1. Introduction
Folate refers to a diverse set of water-soluble vitamers with different oxidation states, one-carbon substitutions, and numbers of conjugated glutamates [
1,
2] (
Table S1). Dietary sources of folate include leafy green vegetables, legumes, and fortified grain products. Folate bioavailability depends not only on dietary intake but also on cellular mechanisms mediating absorption and metabolism, as well as genetic polymorphisms of key enzymes or carriers in the folate pathway [
3]. For example, folate hydrolase (
FOLH1, also known as glutamate carboxypeptidase II, GCP-II) is an enzyme located on the intestinal brush border that must cleave the polyglutamated tail from naturally occurring food folate before it can be absorbed [
4,
5].
Folate is critical for a variety of cellular pathways [
2], including the production of nucleotides and neurotransmitters [
1], histone methylation and thus regulation of gene expression [
6], and the methylation of homocysteine to methionine [
7]. Given that folate requirements are higher during times of increased cell division to accommodate key cellular processes, it follows that folate is vital during pregnancy [
8]. Perhaps the most well studied benefit of folate is its reduction of risk for neural tube defects (NTDs), which can occur in developing embryos before the neural tube closes between 23 and 27 days after conception [
2,
7]. Furthermore, reduced periconceptional folic acid intake has also been associated with higher risk for other conditions, such as severe language delay and autism [
9,
10].
Following evidence that supplemental folic acid reduces the risk of NTD-affected pregnancy [
11,
12], in 1992 the U.S. Public Health Service advised all women of child-bearing age to consume at least 400 mcg of folic acid each day [
13]. Subsequently, the U.S. Food and Drug Administration mandated the fortification of enriched grain products with 140 mcg of folic acid per 100 g of cereal product beginning in January of 1998. Similar interventions have since been widely implemented in other Western hemisphere countries [
14]. Fortified grain products and supplements (vitamins) contain synthetic folic acid, which has a higher bioavailability than naturally occurring food folate since it does not contain a polyglutamated tail [
4]. To accommodate for the fact that synthetic folic acid is 1.7 times more bioavailable than naturally occurring food folate [
15,
16], folate levels are expressed as dietary folate equivalents (DFE), where 1 mcg of food folate equates to 1 mcg DFE, but 1 mcg of synthetic folic acid is 1.7 mcg DFE [
17].
While the FDA predicted that mandatory grain fortification would increase folic acid consumption by about 100 mcg DFE per day [
18,
19], actual folic acid intake from fortified foods among non-supplement users increased by nearly 200 mcg DFE per day within a year after fortification was implemented [
20]. Consequently the percentage of individuals with folate intake below the Estimated Average Requirement (EAR; defined as the daily intake of a nutrient adequate to meet the requirement of 50 percent of the population) of 320 mcg DFE/day decreased from 49 percent to 7 percent [
20].
Blood folate levels can readily be assessed using serum or red blood cell (RBC) folate concentrations. Serum levels are responsive to immediate folate intake, whereas the RBC concentration reflects long-term folate status and is used by the Institute of Medicine as the primary indicator of folate adequacy [
17]. Serum and RBC concentrations were, respectively, 2.5 and 1.5 times higher post-fortification (1999–2010) than pre-fortification (1988–1994), and the rates of folate deficiency (<10 nmol/L and <340 nmol/L for serum and RBC folate, respectively) fell to below 1 percent [
21]. Furthermore, men and women have been found to differ in terms of intake and subsequent blood folate concentrations. While men have generally been shown to have higher dietary and total folate intakes than women [
22,
23,
24,
25], several studies have shown that baseline serum and RBC folate levels are the same or higher in women [
19,
21,
23,
24,
25]. Additionally, two randomized placebo-controlled trials showed that women had larger increases in RBC folate concentrations than men when given the same doses of supplemental folic acid, suggesting men need to consume more folic acid than women to achieve the same RBC folate concentration [
23,
24,
25]. In addition to sex, several single nucleotide polymorphisms (SNPs) in genes related to folate metabolism have been associated with variance in serum and RBC folate concentrations. For example, the 484T>C variant (rs202676) of
FOLH1, a common missense polymorphism located within exon 2 of the gene, has been associated with lower RBC folate levels despite equivalent folate intake across genotypic groups [
26].
Although the rollout of folic acid fortification was initially associated with both increased blood folate levels and a reduction of between 19 and 50 percent in NTDs [
27,
28], subsequent research has raised concerns about the level of ongoing benefit. Specifically, data from the National Health and Nutrition Examination Survey (NHANES) indicate that median serum and RBC folate concentrations among non-pregnant women of childbearing age decreased by 16 and 8 percent, respectively, between 1999–2000 and 2003–2004 [
29]. Similar decreases (<10%) were observed between 1999–2002 and 2003–2006 [
30]. Most recently, both serum and RBC folate concentrations decreased by 17 and 12 percent, respectively, between their initial peaks in 1999–2000 and 2009–2010 [
21]. Such trends are reflected by changes in prevalence estimates of elevated plasma homocysteine levels, which significantly increased from 4.8% immediately following fortification (1999–2000) to 8.6% later in the post-fortification era (2007–2010); critically, over this same period, prevalence estimates of folate insufficiency based on elevated risk for NTD-affected pregnancy increased significantly from 15 to 23% [
31,
32]. While the prevalence of clinical folate deficiency—based on hematologic indicators of megaloblastic anemia—which is considered the final stage of folate deficiency [
33] remained <1% during this period, the marked increase in folate insufficiency among women of reproductive age is of concern, as maternal folate levels necessary to prevent NTDs may be substantially higher than the cutoff for clinical folate deficiency.
Though the cause for this decrease in RBC folate levels remains unclear, some have suggested this drop reflects a reduction in the amount of folic acid added to fortified grain products [
34]. Specifically, fortified grain products collected and analyzed immediately following fortification contained significantly greater total folic acid than mandated by federal regulations [
35]. Despite this initial overage, subsequent studies suggest the folic acid content in fortified bread decreased between 2001 and 2003 [
36], while other fortified foods may have folic acid content below federal regulations [
37]. Though the use of different assays prevents direct comparison of these findings, it is possible that a decrease in RBC folate levels over time reflects underlying changes in the quantity of folic acid added to foods through fortification.
It is currently unclear whether the population is consuming sufficient folate to meet the EAR of 320 mcg DFE, 330 mcg (DFE) for those under 19 [
17]. One particularly concerning finding is that, even when all sources of folate intake were considered, a sizeable percentage of women of child-bearing age were below the EAR, with 19 percent of 14–18 year olds below 330 mcg (DFE), with 17 percent of 19–30 year olds, and 15 percent of 31–50 year olds below 320 mcg (DFE) per day [
22]. Furthermore, a recent review of 2007–2010 NHANES data found fortified grain products were the sole source of folic acid for 48.4% of women of childbearing age, 32% of whom were considered to be at elevated or high risk for NTD-affected pregnancy [
32]. Given the variable contributions of different dietary sources of folate and folic acid to RBC folate, it is important to monitor changes in the sources of folate intake (naturally occurring food folate, folic acid from fortified foods, or supplemental folic acid) to ensure population dietary needs are met. Such monitoring is especially important given recent evidence showing that without folic acid only 12% of the population had usual intake above the EAR [
18]. Additionally, as the relationship between dietary folate forms and blood folate status remains incompletely characterized, it is important to clarify the contributions made by different folate forms to RBC folate levels and to assess the effects of other factors—including common genetic variants in the folate pathway—that may influence this relationship.
As recent evidence suggests that population blood folate levels may be declining over time, it is increasingly important to identify factors that may interact with folate intake to affect the risk of NTD-affected births or subsequent risk for other neurodevelopmental disorders [
9,
10]. Though larger cohorts have previously examined the interplay of natural and synthetic folate forms on blood folate level, to our knowledge this is the first study to examine the combined influence of the
FOLH1 484T>C polymorphism and dietary folate forms on RBC folate status. We predicted that blood folate level would be more strongly dependent on synthetic folic acid intake (both folic acid from fortified foods and supplemental folic acid) than on naturally occurring food folate intake, given that genetic variants in
FOLH1 across the population might stratify the efficiency of naturally occurring food folate absorption. We also specifically examined the interaction between the 484T>C variant in
FOLH1 and naturally occurring food folate intake. As
FOLH1 is critical for cleaving the polyglutamate tails from naturally occurring food folate we predicted there would be a stronger positive relationship between naturally occurring food folate intake and RBC folate levels for homozygous T-allele individuals than for those carrying the hypofunctional C-allele.
4. Discussion
Understanding why circulating folate levels vary from person to person is critical to ensuring adequate bioavailability, especially among women of childbearing age. This cross-sectional study of folate intake patterns in healthy young adults demonstrates the importance of separately considering the contributions of naturally occurring food folate and synthetic folic acid intake to circulating folate levels, especially when viewed in concert with sex and genetic variation in folate absorption.
On average, study participants reported receiving approximately equal amounts naturally occurring food folate (332 mcg) and folic acid from fortified foods (340 mcg DFE). Furthermore, 60.8% participants did not take supplemental folic acid (56.5% of all women, 66.1% of all men), while the 39.2% who did reported an average supplemental folic acid intake of 322 mcg DFE. Synthetic folic acid was critical to maintaining recommended folate intake, as 58.7% of participants (63.3% of female participants) would have been below the EAR of 320 mcg (330 for those under 19) based solely on naturally occurring food folate intake. This supports other recent evidence of the continued importance of fortification, with reports that the percentage of the population below the EAR fell from 88% with naturally occurring food folate alone to 11% with the addition of fortified folic acid [
18]. In the present study, although fortification helped a large proportion of participants to attain sufficient folate intake, 10.2% of all individuals (13.6% of female participants) remained under the EAR even when considering folate intake from all sources. However, the percentage of participants above and below the EAR should be interpreted as approximates [
48]. Thus, the present results are best understood as providing support for previous research that reiterated the importance of folic acid fortification to ensuring adequate folate levels for NTD prevention [
22].
Consistent with previous reports [
22,
23,
24,
25], male and female participants differed substantially in folate intake, but did not differ in RBC folate levels. Men had significantly greater folate intake than women from all sources except supplements, for which intake did not differ by sex. Additionally, the frequency of inadequate folate intake did not differ significantly between women and men when considering only naturally occurring food folate intake. However, the proportion of women under the EAR was significantly greater when accounting for folic acid intake from fortified foods, and approached significance when considering intake of folate from all sources. It remains uncertain why women demonstrated equivalent RBC folate levels to men despite lower folate intake, although other authors have postulated that this pattern reflects increased average volume of distribution in men or potential differences in lean body mass [
25].
In the second part of our analysis, we examined the relative contributions of naturally occurring food folate, synthetic folate, and a common genetic variant in
FOLH1 to circulating folate levels. Compared to serum folate, RBC folate is thought to provide a better estimate of folate levels within body tissues [
49], where it plays a critical role in methylation reactions that regulate gene expression [
6], homocysteine metabolism [
50], neurotransmitter synthesis and turnover [
1], and a host of other essential processes. However, relative to serum folate, RBC folate measurement may be considerably more vulnerable to small variations in sample storage and analysis, which may make it a less reliable measure of folate status [
46,
51].
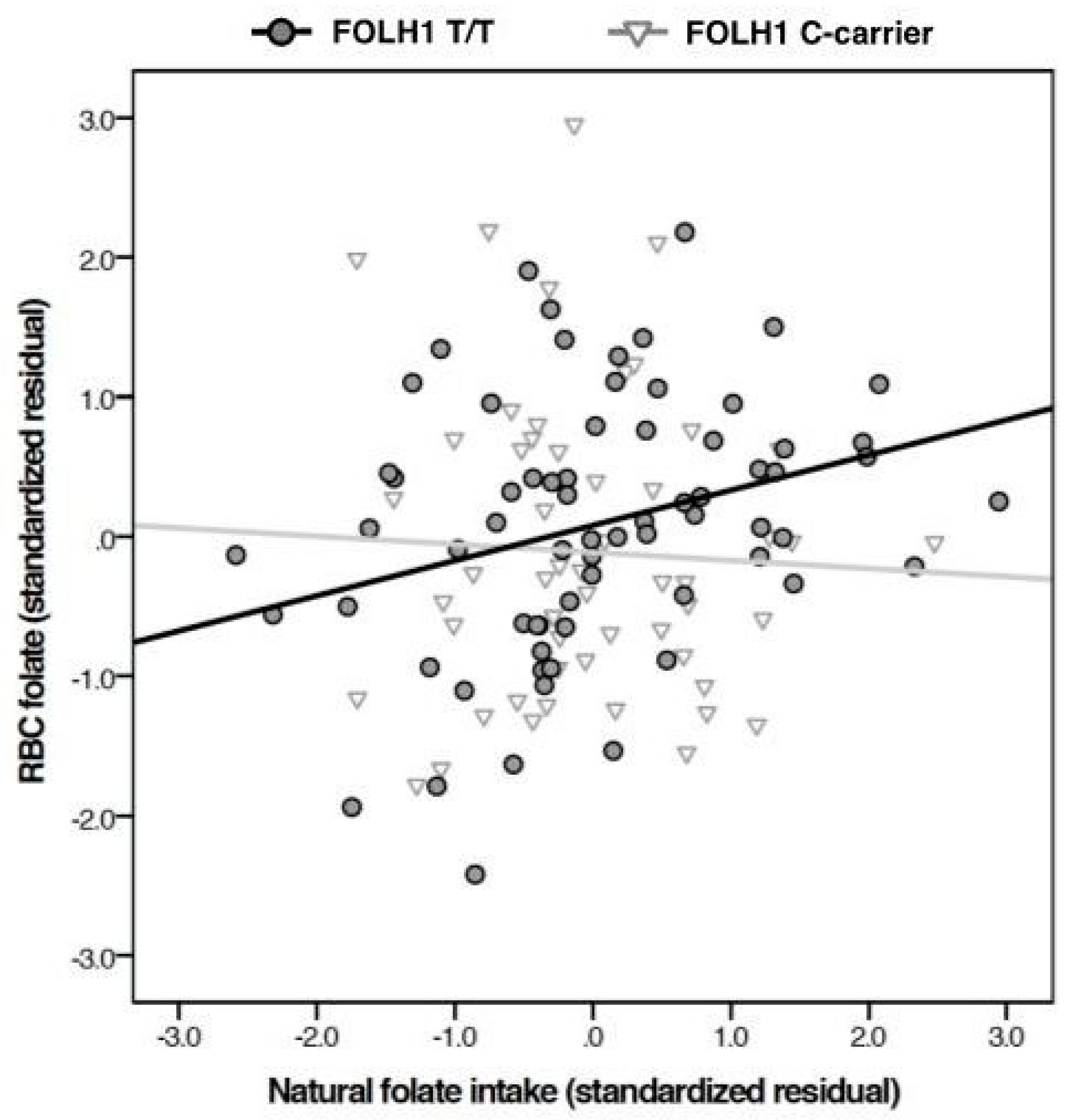
As seen in
Table 4, total synthetic folic acid intake is a stronger predictor of RBC folate level than is intake of naturally occurring food folate. Given that greater RBC folate levels reduce the risk of NTD-affected births [
31,
32], it is possible that greater intake of synthetic folic acid forms afford greater protection against NTDs (and potentially lower the risk for certain conditions such as autism and severe language delay [
9,
10]). Furthermore this difference between synthetic folic acid and naturally occurring food folate forms likely reflects the fact that, unlike synthetic folic acid, naturally occurring food folate must undergo hydrolysis of a polyglutamated tail moiety by
FOLH1, a glutamate carboxypeptidase that lies within the intestinal brush border, before absorption. The 484T>C variant codes for a missense mutation (75Tyr>His) in the
FOLH1 enzyme. The functionality of this variant has not been established using biochemical assays, although structural modeling suggests that the C allele reduces folate binding [
52]. Further, the C allele has previously been associated with reduced blood folate levels [
26], suggesting that it is a hypofunctional variant that limits naturally occurring food folate absorption. As expected,
FOLH1 variants did not influence absorption of synthetic folic acid as such forms lack a polyglutamate tail that must be enzymatically cleaved by
FOLH1 prior to absorption.
In the present sample, which is smaller than those previously examined, the 484T>C genotype by itself did not contribute to RBC folate level. However, in the largest racial group, the interactive effect of genotype with naturally occurring food folate intake on RBC folate levels occurred in the anticipated direction. Specifically, the correlation between naturally occurring food folate and RBC folate was stronger among individuals homozygous for the T allele. This pattern suggests that polyglutamated folate (naturally occurring food folate) is less well absorbed among C allele carriers. This interpretation is consistent with results from previous research, which found those with hypofunctional
FOLH1 484 variants had lower RBC folate levels despite equivalent dietary folate intake [
26].
We repeated our analysis in the largest racial group so as to control for possible population stratification effects [
47], a type of confounding wherein allele frequency varies across subgroups within a population in a manner that might confound the relationship between a genetic variant and a relevant phenotype. Pertinent to this study, the frequency of folate-related alleles has been shown to vary considerably amongst different racial subgroups [
40,
41,
42,
43], which could potentially confound
FOLH1 findings. As such, consistent with prior recommendations [
47], we repeated our analyses in the largest mono-racial group so as to account for differences in frequency of folate-related SNPs amongst different genetic ancestries that could affect folate status [
40,
41,
42,
43,
47].
Though the lack of a main effect of
FOLH1 genotype on RBC folate status may seem surprising, prior research has demonstrated that the effects of certain folate-related SNPs may be attenuated in high-folate intake samples [
53]. As most participants reported considerable intake of synthetic folic acid, which bypasses the
FOLH1 carboxypeptidase and is directly absorbed into the body, the extent to which the
FOLH1 484 T>C polymorphism may influence folate status in this population is likely a function of both the polymorphism but also on the amount of naturally occurring food folate intake in each participant’s diet. For instance, a participant who consumed more naturally occurring food folate but had the hypofunctional
FOLH1 variant might absorb less than participants with the functional
FOLH1 variant. However, this polymorphism would not have as great an effect on blood folate levels for those who derive most of their daily folate from synthetic sources, for which absorption is not
FOLH1 dependent.
After conducting the regression analysis separately by sex, it appears that overall relationships between intake (from either naturally occurring food folate or synthetic folic acid) and genotype may have been driven by the female subsample. The results of our hierarchical linear regression analyses followed the same pattern in both the full sample and the female subsample, with total synthetic folic acid intake and the genotype by naturally occurring food folate interaction significantly predicting RBC folate concentration. However, of the four models, only the inclusion of total synthetic folic acid significantly predicted RBC folate concentrations in males. If significant gender effects are found in subsequent studies of this polymorphism these results could reflect sex differences in folate metabolism, folate absorption,
FOLH1 expression, or, as previously reported, lean body mass [
25]. However, it is essential to note that we did not find a significant three-way interaction between the
FOLH1 484T>C polymorphism, naturally occurring food folate intake, and sex. Therefore, no conclusions regarding sex-differences in the
FOLH1 by naturally occurring food folate intake interaction can be drawn from this work. Though our null finding may be due to a small sample size, our sex-stratified analyses suggest that future studies should perhaps consider possible sex effects.
The
FOLH1 484T>C polymorphism has received little study in the literature, especially when compared to other well-known polymorphisms such as
FOLH1 1561C>T. Specifically, the
FOLH1 1561C>T polymorphism has been associated with both lower and higher RBC folate [
54,
55], serum folate, [
4,
54,
55,
56,
57,
58], and homocysteine levels [
4,
55,
56,
57,
58,
59], though some have failed to find such associations [
60]. Furthermore, this polymorphism was not found to influence the bioavailability of monoglutamyl and polyglutamyl folic acid forms [
54] and likewise has not been found to influence risk for NTDs [
61]. In contrast, the
FOLH1 484 T allele load was positively associated with RBC folate levels in a sample of schizophrenia patients, and was associated with treatment response to L-methylfolate and vitamin B12 supplementation [
26]. Furthermore, this polymorphism has been associated with elevated homocysteine concentrations [
58]. Though to our knowledge no previous study has evaluated the influence of
FOLH1 484T>C polymorphism and dietary folate intake on RBC folate, our finding is nonetheless congruent with these previous results. While additional research is needed to clarify the functional role of the
FOLH1 484T>C polymorphism, together these studies suggest a functional effect of the
FOLH1 484 T>C SNP, such that the T-allele is associated with greater RBC folate levels, greater response of RBC folate to naturally occurring food folate intake, while the hypofunctional variant is associated with NTD risk. However, as this polymorphism remains poorly studied, additional research is needed to clarify the function of this SNP.
There are several important limitations to this study that must be taken into account. First, these results may have limited generalizability due to small sample size, single site design, and recruitment pool (i.e., individuals who were also willing to have an MRI scan). However, the validity of the current study is supported by the fact that the percentage of young women who fell below the EAR when considering folate intake from all sources (13.6%) was similar to that reported in a much larger epidemiologic study (15–19%) [
22]. Furthermore, total folate intake from all sources for male (905 mcg DFE) and female (712 mcg DFE) participants was similar to that previously reported (813 mcg DFE and 724 mcg DFE respectively, [
22]), and the distribution of
FOLH1 T484C alleles is consistent with that reported by larger sequencing studies (see
https://www.ncbi.nlm.nih.gov/SNP/snp_ss.cgi?ss=ss1690360421). Despite the smaller size, this cohort is deeply phenotyped and presents the first interactive effects of the
FOLH1 484T>C polymorphism and dietary intake on blood folate levels.
Second, nutritional intake was determined by self-report, which is subject to recall bias. A recent study that compared food frequency questionnaires to recovery biomarkers indicated substantial variation in the validity of intake self-report among nutrients [
62]. It is unclear what specific factors would contribute to the over- or under-reporting of folate intake, but this remains a theoretical concern, and could potentially contribute to differences between reported folate intake in men and women. Though the use of quantitative estimates from such questionnaires has been previously reported in the literature [
63,
64,
65], the definitive validation of such approaches for measuring long-term folate intake would require observation of the participant’s diet over a prolonged time not feasible for most studies [
48]. Many suggest that such a comparison of quantitative nutrient estimates relative to dietary cutoffs is only an approximate unless individual variation is accounted for by modeling or in calibration studies [
48,
66]. Despite these issues, several DHQs have nonetheless been shown to provide reasonable estimates of nutrient intake [
67,
68,
69,
70]. While the DHQ used in this study has generally been found to be a valid assessment of nutrient intake as a whole [
71], it has specifically been shown to show to provide a reasonable quantitative estimate of folate intake [
72,
73]. Nevertheless, though this measure may be a more reliable estimate of folate intake, it is critical to emphasize that the comparison of our findings with EAR values provides only qualitative evidence supporting the importance of fortification in maintaining dietary folate status. While interpreting our results relative to EAR cutoffs should be made with caution, as they only suggest the qualitative role of fortification in maintaining population folate status [
48], that our results are consistent with those previously reported in larger studies affirms the importance of folic acid fortification intake for meeting population folate requirements [
22].
Third, the strongest model emerging from our analysis still only accounted for less than 14 percent of the variance in RBC folate concentration (29% in female post-hoc analyses), indicating that the majority of variance in RBC folate levels reflects as-yet-to-be-determined factors. Though the significant interaction between naturally occurring food folate intake and the
FOLH1 484T>C polymorphism explained only 2.3% of the variance in RBC folate levels, previous research has shown the individual contributions of SNPs to complex phenotypes such as RBC folate is generally low. For instance, Pvalíková and colleagues found that the well-studied
MTHFR 677C>T variant specifically explained only 1% of the variance in RBC folate levels [
74]. Importantly, recent research done in a sample unexposed to folic acid fortification found
FOLH1 484 C homozygote mothers are at significantly greater risk of multiple-NTD-affected pregnancies compared to those with the functional variant [
75]. Previous work suggests that the effect of certain polymorphisms on RBC folate status may be attenuated in high folate intake populations [
53] while upregulation of
FOLH1 may be part of the physiological response to dietary folate deficiency [
76]. Accordingly, the interaction of
FOLH1 484T>C polymorphism and naturally occurring food folate may be of greater practical importance in populations for which RBC folate status is primarily determined by naturally occurring food folate, such as the high-NTD-risk population described by Guo and colleagues [
75]. Therefore, additional studies are needed to better understand the effect of this polymorphism both on RBC folate levels, but also on risk for NTD-affected pregnancy, especially in populations that rely on naturally occurring food folate.
Finally, as RBC folate assays are not standardized, results obtained using our ECLIA could not be compared to epidemiological cutoffs for RBC folate insufficiency based on metabolic indicators or elevated risk of NTD-affected pregnancy established with other folate assays [
31]. Furthermore, the lack of a standardized reference material for RBC folate measurement is problematic, as the accuracy of RBC measurements cannot be verified [
49]. However, the use of a single assay platform with a coefficient of variation within the accepted range supports the validity of our RBC folate measurements for within study comparisons. Additionally, there is not yet consensus as to whether RBC folate is the preferable determinant of long-term folate status [for a review see 46]. However, RBC folate remains a sensitive indicator of long-term storage, reflecting folate status during erythropoiesis [
77], is significantly correlated with liver folate stores [
78], and has been shown to be a reasonable biomarker of NTD-risk [
79]. Lastly, while previous research found different variants of the
MTHFR 677C>T polymorphism are associated with altered distribution of folate forms within red blood cells [
44], that the distribution of
MTHFR alleles did not significantly differ between
FOLH1 T homozygotes and C-allele carriers suggests
FOLH1 results are likely not influenced by differential
MTHFR-related folate recovery [
45,
46].
Despite its limitations, this study is consistent with previous research findings suggesting that while folic acid fortification and supplementation play a major role in boosting folate intake above EAR, many individuals still struggle to obtain sufficient folate as judged by this factor and as reported in previous work [
22]. Of concern is the fact that 13.6 percent of women (whom were all of child-bearing age) were below the EAR for folate. The neural tube closes approximately four weeks after conception, which is earlier than the average time women recognize they are pregnant, 5.9 weeks [
80]; this problem is compounded by the fact that approximately half of all pregnancies are unplanned [
81]. Thus, it is crucial that all women capable of bearing children have sufficient folate intake regardless of whether they are actively trying to become pregnant, so as to ensure those who become pregnant have optimal periconceptional folate levels. Intake of synthetic folic acid may be particularly important in women who carry the
FOLH1 484C allele, and who thus may have difficulty absorbing sufficient folate from natural sources.
As this variant has previously been associated with elevated risk of multiple-NTD-affected pregnancy [
75], additional research is needed to further clarify both this association and any mechanisms by which
FOLH1 484T>C contributes to NTD risk in different populations. Future research should therefore evaluate this
FOLH1 484T>C polymorphism in both populations exposed and unexposed to mandatory folic acid fortification programs, to better clarify the practical significance of the interaction between naturally occurring food folate and the
FOLH1 484 T>C polymorphism. As some have raised concerns about the tolerable upper limit of folate intake [
82], future work should consider how individuals benefit from fortification if they are already taking a supplement with a high folic acid content. Lastly, the findings in this study support larger-scale investigation of determinants of blood folate, especially in women of childbearing age, as well as obstacles to achieving sufficient folate intake.