Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae
Abstract
1. Introduction
2. Results
2.1. LC-HR-QTOF-MS Analyses
2.2. MC-LR Quantification by LC-QqQ-MS/MS
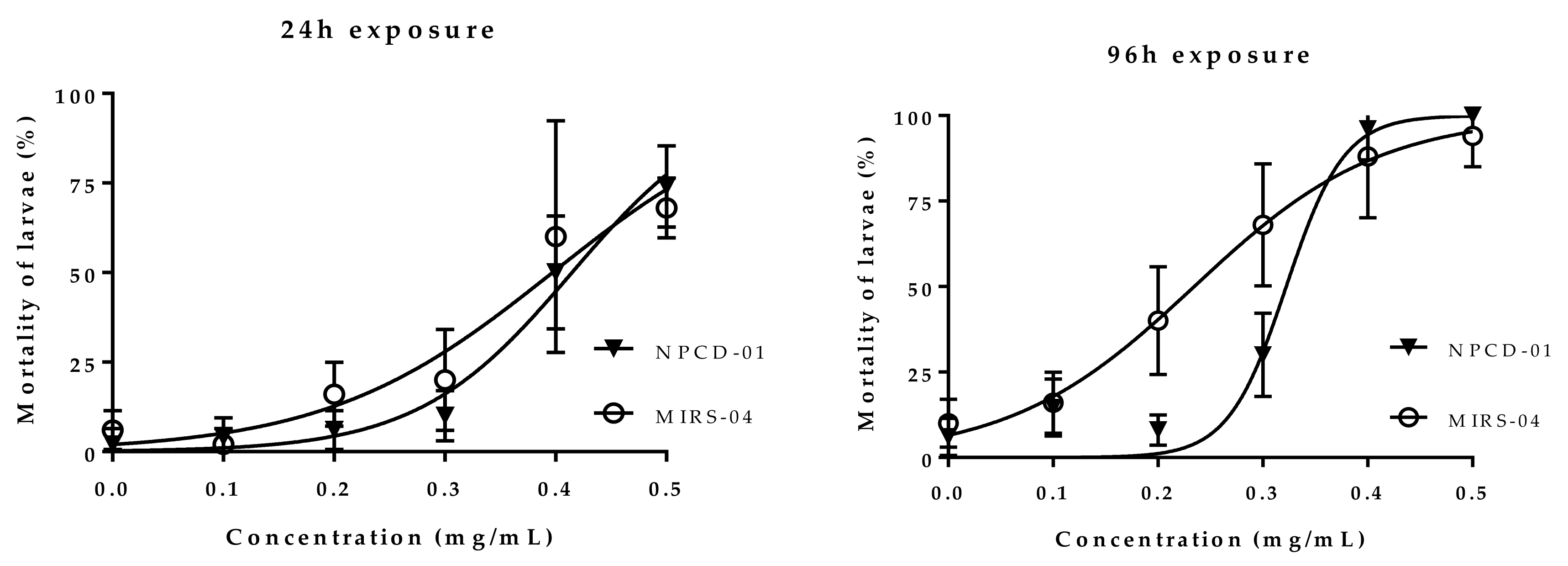
2.3. Acute Toxicity
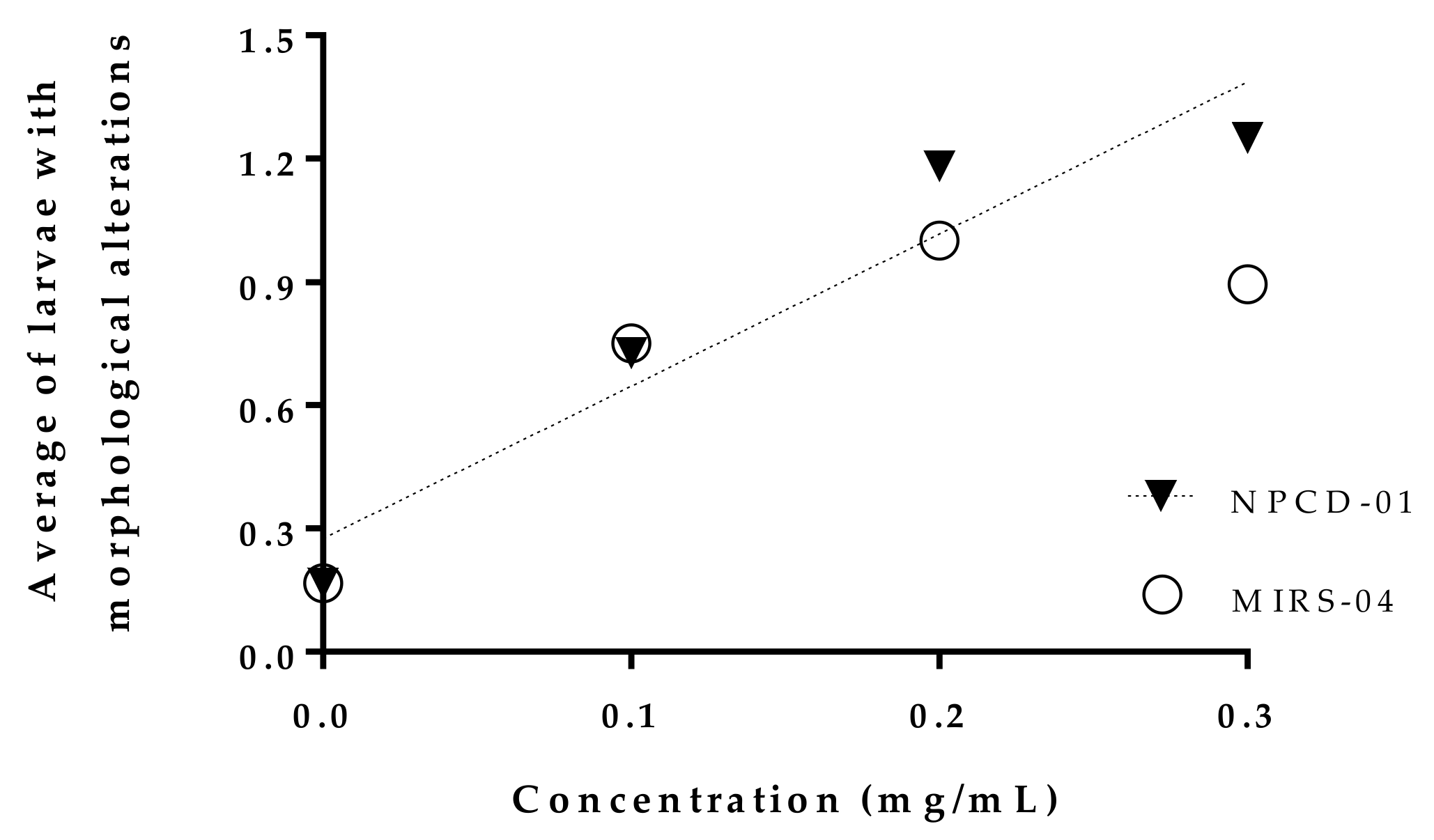
2.4. Morphological Changes
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Cultivation of Microcystis Strains
5.2. Extraction Procedure
5.3. LC-HR-QTOF-MS—Analyses
5.4. Molecular Networking
5.5. Liquid Chromatography–Tandem Mass Spectrometry LC-QqQ-MS/MS Analyses
5.6. Samples Preparation for the Toxicity Tests
5.7. Astyanax Altiparanae Larvae Acquisition
5.8. Acute Effect in Larvae of Astyanax Altiparanae
5.9. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anderson, D.M.; Glibert, P.M.; Burkholder, J.M. Harmful algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 2002, 25, 704–726. [Google Scholar] [CrossRef]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the Chemistry, Toxicology and Genetics of the Cyanobacterial Toxins, Microcystin, Nodularin, Saxitoxin and Cylindrospermopsin. Mar. Drugs 2010, 8, 1650–1680. [Google Scholar] [CrossRef]
- Jaiswal, P.; Singh, P.K.; Prasanna, R. Cyanobacterial bioactive molecules—An overview of their toxic properties. Can. J. Microbiol. 2008, 54, 701–717. [Google Scholar] [CrossRef] [PubMed]
- Leflaive, J.; Ten-Hage, L. Algal and cyanobacterial secondary metabolites in freshwaters: A comparison of allelopathic compounds and toxins. Freshw. Biol. 2007, 52, 199–214. [Google Scholar] [CrossRef]
- Lifshits, M.; Carmeli, S. Metabolites of Microcystis aeruginosa Bloom Material from Lake Kinneret, Israel. J. Nat. Prod. 2012, 75, 209–219. [Google Scholar] [CrossRef]
- Oren, A.; Gunde-Cimerman, N. Mycosporines and mycosporine-like amino acids: UV protectants or multipurpose secondary metabolites? FEMS Microbiol. Lett. 2007, 269, 1–10. [Google Scholar] [CrossRef]
- Gademann, K.; Portmann, C. Secondary metabolites from cyanobacteria: Complex structures and powerful bioactivities. Curr. Org. Chem. 2008, 12, 326–341. [Google Scholar] [CrossRef]
- Volk, R.-B.; Furkert, F.H. Antialgal, antibacterial and antifungal activity of two metabolites produced and excreted by cyanobacteria during growth. Microbiol. Res. 2006, 161, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Legrand, C.; Rengefors, K.; Fistarol, G.O.; Graneli, E. Allelopathy in phytoplankton-biochemical, ecological and evolutionary aspects. Phycologia 2003, 42, 406–419. [Google Scholar] [CrossRef]
- Jaja-Chimedza, A.; Saez, C.; Sanchez, K.; Gantar, M.; Berry, J.P. Identification of teratogenic polymethoxy-1-alkenes from Cylindrospermopsis raciborskii, and taxonomically diverse freshwater cyanobacteria and green algae. Harmful Algae 2015, 49, 156–161. [Google Scholar] [CrossRef]
- Fischer, W.J.; Hitzfeld, B.C.; Tencalla, F.; Eriksson, J.E.; Mikhailov, A.; Dietrich, D.R. Microcystin-LR toxicodynamics, induced pathology, and immunohistochemical localization in livers of blue-green algae exposed rainbow trout (Oncorhynchus mykiss). Toxicol. Sci. 2000, 54, 365–373. [Google Scholar] [CrossRef]
- Harada, K.-I. Production of secondary metabolites by freshwater cyanobacteria. Chem. Pharm. Bull. 2004, 52, 889–899. [Google Scholar] [CrossRef]
- Harada, K.I.; Nakano, T.; Fujii, K.; Shirai, M. Comprehensive analysis system using liquid chromatography-mass spectrometry for the biosynthetic study of peptides produced by cyanobacteria. J. Chromatogr. A 2004, 1033, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Namikoshi, M.; Sun, F.; Choi, B.W.; Rinehart, K.L.; Carmichael, W.W.; Evans, W.R.; Beasley, V.R. Seven More Microcystins from Homer Lake Cells: Application of the General Method for Structure Assignment of Peptides Containing α,β-Dehydroamino Acid Unit(s). J. Org. Chem. 1995, 60, 3671–3679. [Google Scholar] [CrossRef]
- Lombardo, M.; Cristina, R.; Pinto, F.; Maria, S.; Vieira, J.; Yukio Honda, R.; Pimenta, A.; Bemquerer, M.; Carvalho, L.; Kiyota, S. Isolation and structural characterization of microcystin-LR and three minor oligopeptides simultaneously produced by Radiocystis feernandoi (Chroococcales, Cyanobacteriae): A Brazilian toxic cyanobacterium. Toxicon 2006, 47, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, T.; Szafraniec, L.; Hunt, D.F.; Shabanowitz, J.; Yates, J.R.; Hauer, C.R.; Carmichael, W.W.; Skulberg, O.; Codd, G.A.; Missler, S. Structural characterization of toxic cyclic peptides from blue-green algae by tandem mass spectrometry. Proc. Natl. Acad. Sci. USA 1989, 86, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Neumann, U.; Forchert, A.; Flury, T.; Weckesser, J. Microginin FR1, a linear peptide from a water bloom of Microcystis species. FEMS Microbiol. Lett. 1997, 153, 475–478. [Google Scholar] [CrossRef]
- Carneiro, R.L.; Dörr, F.F.A.; Dörr, F.F.A.; Bortoli, S.; Delherbe, N.; Vásquez, M.; Pinto, E. Co-occurrence of microcystin and microginin congeners in Brazilian strains of Microcystis sp. FEMS Microbiol. Ecol. 2012, 82, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Okino, T.; Matsuda, H.; Murakami, M.; Yamaguchi, K. Microginin, an angiotensin-converting enzyme inhibitor from the blue-green alga Microcystis aeruginosa. Tetrahedron Lett. 1993, 34, 501–504. [Google Scholar] [CrossRef]
- Welker, M.; Maršálek, B.; Šejnohová, L.; von Döhren, H. Detection and identification of oligopeptides in Microcystis (cyanobacteria) colonies: Toward an understanding of metabolic diversity. Peptides 2006, 27, 2090–2103. [Google Scholar] [CrossRef]
- Silva-Stenico, M.E.; Silva, C.S.P.; Lorenzi, A.S.; Shishido, T.K.; Etchegaray, A.; Lira, S.P.; Moraes, L.A.B.; Fiore, M.F. Non-ribosomal peptides produced by Brazilian cyanobacterial isolates with antimicrobial activity. Microbiol. Res. 2011, 166, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Sandvik, M.; Nonga, H.E.; Rundberget, T.; Wilkins, A.L.; Rise, F.; Ballot, A. Identification of microcystins in a Lake Victoria cyanobacterial bloom using LC–MS with thiol derivatization. Toxicon 2013, 70, 21–31. [Google Scholar] [CrossRef]
- Reshef, V.; Carmeli, S. Protease inhibitors from a water bloom of the cyanobacterium Microcystis aeruginosa. Tetrahedron 2001, 57, 2885–2894. [Google Scholar] [CrossRef]
- Dörr, F.A.; Pinto, E.; Soares, R.M.; de Oliveira e Azevedo, S.M.F. Microcystins in South American aquatic ecosystems: Occurrence, toxicity and toxicological assays. Toxicon 2010, 56, 1247–1256. [Google Scholar] [CrossRef]
- Bittencourt-Oliveira, M.C.; Oliveira, M.C.; Pinto, E. Diversity of microcystin-producing genotypes in Brazilian strains of Microcystis (Cyanobacteria). Braz. J. Biol. 2011, 71, 209–216. [Google Scholar] [CrossRef]
- Dos Anjos, F.M.; do Carmo Bittencourt-Oliveira, M.; Zajac, M.P.; Hiller, S.; Christian, B.; Erler, K.; Luckas, B.; Pinto, E. Detection of harmful cyanobacteria and their toxins by both PCR amplification and LC-MS during a bloom event. Toxicon 2006, 48, 239–245. [Google Scholar] [CrossRef]
- Paiva, F.C.R.; Ferreira, G.M.; Trossini, G.H.G.; Pinto, E. Identification, In Vitro Testing and Molecular Docking Studies of Microginins’ Mechanism of Angiotensin-Converting Enzyme Inhibition. Molecules 2017, 22, 1884. [Google Scholar] [CrossRef]
- Kind, T.; Tsugawa, H.; Cajka, T.; Ma, Y.; Lai, Z.; Mehta, S.S.; Wohlgemuth, G.; Barupal, D.K.; Showalter, M.R.; Arita, M.; et al. Identification of small molecules using accurate mass MS/MS search. Mass Spectrom. Rev. 2018, 37, 513–532. [Google Scholar] [CrossRef]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.-T.D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat. Biotechnol. 2016, 34, 828. [Google Scholar] [CrossRef]
- Ding, C.; Pang, L.; Liang, Z.-X.; Goh, K.; Glukhov, E.; Gerwick, W.; Tan, L. MS/MS-Based Molecular Networking Approach for the Detection of Aplysiatoxin-Related Compounds in Environmental Marine Cyanobacteria. Mar. Drugs 2018, 16, 505. [Google Scholar] [CrossRef] [PubMed]
- Laughinghouse, H.D.; Prá, D.; Silva-Stenico, M.E.; Rieger, A.; Frescura, V.D.S.; Fiore, M.F.; Tedesco, S.B. Biomonitoring genotoxicity and cytotoxicity of Microcystis aeruginosa (Chroococcales, Cyanobacteria) using the Allium cepa test. Sci. Total Environ. 2012, 432, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.-L.; Utsumi, M. An overview of the accumulation of microcystins in aquatic ecosystems. J. Environ. Manag. 2018, 213, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; Dhanji-Rapkova, M.; O’Neill, A.; Coates, L.; Lewis, A.; Lewis, K. Analysis of Microcystins in Cyanobacterial Blooms from Freshwater Bodies in England. Toxins 2018, 10, 39. [Google Scholar] [CrossRef]
- Preece, E.P.; Hardy, F.J.; Moore, B.C.; Bryan, M. A review of microcystin detections in Estuarine and Marine waters: Environmental implications and human health risk. Harmful Algae 2017, 61, 31–45. [Google Scholar] [CrossRef]
- Zafrir, E.; Carmeli, S. Eight novel serine protease inhibitors from a water bloom of the cyanobacterium Microcystis sp. Tetrahedron 2010, 66, 9194–9202. [Google Scholar] [CrossRef]
- Sanz, M.; Andreote, A.P.D.; Fiore, M.F.; Dörr, F.A.; Pinto, E. Structural characterization of new peptide variants produced by cyanobacteria from the Brazilian Atlantic coastal Forest using liquid chromatography coupled to quadrupole time-of-flight tandem mass spectrometry. Mar. Drugs 2015, 13, 3892–3919. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Pietsch, C.; Wiegand, C.; Amé, M.V.; Nicklisch, A.; Wunderlin, D.; Pflugmacher, S. The effects of a cyanobacterial crude extract on different aquatic organisms: Evidence for cyanobacterial toxin modulating factors. Environ. Toxicol. 2001, 16, 535–542. [Google Scholar] [CrossRef]
- Davis, T.; Berry, D.; Boyer, G.; Gobler, C. The Effects of Temperature and Nutrients on the Growth and Dynamics of Toxic and Non-Toxic Strains of Microcystis during Cyanobacteria Blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Janssen, E.M.-L. Cyanobacterial peptides beyond microcystins—A review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Pancrace, C.; Ishida, K.; Briand, E.; Pichi, D.G.; Weiz, A.R.; Guljamow, A.; Scalvenzi, T.; Sassoon, N.; Hertweck, C.; Dittmann, E.; et al. Unique Biosynthetic Pathway in Bloom-Forming Cyanobacterial Genus Microcystis Jointly Assembles Cytotoxic Aeruginoguanidines and Microguanidines. ACS Chem. Biol. 2019, 14, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Sharif, D.I.; Gallon, J.; Smith, C.J.; Dudley, E. Quorum sensing in Cyanobacteria: N-octanoyl-homoserine lactone release and response, by the epilithic colonial cyanobacterium Gloeothece PCC6909. ISME J. 2008, 2, 1171. [Google Scholar] [CrossRef]
- Gross, E.M. Allelopathy of Aquatic Autotrophs. CRC Crit. Rev. Plant Sci. 2003, 22, 313–339. [Google Scholar] [CrossRef]
- Leão, P.N.; Pereira, A.R.; Liu, W.-T.; Ng, J.; Pevzner, P.A.; Dorrestein, P.C.; König, G.M.; Vasconcelos, V.M.; Gerwick, W.H. Synergistic allelochemicals from a freshwater cyanobacterium. Proc. Natl. Acad. Sci. USA 2010, 107, 11183–11188. [Google Scholar] [CrossRef] [PubMed]
- Rounge, T.B.; Rohrlack, T.; Nederbragt, A.J.; Kristensen, T.; Jakobsen, K.S. A genome-wide analysis of nonribosomal peptide synthetase gene clusters and their peptides in a Planktothrix rubescens strain. BMC Genom. 2009, 10, 396. [Google Scholar] [CrossRef]
- Sanz, M.; Salinas, R.K.; Pinto, E. Namalides B and C and Spumigins K-N from the Cultured Freshwater Cyanobacterium Sphaerospermopsis torques-reginae. J. Nat. Prod. 2017, 80, 2492–2501. [Google Scholar] [CrossRef]
- Welker, M.; von Döhren, H. Cyanobacterial peptides—Nature’s own combinatorial biosynthesis. FEMS Microbiol. Rev. 2006, 30. [Google Scholar] [CrossRef]
- Fernandes, C.A.; Martins-Santos, I.C. Mapping of the 18S and 5S ribosomal RNA genes in Astyanax altiparanae Garutti & Britski, 2000 (Teleostei, Characidae) from the upper Paraná river basin, Brazil. Genet. Mol. Biol. 2006, 29, 464–468. [Google Scholar] [CrossRef]
- Maroneze, D.M.; Tupinambás, T.H.; Alves, C.B.M.; Vieira, F.; Pompeu, P.S.; Callisto, M. Fish as ecological tools to complement biodiversity inventories of benthic macroinvertebrates. Hydrobiologia 2011, 673, 29–40. [Google Scholar] [CrossRef]
- Orsi, M.; Daniel Carvalho, E.; Foresti, F. Population biology of Astyanax altiparanae Garutti & Britski, 2000 (teleostei, characidae) of the medium river Paranapanema. Rev. Bras. Zool. 2004, 21, 207–218. [Google Scholar] [CrossRef]
- Schulz, U.H.; Martins-Junior, H. Astyanax fasciatus as bioindicator of water pollution of Rio dos Sinos, RS, Brazil. Braz. J. Biol. 2001, 61, 615–622. [Google Scholar] [CrossRef]
- Vieira, C.E.D.; Almeida, M.D.S.; Galindo, B.A.; Pereira, L.; Martinez, C.B.D.R. Integrated biomarker response index using a Neotropical fish to assess the water quality in agricultural areas. Neotrop. Ichthyol. 2014, 12, 153–164. [Google Scholar] [CrossRef]
- Vilella, F.S.; Becker, F.G.; Hartz, S.M. Diet of Astyanax species (Teleostei, Characidae) in an Atlantic Forest River in Southern Brazil. Braz. Arch. Biol. Technol. 2002, 45, 223–232. [Google Scholar] [CrossRef]
- Oberemm, A.; Becker, J.; Codd, G.A.; Steinberg, C. Effects of cyanobacterial toxins and aqueous crude extracts of cyanobacteria on the development of fish and amphibians. Environ. Toxicol. 1999, 14, 77–88. [Google Scholar] [CrossRef]
- Oberemm, A.; Fastner, J.; Steinberg, C.E.W. Effects of microcystin-LR and cyanobacterial crude extracts on embryo-larval development of zebrafish (Danio rerio). Water Res. 1997, 31, 2918–2921. [Google Scholar] [CrossRef]
- Liu, Y.; Song, L.; Li, X.; Liu, T. The toxic effects of microcystin-LR on embryo-larval and juvenile development of loach, Misguruns mizolepis Gunthe. Toxicon 2002, 40, 395–399. [Google Scholar] [CrossRef]
- Palíková, M.; Navrátil, S.; Maršálek, B.; Bláha, L. Toxicity of Crude Extracts of Cyanobacteria for Embryos and Larvae of Carp (Cyprinus carpio L.). Acta Vet. Brno 2003, 72, 437–443. [Google Scholar] [CrossRef][Green Version]
- Lecoz, N.; Malécot, M.; Quiblier, C.; Puiseux-Dao, S.; Bernard, C.; Crespeau, F.; Edery, M. Effects of cyanobacterial crude extracts from Planktothrix agardhii on embryo-larval development of medaka fish, Oryzias latipes. Toxicon 2008, 51, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Cazenave, J.; De Los Ángeles Bistoni, M.; Zwirnmann, E.; Wunderlin, D.A.; Wiegand, C. Attenuating effects of natural organic matter on microcystin toxicity in zebra fish (Danio rerio) embryos—Benefits and costs of microcystin detoxication. Environ. Toxicol. 2006, 21, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, C.; Thermes, V.; De Luze, A.; Puiseux-Dao, S.; Bernard, C.; Joly, J.S.; Bourrat, F.; Edery, M. Effects of microcystin-LR on development of medaka fish embryos (Oryzias latipes). Toxicon 2004, 43, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Li, L.; Xue, T.; Long, M.; Su, Y.; Wu, N. Damage and recovery of the ovary in female zebrafish i.p.-injected with MC-LR. Aquat. Toxicol. 2014, 155, 110–118. [Google Scholar] [CrossRef]
- Malbrouck, C.; Kestemont, P. Effects of microcystins on fish. Environ. Toxicol. Chem. 2006, 25, 72–86. [Google Scholar] [CrossRef]
- Baganz, D.; Staaks, G.; Steinberg, C. Impact of the cyanobacteria toxin, microcystin-LR on behaviour of zebrafish, Danio rerio. Water Res. 1998, 32, 948–952. [Google Scholar] [CrossRef]
- Lee, J.; Lee, S.; Jiang, X. Cyanobacterial toxins in freshwater and food: Important sources of exposure to humans. Annu. Rev. Food Sci. Technol. 2017, 8, 281–304. [Google Scholar] [CrossRef] [PubMed]
- Flores, N.M.; Miller, T.R.; Stockwell, J.D. A Global Analysis of the Relationship between Concentrations of Microcystins in Water and Fish. Front. Mar. Sci. 2018, 5, 30. [Google Scholar] [CrossRef]
- Forchert, A.; Neumann, U.; Papendorf, O. New Cyanobacterial Substances with Bioactive Properties. In Cyanotoxins—Occurence, Causes, Consequences; Springer Science & Business Media: New York, NY, USA, 2001; pp. 295–315. [Google Scholar]
- El Ghazali, I.; Saqrane, S.; Carvalho, A.P.; Ouahid, Y.; Oudra, B.; Del Campo, F.F.; Vasconcelos, V. Compensatory growth induced in zebrafish larvae after pre-exposure to a Microcystis aeruginosa natural bloom extract containing Microcystins. Int. J. Mol. Sci. 2009, 10, 133–146. [Google Scholar] [CrossRef]
- Saraf, S.R.; Frenkel, A.; Harke, M.J.; Jankowiak, J.G.; Gobler, C.J.; McElroy, A.E. Effects of Microcystis on development of early life stage Japanese medaka (Oryzias latipes): Comparative toxicity of natural blooms, cultured Microcystis and microcystin-LR. Aquat. Toxicol. 2018, 194, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Sun, H.; Xie, P.; Wang, J.; Zhang, G.; Chen, N.; Yan, W.; Li, G. The role of apoptosis in MCLR-induced developmental toxicity in zebrafish embryos. Aquat. Toxicol. 2014, 149, 25–32. [Google Scholar] [CrossRef]
- Zi, J.; Pan, X.; MacIsaac, H.J.; Yang, J.; Xu, R.; Chen, S.; Chang, X. Cyanobacteria blooms induce embryonic heart failure in an endangered fish species. Aquat. Toxicol. 2018, 194, 78–85. [Google Scholar] [CrossRef]
- Wiegand, C.; Pflugmacher, S.; Beattie, K.A.; Steinberg, C.; Pflugmacher, S.; Oberemm, A.; Meems, N.; Beattie, K.A. Uptake and Effects of Microcystin-LR on Detoxication Enzymes of Early Life Stages of the Zebra Fish (Danio rerio) Uptake and Effects of Microcystin-LR on Detoxication Enzymes of Early Life Stages of the Zebra Fish (Danio rerio). Environ. Toxicol. 1999, 14, 89–95. [Google Scholar] [CrossRef]
- Morse, A.N.C.; Froyd, C.A.; Morse, D.E. Molecules from cyanobacteria and red algae that induce larval settlement and metamorphosis in the mollusc Haliotis rufescens. Mar. Biol. 1984, 81, 293–298. [Google Scholar] [CrossRef]
- Rogers, E.D.; Henry, T.B.; Twiner, M.J.; Gouffon, J.S.; McPherson, J.T.; Boyer, G.L.; Sayler, G.S.; Wilhelm, S.W. Global Gene Expression Profiling in Larval Zebrafish Exposed to Microcystin-LR and Microcystis Reveals Endocrine Disrupting Effects of Cyanobacteria. Environ. Sci. Technol. 2011, 45, 1962–1969. [Google Scholar] [CrossRef]
- Gorham, P.R.; McLachlan, J.; Hammer, U.T.; Kim, W. Isolation and culture of toxic strains of Anabaena flos-aquae (Lyngb.) Breb. Verh. Int. Ver. Theor. Angew. Limnol. 1964, 15, 1964. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Bortoli, S.; Oliveira-Silva, D.; Krüger, T.; Dörr, F.A.; Colepicolo, P.; Volmer, D.A.; Pinto, E. Growth and microcystin production of a Brazilian Microcystis aeruginosa strain (LTPNA 02) under different nutrient conditions. Rev. Bras. Farmacogn. 2014, 24, 389–398. [Google Scholar] [CrossRef]
- Woynarovich, E.; Horváth, L. Propagaçao artificial de peixes de águas tropicais manual: De extensao. In Propagaçao Artificial de Peixes de Águas Tropicais Manual: De Extensao; FAO/CODEVASF/CNPq: Brasilia, Brazil, 1983. [Google Scholar]
- Dos Santos, M.P.; Yasui, G.S.; Xavier, P.L.P.; de Macedo Adamov, N.S.; do Nascimento, N.F.; Fujimoto, T.; Senhorini, J.A.; Nakaghi, L.S.O. Morphology of gametes, post-fertilization events and the effect of temperature on the embryonic development of Astyanax altiparanae (Teleostei, Characidae). Zygote 2016, 24, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Yasui, G.S.; Senhorini, J.A.; Shimoda, E.; Pereira-Santos, M.; Nakaghi, L.S.O.; Fujimoto, T.; Arias-Rodriguez, L.; Silva, L.A. Improvement of gamete quality and its short-term storage: An approach for biotechnology in laboratory fish. Animal 2015, 9, 464–470. [Google Scholar] [CrossRef]
- Core Team R. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; ISBN 3-900051-07-0. [Google Scholar]
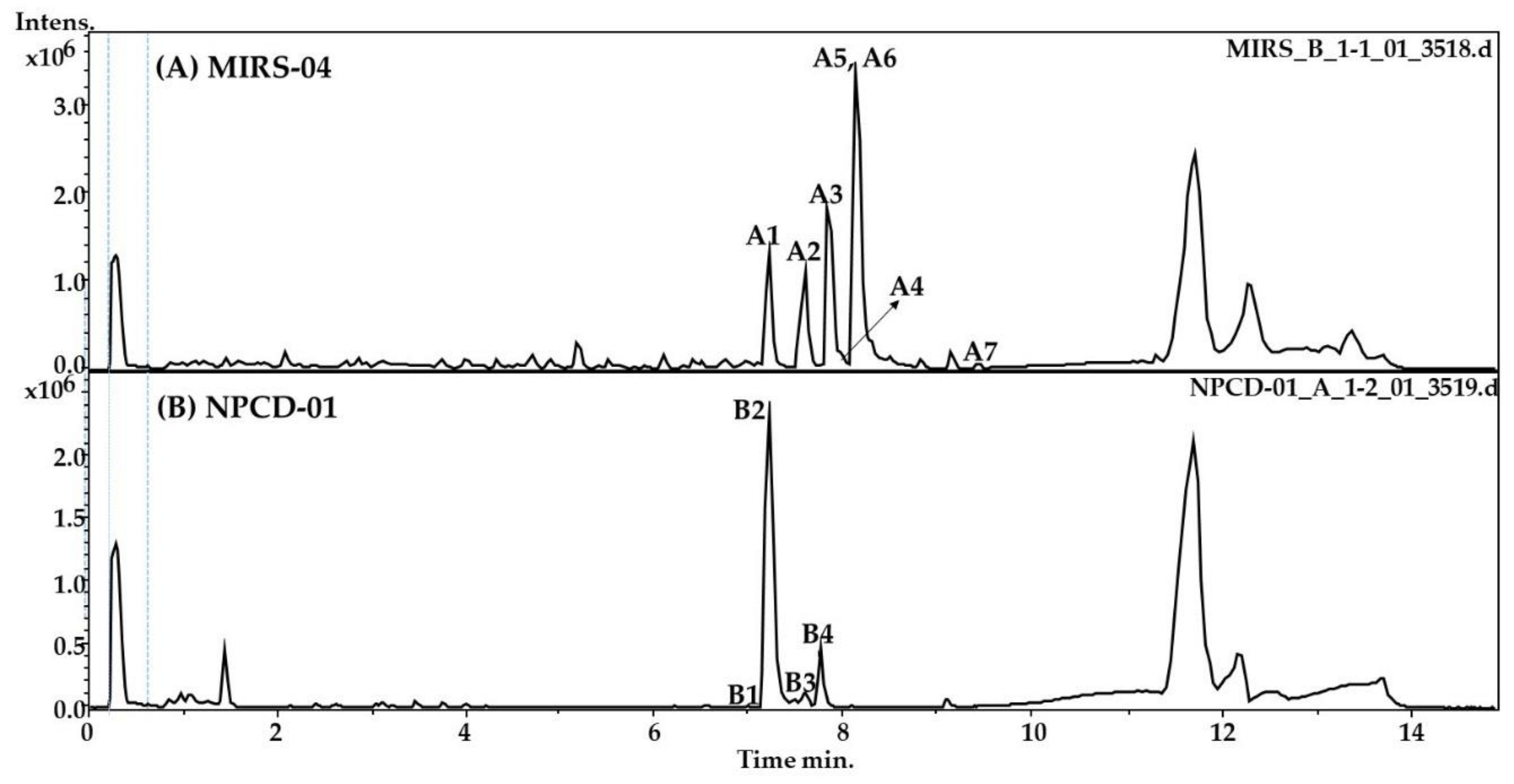
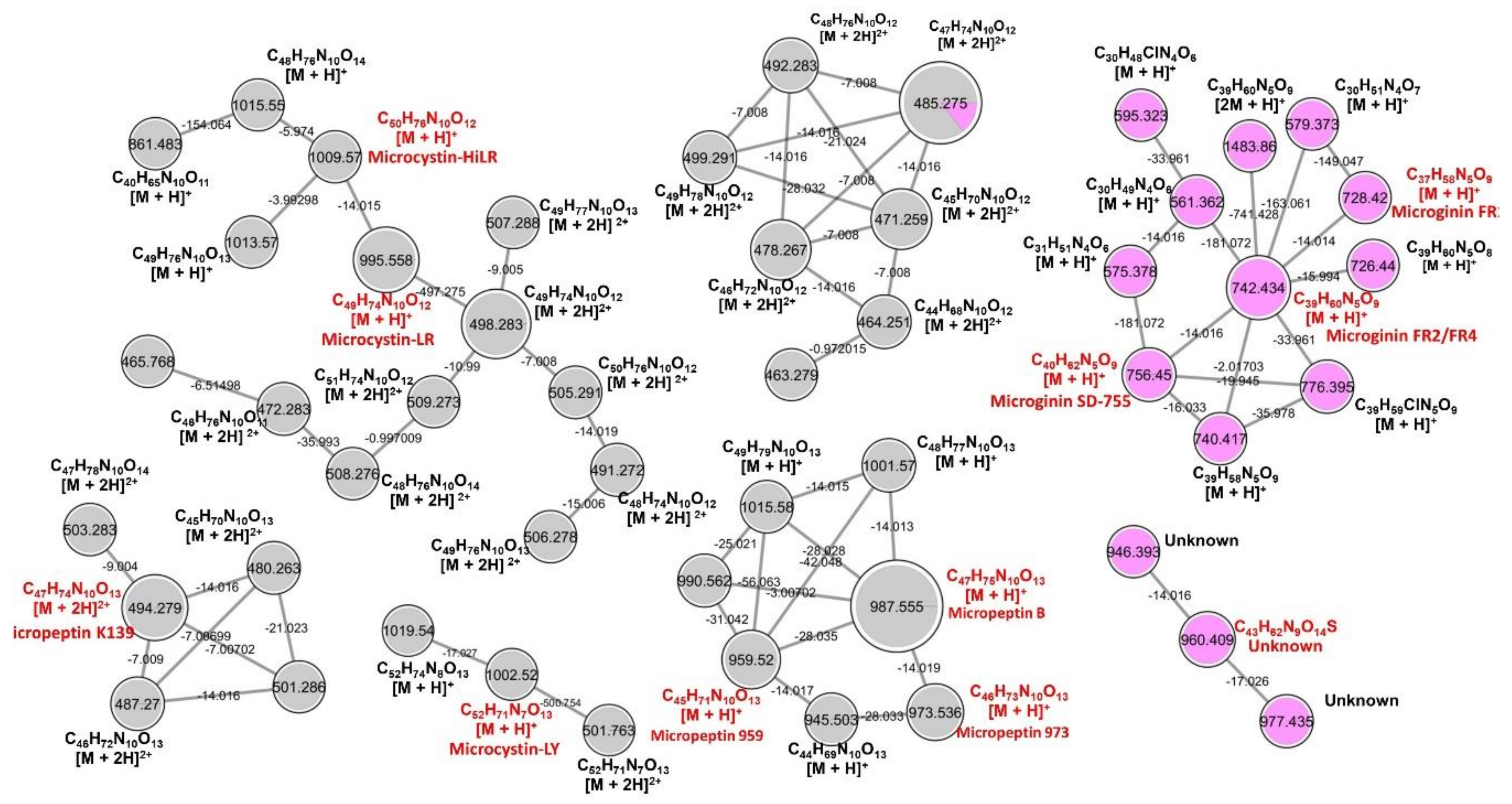
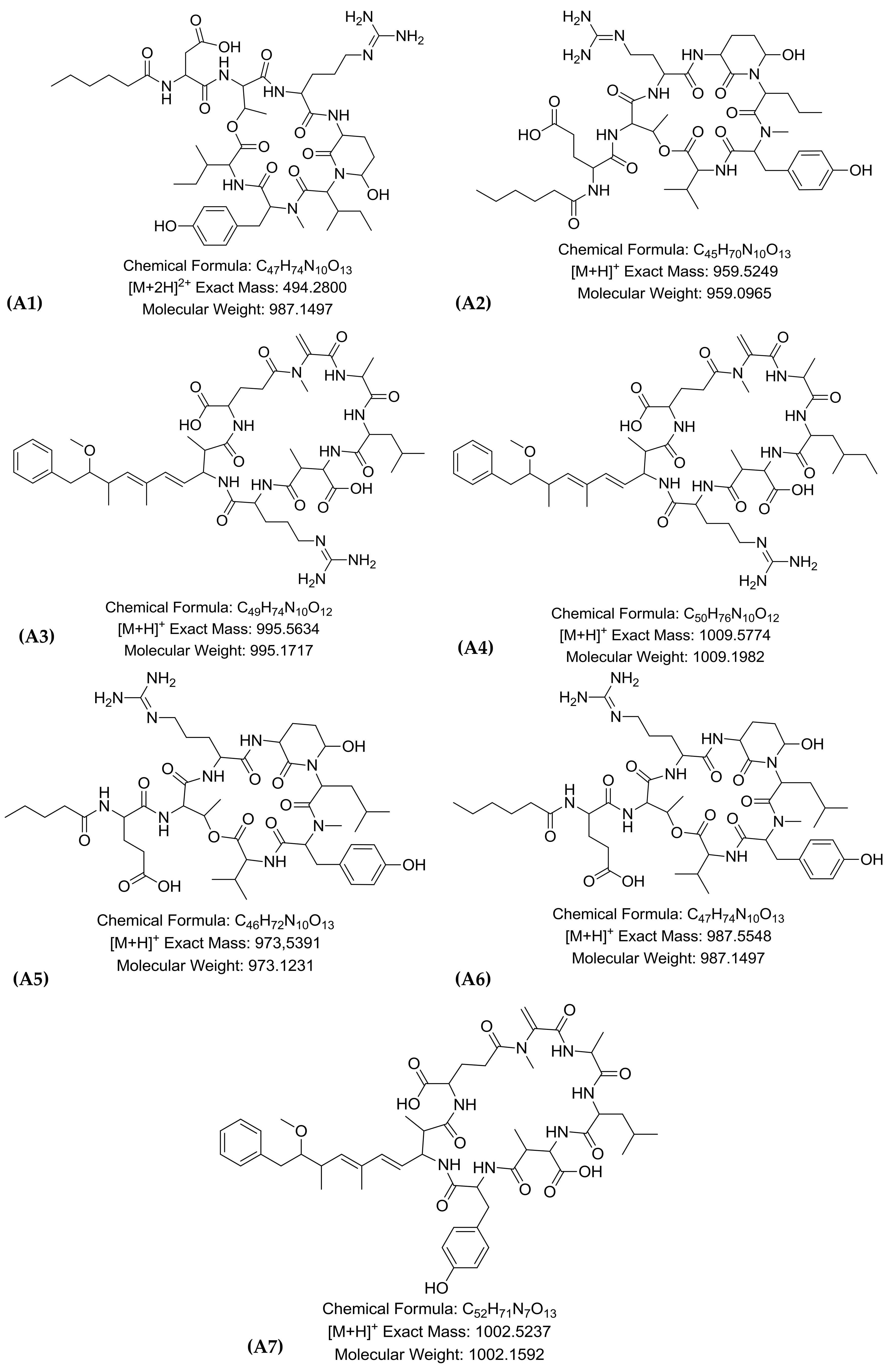
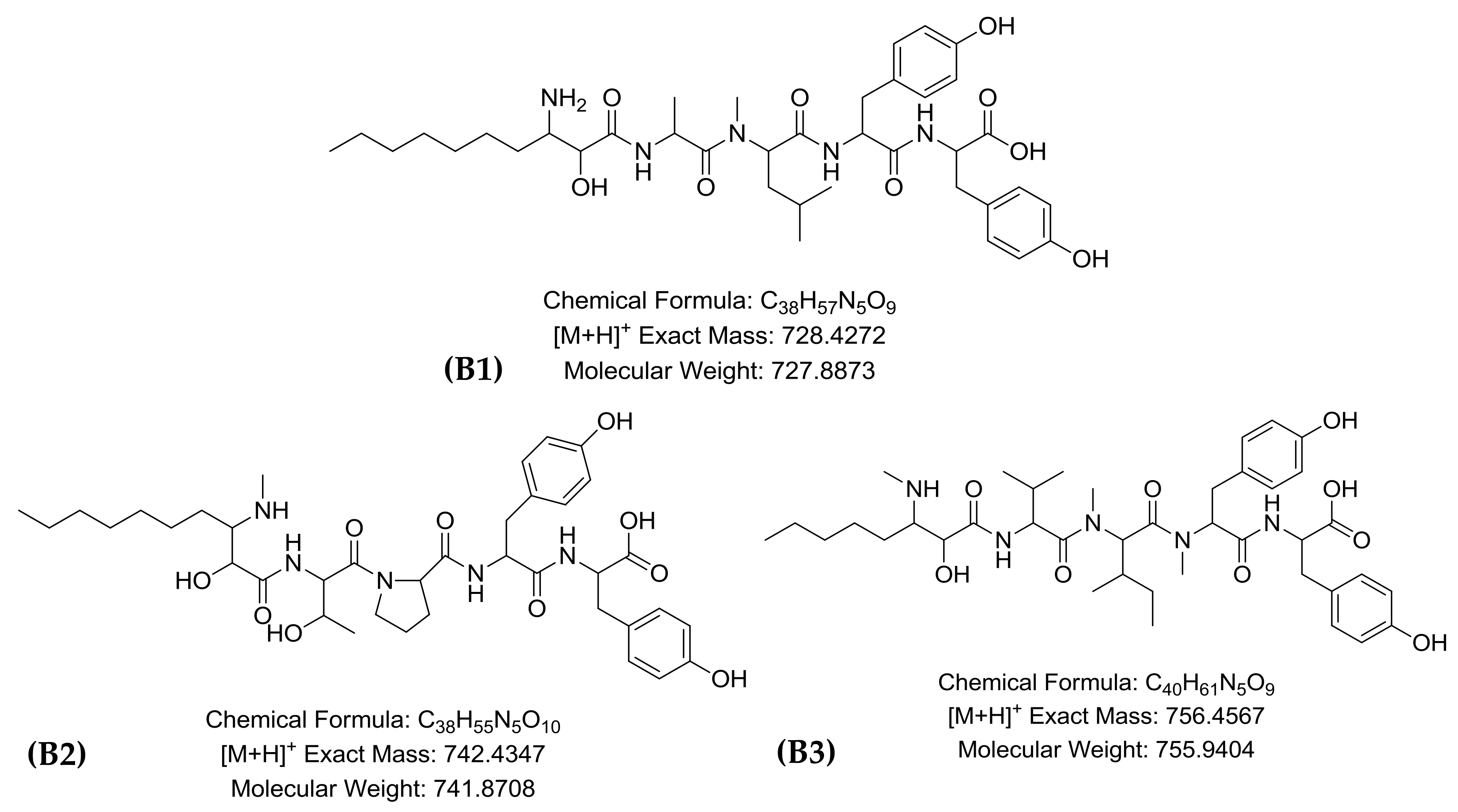

| Peak | RT * | Theoretical m/z | Experimental m/z | Ion Formula | Error [ppm] | mSigma | Compound Name | Reference |
|---|---|---|---|---|---|---|---|---|
| A1 | 7.2 | 494.2800 | 494.2797 | C47H76N10O13 | −0.6 | 2.3 | Micropeptin K139 * | [12] |
| A2 | 7.6 | 959.5249 | 959.5197 | C45H71N10O13 | −2.0 | 10.0 | Micropeptin 959 | [21] |
| A3 | 7.9 | 995.5634 | 995.5560 | C49H75N10O12 | −0.3 | 16.5 | Microcystin–LR | [16] |
| A4 | 8 | 1009.5774 | 1009.5717 | C50H77N10O12 | −0.7 | 20 | Microcystin HilR | [14] |
| A5 | 8.0 | 973.5391 | 973.5353 | C46H73N10O13 | 4.1 | 8.6 | Micropeptin 973 | [13] |
| A6 | 8.2 | 987.5548 | 987.5510 | C47H75N10O13 | −0.8 | 11.4 | Micropeptin B | [12] |
| A7 | 9.5 | 1002.5237 | 1002.5183 | C52H72N7O13 | 2.1 | 5.8 | Microcystin-LY | [22] |
| B1 | 7.0 | 728.4272 | 728.4229 | C38H58N5O9 | 5.2 | 14 | Microginin FR1 | [17] |
| B2 | 7.2 | 742.4347 | 742.4379 | C39H60N5O9 | 0.7 | 1.9 | Microginin FR2/FR4 | [20] |
| B3 | 7.6 | 756.4567 | 756.4542 | C40H62N5O9 | −3.2 | 5.3 | Microginin SD-755 | [23] |
| B4 | 7.8 | 960.4188 | 960.4131 | C43H62N9O14S | 3.5 | 2.0 | Unknown | [20] |
| Treatments | N Total of Larvae | N Total of Larvae M.A (+) * | Types of Morphological Alterations * Observed | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ODS % | ODS% | OPC% | OPC% | CP% | CP% | CT% | CT% | |||
| Control | 24 | 3 | 3 | 12.5 | 1 | 4.2 | 0 | 0 | 0 | 0 |
| 0.1 mg/mL-mirs-04 | 28 | 16 | 12 | 42.9 | 4 | 14.3 | 6 | 21.4 | 0 | 0 |
| 0.2 mg/mL-mirs-04 | 19 | 14 | 7 | 36.8 | 4 | 21.1 | 5 | 26.3 | 1 | 5.3 |
| 0.3 mg/mL-mirs-04 | 11 | 10 | 9 | 81.8 | 2 | 18.2 | 2 | 18.2 | 0 | 0 |
| 0.1 mg/mL-npcd-01 | 14 | 11 | 11 | 78.6 | 0 | 0 | 2 | 14.3 | 0 | 0 |
| 0.2 mg/mL-npcd-01 | 22 | 13 | 10 | 45.5 | 3 | 13.6 | 2 | 9.1 | 0 | 0 |
| 0.3 mg/mL-npcd-01 | 24 | 22 | 22 | 91.7 | 3 | 12.5 | 4 | 16.7 | 1 | 4.2 |
| Total | 142 | 89 | 74 | 52.1 | 17 | 12 | 21 | 14.8 | 2 | 1.4 |
| Treatments | ODS (+) * | ODS (−) * | N Total of Larvae | Frequency of Occurrence (%) |
|---|---|---|---|---|
| Control | 3 | 21 | 24 | 12.5 a |
| 0.1 mg/mL-MIRS-04 | 12 | 16 | 28 | 42.9 b, f, g |
| 0.2 mg/mL-MIRS-04 | 7 | 12 | 19 | 36.8 c, d, e, g |
| 0.3 mg/mL-MIRS-04 | 9 | 2 | 11 | 81.8 c, d |
| 0.1 mg/mL-NPCD-01 | 11 | 3 | 14 | 78.6 c, d, e |
| 0.2 mg/mL-NPCD-01 | 10 | 12 | 22 | 45.5 f, g |
| 0.3 mg/mL-NPCD-01 | 22 | 2 | 24 | 91.7 b, c, f, g |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, K.; Gomes, A.; Calado, L.; Yasui, G.; Assis, D.; Henry, T.; Fonseca, A.; Pinto, E. Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae. Toxins 2019, 11, 220. https://doi.org/10.3390/toxins11040220
Fernandes K, Gomes A, Calado L, Yasui G, Assis D, Henry T, Fonseca A, Pinto E. Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae. Toxins. 2019; 11(4):220. https://doi.org/10.3390/toxins11040220
Chicago/Turabian StyleFernandes, Kelly, Andreia Gomes, Leonardo Calado, George Yasui, Diego Assis, Theodore Henry, Ana Fonseca, and Ernani Pinto. 2019. "Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae" Toxins 11, no. 4: 220. https://doi.org/10.3390/toxins11040220
APA StyleFernandes, K., Gomes, A., Calado, L., Yasui, G., Assis, D., Henry, T., Fonseca, A., & Pinto, E. (2019). Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae. Toxins, 11(4), 220. https://doi.org/10.3390/toxins11040220







