Staphylococcus aureus Pneumonia: Preceding Influenza Infection Paves the Way for Low-Virulent Strains
Abstract
1. Introduction
2. Results
2.1. The Genetic Analysis of Staphylococcal Isolates Showed That Primary Pneumonia Is Associated with Certain Exotoxins and Proteases
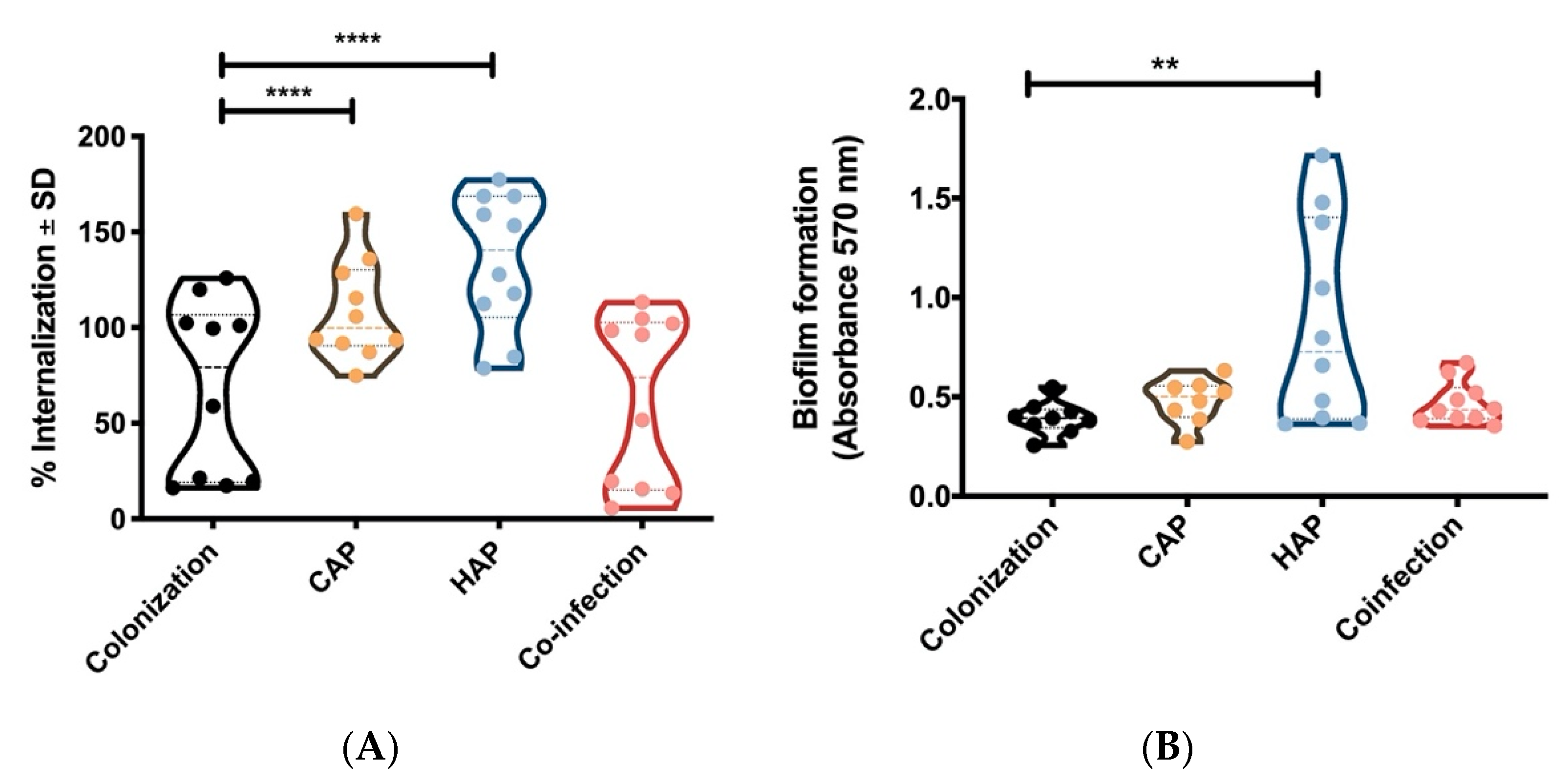
2.2. Strains Obtained From Primary Pneumonia Are More Invasive in Host Cells Than Strains from Colonization and Co-Infection
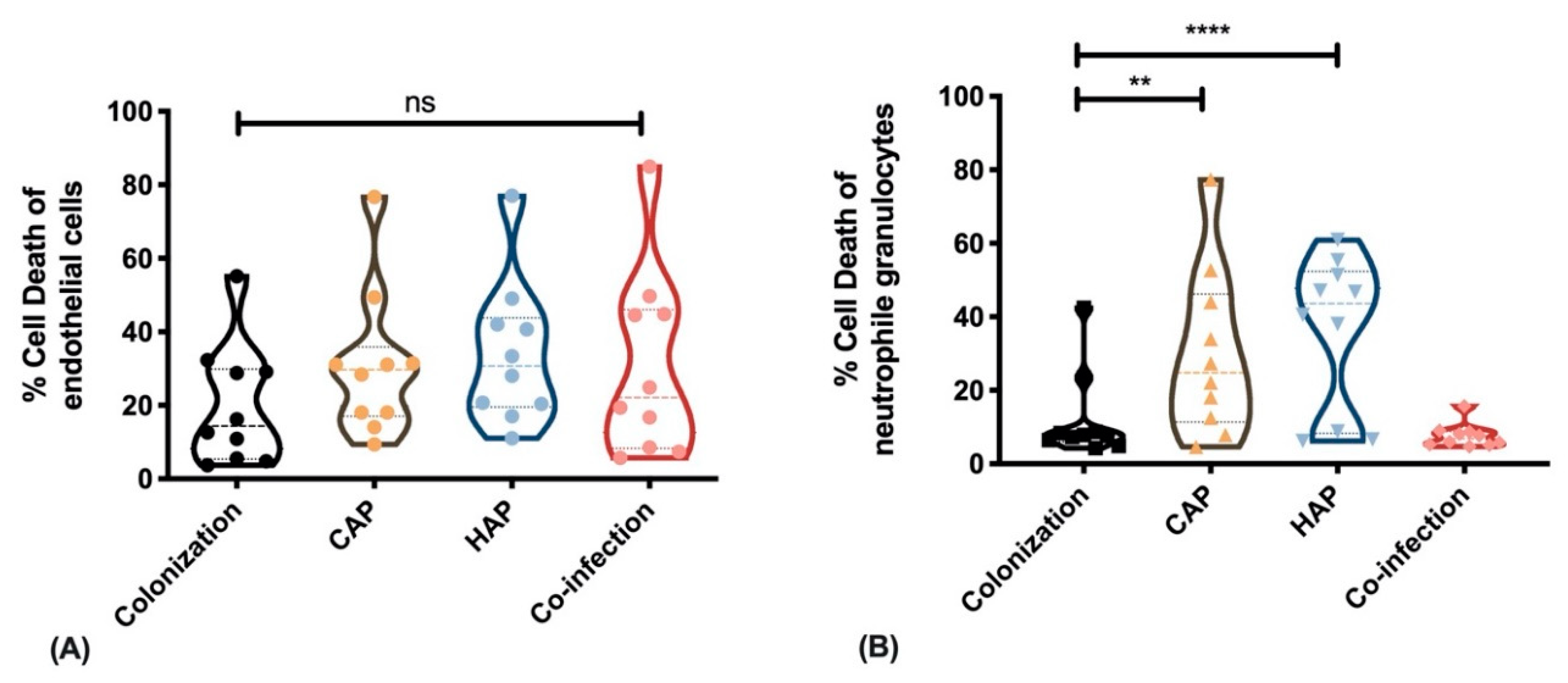
2.3. Strains Obtained from Primary Pneumonia Exhibit Higher Cytotoxicity against Immune Cells Than Strains from Colonization and Co-Infection
3. Discussion
4. Materials and Methods
4.1. Study Design and Informed Consent
4.2. Sample Preparation
4.3. Functional Cell-Based Assays
4.4. Genotyping
4.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Deinhardt-Emmer, S.; Sachse, S.; Geraci, J.; Fischer, C.; Kwetkat, A.; Dawczynski, K.; Tuchscherr, L.; Loffler, B. Virulence patterns of Staphylococcus aureus strains from nasopharyngeal colonization. J. Hosp. Infect. 2018, 100, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Wertheim, H.F.; Melles, D.C.; Vos, M.C.; van Leeuwen, W.; van Belkum, A.; Verbrugh, H.A.; Nouwen, J.L. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect. Dis. 2005, 5, 751–762. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Grousd, J.A.; Rich, H.E.; Alcorn, J.F. Host-Pathogen Interactions in Gram-Positive Bacterial Pneumonia. Clin. Microbiol. Rev. 2019, 32, e00107–e00118. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Self, W.H.; Wunderink, R.G.; Fakhran, S.; Balk, R.; Bramley, A.M.; Reed, C.; Grijalva, C.G.; Anderson, E.J.; Courtney, D.M.; et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N. Engl. J. Med. 2015, 373, 415–427. [Google Scholar] [CrossRef]
- von Baum, H.; Schweiger, B.; Welte, T.; Marre, R.; Suttorp, N.; Pletz, M.W.; Ewig, S. How deadly is seasonal influenza-associated pneumonia? The German Competence Network for Community-Acquired Pneumonia. Eur. Respir. J. 2011, 37, 1151–1157. [Google Scholar] [CrossRef]
- Hanson, K.E.; Couturier, M.R. Multiplexed Molecular Diagnostics for Respiratory, Gastrointestinal, and Central Nervous System Infections. Clin. Infect. Dis. 2016, 63, 1361–1367. [Google Scholar] [CrossRef]
- Peteranderl, C.; Herold, S.; Schmoldt, C. Human Influenza Virus Infections. Semin. Respir. Crit. Care Med. 2016, 37, 487–500. [Google Scholar] [CrossRef]
- Bhat, N.; Wright, J.G.; Broder, K.R.; Murray, E.L.; Greenberg, M.E.; Glover, M.J.; Likos, A.M.; Posey, D.L.; Klimov, A.; Lindstrom, S.E.; et al. Influenza-Associated Deaths among Children in the United States, 2003–2004. N. Engl. J. Med. 2005, 353, 2559–2567. [Google Scholar] [CrossRef]
- Grousd, J.A.; Richardson, A.; Cooper, V.; Alcorn, J.F. Adhesion requirements of methicillin-resistant Staphylococcus aureus in bacterial pneumonia and influenza super-infection. J. Immunol. 2019, 202, 190.49. [Google Scholar]
- McCullers, J.A. The co-pathogenesis of influenza viruses with bacteria in the lung. Nat. Rev. Microbiol. 2014, 12, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Lina, G.; Piemont, Y.; Godail-Gamot, F.; Bes, M.; Peter, M.O.; Gauduchon, V.; Vandenesch, F.; Etienne, J. Involvement of Panton-Valentine leukocidin-producing Staphylococcus aureus in primary skin infections and pneumonia. Clin. Infect. Dis. 1999, 29, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Loffler, B.; Niemann, S.; Ehrhardt, C.; Horn, D.; Lanckohr, C.; Lina, G.; Ludwig, S.; Peters, G. Pathogenesis of Staphylococcus aureus necrotizing pneumonia: The role of PVL and an influenza coinfection. Expert Rev. Anti-Infect. Ther. 2013, 11, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Berube, B.J.; Bubeck Wardenburg, J. Staphylococcus aureus alpha-toxin: Nearly a century of intrigue. Toxins 2013, 5, 1140–1166. [Google Scholar] [CrossRef]
- Diep, B.A.; Chan, L.; Tattevin, P.; Kajikawa, O.; Martin, T.R.; Basuino, L.; Mai, T.T.; Marbach, H.; Braughton, K.R.; Whitney, A.R.; et al. Polymorphonuclear leukocytes mediate Staphylococcus aureus Panton-Valentine leukocidin-induced lung inflammation and injury. Proc. Natl. Acad. Sci. USA 2010, 107, 5587–5592. [Google Scholar] [CrossRef]
- Bloes, D.A.; Haasbach, E.; Hartmayer, C.; Hertlein, T.; Klingel, K.; Kretschmer, D.; Planz, O.; Peschel, A. Phenol-Soluble Modulin Peptides Contribute to Influenza A Virus-Associated Staphylococcus aureus Pneumonia. Infect. Immun. 2017, 85, e00620-17. [Google Scholar] [CrossRef]
- Tam, K.; Torres, V.J. Staphylococcus aureus Secreted Toxins and Extracellular Enzymes. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef]
- Grumann, D.; Nübel, U.; Bröker, B.M. Staphylococcus Aureus Toxins—Their Functions and Genetics; Robert Koch-Institut: Berlin, Germany, 2013; Volume 21, pp. 583–592. [Google Scholar]
- Foster, T.J. The remarkably multifunctional fibronectin binding proteins of Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1923–1931. [Google Scholar] [CrossRef]
- Fraunholz, M.; Sinha, B. Intracellular Staphylococcus aureus: Live-in and let die. Front. Cell. Infect. Microbiol. 2012, 2, 43. [Google Scholar] [CrossRef]
- Loffler, B.; Hussain, M.; Grundmeier, M.; Bruck, M.; Holzinger, D.; Varga, G.; Roth, J.; Kahl, B.C.; Proctor, R.A.; Peters, G. Staphylococcus aureus panton-valentine leukocidin is a very potent cytotoxic factor for human neutrophils. PLoS Pathog. 2010, 6, e1000715. [Google Scholar] [CrossRef]
- Suttorp, N.; Welte, T.; Marre, R.; Stenger, S.; Pletz, M.; Rupp, J.; Schutte, H.; Rohde, G. [CAPNETZ. The competence network for community-acquired pneumonia (CAP)]. Bundesgesundheitsblatt Gesundh. Gesundh. 2016, 59, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Kalinka, J.; Hachmeister, M.; Geraci, J.; Sordelli, D.; Hansen, U.; Niemann, S.; Oetermann, S.; Peters, G.; Loffler, B.; Tuchscherr, L. Staphylococcus aureus isolates from chronic osteomyelitis are characterized by high host cell invasion and intracellular adaptation, but still induce inflammation. Int. J. Med Microbiol. 2014, 304, 1038–1049. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.; Francois, P.; Que, Y.A.; Hussain, M.; Heilmann, C.; Moreillon, P.; Lew, D.; Krause, K.H.; Peters, G.; Herrmann, M. Heterologously expressed Staphylococcus aureus fibronectin-binding proteins are sufficient for invasion of host cells. Infect. Immun. 2000, 68, 6871–6878. [Google Scholar] [CrossRef] [PubMed]
- Arciola, C.R.; Campoccia, D.; Montanaro, L. Implant infections: Adhesion, biofilm formation and immune evasion. Nat. Rev. Microbiol. 2018, 16, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Spaulding, A.R.; Salgado-Pabón, W.; Merriman, J.A.; Stach, C.S.; Ji, Y.; Gillman, A.N.; Peterson, M.L.; Schlievert, P.M. Vaccination against Staphylococcus aureus pneumonia. J. Infect. Dis. 2014, 209, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Horn, J.; Stelzner, K.; Rudel, T.; Fraunholz, M. Inside job: Staphylococcus aureus host-pathogen interactions. Int. J. Med. Microbiol. 2018, 308, 607–624. [Google Scholar] [CrossRef]
- Geraci, J.; Neubauer, S.; Pöllath, C.; Hansen, U.; Rizzo, F.; Krafft, C.; Westermann, M.; Hussain, M.; Peters, G.; Pletz, M.W.; et al. The Staphylococcus aureus extracellular matrix protein (Emp) has a fibrous structure and binds to different extracellular matrices. Sci. Rep. 2017, 7, 13665. [Google Scholar] [CrossRef]
- Josse, J.; Laurent, F.; Diot, A. Staphylococcal Adhesion and Host Cell Invasion: Fibronectin-Binding and Other Mechanisms. Front. Microbiol. 2017, 8, 2433. [Google Scholar] [CrossRef]
- Klinger-Strobel, M.; Stein, C.; Forstner, C.; Makarewicz, O.; Pletz, M.W. Effects of colistin on biofilm matrices of Escherichia coli and Staphylococcus aureus. Int. J. Antimicrob. Agents 2017, 49, 472–479. [Google Scholar] [CrossRef]
- Tuchscherr, L.; Pollath, C.; Siegmund, A.; Deinhardt-Emmer, S.; Hoerr, V.; Svensson, C.M.; Thilo Figge, M.; Monecke, S.; Loffler, B. Clinical S. aureus Isolates Vary in Their Virulence to Promote Adaptation to the Host. Toxins 2019, 11, 135. [Google Scholar] [CrossRef]
- Parker, D. Humanized Mouse Models of Staphylococcus aureus Infection. Front. Immunol. 2017, 8, 512. [Google Scholar] [CrossRef] [PubMed]
- Tuffs, S.W.; James, D.B.A.; Bestebroer, J.; Richards, A.C.; Goncheva, M.I.; O’Shea, M.; Wee, B.A.; Seo, K.S.; Schlievert, P.M.; Lengeling, A.; et al. The Staphylococcus aureus superantigen SElX is a bifunctional toxin that inhibits neutrophil function. PLOS Pathog. 2017, 13, e1006461. [Google Scholar] [CrossRef] [PubMed]
- Parker, D.; Prince, A. Immunopathogenesis of Staphylococcus aureus pulmonary infection. Semin. Immunopathol. 2012, 34, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Paharik, A.E.; Salgado-Pabon, W.; Meyerholz, D.K.; White, M.J.; Schlievert, P.M.; Horswill, A.R. The Spl Serine Proteases Modulate Staphylococcus aureus Protein Production and Virulence in a Rabbit Model of Pneumonia. mSphere 2016, 1, e00208-16. [Google Scholar] [CrossRef] [PubMed]
- Linge, H.M.; Andersson, C.; Nordin, S.L.; Olin, A.I.; Petersson, A.C.; Morgelin, M.; Welin, A.; Bylund, J.; Bjermer, L.; Erjefalt, J.; et al. Midkine is expressed and differentially processed during chronic obstructive pulmonary disease exacerbations and ventilator-associated pneumonia associated with Staphylococcus aureus infection. Mol. Med. 2013, 19, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Laarman, A.J.; Ruyken, M.; Malone, C.L.; van Strijp, J.A.G.; Horswill, A.R.; Rooijakkers, S.H.M. Staphylococcus aureus Metalloprotease Aureolysin Cleaves Complement C3 To Mediate Immune Evasion. J. Immunol. 2011, 186, 6445. [Google Scholar] [CrossRef]
- Sharma-Kuinkel, B.K.; Mongodin, E.F.; Myers, J.R.; Vore, K.L.; Canfield, G.S.; Fraser, C.M.; Rude, T.H.; Fowler, V.G., Jr.; Gill, S.R. Potential Influence of Staphylococcus aureus Clonal Complex 30 Genotype and Transcriptome on Hematogenous Infections. In Open Forum Infectious Diseases; Oxford University Press: Oxford, UK, 2015; Volume 2, p. ofv093. [Google Scholar] [CrossRef]
- Bowers, J.R.; Driebe, E.M.; Albrecht, V.; McDougal, L.K.; Granade, M.; Roe, C.C.; Lemmer, D.; Rasheed, J.K.; Engelthaler, D.M.; Keim, P.; et al. Improved Subtyping of Staphylococcus aureus Clonal Complex 8 Strains Based on Whole-Genome Phylogenetic Analysis. mSphere 2018, 3, e00464-17. [Google Scholar] [CrossRef]
- Kurt, K.; Rasigade, J.-P.; Laurent, F.; Goering, R.V.; Žemličková, H.; Machova, I.; Struelens, M.J.; Zautner, A.E.; Holtfreter, S.; Bröker, B.; et al. Subpopulations of Staphylococcus aureus Clonal Complex 121 Are Associated with Distinct Clinical Entities. PLoS ONE 2013, 8, e58155. [Google Scholar] [CrossRef]
- Hagel, S.; Ludewig, K.; Moeser, A.; Baier, M.; Loffler, B.; Schleenvoigt, B.; Forstner, C.; Pletz, M.W. Characteristics and management of patients with influenza in a German hospital during the 2014/2015 influenza season. Infection 2016, 44, 667–672. [Google Scholar] [CrossRef]
- Morgene, M.F.; Botelho-Nevers, E.; Grattard, F.; Pillet, S.; Berthelot, P.; Pozzetto, B.; Verhoeven, P.O. Staphylococcus aureus colonization and non-influenza respiratory viruses: Interactions and synergism mechanisms. Virulence 2018, 9, 1354–1363. [Google Scholar] [CrossRef]
- Welte, T.; Suttorp, N.; Marre, R. CAPNETZ-community-acquired pneumonia competence network. Infection 2004, 32, 234–238. [Google Scholar] [CrossRef] [PubMed]
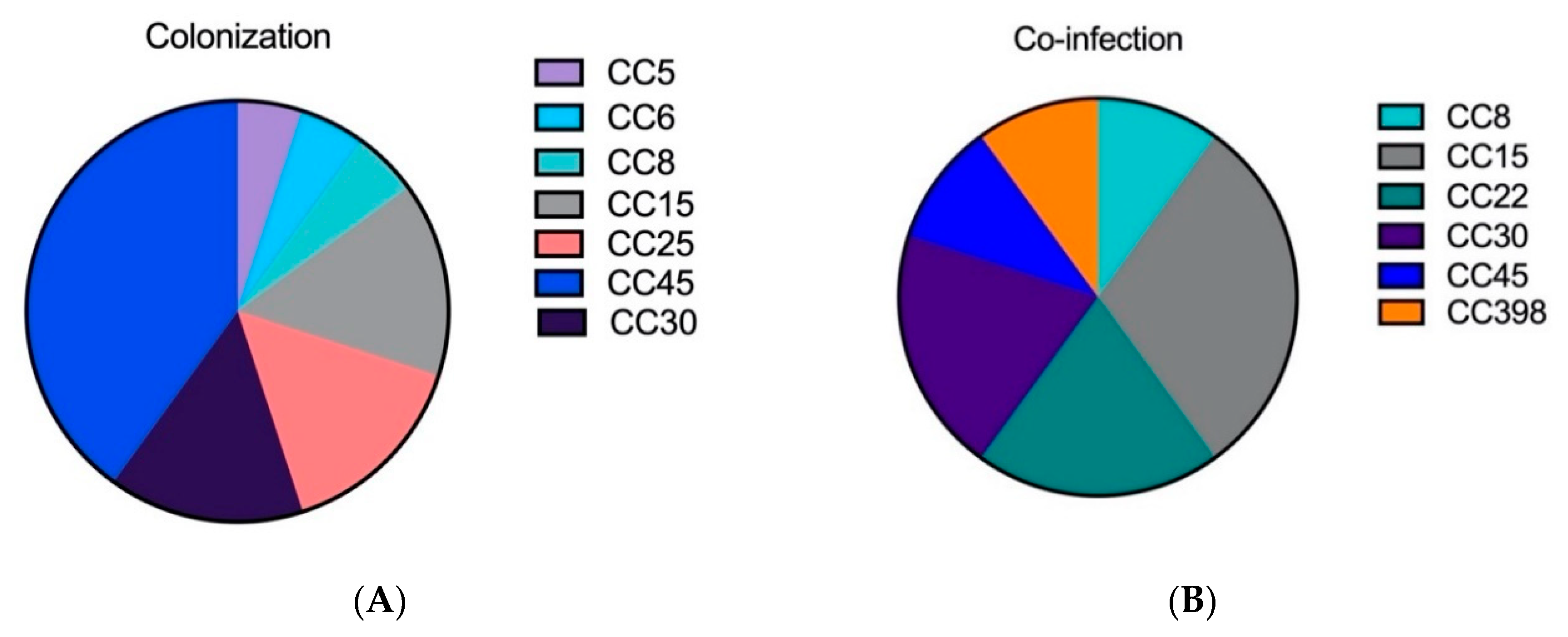
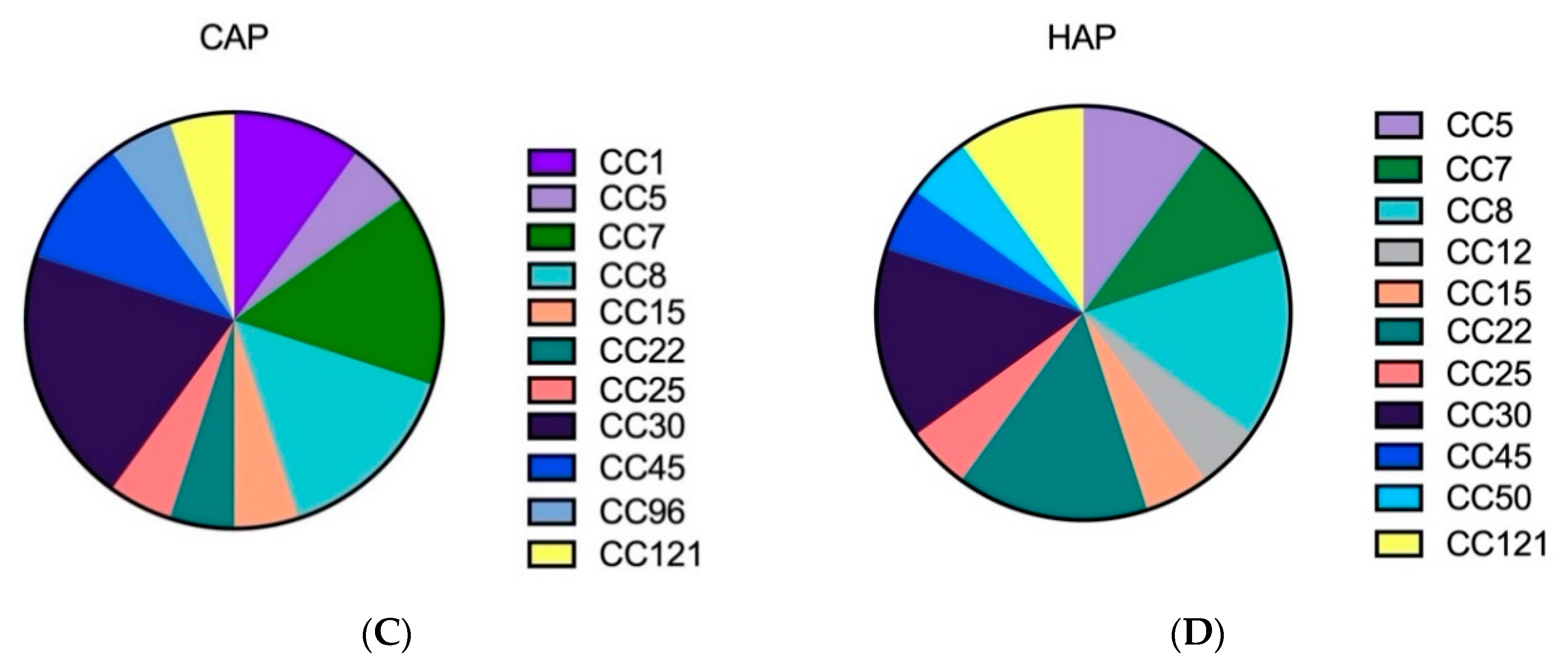
| Characterization | Colonization | CAP | HAP | Co-Infection |
|---|---|---|---|---|
| Age (mean ± SD) | 61.80 (±19.32) years | 64.40 (±9.58) years | 62.40 (±6.34) years | 58.10 (±6.99) years |
| Male Sex | 60% | 70% | 55% | 40% |
| Specimen | 100% respiratory specimens | 50% respiratory specimens 50% BAL | 15% respiratory specimens 85% BAL | 10% respiratory specimens 90% BAL |
| Mortality | 0% | 20% | 15% | 60% |
| Virulence Factors | % Positive Isolates | |||||
|---|---|---|---|---|---|---|
| Group | Gene | Description | Colonization (n = 20) | CAP (n = 20) | HAP (n = 20) | Co-Infection (n = 10) |
| Hemolysin gamma and Leukocidins | Tst1 | Toxic Shock Toxin 1 | 10 | 25 | 15 | 20 |
| lukX | Leukocidin/Hemolysin Toxin Family Protein X | 100 | 100 | 100 | 90 | |
| lukY | Leukocidin/Hemolysin Toxin Family Protein Y | 100 | 100 | 100 | 100 | |
| lukE, lukD | Leukocidin E/D | 45 | 65 | 70 | 30 | |
| lukF, lukS | Hemolysin gamma (comp. B, C) | 100 | 100 | 100 | 100 | |
| lukF/S-PV | Panton Valentine Leukocidin | 5 | 0 | 0 | 0 | |
| Hemolysin | hla | Hemolysin alpha | 100 | 95 | 100 | 100 |
| hlb | Hemolysin beta | 65 | 80 | 90 | 60 | |
| hlIII | Putative membrane proteins | 100 | 100 | 100 | 100 | |
| Staphylococcal Superantigen | ssl02 | Staphylococcal Superantigen-like Protein 2 | 50 | 65 | 80 * | 40 |
| ssl03 | Staphylococcal Superantigen-like Protein 3 | 45 | 70 | 90 ** | 30 | |
| ssl07 | Staphylococcal Superantigen-like Protein 7 | 45 | 60 | 60 | 30 | |
| ssl08 | Staphylococcal Superantigen-like Protein 8 | 40 | 70 | 70 | 30 | |
| ssl09 | Staphylococcal Superantigen-like Protein 9 | 50 | 65 | 80 * | 40 | |
| ssl11 | Staphylococcal Superantigen-like Protein 11 | 55 | 15 | 20 | 0 | |
| Hlb-modificated phages and proteases | chp | Chemotaxis-inhibiting protein | 75 | 60 | 55 | 80 |
| aur | Aureolysin | 60 | 90 * | 100 ** | 100 | |
| splA | Serinprotease A | 45 | 65 | 70 | 30 | |
| splB | Serinprotease B | 45 | 70 | 70 | 30 | |
| sspB/A/P | Staphopain B/A/P, Protease | 100 | 100 | 100 | 100 | |
| Adhesion factors | fnbpA | Fibronectin-binding protein A | 100 | 100 | 100 | 100 |
| fnbpB | Fibronectin-binding protein B | 85 | 70 | 80 | 50 | |
| clfA | Clumping factor A | 100 | 100 | 100 | 100 | |
| clfB | Clumping factor B | 100 | 100 | 100 | 100 | |
| cna | Collagen binding adhesin | 65 | 60 | 50 | 60 | |
| ebh | Cell wall associated fibronectin-binding protein | 100 | 90 | 95 | 70 | |
| ebpS | Cell surface elastin binding protein | 100 | 100 | 100 | 100 | |
| eno | enolase | 100 | 100 | 100 | 100 | |
| sasG | S. aureus surface protein G | 30 | 45 | 45 | 50 | |
| vwb | Von Willebrand factor binding protein | 100 | 100 | 100 | 100 | |
| eap | Extracellular adherence protein | 100 | 95 | 100 | 100 | |
| sdrC | Ser-Asp rich fibrinogen-/bone sialoprotein-binding protein C | 100 | 100 | 100 | 100 | |
| sdrD | Ser-Asp rich fibrinogen-/bone sialoprotein-binding protein D | 70 | 90 | 90 | 80 | |
| Capsule/Biofilm | Cap 1 | Capsule type 1 | 0 | 0 | 0 | 0 |
| Cap 5 | Capsule type 5 | 30 | 30 | 40 | 40 | |
| Cap 8 | Capsule type 8 | 75 | 70 | 60 | 60 | |
| ica A | intercellular adhesion protein A | 100 | 100 | 100 | 100 | |
| ica C | intercellular adhesion protein C | 100 | 100 | 100 | 100 | |
| ica D | biofilm polysaccharide intercellular adhesin (PIA) synthesis protein D | 100 | 100 | 100 | 100 | |
| bap | surface protein involved in biofilm formation | 0 | 0 | 0 | 0 | |
| bbp | bone sialoprotein-binding protein | 85 | 90 | 100 | 90 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deinhardt-Emmer, S.; Haupt, K.F.; Garcia-Moreno, M.; Geraci, J.; Forstner, C.; Pletz, M.; Ehrhardt, C.; Löffler, B. Staphylococcus aureus Pneumonia: Preceding Influenza Infection Paves the Way for Low-Virulent Strains. Toxins 2019, 11, 734. https://doi.org/10.3390/toxins11120734
Deinhardt-Emmer S, Haupt KF, Garcia-Moreno M, Geraci J, Forstner C, Pletz M, Ehrhardt C, Löffler B. Staphylococcus aureus Pneumonia: Preceding Influenza Infection Paves the Way for Low-Virulent Strains. Toxins. 2019; 11(12):734. https://doi.org/10.3390/toxins11120734
Chicago/Turabian StyleDeinhardt-Emmer, Stefanie, Karoline Frieda Haupt, Marina Garcia-Moreno, Jennifer Geraci, Christina Forstner, Mathias Pletz, Christina Ehrhardt, and Bettina Löffler. 2019. "Staphylococcus aureus Pneumonia: Preceding Influenza Infection Paves the Way for Low-Virulent Strains" Toxins 11, no. 12: 734. https://doi.org/10.3390/toxins11120734
APA StyleDeinhardt-Emmer, S., Haupt, K. F., Garcia-Moreno, M., Geraci, J., Forstner, C., Pletz, M., Ehrhardt, C., & Löffler, B. (2019). Staphylococcus aureus Pneumonia: Preceding Influenza Infection Paves the Way for Low-Virulent Strains. Toxins, 11(12), 734. https://doi.org/10.3390/toxins11120734








