Ergot Alkaloids at Doses Close to EU Regulatory Limits Induce Alterations of the Liver and Intestine
Abstract
1. Introduction
2. Results
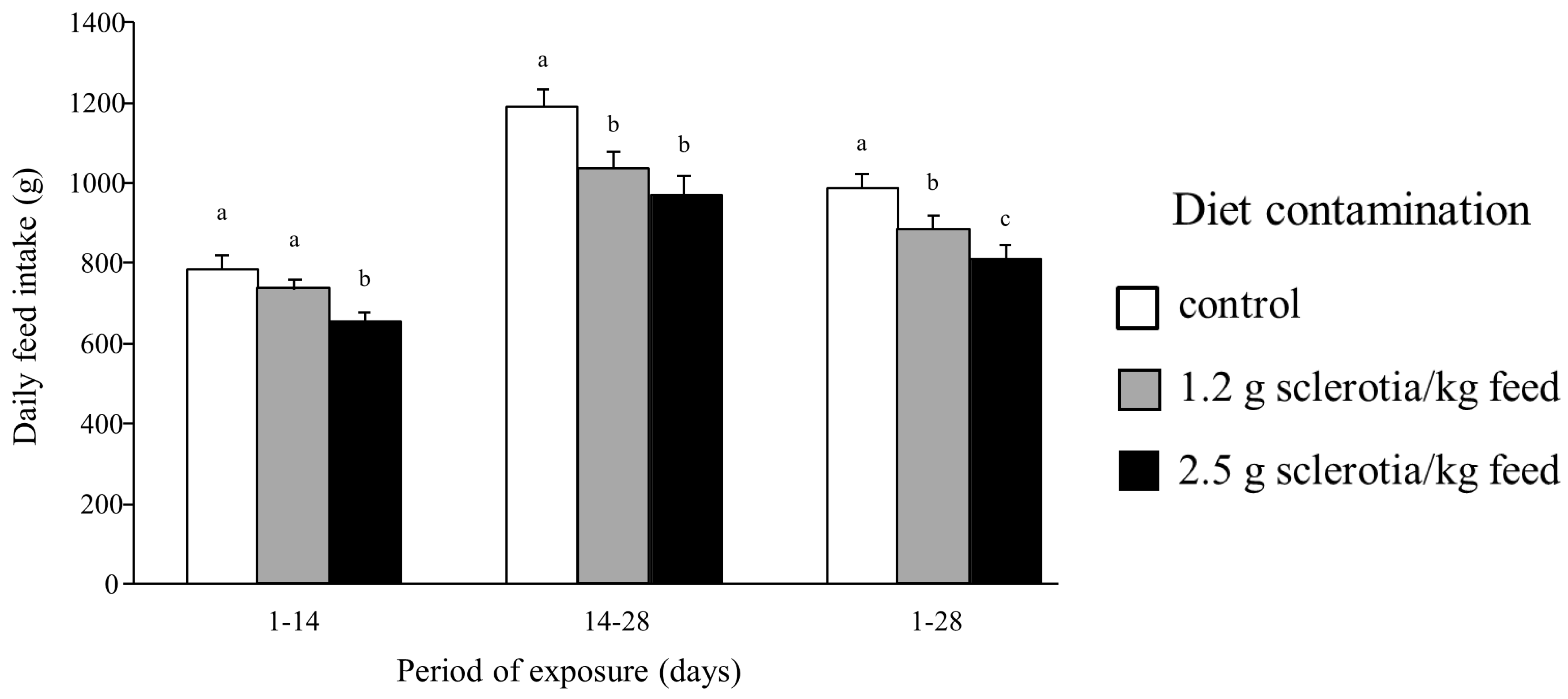
2.1. Effects of Ergot Alkaloids on Clinical Signs, Growth and Feed Intake
2.2. Effects of Ergot Alkaloids on Hematology and Blood Chemistry
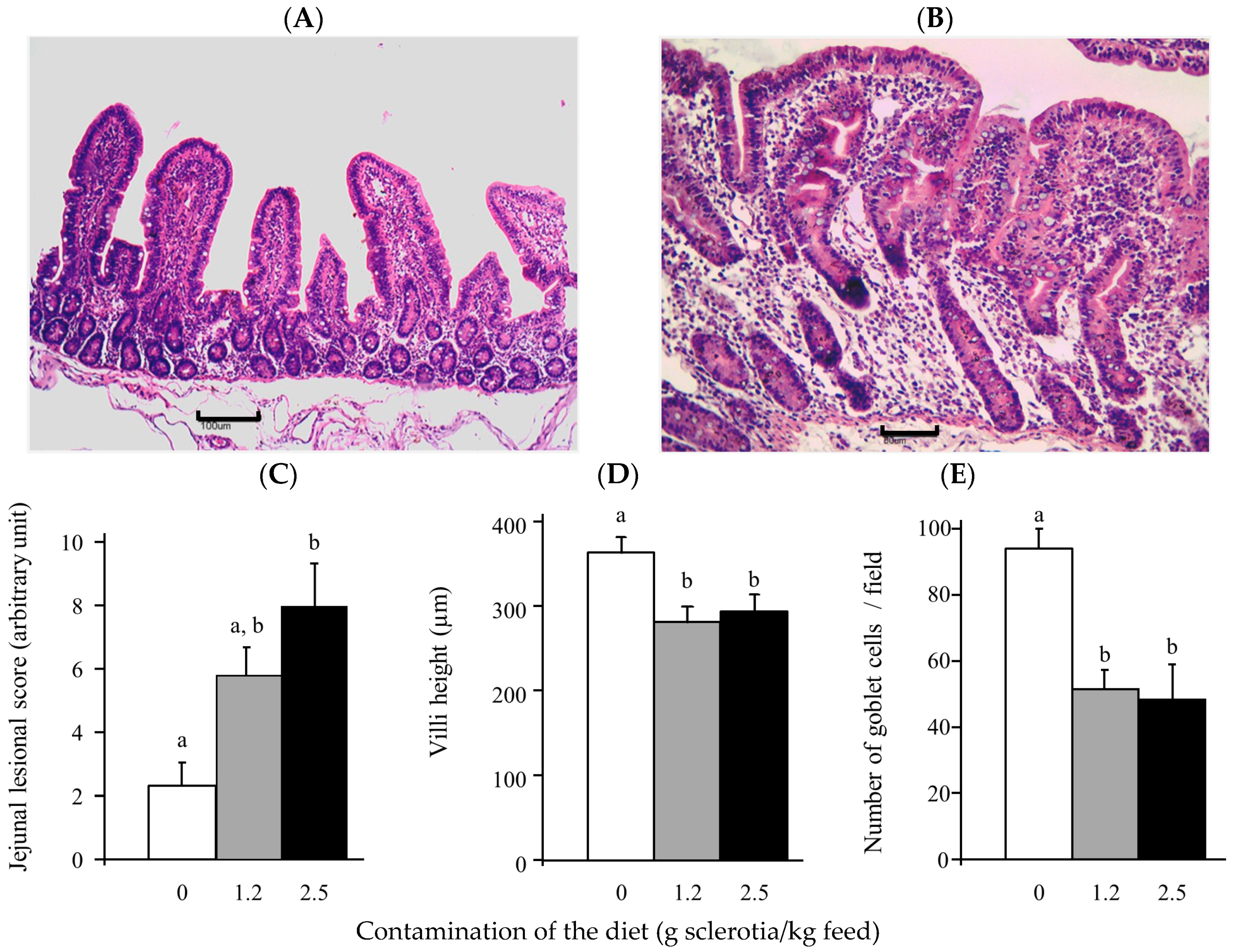
2.3. Alterations of Tissue Morphology Caused by Ergot Alkaloids
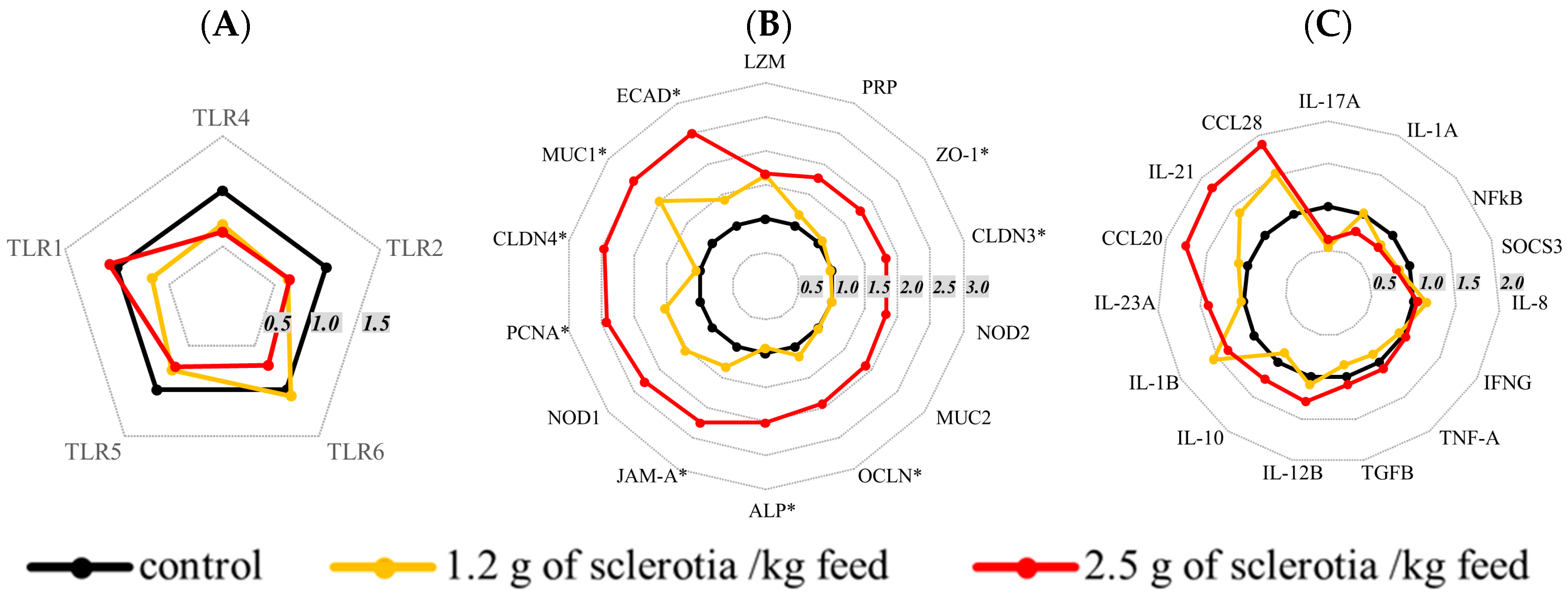
2.4. Effects of Ergot Alkaloids on mRNA Expression in the Jejunum
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Experimental Diets
5.2. Ergot Alkaloid Composition of the Diets
5.3. Animals
5.4. Experimental Setting and Sample Collection
5.5. Hematology and Blood Chemistry
5.6. Histomorphometrical Analysis
5.7. Expression of mRNA by Real-Time PCR
5.8. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- EFSA. Scientific opinion on ergot alkaloids in food and feed. EFSA J. 2012, 10, 2798. [Google Scholar]
- Klotz, J.L. Activities and effects of ergot alkaloids on livestock physiology and production. Toxins 2015, 7, 2801–2821. [Google Scholar] [CrossRef] [PubMed]
- Canty, M.J.; Fogarty, U.; Sheridan, M.K.; Ensley, S.M.; Schrunk, D.E.; More, S.J. Ergot alkaloid intoxication in perennial ryegrass (Lolium perenne): An emerging animal health concern in Ireland? Ir. Vet. J. 2014, 67, 21. [Google Scholar] [CrossRef] [PubMed]
- Korn, A.K.; Gross, M.; Usleber, E.; Thom, N.; Köhler, K.; Erhardt, G. Dietary ergot alkaloids as a possible cause of tail necrosis in rabbits. Mycotoxin Res. 2014, 30, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Strickland, J.R.; Looper, M.L.; Matthews, J.C.; Rosenkrans, C.F., Jr.; Flythe, M.D.; Brown, K.R. Board-invited review: St. Anthony’s Fire in livestock: Causes, mechanisms, and potential solutions. J. Anim. Sci. 2011, 89, 1603–1626. [Google Scholar] [CrossRef] [PubMed]
- Di Mavungu, J.D.; Malysheva, S.V.; Sanders, M.; Larionova, D.; Robbens, J.; Dubruel, P.; Van Peteghem, C.; De Saeger, S. Development and validation of a new LC-MS/MS method for the simultaneous determination of six major ergot alkaloids and their corresponding epimers. Application to some food and feed commodities. Food Chem. 2012, 135, 292–303. [Google Scholar] [CrossRef]
- Scott, P. Ergot alkaloids: Extent of human and animal exposure. World Mycotoxin J. 2009, 2, 141–149. [Google Scholar] [CrossRef]
- Krska, R.; Crews, C. Significance, chemistry and determination of ergot alkaloids: A review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2008, 25, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Tittlemier, S.A.; Drul, D.; Roscoe, M.; McKendry, T. Occurrence of Ergot and Ergot Alkaloids in Western Canadian Wheat and Other Cereals. J. Agric. Food Chem. 2015, 63, 6644–6650. [Google Scholar] [CrossRef] [PubMed]
- Topi, D.; Jakovac-Strajn, B.; Pavsic-Vrtac, K.; Tavcar-Kalcher, G. Occurrence of ergot alkaloids in wheat from Albania. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Orlando, B.; Maumené, C.; Piraux, F. Ergot and ergot alkaloids in French cereals: Occurrence, pattern and agronomic practices for managing the risk. World Mycotoxin J. 2017, 10, 327–338. [Google Scholar] [CrossRef]
- Codex Alimentarius. Codex standard for wheat and durum wheat. Codex Stand. 1995, 199–1995. [Google Scholar]
- European-Union Directive 2002/32/EC. Off. J. Eur. Communities 2002, 10–21.
- Walters, E.M.; Wolf, E.; Whyte, J.J.; Mao, J.; Renner, S.; Nagashima, H.; Kobayashi, E.; Zhao, J.; Wells, K.D.; Critser, J.K.; et al. Completion of the swine genome will simplify the production of swine as a large animal biomedical model. BMC Med. Genomics 2012, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.; Payros, D.; Pinton, P.; Theodorou, V.; Mercier-Bonin, M.; Oswald, I.P.; Théodorou, V.; Mercier-Bonin, M.; Oswald, I.P. Impact of mycotoxins on the intestine: Are mucus and microbiota new targets? Crit. Rev. Toxicol. 2017, 20, 249–275. [Google Scholar] [CrossRef] [PubMed]
- Ingawale, D.K.; Mandlik, S.K.; Naik, S.R. Models of hepatotoxicity and the underlying cellular, biochemical and immunological mechanism(s): A critical discussion. Env. Toxicol Pharmacol 2014, 37, 118–133. [Google Scholar] [CrossRef] [PubMed]
- Danicke, S.; Diers, S. Effects of ergot alkaloids on liver function of piglets as evaluated by the (13)C-methacetin and (13)C-alpha-ketoisocaproic acid breath test. Toxins 2013, 5, 139–161. [Google Scholar] [CrossRef] [PubMed]
- Moubarak, A.; Rosenkrans, C.J.; Johnson, Z.B. Modulation of cytochrome P450 metabolism by ergonovine and dihydroergotamine. Vet. Hum. Toxicol. 2003, 45, 6–9. [Google Scholar] [PubMed]
- Speijers, G.J.A. Subchronic Toxicity Experiment with Rats Fed a Diet Containing Ergotamine-Tartrate; RIVM-report n°618312002; National Institute for Public Health the Environment Protection: Bilthoven, The Netherlands, 1993. [Google Scholar]
- Mainka, S.; Dänicke, S.; Böhme, H.; Ueberschär, K.-H.; Polten, S.; Hüther, L.; Danicke, S.; Bohme, H.; Ueberschar, K.H.; Polten, S.; et al. The influence of ergot-contaminated feed on growth and slaughtering performance, nutrient digestibility and carry over of ergot alkaloids in growing-finishing pigs. Arch. Anim. Nutr. 2005, 59, 377–395. [Google Scholar] [CrossRef] [PubMed]
- Mainka, S.; Danicke, S.; Bohme, H.; Ueberschar, K.H.; Liebert, F.; Dänicke, S.; Böhme, H.; Ueberschär, K.H.; Liebert, F.; Danicke, S.; et al. On the composition of ergot and the effects of feeding two different ergot sources on piglets. Anim. Feed Sci. Technol. 2007, 139, 52–68. [Google Scholar] [CrossRef]
- Spiller, R. Recent advances in understanding the role of serotonin in gastrointestinal motility in functional bowel disorders: Alterations in 5-HT signalling and metabolism in human disease. Neurogastroenterol. Motil. 2007, 19 (Suppl. 2), 25–31. [Google Scholar] [CrossRef] [PubMed]
- Mulac, D.; Lepski, S.; Ebert, F.; Schwerdtle, T.; Humpf, H.U. Cytotoxicity and fluorescence visualization of ergot alkaloids in human cell lines. J. Agric. Food Chem. 2013, 61, 462–471. [Google Scholar] [CrossRef] [PubMed]
- McGuckin, M.A.; Linden, S.K.; Sutton, P.; Florin, T.H.; Lindén, S.K.; Sutton, P.; Florin, T.H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011, 9, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Akbari, P.; Braber, S.; Gremmels, H.; Koelink, P.J.; Verheijden, K.A.; Garssen, J.; Fink-Gremmels, J. Deoxynivalenol: A trigger for intestinal integrity breakdown. FASEB J. 2014, 28, 2414–2429. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Braicu, C.; Nougayrede, J.-P.P.; Laffitte, J.; Taranu, I.; Oswald, I.P.P. Deoxynivalenol Impairs Porcine Intestinal Barrier Function and Decreases the Protein Expression of Claudin-4 through a Mitogen-Activated Protein Kinase-Dependent Mechanism. J. Nutr. 2010, 140, 1956–1962. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Pérez-Cano, F.J.; Castellote, C.; Franch, A.; Castell, M. The bowel: A key component of the immune system. Rev. Esp. Enferm. Dig. 2008, 100, 29–34. [Google Scholar] [PubMed]
- Gourbeyre, P.; Berri, M.; Lippi, Y.; Meurens, F.; Vincent-Naulleau, S.; Laffitte, J.; Rogel-Gaillard, C.; Pinton, P.; Oswald, I.P.P. Pattern recognition receptors in the gut: Analysis of their expression along the intestinal tract and the crypt/villus axis. Physiol. Rep. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kawai, T.; Akira, S. Pathogen recognition by the innate immune system. Int. Rev. Immunol. 2001, 30, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.A.; Moraes, L.E.; Murray, J.D.; Owens, S.D. Hematologic and biochemical reference intervals for specific pathogen free 6-week-old Hampshire-Yorkshire crossbred pigs. J. Anim. Sci. Biotechnol. 2014, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.; Harvey, J.W. Veterinary Laboratory Medicine: Interpretation and Diagnosis, 2nd ed.; W. B. Saunders Co.: Philadelphia, Pennsylvania, 1998; pp. 157–186. [Google Scholar]
- Kerr, M. Veterinary Laboratory Medecine, 2nd ed.; Blackwell Science Ltd.: London, UK, 2002; p. 392. [Google Scholar] [CrossRef]
- Lucioli, J.; Pinton, P.; Callu, P.; Laffitte, J.; Grosjean, F.; Kolf-Clauw, M.; Oswald, I.P.P.; Bracarense, A.P.F.R.L.P. The food contaminant deoxynivalenol activates the mitogen activated protein kinases in the intestine: Interest of ex vivo models as an alternative to in vivo experiments. Toxicon 2013, 66, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Bracarense, A.P.; Schwartz, H.E.; Trumel, C.; Cossalter, A.M.; Schatzmayr, G.; Kolf-Clauw, M.; Moll, W.D.; Oswald, I.P. The low intestinal and hepatic toxicity of hydrolyzed fumonisin B(1) correlates with its inability to alter the metabolism of sphingolipids. Biochem. Pharmacol. 2012, 83, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Gerez, J.R.R.; Pinton, P.; Callu, P.; Grosjean, F.; Oswald, I.P.P.; Bracarense, A.P.F.L.P. Deoxynivalenol alone or in combination with nivalenol andzearalenone induce systemic histological changes in pigs. Exp. Toxicol. Pathol. 2015, 67, 89–98. [Google Scholar] [CrossRef] [PubMed]
- García, G.R.; Payros, D.; Pinton, P.; Dogi, C.A.; Laffitte, J.; Neves, M.; González Pereyra, M.L.; Cavaglieri, L.R.; Oswald, I.P. Intestinal toxicity of deoxynivalenol is limited by Lactobacillus rhamnosus RC007 in pig jejunum explants. Arch. Toxicol. 2018, 92, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Mimoun, S.; Murate, L.S.; Loiseau, N.; Lippi, Y.; Bracarense, A.F.L.; Schatzmayr, G.; He, J.W.; Zhou, T.; Moll, W.D.; et al. Microbial biotransformation of DON: Molecular basis for reduced toxicity. Sci. Rep. 2016, 6, 29105. [Google Scholar] [CrossRef] [PubMed]
- Cano, P.M.; Seeboth, J.; Meurens, F.; Cognie, J.; Abrami, R.; Oswald, I.P.; Guzylack-Piriou, L. Deoxynivalenol as a new factor in the persistence of intestinal inflammatory diseases: An emerging hypothesis through possible modulation of Th17-mediated response. PLoS ONE 2013, 8, e53647. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.; Jensen, J.; Orntoft, T. Normalization of Real Time Quantitative Reverse Transcription—PCR Data: A Model—Based Variance Estimation Approach to Identify Genes Suited for Normalization, Applied to Bladder and Colon Cancer Data Sets. Cancer Res. 2004, 64, 5245. [Google Scholar] [CrossRef] [PubMed]
- Ruijter, J.M.; Ramakers, C.; Hoogaars, W.M.H.; Karlen, Y.; Bakker, O.; van den hoff, M.J.B.; Moorman, A.F.M. Amplification efficiency: Linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res. 2009, 37. [Google Scholar] [CrossRef] [PubMed]
- Alassane-Kpembi, I.; Gerez, J.R.; Cossalter, A.-M.; Neves, M.; Laffitte, J.; Naylies, C.; Lippi, Y.; Kolf-Clauw, M.; Bracarense, A.P.L.; Pinton, P.; et al. Intestinal toxicity of the type B trichothecene mycotoxin fusarenon-X: Whole transcriptome profiling reveals new signaling pathways. Sci. Rep. 2017, 7, 7530. [Google Scholar] [CrossRef] [PubMed]
- Pinton, P.; Graziani, F.; Pujol, A.; Nicoletti, C.; Paris, O.; Ernouf, P.; Di Pasquale, E.; Perrier, J.; Oswald, I.P.P.; Maresca, M.; et al. Deoxynivalenol inhibits the expression by goblet cells of intestinal mucins through a PKR and MAP kinase-dependent repression of the resistin-like molecule beta. Mol. Nutr. Food Res. 2015, 59, 1076–1087. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, K.; Tagami, M.; Takenaga, F.; Yamori, Y.; Itoh, S. Hypoxia-induced changes in tight junction permeability of brain capillary endothelial cells are associated with IL-1beta and nitric oxide. Neurobiol. Dis. 2004, 17, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.; Horgan, G.; Dempfle, L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002, 30, e36. [Google Scholar] [CrossRef] [PubMed]

| Parameters | Contamination of the Diet (g of Sclerotia/kg Feed) | ||
|---|---|---|---|
| 0 | 1.2 | 2.5 | |
| Red blood cells (T/L) | 7.75 ± 0.12 | 7.80 ± 0.13 | 8.08 ± 0.13 |
| Hemoglobin (g/dL) | 10.45 ± 0.22 a,b | 10.09 ± 0.21 a | 10.97 ± 0.19 b |
| Hematocrit (%) | 36.6 ± 0.70 a,b | 35.52 ± 0.87 a | 38.31 ± 0.63 b |
| Mean corpuscular volume (fL) | 47.46 ± 0.93 | 45.67 ± 0.93 | 47.58 ± 0.91 |
| White blood cells (103/mm3) | 14.5 ± 0.6 | 14.2 ± 0.9 | 16.5 ± 0.9 |
| Neutrophils (%) | 40.92 ± 2.51 a | 30.67± 1.84 b | 36.17 ± 2.55 a,b |
| Eosinophils (%) | 0.38 ± 0.15 | 0.54 ± 0.30 | 0.29 ± 0.14 |
| Basophils (%) | 0.08 ± 0.08 | 0.08 ± 0.08 | 0.00 ± 0.00 |
| Lymphocytes (%) | 53.96 ± 2.66 a | 63.96 ± 1.95 b | 58.79 ± 2.77 a,b |
| Monocytes (%) | 4.67 ± 0.42 | 4.75 ± 0.68 | 4.75 ± 0.63 |
| Platelets/mm3 | 521.58 ± 32.67 | 465.00 ± 28.08 | 533.04 ± 32.39 |
| Parameters | Contamination of the Diet (g of Sclerotia/kg Feed) | ||
|---|---|---|---|
| 0 | 1.2 | 2.5 | |
| Alkaline phosphatase (U/L) | 275.7 ± 24.5 | 237.6 ± 16.7 | 228.6 ± 16.1 |
| Alanine aminotransferase (U/L) | 37.3 ± 1.9 | 31.74 ± 1.5 | 33.32 ± 1.6 |
| Amylase (U/L) | 1783 ± 123 | 1815 ± 99 | 2074 ± 122 |
| Aspartate aminotransferase (U/L) | 56.0 ± 6.8 | 42.6 ± 5.4 | 44.4 ± 3.94 |
| Creatine kinase (U/L) | 3377 ± 396 a | 1924 ± 297 b | 1300 ± 252 b |
| Lactate dehydrogenase (U/L) | 962.8 ± 50.5 | 968.3 ± 57.7 | 1013.3 ± 72.5 |
| Lipase (U/L) | 6.45 ± 0.5 | 6.9 ± 0.3 | 5.9 ± 0.8 |
| Albumin (μmol/L) | 533.0 ± 9.7 | 530.4 ± 11.7 | 522.4 ± 13.3 |
| T bilirubine (μmol/L) | 11.2 ± 1.2 | 11.0 ± 1.2 | 7.4 ± 1.3 |
| Cholesterol (mmol/L) | 3.0 ± 0.1 a | 2.6 ± 0.1 b | 2.8 ± 0.1 a |
| Creatinine (μmol/L) | 61.9 ± 6.2 | 63.3 ± 6.5 | 71.7 ± 6.4 |
| Glucose PAP (mmol/L) | 4.4 ± 0.2 a | 4.7 ± 0.3 a | 5.4 ± 0.3 b |
| Phosphorus (mmol/L) | 3.1 ± 0.1 | 3.2 ± 0.1 | 3.0 ± 0.1 |
| Total proteins (g/L) | 63.5 ± 7.8 | 59.1 ± 7.4 | 62.5 ± 8.2 |
| Urea (mmol/L) | 4.7 ± 0.4 | 4.3 ± 0.3 | 3.5 ± 0.4 |
| Parameters | Contamination of the Diet (g of Sclerotia/kg Feed) | ||
|---|---|---|---|
| 0 | 1.2 | 2.5 | |
| Total nitrogen | 20.1 | 20.2 | 20.3 |
| Raw cellulose | 2.6 | 2.7 | 2.8 |
| Lipids | 4.3 | 4.4 | 4.4 |
| Minerals | 6.0 | 6.1 | 6.0 |
| Alkaloids (mg/kg Feed) | Contamination of the Diet (g of Sclerotia/kg Feed) | |
|---|---|---|
| 1.2 | 2.5 | |
| Ergotamine | 0.52 | 1.03 |
| Ergotaminine | 0.24 | 0.58 |
| Ergosine | 0.29 | 0.58 |
| Ergosinine | 0.16 | 0.34 |
| Ergocristine | 0.26 | 0.47 |
| Ergocristinine | 0.18 | 0.40 |
| Ergometrine | 0.17 | 0.44 |
| Ergometrinine | 0.06 | 0.12 |
| Ergocornine | 0.15 | 0.30 |
| Ergocorninine | 0.11 | 0.29 |
| Ergocryptine | 0.13 | 0.30 |
| Ergocryptinine | 0.09 | 0.21 |
| Total alkaloids | 2.4 | 5.1 |
| Target Gene | Primer Sequence (5′–3′) | mRNA | Reference | |
|---|---|---|---|---|
| Alkaline phosphatase (ALP) | F | AAGCTCCGTTTTTGGCCTG | ENSSSCT00000037252.1 | [28] |
| R | GGAGGTATATGGCTTGAGATCCA | |||
| Beta-2-microglobulin (B2M) | F | TTCTACCTTCTGGTCCACACTGA | NM_213978.1 | [41] |
| R | TCATCCAACCCAGATGCA | |||
| C-C Motif Chemokine Ligand 20 (CCL20) | F | GCTCCTGGCTGCTTTGATGTC | NM_001024589 | Present study |
| R | CATTGGCGAGCTGCTGTGTG | |||
| C-C Motif Chemokine Ligand 28 (CCL28) | F | GGCTGCTGTCATCCTTCATGT | ENSSSCT00000018375 | Present study |
| R | TGAGGGCTGACACAGATTCTTCT | |||
| Claudin 3 (CLDN3) | F | CTGCTCTGCTGCTCGTGCCC | AY625258.1 | Present study |
| R | TCATACGTAGTCCTTGCGGTCGTAG | |||
| Claudin 4 (CLDN4) | F | CTGCTTTGCTGCAACTGCC | NM_001161637.1 | [26] |
| R | TCAACGGTAGCACCTTACACGTAGT | |||
| E-cadherin (ECAD) | F | ACCACCGCCATCAGGACTC | NM_001163060.1 | Present study |
| R | TGGGAGCTGGGAAACGTG | |||
| Interferon gamma (IFNG) | F | TGGTAGCTCTGGGAAACTGAATG | NM_213948 | [28] |
| R | GGCTTTGCGCTGGATCTG | |||
| Interleukin 1A (IL-1A) | F | TCAGCCGCCCATCCA | NM_214029.1 | [38] |
| R | AGCCCCCGGTGCCATGT | |||
| Interleukin 1B (IL-1B) | F | ATGCTGAAGGCTCTCCACCTC | NM_214055 | [28] |
| R | TTGTTGCTATCATCTCCTTGCAC | |||
| Interleukin 8 (IL-8) | F | GCTCTCTGTGAGGCTGCAGTTC | NM_213867.1 | [38] |
| R | AAGGTGTGGAATGCGTATTTATGC | |||
| Interleukin 10 (IL-10) | F | GGCCCAGTGAAGAGTTTCTTTC | NM_214041 | [38] |
| R | CAACAAGTCGCCCATCTGGT | |||
| Interleukin 12B (IL-12B) | F | GGTTTCAGACCCGACGAACTCT | NM_214013.1 | [38] |
| R | CATATGGCCACAATGGGAGATG | |||
| Interleukin 17A (IL-17A) | F | CCAGACGGCCCTCAGATTAC | NM_001005729.1 | [38] |
| R | GGTCCTCGTTGCGTTGGA | |||
| Interleukin 21 (IL-21) | F | GGCACAGTGGCCCATAAATC | NM_214415 | Present study |
| R | GCAGCAATTCAGGGTCCAAG | |||
| Interleukin 23A (IL-23A) | F | TTCTCTACACCCTGATGGCTCTG | ENSSSCT00000047550.1 | Present study |
| R | TCGGGCTGCAAGAGTTGC | |||
| Junctional adhesion molecule A (JAM-A) | F | CGTGCCTTCATCAACTCTTCCTAT | NM_001128444.1 | Present study |
| R | CACAAGTGTAATCTCCAGCATCAGA | |||
| Lysozyme (LZM) | F | GGTCTATGATCGGTGCGAGTTC | NM_214392.2 | [28] |
| R | TCCATGCCAGACTTTTTCAGAAT | |||
| Mucin 1 (MUC1) | F | GCATTACAAACCTCCAGTTTACCT | AY243508.1 | [42] |
| R | CCCAGAAGCCCGTCTTCTTT | |||
| Mucin 2 (MUC2) | F | GCAGCCTGTGCGAGGAA | XM_003122394.1 | [42] |
| R | TGTCATCATACACAGTGCCTTCTG | |||
| Occludin (OCLN) | F | AGCTGGAGGAAGACTGGATCAG | U79554.1 | [43] |
| R | TGCAGGCCACTGTCAAAATT | |||
| Nuclear Factor Kappa B (NFkB) | F | CCTCCACAAGGCAGCAAATAG | ENSSSCT00000033438 | Present study |
| R | TCCACACCGCTGTCACAGA | |||
| Nuclear oligomerization domain 1 (NOD1) | F | TGGGCTGCGTCCTGTTCA | AB_187219.1 | [28] |
| R | GGTGACCCTGACCGATGT | |||
| Nuclear oligomerization domain 2 (NOD2) | F | GAGCGCATCCTCTTAACTTTC | AB426547.1 | [28] |
| R | ACGCTCGTGATCCGTGAAC | |||
| Proliferating cell nuclear antigen (PCNA) | F | GTTGATAAAGAGGAGGAAGCAGTT | NM_001291925.1 | [28] |
| R | TGGCTTTTGTAAAGAAGTTCAGGTAC | |||
| Peptidylprolyl isomerase A (cyclophilin A) | F | CCCACCGTCTTCTTCGACAT | NM_214353.1 | [38] |
| R | TCTGCTGTCTTTGGAACTTTGTCT | |||
| Prion protein (PRP) | F | TTTGTGCATGACTGCGTCAAC | NM_001008687.1 | Present study |
| R | CGTGGTCACTGTGTGCTGCT | |||
| Ribosomal protein L32 (RPL32) | F | AGTTCATCCGGCACCAGTCA | NM_001001636.1 | [26] |
| R | GAACCTTCTCCGCACCCTGT | |||
| Suppressor of Cytokine Signaling 3 (SOCS3) | F | CTTCACGCTCAGCGTCAAG | HM045422.1 | Present study |
| R | CTTGAGCACGCAGTCGAAG | |||
| Transforming growth factor beta (TGFB) | F | GAAGCGCATCGAGGCCATTC | NM_214015 | [28] |
| R | GGCTCCGGTTCGACACTTTC | |||
| Toll-like receptor 1 (TLR1) | F | TGCTGGATGCTAACGGATGTC | AB219564.1 | [28] |
| R | AAGTGGTTTCAATGTTGTTCAAAGTC | |||
| Toll-like receptor 2 (TLR2) | F | TCACTTGTCTAACTTATCATCCTCTTG | AB085935.1 | [28] |
| R | TCAGCGAAGGTGTCATTATTGC | |||
| Toll-like receptor 4 (TLR4) | F | GCCATCGCTGCTAACATCATC | AB188301.2 | [28] |
| R | CTCATACTCAAAGATACACCATCGG | |||
| Toll-like receptor 5 (TLR5) | F | CCTTCCTGCTTCTTTGATGG | NM_001348771 | [28] |
| R | CTGTGACCGTCCTGATGTAG | |||
| Toll-like receptor 6 (TLR6) | F | AACCTACTGTCATAAGCCTTCATTC | AB085936.1 | [28] |
| R | GTCTACCACAAATTCACTTTCTTCAG | |||
| Tumor Necrosis Factor alpha (TNF-A) | F | ACTGCACTTCGAGGTTATCGG | NM_214022 | [28] |
| R | GGCGACGGGCTTATCTGA | |||
| Zonula occludens 1 (ZO-1) | F | ATAACATCAGCACAGTGCCTAAAGC | AJ318101.1 | Present study |
| R | GTTGCTGTTAAACACGCCTCG |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maruo, V.M.; Bracarense, A.P.; Metayer, J.-P.; Vilarino, M.; Oswald, I.P.; Pinton, P. Ergot Alkaloids at Doses Close to EU Regulatory Limits Induce Alterations of the Liver and Intestine. Toxins 2018, 10, 183. https://doi.org/10.3390/toxins10050183
Maruo VM, Bracarense AP, Metayer J-P, Vilarino M, Oswald IP, Pinton P. Ergot Alkaloids at Doses Close to EU Regulatory Limits Induce Alterations of the Liver and Intestine. Toxins. 2018; 10(5):183. https://doi.org/10.3390/toxins10050183
Chicago/Turabian StyleMaruo, Viviane Mayumi, Ana Paula Bracarense, Jean-Paul Metayer, Maria Vilarino, Isabelle P. Oswald, and Philippe Pinton. 2018. "Ergot Alkaloids at Doses Close to EU Regulatory Limits Induce Alterations of the Liver and Intestine" Toxins 10, no. 5: 183. https://doi.org/10.3390/toxins10050183
APA StyleMaruo, V. M., Bracarense, A. P., Metayer, J.-P., Vilarino, M., Oswald, I. P., & Pinton, P. (2018). Ergot Alkaloids at Doses Close to EU Regulatory Limits Induce Alterations of the Liver and Intestine. Toxins, 10(5), 183. https://doi.org/10.3390/toxins10050183






