Label-Free Optical Detection of Mycotoxins Using Specific Aptamers Immobilized on Gold Nanostructures
Abstract
:1. Introduction
2. Results and Discussion
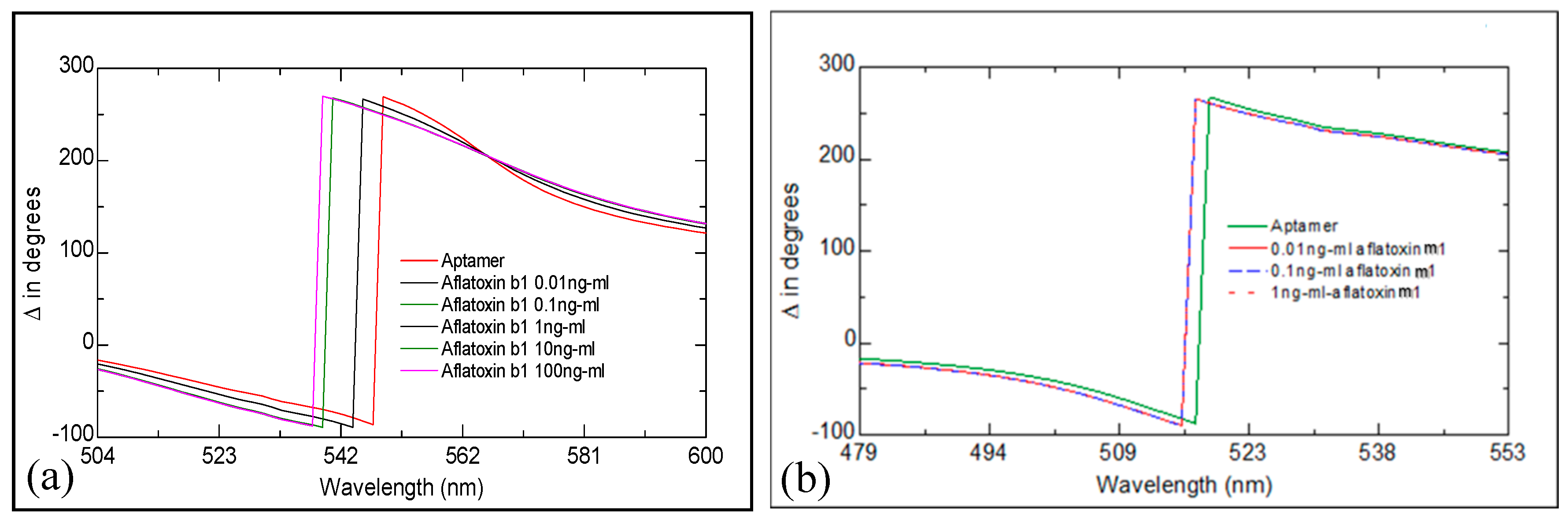
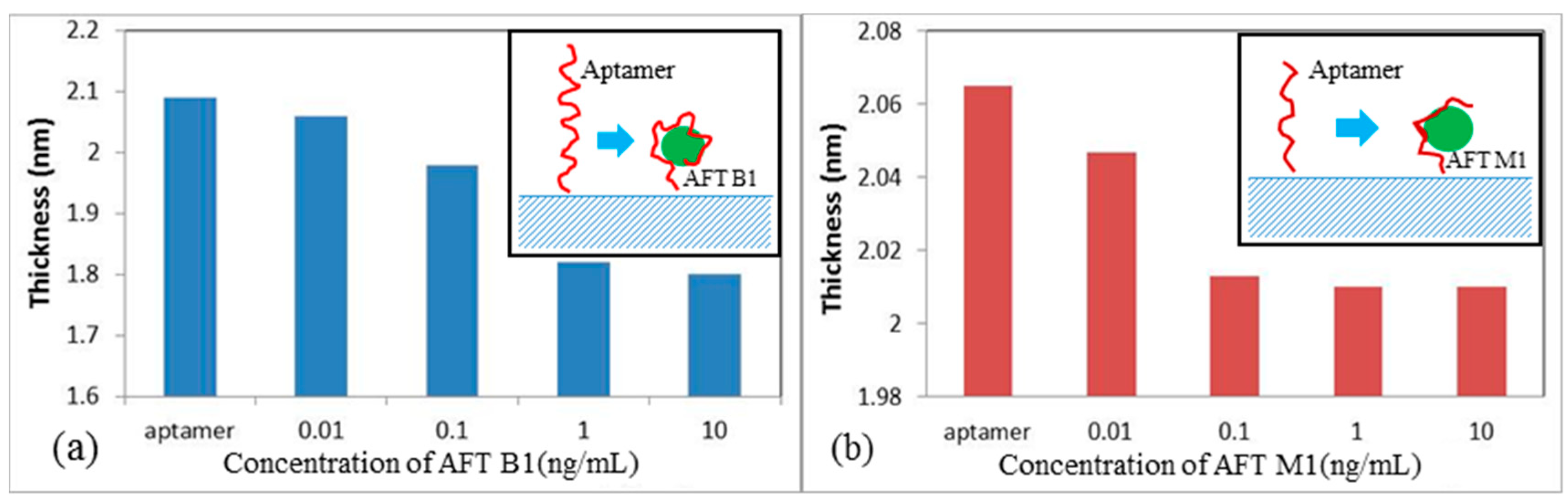
2.1. Study of Aflatoxin/Aptamer Binding Using LSPR/TIRE
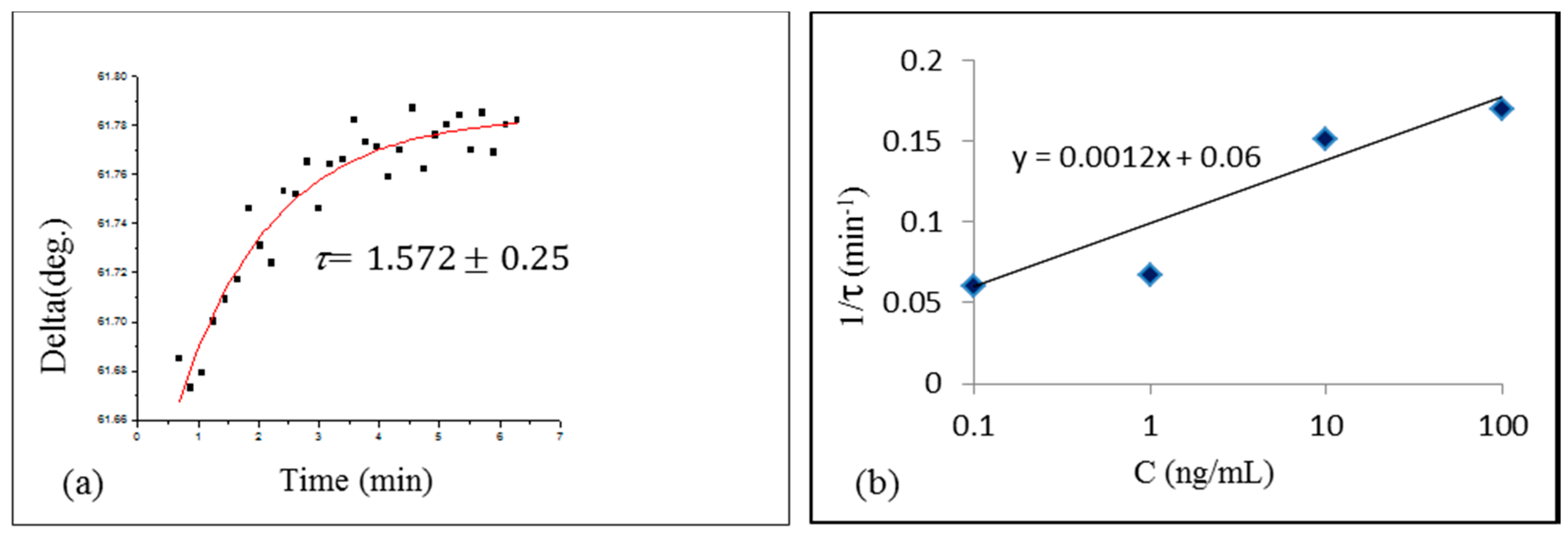
2.2. Study of Aflatoxin-Aptamer Binding Kinetics
3. Conclusions
4. Experimental Details
4.1. TIRE Experimental Set-Up
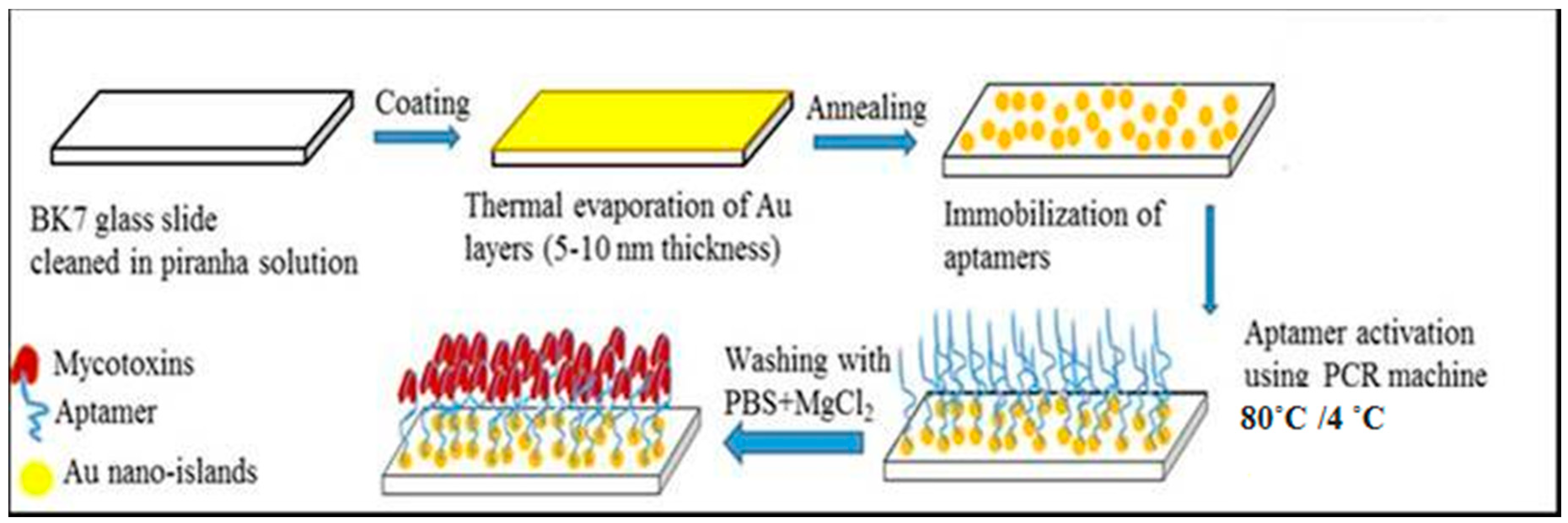
4.2. Preparation of LSPR Transducers
4.3. Aptamers and other Chemicals
4.4. Immobilization of Aptamers on Gold
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Peraica, M.; Radic, B.; Lucic, A.; Pavlovic, M. Toxic effects of mycotoxins in humans. Bull. World Health Organ. 1999, 77, 754–766. [Google Scholar] [PubMed]
- Worldwide Mycotoxin Regulations—Romer Labs. 2016. Available online: https://www.romerlabs.com/en/knowledge-center/knowledge-library/articles/news/worldwide-mycotoxin-regulations/ (accessed on 16 June 2016).
- Nabok, A. Comparative Studies on Optical Biosensors for Detection of Bio-Toxins. In Advanced Sciences and Technologies in Security Applications, Biosensors for Security and Bioterrorism Applications; Nikolelis, D.P., Nikoleli, G.P., Eds.; Springer: Cham, Switzerland, 2016; pp. 491–508. [Google Scholar]
- Nabok, A.V.; Tsargorodskaya, A.; Hassana, A.K.; Starodub, N.F. Total internal reflection ellipsometry and SPR detection of low molecular weight environmental toxins. Appl. Surf. Sci. 2005, 246, 381–386. [Google Scholar] [CrossRef]
- Nabok, A.; Tsargorodskaya, A. The method of total internal reflection ellipsometry for thin film characterisation and sensing. Thin Solid Films 2008, 516, 8993–9001. [Google Scholar] [CrossRef]
- Nabok, A.V.; Tsargorodskaya, A.; Holloway, A.; Starodub, N.F.; Gojster, O. Registration of T-2 mycotoxin with total internal reflection ellipsometry and QCM impedance methods. Biosensors. Bioelectron. 2007, 22, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Nabok, A.; Tsargorodskaya, A.; Mustafa, M.K.; Szekacs, I.; Starodub, N.F.; Szekacs, A. Detection of low molecular weight toxins using optical phase detection techniques. Procedia Chem. 2009, 1, 1491–1494. [Google Scholar] [CrossRef]
- Nabok, A.V.; Mustafa, M.K.; Tsargorodskaya, A.; Starodub, N.F. Detection of Aflatoxin B1 with a label free ellipsometry immunosensor. BioNanoScience 2011, 1, 38–45. [Google Scholar] [CrossRef]
- Lopatynskyi, A.M.; Lopatynska, O.G.; Guo, L.J.; Chegel, V.I. Localized surface plasmon resonance biosensor—Part I: Theoretical study of sensitivity—Extended Mie approach. IEEE Sens. 2011, 11, 361–369. [Google Scholar] [CrossRef]
- Karakouz, T.; Tesler, A.B.; Bendikov, T.A.; Vaskevich, A.; Rubinstein, I. Highly stable localized plasmon transducers obtained by thermal embedding of gold island films on glass. Adv. Mater. 2008, 20, 3893–3899. [Google Scholar] [CrossRef]
- Sepúlveda, B.; Angelomé, P.C.; Lechuga, L.M.; Liz-Marzán, L.M. LSPR-based nanobiosensors. Nano Today 2009, 4, 244–251. [Google Scholar] [CrossRef]
- Swierczewska, M.; Liu, G.; Lee, S.; Chen, X. High-sensitivity nanosensors for biomarker detection. Chem. Soc. Rev. 2011, 41, 2641–2655. [Google Scholar] [CrossRef] [PubMed]
- Al-Rubaye, A.G.; Nabok, A.; Tsargorodska, A. Spectroscopic ellipsometry study of gold nanostructures for LSPR bio-sensing applications. Sens. Bio-Sens. Res. 2017, 12, 30–35. [Google Scholar] [CrossRef]
- Doron-Mor, I.; Cohen, H.; Barkay, Z.; Shanzer, A.; Vaskevich, A.; Rubinstein, I. Sensitivity of transmission surface plasmon resonance (T-SPR) spectroscopy: Self-assembled multilayers on evaporated gold island films. Chem. Eur. J. 2005, 11, 5555–5562. [Google Scholar] [CrossRef] [PubMed]
- Al-Rubaye, A.; Nabok, A.; Abu-Ali, H.; Szekacs, A.; Takacs, E. LSPR/TIRE bio-sensing platform for detection of low molecular weight toxins. In Proceedings of the IEEE SENSORS, Glasgow, UK, 29 October–1 November 2017. [Google Scholar]
- Tombelli, S.; Minunni, M.; Mascini, M. Analytical applications of aptamers. Biosens. Bioelectron. 2005, 20, 2424–2434. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wang, W.; Chen, X.; Yu, X.; Wu, S.; Duan, N.; Wang, Z. Selection, identification, and application of Aflatoxin B1 aptamer. Eur. Food Res. Technol. 2014, 238, 919–925. [Google Scholar] [CrossRef]
- Rhouati, A.; Yang, C.; Hayat, A.; Marty, J.-L. Aptamers: A promising tool for Ochratoxin A detection in food analysis. Toxins 2013, 5, 1988–2008. [Google Scholar] [CrossRef] [PubMed]
- Al Rubaye, A.; Nabok, A.; Catanante, G.; Marty, J.L.; Takacs, E.; Szekacs, A. Detection of ochratoxin A in aptamer assay using total internal reflection ellipsometry. Sens. Actuators B Chem. 2018, 263, 248–251. [Google Scholar] [CrossRef]
- Malhotra, S.; Pandey, A.K.; Rajput, Y.S.; Sharma, R.J. Selection of aptamers for aflatoxin M1 and their characterization. J. Mol. Recognit. 2014, 27, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, S.; Obubuafo, A.; Soper, S.A.; Spivak, D.A. Surface immobilization methods for aptamer diagnostic applications. Anal. Bioanal. Chem. 2008, 390, 1009–1021. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghanim Al-Rubaye, A.; Nabok, A.; Catanante, G.; Marty, J.-L.; Takács, E.; Székács, A. Label-Free Optical Detection of Mycotoxins Using Specific Aptamers Immobilized on Gold Nanostructures. Toxins 2018, 10, 291. https://doi.org/10.3390/toxins10070291
Ghanim Al-Rubaye A, Nabok A, Catanante G, Marty J-L, Takács E, Székács A. Label-Free Optical Detection of Mycotoxins Using Specific Aptamers Immobilized on Gold Nanostructures. Toxins. 2018; 10(7):291. https://doi.org/10.3390/toxins10070291
Chicago/Turabian StyleGhanim Al-Rubaye, Ali, Alexei Nabok, Gaelle Catanante, Jean-Louis Marty, Eszter Takács, and András Székács. 2018. "Label-Free Optical Detection of Mycotoxins Using Specific Aptamers Immobilized on Gold Nanostructures" Toxins 10, no. 7: 291. https://doi.org/10.3390/toxins10070291
APA StyleGhanim Al-Rubaye, A., Nabok, A., Catanante, G., Marty, J.-L., Takács, E., & Székács, A. (2018). Label-Free Optical Detection of Mycotoxins Using Specific Aptamers Immobilized on Gold Nanostructures. Toxins, 10(7), 291. https://doi.org/10.3390/toxins10070291







