The Relationship Analysis on Corn Stalk Rot and Ear Rot According to Fusarium Species and Fumonisin Contamination in Kernels
Abstract
:1. Introduction
2. Results
2.1. Frequency of Fusarium spp. in Samples
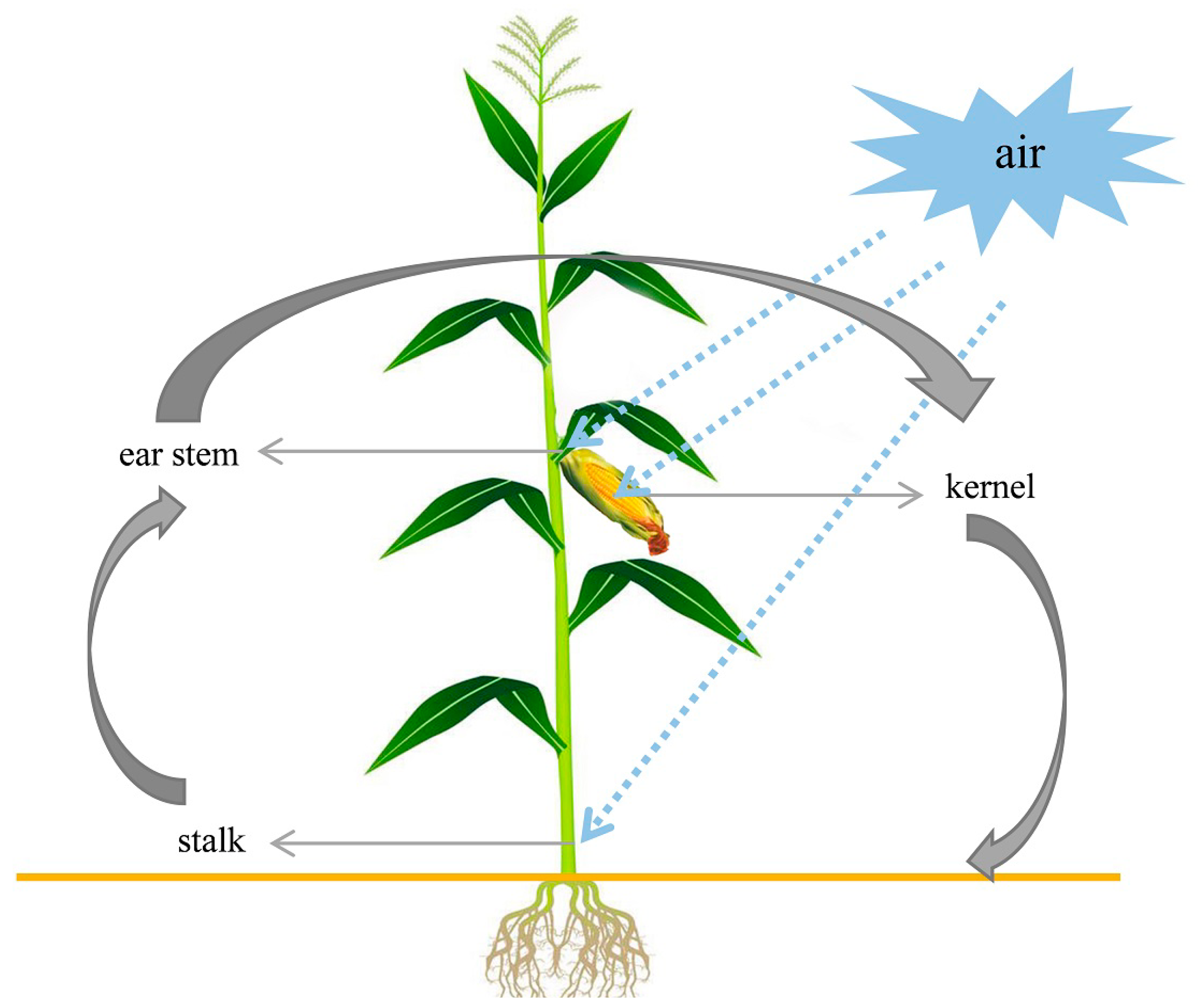
2.2. Fusarium Species in Stems and Ears from Corn Stalk Rot Samples
2.3. Frequency of Fusarium spp. in Different Parts of Corn
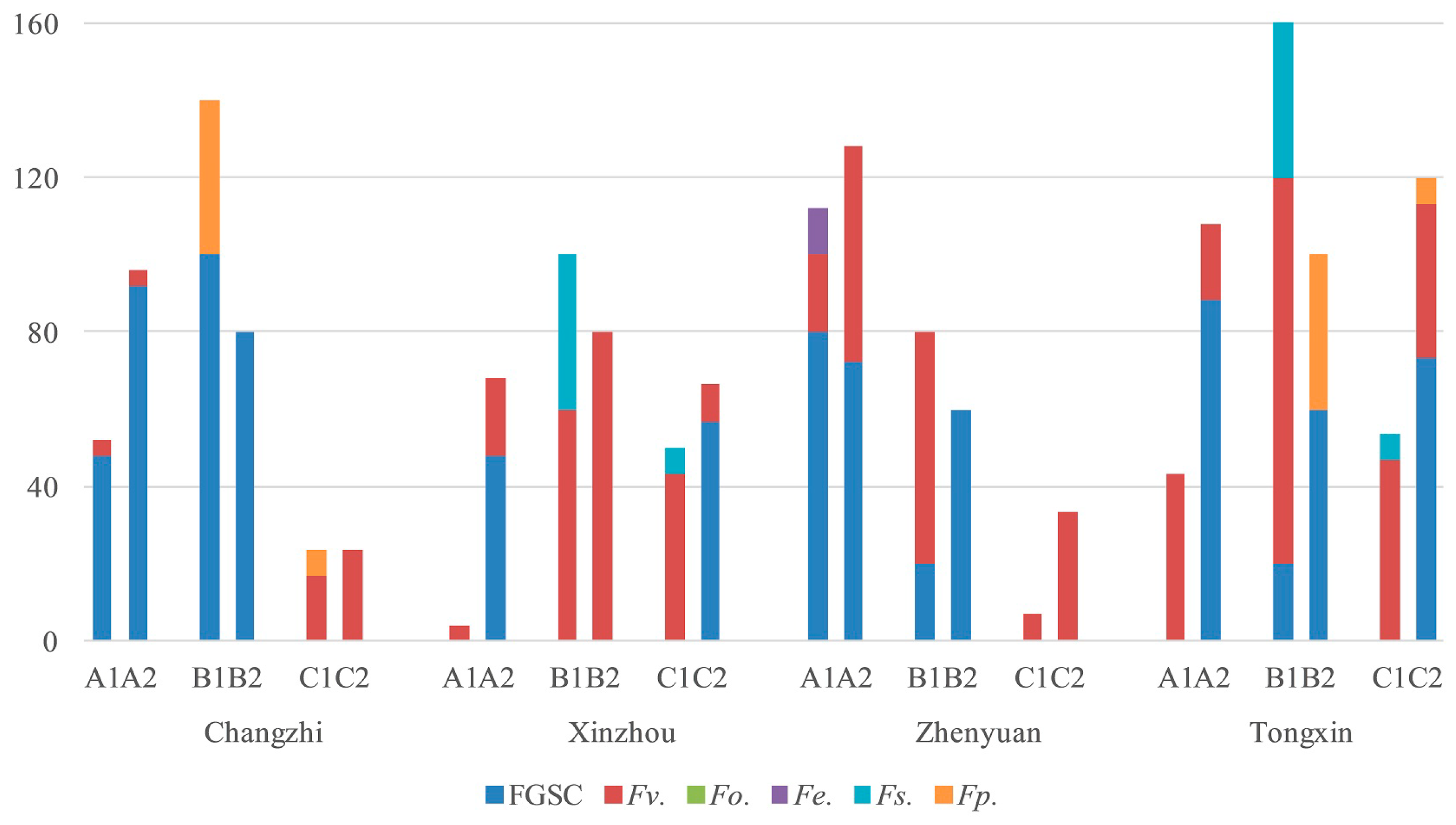
2.4. Fusarium Species in the Different Regions
2.5. Analysis of Fusarium spp. in Different Maize Varieties
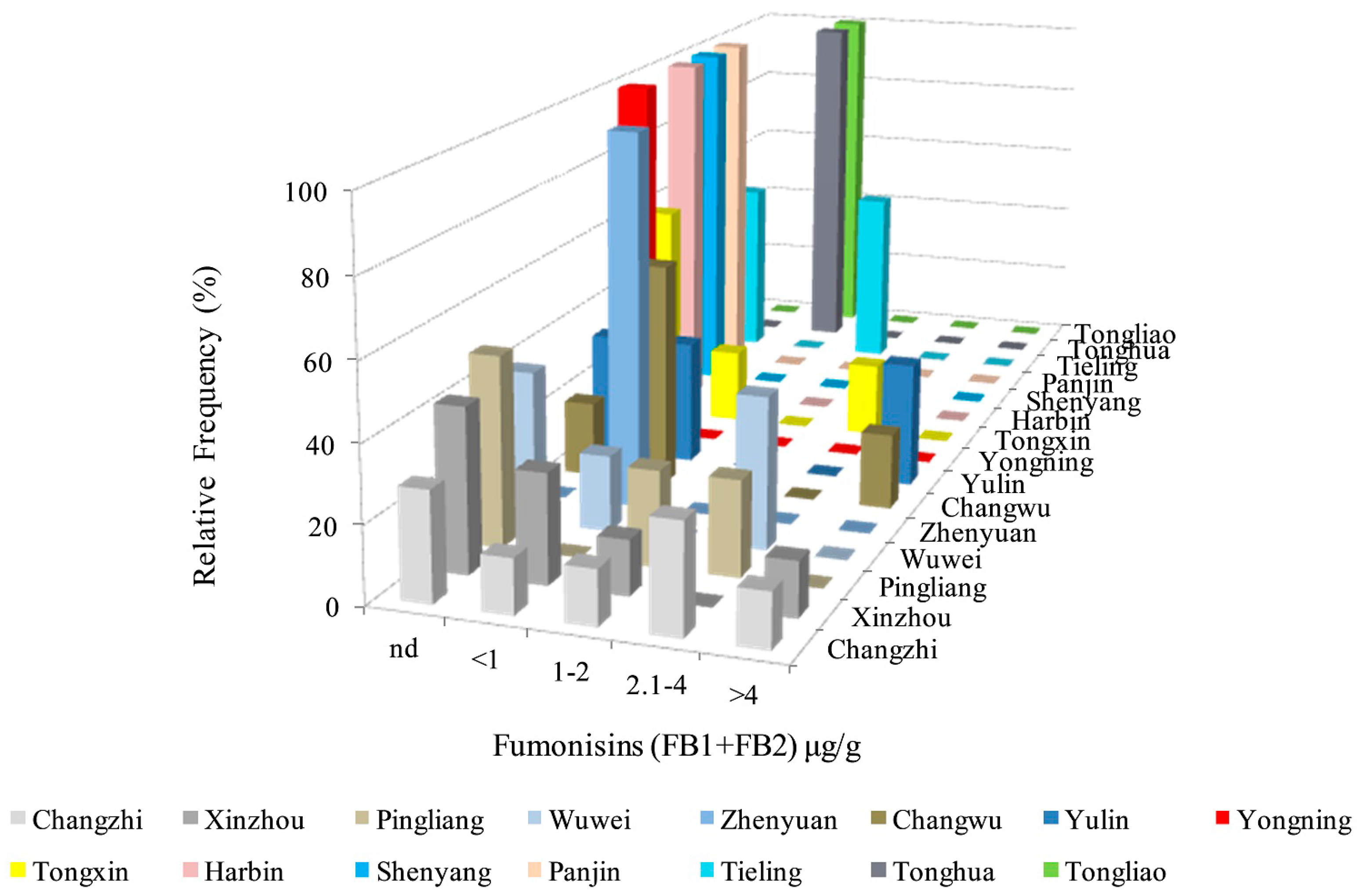
2.6. Analysis of FBs in Kernels
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Fungus Isolation and Identification
4.2. Identification of Pathogenic Fungi
4.3. Detection of Fumonisin FB1 and FB2
4.4. Chemicals and Reagents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, W.J.; He, P.; Jin, J.Y. Effect of potassium on ultrastructure of maize stalk pith and young root and their relation to stalk tot resistance. Agric. Sci. China 2010, 9, 1467–1474. [Google Scholar] [CrossRef]
- Yu, C.J.; Saravanakumar, K.; Xia, H.; Gao, J.X.; Fu, K.H.; Sun, J.N.; Dou, K.; Chen, J. Occurrence and virulence of Fusarium spp. associated with stalk rot of maize in North-East China. Physiol. Mol. Plant Pathol. 2017, 98, 1–8. [Google Scholar] [CrossRef]
- Wang, C.; Yang, Q.; Wang, W.X.; Li, Y.P.; Guo, Y.L.; Zhang, D.F.; Ma, X.N.; Song, W.; Zhao, J.R.; Xu, M.L. A transposon-directed epigenetic change in ZmCCT underlies quantitative resistance to Gibberella stalk rot in maize. New Phytol. 2017, 215, 1503–1515. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.H.; Chen, M.L.; Ma, T.; Gao, H.; Lu, P.A. Investigation on occurrence factors of corn stem base rot in Heilongjiang province in 2014. Mod. Agric. 2015, 6, 68–69. [Google Scholar]
- Wang, L.F.; Xu, G.J.; Zhang, S.L.; Zhang, J.K.; Lu, R.Q.; Li, C.J.; Wang, H.J.; Li, F.Z. Analysis of the resistance to stalk rot and yield loss evaluation of 25 maize varieties. J. Maize Sci. 2015, 23, 12–17. [Google Scholar]
- Li, X.J.; Zhang, G.G.; Wu, M.Q.; Hou, T.R.; Li, N.; Qiu, M. Effect of stem rot disease at late maturity on thousand-grain weight of summer corn. Shandong Agric. Sci. 2015, 47, 93–95. [Google Scholar]
- Wang, Z.Y.; Wang, X.M. Current status and management strategies for corn pets and diseases in China. Plant Prot. 2019, 45, 1–11. [Google Scholar]
- Pronczuk, M.; Pronczuk, S.; Messyasz, M. Pathogenicity of Fusarium spp. contributing to the stalk rot of maize in Poland. Mycotoxin Res. 1991, 7, 97–101. [Google Scholar] [CrossRef]
- Mathew, R.; Singh, K.K.; Paul, B. Pythium campanulatum sp. nov., isolated from the rhizosphere of maize, its taxonomy, ITS region of rDNA, and comparison with related species. FEMS Microbiol. Lett. 2003, 226, 9–14. [Google Scholar] [CrossRef]
- Sinha, S.K.; Prasad, M. Bacterial stalk rot of maize, its symptoms and host-range, Zentralblatt fur Bakteriol. Parasitenkd. Infekt. Hyg. Zweite natur-wissenschaftliche Abt Allg. Landwirtsch. Tech. Mikrobiol. 1997, 132, 81–88. [Google Scholar]
- El-Helaly, A.F.; Abo-El-Dahab, M.K.; El-Goorani, M.A.; Gabr, M.R. Identification of Erwinia sp., causing stalk rot of maize in Egypt. Zent. Bakteriol. Parasitenkd. Infekt. Hyg. Zweite Nat. Abt. Mikrobiol. Landwirtsch. Technol. Umweltschutzes 1978, 133, 680–685. [Google Scholar] [CrossRef]
- Ma, C.Y.; Yao, L.S.; Du, F.L.; Xu, M.L. Research progress on resistance of maize to stem rot. J. Maize Sci. 2018, 10, 131–137. [Google Scholar]
- Zhang, M.Z.; Wang, S.Z.; Wang, Z.Y.; Zhou, T. A preliminary study on the pathogens of corn bacterial wilt in Henan province. J. Henan Agric. Univ. 1988, 22, 135–148. [Google Scholar]
- Andreas, G.; Erich-Christian, O.; Ulrike, S.; Cees, W.; Ineke, V.; Heinz-Wilhelm, D. Biodiversity of Fusarium species causing ear rot of maize in Germany. Cereal Res. Commun. 2008, 36, 617–622. [Google Scholar]
- Qin, Z.H.; Ren, X.; Jiang, K.; Wu, X.F.; Yang, Z.H.; Wang, X.M. Identification of Fusarium species and F. graminearum species complex causing maize ear rot in China. Acta Phytophysiol. Sin. 2014, 41, 589–596. [Google Scholar]
- Ammar, M.; Merfat, A.; Walid, N.; Paul, H.V.; Mohammad, H. Morphological and molecular characterization of Fusarium isolated from maize in Syria. J. Phytopathol. 2013, 161, 452–458. [Google Scholar]
- Rahjoo, V.; Zad, J.; Javan-Nikkhah, A.; Mirzadi, G.A.; Okhovvat, S.M.; Bihamta, M.R.; Razzaghian, J.; Klemsdal, S.S. Morphological and molecular identification of Fusarium isolated from maize ears in Iran. J. Plant Pathol. 2008, 90, 463–468. [Google Scholar]
- Shi, J.; Wang, Z.Y.; He, K.L. Occurrence trend and cause analysis of summer maize diseases and insect pests in Huang-Huai-Hai region. Plant Prot. 2005, 31, 63–65. [Google Scholar]
- Guo, M.K.; Wang, X.M.; He, S.Q.; Liu, Y.G.; Jin, S.L.; Cao, S.Q.; Wei, H.Y. Occurrence of maize kernel rot and corn stalk rot in Gansu in 2009. Plant Prot. 2011, 37, 134–137. [Google Scholar]
- Morales-Rodríguez, I.; de Yañz-Morales, M.J.; Silva-Rojas, H.V.; García-de-los-Santos, G.; Guzman-de-Pena, D.A. Biodiversity of Fusarium species in Mexico associated with ear rot in maize, and their identification using a phylogenetic approach. Mycopathologia 2007, 163, 31–39. [Google Scholar] [CrossRef]
- Chulze, S.N.; Ramirez, M.L.; Torres, A.; Leslie, J.F. Genetic variation in Fusarium section Liseola from No-Till maize in Argentina. Appl. Environ. Microbiol. 2000, 66, 5312–5315. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.W.; Dyer, R.B.; McCormick, S.P.; Kendra, D.F.; Plattner, R.D. Functional demarcation of the Fusarium core trichothecene gene cluster. Fungal Genet. Biol. 2004, 41, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; Proctor, R.H.; McCormick, S.P. Genes, gene clusters, and biosynthesis of trichothecenes and fumonisins in Fusarium. Toxin Rev. 2009, 28, 198–215. [Google Scholar] [CrossRef]
- Yin, S.; Guo, X.; Li, J.; Fan, L.; Hu, H. Fumonisin B1 induces autophagic cell death via activation of ERN1-MAPK8/9/10 pathway in monkey kidney MARC-145 cells. Arch. Toxicol. 2016, 90, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Lino, C.M.; Silva, L.J.G.; Pena, A.L.S.; Silveira, M.I. Determination of fumonisins B1 and B2 in Portuguese maize and maize-based samples by HPLC with fluorescence detection. Anal Bioanal Chem. 2006, 384, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Li, R.; Guo, C.C.; Pang, M.H.; Liu, Y.C.; Dong, J.G. Natural incidence of Fusarium species and fumonisins B1 and B2 associated with maize kernels from nine provinces in China in 2012. Food Addit. Contam. 2015, 32, 503–511. [Google Scholar] [CrossRef]
- Covarelli, L.; Stifano, S.; Beccari, G.; Raggi, L.; Lattanzio, V.M.; Albertini, E. Characterization of Fusarium verticillioides strains isolated from maize in Italy: Fumonisin production, pathogenicity and genetic variability. Food Microbiol. 2012, 31, 17–24. [Google Scholar] [CrossRef]
- Gelderblom, W.C.; Jaskiewicz, K.; Marasas, W.F.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P. Fumonisins-novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1998, 54, 1806–1811. [Google Scholar]
- European Commission. Commission regulation (EC) no. 856/2005 of 6 June 2005 amending regulation (EC) no. 466/2001 as regards Fusarium toxins. Off. J. Eur. Union 2005, 143, 3–8. [Google Scholar]
- Food and Drug Administration USA. Fumonisin levels in human foods and animal feeds. Center for food safety and applied nutrition. Center for veterinary medicine. Fed. Regist. 2001, 66, 218. [Google Scholar]
- Duan, C.X.; Wang, X.M.; Song, F.J.; Sun, S.L.; Zhou, D.N.; Zhu, Z.D. Advance in research on maize resistance to ear rot. Sci. Agric. Sin. 2015, 48, 2152–2164. [Google Scholar]
- Ren, J.P.; Wu, X.L.; Sun, X.H. A study on infection cycle of pathogen of corn ear rot and stalk rot in Jilin Province. Maize Sci. 1995, 10, 25–28. [Google Scholar]
- Chen, J. Status and perspective on research of ear rot and stalk rot in maize. J. Shenyang Agric. Univ. 2000, 31, 393–401. [Google Scholar]
- Zou, Q.D.; Xu, Y.; Wang, L.; Chen, J. Relationship of infection cycle between ear rot and stalk rot in maize caused by Fusarium. J. Shenyang Agric. Univ. 2000, 31, 487–489. [Google Scholar]
- Rahayu, D.; Rahayu, W.P.; Lioe, H.N. The effect of temperature and humidity on the growth of Fusarium verticillioides BIO 957 and fumonisin B1 productions. Agritech 2015, 35, 156–163. [Google Scholar] [CrossRef]
- Sewarm, V.; Mshicileli, N.; Shephard, G.S.; Vismer, H.F.; Rheeder, J.P.; Lee, Y.W.; Leslie, J.F.; Marasas, W.F. Production of fumonisin B and C analogues by several Fusarium species. J. Agric. Food Chem. 2005, 53, 4861–4866. [Google Scholar] [CrossRef]
- Cook, R.J. The incidence of stalk rot (Fusarium spp.) on maize hybrids and its effect on yield of maize in Britain. Ann. Appl. Biol. 1978, 88, 23–30. [Google Scholar] [CrossRef]
- Ledencan, T.; Sumic, D.; Brkic, I.; Jambrovic, A.; Zdunic, Z. Resistance of maize inbreds and their hybrids to Fusarium stalk rot. Czech J. Genet. Plant Breed 2003, 39, 15–20. [Google Scholar] [CrossRef]
- Jiang, Y.Y.; Du, J.; Chi, Y.P.; Liu, X.Y.; Liu, H.W.; Li, W.L. Occurrence and effective control of maize stalk rot. J. Northeast Agric. Sci. 2018, 43, 24–27. [Google Scholar]
- Li, Y.Y.; Feng, G.; Wang, L.; Wu, Y.J. Yield comparison of Zhengdan 958 and Xianyu 335 in different regions. China Seed Ind. 2009, 8, 36–37. [Google Scholar]
- Mulè, G.; Susca, A.; Stea, G.; Moretti, A. A species-specific PCR assay based on the calmodulin partial gene for identifcation of Fusarium verticillioides, F. proliferatum and F. subglutinans. Eur. J. Plant Pathol. 2004, 110, 495–502. [Google Scholar] [CrossRef]
- Nicholson, P.; Simpson, D.R.; Weston, G.; Rezanoor, H.N.; Lees, A.K.; Parry, D.W.; Joyce, D. Detection and quantification of Fusarium culmorum and Fusarium graminearumin cereals using PCR assays. Physiol. Mol. Plant Pathol. 1998, 53, 17–37. [Google Scholar] [CrossRef]
- Mishra, P.K.; Fox, R.T.V.; Culham, A. Development of a PCR-based assay for rapid and reliable identification of pathogenic Fusariam. FEMS Microbiol. Lett. 2003, 218, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.Z.; Lu, X.H.; Tao, B.; Pang, M.H.; Liu, Y.C.; Dong, J.G. Natural occurrence of fumonisins B1 and B2 in corn from three main production provinces in China. J. Food Prot. 2011, 74, 1374–1378. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.C.; Zhu, W.F.; Fu, M.; Pang, M.H.; Liu, Y.C.; Dong, J.G. Occurrence of Fusarium species and fumonisins associated with maize kernels from Gansu province. Plant Prot. 2015, 42, 942–948. [Google Scholar]
- Liu, Y.X.; Jiang, Y.; Li, R.J.; Pang, M.H.; Liu, Y.C.; Dong, J.G. Natural occurrence of fumonisins B1 and B2 in maize from eight provinces of China in 2014. Food Addit. Contam. Part B Surveill. 2017, 10, 113–117. [Google Scholar] [CrossRef] [PubMed]
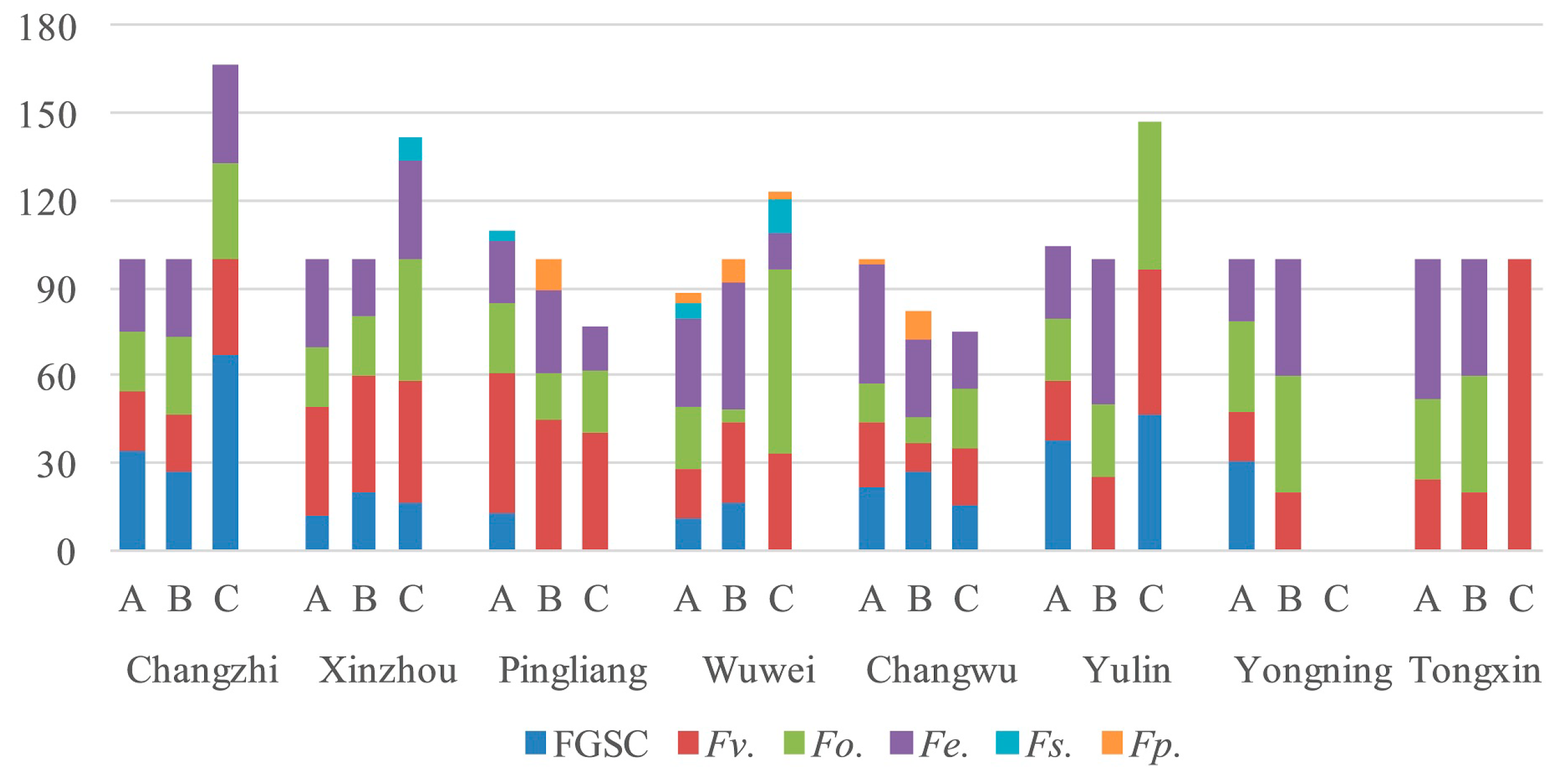
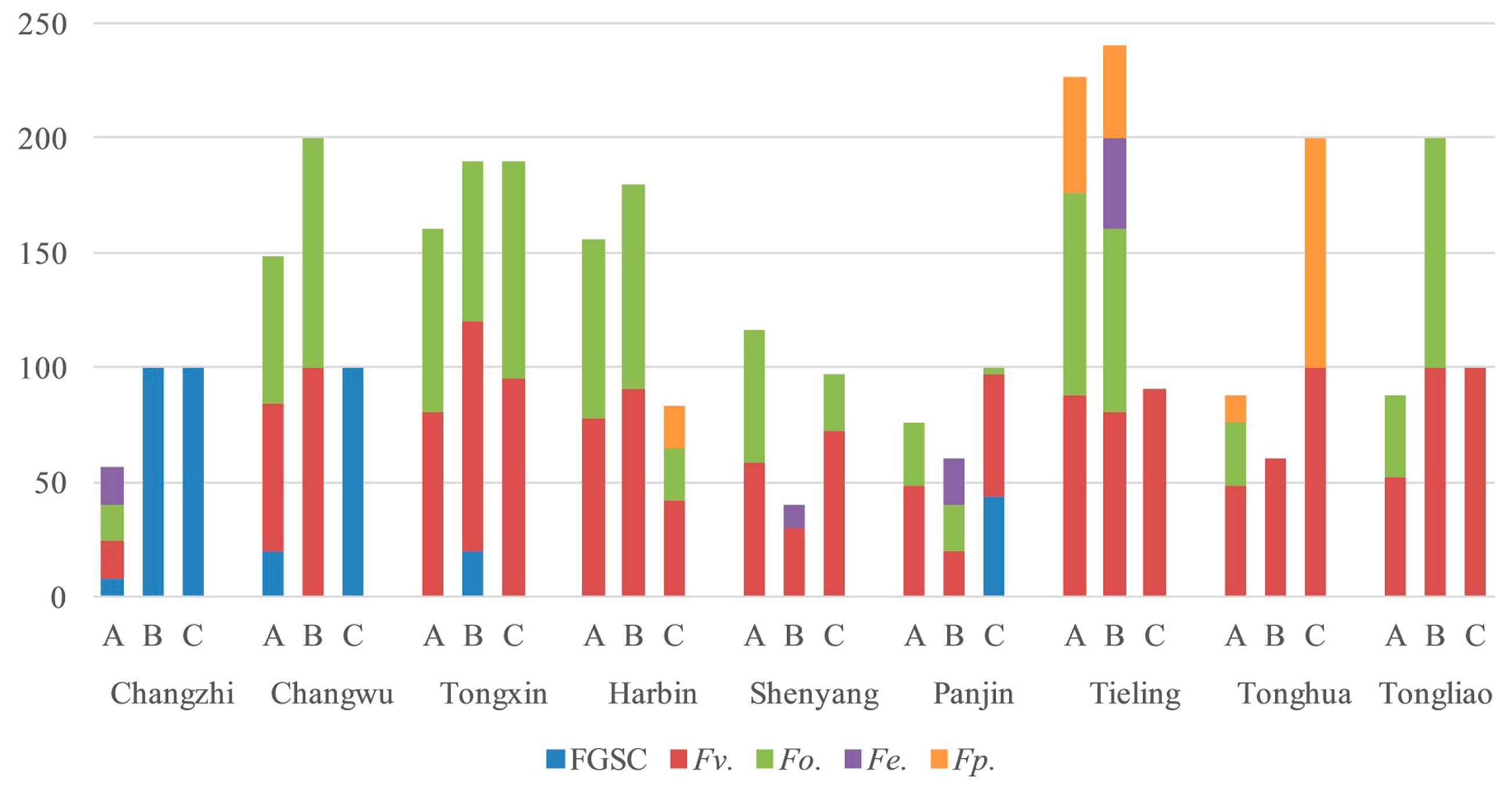
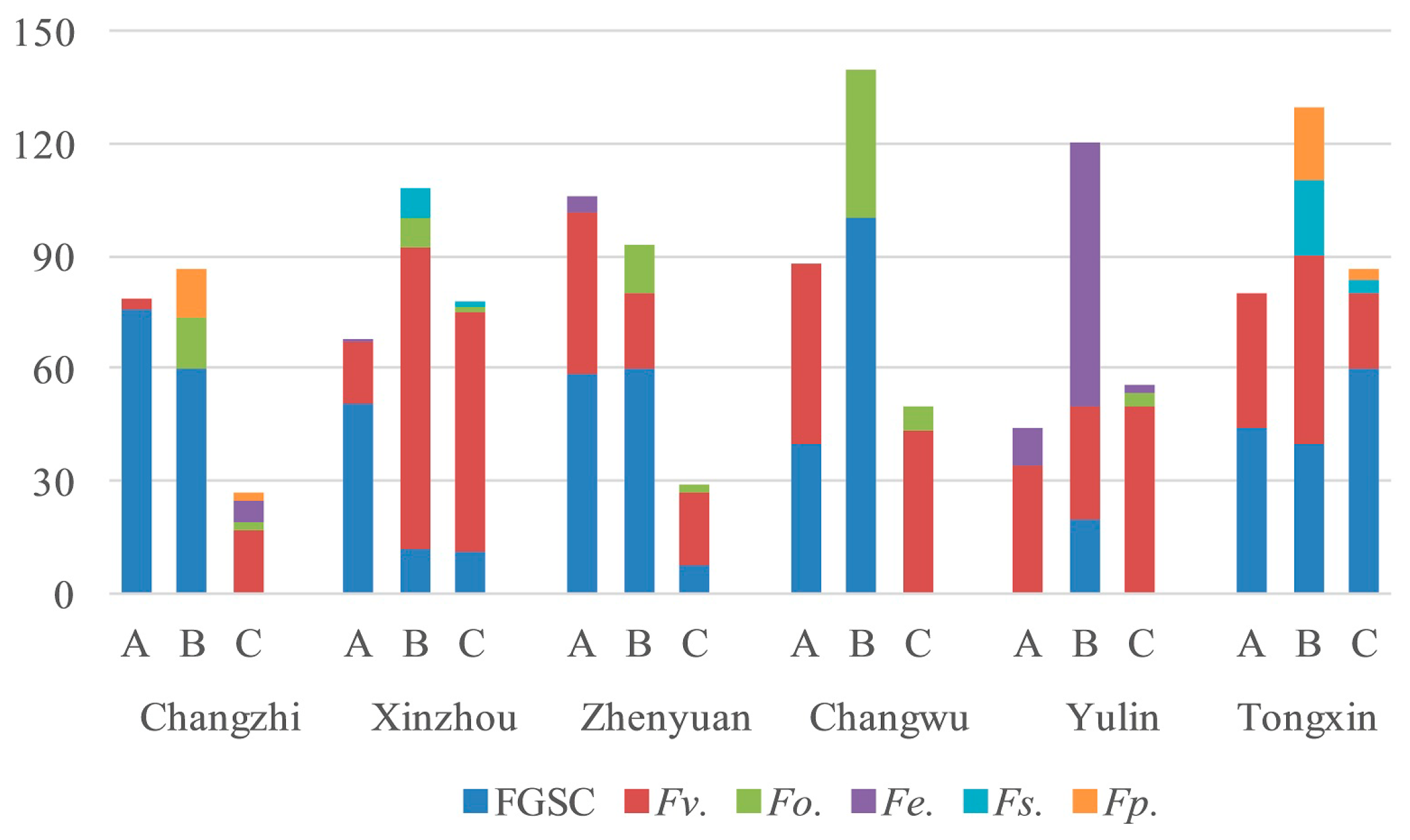

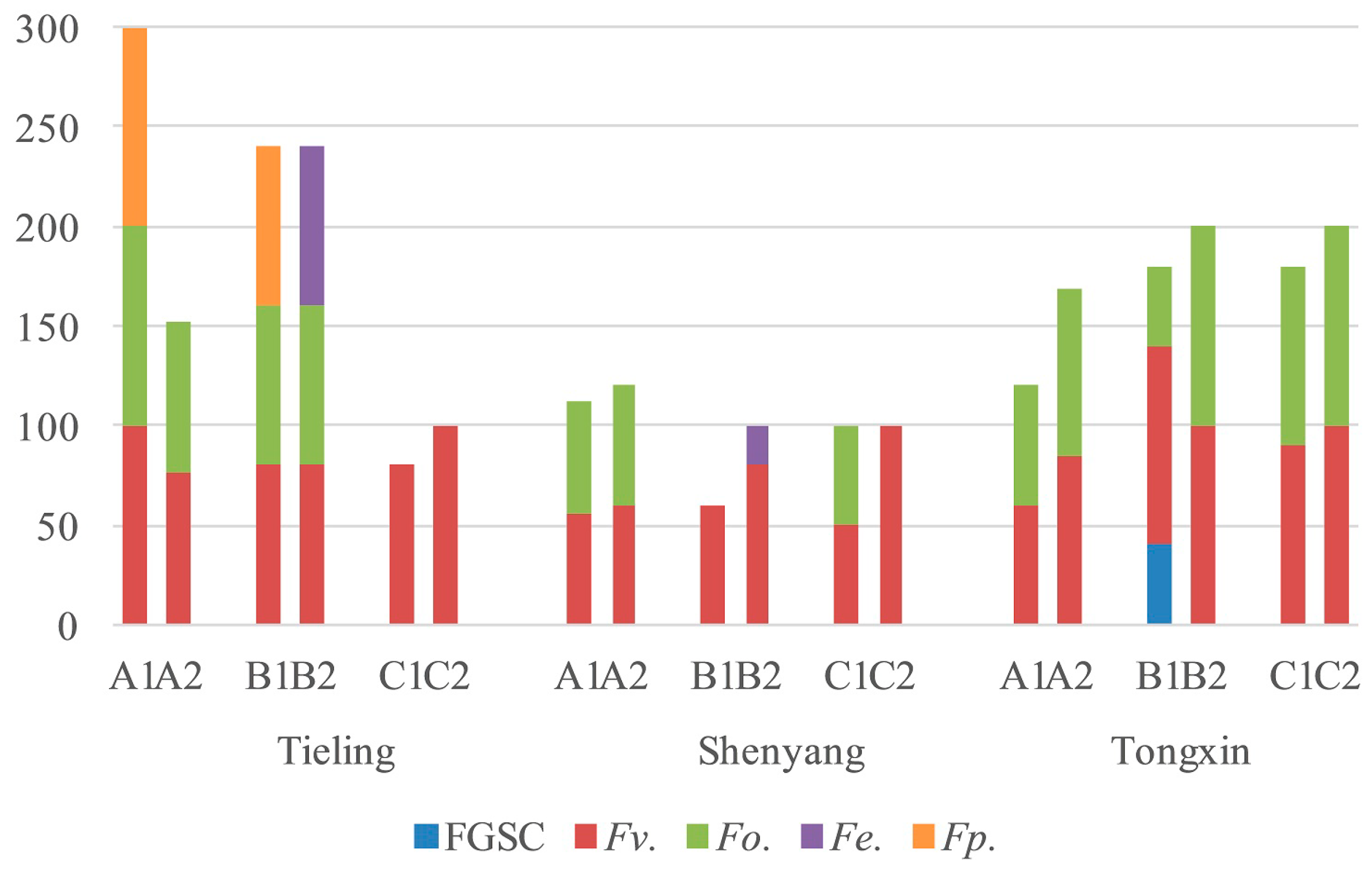
| Fusarium spp. | Number of Strains Isolated | Total | ||
|---|---|---|---|---|
| Corn Kernels | Ear Stems | Stalks | ||
| F. verticillioides | 668 | 105 | 428 | 1201 |
| F. oxysporum | 318 | 59 | 291 | 668 |
| FGSC | 240 | 52 | 282 | 574 |
| F. equiseti | 123 | 44 | 151 | 318 |
| F. proliferatum | 49 | 13 | 33 | 95 |
| F. subglutinans | 27 | 4 | 9 | 40 |
| Region | No. of Total Samples | Isolation Frequency of Fusarium spp. | |||||
|---|---|---|---|---|---|---|---|
| F. verticillioides | F. oxysporum | F. proliferatum | FGSC | F. equiseti | F. subglutinans | ||
| Wuwei | 5 | 25.3% | 40.3% | 3.3% | 5.7% | 21.3% | 8.0% |
| Pingliang | 4 | 42.1% | 21.7% | 0.8% | 5.0% | 17.9% | 1.2% |
| Changzhi | 3 | 26.7% | 27.2% | 0.0% | 49.4% | 28.9% | 0.0% |
| Changwu | 3 | 25.0% | 21.7% | 1.1% | 21.7% | 32.2% | 0.0% |
| Xinzhou | 2 | 39.2% | 30.8% | 0.0% | 15.0% | 30.8% | 4.2% |
| Yulin | 1 | 35.0% | 35.0% | 0.0% | 38.3% | 11.7% | 0.0% |
| Yongning | 1 | 8.3% | 15.0% | 0.0% | 11.7% | 11.7% | 0.0% |
| Tongxin | 1 | 61.7% | 15.0% | 0.0% | 0.0% | 23.3% | 0.0% |
| Region | No. of Total Samples | Isolation Frequency of Fusarium spp. | ||||
|---|---|---|---|---|---|---|
| F. verticillioides | F. oxysporum | F. proliferatum | FGSC | F. equiseti | ||
| Harbin | 2 | 60.8% | 51.7% | 9.2% | 0.0% | 0.0% |
| Tieling | 2 | 88.3% | 43.3% | 24.2% | 0.0% | 3.3% |
| Shenyang | 2 | 67.5% | 36.7% | 0.0% | 0.0% | 0.8% |
| Tongxin | 2 | 85.8% | 83.3% | 0.0% | 1.7% | 0.0% |
| Changwu | 1 | 35.0% | 35.0% | 0.0% | 58.3% | 0.0% |
| Changzhi | 1 | 6.7% | 6.7% | 0.0% | 61.7% | 10.0% |
| Panjin | 1 | 48.3% | 15.0% | 0.0% | 21.7% | 8.3% |
| Tonghua | 1 | 75.0% | 11.7% | 55.0% | 0.0% | 0.0% |
| Tongliao | 1 | 80.0% | 23.3% | 0.0% | 0.0% | 0.0% |
| Region | No. of Total Samples | Isolation Frequency of Fusarium spp. | |||||
|---|---|---|---|---|---|---|---|
| F. verticillioides | F. oxysporum | F. proliferatum | FGSC | F. equiseti | F. subglutinans | ||
| Xinzhou | 5 | 45.7% | 1.3% | 0.0% | 28.0% | 0.0% | 1.3% |
| Zhenyuan | 3 | 26.7% | 2.2% | 0.0% | 30.0% | 1.7% | 0.0% |
| Changzhi | 3 | 9.4% | 2.2% | 2.2% | 38.3% | 2.8% | 0.0% |
| Yulin | 2 | 41.7% | 1.7% | 0.0% | 1.7% | 11.7% | 0.0% |
| Tongxin | 2 | 29.2% | 0.0% | 3.3% | 51.7% | 0.0% | 3.3% |
| Changwu | 1 | 41.7% | 6.7% | 0.0% | 25.0% | 0.0% | 0.0% |
| Region | No. of Total Samples | Fumonisins | No. of Positive Samples (%) | Range (μg/g) | Mean (μg/g) |
|---|---|---|---|---|---|
| Changzhi | 3 | FB1 | 1/3(33.3) | nd–0.3 | 0.3 |
| FB2 | 3/3(100.0) | 1.7–4.5 | 2.8 | ||
| FB1 + FB2 | 3/3(100.0) | 1.7–4.5 | 2.9 | ||
| Xinzhou | 2 | FB1 | 0/2(0.0) | nd | - |
| FB2 | 1/2(50.0) | nd–1.7 | 1.7 | ||
| FB1 + FB2 | 1/2(50.0) | nd–1.73 | 1.7 | ||
| Pingliang | 4 | FB1 | 0/4(0.0) | nd | - |
| FB2 | 2/4(50.0) | nd–2.9 | 2.1 | ||
| FB1 + FB2 | 2/4(50.0) | nd–2.9 | 2.1 | ||
| Wuwei | 5 | FB1 | 0/5(0.0) | nd | - |
| FB2 | 3/5 (60.0) | nd–3.3 | 2.1 | ||
| FB1 + FB2 | 3/5 (60.0) | nd–3.3 | 2.1 | ||
| Changwu | 3 | FB1 | 2/3(66.7) | nd–1.2 | 0.8 |
| FB2 | 1/3(33.3) | nd–4.3 | 4.3 | ||
| FB1 + FB2 | 2/3(66.7) | nd–5.4 | 2.9 | ||
| Yulin | 1 | FB1 | 1/1(100.0) | 0.3 | 0.3 |
| FB2 | 0/1(0.0) | nd | - | ||
| FB1 + FB2 | 1/1(100.0) | 0.3 | 0.3 | ||
| Tongxin | 1 | FB1 | 0/1(0.0) | nd | - |
| FB2 | 1/1(100.0) | 2.5 | 2.5 | ||
| FB1 + FB2 | 1/1(100.0) | 2.5 | 2.5 | ||
| Total | 19 | FB1 | 4/19(21.1) | nd–1.2 | 0.6 |
| FB2 | 11/19(57.9) | nd–4.5 | 2.5 | ||
| FB1 + FB2 | 13/19(68.4) | nd–5.4 | 2.3 |
| Region | No. of Total Samples | Fumonisins | No. of Positive Samples (%) | Range (μg/g) | Mean (μg/g) |
|---|---|---|---|---|---|
| Changwu | 1 | FB1 | 1/1(100.0) | 0.2 | 0.2 |
| FB2 | 0/1(0.0) | nd | - | ||
| FB1 + FB2 | 1/1(100.0) | 0.2 | 0.2 | ||
| Changzhi | 1 | FB1 | 1/1(100.0) | 0.1 | 0.1 |
| FB2 | 0/1(0.0) | nd | - | ||
| FB1 + FB2 | 1/1(100.0) | 0.1 | 0.1 | ||
| Tieling | 2 | FB1 | 1/2(50.0) | nd–0.5 | 0.5 |
| FB2 | 1/2(50.0) | nd–0.5 | 0.5 | ||
| FB1 + FB2 | 1/2(50.0) | nd–1.1 | 1.1 | ||
| Tonghua | 1 | FB1 | 1/1(100.0) | 0.1 | 0.1 |
| FB2 | 0/1(0.0) | nd | - | ||
| FB1 + FB2 | 1/1(100.0) | 0.1 | 0.1 | ||
| Tongliao | 1 | FB1 | 1/1(100.0) | 0.1 | 0.1 |
| FB2 | 0/1(0.0) | nd | - | ||
| FB1 + FB2 | 1/1(100.0) | 0.1 | 0.1 | ||
| Total | 6 | FB1 | 5/6(83.3) | nd–0.5 | 0.2 |
| FB2 | 1/6(16.7) | 0.5 | 0.5 | ||
| FB1 + FB2 | 5/6(83.3) | nd–1.1 | 0.3 |
| Region | No. of Total Samples | Fumonisins | No.of Positive Samples (%) | Range (μg/g) | Mean (μg/g) |
|---|---|---|---|---|---|
| Yulin | 2 | FB1 | 1/2(50.0) | nd–5.4 | 5.4 |
| FB2 | 1/2(50.0) | nd–4.7 | 4.7 | ||
| FB1 + FB2 | 1/2(50.0) | nd–10.1 | 10.1 | ||
| Changwu | 1 | FB1 | 1/1(100.0) | 0.3 | 0.3 |
| FB2 | 1/1(100.0) | 0.2 | 0.2 | ||
| FB1 + FB2 | 1/1(100.0) | 0.5 | 0.5 | ||
| Changzhi | 3 | FB1 | 1/3(33.3) | 2.7 | 2.7 |
| FB2 | 1/3(33.3) | 0.7 | 0.7 | ||
| FB1 + FB2 | 1/3(33.3) | 3.4 | 3.4 | ||
| Xinzhou | 5 | FB1 | 2/5(40.0) | nd–3.8 | 1.9 |
| FB2 | 2/5 (40.0) | nd–2.6 | 1.4 | ||
| FB1 + FB2 | 3/5 (60.0) | nd–6.5 | 2.2 | ||
| Tongxin | 2 | FB1 | 1/2(50.0) | nd–0.1 | 0.1 |
| FB2 | 0/2(0.0) | nd | - | ||
| FB1 + FB2 | 1/2(50.0) | nd–0.1 | 0.1 | ||
| Total | 13 | FB1 | 9/13(69.2) | nd–5.4 | 1.4 |
| FB2 | 6/13(46.2) | nd–4.7 | 1.4 | ||
| FB1 + FB2 | 10/13(76.9) | nd–10.1 | 2.1 |
| Fungi | Primer | Sequences (5′-3′) | Product Size (bp) | Tm (°C) | Ref. |
|---|---|---|---|---|---|
| F. verticillioides | VER1 VER2 | CTTCCTGCGATGTTTCTCC AATTGGCCATTGGTATTATATATCTA | 578 | 56 | [41] |
| F. proliferatum | PRO1 PRO2 | CTTTCCGCCAAGTTTCTTC TGTCAGTAACTCGACGTTGTTG | 585 | 56 | [41] |
| F. subglutinans | SUB1 SUB2 | CTGTCGCTAACCTCTTTATCCA CAGTATGGACGTTGGTATTATATCTAA | 631 | 56 | [41] |
| FGSC | Fg16NF Fg16NR | ACAGATGACAAGATTCAGGCACA TTCTTTGACATCTGTTCAACCCA | 280 | 57 | [42] |
| F. oxysporum | FoF1 FoF2 | ACATACCACTTGTTGCCTCG CGCCAATCAATTTGAGGAACG | 340 | 58 | [43] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Qu, Q.; Cao, Z.; Guo, Z.; Jia, H.; Liu, N.; Wang, Y.; Dong, J. The Relationship Analysis on Corn Stalk Rot and Ear Rot According to Fusarium Species and Fumonisin Contamination in Kernels. Toxins 2019, 11, 320. https://doi.org/10.3390/toxins11060320
Li L, Qu Q, Cao Z, Guo Z, Jia H, Liu N, Wang Y, Dong J. The Relationship Analysis on Corn Stalk Rot and Ear Rot According to Fusarium Species and Fumonisin Contamination in Kernels. Toxins. 2019; 11(6):320. https://doi.org/10.3390/toxins11060320
Chicago/Turabian StyleLi, Lina, Qing Qu, Zhiyan Cao, Zhengyu Guo, Hui Jia, Ning Liu, Yanhui Wang, and Jingao Dong. 2019. "The Relationship Analysis on Corn Stalk Rot and Ear Rot According to Fusarium Species and Fumonisin Contamination in Kernels" Toxins 11, no. 6: 320. https://doi.org/10.3390/toxins11060320
APA StyleLi, L., Qu, Q., Cao, Z., Guo, Z., Jia, H., Liu, N., Wang, Y., & Dong, J. (2019). The Relationship Analysis on Corn Stalk Rot and Ear Rot According to Fusarium Species and Fumonisin Contamination in Kernels. Toxins, 11(6), 320. https://doi.org/10.3390/toxins11060320





