Effects of Oral Administration of Silymarin in a Juvenile Murine Model of Non-alcoholic Steatohepatitis
Abstract
:1. Introduction
2. Materials and Methods
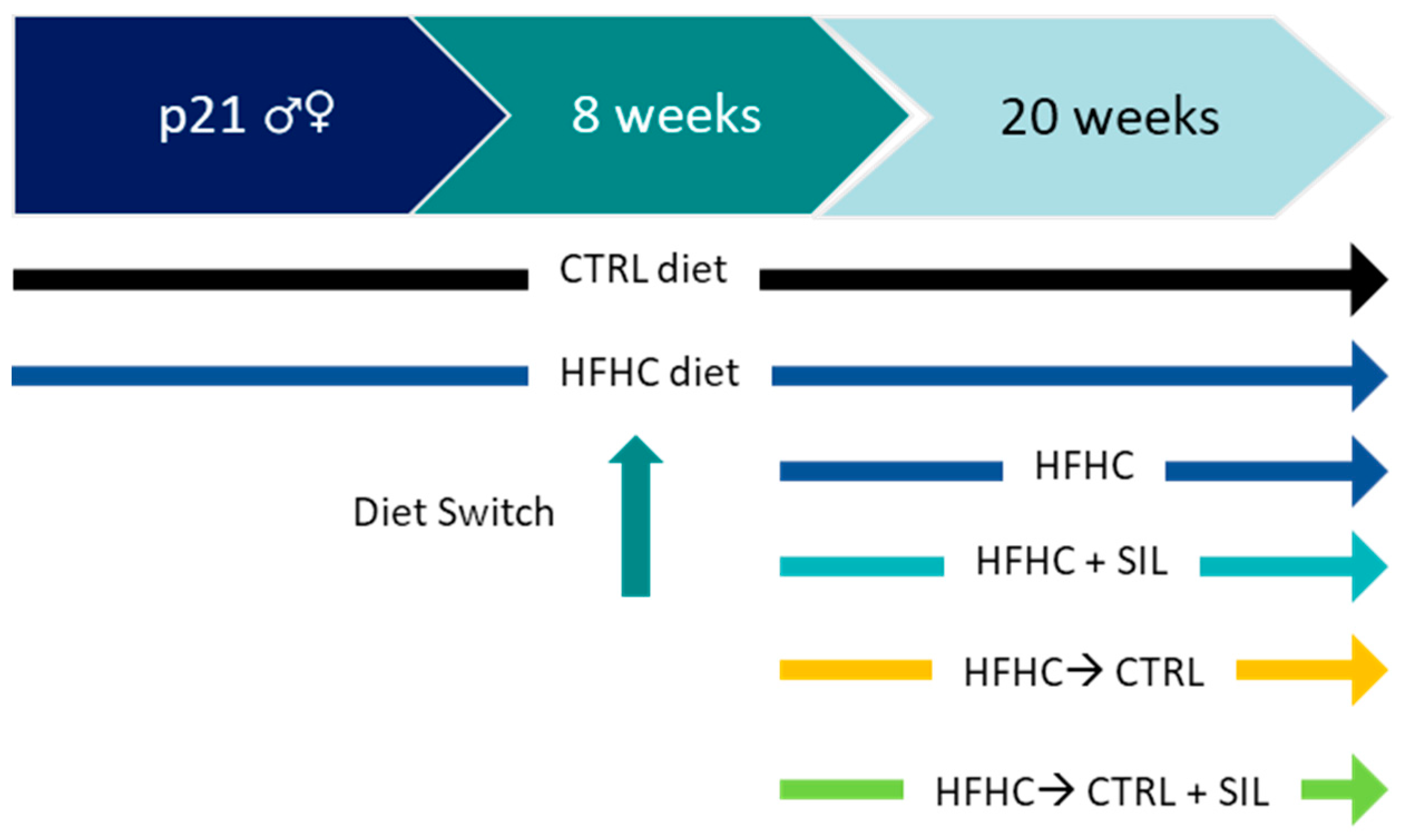
2.1. Experimental Set-Up
2.1.1. Animal Model (In Vivo)
2.1.2. HFHC + SILymarin Food Formulation
2.1.3. Cellular In Vitro Model
2.2. Intracellular Fat Quantification
2.3. Body, Liver, Perigonadal Adipose Tissue Weight, and Food Intake
2.4. Assessment of Glucose Homeostasis, Serum Insulin, and Insulin Resistance
2.5. Biochemical Parameters
2.6. Histopathological Analysis
2.7. Collagen Determination after SIRIUS Red/Fast Green Staining
2.8. Oxidative Stress Determination
2.8.1. Assessment of Lipid Peroxidation (MDA)
2.8.2. GSH/GSSG
2.8.3. Quantification of Reactive Oxygen Species (ROS)
2.9. RNA Extraction and cDNA Synthesis
2.10. Gene Expression Analysis by Real Time RT-PCR
2.11. Assessment of Apoptosis
2.11.1. In Vivo
2.11.2. In Vitro
2.12. Statistical Analysis
3. Results
3.1. Effect on the Body, Liver, and Adipose Tissue Weight
3.2. Effect on Glucose Homeostasis
3.3. Effects on Lipid Profile
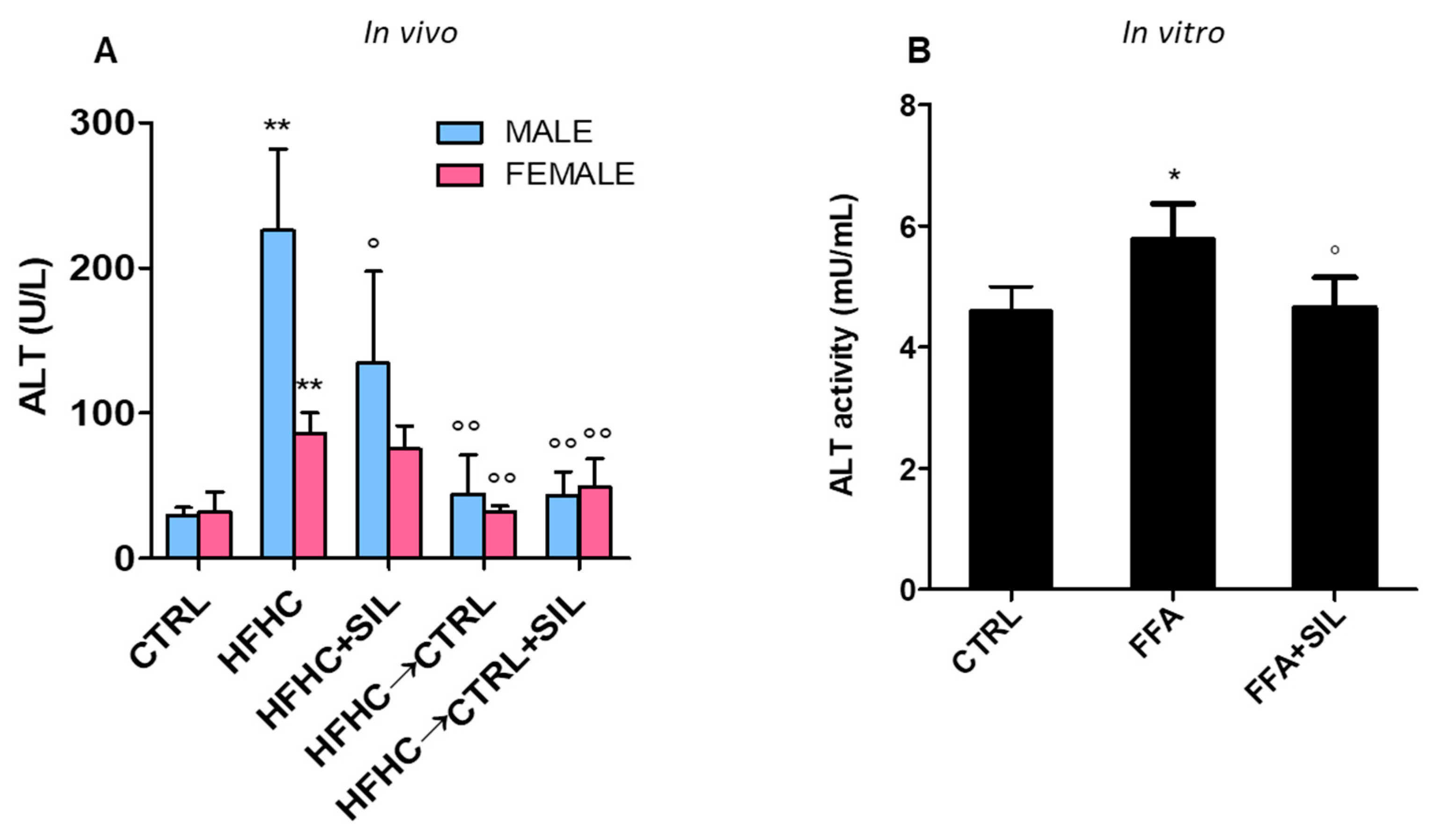
3.4. Effect on Transaminases Activity
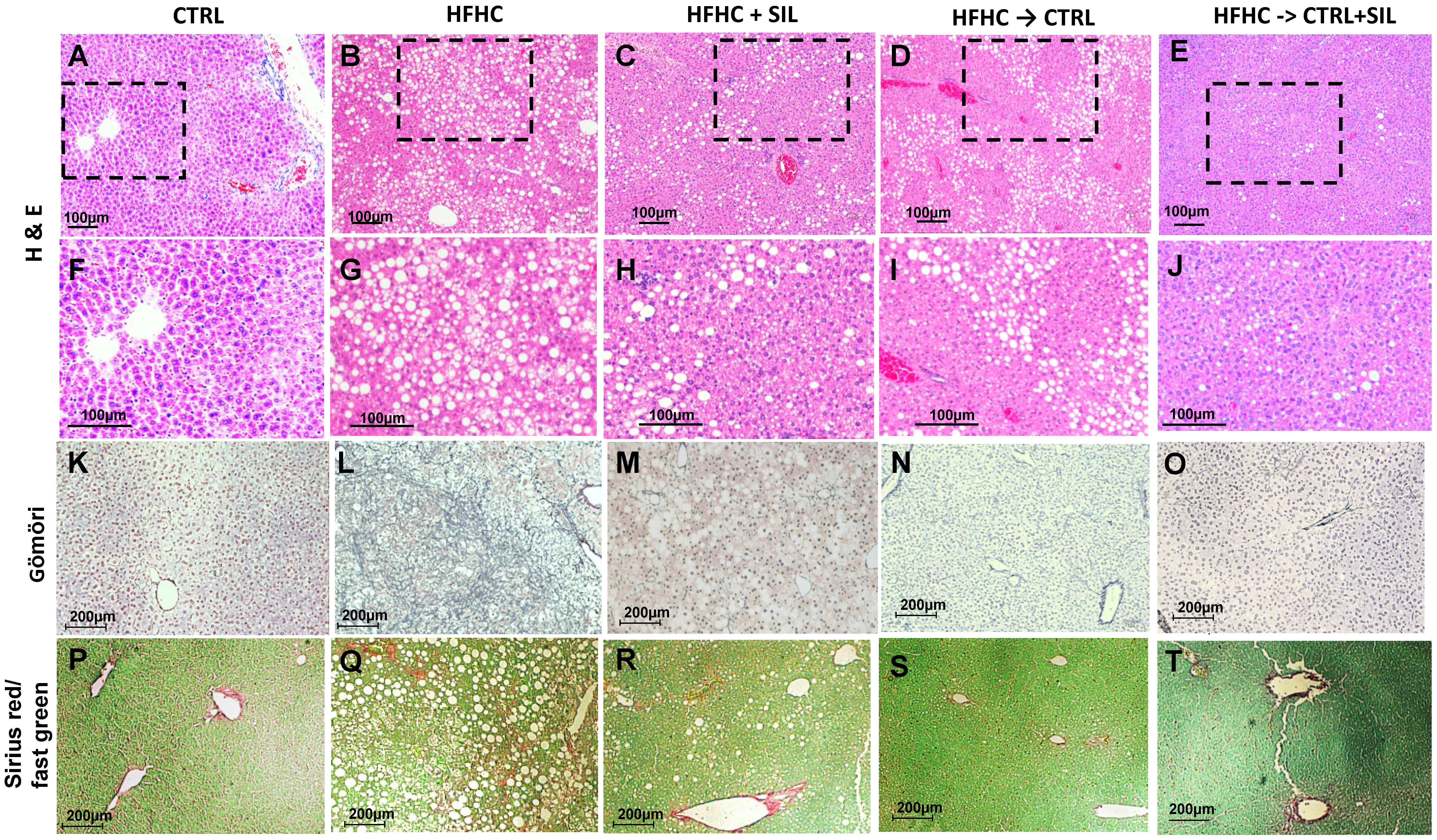
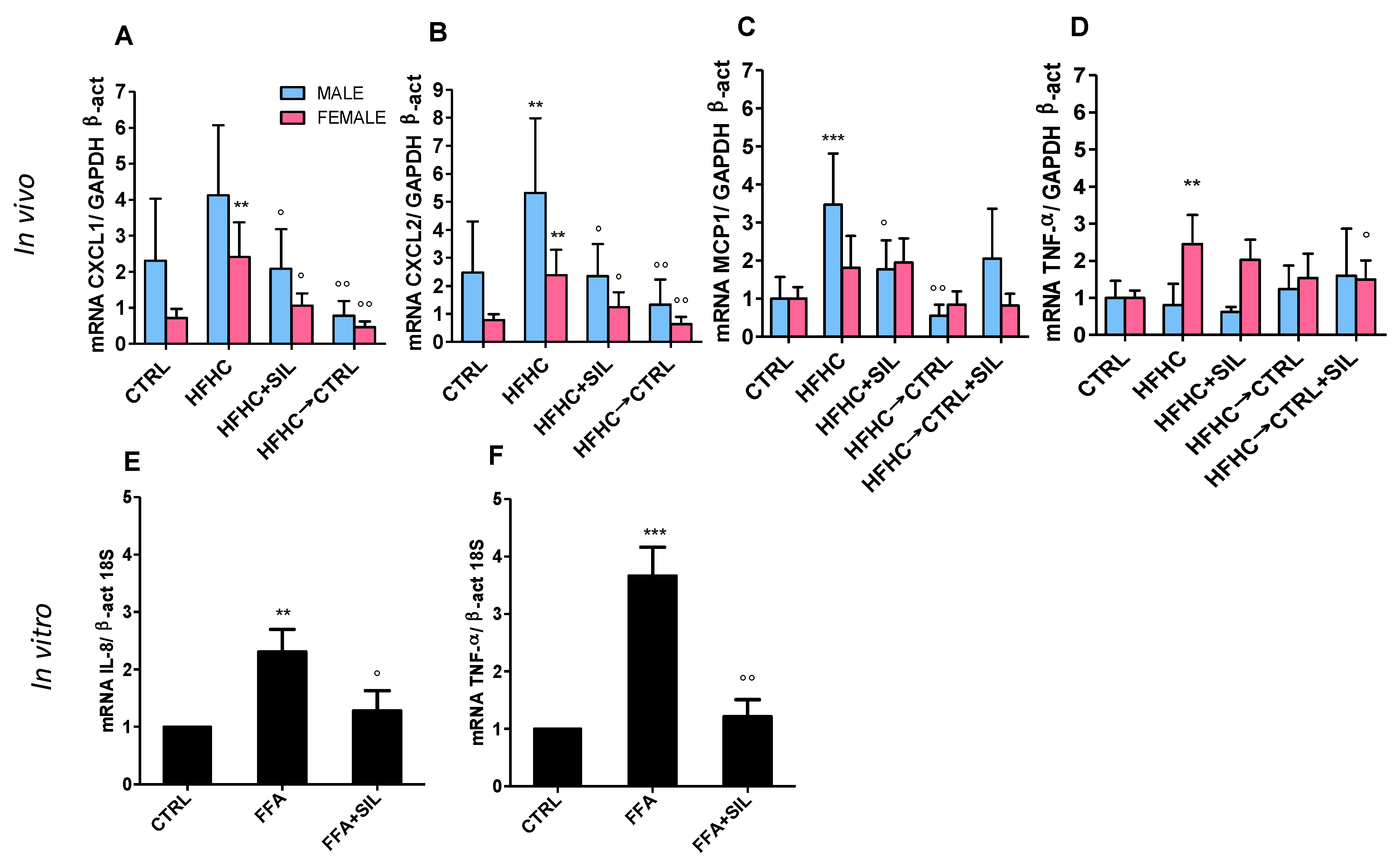
3.5. Hepatic Histological and Molecular Features Upon Silymarin Supplementation
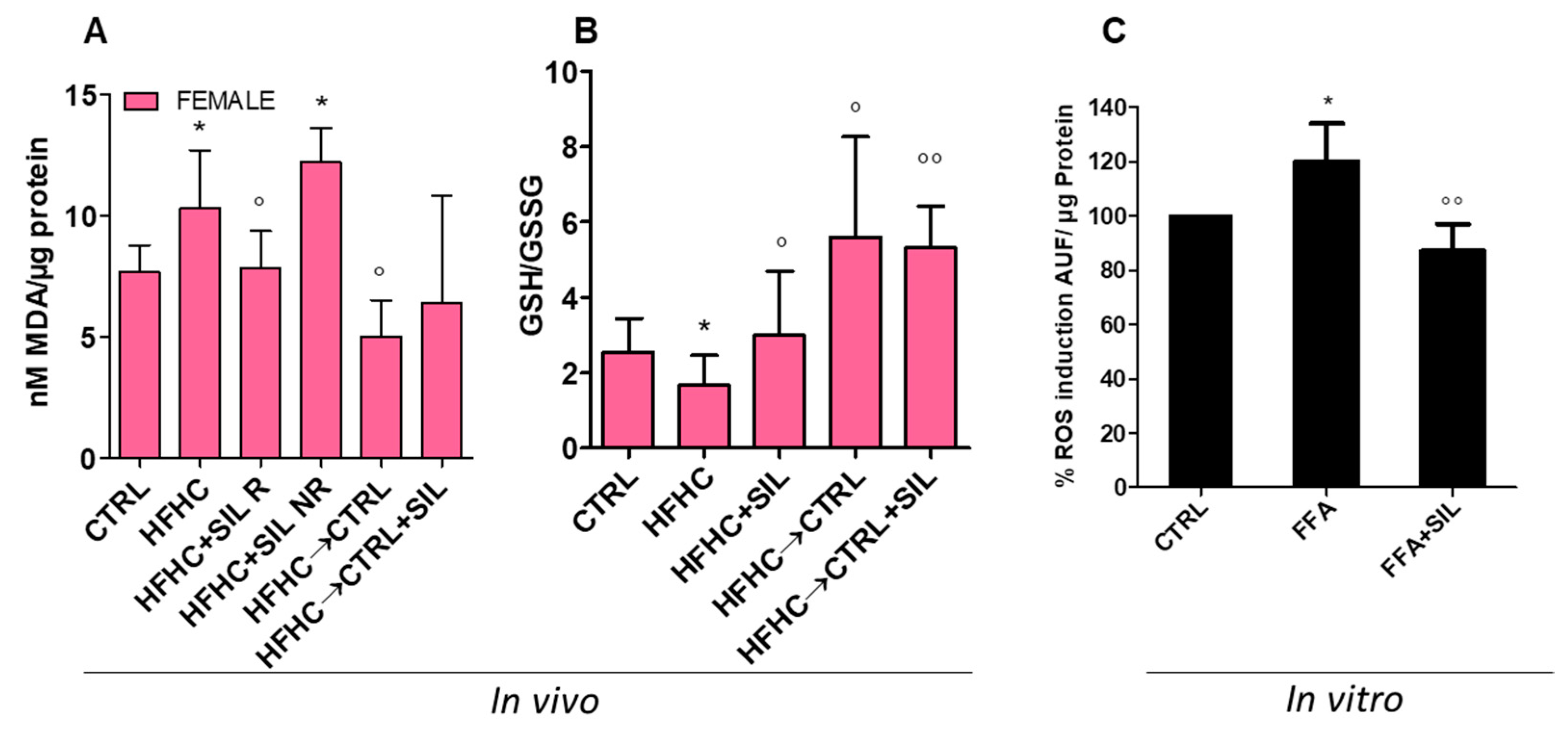
3.6. Antioxidant Properties
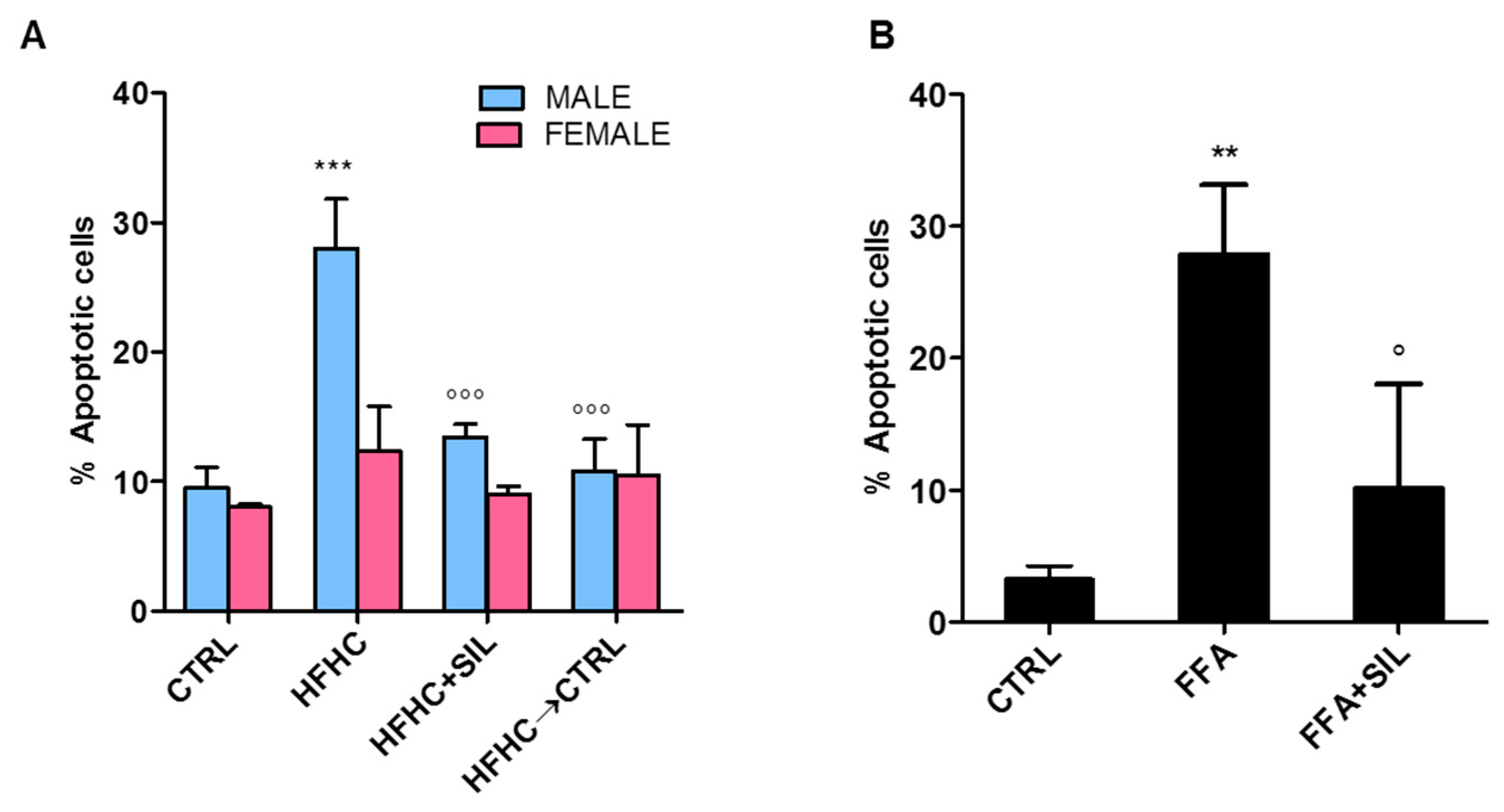
3.7. Anti-Apoptotic Effect
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Younossi, Z.M.; Blissett, D.; Blissett, R.; Henry, L.; Stepanova, M.; Younossi, Y.; Racila, A.; Hunt, S.; Beckerman, R. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatolory 2016, 64, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Manco, M.; Marcellini, M.; Devito, R.; Comparcola, D.; Sartorelli, M.R.; Nobili, V. Metabolic syndrome and liver histology in paediatric non-alcoholic steatohepatitis. Int. J. Obes. 2008, 32, 381–387. [Google Scholar] [CrossRef] [PubMed]
- De Bruyne, R.M.L.; Fitzpatrick, E.; Dhawan, A. Fatty liver disease in children: Eat now pay later. Hepatol. Int. 2010, 4, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Godos, J.; Salomone, F. Lifestyle changes for the treatment of nonalcoholic fatty liver disease: A review of observational studies and intervention trials. Ther. Adv. Gastroenterol. 2016, 9, 392–407. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for non-alcoholic fatty liver disease: Evidence and plausible mechanisms. Liver Int. 2017, 37, 936–949. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.C.; Suh, S.; Park, S.E.; Rhee, E.J.; Park, C.Y.; Oh, K.W.; Park, S.W.; Kim, S.W.; Hur, K.Y.; Kim, J.H.; et al. Regular exercise is associated with a reduction in the risk of NAFLD and decreased liver enzymes in individuals with NAFLD independent of obesity in Korean adults. PLoS ONE 2012, 7, e46819. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Guo, R.; Fung, M.L.; Liong, E.C.; Tipoe, G.L. Therapeutic approaches to non-alcoholic fatty liver disease: Past achievements and future challenges. Hepatobiliary Pancreat. Dis. Int. 2013, 12, 125–135. [Google Scholar] [CrossRef]
- Bistrian, B.R. Dietary composition during weight-loss maintenance. JAMA J. Am. Med. Assoc. 2012, 308, 1087–1088. [Google Scholar] [CrossRef] [PubMed]
- Hackett, E.S.; Twedt, D.C.; Gustafson, D.L. Milk thistle and its derivative compounds: A review of opportunities for treatment of liver disease. J. Vet. Intern. Med. 2013, 27, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Capasso, R.; Milic, N.; Capasso, F. Milk thistle in liver diseases: Past, present, future. Phytother. Res. 2010, 24, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk thistle (Silybum marianum) for the therapy of liver disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Saller, R.; Meier, R.; Brignoli, R. The use of silymarin in the treatment of liver diseases. Drugs 2001, 61, 2035–2063. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, S.C.; Girish, C. Hepatoprotective herbal drug, silymarin from experimental pharmacology to clinical medicine. Indian J. Med. Res. 2006, 124, 491–504. [Google Scholar] [PubMed]
- Katiyar, S.K. Silymarin and skin cancer prevention: Anti-inflammatory, antioxidant and immunomodulatory effects. Int. J. Oncol. 2005, 26, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, H.; Abascal, K.; Yarnell, E.; Ladas, E. Clinical applications of Silybum marianum in oncology. Integr. Cancer Ther. 2007, 6, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Agarwal, C.; Ichikawa, H.; Singh, R.P.; Aggarwal, B.B. Anticancer potential of silymarin: From bench to bed side. Anticancer Res. 2006, 26, 4457–4498. [Google Scholar] [PubMed]
- Deep, G.; Agarwal, R. Antimetastatic efficacy of silibinin: Molecular mechanisms and therapeutic potential against cancer. Cancer Metastasis Rev. 2010, 29, 447–463. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Kim, S.G.; Kim, H.K.; Lee, T.-H.; Jeong, Y.-I.; Lee, C.-M.; Yoon, M.-S.; Na, Y.J.; Suh, D.-S.; Park, N.C.; et al. Silibinin polarizes Th1/Th2 immune responses through the inhibition of immunostimulatory function of dendritic cells. J. Cell. Physiol. 2007, 210, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Krecman, V.; Skottová, N.; Walterová, D.; Ulrichová, J.; Simánek, V. Silymarin inhibits the development of diet-induced hypercholesterolemia in rats. Planta Med. 1998, 64, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Skottová, N.; Krecman, V. Silymarin as a potential hypocholesterolaemic drug. Physiol. Res. Acad. Sci. Bohemoslov. 1998, 47, 1–7. [Google Scholar]
- Loguercio, C.; Festi, D. Silybin and the liver: From basic research to clinical practice. World J. Gastroenterol. 2011, 17, 2288–2301. [Google Scholar] [CrossRef] [PubMed]
- Fraschini, F.; Demartini, D.; Esposti, D. Pharmacology of Silymarin. Clin. Drug Investig. 2002, 22, 51–65. [Google Scholar] [CrossRef]
- Voinovich, D.; Perissutti, B.; Grassi, M.; Passerini, N.; Bigotto, A. Solid state mechanochemical activation of Silybum marianum dry extract with betacyclodextrins: Characterization and bioavailability of the coground systems. J. Pharm. Sci. 2009, 98, 4119–4129. [Google Scholar] [CrossRef] [PubMed]
- Salamone, F.; Galvano, F.; Marino Gammazza, A.; Marino, A.; Paternostro, C.; Tibullo, D.; Bucchieri, F.; Mangiameli, A.; Parola, M.; Bugianesi, E.; et al. Silibinin improves hepatic and myocardial injury in mice with nonalcoholic steatohepatitis. Dig. Liver Dis. 2012, 44, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Andreone, P.; Brisc, C.; Brisc, M.C.; Bugianesi, E.; Chiaramonte, M.; Cursaro, C.; Danila, M.; de Sio, I.; Floreani, A.; et al. Silybin combined with phosphatidylcholine and vitamin E in patients with nonalcoholic fatty liver disease: A randomized controlled trial. Free Radic. Biol. Med. 2012, 52, 1658–1665. [Google Scholar] [CrossRef] [PubMed]
- Cacciapuoti, F.; Scognamiglio, A.; Palumbo, R.; Forte, R.; Cacciapuoti, F. Silymarin in non alcoholic fatty liver disease. World J. Hepatol. 2013, 5, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Rosso, N.; Marin, V.; Giordani, A.; Persiani, S.; Sala, F.; Cavicchioli, L.; Rovati, L.C.; Tiribelli, C. The pros and the cons for the use of Silybin-rich oral formulations in treatment of liver damage (NAFLD in particular). Curr. Med. Chem. 2015, 22, 2954–2971. [Google Scholar] [CrossRef] [PubMed]
- Africa, J.A.; Newton, K.P.; Schwimmer, J.B. Lifestyle Interventions Including Nutrition, Exercise, and Supplements for Nonalcoholic Fatty Liver Disease in Children. Dig. Dis. Sci. 2016, 61, 1375–1386. [Google Scholar] [CrossRef] [PubMed]
- Marin, V.; Rosso, N.; Dal Ben, M.; Raseni, A.; Boschelle, M.; Degrassi, C.; Nemeckova, I.; Nachtigal, P.; Avellini, C.; Tiribelli, C.; et al. An Animal Model for the Juvenile Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis. PLoS ONE 2016, 11, e0158817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavez-Tapia, N.C.; Rosso, N.; Tiribelli, C. Effect of intracellular lipid accumulation in a new model of non-alcoholic fatty liver disease. BMC Gastroenterol. 2012, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Kroll, D.J.; Shaw, H.S.; Oberlies, N.H. Milk thistle nomenclature: Why it matters in cancer research and pharmacokinetic studies. Integr. Cancer Ther. 2007, 6, 110–119. [Google Scholar] [CrossRef] [PubMed]
- McMillian, M.K.; Grant, E.R.; Zhong, Z.; Parker, J.B.; Li, L.; Zivin, R.A.; Burczynski, M.E.; Johnson, M.D. Nile Red binding to HepG2 cells: An improved assay for in vitro studies of hepatosteatosis. In Vitro Mol. Toxicol. 2001, 14, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, P.; Mayer, E.P.; Fowler, S.D. Nile red: A selective fluorescent stain for intracellular lipid droplets. J. Cell Biol. 1985, 100, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Altunkaynak, B.Z.; Altunkaynak, M.E. Relationship of body weight and volume of liver. A morphometrical and stereological study. Saudi Med. J. 2007, 28, 891–895. [Google Scholar] [PubMed]
- Akagiri, S.; Naito, Y.; Ichikawa, H.; Mizushima, K.; Takagi, T.; Handa, O.; Kokura, S.; Yoshikawa, T. A Mouse Model of Metabolic Syndrome; Increase in Visceral Adipose Tissue Precedes the Development of Fatty Liver and Insulin Resistance in High-Fat Diet-Fed Male KK/Ta Mice. J. Clin. Biochem. Nutr. 2008, 42, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.-C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Nonalcoholic Steatohepatitis Clinical Research Network Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Brunt, E.M.; Kleiner, D.E.; Wilson, L.A.; Belt, P.; Neuschwander-Tetri, B.A.; NASH Clinical Research Network (CRN). Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: Distinct clinicopathologic meanings. Hepatology 2011, 53, 810–820. [Google Scholar] [CrossRef] [PubMed]
- López-De León, A.; Rojkind, M. A simple micromethod for collagen and total protein determination in formalin-fixed paraffin-embedded sections. J. Histochem. Cytochem. 1985, 33, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H.; Schaur, R.J.; Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Hodges, D.M.; DeLong, J.M.; Forney, C.F.; Prange, R.K. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Zitka, O.; Skalickova, S.; Gumulec, J.; Masarik, M.; Adam, V.; Hubalek, J.; Trnkova, L.; Kruseova, J.; Eckschlager, T.; Kizek, R. Redox status expressed as GSH:GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncol. Lett. 2012, 4, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Held, P. Fluorimetric Quantitation of Protein using the Reactive Compound Fluorescamine. Nat. Methods Appl. Notes 2006. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Feldstein, A.E.; Canbay, A.; Angulo, P.; Taniai, M.; Burgart, L.J.; Lindor, K.D.; Gores, G.J. Hepatocyte apoptosis and fas expression are prominent features of human nonalcoholic steatohepatitis. Gastroenterology 2003, 125, 437–443. [Google Scholar] [CrossRef]
- European Health Interview Survey Almost 1 Adult in 6 in the EU is Considered Obese. Available online: http://ec.europa.eu/eurostat/documents/2995521/7700898/3-20102016-BP-EN.pdf/c26b037b-d5f3-4c05-89c1-00bf0b98d646 (accessed on 3 July 2017).
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Shoelson, S.E.; Herrero, L.; Naaz, A. Obesity, inflammation, and insulin resistance. Gastroenterology 2007, 132, 2169–2180. [Google Scholar] [CrossRef] [PubMed]
- Schwimmer, J.B.; Deutsch, R.; Kahen, T.; Lavine, J.E.; Stanley, C.; Behling, C. Prevalence of fatty liver in children and adolescents. Pediatrics 2006, 118, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
- Berardis, S.; Sokal, E. Pediatric non-alcoholic fatty liver disease: An increasing public health issue. Eur. J. Pediatr. 2014, 173, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Rodas, M.C.; Valenzuela, R.; Videla, L.A. Relevant Aspects of Nutritional and Dietary Interventions in Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2015, 16, 25168–25198. [Google Scholar] [CrossRef] [PubMed]
- Centis, E.; Moscatiello, S.; Bugianesi, E.; Bellentani, S.; Fracanzani, A.L.; Calugi, S.; Petta, S.; Dalle Grave, R.; Marchesini, G. Stage of change and motivation to healthier lifestyle in non-alcoholic fatty liver disease. J. Hepatol. 2013, 58, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Saller, R.; Melzer, J.; Reichling, J.; Brignoli, R.; Meier, R. An updated systematic review of the pharmacology of silymarin. Forsch. Komplementarmed. 2007, 14, 70–80. [Google Scholar] [CrossRef]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2016, 152, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Yang, Z.; Sun, J.; Tian, J.; Chang, Z.; Li, X.; Zhang, B.; Ye, Y.; Ji, H.; Yu, E.; et al. Silymarin inhibits adipogenesis in the adipocytes in grass carp Ctenopharyngodon idellus in vitro and in vivo. Fish Physiol. Biochem. 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Linton, M.F.; Yancey, P.G.; Davies, S.S.; Jerome, W.G.J.; Linton, E.F.; Vickers, K.C. The Role of Lipids and Lipoproteins in Atherosclerosis. In Endotext; de Groot, L.J., Chrousos, G., Dungan, K., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Hayek, T.; Ito, Y.; Azrolan, N.; Verdery, R.B.; Aalto-Setälä, K.; Walsh, A.; Breslow, J.L. Dietary fat increases high density lipoprotein (HDL) levels both by increasing the transport rates and decreasing the fractional catabolic rates of HDL cholesterol ester and apolipoprotein (Apo) A-I. Presentation of a new animal model and mechanistic studies in human Apo A-I transgenic and control mice. J. Clin. Investig. 1993, 91, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Haukeland, J.W.; Damås, J.K.; Konopski, Z.; Løberg, E.M.; Haaland, T.; Goverud, I.; Torjesen, P.A.; Birkeland, K.; Bjøro, K.; Aukrust, P. Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J. Hepatol. 2006, 44, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Pinzani, M.; Marra, F. Cytokine receptors and signaling in hepatic stellate cells. Semin. Liver Dis. 2001, 21, 397–416. [Google Scholar] [CrossRef] [PubMed]
- Kren, V.; Walterová, D. Silybin and silymarin—New effects and applications. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czechoslov. 2005, 149, 29–41. [Google Scholar] [CrossRef]
- Valenzuela, A.; Garrido, A. Biochemical bases of the pharmacological action of the flavonoid silymarin and of its structural isomer silibinin. Biol. Res. 1994, 27, 105–112. [Google Scholar] [PubMed]
- Aghazadeh, S.; Amini, R.; Yazdanparast, R.; Ghaffari, S.H. Anti-apoptotic and anti-inflammatory effects of Silybum marianum in treatment of experimental steatohepatitis. Exp. Toxicol. Pathol. 2011, 63, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Guicciardi, M.E.; Gores, G.J. Life and death by death receptors. FASEB J. 2009, 23, 1625–1637. [Google Scholar] [CrossRef] [PubMed]
- Trappoliere, M.; Caligiuri, A.; Schmid, M.; Bertolani, C.; Failli, P.; Vizzutti, F.; Novo, E.; di Manzano, C.; Marra, F.; Loguercio, C.; et al. Silybin, a component of sylimarin, exerts anti-inflammatory and anti-fibrogenic effects on human hepatic stellate cells. J. Hepatol. 2009, 50, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Salamone, F.; Galvano, F.; Cappello, F.; Mangiameli, A.; Barbagallo, I.; Li Volti, G. Silibinin modulates lipid homeostasis and inhibits nuclear factor kappa B activation in experimental nonalcoholic steatohepatitis. Transl. Res. J. Lab. Clin. Med. 2012, 159, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Mills, P.R.; Keach, J.C.; Lafferty, H.D.; Stahler, A.; et al. Liver Fibrosis, but No Other Histologic Features, Is Associated With Long-term Outcomes of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterol. 2015, 149, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Hecht, L.; Weiss, R. Nonalcoholic fatty liver disease and type 2 diabetes in obese children. Curr. Diab. Rep. 2014, 14, 448. [Google Scholar] [CrossRef] [PubMed]
- Ladas, E.; Kroll, D.; Oberlies, N.; Cheng, B.; Hughes, D.; Rheingold, S.; Kelly, K. A randomized controlled, double-blind pilot study of milk thistle for the treatment of hepatotoxicity in childhood acute lymphoblastic leukemia (ALL). Cancer 2010, 116, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Kassem, L.; Abdelrahim, M.; Naguib, H. Investigating the Efficacy and Safety of Silymarin in Management of Hyperbilirubinemia in Neonatal Jaundice. Med. Sci. 2013, 2, 575–590. [Google Scholar] [CrossRef]
- Home—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/home (accessed on 22 August 2017).


| Mouse Genes | Accession Number | Forward | Reverse |
| Gapdh | NM_008084 | CCAGTATGACTCCACTCACG | CTCGCTCCTGGAAGATGGTG |
| β-Actin | NM_007393 | CCTTCTTGGGTATGGAATCCTGTG | CAGCACTGTGTTGGCATAGAGG |
| Dgat-2 | NM_026384 | CCAAGAAAGGTGGCAGGA | ATGGGAAAGTAGTCTCGGAAG |
| Mcp-1 | NM_011333 | TGTGACTCGGACTGTGAT | CATTGAAAGTGTTGAATCTGGAT |
| Tnf-α | NM_013693 | AGACCCTCACACTCAGAT | CTTGGTGGTTTGCTACGA |
| Cxcl-1 | NM_008176 | CATAGCCACACTCAAGAA | GGACACCTTTTAGCATCT |
| Cxcl-2 | NM_009140 | ACTTCAAGAACATCCAGAG | TTGAGAGTGGCTATGACT |
| α-Sma | NM_007392.2 | GGCATCAATCACTTCAAC | TCTGGTCACCTGTATGTA |
| Col-1a1 | NM_007742.3 | AAGAAGACATCCCTGAAG | ATACAGATCAAGCATACCT |
| Human Genes | Accession Number | Forward | Reverse |
| β-Actin | NM_001101.3 | CGCCGCCAGCTCACCATG | CACGATGGAGGGGAAGACGG |
| 18S | NR_003286.2 | TAACCCGTTGAACCCATT | CCATCCAATCGGTAGTAGCG |
| IL-8 | NM_000584 | GACATACTCCAAACCTTTCCAC | CTTCTCCACAACCCTCTGC |
| TNF-α | NM_000594 | GTGAGGAGGACGAACATC | GAGCCAGAAGAGGTTGAG |
| MCP-1 | NM_002982 | CCAGTCACCTGCTGTTAT | CAATGGTCTTGAAGATCACA |
| Parameters | MALES | FEMALES | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL + SIL | Control | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL + SIL | |
| Body weight (g) | 29.2 ± 2.6 | 43.4 ± 1.8 *** | 43.7 ± 4.6 | 34.4 ± 2.6 ### | 30.3 ± 3.3 ### | 23.4 ± 1.0 | 35.4 ± 4.3 ** | 31.8 ± 3.4 | 23.5 ± 1.5 ## | 23.9 ± 1.9 ## |
| BMI (kg/m2) | 3.2 ± 0.3 | 4.8 ± 0.2 *** | 4.8 ± 0.5 | 3.7 ± 0.3 ### | 3.3 ± 0.4 | 2.9 ± 0.1 | 4.4 ± 0.5 *** | 3.9 ± 0.4 | 2.9 ± 0.2 ### | 2.9 ± 0.1 ## |
| Liver weight (g) | 0.96 ± 0.08 | 1.8 ± 0.2 *** | 1.8 ± 0.1 | 1.1 ± 0.1 ## | 1.00 ± 0.04 ## | 0.88 ± 0.05 | 1.2 ± 0.1 * | 1.09 ± 0.15 # | 0.9 ± 0.1 | 0.94 ± 0.01 ## |
| Perigonadal Fat Pads (g) | 0.98 ± 0.08 | 2.4 ± 0.3 *** | 2.3 ± 0.2 # | 1.3 ± 0.28 ## | 1.0 ± 0.4 ## | 0.32 ± 0.01 | 1.5 ± 0.6 ** | 0.9 ± 0.3 # | 0.32 ± 0.09 ### | 0.27 ± 0.07 ### |
| Parameters | MALES | FEMALES | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL + SIL | Control | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL + SIL | |
| Glucose (mg/dL) | 164 ± 20 | 260 ± 24 *** | 226 ± 17 # | 167 ± 13 ## | 187 ± 17 ## | 141 ± 10 | 193 ± 25 ** | 181 ± 31 | 134 ± 6 ### | 142 ± 12 ### |
| Insulinemia (µU/mL) | 1.9 ± 0.4 | 4.1 ± 1.1 ** | 3.6 ± 1.3 | 2.0 ± 1.2 ## | 1.5 ± 0.7 ### | 0.9 ± 0.1 | 2.5 ± 1.7 | 2.1 ± 0.5 | 1.3 ± 0.3 # | 1.2 ± 0.4 # |
| HOMA-IR | 1.4 ± 0.4 | 2.8 ± 0.7 * | 2.0 ± 0.8 | 0.6 ± 0.1 ## | 0.7 ± 0.4 ## | 0.3 ± 0.004 | 1.3 ± 1.0 | 0.9 ± 0.2 | 0.4 ± 0.1 | 0.4 ± 0.2 |
| MALES | FEMALES | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | CTRL | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL +SIL | CTRL | HFHC | HFHC + SIL | HFHC→CTRL | HFHC→CTRL +SIL |
| Steatosis Grade (0–3) | 2 (100%) | 3 (100%) | 3 (60%) 2 (40%) | 2 (50%) 1 (50%) | 1 (60%) 2 (40%) | 1 (33%) 2 (66%) | 1 (20%) 2 (80%) | 2 (100%) | 1 (75%) 2 (25%) | 2(100%) |
| Location (score 0–3) | 3 (100%) | 3 (40%) 0 (60%) | 3 (60%) 2 (40%) | 3 (75%) 2(25%) | 2 (100%) | 3 (100%) | 0 (20%) 2 (60%) | 0 (33.3%) 2 (66.6%) | 2 (100%) | 2 (100%) |
| Portal Inflammation (score 0–1) | 1 (100%) | 0 (100%) | 0 (88%) 1 (12%) | 1 (50%) 0 (50%) | 0 (100%) | 0 (100%) | 0 (60%) 1 (40%) | 0 (50%) 1 (50%) | 0 (25%) 1 (75%) | 0 (100%) |
| Lobular Inflammation Score (0–3) | 0 (20%) 1 (80%) | 0 (80%) 1 (20%) | 0 (40%) 1 (60%) | 1 (75%) 0 (25%) | 1 (100%) | 1 (66%) 0 (33%) | 0 (40%) 1 (40%) 2 (20%) | 1 (100%) | 0 (25%) 1 (75%) | 0 (100%) |
| Fibrosis Stage (0–4) | 1a (80%) | 1a (60%) 1c (40%) | 1a (50%) 1c (25%) 0 (25%) | 1a (50%) 1c (25%) 0 (25%) | 1a (100%) | 1a (83.3%) 1b (16.6%) | 1a (40%) 1c (60%) | 1a (83.3%) 1b (16.6%) | 0 (25%) 1a (25%) 1b (25%) 1c (25%) | 1a (100%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marin, V.; Gazzin, S.; Gambaro, S.E.; Dal Ben, M.; Calligaris, S.; Anese, M.; Raseni, A.; Avellini, C.; Giraudi, P.J.; Tiribelli, C.; et al. Effects of Oral Administration of Silymarin in a Juvenile Murine Model of Non-alcoholic Steatohepatitis. Nutrients 2017, 9, 1006. https://doi.org/10.3390/nu9091006
Marin V, Gazzin S, Gambaro SE, Dal Ben M, Calligaris S, Anese M, Raseni A, Avellini C, Giraudi PJ, Tiribelli C, et al. Effects of Oral Administration of Silymarin in a Juvenile Murine Model of Non-alcoholic Steatohepatitis. Nutrients. 2017; 9(9):1006. https://doi.org/10.3390/nu9091006
Chicago/Turabian StyleMarin, Veronica, Silvia Gazzin, Sabrina E. Gambaro, Matteo Dal Ben, Sonia Calligaris, Monica Anese, Alan Raseni, Claudio Avellini, Pablo J. Giraudi, Claudio Tiribelli, and et al. 2017. "Effects of Oral Administration of Silymarin in a Juvenile Murine Model of Non-alcoholic Steatohepatitis" Nutrients 9, no. 9: 1006. https://doi.org/10.3390/nu9091006
APA StyleMarin, V., Gazzin, S., Gambaro, S. E., Dal Ben, M., Calligaris, S., Anese, M., Raseni, A., Avellini, C., Giraudi, P. J., Tiribelli, C., & Rosso, N. (2017). Effects of Oral Administration of Silymarin in a Juvenile Murine Model of Non-alcoholic Steatohepatitis. Nutrients, 9(9), 1006. https://doi.org/10.3390/nu9091006






