Deleting Death and Dialysis: Conservative Care of Cardio-Vascular Risk and Kidney Function Loss in Chronic Kidney Disease (CKD)
Abstract
1. Introduction
1.1. Traditional and Non-Traditional Risk Factors of Renal Cardiovascular Disease
1.2. Concept of This Publication
1.3. Search Strategy
2. Lifestyle Correction
2.1. Cessation of Smoking
2.2. Exercise
2.3. Reduction of Overweight and Obesity
2.4. Improving Environmental Factors
2.5. Conclusions
3. Dietary Interventions
3.1. Protein Restriction
3.2. Sodium Restriction
3.3. Potassium Rich Diet
3.4. Phosphorus Restriction
3.5. Enhanced Polyunsaturated Fatty Acid (PUFA) Intake
3.6. Sugar Intake Restriction
3.7. Correction of Intestinal Dysbiosis
3.8. Diet: General Conclusions
4. Pharmacological Treatment
4.1. Antihypertensive Treatment
4.1.1. Intensive Treatment
4.1.2. Angiotensin Converting Enzyme Inhibitors (ACEi) and Angiotensin Receptor Blockers (ARB)
4.1.3. Mineralocorticoid Receptor Antagonists
4.1.4. Hyperkalemia
4.1.5. Beta-Blockade
4.2. Interventions Other than Antihypertensive Treatment
4.2.1. Glycemia Control
4.2.2. Treatment of CKD-MBD
4.2.3. Treatment of Dyslipidemia
4.2.4. Anti-Inflammatory Therapies
4.2.5. Correction of Hypomagnesemia
4.2.6. Correction of Metabolic Acidosis
4.2.7. Aryl Hydrocarbon Receptor (AhR) Blockade
4.2.8. Anticoagulation
4.2.9. Prevention of Kidney Fibrosis
4.2.10. AST-120 (KremezinR)
4.3. Targeting Specific Uremic Retention Solutes
4.3.1. Advanced Glycation End Product (AGE) Reduction
4.3.2. Cytokine Antagonism
4.3.3. Endothelin A Receptor Blockade
4.3.4. Homocysteine Lowering
4.3.5. Uric Acid Lowering
4.3.6. Conclusion
5. General Conclusions
Conflicts of Interest
References
- Vanholder, R.; Fouque, D.; Glorieux, G.; Heine, G.H.; Kanbay, M.; Mallamaci, F.; Massy, Z.A.; Ortiz, A.; Rossignol, P.; Wiecek, A.; et al. Clinical management of the uraemic syndrome in chronic kidney disease. Lancet Diabetes Endocrinol. 2016, 4, 360–373. [Google Scholar] [CrossRef]
- Meyer, T.W.; Hostetter, T.H. Uremia. N. Engl. J. Med. 2007, 357, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.; Covic, A.; Fliser, D.; Fouque, D.; Goldsmith, D.; Kanbay, M.; Mallamaci, F.; Massy, Z.A.; Rossignol, P.; Vanholder, R.; et al. Epidemiology, contributors to, and clinical trials of mortality risk in chronic kidney failure. Lancet 2014, 383, 1831–1843. [Google Scholar] [CrossRef]
- Weiner, D.E.; Tighiouart, H.; Elsayed, E.F.; Griffith, J.L.; Salem, D.N.; Levey, A.S.; Sarnak, M.J. The Framingham predictive instrument in chronic kidney disease. J. Am. Coll. Cardiol. 2007, 50, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; Coresh, J.; Sang, Y.; Chalmers, J.; Fox, C.; Guallar, E.; Jafar, T.; Jassal, S.K.; Landman, G.W.; Muntner, P.; et al. Estimated glomerular filtration rate and albuminuria for prediction of cardiovascular outcomes: A collaborative meta-analysis of individual participant data. Lancet Diabetes Endocrinol. 2015. [Google Scholar] [CrossRef]
- Roberts, M.A.; Polkinghorne, K.R.; McDonald, S.P.; Ierino, F.L. Secular trends in cardiovascular mortality rates of patients receiving dialysis compared with the general population. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2011, 58, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.R. What are the key challenges we face in kidney transplantation today? Transplant. Res. 2013, 2, S1. [Google Scholar] [CrossRef] [PubMed]
- Chronic Kidney Disease Prognosis Consortium; Matsushita, K.; van der Velde, M.; Astor, B.C.; Woodward, M.; Levey, A.S.; de Jong, P.E.; Coresh, J.; Gansevoort, R.T. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 2010, 375, 2073–2081. [Google Scholar] [PubMed]
- Vanholder, R.; Massy, Z.; Argiles, A.; Spasovski, G.; Verbeke, F.; Lameire, N. Chronic kidney disease as cause of cardiovascular morbidity and mortality. Nephrol. Dial. Transplant. 2005, 20, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Jun, M.; Lv, J.; Perkovic, V.; Jardine, M.J. Managing cardiovascular risk in people with chronic kidney disease: A review of the evidence from randomized controlled trials. Ther. Adv. Chronic Dis. 2011, 2, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; Pletinck, A.; Schepers, E.; Glorieux, G. Biochemical and Clinical Impact of Organic Uremic Retention Solutes: A Comprehensive Update. Toxins 2018, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Vanmassenhove, J.; Vanholder, R.; Lameire, N. Points of Concern in Post Acute Kidney Injury Management. Nephron 2017. [Google Scholar] [CrossRef] [PubMed]
- Lameire, N.H.; Bagga, A.; Cruz, D.; De Maeseneer, J.; Endre, Z.; Kellum, J.A.; Liu, K.D.; Mehta, R.L.; Pannu, N.; Van Biesen, W.; et al. Acute kidney injury: An increasing global concern. Lancet 2013, 382, 170–179. [Google Scholar] [CrossRef]
- Joannidis, M.; Druml, W.; Forni, L.G.; Groeneveld, A.B.J.; Honore, P.M.; Hoste, E.; Ostermann, M.; Oudemans-van Straaten, H.M.; Schetz, M. Prevention of acute kidney injury and protection of renal function in the intensive care unit: Update 2017: Expert opinion of the Working Group on Prevention, AKI section, European Society of Intensive Care Medicine. Intensive Care Med. 2017, 43, 730–749. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; Annemans, L.; Brown, E.; Gansevoort, R.; Gout-Zwart, J.J.; Lameire, N.; Morton, R.L.; Oberbauer, R.; Postma, M.J.; Tonelli, M.; et al. Reducing the costs of chronic kidney disease while delivering quality health care: A call to action. Nat. Rev. Nephrol. 2017, 13, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, J.A.; Barua, R.S. The pathophysiology of cigarette smoking and cardiovascular disease: An update. J. Am. Coll. Cardiol. 2004, 43, 1731–1737. [Google Scholar] [CrossRef] [PubMed]
- Messner, B.; Bernhard, D. Smoking and cardiovascular disease: Mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Kassi, E.; Dalamaga, M.; Faviou, E.; Hroussalas, G.; Kazanis, K.; Nounopoulos, C.; Dionyssiou-Asteriou, A. Circulating oxidized LDL levels, current smoking and obesity in postmenopausal women. Atherosclerosis 2009, 205, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Maggi, E.; Bellazzi, R.; Falaschi, F.; Frattoni, A.; Perani, G.; Finardi, G.; Gazo, A.; Nai, M.; Romanini, D.; Bellomo, G. Enhanced LDL oxidation in uremic patients: An additional mechanism for accelerated atherosclerosis? Kidney Int. 1994, 45, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Elliot, S.J.; Karl, M.; Berho, M.; Xia, X.; Pereria-Simon, S.; Espinosa-Heidmann, D.; Striker, G.E. Smoking induces glomerulosclerosis in aging estrogen-deficient mice through cross-talk between TGF-beta1 and IGF-I signaling pathways. J. Am. Soc. Nephrol. 2006, 17, 3315–3324. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, R.; Habib, H.; Lahdo, A.; Al Ali, R.; Varjabedian, L.; Atalla, G.; Kassis Akl, N.; Aldakheel, S.; Alahdab, S.; Albitar, S. Association between smoking and chronic kidney disease: A case control study. BMC Public Health 2010, 10, 731. [Google Scholar] [CrossRef] [PubMed]
- Ejerblad, E.; Fored, C.M.; Lindblad, P.; Fryzek, J.; Dickman, P.W.; Elinder, C.G.; McLaughlin, J.K.; Nyren, O. Association between smoking and chronic renal failure in a nationwide population-based case-control study. J. Am. Soc. Nephrol. 2004, 15, 2178–2185. [Google Scholar] [CrossRef] [PubMed]
- Hallan, S.I.; Orth, S.R. Smoking is a risk factor in the progression to kidney failure. Kidney Int. 2011, 80, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Orth, S.R.; Stockmann, A.; Conradt, C.; Ritz, E.; Ferro, M.; Kreusser, W.; Piccoli, G.; Rambausek, M.; Roccatello, D.; Schafer, K.; et al. Smoking as a risk factor for end-stage renal failure in men with primary renal disease. Kidney Int. 1998, 54, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wang, L.; Ma, Z.; Zhong, L.; Wang, Y.; Gao, Y.; He, L.; Su, X. Cigarette smoking and chronic kidney disease in the general population: A systematic review and meta-analysis of prospective cohort studies. Nephrol. Dial. Transplant. 2017, 32, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; D’Agati, V.D. Nodular glomerulosclerosis in the nondiabetic smoker. J. Am. Soc. Nephrol. 2007, 18, 2032–2036. [Google Scholar] [CrossRef] [PubMed]
- Shinzawa, M.; Tanaka, S.; Tokumasu, H.; Takada, D.; Tsukamoto, T.; Yanagita, M.; Kawakami, K. Maternal Smoking during Pregnancy, Household Smoking after the Child’s Birth, and Childhood Proteinuria at Age 3 Years. Clin. J. Am. Soc. Nephrol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Nakagawa, H.; Murakami, Y.; Kitamura, A.; Kiyama, M.; Sakata, K.; Tsuji, I.; Miura, K.; Ueshima, H.; Okamura, T.; et al. Smoking increases the risk of all-cause and cardiovascular mortality in patients with chronic kidney disease. Kidney Int. 2015, 88, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Staplin, N.; Haynes, R.; Herrington, W.G.; Reith, C.; Cass, A.; Fellstrom, B.; Jiang, L.; Kasiske, B.L.; Krane, V.; Levin, A.; et al. Smoking and Adverse Outcomes in Patients With CKD: The Study of Heart and Renal Protection (SHARP). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 68, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Parrott, S.; Godfrey, C. Economics of smoking cessation. BMJ 2004, 328, 947–949. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, S.; Welte, R.; Beiche, A.; Koenig, H.H.; Buesch, K.; Leidl, R. Mortality, morbidity and costs attributable to smoking in Germany: Update and a 10-year comparison. Tob. Control 2006, 15, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Alam, M.F.; Jarvis, P.S. An analysis of the economic impact of smoking cessation in Europe. BMC Public Health 2013, 13, 390. [Google Scholar] [CrossRef] [PubMed]
- Van Baal, P.H.; Polder, J.J.; de Wit, G.A.; Hoogenveen, R.T.; Feenstra, T.L.; Boshuizen, H.C.; Engelfriet, P.M.; Brouwer, W.B. Lifetime medical costs of obesity: Prevention no cure for increasing health expenditure. PLoS Med. 2008, 5, e29. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.Y.; Schafferer, C.; Lee, J.M.; Ho, L.M.; Hsieh, C.J. The effects of a rise in cigarette price on cigarette consumption, tobacco taxation revenues, and of smoking-related deaths in 28 EU countries—Applying threshold regression modelling. BMC Public Health 2017, 17, 676. [Google Scholar] [CrossRef] [PubMed]
- Myers, J. Cardiology patient pages. Exercise and cardiovascular health. Circulation 2003, 107, e2–e5. [Google Scholar] [CrossRef] [PubMed]
- Dalal, H.M.; Doherty, P.; Taylor, R.S. Cardiac rehabilitation. BMJ 2015, 351, h5000. [Google Scholar] [CrossRef] [PubMed]
- Villella, M.; Villella, A. Exercise and cardiovascular diseases. Kidney Blood Press. Res. 2014, 39, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Goel, K.; Lennon, R.J.; Tilbury, R.T.; Squires, R.W.; Thomas, R.J. Impact of cardiac rehabilitation on mortality and cardiovascular events after percutaneous coronary intervention in the community. Circulation 2011, 123, 2344–2352. [Google Scholar] [CrossRef] [PubMed]
- Lubitz, J.; Cai, L.; Kramarow, E.; Lentzner, H. Health, life expectancy, and health care spending among the elderly. N. Engl. J. Med. 2003, 349, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Buttar, H.S.; Li, T.; Ravi, N. Prevention of cardiovascular diseases: Role of exercise, dietary interventions, obesity and smoking cessation. Exp. Clin. Cardiol. 2005, 10, 229–249. [Google Scholar] [PubMed]
- Garrett, S.; Elley, C.R.; Rose, S.B.; O’Dea, D.; Lawton, B.A.; Dowell, A.C. Are physical activity interventions in primary care and the community cost-effective? A systematic review of the evidence. Br. J. Gen. Pract. J. R. Coll. Gen. Pract. 2011, 61, e125–e133. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, A.J.; Critchley, J.A.; Tai, S.S.; Buckingham, K.; Westley, D.; Harridge, S.D.; Smith, C.; Gottlieb, J.M. Exercise Evaluation Randomised Trial (EXERT): A randomised trial comparing GP referral for leisure centre-based exercise, community-based walking and advice only. Health Technol. Assess. 2007, 11, 1–165. [Google Scholar] [CrossRef] [PubMed]
- Johansen, K.L.; Painter, P. Exercise in individuals with CKD. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 59, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Kosmadakis, G.C.; Bevington, A.; Smith, A.C.; Clapp, E.L.; Viana, J.L.; Bishop, N.C.; Feehally, J. Physical exercise in patients with severe kidney disease. Nephron Clin. Pract. 2010, 115, c7–c16. [Google Scholar] [CrossRef] [PubMed]
- Ikizler, T.A.; Robinson-Cohen, C.; Ellis, C.; Headley, S.A.E.; Tuttle, K.; Wood, R.J.; Evans, E.E.; Milch, C.M.; Moody, K.A.; Germain, M.; et al. Metabolic Effects of Diet and Exercise in Patients with Moderate to Severe CKD: A Randomized Clinical Trial. J. Am. Soc. Nephrol. 2018, 29, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Dunkler, D.; Kohl, M.; Heinze, G.; Teo, K.K.; Rosengren, A.; Pogue, J.; Gao, P.; Gerstein, H.; Yusuf, S.; Oberbauer, R.; et al. Modifiable lifestyle and social factors affect chronic kidney disease in high-risk individuals with type 2 diabetes mellitus. Kidney Int. 2015, 87, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Dunkler, D.; Kohl, M.; Teo, K.K.; Heinze, G.; Dehghan, M.; Clase, C.M.; Gao, P.; Yusuf, S.; Mann, J.F.; Oberbauer, R.; et al. Population-Attributable Fractions of Modifiable Lifestyle Factors for CKD and Mortality in Individuals With Type 2 Diabetes: A Cohort Study. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 68, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Barcellos, F.C.; Santos, I.S.; Umpierre, D.; Bohlke, M.; Hallal, P.C. Effects of exercise in the whole spectrum of chronic kidney disease: A systematic review. Clin. Kidney J. 2015, 8, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Heiwe, S.; Jacobson, S.H. Exercise training in adults with CKD: A systematic review and meta-analysis. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2014, 64, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and cardiovascular disease: Pathophysiology, evaluation, and effect of weight loss: An update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation 2006, 113, 898–918. [Google Scholar] [PubMed]
- Lavie, C.J.; Milani, R.V.; Ventura, H.O. Obesity and cardiovascular disease: Risk factor, paradox, and impact of weight loss. J. Am. Coll. Cardiol. 2009, 53, 1925–1932. [Google Scholar] [CrossRef] [PubMed]
- Burke, G.L.; Bertoni, A.G.; Shea, S.; Tracy, R.; Watson, K.E.; Blumenthal, R.S.; Chung, H.; Carnethon, M.R. The impact of obesity on cardiovascular disease risk factors and subclinical vascular disease: The Multi-Ethnic Study of Atherosclerosis. Arch. Intern. Med. 2008, 168, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Schneider, H.; Golay, A.; Keller, U. Economic burden of obesity and its comorbidities in Switzerland. Soz. Praventivmed. 2005, 50, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Anis, A.H.; Zhang, W.; Bansback, N.; Guh, D.P.; Amarsi, Z.; Birmingham, C.L. Obesity and overweight in Canada: An updated cost-of-illness study. Obes. Rev. Off. J. Int. Assoc. Stud. Obes. 2010, 11, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Verhaeghe, N.; De Greve, O.; Annemans, L. The potential health and economic effect of a Body Mass Index decrease in the overweight and obese population in Belgium. Public Health 2016, 134, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Juul, F.; Hemmingsson, E. Trends in consumption of ultra-processed foods and obesity in Sweden between 1960 and 2010. Public Health Nutr. 2015, 18, 3096–3107. [Google Scholar] [CrossRef] [PubMed]
- Cummings, D.E.; Cohen, R.V. Bariatric/Metabolic Surgery to Treat Type 2 Diabetes in Patients With a BMI <35 kg/m2. Diabetes Care 2016, 39, 924–933. [Google Scholar]
- Courcoulas, A.P.; Yanovski, S.Z.; Bonds, D.; Eggerman, T.L.; Horlick, M.; Staten, M.A.; Arterburn, D.E. Long-term outcomes of bariatric surgery: A National Institutes of Health symposium. JAMA Surg. 2014, 149, 1323–1329. [Google Scholar] [CrossRef] [PubMed]
- Johansson, K.; Sundstrom, J.; Neovius, K.; Rossner, S.; Neovius, M. Long-term changes in blood pressure following orlistat and sibutramine treatment: A meta-analysis. Obes. Rev. Off. J. Int. Assoc. Stud. Obes. 2010, 11, 777–791. [Google Scholar] [CrossRef] [PubMed]
- Yanovski, S.Z.; Yanovski, J.A. Long-term drug treatment for obesity: A systematic and clinical review. JAMA 2014, 311, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Danaei, G.; Lu, Y.; Singh, G.; Stevens, G.; Cowan, M.; Farzadfar, F.; Lin, J.; Finucane, M.; Rao, M.; Khang, Y.; et al. Cardiovascular disease, chronic kidney disease, and diabetes mortality burden of cardiometabolic risk factors from 1980 to 2010: A comparative risk assessment. Lancet Diabetes Endocrinol. 2014, 2, 634–647. [Google Scholar]
- Hall, M.E.; do Carmo, J.M.; da Silva, A.A.; Juncos, L.A.; Wang, Z.; Hall, J.E. Obesity, hypertension, and chronic kidney disease. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Babayev, R.; Whaley-Connell, A.; Kshirsagar, A.; Klemmer, P.; Navaneethan, S.; Chen, S.C.; Li, S.; McCullough, P.A.; Bakris, G.; Bomback, A.; et al. Association of race and body mass index with ESRD and mortality in CKD stages 3–4: Results from the Kidney Early Evaluation Program (KEEP). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2013, 61, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.L.; Molnar, M.Z.; Naseer, A.; Mikkelsen, M.K.; Kalantar-Zadeh, K.; Kovesdy, C.P. Association of age and BMI with kidney function and mortality: A cohort study. Lancet Diabetes Endocrinol. 2015, 3, 704–714. [Google Scholar] [CrossRef]
- Navaneethan, S.D.; Yehnert, H.; Moustarah, F.; Schreiber, M.J.; Schauer, P.R.; Beddhu, S. Weight loss interventions in chronic kidney disease: A systematic review and meta-analysis. Clin. J. Am. Soc. Nephrol. 2009, 4, 1565–1574. [Google Scholar] [CrossRef] [PubMed]
- Bolignano, D.; Zoccali, C. Effects of weight loss on renal function in obese CKD patients: A systematic review. Nephrol. Dial. Transplant. 2013, 28 (Suppl. 4), iv82–iv98. [Google Scholar] [CrossRef] [PubMed]
- Friedman, A.N.; Wahed, A.S.; Wang, J.; Courcoulas, A.P.; Dakin, G.; Hinojosa, M.W.; Kimmel, P.L.; Mitchell, J.E.; Pomp, A.; Pories, W.J.; et al. Effect of Bariatric Surgery on CKD Risk. J. Am. Soc. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Ahmadi, S.F.; Streja, E.; Molnar, M.Z.; Flegal, K.M.; Gillen, D.; Kovesdy, C.P.; Kalantar-Zadeh, K. Obesity paradox in end-stage kidney disease patients. Prog. Cardiovasc. Dis. 2014, 56, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Chrysant, S.G.; Chrysant, G.S. New insights into the true nature of the obesity paradox and the lower cardiovascular risk. J. Am. Soc. Hypertens. JASH 2013, 7, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.K.; Lock, K.; Teo, K.; Subramanian, S.V.; McKee, M.; Yusuf, S. Environmental and societal influences acting on cardiovascular risk factors and disease at a population level: A review. Int. J. Epidemiol. 2009, 38, 1580–1594. [Google Scholar] [CrossRef] [PubMed]
- Soderland, P.; Lovekar, S.; Weiner, D.E.; Brooks, D.R.; Kaufman, J.S. Chronic kidney disease associated with environmental toxins and exposures. Adv. Chronic Kidney Dis. 2010, 17, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Bowe, B.; Xie, Y.; Li, T.; Yan, Y.; Xian, H.; Al-Aly, Z. Particulate Matter Air Pollution and the Risk of Incident CKD and Progression to ESRD. J. Am. Soc. Nephrol. 2018, 29, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Jayasumana, C.; Gunatilake, S.; Senanayake, P. Glyphosate, hard water and nephrotoxic metals: Are they the culprits behind the epidemic of chronic kidney disease of unknown etiology in Sri Lanka? Int. J. Environ. Res. Public Health 2014, 11, 2125–2147. [Google Scholar] [CrossRef] [PubMed]
- Diamond, J.R. Effects of dietary interventions on glomerular pathophysiology. Am. J. Physiol. 1990, 258, F1–F8. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Aparicio, M. Eleven reasons to control the protein intake of patients with chronic kidney disease. Nat. Clin. Pract. Nephrol. 2007, 3, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Klahr, S.; Levey, A.S.; Beck, G.J.; Caggiula, A.W.; Hunsicker, L.; Kusek, J.W.; Striker, G. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of Diet in Renal Disease Study Group. N. Engl. J. Med. 1994, 330, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Greene, T.; Sarnak, M.J.; Wang, X.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; Kopple, J.D. Effect of dietary protein restriction on the progression of kidney disease: Long-term follow-up of the Modification of Diet in Renal Disease (MDRD) Study. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2006, 48, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Kopple, J.D.; Wang, X.; Beck, G.J.; Collins, A.J.; Kusek, J.W.; Greene, T.; Levey, A.S.; Sarnak, M.J. Effect of a very low-protein diet on outcomes: Long-term follow-up of the Modification of Diet in Renal Disease (MDRD) Study. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2009, 53, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Ikizler, T.A.; Greene, J.H.; Wingard, R.L.; Parker, R.A.; Hakim, R.M. Spontaneous dietary protein intake during progression of chronic renal failure. J. Am. Soc. Nephrol. 1995, 6, 1386–1391. [Google Scholar] [PubMed]
- Cianciaruso, B.; Pota, A.; Bellizzi, V.; Di Giuseppe, D.; Di Micco, L.; Minutolo, R.; Pisani, A.; Sabbatini, M.; Ravani, P. Effect of a low- versus moderate-protein diet on progression of CKD: Follow-up of a randomized controlled trial. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2009, 54, 1052–1061. [Google Scholar] [CrossRef] [PubMed]
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-Supplemented Vegetarian Very Low-Protein Diet and CKD Progression. J. Am. Soc. Nephrol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bellizzi, V.; Di Iorio, B.R.; De Nicola, L.; Minutolo, R.; Zamboli, P.; Trucillo, P.; Catapano, F.; Cristofano, C.; Scalfi, L.; Conte, G.; et al. Very low protein diet supplemented with ketoanalogs improves blood pressure control in chronic kidney disease. Kidney Int. 2007, 71, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.R.; Di Micco, L.; Marzocco, S.; De Simone, E.; De Blasio, A.; Sirico, M.L.; Nardone, L.; Ubi Study Group. Very Low-Protein Diet (VLPD) Reduces Metabolic Acidosis in Subjects with Chronic Kidney Disease: The “Nutritional Light Signal” of the Renal Acid Load. Nutrients 2017, 9, 69. [Google Scholar] [CrossRef] [PubMed]
- Nezu, U.; Kamiyama, H.; Kondo, Y.; Sakuma, M.; Morimoto, T.; Ueda, S. Effect of low-protein diet on kidney function in diabetic nephropathy: Meta-analysis of randomised controlled trials. BMJ Open 2013, 3, e002934. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Laville, M. Low protein diets for chronic kidney disease in non diabetic adults. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef] [PubMed]
- Clase, C.M.; Smyth, A. Chronic kidney disease. BMJ Clin. Evid. 2015, 6, 2004. [Google Scholar]
- Chaturvedi, S.; Jones, C. Protein restriction for children with chronic renal failure. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Kalantar-Zadeh, K.; Kopple, J.; Cano, N.; Chauveau, P.; Cuppari, L.; Franch, H.; Guarnieri, G.; Ikizler, T.A.; Kaysen, G.; et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney Int. 2008, 73, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Noce, A.; Vidiri, M.F.; Marrone, G.; Moriconi, E.; Bocedi, A.; Capria, A.; Rovella, V.; Ricci, G.; De Lorenzo, A.; Di Daniele, N. Is low-protein diet a possible risk factor of malnutrition in chronic kidney disease patients? Cell Death Discov. 2016, 2, 16026. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Hu, F.B.; Curhan, G.C. Associations of diet with albuminuria and kidney function decline. Clin. J. Am. Soc. Nephrol. 2010, 5, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Hai, X.; Landeras, V.; Dobre, M.A.; DeOreo, P.; Meyer, T.W.; Hostetter, T.H. Mechanism of Prominent Trimethylamine Oxide (TMAO) Accumulation in Hemodialysis Patients. PLoS ONE 2015, 10, e0143731. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.T.; Ramezani, A.; Manal, A.; Raj, D.S. Trimethylamine N-Oxide: The Good, the Bad and the Unknown. Toxins 2016, 8, 326. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Atabak, S.; Esmaillzadeh, A. Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy: A longitudinal randomized clinical trial. Diabetes Care 2008, 31, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Goraya, N.; Simoni, J.; Jo, C.H.; Wesson, D.E. A comparison of treating metabolic acidosis in CKD stage 4 hypertensive kidney disease with fruits and vegetables or sodium bicarbonate. Clin. J. Am. Soc. Nephrol. 2013, 8, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Scialla, J.J.; Appel, L.J.; Wolf, M.; Yang, W.; Zhang, X.; Sozio, S.M.; Miller, E.R., 3rd; Bazzano, L.A.; Cuevas, M.; Glenn, M.J.; et al. Plant protein intake is associated with fibroblast growth factor 23 and serum bicarbonate levels in patients with chronic kidney disease: The Chronic Renal Insufficiency Cohort study. J. Ren. Nutr. 2012, 22, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Iwase, M.; Ohkuma, T.; Ogata-Kaizu, S.; Ide, H.; Kikuchi, Y.; Idewaki, Y.; Joudai, T.; Hirakawa, Y.; Uchida, K.; et al. Impact of dietary fiber intake on glycemic control, cardiovascular risk factors and chronic kidney disease in Japanese patients with type 2 diabetes mellitus: The Fukuoka Diabetes Registry. Nutr. J. 2013, 12, 159. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, V.M.; Wei, G.; Baird, B.C.; Murtaugh, M.; Chonchol, M.B.; Raphael, K.L.; Greene, T.; Beddhu, S. High dietary fiber intake is associated with decreased inflammation and all-cause mortality in patients with chronic kidney disease. Kidney Int. 2012, 81, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.P.; Luo, F.J.; Plummer, N.S.; Hostetter, T.H.; Meyer, T.W. The production of p-cresol sulfate and indoxyl sulfate in vegetarians versus omnivores. Clin. J. Am. Soc. Nephrol. 2012, 7, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Salmean, Y.A.; Segal, M.S.; Palii, S.P.; Dahl, W.J. Fiber supplementation lowers plasma p-cresol in chronic kidney disease patients. J. Ren. Nutr. 2015, 25, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Kandouz, S.; Mohamed, A.S.; Zheng, Y.; Sandeman, S.; Davenport, A. Reduced protein bound uraemic toxins in vegetarian kidney failure patients treated by haemodiafiltration. Hemodial. Int. 2016, 20, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Rysz, J.; Franczyk, B.; Cialkowska-Rysz, A.; Gluba-Brzozka, A. The Effect of Diet on the Survival of Patients with Chronic Kidney Disease. Nutrients 2017, 9, 495. [Google Scholar] [CrossRef] [PubMed]
- Chauveau, P.; Aparicio, M.; Bellizzi, V.; Campbell, K.; Hong, X.; Johansson, L.; Kolko, A.; Molina, P.; Sezer, S.; Wanner, C.; et al. Mediterranean diet as the diet of choice for patients with chronic kidney disease. Nephrol. Dial. Transplant. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Vennegoor, M.; ter Wee, P.; Wanner, C.; Basci, A.; Canaud, B.; Haage, P.; Konner, K.; Kooman, J.; Martin-Malo, A.; et al. EBPG guideline on nutrition. Nephrol. Dial. Transplant. 2007, 22 (Suppl. 2), ii45–ii87. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Yusuf, S.; Mente, A.; Gao, P.; Mann, J.F.; Teo, K.; McQueen, M.; Sleight, P.; Sharma, A.M.; Dans, A.; et al. Urinary sodium and potassium excretion and risk of cardiovascular events. JAMA 2011, 306, 2229–2238. [Google Scholar] [CrossRef] [PubMed]
- Mente, A.; O’Donnell, M.; Rangarajan, S.; Dagenais, G.; Lear, S.; McQueen, M.; Diaz, R.; Avezum, A.; Lopez-Jaramillo, P.; Lanas, F.; et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: A pooled analysis of data from four studies. Lancet 2016, 388, 465–475. [Google Scholar] [CrossRef]
- Stolarz-Skrzypek, K.; Kuznetsova, T.; Thijs, L.; Tikhonoff, V.; Seidlerova, J.; Richart, T.; Jin, Y.; Olszanecka, A.; Malyutina, S.; Casiglia, E.; et al. Fatal and nonfatal outcomes, incidence of hypertension, and blood pressure changes in relation to urinary sodium excretion. JAMA 2011, 305, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.C.; Moran, J.; Forsblom, C.; Harjutsalo, V.; Thorn, L.; Ahola, A.; Waden, J.; Tolonen, N.; Saraheimo, M.; Gordin, D.; et al. The association between dietary sodium intake, ESRD, and all-cause mortality in patients with type 1 diabetes. Diabetes Care 2011, 34, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Adrogue, H.J.; Madias, N.E. Sodium and potassium in the pathogenesis of hypertension. N. Engl. J. Med. 2007, 356, 1966–1978. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Tighiouart, H.; Levey, A.S.; Beck, G.J.; Sarnak, M.J. Urinary sodium excretion and kidney failure in nondiabetic chronic kidney disease. Kidney Int. 2014, 86, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Dunkler, D.; Gao, P.; Teo, K.K.; Yusuf, S.; O’Donnell, M.J.; Mann, J.F.; Clase, C.M.; Ontarget and investigators. The relationship between estimated sodium and potassium excretion and subsequent renal outcomes. Kidney Int. 2014, 86, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Li, Y.; Yang, Z.; Luo, J. Low dietary sodium intake increases the death risk in peritoneal dialysis. Clin. J. Am. Soc. Nephrol. 2010, 5, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Lambers Heerspink, H.J.; Holtkamp, F.A.; Parving, H.H.; Navis, G.J.; Lewis, J.B.; Ritz, E.; de Graeff, P.A.; de Zeeuw, D. Moderation of dietary sodium potentiates the renal and cardiovascular protective effects of angiotensin receptor blockers. Kidney Int. 2012, 82, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Slagman, M.C.; Waanders, F.; Hemmelder, M.H.; Woittiez, A.J.; Janssen, W.M.; Lambers Heerspink, H.J.; Navis, G.; Laverman, G.D. Moderate dietary sodium restriction added to angiotensin converting enzyme inhibition compared with dual blockade in lowering proteinuria and blood pressure: Randomised controlled trial. BMJ 2011, 343, d4366. [Google Scholar] [CrossRef] [PubMed]
- Vegter, S.; Perna, A.; Postma, M.J.; Navis, G.; Remuzzi, G.; Ruggenenti, P. Sodium intake, ACE inhibition, and progression to ESRD. J. Am. Soc. Nephrol. 2012, 23, 165–173. [Google Scholar] [CrossRef] [PubMed]
- McMahon, E.J.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Johnson, D.W.; Campbell, K.L. A randomized trial of dietary sodium restriction in CKD. J. Am. Soc. Nephrol. 2013, 24, 2096–2103. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., III; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.L.; Johnson, D.W.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Whitehead, J.P.; Dimeski, G.; McMahon, E. A randomized trial of sodium-restriction on kidney function, fluid volume and adipokines in CKD patients. BMC Nephrol. 2014, 15, 57. [Google Scholar] [CrossRef] [PubMed]
- Parving, H.H.; Lehnert, H.; Brochner-Mortensen, J.; Gomis, R.; Andersen, S.; Arner, P. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N. Engl. J. Med. 2001, 345, 870–878. [Google Scholar] [CrossRef] [PubMed]
- McMahon, E.J.; Campbell, K.L.; Bauer, J.D.; Mudge, D.W. Altered dietary salt intake for people with chronic kidney disease. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Jones-Burton, C.; Mishra, S.I.; Fink, J.C.; Brown, J.; Gossa, W.; Bakris, G.L.; Weir, M.R. An in-depth review of the evidence linking dietary salt intake and progression of chronic kidney disease. Am. J. Nephrol. 2006, 26, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Cupisti, A.; D’Alessandro, C.; Valeri, A.; Capitanini, A.; Meola, M.; Betti, G.; Barsotti, G. Food intake and nutritional status in stable hemodialysis patients. Ren. Fail. 2010, 32, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Montford, J.R.; Linas, S. How Dangerous Is Hyperkalemia? J. Am. Soc. Nephrol. 2017, 28, 3155–3165. [Google Scholar] [CrossRef] [PubMed]
- Tyson, C.C.; Kuchibhatla, M.; Patel, U.D.; Pun, P.H.; Chang, A.; Nwankwo, C.; Joseph, M.A.; Svetkey, L.P. Impact of Kidney Function on Effects of the Dietary Approaches to Stop Hypertension (Dash) Diet. J. Hypertens. Open Access 2014, 3, 1000168. [Google Scholar]
- Calvo, M.S.; Moshfegh, A.J.; Tucker, K.L. Assessing the health impact of phosphorus in the food supply: Issues and considerations. Adv. Nutr. 2014, 5, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.; Sayre, S.S.; Leon, J.B.; Machekano, R.; Love, T.E.; Porter, D.; Marbury, M.; Sehgal, A.R. Effect of food additives on hyperphosphatemia among patients with end-stage renal disease: A randomized controlled trial. JAMA 2009, 301, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Chue, C.D.; Edwards, N.C.; Davis, L.J.; Steeds, R.P.; Townend, J.N.; Ferro, C.J. Serum phosphate but not pulse wave velocity predicts decline in renal function in patients with early chronic kidney disease. Nephrol. Dial. Transplant. 2011, 26, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Ruggenenti, P.; Perna, A.; Leonardis, D.; Tripepi, R.; Tripepi, G.; Mallamaci, F.; Remuzzi, G.; Group, R.S. Phosphate may promote CKD progression and attenuate renoprotective effect of ACE inhibition. J. Am. Soc. Nephrol. 2011, 22, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.R.; Bellizzi, V.; Bellasi, A.; Torraca, S.; D’Arrigo, G.; Tripepi, G.; Zoccali, C. Phosphate attenuates the anti-proteinuric effect of very low-protein diet in CKD patients. Nephrol. Dial. Transplant. 2013, 28, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Sim, J.J.; Bhandari, S.K.; Smith, N.; Chung, J.; Liu, I.L.; Jacobsen, S.J.; Kalantar-Zadeh, K. Phosphorus and risk of renal failure in subjects with normal renal function. Am. J. Med. 2013, 126, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Lynch, K.E.; Lynch, R.; Curhan, G.C.; Brunelli, S.M. Prescribed dietary phosphate restriction and survival among hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 620–629. [Google Scholar] [CrossRef] [PubMed]
- Shinaberger, C.S.; Greenland, S.; Kopple, J.D.; Van Wyck, D.; Mehrotra, R.; Kovesdy, C.P.; Kalantar-Zadeh, K. Is controlling phosphorus by decreasing dietary protein intake beneficial or harmful in persons with chronic kidney disease? Am. J. Clin. Nutr. 2008, 88, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B.; et al. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed]
- Mollsten, A.V.; Dahlquist, G.G.; Stattin, E.L.; Rudberg, S. Higher intakes of fish protein are related to a lower risk of microalbuminuria in young Swedish type 1 diabetic patients. Diabetes Care 2001, 24, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Guebre-Egziabher, F.; Debard, C.; Drai, J.; Denis, L.; Pesenti, S.; Bienvenu, J.; Vidal, H.; Laville, M.; Fouque, D. Differential dose effect of fish oil on inflammation and adipose tissue gene expression in chronic kidney disease patients. Nutrition 2013, 29, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, K.; Terashima, Y.; Itomura, M.; Sawazaki, S.; Inagaki, H.; Kuroda, M.; Tomita, S.; Hirata, H.; Inadera, H.; Hamazaki, T. Docosahexaenoic acid is an independent predictor of all-cause mortality in hemodialysis patients. Am. J. Nephrol. 2011, 33, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Terashima, Y.; Hamazaki, K.; Itomura, M.; Tomita, S.; Kuroda, M.; Hirata, H.; Hamazaki, T.; Inadera, H. Inverse association between docosahexaenoic acid and mortality in patients on hemodialysis during over 10 years. Hemodial. Int. 2014, 18, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Kutner, N.G.; Clow, P.W.; Zhang, R.; Aviles, X. Association of fish intake and survival in a cohort of incident dialysis patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2002, 39, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Okano, K.; Tsuruta, Y.; Tsuruta, Y.; Tsuchiya, K.; Akiba, T.; Nitta, K. Eicosapentaenoic Acid (EPA) Decreases the All-Cause Mortality in Hemodialysis Patients. Intern. Med. 2015, 54, 3133–3137. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Li, M.S.; Lin, M.; Zhao, T.Y.; Gao, P. Effect of fish oil supplement in maintenance hemodialysis patients: A systematic review and meta-analysis of published randomized controlled trials. Eur. J. Clin. Pharmacol. 2016, 72, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Svensson, M.; Schmidt, E.B.; Jorgensen, K.A.; Christensen, J.H.; Group, O.S. N-3 fatty acids as secondary prevention against cardiovascular events in patients who undergo chronic hemodialysis: A randomized, placebo-controlled intervention trial. Clin. J. Am. Soc. Nephrol. 2006, 1, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Wang, L.N. omega-3 fatty acids therapy for IgA nephropathy: A meta-analysis of randomized controlled trials. Clin. Nephrol. 2012, 77, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.H.; Chiou, Y.Y.; Hung, P.H.; Chiang, P.C.; Wang, S.T. Omega-3 fatty acids ameliorate proteinuria but not renal function in IgA nephropathy: A meta-analysis of randomized controlled trials. Nephron Clin. Pract. 2012, 121, c30–c35. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Curhan, G.C. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin. J. Am. Soc. Nephrol. 2011, 6, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Yuzbashian, E.; Asghari, G.; Hosseinpour-Niazi, S.; Azizi, F. Consumption of sugar sweetened beverage is associated with incidence of metabolic syndrome in Tehranian children and adolescents. Nutr. Metab. 2015, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Cheungpasitporn, W.; Thongprayoon, C.; O’Corragain, O.A.; Edmonds, P.J.; Kittanamongkolchai, W.; Erickson, S.B. Associations of sugar-sweetened and artificially sweetened soda with chronic kidney disease: A systematic review and meta-analysis. Nephrology 2014, 19, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Dwyer, J.; Fulgoni, V.L., III; King, J.C.; Leveille, G.A.; MacDonald, R.S.; Ordovas, J.; Schnakenberg, D. Processed foods: Contributions to nutrition. Am. J. Clin. Nutr. 2014, 99, 1525–1542. [Google Scholar] [CrossRef] [PubMed]
- Meier, T.; Senftleben, K.; Deumelandt, P.; Christen, O.; Riedel, K.; Langer, M. Healthcare Costs Associated with an Adequate Intake of Sugars, Salt and Saturated Fat in Germany: A Health Econometrical Analysis. PLoS ONE 2015, 10, e0135990. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Sanchez-Lozada, L.G.; Nakagawa, T. The effect of fructose on renal biology and disease. J. Am. Soc. Nephrol. 2010, 21, 2036–2039. [Google Scholar] [CrossRef] [PubMed]
- Brymora, A.; Flisinski, M.; Johnson, R.J.; Goszka, G.; Stefanska, A.; Manitius, J. Low-fructose diet lowers blood pressure and inflammation in patients with chronic kidney disease. Nephrol. Dial. Transplant. 2012, 27, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.D.; Sievenpiper, J.L.; de Souza, R.J.; Chiavaroli, L.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Di Buono, M.; et al. The effects of fructose intake on serum uric acid vary among controlled dietary trials. J. Nutr. 2012, 142, 916–923. [Google Scholar] [CrossRef] [PubMed]
- Wagner, Z.; Molnar, M.; Molnar, G.A.; Tamasko, M.; Laczy, B.; Wagner, L.; Csiky, B.; Heidland, A.; Nagy, J.; Wittmann, I. Serum carboxymethyllysine predicts mortality in hemodialysis patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2006, 47, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Schwedler, S.B.; Metzger, T.; Schinzel, R.; Wanner, C. Advanced glycation end products and mortality in hemodialysis patients. Kidney Int. 2002, 62, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Peppa, M.; Cai, W.; Goldberg, T.; Lu, M.; He, C.; Vlassara, H. Restriction of dietary glycotoxins reduces excessive advanced glycation end products in renal failure patients. J. Am. Soc. Nephrol. 2003, 14, 728–731. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Cai, W.; Goodman, S.; Pyzik, R.; Yong, A.; Chen, X.; Zhu, L.; Neade, T.; Beeri, M.; Silverman, J.M.; et al. Protection against loss of innate defenses in adulthood by low advanced glycation end products (AGE) intake: Role of the antiinflammatory AGE receptor-1. J. Clin. Endocrinol. Metab. 2009, 94, 4483–4491. [Google Scholar] [CrossRef] [PubMed]
- Harcourt, B.E.; Sourris, K.C.; Coughlan, M.T.; Walker, K.Z.; Dougherty, S.L.; Andrikopoulos, S.; Morley, A.L.; Thallas-Bonke, V.; Chand, V.; Penfold, S.A.; et al. Targeted reduction of advanced glycation improves renal function in obesity. Kidney Int. 2011, 80, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Johnson, D.W.; Morrison, M.; Pascoe, E.M.; Coombes, J.S.; Forbes, J.M.; Szeto, C.C.; McWhinney, B.C.; Ungerer, J.P.; Campbell, K.L. Synbiotics Easing Renal Failure by Improving Gut Microbiology (SYNERGY): A Randomized Trial. Clin. J. Am. Soc. Nephrol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sirich, T.L.; Plummer, N.S.; Gardner, C.D.; Hostetter, T.H.; Meyer, T.W. Effect of increasing dietary fiber on plasma levels of colon-derived solutes in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2014, 9, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Bliss, D.Z.; Stein, T.P.; Schleifer, C.R.; Settle, R.G. Supplementation with gum arabic fiber increases fecal nitrogen excretion and lowers serum urea nitrogen concentration in chronic renal failure patients consuming a low-protein diet. Am. J. Clin. Nutr. 1996, 63, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, A.; Zarrati Mojarrad, M.; Bahmani, F.; Taghizadeh, M.; Ramezani, M.; Tajabadi-Ebrahimi, M.; Jafari, P.; Esmaillzadeh, A.; Asemi, Z. Probiotic supplementation in diabetic hemodialysis patients has beneficial metabolic effects. Kidney Int. 2017, 91, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Poesen, R.; Evenepoel, P.; de Loor, H.; Delcour, J.A.; Courtin, C.M.; Kuypers, D.; Augustijns, P.; Verbeke, K.; Meijers, B. The Influence of Prebiotic Arabinoxylan Oligosaccharides on Microbiota Derived Uremic Retention Solutes in Patients with Chronic Kidney Disease: A Randomized Controlled Trial. PLoS ONE 2016, 11, e0153893. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.K.; Wu, Y.Y.; Yang, Y.F.; Ting, I.W.; Lin, C.C.; Yen, T.H.; Chen, J.H.; Wang, C.H.; Huang, C.C.; Lin, H.C. The effect of probiotics on serum levels of cytokine and endotoxin in peritoneal dialysis patients: A randomised, double-blind, placebo-controlled trial. Benef. Microbes 2015, 6, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Guida, B.; Germano, R.; Trio, R.; Russo, D.; Memoli, B.; Grumetto, L.; Barbato, F.; Cataldi, M. Effect of short-term synbiotic treatment on plasma p-cresol levels in patients with chronic renal failure: A randomized clinical trial. Nutr. Metab. Cardiovasc. Dis. NMCD 2014, 24, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Pavan, M. Influence of prebiotic and probiotic supplementation on the progression of chronic kidney disease. Minerva Urol. Nefrol. Ital. J. Urol. Nephrol. 2016, 68, 222–226. [Google Scholar]
- Snelson, M.; Clarke, R.E.; Coughlan, M.T. Stirring the Pot: Can Dietary Modification Alleviate the Burden of CKD? Nutrients 2017, 9, 265. [Google Scholar] [CrossRef] [PubMed]
- Laville, M.; Fouque, D. Nutritional aspects in hemodialysis. Kidney Int. 2000, 76, S133–S139. [Google Scholar] [CrossRef]
- Blood Pressure Lowering Treatment Trialists’ Collaboration. Blood pressure-lowering treatment based on cardiovascular risk: A meta-analysis of individual patient data. Lancet 2014, 384, 591–598. [Google Scholar]
- Xie, X.; Atkins, E.; Lv, J.; Bennett, A.; Neal, B.; Ninomiya, T.; Woodward, M.; MacMahon, S.; Turnbull, F.; Hillis, G.S.; et al. Effects of intensive blood pressure lowering on cardiovascular and renal outcomes: Updated systematic review and meta-analysis. Lancet 2016, 387, 435–443. [Google Scholar] [CrossRef]
- Verbeke, F.; Lindley, E.; Van Bortel, L.; Vanholder, R.; London, G.; Cochat, P.; Wiecek, A.; Fouque, D.; Van Biesen, W. A European Renal Best Practice (ERBP) position statement on the Kidney Disease: Improving Global Outcomes (KDIGO) clinical practice guideline for the management of blood pressure in non-dialysis-dependent chronic kidney disease: An endorsement with some caveats for real-life application. Nephrol. Dial. Transplant. 2014, 29, 490–496. [Google Scholar] [PubMed]
- Appel, L.J.; Wright, J.T., Jr.; Greene, T.; Agodoa, L.Y.; Astor, B.C.; Bakris, G.L.; Cleveland, W.H.; Charleston, J.; Contreras, G.; Faulkner, M.L.; et al. Intensive blood-pressure control in hypertensive chronic kidney disease. N. Engl. J. Med. 2010, 363, 918–929. [Google Scholar] [CrossRef] [PubMed]
- Group, A.S.; Cushman, W.C.; Evans, G.W.; Byington, R.P.; Goff, D.C., Jr.; Grimm, R.H., Jr.; Cutler, J.A.; Simons-Morton, D.G.; Basile, J.N.; Corson, M.A.; et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N. Engl. J. Med. 2010, 362, 1575–1585. [Google Scholar]
- Bress, A.P.; Tanner, R.M.; Hess, R.; Colantonio, L.D.; Shimbo, D.; Muntner, P. Generalizability of SPRINT Results to the U.S. Adult Population. J. Am. Coll. Cardiol. 2016, 67, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Group, S.R.; Wright, J.T., Jr.; Williamson, J.D.; Whelton, P.K.; Snyder, J.K.; Sink, K.M.; Rocco, M.V.; Reboussin, D.M.; Rahman, M.; Oparil, S.; et al. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N. Engl. J. Med. 2015, 373, 2103–2116. [Google Scholar] [CrossRef] [PubMed]
- Chertow, G.M.; Beddhu, S.; Lewis, J.B.; Toto, R.D.; Cheung, A.K. Managing Hypertension in Patients with CKD: A Marathon, Not a SPRINT. J. Am. Soc. Nephrol. 2016, 27, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.K.; Rahman, M.; Reboussin, D.M.; Craven, T.E.; Greene, T.; Kimmel, P.L.; Cushman, W.C.; Hawfield, A.T.; Johnson, K.C.; Lewis, C.E.; et al. Effects of Intensive BP Control in CKD. J. Am. Soc. Nephrol. 2017, 28, 2812–2823. [Google Scholar] [CrossRef] [PubMed]
- Magrico, R.; Bigotte Vieira, M.; Viegas Dias, C.; Leitao, L.; Neves, J.S. BP Reduction, Kidney Function Decline, and Cardiovascular Events in Patients without CKD. Clin. J. Am. Soc. Nephrol. 2018, 13, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Yamout, H.; Bakris, G.L. Consequences of Overinterpreting Serum Creatinine Increases when Achieving BP Reduction: Balancing Risks and Benefits of BP Reduction in Hypertension. Clin. J. Am. Soc. Nephrol. 2018, 13, 9–10. [Google Scholar] [CrossRef] [PubMed]
- Ku, E.; Bakris, G.; Johansen, K.L.; Lin, F.; Sarnak, M.J.; Campese, V.M.; Jamerson, K.; Gassman, J.J.; Smogorzewski, M.; Hsu, C.Y. Acute Declines in Renal Function during Intensive BP Lowering: Implications for Future ESRD Risk. J. Am. Soc. Nephrol. 2017, 28, 2794–2801. [Google Scholar] [CrossRef] [PubMed]
- Kjeldsen, S.E.; Mancia, G. Unobserved automated office blood pressure measurement in the Systolic Blood Pressure Intervention Trial (SPRINT): Systolic blood pressure treatment target remains below 140 mmHg. Eur. Heart J. Cardiovasc. Pharmacother. 2016, 2, 79–80. [Google Scholar] [CrossRef] [PubMed]
- Jafar, T.H.; Stark, P.C.; Schmid, C.H.; Landa, M.; Maschio, G.; de Jong, P.E.; de Zeeuw, D.; Shahinfar, S.; Toto, R.; Levey, A.S.; et al. Progression of chronic kidney disease: The role of blood pressure control, proteinuria, and angiotensin-converting enzyme inhibition: A patient-level meta-analysis. Ann. Intern. Med. 2003, 139, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.C.; Wu, H.Y.; Peng, Y.S.; Yang, J.Y.; Chen, H.Y.; Chiu, Y.L.; Hsu, S.P.; Ko, M.J.; Pai, M.F.; Tu, Y.K.; et al. Association of Intensive Blood Pressure Control and Kidney Disease Progression in Nondiabetic Patients With Chronic Kidney Disease: A Systematic Review and Meta-analysis. JAMA Intern. Med. 2017, 177, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Earley, A.; Haynes, S.M.; Uhlig, K. Systematic review: Blood pressure target in chronic kidney disease and proteinuria as an effect modifier. Ann. Intern. Med. 2011, 154, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, R.; Nguyen, H.A.; Benavente, O.; Mete, M.; Howard, B.V.; Mant, J.; Odden, M.C.; Peralta, C.A.; Cheung, A.K.; Nadkarni, G.N.; et al. Association Between More Intensive vs Less Intensive Blood Pressure Lowering and Risk of Mortality in Chronic Kidney Disease Stages 3 to 5: A Systematic Review and Meta-analysis. JAMA Intern. Med. 2017, 177, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Peixoto, A.J.; Santos, S.F.; Zoccali, C. Out-of-office blood pressure monitoring in chronic kidney disease. Blood Press. Monit. 2009, 14, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Peixoto, A.J.; Santos, S.F.; Zoccali, C. Pre- and postdialysis blood pressures are imprecise estimates of interdialytic ambulatory blood pressure. Clin. J. Am. Soc. Nephrol. 2006, 1, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Sarafidis, P.A.; Persu, A.; Agarwal, R.; Burnier, M.; de Leeuw, P.; Ferro, C.; Halimi, J.M.; Heine, G.; Jadoul, M.; Jarraya, F.; et al. Hypertension in dialysis patients: A consensus document by the European Renal and Cardiovascular Medicine (EURECA-m) working group of the European Renal Association—European Dialysis and Transplant Association (ERA-EDTA) and the Hypertension and the Kidney working group of the European Society of Hypertension (ESH). J. Hypertens. 2017, 35, 657–676. [Google Scholar] [PubMed]
- Sarafidis, P.A.; Persu, A.; Agarwal, R.; Burnier, M.; de Leeuw, P.; Ferro, C.J.; Halimi, J.M.; Heine, G.H.; Jadoul, M.; Jarraya, F.; et al. Hypertension in dialysis patients: A consensus document by the European Renal and Cardiovascular Medicine (EURECA-m) working group of the European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) and the Hypertension and the Kidney working group of the European Society of Hypertension (ESH). Nephrol. Dial. Transplant. 2017, 32, 620–640. [Google Scholar] [PubMed]
- Bansal, N.; McCulloch, C.E.; Rahman, M.; Kusek, J.W.; Anderson, A.H.; Xie, D.; Townsend, R.R.; Lora, C.M.; Wright, J.; Go, A.S.; et al. Blood pressure and risk of all-cause mortality in advanced chronic kidney disease and hemodialysis: The chronic renal insufficiency cohort study. Hypertension 2015, 65, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.; Ninomiya, T.; Zoungas, S.; de Zeeuw, D.; Grobbee, D.E.; Jardine, M.J.; Gallagher, M.; Roberts, M.A.; Cass, A.; Neal, B.; et al. Effect of lowering blood pressure on cardiovascular events and mortality in patients on dialysis: A systematic review and meta-analysis of randomised controlled trials. Lancet 2009, 373, 1009–1015. [Google Scholar] [CrossRef]
- Eldehni, M.T.; Odudu, A.; McIntyre, C.W. Randomized clinical trial of dialysate cooling and effects on brain white matter. J. Am. Soc. Nephrol. 2015, 26, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Levin, N.W.; Kotanko, P.; Eckardt, K.U.; Kasiske, B.L.; Chazot, C.; Cheung, A.K.; Redon, J.; Wheeler, D.C.; Zoccali, C.; London, G.M. Blood pressure in chronic kidney disease stage 5D-report from a Kidney Disease: Improving Global Outcomes controversies conference. Kidney Int. 2010, 77, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Miskulin, D.C.; Gassman, J.; Schrader, R.; Gul, A.; Jhamb, M.; Ploth, D.W.; Negrea, L.; Kwong, R.Y.; Levey, A.S.; Singh, A.K.; et al. BP in Dialysis: Results of a Pilot Study. J. Am. Soc. Nephrol. 2018, 29, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.C.; Becker, G.J. Summary of KDIGO guideline. What do we really know about management of blood pressure in patients with chronic kidney disease? Kidney Int. 2013, 83, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Maschio, G.; Alberti, D.; Janin, G.; Locatelli, F.; Mann, J.F.; Motolese, M.; Ponticelli, C.; Ritz, E.; Zucchelli, P. Effect of the angiotensin-converting-enzyme inhibitor benazepril on the progression of chronic renal insufficiency. The Angiotensin-Converting-Enzyme Inhibition in Progressive Renal Insufficiency Study Group. N. Engl. J. Med. 1996, 334, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Hou, F.F.; Zhang, X.; Zhang, G.H.; Xie, D.; Chen, P.Y.; Zhang, W.R.; Jiang, J.P.; Liang, M.; Wang, G.B.; Liu, Z.R.; et al. Efficacy and safety of benazepril for advanced chronic renal insufficiency. N. Engl. J. Med. 2006, 354, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Hou, F.F.; Xie, D.; Zhang, X.; Chen, P.Y.; Zhang, W.R.; Liang, M.; Guo, Z.J.; Jiang, J.P. Renoprotection of Optimal Antiproteinuric Doses (ROAD) Study: A randomized controlled study of benazepril and losartan in chronic renal insufficiency. J. Am. Soc. Nephrol. 2007, 18, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Ruggenenti, P.; Perna, A.; Remuzzi, G.; Gruppo Italiano di Studi Epidemiologici in Nefrologia. ACE inhibitors to prevent end-stage renal disease: When to start and why possibly never to stop: A post hoc analysis of the REIN trial results. Ramipril Efficacy in Nephropathy. J. Am. Soc. Nephrol. 2001, 12, 2832–2837. [Google Scholar] [PubMed]
- Asselbergs, F.W.; Diercks, G.F.; Hillege, H.L.; van Boven, A.J.; Janssen, W.M.; Voors, A.A.; de Zeeuw, D.; de Jong, P.E.; van Veldhuisen, D.J.; van Gilst, W.H.; et al. Effects of fosinopril and pravastatin on cardiovascular events in subjects with microalbuminuria. Circulation 2004, 110, 2809–2816. [Google Scholar] [CrossRef] [PubMed]
- Ruggenenti, P.; Fassi, A.; Ilieva, A.P.; Bruno, S.; Iliev, I.P.; Brusegan, V.; Rubis, N.; Gherardi, G.; Arnoldi, F.; Ganeva, M.; et al. Preventing microalbuminuria in type 2 diabetes. N. Engl. J. Med. 2004, 351, 1941–1951. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.T., Jr.; Bakris, G.; Greene, T.; Agodoa, L.Y.; Appel, L.J.; Charleston, J.; Cheek, D.; Douglas-Baltimore, J.G.; Gassman, J.; Glassock, R.; et al. Effect of blood pressure lowering and antihypertensive drug class on progression of hypertensive kidney disease: Results from the AASK trial. JAMA 2002, 288, 2421–2431. [Google Scholar] [CrossRef] [PubMed]
- Ruggenenti, P.; Fassi, A.; Ilieva, A.; Iliev, I.P.; Chiurchiu, C.; Rubis, N.; Gherardi, G.; Ene-Iordache, B.; Gaspari, F.; Perna, A.; et al. Effects of verapamil added-on trandolapril therapy in hypertensive type 2 diabetes patients with microalbuminuria: The BENEDICT-B randomized trial. J. Hypertens. 2011, 29, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Praga, M.; Andrade, C.F.; Luno, J.; Arias, M.; Poveda, R.; Mora, J.; Prat, M.V.; Rivera, F.; Galceran, J.M.; Ara, J.M.; et al. Antiproteinuric efficacy of losartan in comparison with amlodipine in non-diabetic proteinuric renal diseases: A double-blind, randomized clinical trial. Nephrol. Dial. Transplant. 2003, 18, 1806–1813. [Google Scholar] [CrossRef] [PubMed]
- Imai, E.; Chan, J.C.; Ito, S.; Yamasaki, T.; Kobayashi, F.; Haneda, M.; Makino, H.; ORIENT Study Investigators. Effects of olmesartan on renal and cardiovascular outcomes in type 2 diabetes with overt nephropathy: A multicentre, randomised, placebo-controlled study. Diabetologia 2011, 54, 2978–2986. [Google Scholar] [CrossRef] [PubMed]
- Haller, H.; Ito, S.; Izzo, J.L., Jr.; Januszewicz, A.; Katayama, S.; Menne, J.; Mimran, A.; Rabelink, T.J.; Ritz, E.; Ruilope, L.M.; et al. Olmesartan for the delay or prevention of microalbuminuria in type 2 diabetes. N. Engl. J. Med. 2011, 364, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Brenner, B.M.; Cooper, M.E.; de Zeeuw, D.; Keane, W.F.; Mitch, W.E.; Parving, H.H.; Remuzzi, G.; Snapinn, S.M.; Zhang, Z.; Shahinfar, S.; et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N. Engl. J. Med. 2001, 345, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Remuzzi, G.; Ruggenenti, P.; Perna, A.; Dimitrov, B.D.; de Zeeuw, D.; Hille, D.A.; Shahinfar, S.; Carides, G.W.; Brenner, B.M.; Group, R.S. Continuum of renoprotection with losartan at all stages of type 2 diabetic nephropathy: A post hoc analysis of the RENAAL trial results. J. Am. Soc. Nephrol. 2004, 15, 3117–3125. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Diener, H.C.; Sacco, R.L.; Cotton, D.; Ounpuu, S.; Lawton, W.A.; Palesch, Y.; Martin, R.H.; Albers, G.W.; Bath, P.; et al. Telmisartan to prevent recurrent stroke and cardiovascular events. N. Engl. J. Med. 2008, 359, 1225–1237. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.; Kropelin, T.F.; Hoekman, J.; de Zeeuw, D. Drug-Induced Reduction in Albuminuria Is Associated with Subsequent Renoprotection: A Meta-Analysis. J. Am. Soc. Nephrol. 2015, 26, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Mavridis, D.; Navarese, E.; Craig, J.C.; Tonelli, M.; Salanti, G.; Wiebe, N.; Ruospo, M.; Wheeler, D.C.; Strippoli, G.F. Comparative efficacy and safety of blood pressure-lowering agents in adults with diabetes and kidney disease: A network meta-analysis. Lancet 2015, 385, 2047–2056. [Google Scholar] [CrossRef]
- Xie, X.; Liu, Y.; Perkovic, V.; Li, X.; Ninomiya, T.; Hou, W.; Zhao, N.; Liu, L.; Lv, J.; Zhang, H.; et al. Renin-Angiotensin System Inhibitors and Kidney and Cardiovascular Outcomes in Patients With CKD: A Bayesian Network Meta-analysis of Randomized Clinical Trials. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 67, 728–741. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Blackburn, R.C.; Parke, C.L.; McCullough, K.; Marks, A.; Black, C. Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers for adults with early (stage 1 to 3) non-diabetic chronic kidney disease. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef] [PubMed]
- Investigators, O.; Yusuf, S.; Teo, K.K.; Pogue, J.; Dyal, L.; Copland, I.; Schumacher, H.; Dagenais, G.; Sleight, P.; Anderson, C. Telmisartan, ramipril, or both in patients at high risk for vascular events. N. Engl. J. Med. 2008, 358, 1547–1559. [Google Scholar]
- Li, S.M.; He, W.B.; Chen, J.; Cai, Q.Q.; Huang, F.F.; Zhang, K.; Wang, J.F.; Liu, X.; Huang, H. Combined blockade of renin-angiotensin-aldosterone system reduced all-cause but not cardiovascular mortality in dialysis patients: A mediation analysis and systematic review. Atherosclerosis 2018, 269, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.K.; Kamath, N.S.; El Kossi, M.; El Nahas, A.M. The impact of stopping inhibitors of the renin-angiotensin system in patients with advanced chronic kidney disease. Nephrol. Dial. Transplant. 2010, 25, 3977–3982. [Google Scholar] [CrossRef] [PubMed]
- Hogan, T.J.; Elliott, W.J.; Seto, A.H.; Bakris, G.L. Antihypertensive treatment with and without benazepril in patients with chronic renal insufficiency: A US economic evaluation. Pharmacoeconomics 2002, 20, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Adarkwah, C.C.; Gandjour, A.; Akkerman, M.; Evers, S. To treat or not to treat? Cost-effectiveness of ace inhibitors in non-diabetic advanced renal disease—A Dutch perspective. Kidney Blood Press. Res. 2013, 37, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Atthobari, J.; Asselbergs, F.W.; Boersma, C.; de Vries, R.; Hillege, H.L.; van Gilst, W.H.; Gansevoort, R.T.; de Jong, P.E.; de Jong-van den Berg, L.T.; Postma, M.J.; et al. Cost-effectiveness of screening for albuminuria with subsequent fosinopril treatment to prevent cardiovascular events: A pharmacoeconomic analysis linked to the prevention of renal and vascular endstage disease (PREVEND) study and the prevention of renal and vascular endstage disease intervention trial (PREVEND IT). Clin. Ther. 2006, 28, 432–444. [Google Scholar] [PubMed]
- Palmer, A.J.; Annemans, L.; Roze, S.; Lamotte, M.; Lapuerta, P.; Chen, R.; Gabriel, S.; Carita, P.; Rodby, R.A.; de Zeeuw, D.; et al. Cost-effectiveness of early irbesartan treatment versus control (standard antihypertensive medications excluding ACE inhibitors, other angiotensin-2 receptor antagonists, and dihydropyridine calcium channel blockers) or late irbesartan treatment in patients with type 2 diabetes, hypertension, and renal disease. Diabetes Care 2004, 27, 1897–1903. [Google Scholar] [PubMed]
- Ruggenenti, P.; Cravedi, P.; Remuzzi, G. Mechanisms and treatment of CKD. J. Am. Soc. Nephrol. 2012, 23, 1917–1928. [Google Scholar] [CrossRef] [PubMed]
- Pitt, B.; Filippatos, G.; Gheorghiade, M.; Kober, L.; Krum, H.; Ponikowski, P.; Nowack, C.; Kolkhof, P.; Kim, S.Y.; Zannad, F. Rationale and design of ARTS: A randomized, double-blind study of BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease. Eur. J. Heart Fail. 2012, 14, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; MacDonald, T.M.; Morant, S.; Webb, D.J.; Sever, P.; McInnes, G.; Ford, I.; Cruickshank, J.K.; Caulfield, M.J.; Salsbury, J.; et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): A randomised, double-blind, crossover trial. Lancet 2015, 386, 2059–2068. [Google Scholar] [CrossRef]
- Rossignol, P.; Massy, Z.A.; Azizi, M.; Bakris, G.; Ritz, E.; Covic, A.; Goldsmith, D.; Heine, G.H.; Jager, K.J.; Kanbay, M.; et al. The double challenge of resistant hypertension and chronic kidney disease. Lancet 2015, 386, 1588–1598. [Google Scholar] [CrossRef]
- Edwards, N.C.; Steeds, R.P.; Chue, C.D.; Stewart, P.M.; Ferro, C.J.; Townend, J.N. The safety and tolerability of spironolactone in patients with mild to moderate chronic kidney disease. Br. J. Clin. Pharmacol. 2012, 73, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, S.; Bigazzi, R.; Campese, V.M. Long-term effects of spironolactone on proteinuria and kidney function in patients with chronic kidney disease. Kidney Int. 2006, 70, 2116–2123. [Google Scholar] [CrossRef] [PubMed]
- Bakris, G.L.; Agarwal, R.; Chan, J.C.; Cooper, M.E.; Gansevoort, R.T.; Haller, H.; Remuzzi, G.; Rossing, P.; Schmieder, R.E.; Nowack, C.; et al. Effect of Finerenone on Albuminuria in Patients With Diabetic Nephropathy: A Randomized Clinical Trial. JAMA 2015, 314, 884–894. [Google Scholar] [CrossRef] [PubMed]
- Katayama, S.; Yamada, D.; Nakayama, M.; Yamada, T.; Myoishi, M.; Kato, M.; Nowack, C.; Kolkhof, P.; Yamasaki, Y. A randomized controlled study of finerenone versus placebo in Japanese patients with type 2 diabetes mellitus and diabetic nephropathy. J. Diabetes Its Complic. 2017, 31, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Currie, G.; Taylor, A.H.; Fujita, T.; Ohtsu, H.; Lindhardt, M.; Rossing, P.; Boesby, L.; Edwards, N.C.; Ferro, C.J.; Townend, J.N.; et al. Effect of mineralocorticoid receptor antagonists on proteinuria and progression of chronic kidney disease: A systematic review and meta-analysis. BMC Nephrol. 2016, 17, 127. [Google Scholar] [CrossRef] [PubMed]
- Bolignano, D.; Palmer, S.C.; Navaneethan, S.D.; Strippoli, G.F. Aldosterone antagonists for preventing the progression of chronic kidney disease. Cochrane Database Syst. Rev. 2014, 4, CD007004. [Google Scholar] [CrossRef] [PubMed]
- Navaneethan, S.D.; Nigwekar, S.U.; Sehgal, A.R.; Strippoli, G.F. Aldosterone antagonists for preventing the progression of chronic kidney disease: A systematic review and meta-analysis. Clin. J. Am. Soc. Nephrol. 2009, 4, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Chua, D.; Lo, A.; Lo, C. Spironolactone use in heart failure patients with end-stage renal disease on hemodialysis: Is it safe? Clin. Cardiol. 2010, 33, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Mori, Y.; Kageyama, S.; Arihara, K.; Sugiyama, T.; Ohmura, H.; Yakushigawa, T.; Sugiyama, H.; Shimada, Y.; Nojima, Y.; et al. Spironolactone reduces cardiovascular and cerebrovascular morbidity and mortality in hemodialysis patients. J. Am. Coll. Cardiol. 2014, 63, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Ponda, M.P.; Hostetter, T.H. Aldosterone antagonism in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2006, 1, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Einhorn, L.M.; Zhan, M.; Hsu, V.D.; Walker, L.D.; Moen, M.F.; Seliger, S.L.; Weir, M.R.; Fink, J.C. The frequency of hyperkalemia and its significance in chronic kidney disease. Arch. Intern. Med. 2009, 169, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.R.; Bakris, G.L.; Bushinsky, D.A.; Mayo, M.R.; Garza, D.; Stasiv, Y.; Wittes, J.; Christ-Schmidt, H.; Berman, L.; Pitt, B.; et al. Patiromer in patients with kidney disease and hyperkalemia receiving RAAS inhibitors. N. Engl. J. Med. 2015, 372, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.R.; Bakris, G.L.; Gross, C.; Mayo, M.R.; Garza, D.; Stasiv, Y.; Yuan, J.; Berman, L.; Williams, G.H. Treatment with patiromer decreases aldosterone in patients with chronic kidney disease and hyperkalemia on renin-angiotensin system inhibitors. Kidney Int. 2016, 90, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Packham, D.K.; Rasmussen, H.S.; Lavin, P.T.; El-Shahawy, M.A.; Roger, S.D.; Block, G.; Qunibi, W.; Pergola, P.; Singh, B. Sodium zirconium cyclosilicate in hyperkalemia. N. Engl. J. Med. 2015, 372, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Emmett, M.; Mehta, A. Gastrointestinal potassium binding-more than just lowering serum [K(+)]: Patiromer, potassium balance, and the renin angiotensin aldosterone axis. Kidney Int. 2016, 90, 484–486. [Google Scholar] [CrossRef] [PubMed]
- Lepage, L.; Dufour, A.C.; Doiron, J.; Handfield, K.; Desforges, K.; Bell, R.; Vallee, M.; Savoie, M.; Perreault, S.; Laurin, L.P.; et al. Randomized Clinical Trial of Sodium Polystyrene Sulfonate for the Treatment of Mild Hyperkalemia in CKD. Clin. J. Am. Soc. Nephrol. 2015, 10, 2136–2142. [Google Scholar] [CrossRef] [PubMed]
- Sterns, R.H.; Grieff, M.; Bernstein, P.L. Treatment of hyperkalemia: Something old, something new. Kidney Int. 2016, 89, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.A.; Baker, T.P.; Nguyen, A.; Sebastianelli, M.E.; Stewart, H.L.; Oliver, D.K.; Abbott, K.C.; Yuan, C.M. Association of prescription of oral sodium polystyrene sulfonate with sorbitol in an inpatient setting with colonic necrosis: A retrospective cohort study. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 60, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.; Ligtenberg, G.; Klein, I.I.; Koomans, H.A.; Blankestijn, P.J. Sympathetic hyperactivity in chronic kidney disease: Pathogenesis, clinical relevance, and treatment. Kidney Int. 2004, 65, 1568–1576. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Mallamaci, F.; Parlongo, S.; Cutrupi, S.; Benedetto, F.A.; Tripepi, G.; Bonanno, G.; Rapisarda, F.; Fatuzzo, P.; Seminara, G.; et al. Plasma norepinephrine predicts survival and incident cardiovascular events in patients with end-stage renal disease. Circulation 2002, 105, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Bakris, G.L.; Hart, P.; Ritz, E. Beta blockers in the management of chronic kidney disease. Kidney Int. 2006, 70, 1905–1913. [Google Scholar] [CrossRef] [PubMed]
- Bleyer, A.J.; Hartman, J.; Brannon, P.C.; Reeves-Daniel, A.; Satko, S.G.; Russell, G. Characteristics of sudden death in hemodialysis patients. Kidney Int. 2006, 69, 2268–2273. [Google Scholar] [CrossRef] [PubMed]
- Hannedouche, T.; Landais, P.; Goldfarb, B.; el Esper, N.; Fournier, A.; Godin, M.; Durand, D.; Chanard, J.; Mignon, F.; Suo, J.M.; et al. Randomised controlled trial of enalapril and beta blockers in non-diabetic chronic renal failure. BMJ 1994, 309, 833–837. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Lacy, P.S.; Thom, S.M.; Cruickshank, K.; Stanton, A.; Collier, D.; Hughes, A.D.; Thurston, H.; O’Rourke, M.; Investigators, C.; et al. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: Principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation 2006, 113, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- Apperloo, A.J.; de Zeeuw, D.; Sluiter, H.E.; de Jong, P.E. Differential effects of enalapril and atenolol on proteinuria and renal haemodynamics in non-diabetic renal disease. BMJ 1991, 303, 821–824. [Google Scholar] [CrossRef] [PubMed]
- Neutel, J.M.; Schnaper, H.; Cheung, D.G.; Graettinger, W.F.; Weber, M.A. Antihypertensive effects of beta-blockers administered once daily: 24-hour measurements. Am. Heart J. 1990, 120, 166–171. [Google Scholar] [CrossRef]
- Shireman, T.I.; Mahnken, J.D.; Phadnis, M.A.; Ellerbeck, E.F. Effectiveness comparison of cardio-selective to non-selective beta-blockers and their association with mortality and morbidity in end-stage renal disease: A retrospective cohort study. BMC Cardiovasc. Disord. 2016, 16, 60. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.A.; Dixon, S.N.; Fleet, J.L.; Roberts, M.A.; Hackam, D.G.; Oliver, M.J.; Suri, R.S.; Quinn, R.R.; Ozair, S.; Beyea, M.M.; et al. beta-Blocker dialyzability and mortality in older patients receiving hemodialysis. J. Am. Soc. Nephrol. 2015, 26, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Kovesdy, C.P. Management of Hyperkalemia: An Update for the Internist. Am. J. Med. 2015, 128, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Bakris, G.L.; Fonseca, V.; Katholi, R.E.; McGill, J.B.; Messerli, F.H.; Phillips, R.A.; Raskin, P.; Wright, J.T., Jr.; Oakes, R.; Lukas, M.A.; et al. Metabolic effects of carvedilol vs metoprolol in patients with type 2 diabetes mellitus and hypertension: A randomized controlled trial. JAMA 2004, 292, 2227–2236. [Google Scholar] [CrossRef] [PubMed]
- Cice, G.; Ferrara, L.; D’Andrea, A.; D’Isa, S.; Di Benedetto, A.; Cittadini, A.; Russo, P.E.; Golino, P.; Calabro, R. Carvedilol increases two-year survivalin dialysis patients with dilated cardiomyopathy: A prospective, placebo-controlled trial. J. Am. Coll. Cardiol. 2003, 41, 1438–1444. [Google Scholar] [CrossRef]
- Ghali, J.K.; Wikstrand, J.; Van Veldhuisen, D.J.; Fagerberg, B.; Goldstein, S.; Hjalmarson, A.; Johansson, P.; Kjekshus, J.; Ohlsson, L.; Samuelsson, O.; et al. The influence of renal function on clinical outcome and response to beta-blockade in systolic heart failure: Insights from Metoprolol CR/XL Randomized Intervention Trial in Chronic HF (MERIT-HF). J. Card. Fail. 2009, 15, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Castagno, D.; Jhund, P.S.; McMurray, J.J.; Lewsey, J.D.; Erdmann, E.; Zannad, F.; Remme, W.J.; Lopez-Sendon, J.L.; Lechat, P.; Follath, F.; et al. Improved survival with bisoprolol in patients with heart failure and renal impairment: An analysis of the cardiac insufficiency bisoprolol study II (CIBIS-II) trial. Eur. J. Heart Fail. 2010, 12, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Wali, R.K.; Iyengar, M.; Beck, G.J.; Chartyan, D.M.; Chonchol, M.; Lukas, M.A.; Cooper, C.; Himmelfarb, J.; Weir, M.R.; Berl, T.; et al. Efficacy and safety of carvedilol in treatment of heart failure with chronic kidney disease: A meta-analysis of randomized trials. Circ. Heart Fail. 2011, 4, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Badve, S.V.; Roberts, M.A.; Hawley, C.M.; Cass, A.; Garg, A.X.; Krum, H.; Tonkin, A.; Perkovic, V. Effects of beta-adrenergic antagonists in patients with chronic kidney disease: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2011, 58, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Diabetes Control and Complications Trial Research Group; Nathan, D.M.; Genuth, S.; Lachin, J.; Cleary, P.; Crofford, O.; Davis, M.; Rand, L.; Siebert, C. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 1993, 329, 977–986. [Google Scholar]
- Writing Team for the Diabetes; Complications Trial/Epidemiology of Diabetes Interventions; Complications Research Group. Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: The Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 2003, 290, 2159–2167. [Google Scholar]
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998, 352, 837–853. [Google Scholar]
- Group, A.C.; Patel, A.; MacMahon, S.; Chalmers, J.; Neal, B.; Billot, L.; Woodward, M.; Marre, M.; Cooper, M.; Glasziou, P.; et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 2008, 358, 2560–2572. [Google Scholar]
- Perkovic, V.; Heerspink, H.L.; Chalmers, J.; Woodward, M.; Jun, M.; Li, Q.; MacMahon, S.; Cooper, M.E.; Hamet, P.; Marre, M.; et al. Intensive glucose control improves kidney outcomes in patients with type 2 diabetes. Kidney Int. 2013, 83, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.G.; Perkovic, V.; Chalmers, J.; Woodward, M.; Li, Q.; Cooper, M.E.; Hamet, P.; Harrap, S.; Heller, S.; MacMahon, S.; et al. Long-term Benefits of Intensive Glucose Control for Preventing End-Stage Kidney Disease: ADVANCE-ON. Diabetes Care 2016, 39, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Action to Control Cardiovascular Risk in Diabetes Study Group; Gerstein, H.C.; Miller, M.E.; Byington, R.P.; Goff, D.C., Jr.; Bigger, J.T.; Buse, J.B.; Cushman, W.C.; Genuth, S.; Ismail-Beigi, F.; et al. Effects of intensive glucose lowering in type 2 diabetes. N. Engl. J. Med. 2008, 358, 2545–2559. [Google Scholar] [PubMed]
- Papademetriou, V.; Lovato, L.; Doumas, M.; Nylen, E.; Mottl, A.; Cohen, R.M.; Applegate, W.B.; Puntakee, Z.; Yale, J.F.; Cushman, W.C.; et al. Chronic kidney disease and intensive glycemic control increase cardiovascular risk in patients with type 2 diabetes. Kidney Int. 2015, 87, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Moen, M.F.; Zhan, M.; Hsu, V.D.; Walker, L.D.; Einhorn, L.M.; Seliger, S.L.; Fink, J.C. Frequency of hypoglycemia and its significance in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2009, 4, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Guideline Development Group. Clinical Practice Guideline on management of patients with diabetes and chronic kidney disease stage 3b or higher (eGFR <45 mL/min). Nephrol. Dial. Transplant. 2015, 30 (Suppl. 2), ii1–ii142. [Google Scholar]
- Neumiller, J.J.; Alicic, R.Z.; Tuttle, K.R. Therapeutic Considerations for Antihyperglycemic Agents in Diabetic Kidney Disease. J. Am. Soc. Nephrol. 2017, 28, 2263–2274. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Ghani, M.; Del Prato, S.; Chilton, R.; DeFronzo, R.A. SGLT2 Inhibitors and Cardiovascular Risk: Lessons Learned From the EMPA-REG OUTCOME Study. Diabetes Care 2016, 39, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Fioretto, P.; Zambon, A.; Rossato, M.; Busetto, L.; Vettor, R. SGLT2 Inhibitors and the Diabetic Kidney. Diabetes Care 2016, 39 (Suppl. 2), S165–S171. [Google Scholar] [CrossRef] [PubMed]
- Cherney, D.Z.; Perkins, B.A.; Soleymanlou, N.; Maione, M.; Lai, V.; Lee, A.; Fagan, N.M.; Woerle, H.J.; Johansen, O.E.; Broedl, U.C.; et al. Renal hemodynamic effect of sodium-glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation 2014, 129, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wanner, C.; Inzucchi, S.E.; Lachin, J.M.; Fitchett, D.; von Eynatten, M.; Mattheus, M.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Zinman, B.; et al. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R.; et al. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.; Desai, M.; Jardine, M.; Balis, D.; Meininger, G.; Perkovic, V. Canagliflozin Slows Progression of Renal Function Decline Independently of Glycemic Effects. J. Am. Soc. Nephrol. 2017, 28, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.; Johnsson, E.; Gause-Nilsson, I.; Cain, V.A.; Sjostrom, C.D. Dapagliflozin reduces albuminuria in patients with diabetes and hypertension receiving renin-angiotensin blockers. Diabetes Obes. Metab. 2016, 18, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Cherney, D.Z.I.; Zinman, B.; Inzucchi, S.E.; Koitka-Weber, A.; Mattheus, M.; von Eynatten, M.; Wanner, C. Effects of empagliflozin on the urinary albumin-to-creatinine ratio in patients with type 2 diabetes and established cardiovascular disease: An exploratory analysis from the EMPA-REG OUTCOME randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017, 5, 610–621. [Google Scholar] [CrossRef]
- Pecoits-Filho, R.; Perkovic, V. Are SGLT2 Inhibitors Ready for Prime Time for CKD? Clin. J. Am. Soc. Nephrol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Foote, C.; Blomster, J.; Toyama, T.; Perkovic, V.; Sundstrom, J.; Neal, B. Effects of sodium-glucose cotransporter-2 inhibitors on cardiovascular events, death, and major safety outcomes in adults with type 2 diabetes: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2016, 4, 411–419. [Google Scholar] [CrossRef]
- Wanner, C.; Lachin, J.M.; Inzucchi, S.E.; Fitchett, D.; Mattheus, M.; George, J.; Woerle, H.J.; Broedl, U.C.; von Eynatten, M.; Zinman, B.; et al. Empagliflozin and Clinical Outcomes in Patients With Type 2 Diabetes Mellitus, Established Cardiovascular Disease, and Chronic Kidney Disease. Circulation 2018, 137, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Ferrannini, E. Euglycemic Diabetic Ketoacidosis: A Predictable, Detectable, and Preventable Safety Concern With SGLT2 Inhibitors. Diabetes Care 2015, 38, 1638–1642. [Google Scholar] [CrossRef] [PubMed]
- Highlights of Prescribing Information docs. Boehringer-Ingelheim.com. Available online: http://docs.boehringer-ingelheim.com/Prescribing%20Information/PIs/Jardiance/jardiance.pdf (accessed on 20 May 2018).
- Watts, N.B.; Bilezikian, J.P.; Usiskin, K.; Edwards, R.; Desai, M.; Law, G.; Meininger, G. Effects of Canagliflozin on Fracture Risk in Patients With Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2016, 101, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P.; Watts, N.B.; Usiskin, K.; Polidori, D.; Fung, A.; Sullivan, D.; Rosenthal, N. Evaluation of Bone Mineral Density and Bone Biomarkers in Patients With Type 2 Diabetes Treated With Canagliflozin. J. Clin. Endocrinol. Metab. 2016, 101, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Mosenzon, O.; Leibowitz, G.; Bhatt, D.L.; Cahn, A.; Hirshberg, B.; Wei, C.; Im, K.; Rozenberg, A.; Yanuv, I.; Stahre, C.; et al. Effect of Saxagliptin on Renal Outcomes in the SAVOR-TIMI 53 Trial. Diabetes Care 2017, 40, 69–76. [Google Scholar] [CrossRef] [PubMed]
- MacIsaac, R.J.; Thomas, M.C. Effects of Diabetes Medications Targeting the Incretin System on the Kidney. Clin. J. Am. Soc. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.R.; Bethel, M.A.; Mentz, R.J.; Thompson, V.P.; Lokhnygina, Y.; Buse, J.B.; Chan, J.C.; Choi, J.; Gustavson, S.M.; Iqbal, N.; et al. Effects of Once-Weekly Exenatide on Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 1228–1239. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.A.; Claggett, B.; Diaz, R.; Dickstein, K.; Gerstein, H.C.; Kober, L.V.; Lawson, F.C.; Ping, L.; Wei, X.; Lewis, E.F.; et al. Lixisenatide in Patients with Type 2 Diabetes and Acute Coronary Syndrome. N. Engl. J. Med. 2015, 373, 2247–2257. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jodar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L.; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Arnouts, P.; Bolignano, D.; Nistor, I.; Bilo, H.; Gnudi, L.; Heaf, J.; van Biesen, W. Glucose-lowering drugs in patients with chronic kidney disease: A narrative review on pharmacokinetic properties. Nephrol. Dial. Transplant. 2014, 29, 1284–1300. [Google Scholar] [CrossRef] [PubMed]
- Inzucchi, S.E.; Lipska, K.J.; Mayo, H.; Bailey, C.J.; McGuire, D.K. Metformin in patients with type 2 diabetes and kidney disease: A systematic review. JAMA 2014, 312, 2668–2675. [Google Scholar] [CrossRef] [PubMed]
- Hung, A.M.; Roumie, C.L.; Greevy, R.A.; Liu, X.; Grijalva, C.G.; Murff, H.J.; Griffin, M.R. Kidney function decline in metformin versus sulfonylurea initiators: Assessment of time-dependent contribution of weight, blood pressure, and glycemic control. Pharmacoepidemiol. Drug Saf. 2013, 22, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Roussel, R.; Travert, F.; Pasquet, B.; Wilson, P.W.; Smith, S.C., Jr.; Goto, S.; Ravaud, P.; Marre, M.; Porath, A.; Bhatt, D.L.; et al. Metformin use and mortality among patients with diabetes and atherothrombosis. Arch. Intern. Med. 2010, 170, 1892–1899. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J.; Paquot, N. Metformin revisited: A critical review of the benefit-risk balance in at-risk patients with type 2 diabetes. Diabetes Metab. 2013, 39, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Crowley, M.J.; Diamantidis, C.J.; McDuffie, J.R.; Cameron, C.B.; Stanifer, J.W.; Mock, C.K.; Wang, X.; Tang, S.; Nagi, A.; Kosinski, A.S.; et al. Clinical Outcomes of Metformin Use in Populations With Chronic Kidney Disease, Congestive Heart Failure, or Chronic Liver Disease: A Systematic Review. Ann. Intern. Med. 2017, 166, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Ma, X.; Feng, W.; Fu, Y.; Lu, Z.; Xu, M.; Shen, Q.; Zhu, Y.; Zhang, Y. Metformin attenuates cardiac fibrosis by inhibiting the TGFbeta1-Smad3 signalling pathway. Cardiovasc. Res. 2010, 87, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Takasaka, N.; Yoshida, M.; Tsubouchi, K.; Minagawa, S.; Araya, J.; Saito, N.; Fujita, Y.; Kurita, Y.; Kobayashi, K.; et al. Metformin attenuates lung fibrosis development via NOX4 suppression. Respir. Res. 2016, 17, 107. [Google Scholar] [CrossRef] [PubMed]
- Rowe, I.; Chiaravalli, M.; Mannella, V.; Ulisse, V.; Quilici, G.; Pema, M.; Song, X.W.; Xu, H.; Mari, S.; Qian, F.; et al. Defective glucose metabolism in polycystic kidney disease identifies a new therapeutic strategy. Nat. Med. 2013, 19, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Takiar, V.; Nishio, S.; Seo-Mayer, P.; King, J.D., Jr.; Li, H.; Zhang, L.; Karihaloo, A.; Hallows, K.R.; Somlo, S.; Caplan, M.J. Activating AMP-activated protein kinase (AMPK) slows renal cystogenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 2462–2467. [Google Scholar] [CrossRef] [PubMed]
- Vervloet, M.G.; Sezer, S.; Massy, Z.A.; Johansson, L.; Cozzolino, M.; Fouque, D. The role of phosphate in kidney disease. Nat. Rev. Nephrol. 2017, 13, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Kestenbaum, B. Con: Phosphate binders in chronic kidney disease. Nephrol. Dial. Transplant. 2016, 31, 189–194. [Google Scholar] [PubMed]
- Liabeuf, S.; Neirynck, N.; Drueke, T.B.; Vanholder, R.; Massy, Z.A. Clinical studies and chronic kidney disease: What did we learn recently? Semin. Nephrol. 2014, 34, 164–179. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Mallamaci, F. Moderator’s view: Phosphate binders in chronic kidney disease patients: A clear ‘No’ at the moment, but stay tuned. Nephrol. Dial. Transplant. 2016, 31, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Tentori, F.; Blayney, M.J.; Albert, J.M.; Gillespie, B.W.; Kerr, P.G.; Bommer, J.; Young, E.W.; Akizawa, T.; Akiba, T.; Pisoni, R.L.; et al. Mortality risk for dialysis patients with different levels of serum calcium, phosphorus, and PTH: The Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2008, 52, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Hayen, A.; Macaskill, P.; Pellegrini, F.; Craig, J.C.; Elder, G.J.; Strippoli, G.F. Serum levels of phosphorus, parathyroid hormone, and calcium and risks of death and cardiovascular disease in individuals with chronic kidney disease: A systematic review and meta-analysis. JAMA 2011, 305, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Kovesdy, C.P.; Kuchmak, O.; Lu, J.L.; Kalantar-Zadeh, K. Outcomes associated with phosphorus binders in men with non-dialysis-dependent CKD. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2010, 56, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Isakova, T.; Gutierrez, O.M.; Chang, Y.; Shah, A.; Tamez, H.; Smith, K.; Thadhani, R.; Wolf, M. Phosphorus binders and survival on hemodialysis. J. Am. Soc. Nephrol. 2009, 20, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Block, G.A.; Wheeler, D.C.; Persky, M.S.; Kestenbaum, B.; Ketteler, M.; Spiegel, D.M.; Allison, M.A.; Asplin, J.; Smits, G.; Hoofnagle, A.N.; et al. Effects of phosphate binders in moderate CKD. J. Am. Soc. Nephrol. 2012, 23, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Winkelmayer, W.C.; Liu, J.; Kestenbaum, B. Comparative effectiveness of calcium-containing phosphate binders in incident U.S. dialysis patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Wald, R.; Rabbat, C.G.; Girard, L.; Garg, A.X.; Tennankore, K.; Tyrwhitt, J.; Smyth, A.; Rathe-Skafel, A.; Gao, P.; Mazzetti, A.; et al. Two phosphAte taRGets in End-stage renal disease Trial (TARGET): A Randomized Controlled Trial. Clin. J. Am. Soc. Nephrol. 2017, 12, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Teixeira-Pinto, A.; Saglimbene, V.; Craig, J.C.; Macaskill, P.; Tonelli, M.; de Berardis, G.; Ruospo, M.; Strippoli, G.F. Association of Drug Effects on Serum Parathyroid Hormone, Phosphorus, and Calcium Levels With Mortality in CKD: A Meta-analysis. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2015, 66, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Gardner, S.; Tonelli, M.; Mavridis, D.; Johnson, D.W.; Craig, J.C.; French, R.; Ruospo, M.; Strippoli, G.F. Phosphate-Binding Agents in Adults With CKD: A Network Meta-analysis of Randomized Trials. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 68, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, M.I.; Sonmez, A.; Saglam, M.; Yaman, H.; Kilic, S.; Eyileten, T.; Caglar, K.; Oguz, Y.; Vural, A.; Yenicesu, M.; et al. Comparison of calcium acetate and sevelamer on vascular function and fibroblast growth factor 23 in CKD patients: A randomized clinical trial. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 59, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Kuczera, P.; Adamczak, M.; Wiecek, A. Fibroblast Growth Factor-23-A Potential Uremic Toxin. Toxins 2016, 8, 369. [Google Scholar] [CrossRef] [PubMed]
- Evenepoel, P.; Rodriguez, M.; Ketteler, M. Laboratory abnormalities in CKD-MBD: Markers, predictors, or mediators of disease? Semin. Nephrol. 2014, 34, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.B.; Cancela, A.L.; Graciolli, F.G.; Dos Reis, L.M.; Draibe, S.A.; Cuppari, L.; Carvalho, A.B.; Jorgetti, V.; Canziani, M.E.; Moyses, R.M. Early control of PTH and FGF23 in normophosphatemic CKD patients: A new target in CKD-MBD therapy? Clin. J. Am. Soc. Nephrol. 2010, 5, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Liabeuf, S.; Ryckelynck, J.P.; El Esper, N.; Urena, P.; Combe, C.; Dussol, B.; Fouque, D.; Vanhille, P.; Frimat, L.; Thervet, E.; et al. Randomized Clinical Trial of Sevelamer Carbonate on Serum Klotho and Fibroblast Growth Factor 23 in CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 1930–1940. [Google Scholar] [CrossRef] [PubMed]
- Chue, C.D.; Townend, J.N.; Moody, W.E.; Zehnder, D.; Wall, N.A.; Harper, L.; Edwards, N.C.; Steeds, R.P.; Ferro, C.J. Cardiovascular effects of sevelamer in stage 3 CKD. J. Am. Soc. Nephrol. 2013, 24, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Isakova, T.; Gutierrez, O.M.; Smith, K.; Epstein, M.; Keating, L.K.; Juppner, H.; Wolf, M. Pilot study of dietary phosphorus restriction and phosphorus binders to target fibroblast growth factor 23 in patients with chronic kidney disease. Nephrol. Dial. Transplant. 2011, 26, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Parra, E.; Gonzalez-Casaus, M.L.; Galan, A.; Martinez-Calero, A.; Navas, V.; Rodriguez, M.; Ortiz, A. Lanthanum carbonate reduces FGF23 in chronic kidney disease Stage 3 patients. Nephrol. Dial. Transplant. 2011, 26, 2567–2571. [Google Scholar] [CrossRef] [PubMed]
- Suki, W.N.; Zabaneh, R.; Cangiano, J.L.; Reed, J.; Fischer, D.; Garrett, L.; Ling, B.N.; Chasan-Taber, S.; Dillon, M.A.; Blair, A.T.; et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 2007, 72, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Block, G.A.; Raggi, P.; Bellasi, A.; Kooienga, L.; Spiegel, D.M. Mortality effect of coronary calcification and phosphate binder choice in incident hemodialysis patients. Kidney Int. 2007, 71, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.; Molony, D.; Bell, C.; Cucciniello, E.; Bellizzi, V.; Russo, D.; Bellasi, A.; Investigators, I.S. Sevelamer versus calcium carbonate in incident hemodialysis patients: Results of an open-label 24-month randomized clinical trial. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2013, 62, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.; Bellasi, A.; Russo, D.; Investigators, I.S. Mortality in kidney disease patients treated with phosphate binders: A randomized study. Clin. J. Am. Soc. Nephrol. 2012, 7, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, X.; Zhou, Y.; Li, S.; Chen, Y.; Wang, Y.; Lou, T. New Conclusions Regarding Comparison of Sevelamer and Calcium-Based Phosphate Binders in Coronary-Artery Calcification for Dialysis Patients: A Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2015, 10, e0133938. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Bolignano, D.; D’Arrigo, G.; Dekker, F.W.; Fliser, D.; Heine, G.H.; Jager, K.J.; Kanbay, M.; Mallamaci, F.; Massy, Z.; et al. Validity of Vascular Calcification as a Screening Tool and as a Surrogate End Point in Clinical Research. Hypertension 2015, 66, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Navaneethan, S.D.; Palmer, S.C.; Craig, J.C.; Elder, G.J.; Strippoli, G.F. Benefits and harms of phosphate binders in CKD: A systematic review of randomized controlled trials. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2009, 54, 619–637. [Google Scholar] [CrossRef] [PubMed]
- Habbous, S.; Przech, S.; Acedillo, R.; Sarma, S.; Garg, A.X.; Martin, J. The efficacy and safety of sevelamer and lanthanum versus calcium-containing and iron-based binders in treating hyperphosphatemia in patients with chronic kidney disease: A systematic review and meta-analysis. Nephrol. Dial. Transplant. 2017, 32, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Sekercioglu, N.; Thabane, L.; Diaz Martinez, J.P.; Nesrallah, G.; Longo, C.J.; Busse, J.W.; Akhtar-Danesh, N.; Agarwal, A.; Al-Khalifah, R.; Iorio, A.; et al. Comparative Effectiveness of Phosphate Binders in Patients with Chronic Kidney Disease: A Systematic Review and Network Meta-Analysis. PLoS ONE 2016, 11, e0156891. [Google Scholar] [CrossRef] [PubMed]
- Jamal, S.A.; Vandermeer, B.; Raggi, P.; Mendelssohn, D.C.; Chatterley, T.; Dorgan, M.; Lok, C.E.; Fitchett, D.; Tsuyuki, R.T. Effect of calcium-based versus non-calcium-based phosphate binders on mortality in patients with chronic kidney disease: An updated systematic review and meta-analysis. Lancet 2013, 382, 1268–1277. [Google Scholar] [CrossRef]
- Ortiz, A.; Sanchez-Nino, M.D. The demise of calcium-based phosphate binders. Lancet 2013, 382, 1232–1234. [Google Scholar] [CrossRef]
- Manns, B.; Klarenbach, S.; Lee, H.; Culleton, B.; Shrive, F.; Tonelli, M. Economic evaluation of sevelamer in patients with end-stage renal disease. Nephrol. Dial. Transplant. 2007, 22, 2867–2878. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, M.; Cipriani, F.; Bellasi, A.; Russo, D.; Di Iorio, B. Sevelamer is cost-saving vs. calcium carbonate in non-dialysis-dependent CKD patients in italy: A patient-level cost-effectiveness analysis of the INDEPENDENT study. Blood Purif. 2014, 37, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.V.; Bose, S.; Finkelstein, E. Incremental cost-utility of sevelamer relative to calcium carbonate for treatment of hyperphosphatemia among pre-dialysis chronic kidney disease patients. BMC Nephrol. 2016, 17, 45. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.; Bartko-Winters, S.; Bernard, L.; Fenton, A.; Hutchison, C.; Di Iorio, B. Economic evaluation of sevelamer for the treatment of hyperphosphatemia in chronic kidney disease patients not on dialysis in the United Kingdom. J. Med. Econ. 2013, 16, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Rascati, K.L.; Keith, M.S.; Hodgkins, P.; Smyth, M.; Goldsmith, D.; Akehurst, R. Cost-effectiveness of lanthanum carbonate versus sevelamer hydrochloride for the treatment of hyperphosphatemia in patients with end-stage renal disease: A US payer perspective. Value Health J. Int. Soc. Pharmacoecon. Outcomes Res. 2011, 14, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Moe, S.M.; Shroff, R.; Tonelli, M.A.; Toussaint, N.D.; et al. Executive summary of the 2017 KDIGO Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) Guideline Update: What’s changed and why it matters. Kidney Int. 2017, 92, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Behets, G.J.; Spasovski, G.; Sterling, L.R.; Goodman, W.G.; Spiegel, D.M.; De Broe, M.E.; D’Haese, P.C. Bone histomorphometry before and after long-term treatment with cinacalcet in dialysis patients with secondary hyperparathyroidism. Kidney Int. 2015, 87, 846–856. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, J.; Danese, M.; Olson, K.; Klassen, P.; Chertow, G.M. Effects of the calcimimetic cinacalcet HCl on cardiovascular disease, fracture, and health-related quality of life in secondary hyperparathyroidism. Kidney Int. 2005, 68, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P.; Chertow, G.M.; Torres, P.U.; Csiky, B.; Naso, A.; Nossuli, K.; Moustafa, M.; Goodman, W.G.; Lopez, N.; Downey, G.; et al. The ADVANCE study: A randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol. Dial. Transplant. 2011, 26, 1327–1339. [Google Scholar] [CrossRef] [PubMed]
- Investigators, E.T.; Chertow, G.M.; Block, G.A.; Correa-Rotter, R.; Drueke, T.B.; Floege, J.; Goodman, W.G.; Herzog, C.A.; Kubo, Y.; London, G.M.; et al. Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N. Engl. J. Med. 2012, 367, 2482–2494. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Abdalla, S.; Chertow, G.M.; Parfrey, P.S.; Block, G.A.; Correa-Rotter, R.; Floege, J.; Herzog, C.A.; London, G.M.; Mahaffey, K.W.; et al. Effects of Cinacalcet on Fracture Events in Patients Receiving Hemodialysis: The EVOLVE Trial. J. Am. Soc. Nephrol. 2015, 26, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.M.; Chertow, G.M.; Parfrey, P.S.; Kubo, Y.; Block, G.A.; Correa-Rotter, R.; Drueke, T.B.; Herzog, C.A.; London, G.M.; Mahaffey, K.W.; et al. Cinacalcet, Fibroblast Growth Factor-23, and Cardiovascular Disease in Hemodialysis: The Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) Trial. Circulation 2015, 132, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Nistor, I.; Craig, J.C.; Pellegrini, F.; Messa, P.; Tonelli, M.; Covic, A.; Strippoli, G.F. Cinacalcet in patients with chronic kidney disease: A cumulative meta-analysis of randomized controlled trials. PLoS Med. 2013, 10, e1001436. [Google Scholar] [CrossRef] [PubMed]
- Komaba, H.; Taniguchi, M.; Wada, A.; Iseki, K.; Tsubakihara, Y.; Fukagawa, M. Parathyroidectomy and survival among Japanese hemodialysis patients with secondary hyperparathyroidism. Kidney Int. 2015, 88, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, M.; Komaba, H.; Fukagawa, M. Parathyroid function in chronic kidney disease: Role of FGF23-Klotho axis. Contrib. Nephrol. 2013, 180, 110–123. [Google Scholar] [PubMed]
- Shalhoub, V.; Shatzen, E.M.; Ward, S.C.; Davis, J.; Stevens, J.; Bi, V.; Renshaw, L.; Hawkins, N.; Wang, W.; Chen, C.; et al. FGF23 neutralization improves chronic kidney disease-associated hyperparathyroidism yet increases mortality. J. Clin. Investig. 2012, 122, 2543–2553. [Google Scholar] [CrossRef] [PubMed]
- Faul, C.; Amaral, A.P.; Oskouei, B.; Hu, M.C.; Sloan, A.; Isakova, T.; Gutierrez, O.M.; Aguillon-Prada, R.; Lincoln, J.; Hare, J.M.; et al. FGF23 induces left ventricular hypertrophy. J. Clin. Investig. 2011, 121, 4393–4408. [Google Scholar] [CrossRef] [PubMed]
- Rossaint, J.; Oehmichen, J.; Van Aken, H.; Reuter, S.; Pavenstadt, H.J.; Meersch, M.; Unruh, M.; Zarbock, A. FGF23 signaling impairs neutrophil recruitment and host defense during CKD. J. Clin. Investig. 2016, 126, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Li, X.H.; Feng, L.; Yang, Z.H.; Liao, Y.H. The Effect of Active Vitamin D on Cardiovascular Outcomes in Predialysis Chronic Kidney Diseases: A Systematic Review and Meta-Analysis. Nephrology 2015. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wan, X.; Huang, Z.; Zeng, F.; Wei, G.; Fang, D.; Deng, W.; Li, Y. Impact of vitamin D on chronic kidney diseases in non-dialysis patients: A meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e61387. [Google Scholar] [CrossRef] [PubMed]
- de Zeeuw, D.; Agarwal, R.; Amdahl, M.; Audhya, P.; Coyne, D.; Garimella, T.; Parving, H.H.; Pritchett, Y.; Remuzzi, G.; Ritz, E.; et al. Selective vitamin D receptor activation with paricalcitol for reduction of albuminuria in patients with type 2 diabetes (VITAL study): A randomised controlled trial. Lancet 2010, 376, 1543–1551. [Google Scholar] [CrossRef]
- Thadhani, R.; Appelbaum, E.; Pritchett, Y.; Chang, Y.; Wenger, J.; Tamez, H.; Bhan, I.; Agarwal, R.; Zoccali, C.; Wanner, C.; et al. Vitamin D therapy and cardiac structure and function in patients with chronic kidney disease: The PRIMO randomized controlled trial. JAMA 2012, 307, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, J.; Cobbe, S.M.; Ford, I.; Isles, C.G.; Lorimer, A.R.; MacFarlane, P.W.; McKillop, J.H.; Packard, C.J. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N. Engl. J. Med. 1995, 333, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Danielson, E.; Fonseca, F.A.; Genest, J.; Gotto, A.M., Jr.; Kastelein, J.J.; Koenig, W.; Libby, P.; Lorenzatti, A.J.; MacFadyen, J.G.; et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 2008, 359, 2195–2207. [Google Scholar] [CrossRef] [PubMed]
- Collins, R.; Reith, C.; Emberson, J.; Armitage, J.; Baigent, C.; Blackwell, L.; Blumenthal, R.; Danesh, J.; Smith, G.D.; DeMets, D.; et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet 2016, 388, 2532–2561. [Google Scholar] [CrossRef]
- Tonelli, M.; Wanner, C. Lipid management in chronic kidney disease: Synopsis of the Kidney Disease: Improving Global Outcomes 2013 clinical practice guideline. Ann. Intern. Med. 2014, 160, 182. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Isles, C.; Curhan, G.C.; Tonkin, A.; Pfeffer, M.A.; Shepherd, J.; Sacks, F.M.; Furberg, C.; Cobbe, S.M.; Simes, J.; et al. Effect of pravastatin on cardiovascular events in people with chronic kidney disease. Circulation 2004, 110, 1557–1563. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Zhang, L.; Lv, J.; Wang, J.; Hou, W.; Xie, X.; Zhang, H. Effect of Statins on Kidney Disease Outcomes: A Systematic Review and Meta-analysis. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 67, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, P.; Zhang, J.; Wang, S.; Zhang, G. The effect of statins on microalbuminuria, proteinuria, progression of kidney function, and all-cause mortality in patients with non-end stage chronic kidney disease: A meta-analysis. Pharmacol. Res. 2016, 105, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Baigent, C.; Landray, M.J.; Reith, C.; Emberson, J.; Wheeler, D.C.; Tomson, C.; Wanner, C.; Krane, V.; Cass, A.; Craig, J.; et al. The effects of lowering LDL cholesterol with simvastatin plus ezetimibe in patients with chronic kidney disease (Study of Heart and Renal Protection): A randomised placebo-controlled trial. Lancet 2011, 377, 2181–2192. [Google Scholar] [CrossRef]
- Upadhyay, A.; Earley, A.; Lamont, J.L.; Haynes, S.; Wanner, C.; Balk, E.M. Lipid-lowering therapy in persons with chronic kidney disease: A systematic review and meta-analysis. Ann. Intern. Med. 2012, 157, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Navaneethan, S.D.; Craig, J.C.; Johnson, D.W.; Perkovic, V.; Hegbrant, J.; Strippoli, G.F. HMG CoA reductase inhibitors (statins) for people with chronic kidney disease not requiring dialysis. Cochrane Database Syst. Rev. 2014, 5, CD007784. [Google Scholar]
- Haynes, R.; Lewis, D.; Emberson, J.; Reith, C.; Agodoa, L.; Cass, A.; Craig, J.C.; de Zeeuw, D.; Feldt-Rasmussen, B.; Fellstrom, B.; et al. Effects of lowering LDL cholesterol on progression of kidney disease. J. Am. Soc. Nephrol. 2014, 25, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Chronic Kidney Disease in Adults | Guidance and Guidelines | NICE. Available online: https://www.nice.org.uk/guidance/qs5/chapter/Quality-statement-3-Statins-for-people-with-CKD#quality-statement-3 (accessed on 20 May 2018).
- Fellstrom, B.C.; Jardine, A.G.; Schmieder, R.E.; Holdaas, H.; Bannister, K.; Beutler, J.; Chae, D.W.; Chevaile, A.; Cobbe, S.M.; Gronhagen-Riska, C.; et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis. N. Engl. J. Med. 2009, 360, 1395–1407. [Google Scholar] [CrossRef] [PubMed]
- Navaneethan, S.D.; Nigwekar, S.U.; Perkovic, V.; Johnson, D.W.; Craig, J.C.; Strippoli, G.F. HMG CoA reductase inhibitors (statins) for dialysis patients. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef]
- Wanner, C.; Krane, V.; Marz, W.; Olschewski, M.; Mann, J.F.; Ruf, G.; Ritz, E. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis. N. Engl. J. Med. 2005, 353, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Massy, Z.A.; de Zeeuw, D. LDL cholesterol in CKD—To treat or not to treat? Kidney Int. 2013, 84, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.S.; Nallamothu, B.K.; Gurm, H.S.; Hayward, R.A.; Vijan, S. Incremental benefit and cost-effectiveness of high-dose statin therapy in high-risk patients with coronary artery disease. Circulation 2007, 115, 2398–2409. [Google Scholar] [CrossRef] [PubMed]
- Stam-Slob, M.C.; van der Graaf, Y.; Greving, J.P.; Dorresteijn, J.A.; Visseren, F.L. Cost-Effectiveness of Intensifying Lipid-Lowering Therapy With Statins Based on Individual Absolute Benefit in Coronary Artery Disease Patients. J. Am. Heart Assoc. 2017, 6, e004648. [Google Scholar] [CrossRef] [PubMed]
- Pandya, A.; Sy, S.; Cho, S.; Weinstein, M.C.; Gaziano, T.A. Cost-effectiveness of 10-Year Risk Thresholds for Initiation of Statin Therapy for Primary Prevention of Cardiovascular Disease. JAMA 2015, 314, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.P.; Simpson, R.J. Statin cost effectiveness in primary prevention: A systematic review of the recent cost-effectiveness literature in the United States. BMC Res. Notes 2012, 5, 373. [Google Scholar] [CrossRef] [PubMed]
- Odden, M.C.; Pletcher, M.J.; Coxson, P.G.; Thekkethala, D.; Guzman, D.; Heller, D.; Goldman, L.; Bibbins-Domingo, K. Cost-effectiveness and population impact of statins for primary prevention in adults aged 75 years or older in the United States. Ann. Intern. Med. 2015, 162, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Kent, S.; Schlackow, I.; Lozano-Kuhne, J.; Reith, C.; Emberson, J.; Haynes, R.; Gray, A.; Cass, A.; Baigent, C.; Landray, M.J.; et al. What is the impact of chronic kidney disease stage and cardiovascular disease on the annual cost of hospital care in moderate-to-severe kidney disease? BMC Nephrol. 2015, 16, 65. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.F.; Japa, S.; Owens, D.K.; Chertow, G.M.; Garber, A.M.; Goldhaber-Fiebert, J.D. Cost-effectiveness of statins for primary cardiovascular prevention in chronic kidney disease. J. Am. Coll. Cardiol. 2013, 61, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Mihaylova, B.; Schlackow, I.; Herrington, W.; Lozano-Kuhne, J.; Kent, S.; Emberson, J.; Reith, C.; Haynes, R.; Cass, A.; Craig, J.; et al. Cost-effectiveness of Simvastatin plus Ezetimibe for Cardiovascular Prevention in CKD: Results of the Study of Heart and Renal Protection (SHARP). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 67, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Markossian, T.; Burge, N.; Ling, B.; Schneider, J.; Pacold, I.; Bansal, V.; Leehey, D.; Stroupe, K.; Chang, A.; Kramer, H. Controversies Regarding Lipid Management and Statin Use for Cardiovascular Risk Reduction in Patients With CKD. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2016, 67, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Dewey, F.E.; Gusarova, V.; Dunbar, R.L.; O’Dushlaine, C.; Schurmann, C.; Gottesman, O.; McCarthy, S.; Van Hout, C.V.; Bruse, S.; Dansky, H.M.; et al. Genetic and Pharmacologic Inactivation of ANGPTL3 and Cardiovascular Disease. N. Engl. J. Med. 2017, 377, 211–221. [Google Scholar] [CrossRef] [PubMed]
- FOURIER: Is Evolocumab Worth the Cost? Available online: https://www.pcronline.com/News/Whats-new-on-PCRonline/2017/FOURIER-Is-evolocumab-worth-the-cost (accessed on 20 May 2018).
- Villa, G.; Lothgren, M.; Kutikova, L.; Lindgren, P.; Gandra, S.R.; Fonarow, G.C.; Sorio, F.; Masana, L.; Bayes-Genis, A.; Hout, B.V. Cost-effectiveness of Evolocumab in Patients With High Cardiovascular Risk in Spain. Clin. Ther. 2017, 39, 771–786. [Google Scholar] [CrossRef] [PubMed]
- Gandra, S.R.; Villa, G.; Fonarow, G.C.; Lothgren, M.; Lindgren, P.; Somaratne, R.; van Hout, B. Cost-Effectiveness of LDL-C Lowering With Evolocumab in Patients With High Cardiovascular Risk in the United States. Clin. Cardiol. 2016, 39, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Krumholz, H.M. Treatment of Cholesterol in 2017. JAMA 2017, 318, 417–418. [Google Scholar] [CrossRef] [PubMed]
- Combadiere, C.; Potteaux, S.; Rodero, M.; Simon, T.; Pezard, A.; Esposito, B.; Merval, R.; Proudfoot, A.; Tedgui, A.; Mallat, Z. Combined inhibition of CCL2, CX3CR1, and CCR5 abrogates Ly6C(hi) and Ly6C(lo) monocytosis and almost abolishes atherosclerosis in hypercholesterolemic mice. Circulation 2008, 117, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Pergola, P.E.; Raskin, P.; Toto, R.D.; Meyer, C.J.; Huff, J.W.; Grossman, E.B.; Krauth, M.; Ruiz, S.; Audhya, P.; Christ-Schmidt, H.; et al. Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N. Engl. J. Med. 2011, 365, 327–336. [Google Scholar] [CrossRef] [PubMed]
- De Zeeuw, D.; Akizawa, T.; Audhya, P.; Bakris, G.L.; Chin, M.; Christ-Schmidt, H.; Goldsberry, A.; Houser, M.; Krauth, M.; Lambers Heerspink, H.J.; et al. Bardoxolone methyl in type 2 diabetes and stage 4 chronic kidney disease. N. Engl. J. Med. 2013, 369, 2492–2503. [Google Scholar] [CrossRef] [PubMed]
- Van Laecke, S.; Van Biesen, W.; Vanholder, R. The paradox of bardoxolone methyl: A call for every witness on the stand? Diabetes Obes. Metab. 2015, 17, 9–14. [Google Scholar] [CrossRef] [PubMed]
- First Alport Patient Enrolls in Phase 3 trial of Bardoxolone Methyl for Kidney Disease. Available online: https://alportsyndromenews.com/2017/08/09/first-alport-syndrome-patient-enrolls-in-phase-3-trial-of-bardoxolone-methyl-for-kidney-disease/ (accessed on 20 May 2018).
- Toto, R.D. Bardoxolone-the Phoenix? J. Am. Soc. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Baigent, C.; Lennon, R. Should We Increase GFR with Bardoxolone in Alport Syndrome? J. Am. Soc. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- De Zeeuw, D.; Bekker, P.; Henkel, E.; Hasslacher, C.; Gouni-Berthold, I.; Mehling, H.; Potarca, A.; Tesar, V.; Heerspink, H.J.; Schall, T.J.; et al. The effect of CCR2 inhibitor CCX140-B on residual albuminuria in patients with type 2 diabetes and nephropathy: A randomised trial. Lancet Diabetes Endocrinol. 2015, 3, 687–696. [Google Scholar] [CrossRef]
- Weir, M.R. CCR2 inhibition: A panacea for diabetic kidney disease? Lancet Diabetes Endocrinol. 2015, 3, 666–667. [Google Scholar] [CrossRef]
- Menne, J.; Eulberg, D.; Beyer, D.; Baumann, M.; Saudek, F.; Valkusz, Z.; Wiecek, A.; Haller, H.; Emapticap Study Group. C-C motif-ligand 2 inhibition with emapticap pegol (NOX-E36) in type 2 diabetic patients with albuminuria. Nephrol. Dial. Transplant. 2017, 32, 307–315. [Google Scholar] [PubMed]
- Kuo, K.L.; Hung, S.C.; Liu, J.S.; Chang, Y.K.; Hsu, C.C.; Tarng, D.C. Add-on Protective Effect of Pentoxifylline in Advanced Chronic Kidney Disease Treated with Renin-Angiotensin-Aldosterone System Blockade—A Nationwide Database Analysis. Sci. Rep. 2015, 5, 17150. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Gonzalez, J.F.; Mora-Fernandez, C.; Muros de Fuentes, M.; Chahin, J.; Mendez, M.L.; Gallego, E.; Macia, M.; del Castillo, N.; Rivero, A.; Getino, M.A.; et al. Effect of pentoxifylline on renal function and urinary albumin excretion in patients with diabetic kidney disease: The PREDIAN trial. J. Am. Soc. Nephrol. 2015, 26, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Wang, L.N.; Li, H.X.; Huang, P.; Qu, L.B.; Chen, F.Y. Pentoxifylline plus ACEIs/ARBs for proteinuria and kidney function in chronic kidney disease: A meta-analysis. J. Int. Med. Res. 2017, 45, 383–398. [Google Scholar] [CrossRef] [PubMed]
- D’Aiuto, F.; Nibali, L.; Parkar, M.; Suvan, J.; Tonetti, M.S. Short-term effects of intensive periodontal therapy on serum inflammatory markers and cholesterol. J. Dent. Res. 2005, 84, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.D.; Eke, P.; Lin, D.; Madianos, P.; Couper, D.; Moss, K.; Elter, J.; Heiss, G.; Offenbacher, S. Associations between IgG antibody to oral organisms and carotid intima-medial thickness in community-dwelling adults. Atherosclerosis 2005, 183, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, L.; Skilton, M.; Maple-Brown, L.; Kapellas, K.; Askie, L.; Hughes, J.; Arrow, P.; Cherian, S.; Fernandes, D.; Pawar, B.; et al. Periodontal disease and chronic kidney disease among Aboriginal adults; an RCT. BMC Nephrol. 2015, 16, 181. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, J.; Rodriguez, M.; Messa, P. Magnesium in chronic kidney disease Stages 3 and 4 and in dialysis patients. Clin. Kidney J. 2012, 5, i39–i51. [Google Scholar] [CrossRef] [PubMed]
- Massy, Z.A.; Drueke, T.B. Magnesium and cardiovascular complications of chronic kidney disease. Nat. Rev. Nephrol. 2015, 11, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Montes de Oca, A.; Guerrero, F.; Martinez-Moreno, J.M.; Madueno, J.A.; Herencia, C.; Peralta, A.; Almaden, Y.; Lopez, I.; Aguilera-Tejero, E.; Gundlach, K.; et al. Magnesium inhibits Wnt/beta-catenin activity and reverses the osteogenic transformation of vascular smooth muscle cells. PLoS ONE 2014, 9, e89525. [Google Scholar] [CrossRef] [PubMed]
- Kircelli, F.; Peter, M.E.; Sevinc Ok, E.; Celenk, F.G.; Yilmaz, M.; Steppan, S.; Asci, G.; Ok, E.; Passlick-Deetjen, J. Magnesium reduces calcification in bovine vascular smooth muscle cells in a dose-dependent manner. Nephrol. Dial. Transplant. 2012, 27, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Louvet, L.; Buchel, J.; Steppan, S.; Passlick-Deetjen, J.; Massy, Z.A. Magnesium prevents phosphate-induced calcification in human aortic vascular smooth muscle cells. Nephrol. Dial. Transplant. 2013, 28, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Meema, H.E.; Oreopoulos, D.G.; Rapoport, A. Serum magnesium level and arterial calcification in end-stage renal disease. Kidney Int. 1987, 32, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Shechter, M.; Sharir, M.; Labrador, M.J.; Forrester, J.; Silver, B.; Bairey Merz, C.N. Oral magnesium therapy improves endothelial function in patients with coronary artery disease. Circulation 2000, 102, 2353–2358. [Google Scholar] [CrossRef] [PubMed]
- de Francisco, A.L.; Leidig, M.; Covic, A.C.; Ketteler, M.; Benedyk-Lorens, E.; Mircescu, G.M.; Scholz, C.; Ponce, P.; Passlick-Deetjen, J. Evaluation of calcium acetate/magnesium carbonate as a phosphate binder compared with sevelamer hydrochloride in haemodialysis patients: A controlled randomized study (CALMAG study) assessing efficacy and tolerability. Nephrol. Dial. Transplant. 2010, 25, 3707–3717. [Google Scholar] [CrossRef] [PubMed]
- Turgut, F.; Kanbay, M.; Metin, M.R.; Uz, E.; Akcay, A.; Covic, A. Magnesium supplementation helps to improve carotid intima media thickness in patients on hemodialysis. Int. Urol. Nephrol. 2008, 40, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, M.; Moeinzadeh, F.; Saadatnia, M.; Shahidi, S.; McGee, J.C.; Minagar, A. Effect of magnesium supplementation on carotid intima-media thickness and flow-mediated dilatation among hemodialysis patients: A double-blind, randomized, placebo-controlled trial. Eur. Neurol. 2013, 69, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Bressendorff, I.; Hansen, D.; Schou, M.; Silver, B.; Pasch, A.; Bouchelouche, P.; Pedersen, L.; Rasmussen, L.M.; Brandi, L. Oral Magnesium Supplementation in Chronic Kidney Disease Stages 3 and 4: Efficacy, Safety, and Effect on Serum Calcification Propensity-A Prospective Randomized Double-Blinded Placebo-Controlled Clinical Trial. Kidney Int. Rep. 2017, 2, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Tzanakis, I.P.; Stamataki, E.E.; Papadaki, A.N.; Giannakis, N.; Damianakis, N.E.; Oreopoulos, D.G. Magnesium retards the progress of the arterial calcifications in hemodialysis patients: A pilot study. Int. Urol. Nephrol. 2014, 46, 2199–2205. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Fang, X.; Wei, X.; Liu, Y.; Jin, Z.; Chen, Q.; Fan, Z.; Aaseth, J.; Hiyoshi, A.; He, J.; et al. Dose-response relationship between dietary magnesium intake, serum magnesium concentration and risk of hypertension: A systematic review and meta-analysis of prospective cohort studies. Nutr. J. 2017, 16, 26. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, Y.; Iwatani, H.; Hamano, T.; Tomida, K.; Kawabata, H.; Kusunoki, Y.; Shimomura, A.; Matsui, I.; Hayashi, T.; Tsubakihara, Y.; et al. Magnesium modifies the association between serum phosphate and the risk of progression to end-stage kidney disease in patients with non-diabetic chronic kidney disease. Kidney Int. 2015, 88, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Van Laecke, S.; Nagler, E.V.; Verbeke, F.; Van Biesen, W.; Vanholder, R. Hypomagnesemia and the risk of death and GFR decline in chronic kidney disease. Am. J. Med. 2013, 126, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, Y.; Fujii, N.; Shoji, T.; Hayashi, T.; Rakugi, H.; Isaka, Y. Hypomagnesemia is a significant predictor of cardiovascular and non-cardiovascular mortality in patients undergoing hemodialysis. Kidney Int. 2014, 85, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Streja, E.; Rhee, C.M.; Mehrotra, R.; Soohoo, M.; Brunelli, S.M.; Kovesdy, C.P.; Kalantar-Zadeh, K. Hypomagnesemia and Mortality in Incident Hemodialysis Patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2015, 66, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, Y.; Hamano, T.; Wada, A.; Hoshino, J.; Masakane, I. Magnesium and Risk of Hip Fracture among Patients Undergoing Hemodialysis. J. Am. Soc. Nephrol. 2017. [Google Scholar] [CrossRef] [PubMed]
- De Roij van Zuijdewijn, C.L.; Grooteman, M.P.; Bots, M.L.; Blankestijn, P.J.; Steppan, S.; Buchel, J.; Groenwold, R.H.; Brandenburg, V.; van den Dorpel, M.A.; Ter Wee, P.M.; et al. Serum Magnesium and Sudden Death in European Hemodialysis Patients. PLoS ONE 2015, 10, e0143104. [Google Scholar] [CrossRef] [PubMed]
- De Francisco, A.L.; Rodríguez, M. Magnesium—Its role in CKD. Nefrologia 2013, 33, 389–399. [Google Scholar]
- Van Laecke, S.; Caluwe, R.; Huybrechts, I.; Nagler, E.V.; Vanholder, R.; Peeters, P.; Van Vlem, B.; Van Biesen, W. Effect of Magnesium Supplements on Insulin Secretion After Kidney Transplantation: A Randomized Controlled Trial. Ann. Transplant. 2017, 22, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Mitch, W.E.; Medina, R.; Grieber, S.; May, R.C.; England, B.K.; Price, S.R.; Bailey, J.L.; Goldberg, A.L. Metabolic acidosis stimulates muscle protein degradation by activating the adenosine triphosphate-dependent pathway involving ubiquitin and proteasomes. J. Clin. Investig. 1994, 93, 2127–2133. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.P.; Hasbargen, J.A. Correction of metabolic acidosis and its effect on albumin in chronic hemodialysis patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 1998, 31, 35–40. [Google Scholar] [CrossRef]
- De Brito-Ashurst, I.; Varagunam, M.; Raftery, M.J.; Yaqoob, M.M. Bicarbonate supplementation slows progression of CKD and improves nutritional status. J. Am. Soc. Nephrol. 2009, 20, 2075–2084. [Google Scholar] [CrossRef] [PubMed]
- Susantitaphong, P.; Sewaralthahab, K.; Balk, E.M.; Jaber, B.L.; Madias, N.E. Short- and long-term effects of alkali therapy in chronic kidney disease: A systematic review. Am. J. Nephrol. 2012, 35, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Kovesdy, C.P.; Anderson, J.E.; Kalantar-Zadeh, K. Association of serum bicarbonate levels with mortality in patients with non-dialysis-dependent CKD. Nephrol. Dial. Transplant. 2009, 24, 1232–1237. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Shoji, S.; Yamakawa, T.; Wada, A.; Suzuki, K.; Iseki, K.; Tsubakihara, Y. Predialysis and Postdialysis pH and Bicarbonate and Risk of All-Cause and Cardiovascular Mortality in Long-term Hemodialysis Patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2015, 66, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Bommer, J.; Locatelli, F.; Satayathum, S.; Keen, M.L.; Goodkin, D.A.; Saito, A.; Akiba, T.; Port, F.K.; Young, E.W. Association of predialysis serum bicarbonate levels with risk of mortality and hospitalization in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2004, 44, 661–671. [Google Scholar] [CrossRef]
- Tentori, F.; Karaboyas, A.; Robinson, B.M.; Morgenstern, H.; Zhang, J.; Sen, A.; Ikizler, T.A.; Rayner, H.; Fissell, R.B.; Vanholder, R.; et al. Association of dialysate bicarbonate concentration with mortality in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2013, 62, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Fernandez, B.; Martin-Cleary, C.; Ortiz, A. Bicarbonate therapy, phosphate binders, and risk for vascular calcification. Kidney Int. 2014, 86, 1056. [Google Scholar] [CrossRef] [PubMed]
- Stevens, E.A.; Mezrich, J.D.; Bradfield, C.A. The aryl hydrocarbon receptor: A perspective on potential roles in the immune system. Immunology 2009, 127, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Mulero-Navarro, S.; Fernandez-Salguero, P.M. New Trends in Aryl Hydrocarbon Receptor Biology. Front. Cell Dev. Biol. 2016, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Gondouin, B.; Cerini, C.; Dou, L.; Sallee, M.; Duval-Sabatier, A.; Pletinck, A.; Calaf, R.; Lacroix, R.; Jourde-Chiche, N.; Poitevin, S.; et al. Indolic uremic solutes increase tissue factor production in endothelial cells by the aryl hydrocarbon receptor pathway. Kidney Int. 2013, 84, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.C.; Dinatale, B.C.; Murray, I.A.; Flaveny, C.A.; Liu, Q.; Laurenzana, E.M.; Lin, J.M.; Strom, S.C.; Omiecinski, C.J.; Amin, S.; et al. The uremic toxin 3-indoxyl sulfate is a potent endogenous agonist for the human aryl hydrocarbon receptor. Biochemistry 2010, 49, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, M.; Tatebe, J.; Watanabe, I.; Yamazaki, J.; Ikeda, T.; Morita, T. Aryl hydrocarbon receptor mediates indoxyl sulfate-induced cellular senescence in human umbilical vein endothelial cells. J. Atheroscler. Thromb. 2014, 21, 904–916. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Osaka, M.; Edamatsu, T.; Itoh, Y.; Yoshida, M. Crucial Role of the Aryl Hydrocarbon Receptor (AhR) in Indoxyl Sulfate-Induced Vascular Inflammation. J. Atheroscler. Thromb. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sallee, M.; Dou, L.; Cerini, C.; Poitevin, S.; Brunet, P.; Burtey, S. The aryl hydrocarbon receptor-activating effect of uremic toxins from tryptophan metabolism: A new concept to understand cardiovascular complications of chronic kidney disease. Toxins 2014, 6, 934–949. [Google Scholar] [CrossRef] [PubMed]
- Brito, J.S.; Borges, N.A.; Esgalhado, M.; Magliano, D.C.; Soulage, C.O.; Mafra, D. Aryl Hydrocarbon Receptor Activation in Chronic Kidney Disease: Role of Uremic Toxins. Nephron 2017, 137, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shivanna, S.; Kolandaivelu, K.; Shashar, M.; Belghasim, M.; Al-Rabadi, L.; Balcells, M.; Zhang, A.; Weinberg, J.; Francis, J.; Pollastri, M.P.; et al. The Aryl Hydrocarbon Receptor is a Critical Regulator of Tissue Factor Stability and an Antithrombotic Target in Uremia. J. Am. Soc. Nephrol. 2016, 27, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Kolachalama, V.B.; Shashar, M.; Alousi, F.; Shivanna, S.; Rijal, K.; Belghasem, M.E.; Walker, J.; Matsuura, S.; Chang, G.H.; Gibson, C.M.; et al. Uremic Solute-Aryl Hydrocarbon Receptor-Tissue Factor Axis Associates with Thrombosis after Vascular Injury in Humans. J. Am. Soc. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Shashar, M.; Belghasem, M.E.; Matsuura, S.; Walker, J.; Richards, S.; Alousi, F.; Rijal, K.; Kolachalama, V.B.; Balcells, M.; Odagi, M.; et al. Targeting STUB1-tissue factor axis normalizes hyperthrombotic uremic phenotype without increasing bleeding risk. Sci. Transl. Med. 2017, 9, eaam8475. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, C.J.; Ciolino, H.P.; Yeh, G.C. The drug salicylamide is an antagonist of the aryl hydrocarbon receptor that inhibits signal transduction induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Cancer Res. 2004, 64, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Stanford, E.A.; Ramirez-Cardenas, A.; Wang, Z.; Novikov, O.; Alamoud, K.; Koutrakis, P.; Mizgerd, J.P.; Genco, C.A.; Kukuruzinska, M.; Monti, S.; et al. Role for the Aryl Hydrocarbon Receptor and Diverse Ligands in Oral Squamous Cell Carcinoma Migration and Tumorigenesis. Mol. Cancer Res. MCR 2016, 14, 696–706. [Google Scholar] [CrossRef] [PubMed]
- Kerkvliet, N.I. AHR-mediated immunomodulation: The role of altered gene transcription. Biochem. Pharmacol. 2009, 77, 746–760. [Google Scholar] [CrossRef] [PubMed]
- Reinecke, H.; Brand, E.; Mesters, R.; Schabitz, W.R.; Fisher, M.; Pavenstadt, H.; Breithardt, G. Dilemmas in the management of atrial fibrillation in chronic kidney disease. J. Am. Soc. Nephrol. 2009, 20, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, D.J.; Bernardo, E.; Capodanno, D.; Vivas, D.; Sabate, M.; Ferreiro, J.L.; Ueno, M.; Jimenez-Quevedo, P.; Alfonso, F.; Bass, T.A.; et al. Impact of chronic kidney disease on platelet function profiles in diabetes mellitus patients with coronary artery disease taking dual antiplatelet therapy. J. Am. Coll. Cardiol. 2010, 55, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Mangiacapra, F.; Cavallari, I.; Barbato, E.; Ricottini, E.; Patti, G.; Vizzi, V.; D’Ambrosio, A.; De Bruyne, B.; Wijns, W.; Di Sciascio, G. Impact of chronic kidney disease on platelet reactivity and outcomes of patients receiving clopidogrel and undergoing percutaneous coronary intervention. Am. J. Cardiol. 2014, 113, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Best, P.J.; Steinhubl, S.R.; Berger, P.B.; Dasgupta, A.; Brennan, D.M.; Szczech, L.A.; Califf, R.M.; Topol, E.J. The efficacy and safety of short- and long-term dual antiplatelet therapy in patients with mild or moderate chronic kidney disease: Results from the Clopidogrel for the Reduction of Events During Observation (CREDO) trial. Am. Heart J. 2008, 155, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Ethier, J.; Bragg-Gresham, J.L.; Piera, L.; Akizawa, T.; Asano, Y.; Mason, N.; Gillespie, B.W.; Young, E.W. Aspirin prescription and outcomes in hemodialysis patients: The Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2007, 50, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.H.; Cho, J.H.; Woo, J.S.; Kim, J.B.; Kim, W.S.; Lee, T.W.; Kim, K.S.; Ihm, C.G.; Kim, W. Platelet reactivity after receiving clopidogrel compared with ticagrelor in patients with kidney failure treated with hemodialysis: A randomized crossover study. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2015, 65, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.C.; Di Micco, L.; Razavian, M.; Craig, J.C.; Perkovic, V.; Pellegrini, F.; Copetti, M.; Graziano, G.; Tognoni, G.; Jardine, M.; et al. Effects of antiplatelet therapy on mortality and cardiovascular and bleeding outcomes in persons with chronic kidney disease: A systematic review and meta-analysis. Ann. Intern. Med. 2012, 156, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Keskar, V.; McArthur, E.; Wald, R.; Harel, Z.; Zimmerman, D.; Molnar, A.O.; Garg, A.X.; Lam, N.N.; McCallum, M.K.; Bota, S.E.; et al. The association of anticoagulation, ischemic stroke, and hemorrhage in elderly adults with chronic kidney disease and atrial fibrillation. Kidney Int. 2017, 91, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.E.; Lazarus, J.M.; Thadhani, R.; Hakim, R.M. Warfarin use associates with increased risk for stroke in hemodialysis patients with atrial fibrillation. J. Am. Soc. Nephrol. 2009, 20, 2223–2233. [Google Scholar] [CrossRef] [PubMed]
- Bonde, A.N.; Lip, G.Y.; Kamper, A.L.; Hansen, P.R.; Lamberts, M.; Hommel, K.; Hansen, M.L.; Gislason, G.H.; Torp-Pedersen, C.; Olesen, J.B. Net clinical benefit of antithrombotic therapy in patients with atrial fibrillation and chronic kidney disease: A nationwide observational cohort study. J. Am. Coll. Cardiol. 2014, 64, 2471–2482. [Google Scholar] [CrossRef] [PubMed]
- Marinigh, R.; Lane, D.A.; Lip, G.Y. Severe renal impairment and stroke prevention in atrial fibrillation: Implications for thromboprophylaxis and bleeding risk. J. Am. Coll. Cardiol. 2011, 57, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.; Szeki, I.; Nash, M.J.; Thachil, J. Anticoagulation in chronic kidney disease patients-the practical aspects. Clin. Kidney J. 2014, 7, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Heine, G.H.; Brandenburg, V. Anticoagulation, atrial fibrillation, and chronic kidney disease-whose side are you on? Kidney Int. 2017, 91, 778–780. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.E.; Giugliano, R.P.; Patel, M.R.; Abramson, S.; Jardine, M.; Zhao, S.; Perkovic, V.; Maddux, F.W.; Piccini, J.P. Nonvitamin K Anticoagulant Agents in Patients With Advanced Chronic Kidney Disease or on Dialysis With AF. J. Am. Coll. Cardiol. 2016, 67, 2888–2899. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Lopez, J.A.; Sterne, J.A.C.; Thom, H.H.Z.; Higgins, J.P.T.; Hingorani, A.D.; Okoli, G.N.; Davies, P.A.; Bodalia, P.N.; Bryden, P.A.; Welton, N.J.; et al. Oral anticoagulants for prevention of stroke in atrial fibrillation: Systematic review, network meta-analysis, and cost effectiveness analysis. BMJ 2017, 359, j5058. [Google Scholar] [CrossRef] [PubMed]
- Lutz, J.; Jurk, K.; Schinzel, H. Direct oral anticoagulants in patients with chronic kidney disease: Patient selection and special considerations. Int. J. Nephrol. Renovasc. Dis. 2017, 10, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Willett, K.C.; Morrill, A.M. Use of direct oral anticoagulants for the prevention and treatment of thromboembolic disease in patients with reduced renal function: A short review of the clinical evidence. Ther. Clin. Risk Manag. 2017, 13, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, S.V.; Nadasdy, T.; Rovin, B.H.; Satoskar, A.A.; Nadasdy, G.M.; Wu, H.M.; Bhatt, U.Y.; Hebert, L.A. Warfarin-related nephropathy occurs in patients with and without chronic kidney disease and is associated with an increased mortality rate. Kidney Int. 2011, 80, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, S.V.; Hebert, L.A. Anticoagulant-Related Nephropathy: Is an AKI Elephant Hiding in Plain View? J. Am. Coll. Cardiol. 2016, 68, 2284–2286. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.; Connolly, S.J. Another Dimension of Safety in the Prescription of Anticoagulants for Nonvalvular Atrial Fibrillation. J. Am. Coll. Cardiol. 2017, 70, 2633–2635. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Tangri, N.; Gersh, B.J.; Sangaralingham, L.R.; Shah, N.D.; Nath, K.A.; Noseworthy, P.A. Renal Outcomes in Anticoagulated Patients With Atrial Fibrillation. J. Am. Coll. Cardiol. 2017, 70, 2621–2632. [Google Scholar] [CrossRef] [PubMed]
- Bohm, M.; Ezekowitz, M.D.; Connolly, S.J.; Eikelboom, J.W.; Hohnloser, S.H.; Reilly, P.A.; Schumacher, H.; Brueckmann, M.; Schirmer, S.H.; Kratz, M.T.; et al. Changes in Renal Function in Patients With Atrial Fibrillation: An Analysis From the RE-LY Trial. J. Am. Coll. Cardiol. 2015, 65, 2481–2493. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.H.; Yeh, Y.H.; See, L.C.; Wang, C.L.; Chang, S.H.; Lee, H.F.; Wu, L.S.; Tu, H.T.; Kuo, C.T. Acute Kidney Injury in Asians With Atrial Fibrillation Treated With Dabigatran or Warfarin. J. Am. Coll. Cardiol. 2016, 68, 2272–2283. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, D.; Goncalves, N.; Pinto, F.J.; Costa, J.; Ferreira, J.J. Risk of renal failure with the non-vitamin K antagonist oral anticoagulants: Systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2015, 24, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Breyer, M.D.; Susztak, K. The next generation of therapeutics for chronic kidney disease. Nat. Rev. Drug Discov. 2016, 15, 568–588. [Google Scholar] [CrossRef] [PubMed]
- Yanagita, M. Inhibitors/antagonists of TGF-beta system in kidney fibrosis. Nephrol. Dial. Transplant. 2012, 27, 3686–3691. [Google Scholar] [CrossRef] [PubMed]
- Loeffler, I.; Wolf, G. Transforming growth factor-beta and the progression of renal disease. Nephrol. Dial. Transplant. 2014, 29 (Suppl. 1), i37–i45. [Google Scholar] [CrossRef] [PubMed]
- Benigni, A.; Zoja, C.; Corna, D.; Zatelli, C.; Conti, S.; Campana, M.; Gagliardini, E.; Rottoli, D.; Zanchi, C.; Abbate, M.; et al. Add-on anti-TGF-beta antibody to ACE inhibitor arrests progressive diabetic nephropathy in the rat. J. Am. Soc. Nephrol. 2003, 14, 1816–1824. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.E.; Smith, D.C.; Branton, M.H.; Penzak, S.R.; Kopp, J.B. Pirfenidone slows renal function decline in patients with focal segmental glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 2007, 2, 906–913. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Ix, J.H.; Mathew, A.V.; Cho, M.; Pflueger, A.; Dunn, S.R.; Francos, B.; Sharma, S.; Falkner, B.; McGowan, T.A.; et al. Pirfenidone for diabetic nephropathy. J. Am. Soc. Nephrol. 2011, 22, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, Q.; Simon, T.C.; Strebeck, F.; Chaudhary, L.; Morrissey, J.; Liapis, H.; Klahr, S.; Hruska, K.A. Bone morphogenic protein-7 (BMP-7), a novel therapy for diabetic nephropathy. Kidney Int. 2003, 63, 2037–2049. [Google Scholar] [CrossRef] [PubMed]
- Li, R.X.; Yiu, W.H.; Tang, S.C. Role of bone morphogenetic protein-7 in renal fibrosis. Front. Physiol. 2015, 6, 114. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Agarwal, R.; Acharya, M.; Berl, T.; Blumenthal, S.; Kopyt, N. A multicenter, randomized, double-blind, placebo-controlled, dose-ranging study of AST-120 (Kremezin) in patients with moderate to severe CKD. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2006, 47, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Itoh, Y.; Tateoka, R.; Ezawa, A.; Murakami, K.; Niwa, T. Metabolomic search for uremic toxins as indicators of the effect of an oral sorbent AST-120 by liquid chromatography/tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2010, 878, 2997–3002. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, J.; Tanaka, T.; Inagi, R. Effect of AST-120 in Chronic Kidney Disease Treatment: Still a Controversy? Nephron 2017, 135, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; Schepers, E.; Pletinck, A.; Nagler, E.V.; Glorieux, G. The uremic toxicity of indoxyl sulfate and p-cresyl sulfate: A systematic review. J. Am. Soc. Nephrol. 2014, 25, 1897–1907. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Vanholder, R.; Niwa, T. AST-120 for the management of progression of chronic kidney disease. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Konishi, K.; Nakano, S.; Tsuda, S.; Nakagawa, A.; Kigoshi, T.; Koya, D. AST-120 (Kremezin) initiated in early stage chronic kidney disease stunts the progression of renal dysfunction in type 2 diabetic subjects. Diabetes Res. Clin. Pract. 2008, 81, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Akizawa, T.; Asano, Y.; Morita, S.; Wakita, T.; Onishi, Y.; Fukuhara, S.; Gejyo, F.; Matsuo, S.; Yorioka, N.; Kurokawa, K.; et al. Effect of a carbonaceous oral adsorbent on the progression of CKD: A multicenter, randomized, controlled trial. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2009, 54, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Sato, E.; Fujiwara, N.; Kawagoe, Y.; Suzuki, T.; Ueda, Y.; Yamagishi, S. Oral adsorbent AST-120 ameliorates tubular injury in chronic renal failure patients by reducing proteinuria and oxidative stress generation. Metab. Clin. Exp. 2011, 60, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.W.; Hsu, K.H.; Sun, C.Y.; Tsai, C.J.; Wu, M.S.; Lee, C.C. Oral adsorbent AST-120 potentiates the effect of erythropoietin-stimulating agents on Stage 5 chronic kidney disease patients: A randomized crossover study. Nephrol. Dial. Transplant. 2014, 29, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Hayashino, Y.; Fukuhara, S.; Akizawa, T.; Asano, Y.; Wakita, T.; Onishi, Y.; Kurokawa, K. Cost-effectiveness of administering oral adsorbent AST-120 to patients with diabetes and advance-stage chronic kidney disease. Diabetes Res. Clin. Pract. 2010, 90, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Berl, T.; Beck, G.J.; Remuzzi, G.; Ritz, E.; Arita, K.; Kato, A.; Shimizu, M. Randomized Placebo-Controlled EPPIC Trials of AST-120 in CKD. J. Am. Soc. Nephrol. 2015, 26, 1732–1746. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Berl, T.; Beck, G.J.; Remuzzi, G.; Ritz, E.; Shimizu, M.; Shobu, Y.; Kikuchi, M. The effects of AST-120 on chronic kidney disease progression in the United States of America: A post hoc subgroup analysis of randomized controlled trials. BMC Nephrol. 2016, 17, 141. [Google Scholar] [CrossRef] [PubMed]
- Schulman, G.; Berl, T.; Beck, G.J.; Remuzzi, G.; Ritz, E.; Shimizu, M.; Kikuchi, M.; Shobu, Y. Risk factors for progression of chronic kidney disease in the EPPIC trials and the effect of AST-120. Clin. Exp. Nephrol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Cha, R.H.; Kang, S.W.; Park, C.W.; Cha, D.R.; Na, K.Y.; Kim, S.G.; Yoon, S.A.; Han, S.Y.; Chang, J.H.; Park, S.K.; et al. A Randomized, Controlled Trial of Oral Intestinal Sorbent AST-120 on Renal Function Deterioration in Patients with Advanced Renal Dysfunction. Clin. J. Am. Soc. Nephrol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Cha, R.H.; Kang, S.W.; Park, C.W.; Cha, D.R.; Na, K.Y.; Kim, S.G.; Yoon, S.A.; Kim, S.; Han, S.Y.; Park, J.H.; et al. Sustained uremic toxin control improves renal and cardiovascular outcomes in patients with advanced renal dysfunction: Post-hoc analysis of the Kremezin Study against renal disease progression in Korea. Kidney Res. Clin. Pract. 2017, 36, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.M.; Sun, H.J.; Wang, F.; Yang, M.; Dong, B.R.; Liu, G.J. Oral adsorbents for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Stinghen, A.E.; Massy, Z.A.; Vlassara, H.; Striker, G.E.; Boullier, A. Uremic Toxicity of Advanced Glycation End Products in CKD. J. Am. Soc. Nephrol. 2016, 27, 354–370. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.J.; Greene, T.; Spitalewiz, S.; Blumenthal, S.; Berl, T.; Hunsicker, L.G.; Pohl, M.A.; Rohde, R.D.; Raz, I.; Yerushalmy, Y.; et al. Pyridorin in type 2 diabetic nephropathy. J. Am. Soc. Nephrol. 2012, 23, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Weickert, M.O.; Qureshi, S.; Kandala, N.B.; Anwar, A.; Waldron, M.; Shafie, A.; Messenger, D.; Fowler, M.; Jenkins, G.; et al. Improved Glycemic Control and Vascular Function in Overweight and Obese Subjects by Glyoxalase 1 Inducer Formulation. Diabetes 2016, 65, 2282–2294. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, N.; Xue, M.; Thornalley, P.J. Methylglyoxal-induced dicarbonyl stress in aging and disease: First steps towards glyoxalase 1-based treatments. Clin. Sci. 2016, 130, 1677–1696. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Rodriguez, E.; Pizarro-Sanchez, S.; Sanz, A.B.; Ramos, A.M.; Sanchez-Nino, M.D.; Martin-Cleary, C.; Fernandez-Fernandez, B.; Ortiz, A. Inflammatory Cytokines as Uremic Toxins: “Ni Son Todos Los Que Estan, Ni Estan Todos Los Que Son”. Toxins 2017, 9, 114. [Google Scholar] [CrossRef] [PubMed]
- Hung, A.M.; Ellis, C.D.; Shintani, A.; Booker, C.; Ikizler, T.A. IL-1beta receptor antagonist reduces inflammation in hemodialysis patients. J. Am. Soc. Nephrol. 2011, 22, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Barnabe, C.; Martin, B.J.; Ghali, W.A. Systematic review and meta-analysis: Anti-tumor necrosis factor alpha therapy and cardiovascular events in rheumatoid arthritis. Arthritis Care Res. 2011, 63, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Lee, C.K.; Cha, H.S.; Choe, J.Y.; Park, E.J.; Kim, J. Effect of anti-tumor necrosis factor alpha treatment of rheumatoid arthritis and chronic kidney disease. Rheumatol. Int. 2015, 35, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Don, B.R.; Kim, K.; Li, J.; Dwyer, T.; Alexander, F.; Kaysen, G.A. The effect of etanercept on suppression of the systemic inflammatory response in chronic hemodialysis patients. Clin. Nephrol. 2010, 73, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Dhaun, N.; MacIntyre, I.M.; Kerr, D.; Melville, V.; Johnston, N.R.; Haughie, S.; Goddard, J.; Webb, D.J. Selective endothelin—A receptor antagonism reduces proteinuria, blood pressure, and arterial stiffness in chronic proteinuric kidney disease. Hypertension 2011, 57, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Dhaun, N.; Melville, V.; Blackwell, S.; Talwar, D.K.; Johnston, N.R.; Goddard, J.; Webb, D.J. Endothelin—A receptor antagonism modifies cardiovascular risk factors in CKD. J. Am. Soc. Nephrol. 2013, 24, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.R.; Littke, T.; Kuranoff, S.; Jurgens, C.; Bruck, H.; Ritz, E.; Philipp, T.; Mitchell, A.; SPP301 (Avosentan) Endothelin Antagonist Evaluation in Diabetic Nephropathy Study Investigators. Avosentan reduces albumin excretion in diabetics with macroalbuminuria. J. Am. Soc. Nephrol. 2009, 20, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Kohan, D.E.; Pollock, D.M. Endothelin antagonists for diabetic and non-diabetic chronic kidney disease. Br. J. Clin. Pharmacol. 2013, 76, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.F.; Green, D.; Jamerson, K.; Ruilope, L.M.; Kuranoff, S.J.; Littke, T.; Viberti, G.; Group, A.S. Avosentan for overt diabetic nephropathy. J. Am. Soc. Nephrol. 2010, 21, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Andress, D.L.; Coll, B.; Pritchett, Y.; Brennan, J.; Molitch, M.; Kohan, D.E. Clinical efficacy of the selective endothelin A receptor antagonist, atrasentan, in patients with diabetes and chronic kidney disease (CKD). Life Sci. 2012, 91, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Kohan, D.E.; Lambers Heerspink, H.J.; Coll, B.; Andress, D.; Brennan, J.J.; Kitzman, D.W.; Correa-Rotter, R.; Makino, H.; Perkovic, V.; Hou, F.F.; et al. Predictors of Atrasentan-Associated Fluid Retention and Change in Albuminuria in Patients with Diabetic Nephropathy. Clin. J. Am. Soc. Nephrol. 2015, 10, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Schievink, B.; de Zeeuw, D.; Smink, P.A.; Andress, D.; Brennan, J.J.; Coll, B.; Correa-Rotter, R.; Hou, F.F.; Kohan, D.; Kitzman, D.W.; et al. Prediction of the effect of atrasentan on renal and heart failure outcomes based on short-term changes in multiple risk markers. Eur. J. Prev. Cardiol. 2016, 23, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Andress, D.L.; Bakris, G.; Brennan, J.J.; Correa-Rotter, R.; Dey, J.; Hou, F.F.; Kitzman, D.W.; Kohan, D.; Makino, H.; et al. Rationale and protocol of the Study Of diabetic Nephropathy with AtRasentan (SONAR) trial: A clinical trial design novel to diabetic nephropathy. Diabetes Obes. Metab. 2018. [Google Scholar] [CrossRef] [PubMed]
- AbbVie Statement on SONAR Study Closure | AbbVie Neswroom. Available online: https://news.abbvie.com/news/media-statements/abbvie-statement-on-sonar-study-closure.htm (accessed on 20 May 2018).
- Jamison, R.L.; Hartigan, P.; Kaufman, J.S.; Goldfarb, D.S.; Warren, S.R.; Guarino, P.D.; Gaziano, J.M.; Veterans Affairs Site Investigators. Effect of homocysteine lowering on mortality and vascular disease in advanced chronic kidney disease and end-stage renal disease: A randomized controlled trial. JAMA 2007, 298, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.F.; Sheridan, P.; McQueen, M.J.; Held, C.; Arnold, J.M.; Fodor, G.; Yusuf, S.; Lonn, E.M.; investigators, H. Homocysteine lowering with folic acid and B vitamins in people with chronic kidney disease—Results of the renal Hope-2 study. Nephrol. Dial. Transplant. 2008, 23, 645–653. [Google Scholar] [CrossRef] [PubMed]
- House, A.A.; Eliasziw, M.; Cattran, D.C.; Churchill, D.N.; Oliver, M.J.; Fine, A.; Dresser, G.K.; Spence, J.D. Effect of B-vitamin therapy on progression of diabetic nephropathy: A randomized controlled trial. JAMA 2010, 303, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Heinz, J.; Kropf, S.; Domrose, U.; Westphal, S.; Borucki, K.; Luley, C.; Neumann, K.H.; Dierkes, J. B vitamins and the risk of total mortality and cardiovascular disease in end-stage renal disease: Results of a randomized controlled trial. Circulation 2010, 121, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Li, J.; Qin, X.; Huang, Y.; Wang, X.; Gottesman, R.F.; Tang, G.; Wang, B.; Chen, D.; He, M.; et al. Efficacy of folic acid therapy in primary prevention of stroke among adults with hypertension in China: The CSPPT randomized clinical trial. JAMA 2015, 313, 1325–1335. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Qin, X.; Li, Y.; Sun, D.; Wang, J.; Liang, M.; Wang, B.; Huo, Y.; Hou, F.F. Efficacy of Folic Acid Therapy on the Progression of Chronic Kidney Disease: The Renal Substudy of the China Stroke Primary Prevention Trial. JAMA Intern. Med. 2016, 176, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Heinz, J.; Kropf, S.; Luley, C.; Dierkes, J. Homocysteine as a risk factor for cardiovascular disease in patients treated by dialysis: A meta-analysis. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2009, 54, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Huo, Y.; Langman, C.B.; Hou, F.; Chen, Y.; Matossian, D.; Xu, X.; Wang, X. Folic acid therapy and cardiovascular disease in ESRD or advanced chronic kidney disease: A meta-analysis. Clin. J. Am. Soc. Nephrol. 2011, 6, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.P.; Stringer, S.J.; Jesky, M.D.; Yadav, P.; Athwal, R.; Dutton, M.; Ferro, C.J.; Cockwell, P. Allopurinol is an independent determinant of improved arterial stiffness in chronic kidney disease: A cross-sectional study. PLoS ONE 2014, 9, e91961. [Google Scholar] [CrossRef] [PubMed]
- Tsuruta, Y.; Nitta, K.; Akizawa, T.; Fukuhara, S.; Saito, A.; Karaboyas, A.; Li, Y.; Port, F.K.; Robinson, B.M.; Pisoni, R.L.; et al. Association between allopurinol and mortality among Japanese hemodialysis patients: Results from the DOPPS. Int. Urol. Nephrol. 2014, 46, 1833–1841. [Google Scholar] [CrossRef] [PubMed]
- Jalal, D.I.; Decker, E.; Perrenoud, L.; Nowak, K.L.; Bispham, N.; Mehta, T.; Smits, G.; You, Z.; Seals, D.; Chonchol, M.; et al. Vascular Function and Uric Acid-Lowering in Stage 3 CKD. J. Am. Soc. Nephrol. 2017, 28, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Borgi, L.; McMullan, C.; Wohlhueter, A.; Curhan, G.C.; Fisher, N.D.; Forman, J.P. Effect of Uric Acid-Lowering Agents on Endothelial Function: A Randomized, Double-Blind, Placebo-Controlled Trial. Hypertension 2017, 69, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Doehner, W.; Schoene, N.; Rauchhaus, M.; Leyva-Leon, F.; Pavitt, D.V.; Reaveley, D.A.; Schuler, G.; Coats, A.J.; Anker, S.D.; Hambrecht, R. Effects of xanthine oxidase inhibition with allopurinol on endothelial function and peripheral blood flow in hyperuricemic patients with chronic heart failure: Results from 2 placebo-controlled studies. Circulation 2002, 105, 2619–2624. [Google Scholar] [CrossRef] [PubMed]
- Feig, D.I.; Soletsky, B.; Johnson, R.J. Effect of allopurinol on blood pressure of adolescents with newly diagnosed essential hypertension: A randomized trial. JAMA 2008, 300, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Gois, P.H.F.; Souza, E.R.M. Pharmacotherapy for hyperuricemia in hypertensive patients. Cochrane Database Syst. Rev. 2017, 4, CD008652. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, M.; de Vinuesa, S.G.; Verdalles, U.; Ruiz-Caro, C.; Ampuero, J.; Rincon, A.; Arroyo, D.; Luno, J. Effect of allopurinol in chronic kidney disease progression and cardiovascular risk. Clin. J. Am. Soc. Nephrol. 2010, 5, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, M.; Garcia de Vinuesa, S.; Verdalles, U.; Verde, E.; Macias, N.; Santos, A.; Perez de Jose, A.; Cedeno, S.; Linares, T.; Luno, J. Allopurinol and progression of CKD and cardiovascular events: Long-term follow-up of a randomized clinical trial. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2015, 65, 543–549. [Google Scholar] [CrossRef] [PubMed]
- McMullan, C.J.; Borgi, L.; Fisher, N.; Curhan, G.; Forman, J. Effect of Uric Acid Lowering on Renin-Angiotensin-System Activation and Ambulatory BP: A Randomized Controlled Trial. Clin. J. Am. Soc. Nephrol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; Patel, S.; Hsu, C.H. Effect of uric acid on plasma levels of 1,25(OH)2D in renal failure. J. Am. Soc. Nephrol. 1993, 4, 1035–1038. [Google Scholar] [PubMed]
- Sampson, A.L.; Singer, R.F.; Walters, G.D. Uric acid lowering therapies for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst. Rev. 2017, 10, CD009460. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Meng, X.; Timofeeva, M.; Tzoulaki, I.; Tsilidis, K.K.; Ioannidis, J.P.; Campbell, H.; Theodoratou, E. Serum uric acid levels and multiple health outcomes: Umbrella review of evidence from observational studies, randomised controlled trials, and Mendelian randomisation studies. BMJ 2017, 357, j2376. [Google Scholar] [CrossRef] [PubMed]
- A Multicenter Clinical Trial of Allopurinol to Prevent Kidney Function Loss in Type 1 Diabetes. Available online: https://clinicaltrials.gov/ct2/show/NCT02017171 (accessed on 20 May 2018).
- White, W.B.; Saag, K.G.; Becker, M.A.; Borer, J.S.; Gorelick, P.B.; Whelton, A.; Hunt, B.; Castillo, M.; Gunawardhana, L.; Investigators, C. Cardiovascular Safety of Febuxostat or Allopurinol in Patients with Gout. N. Engl. J. Med. 2018, 378, 1200–1210. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Nino, M.D.; Zheng-Lin, B.; Valino-Rivas, L.; Sanz, A.B.; Ramos, A.M.; Luno, J.; Goicoechea, M.; Ortiz, A. Lesinurad: What the nephrologist should know. Clin. Kidney J. 2017, 10, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Unni, S.; White, K.; Goodman, M.; Ye, X.; Mavros, P.; Bash, L.D.; Brixner, D. Hypertension control and antihypertensive therapy in patients with chronic kidney disease. Am. J. Hypertens. 2015, 28, 814–822. [Google Scholar] [CrossRef] [PubMed]
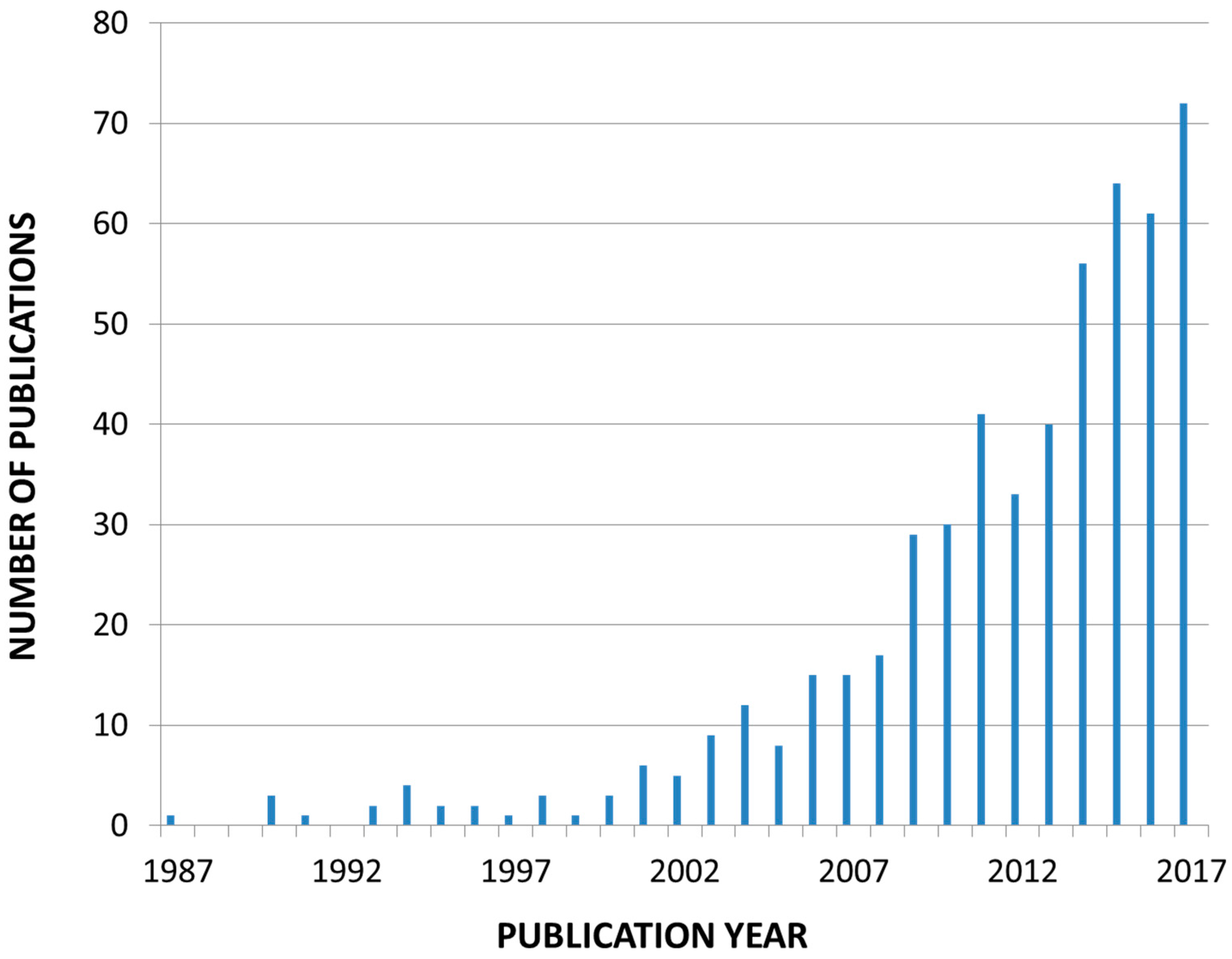
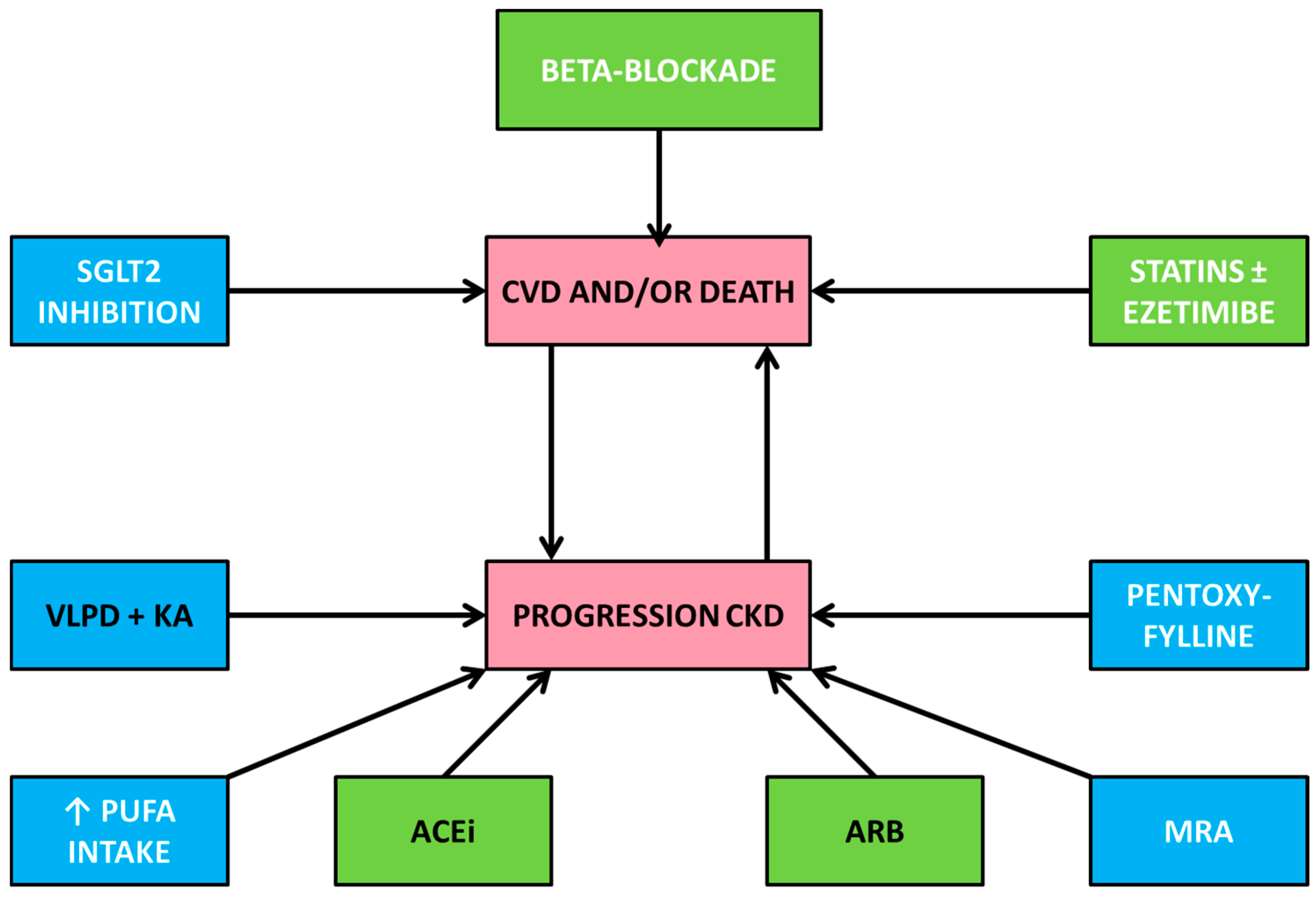
| Stage | GFR a | Definition |
|---|---|---|
| G1 | >90 b | Normal or increased kidney function and urine abnormalities (e.g., hematuria from presumed or proven renal origin or albuminuria c), proven structural kidney abnormalities detected by imaging, genetic trait for kidney disease, electrolyte abnormalities from renal tubular dysfunction, renal histological abnormalities or history of kidney transplantation |
| G2 | 60–89 | Mild reduction in kidney function and urine abnormalities (e.g., hematuria from presumed or proven renal origin or albuminuria c), proven structural kidney abnormalities detected by imaging, genetic trait for kidney disease, electrolyte abnormalities from renal tubular dysfunction, renal histological abnormalities or history of kidney transplantation |
| G3a G3b | 45–59 30–44 | Moderately reduced kidney function |
| G4 | 15–29 | Severely reduced kidney function |
| G5 | <15 | Very severe kidney failure or ESKD, on dialysis or not |
| Cardiovascular Events or Mortality | References | Development of Progression of CKD a | References | Health Economic Impact b | References | |
|---|---|---|---|---|---|---|
| ||||||
| Smoking cessation | + | [28],[29] | + | [21],[22],[23],[24],[25],[29] | + | [30],[31],[32],[33],[34] |
| Exercise | + | [46],[47] | + | [46],[47] | + | [40],[41],[42] |
| Reduction of obesity | + | [63],[64],[65],[66],[67] | + | [53],[54],[55],[33] | ||
| ||||||
| Low protein diet | + | [76],[77],[78],[80],[84],[85],[86],[87] | ||||
| Very low protein diet + KA | ++ | [81],[84],[86] | ||||
| Low Na+ diet | + | [90],[112],[113], [115] c, [116] c,[117] c | ||||
| K+-rich diet d | + | [113],[47] | ||||
| P-restriction | + | [133],[134] | + | [129] e,[130] e,[131],[132] e | ||
| Enhanced PUFA-intake | + | [138],[139],[140] f,[141],[142],[143] | ++ | [144],[145] | ||
| Reduced glucose intake | + | [146],[148] | + | [150] | ||
| Reduced AGE intake | + | [154],[155] | + | [158] | ||
| Correction intestinal dysbiosis | + | [164],[166] |
| CKD 3–5 ND | Hemodialysis | Peritoneal Dialysis | Transplantation First 3 Months | Transplantation > 3 Months | |
|---|---|---|---|---|---|
| Target protein intake (g/kg/d) | 0.6–0.8 (or less + KA) | 1.0–1.2 | 1.0–1.2 | 1.4 | 0.6–0.8 |
| Salt intake (mg/d) * | <6000 | <5000 | <5000 | <6000 | <6000 |
| Potassium intake (mg/d) | 2500 | 2500 | 2500 | Free ** | Free ** |
| Phosphorus intake (mg/d) | <800 | <1000 | <1000 | Free | <800 |
| Cardiovascular Events or Mortality | References | Development of Progression of CKD a | References | Health Economic Impact | References | |
|---|---|---|---|---|---|---|
| ||||||
| Intensive treatment non-diabetic CKD b | + | [172],[173],[175],[177],[183],[184],[185] | + | [172],[173],[175],[177],[182] c,[183],[184] | ||
| Standard treatment ESKD (hemodialysis) d | + | [190],[191] | ||||
| ||||||
| ACEi | + | [201],[202],[203],[211],[212],[213] | +++ | [197],[198],[199],[200],[201],[202],[203],[211],[212],[213] | ++ | [217],[218],[219] |
| ARB | + | [206],[211],[212],[213] | +++ | [121],[198],[204],[205],[206],[207],[206],[211],[212],[213] | + | [220] |
| ACEi + ARB e | + | [215] | + | [211],[214] | ||
| Aldosterone antagonists | + | [215],[233] | ++ f | [226],[227],[228],[229],[230],[231] | ||
| Beta blockers g | +++ | [255],[256],[257],[258],[259] |
| Cardiovascular Events or Mortality | References | Development or Progression of CKD a | References | Health Economic Impact | References | |
|---|---|---|---|---|---|---|
| ||||||
| Intensive glycemia control | + b | [260],[261],[262],[263],[264],[265] | ||||
| SGLT-2 inhibitors | ++ | [274],[276],[281],[282] | + | [274],[275],[276],[277],[278],[279] | ||
| DDP-4 inhibitors | + c | [287] | ||||
| GLP-1RA | + | [291] | + c | [288],[291] | ||
| Metformin | + | [294],[295],[297] | + | [294] | ||
| ||||||
| Phosphate binders (aggregated) d | + | [308],[309],[312],[313],[314] | ||||
| Non-calcium phosphate binders e | + | [323],[324],[325],[326],[328],[329],[314],[331],[332],[330] | + | [334],[335],[336],[337],[338] | ||
| Calcimimetics | + | [341],[343],[345],[346] | ||||
| Parathyroidectomy | + | [347] | ||||
| Calcitriol and vitamin D analogs f | + c | [352],[353],[354],[355] | ||||
| ||||||
| Statins ± ezetemibe (non-dialysis) | +++ | [360],[361],[362],[363],[364],[365] | + c | [361],[362],[366] | + | [378],[379] |
| ||||||
| NFκB inhibition g | + | [389],[390] | ||||
| CCR2 inhibition h | + c | [395],[397] | ||||
| Pentoxifylline i | [398] | ++ | [398],[399],[400] | |||
| ||||||
| Correction of hypomagnesemia | + | [418],[419],[420],[422] | + | [417] | ||
| ||||||
| Acidosis correction (mainly bicarbonate) | + | [427],[428] | ||||
| ||||||
| Antiplatelet therapy | + k | [451],[452],[454] | ||||
| Vitamin K antagonists j | + k | [455],[456],[457] | ||||
| ||||||
| Pirfenidone | + | [476],[477] | ||||
| ||||||
| AST-120 sorbent | + | [494] | + | [485],[486],[487],[488],[490],[491],[492],[493],[494],[495] | + | [489] |
| Cardiovascular Events or Mortality | Development of Progression of CKD a | Health Economic Impact b | |||
|---|---|---|---|---|---|
| Advanced glycation end product reduction | + | [497] | |||
| Cytokine antagonism | + b | [503] | |||
| Endothelin A receptor blockade | + c | [505],[506],[507],[509] | |||
| Homocysteine lowering | + | [515],[516],[517],[518],[521],[522] | + | [520] | |
| Uric acid lowering | + | [524],[530],[531],[532],[534],[535] | + | [530],[531],[534],[535] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vanholder, R.; Van Laecke, S.; Glorieux, G.; Verbeke, F.; Castillo-Rodriguez, E.; Ortiz, A. Deleting Death and Dialysis: Conservative Care of Cardio-Vascular Risk and Kidney Function Loss in Chronic Kidney Disease (CKD). Toxins 2018, 10, 237. https://doi.org/10.3390/toxins10060237
Vanholder R, Van Laecke S, Glorieux G, Verbeke F, Castillo-Rodriguez E, Ortiz A. Deleting Death and Dialysis: Conservative Care of Cardio-Vascular Risk and Kidney Function Loss in Chronic Kidney Disease (CKD). Toxins. 2018; 10(6):237. https://doi.org/10.3390/toxins10060237
Chicago/Turabian StyleVanholder, Raymond, Steven Van Laecke, Griet Glorieux, Francis Verbeke, Esmeralda Castillo-Rodriguez, and Alberto Ortiz. 2018. "Deleting Death and Dialysis: Conservative Care of Cardio-Vascular Risk and Kidney Function Loss in Chronic Kidney Disease (CKD)" Toxins 10, no. 6: 237. https://doi.org/10.3390/toxins10060237
APA StyleVanholder, R., Van Laecke, S., Glorieux, G., Verbeke, F., Castillo-Rodriguez, E., & Ortiz, A. (2018). Deleting Death and Dialysis: Conservative Care of Cardio-Vascular Risk and Kidney Function Loss in Chronic Kidney Disease (CKD). Toxins, 10(6), 237. https://doi.org/10.3390/toxins10060237







