylesterase FumD prevents fumonisin induced alteration of sphingolipid metabolism in turkey and swine. Toxins 2016, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Bracarense, A.F.L.; Schwartz, H.E.; Lucioli, J.; Cossalter, A.; Moll, W.-D.; Schatzmayr, G.; Oswald, I.P. Biotransformation approaches to alleviate the effects induced by Fusarium mycotoxins in Swine. J. Agric. Food Chem. 2013, 61, 6711–6719. [Google Scholar] [CrossRef]
- Grenier, B.; Schwartz, H.E.; Gruber-Dorninger, C.; Dohnal, I.; Aleschko, M.; Schatzmayr, G.; Moll, W.-D.; Applegate, T.J. Enzymatic hydrolysis of fumonisins in the gastrointestinal tract of broiler chickens. Poult Sci. 2017, 96, 4342–4351. [Google Scholar] [CrossRef]
- Mogensen, J.M.; Sørensen, S.M.; Sulyok, M.; van der Westhuizen, L.; Shephard, G.S.; Frisvad, J.C.; Thrane, U.; Krska, R.; Nielsen, K.F. Single-kernel analysis of fumonisins and other fungal metabolites in maize from South African subsistence farmers. Food Addit. Contam. 2011, 28, 1724–1734. [Google Scholar] [CrossRef]
- Janse van Rensburg, J.; Flett, B.C.; Mc Laren, N.W.; Mc Donald, A.H. Sampling variation in the quantification of fumonisins in maize samples. S. Afr. J. Plant. & Soil 2011, 28, 90–96. [Google Scholar]
- Febbraio, F. Biochemical strategies for the detection and detoxification of toxic chemicals in the environment. World J. Biol. Chem. 2017, 8, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Hahn, I.; Nagl, V.; Schwartz, H.E.; Varga, E.; Schwartz, C.; Slavik, V.; Reisinger, N.; Malachová, A.; Cirlini, M.; Generotti, S.; et al. Effects of orally administered fumonisin B1 (FB1), partially hydrolysed FB1, hydrolysed FB1 and N-(1-deoxy-D-fructos-1-yl) FB1 on the sphingolipid metabolism in rats. Food Chem. Toxicol. 2015, 76, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Burger, H.-M.; Shephard, G.S.; Louw, W.; Rheeder, J.P.; Gelderblom, W.C.A. The mycotoxin distribution in maize milling fractions under experimental conditions. Int. J. Food Microbiol. 2013, 165, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Brera, C.; Debegnach, F.; Grossi, S.; Miraglia, M. Effect of industrial processing on the distribution of fumonisin B1 in dry milling corn fractions. J. Food Prot. 2004, 67, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Duncan, K.E.; Howard, R.J. Biology of maize kernel infection by Fusarium verticillioides. Mol. Plant. Microbe Interact. 2010, 23, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Kent, N.L.; Evers, D. Dry Milling. In Kent’s Technology of Cereals. An Introduction for Students of Food Science and Agriculture, 5th ed.; Rosenstrater, K., Evers, A., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 421–514. [Google Scholar]
- Bruggink, H.; Kraak, H.L.; Dijkema, M.H.G.E.; Bekendam, J. Some factors influencing electrolyte leakage from maize (Zea mays L.) kernels. Seed Sci. Res. 1991, 1, 15–20. [Google Scholar] [CrossRef]
- Wu, F.; Munkvold, G.P. Mycotoxins in Ethanol Co-products: Modeling Economic Impacts on the Livestock Industry and Management Strategies. J. Agric. Food Chem. 2008, 56, 3900–3911. [Google Scholar] [CrossRef]
- Ranum, P.; Pena-Rosas, J.P.; Garcia-Casal, M.N. Global maize production, utilization, and consumption. Ann. N. Y. Acad. Sci. 2014, 1312, 105–112. [Google Scholar] [CrossRef]
- Burger, H.-M.; Lombard, M.J.; Shephard, G.S.; Danster-Christians, N.; Gelderblom, W.C.A. Development and evaluation of a sensitive mycotoxin risk assessment model (MYCORAM). Toxicol. Sci. 2014, 141, 387–397. [Google Scholar] [CrossRef]
- Shephard, G.S.; Burger, H.-M.; Rheeder, J.P.; Alberts, J.F.; Gelderblom, W.C.A. The effectiveness of regulatory maximum levels for fumonisin mycotoxins in commercial and subsistence maize crops in South Africa. Food Control. 2019, 97, 77–80. [Google Scholar] [CrossRef]
- FAO. Maximum Levels of Fumonsisin in Maize and Maize Products, Codex Alimentarius Commission, Geneva. Available online: http://www.fao.org/news/story/en/item/238558/icode/ (accessed on 30 May 2019).
- Alberts, J.; Rheeder, J.; Gelderblom, W.; Shephard, G.; Burger, H.-M. Rural subsistence maize farming in South Africa: Risk assessment and intervention models for reduction of exposure to mycotoxins. Toxins 2019, 11, 334. [Google Scholar] [CrossRef]
- Van der Westhuizen, L.; Shephard, G.S.; Burger, H.M.; Rheeder, J.P.; Gelderblom, W.C.; Wild, C.P.; Gong, Y.Y. Fumonisin B1 as a urinary biomarker of exposure in a maize intervention study among South African subsistence farmers. Cancer Epidemiol. Prev. Biomark. 2011, 20, 483–489. [Google Scholar] [CrossRef]
- Van der Westhuizen, L.; Shephard, G.S.; Rheeder, J.P.; Burger, H.-M.; Gelderblom, W.C.A.; Wild, C.P.; Gong, Y.Y. Optimising sorting and washing of home-grown maize to reduce fumonisin contamination under laboratory-controlled conditions. Food Control. 2011, 22, 396–400. [Google Scholar] [CrossRef]
- Cawood, M.E.; Gelderblom, W.C.A.; Vleggaar, R.; Behrend, Y.; Thiel, P.; Marasas, W.F.O. Isolation of the fumonisins: A quantitative approach. J. Agric. Food Chem. 1991, 39, 1958–1962. [Google Scholar] [CrossRef]
- Sewram, V.; Shephard, G.S.; Marasas, W.F.O.; De Castro, M.F.P.M. Improving extraction of fumonisin mycotoxins from Brazilian corn-based infant foods. J. Food Prot. 2003, 66, 854–859. [Google Scholar] [CrossRef]
- United States Department of Health and Human Services, Food and Drug Administration (US FDA). USA Guidance for Industry. Bioanalytical Method Validation. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry (accessed on 30 May 2019).
- NCSS 11 Statistical Software. Available online: http://ncss.com/software/ncss (accessed on 30 May 2019).
| Analyte | Cone Voltage | Precursor | Quantifier (Collision Energy) | Qualifier (Collision Energy) |
|---|---|---|---|---|
| Fumonisin B1 | 50 | 722.3 | 334.3 (40) | 352.3 (38) |
| Fumonisins B2 and B3 | 50 | 706.3 | 318.3 (40) | 336.3 (40) |
| Hydrolysed fumonisin B1 | 25 | 406.6 | 334.3 (25) | 352.4 (20) |
| Analyte | LOQ (µg/kg) | Spike Level (µg/kg) | Recovery (%) | RSDr (%) |
|---|---|---|---|---|
| Fumonisin B1 | 3.5 | 1060 | 84 | 2 |
| Fumonisin B2 | 2.8 | 925 | 66 | 4 |
| Fumonisin B3 | 2.8 | 520 | 79 | 1 |
| Hydrolysed fumonisin B1 | 2.8 | 800 | 80 | 2 |
| FumD Aactivity (U/L) | Total Mean µmol FB1 in Incubation Mixture * | Mean µmol FB1 Converted | Mean µmol FB1 Loss (%) | Total Mean µmol HFB1 in Incubation Mixture | FB1:HFB1 µmol Conversion Ratio |
|---|---|---|---|---|---|
| “Low” FB Contaminated Maize | |||||
| 0 ** | 1.15 ± 0.55 | - | - | 0.02 ± 0.01 | 42.75 ± 12.14 |
| 10 | 0.27 ± 0.14 ab | 0.88 ± 0.14 a | 76.58 ± 11.90 abc | 0.13± 0.05 a | 7.22 ± 2.40 a |
| 100 | 0.18 ± 0.16 ac | 0.98 ± 0.16 a | 84.73 ± 14.14 ab | 0.94 ± 0.62 b | 2.52 ± 0.84 ab |
| 1000 | 0.07 ± 0.03 c | 1.09 ± 0.03 ab | 94.32 ± 2.91 a | 0.99 ± 0.60 b | 1.30 ± 0.58 bc |
| 5000 | 0.06 ± 0.05 c | 1.09 ± 0.05 ab | 94.64 ± 3.98 ab | 1.02 ± 1.00 b | 0.66 ± 0.32 c |
| “High” FB Contaminated Maize | |||||
| 0 ** | 1.74 ± 1.32 | - | - | 0.16 ± 0.10 | 10.23 ± 3.96 |
| 10 | 0.97 ± 0.50 b | 0.76 ± 0.50 a | 55.04 ± 17.68 c | 0.24 ± 0.15 a | 3.4 ± 1.73 abd |
| 100 | 0.71 ± 0.37 b | 1.02 ± 0.37 a | 58.84 ± 21.59 c | 1.17 ± 0.53 b | 1.38 ± 0.72 bc |
| 1000 | 0.21 ± 0.16 ac | 1.52 ± 0.16 b | 87.75 ± 9.31 ab | 1.96 ± 0.89 b | 0.93 ± 0.46 c |
| 5000 | 0.13 ± 0.07 ac | 1.61 ± 0.07 b | 92.61 ± 4.09 ab | 1.68 ± 0.66 b | 1.11 ± 0.54 cd |
| FumD Activity (U/L) | Total Mean HFB1 (µmol) in 100 g Residual Maize Kernels | Total Mean HFB1 (µmol) in 200 mL Residual Solution | Mean µmol HFB1 in Residual Maize Kernels (%) | Mean µmol HFB1 in Residual Solution (%) |
|---|---|---|---|---|
| “Low” FB Contaminated Maize | ||||
| 10 | 0.01 ± 0.01 a | 0.13 ± 0.04 a | 5.65 ± 3.47 a | 94.35 ± 3.47 a |
| 100 | 0.07 ± 0.06 b | 0.87 ± 0.58 b | 7.35 ± 3.28 a | 92.65 ± 3.28 a |
| 1000 | 0.15 ± 0.11 b | 0.83 ± 0.50 b | 15.82 ± 4.48 b | 84.18 ± 4.48 b |
| 5000 | 0.19 ± 0.22 b | 0.83 ± 0.78 b | 16.25 ± 3.86 b | 83.75 ± 3.86 b |
| “High” FB Contaminated Maize | ||||
| 10 | 0.00 a | 0.24 ± 0.14 a | 0.48 ± 0.58 a | 99.52 ± 0.60 a |
| 100 | 0.08 ± 0.06 b | 1.09 ± 0.48 b | 6.75 ± 2.24 b | 93.25 ± 2.24 b |
| 1000 | 0.19 ± 0.09 bc | 1.77 ± 0.86 b | 10.68 ± 4.33 b | 89.32 ± 4.33 b |
| 5000 | 0.50 ± 0.17 c | 1.17 ± 0.65 b | 32.90 ± 11.85 c | 67.10 ± 11.90 c |
| “Low” FB Contaminated Maize | “High” FB Contaminated Maize | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment Duration | 10 min | 1 h | 4 h | 24 h | 10 min | 1 h | 4 h | 24 h | 10 min | 1 h | 4 h | 24 h | 10 min | 1 h | 4 h | 24 h |
| FumD Activity | FB1Hydrolysis (nmol/min/mg Enzyme) | HFB1Formation * (nmol/min/mg Enzyme) | FB1Hydrolysis (nmol/min/mg Enzyme) | HFB1Formation * (nmol/min/mg Enzyme) | ||||||||||||
| 100 U/L | 38.34 (1.54) a | 8.23 (0.96) b | 1.89 (0.04) c | 0.32 (0.01) d | 20.26 (10.75) A | 5.15 (0.80) B | 2.12 (0.56) C | 0.32 (0.12) D | 59.19 (12.08) i | 13.18 (3.24) j | 4.42 (1.03) e | 0.92 (0.04) f | 21.10 (8.56) A | 14.55 (1.46) A | 4.86 (0.82) B | 1.18 (0.16) C |
| 1000 U/L | 3.06 (0.62)e | 0.89 (0.11) f | 0.20 (0.01) g | 0.03 (0.00) h | 2.78 (1.55) C | 0.87 (0.16) E | 0.25 (0.12) D | 0.04 (0.01) F | 4.53 (2.26) e | 1.70 (0.19) k | 0.52 (0.05) l | 0.10 (0.00) m | 3.64 (2.02) B | 1.98 (0.46) C | 0.67 (0.21) E | 0.11 (0.02) D |
| FB1Hydrolysis: HFB1Formation Ratio | FB1Hydrolysis: HFB1Formation Ratio | |||||||||||||||
| 100 U/L | 3.46 (1.93) p | 1.39 (0.62) pq | 0.95 (0.30) qr | 0.93 (0.36) pr | - | - | - | - | 1.94 (0.16) pr | 0.91 (0.20) pq | 0.91 (0.14) qr | 0.95 (0.33) qr | - | - | - | - |
| 1000 U/L | 1.45 (0.94) pqs | 1.04 (0.16) pq | 0.93 (0.38) qr | 0.92 (0.35) qr | - | - | - | - | 1.10 (0.29) pqs | 0.91 (0.26) qrs | 0.84 (0.26) qrs | 0.92 (0.24) qrs | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alberts, J.; Schatzmayr, G.; Moll, W.-D.; Davids, I.; Rheeder, J.; Burger, H.-M.; Shephard, G.; Gelderblom, W. Detoxification of the Fumonisin Mycotoxins in Maize: An Enzymatic Approach. Toxins 2019, 11, 523. https://doi.org/10.3390/toxins11090523
Alberts J, Schatzmayr G, Moll W-D, Davids I, Rheeder J, Burger H-M, Shephard G, Gelderblom W. Detoxification of the Fumonisin Mycotoxins in Maize: An Enzymatic Approach. Toxins. 2019; 11(9):523. https://doi.org/10.3390/toxins11090523
Chicago/Turabian StyleAlberts, Johanna, Gerd Schatzmayr, Wulf-Dieter Moll, Ibtisaam Davids, John Rheeder, Hester-Mari Burger, Gordon Shephard, and Wentzel Gelderblom. 2019. "Detoxification of the Fumonisin Mycotoxins in Maize: An Enzymatic Approach" Toxins 11, no. 9: 523. https://doi.org/10.3390/toxins11090523
APA StyleAlberts, J., Schatzmayr, G., Moll, W.-D., Davids, I., Rheeder, J., Burger, H.-M., Shephard, G., & Gelderblom, W. (2019). Detoxification of the Fumonisin Mycotoxins in Maize: An Enzymatic Approach. Toxins, 11(9), 523. https://doi.org/10.3390/toxins11090523





