Could Mycolactone Inspire New Potent Analgesics? Perspectives and Pitfalls
Abstract
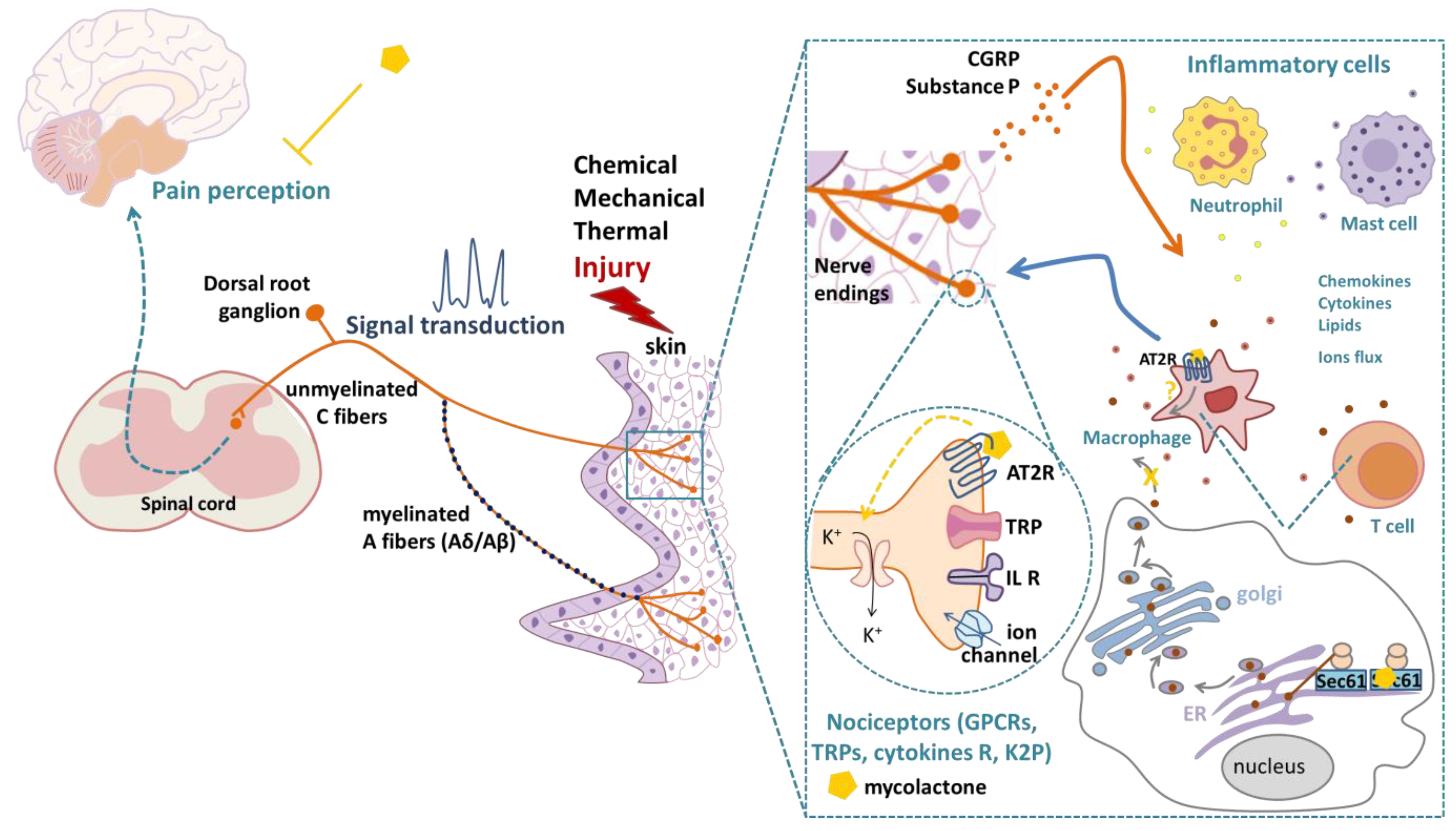
:1. Introduction
2. Mycolactone-Induced Analgesia: Inflammatory Versus Non-Inflammatory Contexts
3. Detection and Synthesis of Mycolactone: Key Starting Points for the Development of Mycolactone-Inspired Potent Analgesics
4. Perspectives for the Use of Mycolactone as an Analgesic
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Phillips, C.J. The Cost and Burden of Chronic Pain. Rev. Pain 2009, 3, 2–5. [Google Scholar] [CrossRef] [Green Version]
- Breivik, H.; Collett, B.; Ventafridda, V.; Cohen, R.; Gallacher, D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur. J. Pain 2006, 10, 287–333. [Google Scholar] [CrossRef]
- American Academy of Pain Medicine. Management of chronic pain syndromes: Issues and interventions. Pain Med. 2005, 6, S1–S21. [Google Scholar] [CrossRef]
- Nicholson, B.; Ross, E.; Weil, A.; Sasaki, J.; Sacks, G. Treatment of chronic moderate-to-severe non-malignant pain with polymer-coated extended-release morphine sulfate capsules. Curr. Med. Res. Opin. 2006, 22, 539–550. [Google Scholar] [CrossRef]
- Deandrea, S.; Montanari, M.; Moja, L.; Apolone, G. Prevalence of undertreatment in cancer pain. A review of published literature. Ann. Oncol. 2008, 19, 1985–1991. [Google Scholar] [CrossRef]
- Geurts, J.W.; Willems, P.C.; Lockwood, C.; van Kleef, M.; Kleijnen, J.; Dirksen, C. Patient expectations for management of chronic non-cancer pain: A systematic review. Health Expect. 2017, 20, 1201–1217. [Google Scholar] [CrossRef]
- Melnikova, I. Pain market. Nat. Rev. Drug Discov. 2010, 9, 589–590. [Google Scholar] [CrossRef]
- Borsook, D.; Hargreaves, R.; Bountra, C.; Porreca, F. Lost but making progress--Where will new analgesic drugs come from? Sci. Transl. Med. 2014, 6, 249sr243. [Google Scholar] [CrossRef]
- Stern, P.; Roberts, L. The future of pain research. Science 2016, 354, 564–565. [Google Scholar] [CrossRef] [Green Version]
- Skolnick, P. The Opioid Epidemic: Crisis and Solutions. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 143–159. [Google Scholar] [CrossRef] [Green Version]
- Basbaum, A.I.; Jessell, T. The Perception of Pain. In Neural Sciences; Kandel, E.R., Schwartz, J., Jessell, T., Eds.; Appleton and Lange: New York, NY, USA, 2000; pp. 472–491. [Google Scholar]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef]
- Woolf, C.J.; Ma, Q. Nociceptors--noxious stimulus detectors. Neuron 2007, 55, 353–364. [Google Scholar] [CrossRef]
- Dubin, A.E.; Patapoutian, A. Nociceptors: The sensors of the pain pathway. J. Clin. Investig. 2010, 120, 3760–3772. [Google Scholar] [CrossRef]
- Ji, R.R.; Chamessian, A.; Zhang, Y.Q. Pain regulation by non-neuronal cells and inflammation. Science 2016, 354, 572–577. [Google Scholar] [CrossRef] [Green Version]
- Abbadie, C.; Bhangoo, S.; De Koninck, Y.; Malcangio, M.; Melik-Parsadaniantz, S.; White, F.A. Chemokines and pain mechanisms. Brain Res. 2009, 60, 125–134. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, S.H.; Lorenzetti, B.B.; Bristow, A.F.; Poole, S. Interleukin-1 beta as a potent hyperalgesic agent antagonized by a tripeptide analogue. Nature 1988, 334, 698–700. [Google Scholar] [CrossRef]
- Woolf, C.J.; Allchorne, A.; Safieh-Garabedian, B.; Poole, S. Cytokines, nerve growth factor and inflammatory hyperalgesia: The contribution of tumour necrosis factor alpha. Br. J. Pharmacol. 1997, 121, 417–424. [Google Scholar] [CrossRef]
- McMahon, S.B.; La Russa, F.; Bennett, D.L. Crosstalk between the nociceptive and immune systems in host defence and disease. Nat. Rev. Neurosci. 2015, 16, 389–402. [Google Scholar] [CrossRef]
- FitzGerald, G.A. Correction: Cox-2 and beyond: Approaches to prostagland in inhibition in human disease. Nat. Rev. Drug Discov. 2003, 2, 1030. [Google Scholar] [CrossRef]
- Baral, P.; Udit, S.; Chiu, I.M. Pain and immunity: Implications for host defence. Nat. Rev. Immunol. 2019. [Google Scholar] [CrossRef]
- Pinho-Ribeiro, F.A.; Verri, W.A., Jr.; Chiu, I.M. Nociceptor Sensory Neuron-Immune Interactions in Pain and Inflammation. Trends Immunol. 2017, 38, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Julius, D.; Nathans, J. Signaling by sensory receptors. Cold Spring Harb. Perspect. Biol. 2012, 4, a005991. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Khan, S.; Kim, Y.S. Insight into Pain Modulation: Nociceptors Sensitization and Therapeutic Targets. Curr. Drug Targets 2019, 20, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Costigan, M.; Woolf, C.J. Pain: Molecular mechanisms. J. Pain 2000, 1, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Gohlke, B.O.; Preissner, R.; Preissner, S. SuperPain--a resource on pain-relieving compounds targeting ion channels. Nucleic Acids Res. 2014, 42, D1107–D1112. [Google Scholar] [CrossRef] [PubMed]
- Rouwette, T.; Avenali, L.; Sondermann, J.; Narayanan, P.; Gomez-Varela, D.; Schmidt, M. Modulation of nociceptive ion channels and receptors via protein-protein interactions: Implications for pain relief. Channels 2015, 9, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Benham, C.D.; Davis, J.B.; Randall, A.D. Vanilloid and TRP channels: A family of lipid-gated cation channels. Neuropharmacology 2002, 42, 873–888. [Google Scholar] [CrossRef]
- Blair, H.A. Capsaicin 8% Dermal Patch: A Review in Peripheral Neuropathic Pain. Drugs 2018, 78, 1489–1500. [Google Scholar] [CrossRef]
- Nozadze, I.; Tsiklauri, N.; Gurtskaia, G.; Tsagareli, M.G. NSAIDs attenuate hyperalgesia induced by TRP channel activation. Data Brief 2016, 6, 668–673. [Google Scholar] [CrossRef] [Green Version]
- Machelska, H.; Celik, M.O. Advances in Achieving Opioid Analgesia Without Side Effects. Front. Pharmacol. 2018, 9, 1388. [Google Scholar] [CrossRef]
- Anand, U.; Facer, P.; Yiangou, Y.; Sinisi, M.; Fox, M.; McCarthy, T.; Bountra, C.; Korchev, Y.E.; Anand, P. Angiotensin II type 2 receptor (AT2 R) localization and antagonist-mediated inhibition of capsaicin responses and neurite outgrowth in human and rat sensory neurons. Eur. J. Pain 2013, 17, 1012–1026. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.S.C.; Dworkin, R.H.; McCarthy, T.D.; Anand, P.; Bountra, C.; McCloud, P.I.; Hill, J.; Cutter, G.; Kitson, G.; Desem, N.; et al. EMA401, an orally administered highly selective angiotensin II type 2 receptor antagonist, as a novel treatment for postherpetic neuralgia: A randomised, double-blind, placebo-controlled phase 2 clinical trial. Lancet 2014, 383, 1637–1647. [Google Scholar] [CrossRef]
- Smith, M.T.; Muralidharan, A. Targeting angiotensin II type 2 receptor pathways to treat neuropathic pain and inflammatory pain. Expert Opin. Ther. Targets 2015, 19, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Yekkirala, A.S.; Roberson, D.P.; Bean, B.P.; Woolf, C.J. Breaking barriers to novel analgesic drug development. Nat. Rev. Drug Discov. 2017, 16, 810. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.N. Results in analgesia--Darwin 1, pharma 0. N. Engl. J. Med. 2013, 369, 2558–2560. [Google Scholar] [CrossRef] [PubMed]
- Kou, J.; Ni, Y.; Li, N.; Wang, J.; Liu, L.; Jiang, Z.H. Analgesic and anti-inflammatory activities of total extract and individual fractions of Chinese medicinal ants Polyrhachis lamellidens. Biol. Pharm. Bull. 2005, 28, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Maatuf, Y.; Geron, M.; Priel, A. The Role of Toxins in the Pursuit for Novel Analgesics. Toxins 2019, 11, 131. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zheng, J. Understand spiciness: Mechanism of TRPV1 channel activation by capsaicin. Protein Cell 2017, 8, 169–177. [Google Scholar] [CrossRef]
- Poewe, W.; Burbaud, P.; Castelnovo, G.; Jost, W.H.; Ceballos-Baumann, A.O.; Banach, M.; Potulska-Chromik, A.; Ferreira, J.J.; Bihari, K.; Ehler, E.; et al. Efficacy and safety of abobotulinumtoxinA liquid formulation in cervical dystonia: A randomized-controlled trial. Mov. Disord. 2016, 31, 1649–1657. [Google Scholar] [CrossRef]
- Tassorelli, C.; Aguggia, M.; De Tommaso, M.; Geppetti, P.; Grazzi, L.; Pini, L.A.; Sarchielli, P.; Tedeschi, G.; Martelletti, P.; Cortelli, P. Onabotulinumtoxin A for the management of chronic migraine in current clinical practice: Results of a survey of sixty-three Italian headache centers. J. Headache Pain 2017, 18, 66. [Google Scholar] [CrossRef]
- Lewis, R.J. Conotoxins as selective inhibitors of neuronal ion channels, receptors and transporters. IUBMB Life 2004, 56, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Hama, A.; Sagen, J. Antinociceptive effects of the marine snail peptides conantokin-G and conotoxin MVIIA alone and in combination in rat models of pain. Neuropharmacology 2009, 56, 556–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malmberg, A.B.; Gilbert, H.; McCabe, R.T.; Basbaum, A.I. Powerful antinociceptive effects of the cone snail venom-derived subtype-selective NMDA receptor antagonists conantokins G and T. Pain 2003, 101, 109–116. [Google Scholar] [CrossRef]
- Yotsu, R.R.; Richardson, M.; Ishii, N. Drugs for treating Buruli ulcer (Mycobacterium ulcerans disease). Cochrane Database Syst. Rev. 2018, 8, CD012118. [Google Scholar] [CrossRef] [PubMed]
- George, K.M.; Chatterjee, D.; Gunawardana, G.; Welty, D.; Hayman, J.; Lee, R.; Small, P.L. Mycolactone: A polyketide toxin from Mycobacterium ulcerans required for virulence. Science 1999, 283, 854–857. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Schreiner, A.J.; Biernaskie, J.; Nickerson, D.; Gabriel, V.A. Treating pain on skin graft donor sites: Review and clinical recommendations. J. Trauma Acute Care Surg. 2017, 83, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Vincent, Q.B.; Ardant, M.F.; Adeye, A.; Goundote, A.; Saint-Andre, J.P.; Cottin, J.; Kempf, M.; Agossadou, D.; Johnson, C.; Abel, L.; et al. Clinical epidemiology of laboratory-confirmed Buruli ulcer in Benin: A cohort study. Lancet Glob. Health 2014, 2, e422–e430. [Google Scholar] [CrossRef]
- Johnson, P.D.; Stinear, T.; Small, P.L.; Pluschke, G.; Merritt, R.W.; Portaels, F.; Huygen, K.; Hayman, J.A.; Asiedu, K. Buruli ulcer (M. ulcerans infection): New insights, new hope for disease control. PLoS Med. 2005, 2, e108. [Google Scholar] [CrossRef] [PubMed]
- Sizaire, V.; Nackers, F.; Comte, E.; Portaels, F. Mycobacterium ulcerans infection: Control, diagnosis, and treatment. Lancet Infect. Dis. 2006, 6, 288–296. [Google Scholar] [CrossRef]
- En, J.; Goto, M.; Nakanaga, K.; Higashi, M.; Ishii, N.; Saito, H.; Yonezawa, S.; Hamada, H.; Small, P.L. Mycolactone is responsible for the painlessness of Mycobacterium ulcerans infection (Buruli ulcer) in a murine study. Infect. Immun. 2008, 76, 2002–2007. [Google Scholar] [CrossRef] [PubMed]
- Marion, E.; Song, O.R.; Christophe, T.; Babonneau, J.; Fenistein, D.; Eyer, J.; Letournel, F.; Henrion, D.; Clere, N.; Paille, V.; et al. Mycobacterial toxin induces analgesia in buruli ulcer by targeting the angiotensin pathways. Cell 2014, 157, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Babonneau, J.; Breard, D.; Reynaert, M.L.; Marion, E.; Guilet, D.; Saint Andre, J.P.; Croue, A.; Brodin, P.; Richomme, P.; Marsollier, L. Mycolactone as Analgesic: Subcutaneous Bioavailability Parameters. Front. Pharmacol. 2019, 10, 378. [Google Scholar] [CrossRef]
- Hall, B.; Simmonds, R. Pleiotropic molecular effects of the Mycobacterium ulcerans virulence factor mycolactone underlying the cell death and immunosuppression seen in Buruli ulcer. Biochem. Soc. Trans. 2014, 42, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Sarfo, F.S.; Phillips, R.; Wansbrough-Jones, M.; Simmonds, R.E. Recent advances: Role of mycolactone in the pathogenesis and monitoring of Mycobacterium ulcerans infection/Buruli ulcer disease. Cell. Microbiol. 2016, 18, 17–29. [Google Scholar] [CrossRef]
- Ogbechi, J.; Hall, B.S.; Sbarrato, T.; Taunton, J.; Willis, A.E.; Wek, R.C.; Simmonds, R.E. Inhibition of Sec61-dependent translocation by mycolactone uncouples the integrated stress response from ER stress, driving cytotoxicity via translational activation of ATF4. Cell Death Dis. 2018, 9, 397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asiedu, K.; Scherpbier, R.; Raviglione, M. Buruli ulcer: Mycobacterium Ulcerans Infection. Available online: https://apps.who.int/iris/bitstream/handle/10665/66164/WHO_CDS_CPE_GBUI_2000.1.pdf (accessed on 20 April 2019).
- van der Werf, T.S.; van der Graaf, W.T.; Tappero, J.W.; Asiedu, K. Mycobacterium ulcerans infection. Lancet 1999, 354, 1013–1018. [Google Scholar] [CrossRef]
- Demangel, C.; High, S. Sec61 blockade by mycolactone: A central mechanism in Buruli ulcer disease. Biol. Cell 2018, 110, 237–248. [Google Scholar] [CrossRef] [Green Version]
- Guenin-Mace, L.; Veyron-Churlet, R.; Thoulouze, M.I.; Romet-Lemonne, G.; Hong, H.; Leadlay, P.F.; Danckaert, A.; Ruf, M.T.; Mostowy, S.; Zurzolo, C.; et al. Mycolactone activation of Wiskott-Aldrich syndrome proteins underpins Buruli ulcer formation. J. Clin. Investig. 2013, 123, 1501–1512. [Google Scholar] [CrossRef] [Green Version]
- Baron, L.; Paatero, A.O.; Morel, J.D.; Impens, F.; Guenin-Mace, L.; Saint-Auret, S.; Blanchard, N.; Dillmann, R.; Niang, F.; Pellegrini, S.; et al. Mycolactone subverts immunity by selectively blocking the Sec61 translocon. J. Exp. Med. 2016, 213, 2885–2896. [Google Scholar] [CrossRef]
- Guenin-Mace, L.; Baron, L.; Chany, A.C.; Tresse, C.; Saint-Auret, S.; Jonsson, F.; Le Chevalier, F.; Bruhns, P.; Bismuth, G.; Hidalgo-Lucas, S.; et al. Shaping mycolactone for therapeutic use against inflammatory disorders. Sci. Transl. Med. 2015, 7, 289ra285. [Google Scholar] [CrossRef] [PubMed]
- Isaac, C.; Mauborgne, A.; Grimaldi, A.; Ade, K.; Pohl, M.; Limatola, C.; Boucher, Y.; Demangel, C.; Guenin-Mace, L. Mycolactone displays anti-inflammatory effects on the nervous system. PLoS Negl. Trop. Dis. 2017, 11, e0006058. [Google Scholar] [CrossRef] [PubMed]
- Bieri, R.; Scherr, N.; Ruf, M.T.; Dangy, J.P.; Gersbach, P.; Gehringer, M.; Altmann, K.H.; Pluschke, G. The Macrolide Toxin Mycolactone Promotes Bim-Dependent Apoptosis in Buruli Ulcer through Inhibition of mTOR. ACS Chem. Biol. 2017, 12, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Kwofie, S.K.; Dankwa, B.; Enninful, K.S.; Adobor, C.; Broni, E.; Ntiamoah, A.; Wilson, M.D. Molecular Docking and Dynamics Simulation Studies Predict Munc18b as a Target of Mycolactone: A Plausible Mechanism for Granule Exocytosis Impairment in Buruli Ulcer Pathogenesis. Toxins 2019, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Goto, M.; Nakanaga, K.; Aung, T.; Hamada, T.; Yamada, N.; Nomoto, M.; Kitajima, S.; Ishii, N.; Yonezawa, S.; Saito, H. Nerve damage in Mycobacterium ulcerans-infected mice: Probable cause of painlessness in buruli ulcer. Am. J. Pathol. 2006, 168, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Ocana, M.; Cendan, C.M.; Cobos, E.J.; Entrena, J.M.; Baeyens, J.M. Potassium channels and pain: Present realities and future opportunities. Eur. J. Pharmacol. 2004, 500, 203–219. [Google Scholar] [CrossRef]
- Hargreaves, K.; Dubner, R.; Brown, F.; Flores, C.; Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988, 32, 77–88. [Google Scholar] [CrossRef]
- Song, O.R.; Kim, H.B.; Jouny, S.; Ricard, I.; Vandeputte, A.; Deboosere, N.; Marion, E.; Queval, C.J.; Lesport, P.; Bourinet, E.; et al. A Bacterial Toxin with Analgesic Properties: Hyperpolarization of DRG Neurons by Mycolactone. Toxins 2017, 9, 227. [Google Scholar] [CrossRef] [PubMed]
- MacIver, M.B.; Amagasu, S.M.; Mikulec, A.A.; Monroe, F.A. Riluzole anesthesia: Use-dependent block of presynaptic glutamate fibers. Anesthesiology 1996, 85, 626–634. [Google Scholar] [CrossRef]
- Duprat, F.; Lesage, F.; Patel, A.J.; Fink, M.; Romey, G.; Lazdunski, M. The neuroprotective agent riluzole activates the two P domain K(+) channels TREK-1 and TRAAK. Mol. Pharmacol. 2000, 57, 906–912. [Google Scholar]
- Noel, J.; Zimmermann, K.; Busserolles, J.; Deval, E.; Alloui, A.; Diochot, S.; Guy, N.; Borsotto, M.; Reeh, P.; Eschalier, A.; et al. The mechano-activated K+ channels TRAAK and TREK-1 control both warm and cold perception. EMBO J. 2009, 28, 1308–1318. [Google Scholar] [CrossRef] [Green Version]
- Mathie, A.; Veale, E.L. Two-pore domain potassium channels: Potential therapeutic targets for the treatment of pain. Pflügers Arch. 2015, 467, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Fornasari, D. Pain pharmacology: Focus on opioids. Clin. Cases Miner. Bone Metab. 2014, 11, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Szelenyi, I. Flupirtine, a re-discovered drug, revisited. Inflamm. Res. 2013, 62, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Han, G.W.; Batyuk, A.; Ishchenko, A.; White, K.L.; Patel, N.; Sadybekov, A.; Zamlynny, B.; Rudd, M.T.; Hollenstein, K.; et al. Structural basis for selectivity and diversity in angiotensin II receptors. Nature 2017, 544, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Danser, A.H.; Anand, P. The angiotensin II type 2 receptor for pain control. Cell 2014, 157, 1504–1506. [Google Scholar] [CrossRef] [PubMed]
- Anand, U.; Yiangou, Y.; Sinisi, M.; Fox, M.; MacQuillan, A.; Quick, T.; Korchev, Y.E.; Bountra, C.; McCarthy, T.; Anand, P. Mechanisms underlying clinical efficacy of Angiotensin II type 2 receptor (AT2R) antagonist EMA401 in neuropathic pain: Clinical tissue and in vitro studies. Mol. Pain 2015, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.T.; Anand, P.; Rice, A.S. Selective small molecule angiotensin II type 2 receptor antagonists for neuropathic pain: Preclinical and clinical studies. Pain 2016, 157 (Suppl. 1), S33–S41. [Google Scholar] [CrossRef]
- Anand, U.; Sinisi, M.; Fox, M.; MacQuillan, A.; Quick, T.; Korchev, Y.; Bountra, C.; McCarthy, T.; Anand, P. Mycolactone-mediated neurite degeneration and functional effects in cultured human and rat DRG neurons: Mechanisms underlying hypoalgesia in Buruli ulcer. Mol. Pain 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Wallinder, C.; Plouffe, B.; Beaudry, H.; Mahalingam, A.K.; Wu, X.; Johansson, B.; Holm, M.; Botoros, M.; Karlen, A.; et al. Design, synthesis, and biological evaluation of the first selective nonpeptide AT2 receptor agonist. J. Med. Chem. 2004, 47, 5995–6008. [Google Scholar] [CrossRef] [PubMed]
- Frias, B.; Merighi, A. Capsaicin, Nociception and Pain. Molecules 2016, 21, 797. [Google Scholar] [CrossRef] [PubMed]
- Takayama, Y.; Uta, D.; Furue, H.; Tominaga, M. Pain-enhancing mechanism through interaction between TRPV1 and anoctamin 1 in sensory neurons. Proc. Natl. Acad. Sci. USA 2015, 112, 5213–5218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, P.; Bley, K. Topical capsaicin for pain management: Therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br. J. Anaesth. 2011, 107, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Benitez, S.; Seltzer, A.; Acosta, C. Nociceptor-like rat dorsal root ganglion neurons express the angiotensin-II AT2 receptor throughout development. Int. J. Dev. Neurosci. 2017, 56, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, A.J.; Copits, B.A.; Mickle, A.D.; Karlsson, P.; Kadunganattil, S.; Haroutounian, S.; Tadinada, S.M.; de Kloet, A.D.; Valtcheva, M.V.; McIlvried, L.A.; et al. Angiotensin II Triggers Peripheral Macrophage-to-Sensory Neuron Redox Crosstalk to Elicit Pain. J. Neurosci. 2018, 38, 7032–7057. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coutanceau, E.; Marsollier, L.; Brosch, R.; Perret, E.; Goossens, P.; Tanguy, M.; Cole, S.T.; Small, P.L.; Demangel, C. Modulation of the host immune response by a transient intracellular stage of Mycobacterium ulcerans: The contribution of endogenous mycolactone toxin. Cell. Microbiol. 2005, 7, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Hall, B.S.; Hill, K.; McKenna, M.; Ogbechi, J.; High, S.; Willis, A.E.; Simmonds, R.E. The pathogenic mechanism of the Mycobacterium ulcerans virulence factor, mycolactone, depends on blockade of protein translocation into the ER. PLoS Pathog. 2014, 10, e1004061. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.; Simmonds, R.E.; High, S. Mechanistic insights into the inhibition of Sec61-dependent co- and post-translational translocation by mycolactone. J. Cell Sci. 2016, 129, 1404–1415. [Google Scholar] [CrossRef] [Green Version]
- Ogbechi, J.; Ruf, M.T.; Hall, B.S.; Bodman-Smith, K.; Vogel, M.; Wu, H.L.; Stainer, A.; Esmon, C.T.; Ahnstrom, J.; Pluschke, G.; et al. Mycolactone-Dependent Depletion of Endothelial Cell Thrombomodulin Is Strongly Associated with Fibrin Deposition in Buruli Ulcer Lesions. PLoS Pathog. 2015, 11, e1005011. [Google Scholar] [CrossRef]
- Morel, J.D.; Paatero, A.O.; Wei, J.; Yewdell, J.W.; Guenin-Mace, L.; Van Haver, D.; Impens, F.; Pietrosemoli, N.; Paavilainen, V.O.; Demangel, C. Proteomics Reveals Scope of Mycolactone-mediated Sec61 Blockade and Distinctive Stress Signature. Mol. Cell Proteom. 2018, 17, 1750–1765. [Google Scholar] [CrossRef] [Green Version]
- Connor, D.H.; Lunn, H.F. Mycobacterium ulcerans infection (with comments on pathogenesis). Int. J. Infect. Dis. 1965, 33, 698–709. [Google Scholar]
- George, K.M.; Barker, L.P.; Welty, D.M.; Small, P.L. Partial purification and characterization of biological effects of a lipid toxin produced by Mycobacterium ulcerans. Infect. Immun. 1998, 66, 587–593. [Google Scholar] [PubMed]
- Hong, H.; Gates, P.J.; Staunton, J.; Stinear, T.; Cole, S.T.; Leadlay, P.F.; Spencer, J.B. Identification using LC-MSn of co-metabolites in the biosynthesis of the polyketide toxin mycolactone by a clinical isolate of Mycobacterium ulcerans. Chem. Commun. 2003, 2822–2823. [Google Scholar] [CrossRef] [PubMed]
- Marion, E.; Prado, S.; Cano, C.; Babonneau, J.; Ghamrawi, S.; Marsollier, L. Photodegradation of the Mycobacterium ulcerans toxin, mycolactones: Considerations for handling and storage. PLoS ONE 2012, 7, e33600. [Google Scholar] [CrossRef] [PubMed]
- Sarfo, F.S.; Le Chevalier, F.; Aka, N.; Phillips, R.O.; Amoako, Y.; Boneca, I.G.; Lenormand, P.; Dosso, M.; Wansbrough-Jones, M.; Veyron-Churlet, R.; et al. Mycolactone diffuses into the peripheral blood of Buruli ulcer patients--implications for diagnosis and disease monitoring. PLoS Negl. Trop. Dis. 2011, 5, e1237. [Google Scholar] [CrossRef] [PubMed]
- Sarfo, F.S.; Phillips, R.O.; Zhang, J.; Abass, M.K.; Abotsi, J.; Amoako, Y.A.; Adu-Sarkodie, Y.; Robinson, C.; Wansbrough-Jones, M.H. Kinetics of mycolactone in human subcutaneous tissue during antibiotic therapy for Mycobacterium ulcerans disease. BMC Infect. Dis. 2014, 14, 202. [Google Scholar] [CrossRef] [PubMed]
- Gunawardana, G.; Chatterjee, D.; George, K.M.; Brennan, P.; Whittern, D.; Small, P.L.C. Characterization of Novel Macrolide Toxins, Mycolactones A and B, from a Human Pathogen, Mycobacterium ulcerans. J. Am. Chem. Soc. 1999, 121, 6092–6093. [Google Scholar] [CrossRef]
- Fattori, V.; Zarpelon, A.C.; Staurengo-Ferrari, L.; Borghi, S.M.; Zaninelli, T.H.; Da Costa, F.B.; Alves-Filho, J.C.; Cunha, T.M.; Cunha, F.Q.; Casagrande, R.; et al. Budlein A, a Sesquiterpene Lactone from Viguiera robusta, Alleviates Pain and Inflammation in a Model of Acute Gout Arthritis in Mice. Front. Pharmacol. 2018, 9, 1076. [Google Scholar] [CrossRef]
- Ahmad, M.; Saeed, F.; Mehjabeen; Jahan, N. Neuro-pharmacological and analgesic effects of Arnica montana extract. Int. J. Pharm. Pharm. Sci. 2013, 5, 590–593. [Google Scholar]
- Gounder, R. Kava consumption and its health effects. Pac. Health Dialog. 2006, 13, 131–135. [Google Scholar]
- El-Mahmoudy, A.; Gheith, I. The anti-nociceptive potential of tilmicosin against chemical-induced but not thermal-induced pain in mice. Int. J. Immunopathol. Pharmacol. 2016, 29, 9–16. [Google Scholar] [CrossRef]
- Portaels, F.; Chemlal, K.; Elsen, P.; Johnson, P.D.; Hayman, J.A.; Hibble, J.; Kirkwood, R.; Meyers, W.M. Mycobacterium ulcerans in wild animals. Rev. Sci. Tech. 2001, 20, 252–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stinear, T.P.; Mve-Obiang, A.; Small, P.L.; Frigui, W.; Pryor, M.J.; Brosch, R.; Jenkin, G.A.; Johnson, P.D.; Davies, J.K.; Lee, R.E.; et al. Giant plasmid-encoded polyketide synthases produce the macrolide toxin of Mycobacterium ulcerans. Proc. Natl. Acad. Sci. USA 2004, 101, 1345–1349. [Google Scholar] [CrossRef]
- Porter, J.L.; Tobias, N.J.; Pidot, S.J.; Falgner, S.; Tuck, K.L.; Vettiger, A.; Hong, H.; Leadlay, P.F.; Stinear, T.P. The cell wall-associated mycolactone polyketide synthases are necessary but not sufficient for mycolactone biosynthesis. PLoS ONE 2013, 8, e70520. [Google Scholar] [CrossRef] [PubMed]
- Kubicek-Sutherland, J.Z.; Vu, D.M.; Anderson, A.S.; Sanchez, T.C.; Converse, P.J.; Marti-Arbona, R.; Nuermberger, E.L.; Swanson, B.I.; Mukundan, H. Understanding the Significance of Biochemistry in the Storage, Handling, Purification, and Sampling of Amphiphilic Mycolactone. Toxins 2019, 11, 202. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Spencer, J.B.; Porter, J.L.; Leadlay, P.F.; Stinear, T. A novel mycolactone from a clinical isolate of Mycobacterium ulcerans provides evidence for additional toxin heterogeneity as a result of specific changes in the modular polyketide synthase. Chembiochem 2005, 6, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Mve-Obiang, A.; Lee, R.E.; Portaels, F.; Small, P.L. Heterogeneity of mycolactones produced by clinical isolates of Mycobacterium ulcerans: Implications for virulence. Infect. Immun. 2003, 71, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Kishi, Y. Chemistry of mycolactones, the causative toxins of Buruli ulcer. Proc. Natl. Acad. Sci. USA 2011, 108, 6703–6708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fidanze, S.; Song, F.; Szlosek-Pinaud, M.; Small, P.L.C.; Kishi, Y. Complete Structure of the Mycolactones. J. Am. Chem. Soc. 2001, 123, 10117–10118. [Google Scholar] [CrossRef] [PubMed]
- Mve-Obiang, A.; Lee, R.E.; Umstot, E.S.; Trott, K.A.; Grammer, T.C.; Parker, J.M.; Ranger, B.S.; Grainger, R.; Mahrous, E.A.; Small, P.L. A newly discovered mycobacterial pathogen isolated from laboratory colonies of Xenopus species with lethal infections produces a novel form of mycolactone, the Mycobacterium ulcerans macrolide toxin. Infect. Immun. 2005, 73, 3307–3312. [Google Scholar] [CrossRef] [PubMed]
- Yip, M.J.; Porter, J.L.; Fyfe, J.A.; Lavender, C.J.; Portaels, F.; Rhodes, M.; Kator, H.; Colorni, A.; Jenkin, G.A.; Stinear, T. Evolution of Mycobacterium ulcerans and other mycolactone-producing mycobacteria from a common Mycobacterium marinum progenitor. J. Bacteriol. 2007, 189, 2021–2029. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Miyamoto, Y.; Ogura, Y.; Hayashi, T.; Hoshino, Y. Complete Chromosome Sequence of a Mycolactone-Producing Mycobacterium, Mycobacterium pseudoshottsii. Genome Announc. 2017, 5, e01363-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pidot, S.J.; Asiedu, K.; Kaser, M.; Fyfe, J.A.; Stinear, T.P. Mycobacterium ulcerans and other mycolactone-producing mycobacteria should be considered a single species. PLoS Negl. Trop. Dis. 2010, 4, e663. [Google Scholar] [CrossRef] [PubMed]
- Cregg, R.; Russo, G.; Gubbay, A.; Branford, R.; Sato, H. Pharmacogenetics of analgesic drugs. Br. J. Pain 2013, 7, 189–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gersbach, P.; Jantsch, A.; Feyen, F.; Scherr, N.; Dangy, J.P.; Pluschke, G.; Altmann, K.H. A ring-closing metathesis (RCM)-based approach to mycolactones A/B. Chemistry 2011, 17, 13017–13031. [Google Scholar] [CrossRef] [PubMed]
- Chany, A.C.; Casarotto, V.; Schmitt, M.; Tarnus, C.; Guenin-Mace, L.; Demangel, C.; Mirguet, O.; Eustache, J.; Blanchard, N. A diverted total synthesis of mycolactone analogues: An insight into Buruli ulcer toxins. Chemistry 2011, 17, 14413–14419. [Google Scholar] [CrossRef] [PubMed]
- Gehringer, M.; Altmann, K.H. The chemistry and biology of mycolactones. Beilstein J. Org. Chem. 2017, 13, 1596–1660. [Google Scholar] [CrossRef] [Green Version]
- Scherr, N.; Gersbach, P.; Dangy, J.P.; Bomio, C.; Li, J.; Altmann, K.H.; Pluschke, G. Structure-activity relationship studies on the macrolide exotoxin mycolactone of Mycobacterium ulcerans. PLoS Negl. Trop. Dis. 2013, 7, e2143. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Stinear, T.; Porter, J.; Demangel, C.; Leadlay, P.F. A novel mycolactone toxin obtained by biosynthetic engineering. Chembiochem. 2007, 8, 2043–2047. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.H.; Xiao, Y.; Rowe, M.P.; Cummins, T.R.; Zakon, H.H. Voltage-gated sodium channel in grasshopper mice defends against bark scorpion toxin. Science 2013, 342, 441–446. [Google Scholar] [CrossRef]
- Guenin-Macé, L.; Ruf, M.-T.; Pluschke, G.; Demangel, C. Mycolactone: More than Just a Cytotoxin. In Buruli ulcer: Mycobacterium Ulcerans Disease; Pluschke, G., Röltgen, K., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 117–134. [Google Scholar] [CrossRef] [Green Version]
- Marsollier, L.; Stinear, T.; Aubry, J.; Saint Andre, J.P.; Robert, R.; Legras, P.; Manceau, A.L.; Audrain, C.; Bourdon, S.; Kouakou, H.; et al. Aquatic plants stimulate the growth of and biofilm formation by Mycobacterium ulcerans in axenic culture and harbor these bacteria in the environment. Appl. Environ. Microbiol. 2004, 70, 1097–1103. [Google Scholar] [CrossRef]
- Marsollier, L.; Brodin, P.; Jackson, M.; Kordulakova, J.; Tafelmeyer, P.; Carbonnelle, E.; Aubry, J.; Milon, G.; Legras, P.; Andre, J.P.; et al. Impact of Mycobacterium ulcerans biofilm on transmissibility to ecological niches and Buruli ulcer pathogenesis. PLoS Pathog. 2007, 3, e62. [Google Scholar] [CrossRef] [PubMed]
- Senanayake, T.H.; Lu, Y.; Bohling, A.; Raja, S.; Band, H.; Vinogradov, S.V. Encapsulation of poorly soluble drugs in polymer-drug conjugates: Effect of dual-drug nanoformulations on cancer therapy. Pharm. Res. 2014, 31, 1605–1615. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J.; Dutertre, S.; Vetter, I.; Christie, M.J. Conus venom peptide pharmacology. Pharmacol. Rev. 2012, 64, 259–298. [Google Scholar] [CrossRef] [PubMed]
- McGivern, J.G. Ziconotide: A review of its pharmacology and use in the treatment of pain. Neuropsychiatr. Dis. Treat. 2007, 3, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Chawla, R.; Goyal, M. Topical anesthesia. J. Anaesthesiol. Clin. Pharmacol. 2015, 31, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Groninger, H.; Schisler, R.E. Topical capsaicin for neuropathic pain #255. J. Palliat. Med. 2012, 15, 946–947. [Google Scholar] [CrossRef] [PubMed]
- El-Maddawy, Z.; El-Ashmawy, I. Hepato-Renal and Hematological Effects of Diclofenac Sodium in Rats. Glob. J. Pharmacol. 2013, 7, 123–132. [Google Scholar]
- Wrighton, L.J.; O’Bosky, K.R.; Namm, J.P.; Senthil, M. Postoperative management after hepatic resection. World J. Gastrointest. Oncol. 2012, 3, 41–47. [Google Scholar] [CrossRef]
- Chapman, C.R.; Donaldson, G.W.; Davis, J.J.; Bradshaw, D.H. Improving individual measurement of postoperative pain: The pain trajectory. J. Pain 2011, 12, 257–262. [Google Scholar] [CrossRef]
- Boros, M.; Benko, R.; Bolcskei, K.; Szolcsanyi, J.; Bartho, L.; Petho, G. Effects of reference analgesics and psychoactive drugs on the noxious heat threshold of mice measured by an increasing-temperature water bath. Basic Clin. Pharmacol. Toxicol. 2013, 113, 385–390. [Google Scholar] [CrossRef]
- Chan, S.K.; Lai, P.B.; Li, P.T.; Wong, J.; Karmakar, M.K.; Lee, K.F.; Gin, T. The analgesic efficacy of continuous wound instillation with ropivacaine after open hepatic surgery. Anaesthesia 2010, 65, 1180–1186. [Google Scholar] [CrossRef] [PubMed]
- Ho, A.M.; Karmakar, M.K.; Cheung, M.; Lam, G.C. Right thoracic paravertebral analgesia for hepatectomy. Br. J. Anaesth. 2004, 93, 458–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brutcher, R.E.; Kurihara, C.; Bicket, M.C.; Moussavian-Yousefi, P.; Reece, D.E.; Solomon, L.M.; Griffith, S.R.; Jamison, D.E.; Cohen, S.P. Compounded Topical Pain Creams to Treat Localized Chronic Pain: A Randomized Controlled Trial. Ann. Intern. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
| Molecule | Structure | Biological Effect |
|---|---|---|
| Angiotensin II |  | Natural AT1R and AT2R agonist, involved in haemodynamic effects. AngII induces TRPV1 sensitization, promotes pro-inflammatory responses, regulates gene expression and participates in many cell processes and signalling pathways. |
| Mycolactone |  | AT2R ligand. Main factor of virulence of M.ulcerans; the toxin induces analgesia, sustaining the painlessness of the ulcerative lesions in Buruli ulcer (BU). Analgesia is mediated by K+-dependent hyperpolarization through AT2R activation. Sec61-dependent anti-inflammatory activity on the immune and nervous systems could also contribute to BU-associated analgesia. |
| Compound 21 (C21) |  | First synthetic selective agonist of AT2R; therapeutic potential for heart failure, nephroprotection, anti-inflammation, stroke and some dermatological applications. Does not interfere with mycolactone in binding to AT2R. |
| EMA401 |  | AT2R antagonist. Potent pain killer. Decreases TRPV1 expression. In a phase II clinical trial, the efficacy of EMA401 (100 mg, twice a day) was demonstrated in neuropathic pain, in comparison to placebo, after 3–4 weeks of oral administration. |
| Capsaicin |  | Activates TRPV1. Used in chemical pain models. At low doses, promotes analgesia through the desensitization of the TRPV1 receptor. |
| Conotoxin |  | Toxin extracted from cone snails. Promotes analgesia. Inhibits the release of pro-nociceptive neurochemicals such as glutamate, CGRP, and substance P. Ziconotide (Prialt) is the synthetic compound inspired by conotoxins. This compound is an atypical agent for the management of severe and chronic pains. Intrathecal (IT) administration of Ziconotide appears 1000 times more effective than IT morphine delivery. |
| Botulinium toxin A |  | Toxin extracted from Clostridium botulinum, induces analgesia. Prevents the release of neurotransmitters CGRP and substance P and the expression of TRPV1. |
| Pain | Mycolactone-Induced Analgesia | ||||
|---|---|---|---|---|---|
| Pain Model | Dose-Time Effect | Suggested Mechanism | MycoL | Ref. | |
| Non inflammatory pain | Von Frey filament mechanical pain | 100 µg into mouse footpad, 28 days: ↓response to pressure (g) | Nerve damage | Purified strain 1615 | [51] |
| Tail-flick adapted thermal pain | 5 µg into mouse footpad, 2 h→48 h:↑latency in withdrawal of the footpad in response to noxious thermal stimulus | K+-dependent hyperpolarization of neurons through AT2R, a mechanism further confirmed in DRG neurons [69] | Purified strain 1615, ∑ mycoL | [52] | |
| Hargreaves plantar test thermal pain | 1,2,4 µg into mouse footpad:↑latency in withdrawal of the footpad in response to noxious thermal stimulus, analgesia 2 h→48 h for the highest dose | Basal state reached after 48 h indicating no nerve damages; K+-dependent hyperpolarization of neurons through AT2R | Purified strain 1615 | [53] | |
| Early phase of formalin-induced chemical pain (0–5 min) | IP injection of mycoL purified (0.5 mg/kg) or 5b (5 mg/kg) 1 h before subcutaneous injection of 10 µL of formalin solution (5%) into mouse hindpaw: no↓in pain score (paw licking duration and body tremor number) | Efficiency at inflammatory stages | Purified strain 1615, ∑ 5b | [62] | |
| Inflammatory pain | Second phase of formalin-induced chemical pain (10–40 min) | IP injection of mycoL purified (0.5 mg/kg) or 5b (5 mg/kg) 1 h before subcutaneous injection of 10 µL of formalin solution (5%) into mouse hindpaw: significant↓in pain score anti-inflammatory effect on DRG neurons stimulated by LPS (16 h) | Analgesic effect via AT2R, suppression of inflammatory cytokine production, or both mechanisms | Purified strain 1615, ∑ 5b | [62] [63] |
| Neuropathic pain | Chronic constriction injury of the sciatic nerve | Intrathecal injection of 100 ng mycoL 3 days to rats at day 2 post-operation: ↓expression levels of pro-inflammatory cytokines in mycoL-treated sham-operated rats | inflammatory cytokines inhibition through the blockade of Sec61 translocon (see also [61]) | Purified strain 1615 | [63] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reynaert, M.-L.; Dupoiron, D.; Yeramian, E.; Marsollier, L.; Brodin, P. Could Mycolactone Inspire New Potent Analgesics? Perspectives and Pitfalls. Toxins 2019, 11, 516. https://doi.org/10.3390/toxins11090516
Reynaert M-L, Dupoiron D, Yeramian E, Marsollier L, Brodin P. Could Mycolactone Inspire New Potent Analgesics? Perspectives and Pitfalls. Toxins. 2019; 11(9):516. https://doi.org/10.3390/toxins11090516
Chicago/Turabian StyleReynaert, Marie-Line, Denis Dupoiron, Edouard Yeramian, Laurent Marsollier, and Priscille Brodin. 2019. "Could Mycolactone Inspire New Potent Analgesics? Perspectives and Pitfalls" Toxins 11, no. 9: 516. https://doi.org/10.3390/toxins11090516
APA StyleReynaert, M.-L., Dupoiron, D., Yeramian, E., Marsollier, L., & Brodin, P. (2019). Could Mycolactone Inspire New Potent Analgesics? Perspectives and Pitfalls. Toxins, 11(9), 516. https://doi.org/10.3390/toxins11090516





