Validation of Predicted Virulence Factors in Listeria monocytogenes Identified Using Comparative Genomics
Abstract
1. Introduction
2. Results
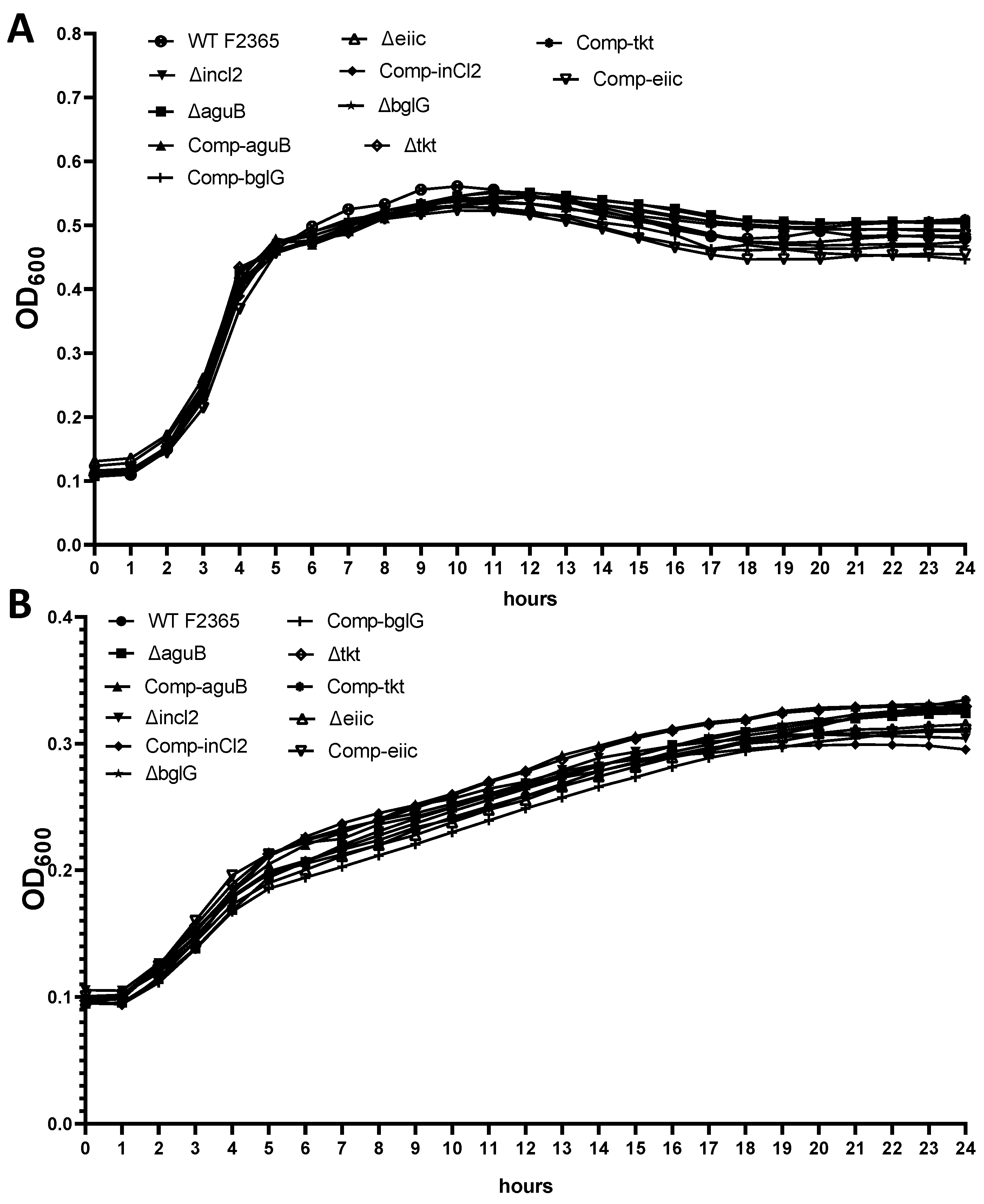
2.1. Growth of Mutants
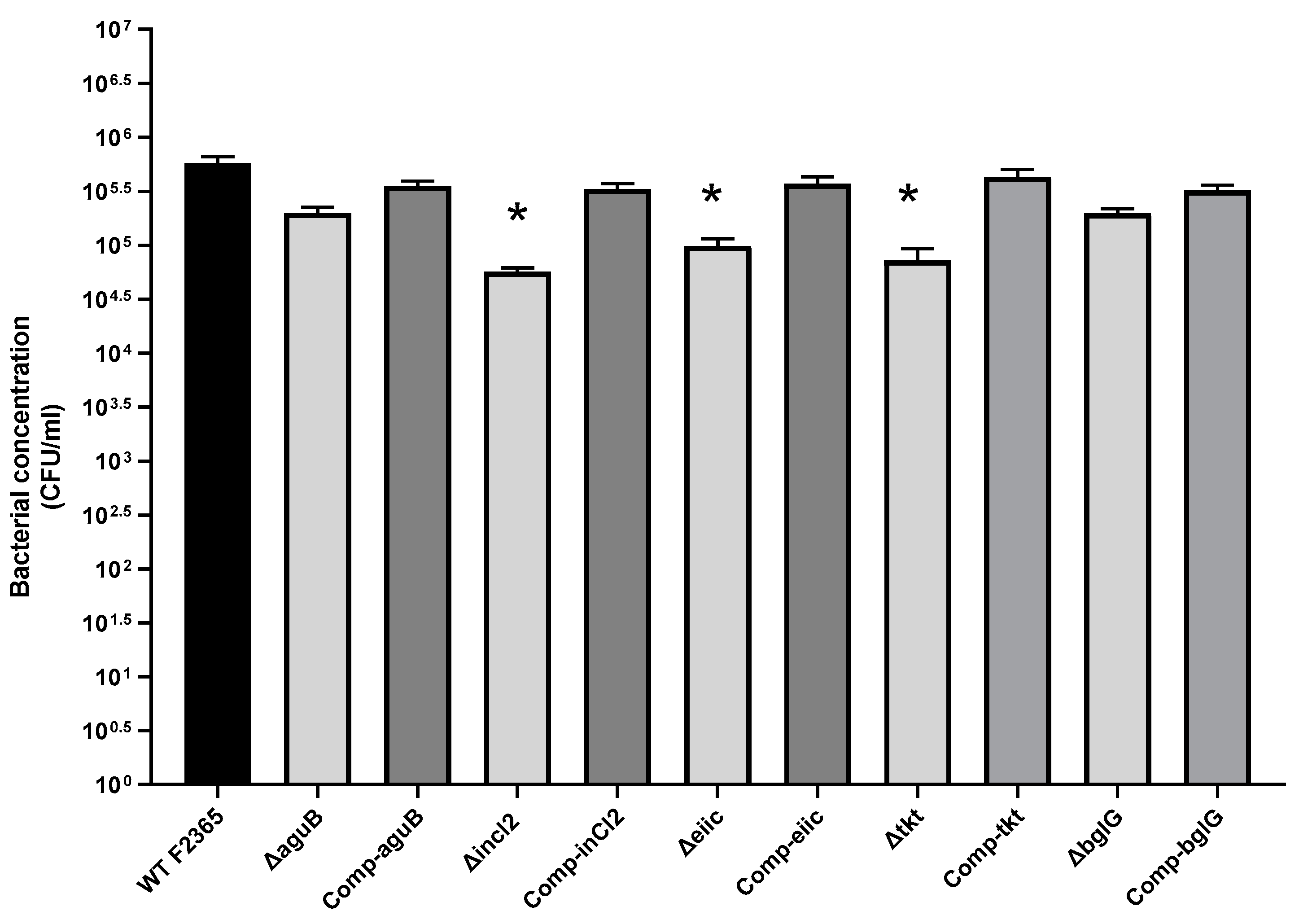
2.2. Role of aguB, inlC2, eiic, tkt, and bglG in L. Monocytogenes Adhesion
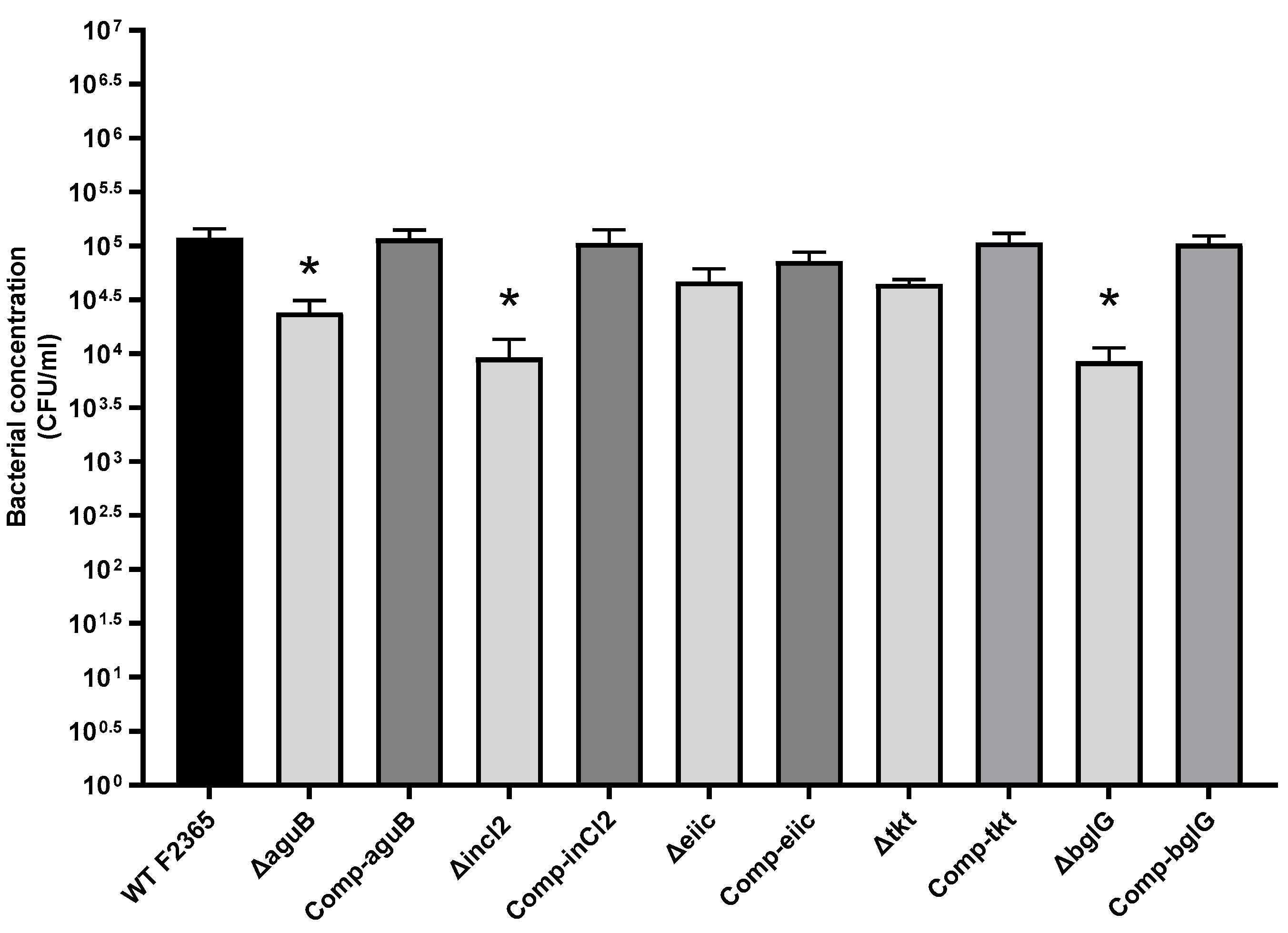
2.3. Role of aguB, inlC2, eiic, tkt, and bglG in L. Monocytogenes Invasion
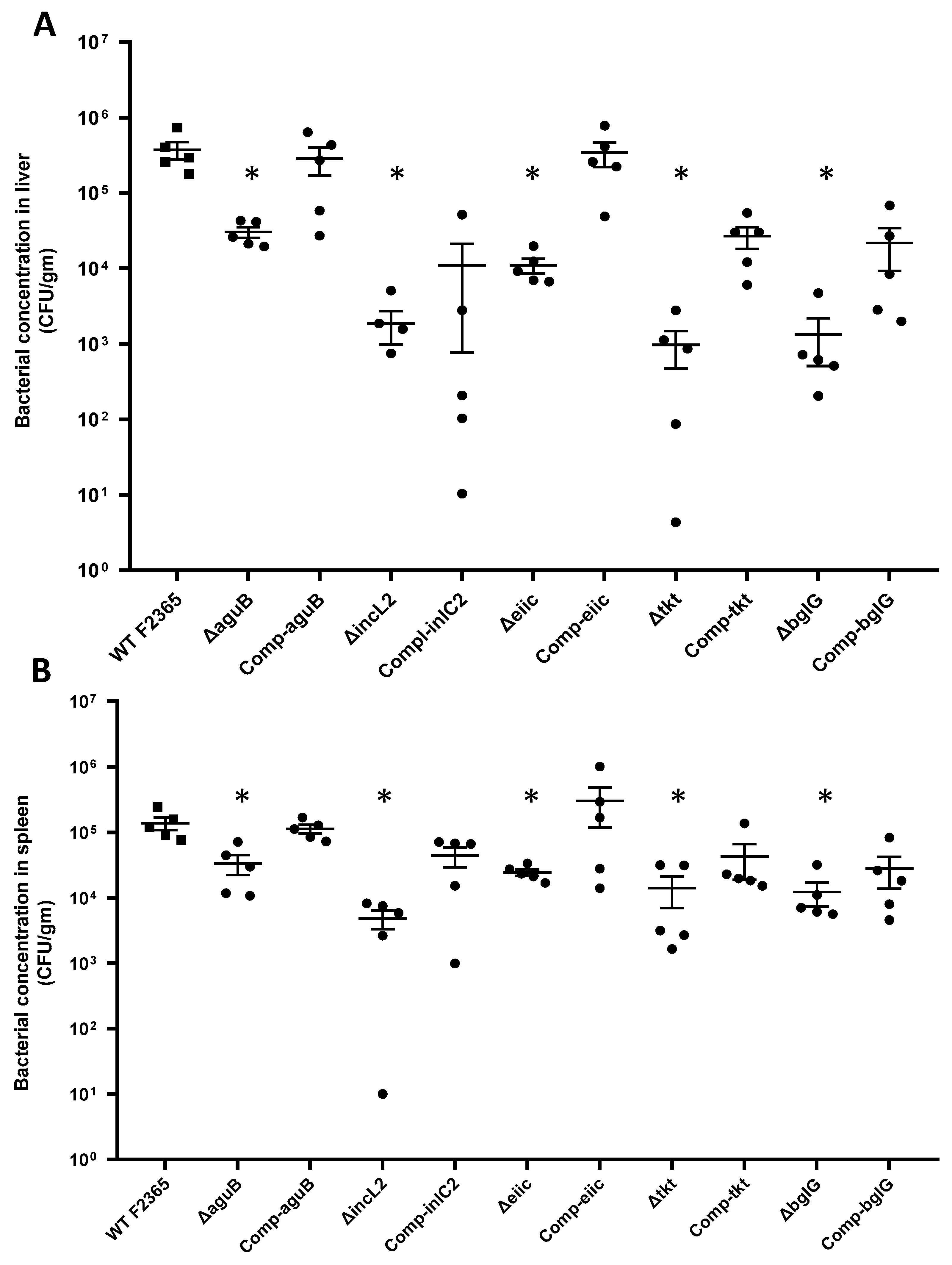
2.4. Virulence and Colonization of Mutant Strains in Mice
3. Discussion
4. Material and Methods
4.1. Ethics Statement
4.2. Bacterial Strains and Growth Conditions
4.3. Construction of L. Monocytogenes Mutants
4.4. Complementation of the Mutant Strains
4.5. Growth Kinetics of Mutants
4.6. Adherence and Invasion Assay
4.7. Virulence in Mice
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-related illness and death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar]
- Norton, D.M.; Scarlett, J.M.; Horton, K.; Sue, D.; Thimothe, J.; Boor, K.J.; Wiedmann, M. Characterization and pathogenic potential of Listeria monocytogenes isolates from the smoked fish industry. Appl. Environ. Microbiol. 2001, 67, 646–653. [Google Scholar] [CrossRef]
- Liu, D.; Lawrence, M.L.; Wiedmann, M.; Gorski, L.; Mandrell, R.E.; Ainsworth, A.J.; Austin, F.W. Listeria monocytogenes subgroups IIIA, IIIB, and IIIC delineate genetically distinct populations with varied pathogenic potential. J. Clin. Microbiol. 2006, 44, 4229–4233. [Google Scholar] [CrossRef]
- Hamon, M.A.; Batsche, E.; Regnault, B.; Tham, T.N.; Seveau, S.; Muchardt, C.; Cossart, P. Histone modifications induced by a family of bacterial toxins. Proc. Natl. Acad. Sci. USA 2007, 104, 13467–13472. [Google Scholar]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef]
- Orsi, R.H.; Wiedmann, M. Characteristics and distribution of Listeria spp., including Listeria species newly described since 2009. Appl. Microbiol. Biotechnol. 2016, 100, 5273–5287. [Google Scholar]
- Cocolin, L.; Rantsiou, K.; Iacumin, L.; Cantoni, C.; Comi, G. Direct identification in food samples of Listeria spp. and Listeria monocytogenes by molecular methods. Appl. Environ. Microbiol. 2002, 68, 6273–6282. [Google Scholar] [CrossRef]
- Scharff, R.L. Economic Burden from Health Losses Due to Foodborne Illness in the United States. J. Food Prot. 2012, 75, 123–131. [Google Scholar] [CrossRef]
- Guillet, C.; Join-Lambert, O.; Le Monnier, A.; Leclercq, A.; Mechai, F.; Mamzer-Bruneel, M.F.; Bielecka, M.K.; Scortti, M.; Disson, O.; Berche, P.; et al. Human listeriosis caused by Listeria ivanovii. Emerg. Infect. Dis. 2010, 16, 136–138. [Google Scholar] [CrossRef]
- Snapir, Y.M.; Vaisbein, E.; Nassar, F. Low virulence but potentially fatal outcome-Listeria ivanovii. Eur. J. Intern. Med. 2006, 17, 286–287. [Google Scholar]
- Sauders, B.D.; Overdevest, J.; Fortes, E.; Windham, K.; Schukken, Y.; Lembo, A.; Wiedmann, M. Diversity of Listeria species in urban and natural environments. Appl. Environ. Microbiol. 2012, 78, 4420–4433. [Google Scholar]
- Paul, D.; Steele, C.; Donaldson, J.R.; Banes, M.M.; Kumar, R.; Bridges, S.M.; Arick, M., 2nd; Lawrence, M.L. Genome comparison of Listeria monocytogenes serotype 4a strain HCC23 with selected lineage I and lineage II L. monocytogenes strains and other Listeria strains. Genom. Data 2014, 2, 219–225. [Google Scholar] [CrossRef]
- Begley, M.; Bron, P.A.; Heuston, S.; Casey, P.G.; Englert, N.; Wiesner, J.; Jomaa, H.; Gahan, C.G.; Hill, C. Analysis of the isoprenoid biosynthesis pathways in Listeria monocytogenes reveals a role for the alternative 2-C-methyl-D-erythritol 4-phosphate pathway in murine infection. Infect. Immun. 2008, 76, 5392–5401. [Google Scholar] [CrossRef]
- Raffelsbauer, D.; Bubert, A.; Engelbrecht, F.; Scheinpflug, J.; Simm, A.; Hess, J.; Kaufmann, S.H.; Goebel, W. The gene cluster inlC2DE of Listeria monocytogenes contains additional new internalin genes and is important for virulence in mice. Mol. Gen. Genet. 1998, 260, 144–158. [Google Scholar]
- Chen, J.; Cheng, C.; Xia, Y.; Zhao, H.; Fang, C.; Shan, Y.; Wu, B.; Fang, W. Lmo0036, an ornithine and putrescine carbamoyltransferase in Listeria monocytogenes, participates in arginine deiminase and agmatine deiminase pathways and mediates acid tolerance. Microbiology 2011, 157 Pt 11, 3150–3161. [Google Scholar]
- Cheng, C.; Chen, J.; Fang, C.; Xia, Y.; Shan, Y.; Liu, Y.; Wen, G.; Song, H.; Fang, W. Listeria monocytogenes aguA1, but not aguA2, encodes a functional agmatine deiminase: Biochemical characterization of its catalytic properties and roles in acid tolerance. J. Biol. Chem. 2013, 288, 26606–26615. [Google Scholar]
- Bonazzi, M.; Lecuit, M.; Cossart, P. Listeria monocytogenes internalin and E-cadherin: From structure to pathogenesis. Cell. Microbiol. 2009, 11, 693–702. [Google Scholar] [CrossRef]
- Bierne, H.; Sabet, C.; Personnic, N.; Cossart, P. Internalins: A complex family of leucine-rich repeat-containing proteins in Listeria monocytogenes. Microbes Infect. 2007, 9, 1156–1166. [Google Scholar]
- McCoy, J.G.; Levin, E.J.; Zhou, M. Structural insight into the PTS sugar transporter EIIC. Biochim. Biophys. Acta 2015, 1850, 577–585. [Google Scholar]
- Jung, Y.M.; Lee, J.N.; Shin, H.D.; Lee, Y.H. Role of tktA gene in pentose phosphate pathway on odd-ball biosynthesis of poly-beta-hydroxybutyrate in transformant Escherichia coli harboring phbCAB operon. J. Biosci. Bioeng. 2004, 98, 224–227. [Google Scholar] [CrossRef]
- Nussbaum-Shochat, A.; Amster-Choder, O. BglG, the transcriptional antiterminator of the bgl system, interacts with the beta’ subunit of the Escherichia coli RNA polymerase. Proc. Natl. Acad. Sci. USA 1999, 96, 4336–4341. [Google Scholar] [CrossRef]
- Raveh, H.; Lopian, L.; Nussbaum-Shochat, A.; Wright, A.; Amster-Choder, O. Modulation of transcription antitermination in the bgl operon of Escherichia coli by the PTS. Proc. Natl. Acad. Sci. USA 2009, 106, 13523–13528. [Google Scholar] [CrossRef]
- Glaser, P.; Frangeul, L.; Buchrieser, C.; Rusniok, C.; Amend, A.; Baquero, F.; Berche, P.; Bloecker, H.; Brandt, P.; Chakraborty, T.; et al. Comparative genomics of Listeria species. Science 2001, 294, 849–852. [Google Scholar]
- Casey, A.; Jordan, K.; Coffey, A.; Fox, E.M.; McAuliffe, O. Comparative Genomic Analysis of Two Serotype 1/2b Listeria monocytogenes Isolates from Analogous Environmental Niches Demonstrates the Influence of Hypervariable Hotspots in Defining Pathogenesis. Front. Nutr. 2016, 3, 54. [Google Scholar]
- Husna, A.U.; Wang, N.; Cobbold, S.A.; Newton, H.J.; Hocking, D.M.; Wilksch, J.J.; Scott, T.A.; Davies, M.R.; Hinton, J.C.; Tree, J.J.; et al. Methionine biosynthesis and transport are functionally redundant for the growth and virulence of Salmonella Typhimurium. J. Biol. Chem. 2018, 293, 9506–9519. [Google Scholar] [CrossRef]
- Drevets, D.A.; Sawyer, R.T.; Potter, T.A.; Campbell, P.A. Listeria monocytogenes infects human endothelial cells by two distinct mechanisms. Infect. Immun. 1995, 63, 4268–4276. [Google Scholar]
- Linnan, M.J.; Mascola, L.; Lou, X.D.; Goulet, V.; May, S.; Salminen, C.; Hird, D.W.; Yonekura, M.L.; Hayes, P.; Weaver, R.; et al. Epidemic listeriosis associated with Mexican-style cheese. N. Engl. J. Med. 1988, 319, 823–828. [Google Scholar] [CrossRef]
- Tilney, L.G.; Portnoy, D.A. Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes. J. Cell Biol. 1989, 109 Pt 1, 1597–1608. [Google Scholar] [CrossRef]
- Portnoy, D.A.; Chakraborty, T.; Goebel, W.; Cossart, P. Molecular determinants of Listeria monocytogenes pathogenesis. Infect. Immun. 1992, 60, 1263–1267. [Google Scholar]
- Sobyanin, K.A.; Sysolyatina, E.V.; Chalenko, Y.M.; Kalinin, E.V.; Ermolaeva, S.A. Route of Injection Affects the Impact of InlB Internalin Domain Variants on Severity of Listeria monocytogenes Infection in Mice. BioMed Res. Int. 2017, 2101575. [Google Scholar] [CrossRef]
- Cousens, L.P.; Wing, E.J. Innate defenses in the liver during Listeria infection. Immunol. Rev. 2000, 174, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Neuenhahn, M.; Busch, D.H. Unique functions of splenic CD8α+ dendritic cells during infection with intracellular pathogens. Immunol. Lett. 2007, 114, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Hoelzer, K.; Pouillot, R.; Dennis, S. Animal models of listeriosis: A comparative review of the current state of the art and lessons learned. Vet. Res. 2012, 43, 18. [Google Scholar] [CrossRef] [PubMed]
- Kernbauer, E.; Maier, V.; Rauch, I.; Muller, M.; Decker, T. Route of Infection Determines the Impact of Type I Interferons on Innate Immunity to Listeria monocytogenes. PLoS ONE 2013, 8, e65007. [Google Scholar] [CrossRef] [PubMed]
- Griswold, A.R.; Jameson-Lee, M.; Burne, R.A. Regulation and physiologic significance of the agmatine deiminase system of Streptococcus mutans UA159. J. Bacteriol. 2006, 188, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Llacer, J.L.; Polo, L.M.; Tavarez, S.; Alarcon, B.; Hilario, R.; Rubio, V. The gene cluster for agmatine catabolism of Enterococcus faecalis: Study of recombinant putrescine transcarbamylase and agmatine deiminase and a snapshot of agmatine deiminase catalyzing its reaction. J. Bacteriol. 2007, 189, 1254–1265. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Liu, Y.; Paoli, G.C. How does Listeria monocytogenes combat acid conditions? Can. J. Microb. 2012, 59, 141–152. [Google Scholar] [CrossRef]
- Lucas, P.M.; Blancato, V.S.; Claisse, O.; Magni, C.; Lolkema, J.S.; Lonvaud-Funel, A. Agmatine deiminase pathway genes in Lactobacillus brevis are linked to the tyrosine decarboxylation operon in a putative acid resistance locus. Microbiology 2007, 153 Pt 7, 2221–2230. [Google Scholar] [CrossRef]
- Ryan, S.; Begley, M.; Gahan, C.G.; Hill, C. Molecular characterization of the arginine deiminase system in Listeria monocytogenes: Regulation and role in acid tolerance. Environ. Microbiol. 2009, 11, 432–445. [Google Scholar] [CrossRef]
- Jelsbak, L.; Thomsen, L.E.; Wallrodt, I.; Jensen, P.R.; Olsen, J.E. Polyamines are required for virulence in Salmonella enterica serovar Typhimurium. PLoS ONE 2012, 7, e36149. [Google Scholar] [CrossRef]
- Gaillard, J.L.; Berche, P.; Frehel, C.; Gouln, E.; Cossart, P. Entry of L. monocytogenes into cells is mediated by internalin, a repeat protein reminiscent of surface antigens from gram-positive cocci. Cell 1991, 65, 1127–1141. [Google Scholar] [CrossRef]
- Rajabian, T.; Gavicherla, B.; Heisig, M.; Muller-Altrock, S.; Goebel, W.; Gray-Owen, S.D.; Ireton, K. The bacterial virulence factor InlC perturbs apical cell junctions and promotes cell-to-cell spread of Listeria. Nat. Cell Biol. 2009, 11, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Hain, T.; Hossain, H.; Chatterjee, S.S.; Machata, S.; Volk, U.; Wagner, S.; Brors, B.; Haas, S.; Kuenne, C.T.; Billion, A.; et al. Temporal transcriptomic analysis of the Listeria monocytogenes EGD-e sigmaB regulon. BMC Microbiol. 2008, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Kazmierczak, M.J.; Mithoe, S.C.; Boor, K.J.; Wiedmann, M. Listeria monocytogenes sigma B regulates stress response and virulence functions. J. Bacteriol. 2003, 185, 5722–5734. [Google Scholar] [CrossRef] [PubMed]
- Dramsi, S.; Dehoux, P.; Lebrun, M.; Goossens, P.L.; Cossart, P. Identification of four new members of the internalin multigene family of Listeria monocytogenes EGD. Infect. Immun. 1997, 65, 1615–1625. [Google Scholar] [PubMed]
- Jia, Y.; Nightingale, K.K.; Boor, K.J.; Ho, A.; Wiedmann, M.; McGann, P. Distribution of internalin gene profiles of Listeria monocytogenes isolates from different sources associated with phylogenetic lineages. Foodborne Pathog. Dis. 2007, 4, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.L.; Dan, H.; Lin, M. Novel protein targets of the humoral immune response to Listeria monocytogenes infection in rabbits. J. Med. Microbiol. 2007, 56 Pt 7, 888–895. [Google Scholar] [CrossRef]
- Jiang, J.; Chen, J.; Cheng, C.; Hu, H.; Bai, F.; Chen, N.; Yan, G.; Fang, W. Disruption of InlC2 enhances the internalization of Listeria monocytogenes by epithelial cells. World J. Microbiol. Biotechnol. 2011, 27, 2155–2161. [Google Scholar] [CrossRef]
- Gözel, B.; Monney, C.; Aguilar-Bultet, L.; Rupp, S.; Frey, J.; Oevermann, A. Hyperinvasiveness of Listeria monocytogenes sequence type 1 is independent of lineage I-specific genes encoding internalin-like proteins. MicrobiologyOpen 2019, 8, e00790. [Google Scholar] [CrossRef] [PubMed]
- Bierne, H.; Cossart, P. Listeria monocytogenes surface proteins: From genome predictions to function. Microbiol. Mol. Biol. Rev. 2007, 71, 377–397. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.L.; Dan, H.; Lin, M. InlA and InlC2 of Listeria monocytogenes Serotype 4b Are Two Internalin Proteins Eliciting Humoral Immune Responses Common to Listerial Infection of Various Host Species. Curr. Microbiol. 2008, 56, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Grisafi, P.L.; Scholle, A.; Sugiyama, J.; Briggs, C.; Jacobson, G.R.; Lengeler, J.W. Deletion mutants of the Escherichia coli K-12 mannitol permease: Dissection of transport-phosphorylation, phospho-exchange, and mannitol-binding activities. J. Bacteriol. 1989, 171, 2719–2727. [Google Scholar] [CrossRef] [PubMed]
- Postma, P.W.; Lengeler, J.W.; Jacobson, G.R. Phosphoenolpyruvate:carbohydrate phosphotransferase systems of bacteria. Microbiol. Rev. 1993, 57, 543–594. [Google Scholar]
- Boddicker, J.D.; Anderson, R.A.; Jagnow, J.; Clegg, S. Signature-tagged mutagenesis of Klebsiella pneumoniae to identify genes that influence biofilm formation on extracellular matrix material. Infect. Immun. 2006, 74, 4590–4597. [Google Scholar] [CrossRef] [PubMed]
- Kilic, A.O.; Tao, L.; Zhang, Y.; Lei, Y.; Khammanivong, A.; Herzberg, M.C. Involvement of Streptococcus gordonii beta-glucoside metabolism systems in adhesion, biofilm formation, and in vivo gene expression. J. Bacteriol. 2004, 186, 4246–4253. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.C.; Peng, H.L.; Chang, H.Y. Identification of genes induced in vivo during Klebsiella pneumoniae CG43 infection. Infect. Immun. 2001, 69, 7140–7145. [Google Scholar] [CrossRef] [PubMed]
- Kochetov, G.A. Transketolase: Structure and mechanism of action. Biokhimiia 1986, 51, 2010–2029. [Google Scholar] [PubMed]
- Datta, A.G.; Racker, E. Mechanism of action of transketolase. II. The substrate-enzyme intermediate. J. Biol. Chem. 1961, 236, 624–628. [Google Scholar]
- Zhao, G.; Winkler, M.E. An Escherichia coli K-12 tktA tktB mutant deficient in transketolase activity requires pyridoxine (vitamin B6) as well as the aromatic amino acids and vitamins for growth. J. Bacteriol. 1994, 176, 6134–6138. [Google Scholar] [CrossRef]
- Kovarova, J.; Pountain, A.W.; Wildridge, D.; Weidt, S.; Bringaud, F.; Burchmore, R.J.S.; Achcar, F.; Barrett, M.P. Deletion of transketolase triggers a stringent metabolic response in promastigotes and loss of virulence in amastigotes of Leishmania mexicana. PLoS Pathog. 2018, 14, e1006953. [Google Scholar] [CrossRef]
- Tuntufye, H.N.; Lebeer, S.; Gwakisa, P.S.; Goddeeris, B.M. Identification of Avian Pathogenic Escherichia coli Genes That Are Induced In Vivo during Infection in Chickens. Appl. Environ. Microbiol. 2012, 78, 3343–3351. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Tivendale, K.A.; Liu, P.; Feng, Y.; Wannemuehler, Y.; Cai, W.; Mangiamele, P.; Johnson, T.J.; Constantinidou, C.; Penn, C.W.; et al. Transcriptome analysis of avian pathogenic Escherichia coli O1 in chicken serum reveals adaptive responses to systemic infection. Infect. Immun. 2011, 79, 1951–1960. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Laturnus, C.; Ewers, C.; Wieler, L.H. Identification of genes required for avian Escherichia coli septicemia by signature-tagged mutagenesis. Infect. Immun. 2005, 73, 2818–2827. [Google Scholar] [CrossRef] [PubMed]
- Kolly, G.S.; Sala, C.; Vocat, A.; Cole, S.T. Assessing essentiality of transketolase in Mycobacterium tuberculosis using an inducible protein degradation system. FEMS Microbiol. Lett. 2014, 358, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Domain, F.; Bina, X.R.; Levy, S.B. Transketolase A, an enzyme in central metabolism, derepresses the marRAB multiple antibiotic resistance operon of Escherichia coli by interaction with MarR. Mol. Microbiol. 2007, 66, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Bigot, A.; Pagniez, H.; Botton, E.; Frehel, C.; Dubail, I.; Jacquet, C.; Charbit, A.; Raynaud, C. Role of FliF and FliI of Listeria monocytogenes in flagellar assembly and pathogenicity. Infect. Immun. 2005, 73, 5530–5539. [Google Scholar] [CrossRef] [PubMed]
- Schnetz, K.; Rak, B. Regulation of the bgl operon of Escherichia coli by transcriptional antitermination. EMBO J. 1988, 7, 3271–3277. [Google Scholar] [CrossRef]
- Mahadevan, S.; Wright, A. A bacterial gene involved in transcription antitermination: Regulation at a rho-independent terminator in the bgl operon of E. coli. Cell 1987, 50, 485–494. [Google Scholar] [CrossRef]
- Amster-Choder, O.; Wright, A. Transcriptional regulation of the bgl operon of Escherichia coli involves phosphotransferase system-mediated phosphorylation of a transcriptional antiterminator. J. Cell. Biochem. 1993, 51, 83–90. [Google Scholar] [CrossRef]
- Rothe, F.M.; Bahr, T.; Stulke, J.; Rak, B.; Gorke, B. Activation of Escherichia coli antiterminator BglG requires its phosphorylation. Proc. Natl. Acad. Sci. USA 2012, 109, 15906–15911. [Google Scholar] [CrossRef]
- Glenn, S.M. Genes Involved in Attachment of Listeria Monocytogenes to Abiotic Surfaces. Ph.D. Thesis, University of Leicester, Leicester, UK, 2012. [Google Scholar]
- Gorski, L.; Palumbo, J.D.; Mandrell, R.E. Attachment of Listeria monocytogenes to radish tissue is dependent upon temperature and flagellar motility. Appl. Environ. Microbiol. 2003, 69, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Ray, M.A.; Johnston, N.A.; Verhulst, S.; Trammell, R.A.; Toth, L.A. Identification of markers for imminent death in mice used in longevity and aging research. J. Am. Assoc. Lab. Anim. Sci. 2010, 49, 282–288. [Google Scholar] [PubMed]
- Gordon, C.J. Effect of cage bedding on temperature regulation and metabolism of group-housed female mice. Comp. Med. 2004, 54, 63–68. [Google Scholar] [PubMed]
- Overton, J.M.; Williams, T.D. Behavioral and physiologic responses to caloric restriction in mice. Physiol. Behav. 2004, 81, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Nemzek, J.A.; Xiao, H.Y.; Minard, A.E.; Bolgos, G.L.; Remick, D.G. Humane endpoints in shock research. Shock 2004, 21, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Krarup, A.; Chattopadhyay, P.; Bhattacharjee, A.K.; Burge, J.R.; Ruble, G.R. Evaluation of surrogate markers of impending death in the galactosamine-sensitized murine model of bacterial endotoxemia. Lab. Anim. Sci. 1999, 49, 545–550. [Google Scholar]
- Abdelhamed, H.; Lawrence, M.L.; Karsi, A. A novel suicide plasmid for efficient gene mutation in Listeria monocytogenes. Plasmid 2015, 81, 1–8. [Google Scholar] [CrossRef]
- Nelson, K.E.; Fouts, D.E.; Mongodin, E.F.; Ravel, J.; DeBoy, R.T.; Kolonay, J.F.; Rasko, D.A.; Angiuoli, S.V.; Gill, S.R.; Paulsen, I.T.; et al. Whole genome comparisons of serotype 4b and 1/2a strains of the food-borne pathogen Listeria monocytogenes reveal new insights into the core genome components of this species. Nucleic Acids Res. 2004, 32, 2386–2395. [Google Scholar] [CrossRef]
- Lauer, P.; Chow, M.Y.; Loessner, M.J.; Portnoy, D.A.; Calendar, R. Construction, characterization, and use of two Listeria monocytogenes site-specific phage integration vectors. J. Bacteriol. 2002, 184, 4177–4186. [Google Scholar] [CrossRef]
- Horton, R.M.; Hunt, H.D.; Ho, S.N.; Pullen, J.K.; Pease, L.R. Engineering hybrid genes without the use of restriction enzymes: Gene splicing by overlap extension. Gene 1989, 77, 61–68. [Google Scholar] [CrossRef]
- Monk, I.R.; Shah, I.M.; Xu, M.; Tan, M.W.; Foster, T.J. Transforming the Untransformable. Application of Direct Transformation To Manipulate Genetically Staphylococcus aureus and Staphylococcus epidermidis. mBio 2012, 3, e00277-11. [Google Scholar] [CrossRef] [PubMed]
- Bae, T.; Schneewind, O. Allelic replacement in Staphylococcus aureus with inducible counter-selection. Plasmid 2006, 55, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Premaratne, R.J.; Lin, W.J.; Johnson, E.A. Development of an improved chemically defined minimal medium for Listeria monocytogenes. Appl. Environ. Microbiol. 1991, 57, 3046–3048. [Google Scholar] [PubMed]
- Cowart, R.E.; Lashmet, J.; McIntosh, M.E.; Adams, T.J. Adherence of a virulent strain of Listeria monocytogenes to the surface of a hepatocarcinoma cell line via lectin-substrate interaction. Arch. Microbiol. 1990, 153, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.; Turaga, G.; Abdelhamed, H.; Banes, M.M.; Wills, R.W.; Lawrence, M.L. Listeria monocytogenes PdeE, a phosphodiesterase that contributes to virulence and has hydrolytic activity against cyclic mononucleotides and cyclic dinucleotides. Microb. Pathog. 2017, 110, 399–408. [Google Scholar] [CrossRef]
- Kortebi, M.; Milohanic, E.; Mitchell, G.; Pechoux, C.; Prevost, M.C.; Cossart, P.; Bierne, H. Listeria monocytogenes switches from dissemination to persistence by adopting a vacuolar lifestyle in epithelial cells. PLoS Pathog. 2017, 13, e1006734. [Google Scholar] [CrossRef]
- Maurin, M.; Raoult, D. Use of aminoglycosides in treatment of infections due to intracellular bacteria. Antimicrob. Agents Chemother. 2001, 45, 2977–2986. [Google Scholar] [CrossRef]
- Vaudaux, P.; Waldvogel, F.A. Gentamicin antibacterial activity in the presence of human polymorphonuclear leukocytes. Antimicrob. Agents Chemother. 1979, 16, 743–749. [Google Scholar] [CrossRef][Green Version]
| Bacterial Strains, Plasmid | Description | Source/Reference |
|---|---|---|
| E. coli | ||
| DH5α and Top10 | Competent cells | Invitrogen (Carlsbad, CA, USA) |
| L. monocytogenes | ||
| F2365 | wild-type serotype 4b strain | [79] |
| F2365ΔaguB | F2365ΔaguB mutant strain | This study |
| F2365ΔinlC2 | F2365ΔinlC2 mutant strain | This study |
| F2365Δeiic | F2365Δeiic mutant strain | This study |
| F2365Δtkt | F2365Δtkt mutant strain | This study |
| F2365ΔbglG | F2365ΔbglG mutant strain | This study |
| F2365ΔaguB::aguB | F2365ΔaguB::pPL2-aguB complemented strain | This study |
| F2365ΔinlC2::inlC2 | F2365ΔinlC2::pPL2-inlC2 complemented strain | This study |
| F2365Δeiic::eiic | F2365Δeiic::pPL2-eiic complemented strain | This study |
| F2365Δtkt::tkt | F2365Δtkt::pPL2-tkt complemented strain | This study |
| F2365ΔbglG::bglG | F2365ΔbglG::pPL2-bglG complemented strain | This study |
| Plasmids | ||
| pHoss1 | 8995 bp, pMAD::secY antisense, ΔbgaB | [78] |
| pPL2 | 6123 bp, PSA attPP, chlr | [80] |
| pLmΔaguB | pHoss1::ΔaguB | This study |
| pLmΔinlC2 | pHoss1::ΔinlC2 | This study |
| pLmΔeiic | pHoss1::Δeiic | This study |
| pLmΔtkt | pHoss1::Δtkt | This study |
| pLmΔbglG | pHoss1::ΔbglG | This study |
| pPl2-aguB | pPL2::aguB | This study |
| pPL2-inlC2 | pPL2::inlC2 | This study |
| pPL2-eiic | pPL2::eiic | This study |
| pPL2-tkt | pPL2::tkt | This study |
| pPL2-bglG | pPL2::bglG | This study |
| Mutants | Locus Tag | Encoded Protein | Function |
|---|---|---|---|
| F2365ΔaguB | LMOf2365_0045 | Putrescine carbamoyltransferase | Catalyzes the formation of putrescine from carbamoyl-putrescine during agmatine degradation |
| F2365ΔinlC2 | LMOf2365_0281 | Internalin C2 | Virulence, modulate host inflammation |
| F2365Δeiic | LMOf2365_0661 | Fructose-like permease EIIC subunit 2 | Putative fructose-like permease EIIC subunit 2 phosphotransferase system (PTS) enzyme |
| F2365Δtkt | LMOf2365_1054 | Transketolase_C | Transketolase, C-terminal subunit, putative transketolase, N-terminal subunit |
| F2365ΔbglG | LMOf2365_2763 | Transcription antiterminator, BglG family | Beta-glucoside operon family transcription antiterminator |
| Primers | Description | Sequence (5’→3’) b | RE a |
|---|---|---|---|
| AguB_F01 | A | AAGTCGACTCCGTTCCAGTAGTCGCTCTA | SalI |
| AguB_R938 | B | CGGAATCACCCTGTAACTCGT | |
| AguB_F833 | C | ACGAGTTACAGGGTGATTCCGCGTTTTAGTTGTGGAATCTGC | |
| AguB_F01 | D | AACCATGGTTTCGCTGCATACATTGCTAC | NcoI |
| AguB_Seq | ATTGCGGAGTTGAAAGGCAAT | ||
| InlC2_F02 | A | AAGTCGACTTCATGGACCAAGCTACCAAT | SalI |
| InlC2_R954 | B | ACCCTTCTGTGCGAAAGATGT | |
| InlC2_F933 | C | ACATCTTTCGCACAGAAGGGTGGCAATTAGCTTTTGGGTAGG | |
| InlC2_R02 | D | AACCATGGATATTCGGGCTTGCATAAACA | NcoI |
| InlC2_seq | CGAATCAGAATAAACTGTTGC | ||
| Eiic_F01 | A | AAGTCGACGCAAAAGTGACAACCCCACTA | SalI |
| Eiic_R967 | B | TGGACAAATTCTTCCTCTTCA | |
| Eiic_F964 | C | TGAAGAGGAAGAATTTGTCCACGAGGAAGCAGATTGTGTCAT | |
| Eiic_F01 | D | AACCATGGCGCCATTATGCTCTTTCAAAC | NcoI |
| Eiic_Seq | TATGGTTGGTTCGATTGTAGG | ||
| Tkt_F01 | A | AAGTCGACAATTGCCGTCTATCTGATCCA | SalI |
| Tkt_R900 | B | CCTTTCTCTATTCACCGCGTA | |
| Tkt_F900 | C | TACGCGGTGAATAGAGAAAGGCCTTGGAGACGAAAGAAGATG | |
| Tkt_R01 | D | ATACCATGGCAATGTGTCTCCACAAGAACG | NcoI |
| Tkt_seq | GTTACTGGGTAAAGCGAGAGG | ||
| BglG_F01 | A | AAGTCGACCCGGTTGCCCTATATTTTAGC | SalI |
| BglG_R975 | B | CGCTGTCAATGGGTTTTGTTA | |
| BglG_F936 | C | TAACAAAACCCATTGACAGCGGCATTACCAGACTACGGTTTCA | |
| BglG_F01 | D | AACCATGGCTGCTTGGCTCATATTGGAAA | NcoI |
| BglG_Seq | GGGTATTATTGCTTGGATATGA | ||
| AguB_Comp_F01 | AAAGAGCTCATGGTTGAGGTGATAGAAATGA | SacI | |
| AguB_Comp_R01 | AAAGTCGACATCTTATAAGCCAGCGCCATT | SalI | |
| InlC2_Comp-F01 | AAAGAGCTCAATGGTAGCTGCTATTCTCGGTA | SacI | |
| InlC2_Comp-R01 | AAAGTCGACCACTTTGATTGTTTTGCGGAG | SalI | |
| EiiC_Comp_F01 | AAAGAGCTCGGAGGATAACTAAATGAGAACGCTTA | SacI | |
| Eiic_Comp_R01 | AAAGTCGACTCATCCTTTCTAAATGTCTTCAA | SalI | |
| Tkt_Comp-F01 | AAAGAGCTCTACGCGGTGAATAGAGAAAGG | SacI | |
| Tkt_Comp-R01 | AAAGTCGACCTTCATCTTCTTTCGTCTCCAAGG | SalI | |
| BglG_Comp_F01 | AAAGAGCTCTGGTGATTTGTTTGAGAATTGAG | SacI | |
| BglG_Comp_F01 | AAAGTCGACTCGCGGTAACAAGCCTATTAGT | SalI |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelhamed, H.; Lawrence, M.L.; Ramachandran, R.; Karsi, A. Validation of Predicted Virulence Factors in Listeria monocytogenes Identified Using Comparative Genomics. Toxins 2019, 11, 508. https://doi.org/10.3390/toxins11090508
Abdelhamed H, Lawrence ML, Ramachandran R, Karsi A. Validation of Predicted Virulence Factors in Listeria monocytogenes Identified Using Comparative Genomics. Toxins. 2019; 11(9):508. https://doi.org/10.3390/toxins11090508
Chicago/Turabian StyleAbdelhamed, Hossam, Mark L. Lawrence, Reshma Ramachandran, and Attila Karsi. 2019. "Validation of Predicted Virulence Factors in Listeria monocytogenes Identified Using Comparative Genomics" Toxins 11, no. 9: 508. https://doi.org/10.3390/toxins11090508
APA StyleAbdelhamed, H., Lawrence, M. L., Ramachandran, R., & Karsi, A. (2019). Validation of Predicted Virulence Factors in Listeria monocytogenes Identified Using Comparative Genomics. Toxins, 11(9), 508. https://doi.org/10.3390/toxins11090508





