Prevention and Therapy of Type 2 Diabetes—What Is the Potential of Daily Water Intake and Its Mineral Nutrients?
Abstract
1. Introduction
2. Methods
2.1. Overview
2.2. Study Search
2.3. Study Inclusion and Exclusion Criteria
- (1)
- Types of study: RCTs were the focus of this review to restrict potential sources of error and facilitate inference about possible causal mechanisms. They were included regardless of quality measures, such as double-blind design, complete protocol adherence, patient attrition, similarity of treatment and control groups at baseline and intention-to-treat analysis or sample size, target population or unit of analysis (e.g., individual or group-level data). They were only eligible if they were published as full paper articles. No language restrictions were made.
- (2)
- Types of participants: individuals of all ages (whether healthy or with or without type 2 diabetes, at risk of type 2 diabetes or other diseases, as having cardiovascular risk factors (CVRF), being overweight or obese or not, with hyperlipemia or not; excluded: type 1 diabetes).
- (3)
- Types of intervention: plain or drinking water, served at any temperature, whether tap or bottled water, high or low mineralized, carbonated or uncarbonated water.
- (4)
- Types of comparisons: interventions or control groups with drinking water tested alone, as a single intervention or as main part of a multi-component intervention for at least four weeks.
- (5)
- Types of outcome: at least one parameter for glycemic control as shown in Table 2 independent whether it was a primary or secondary outcome of the study.
2.4. Data Extraction and Risk of Bias Evaluation
3. Results
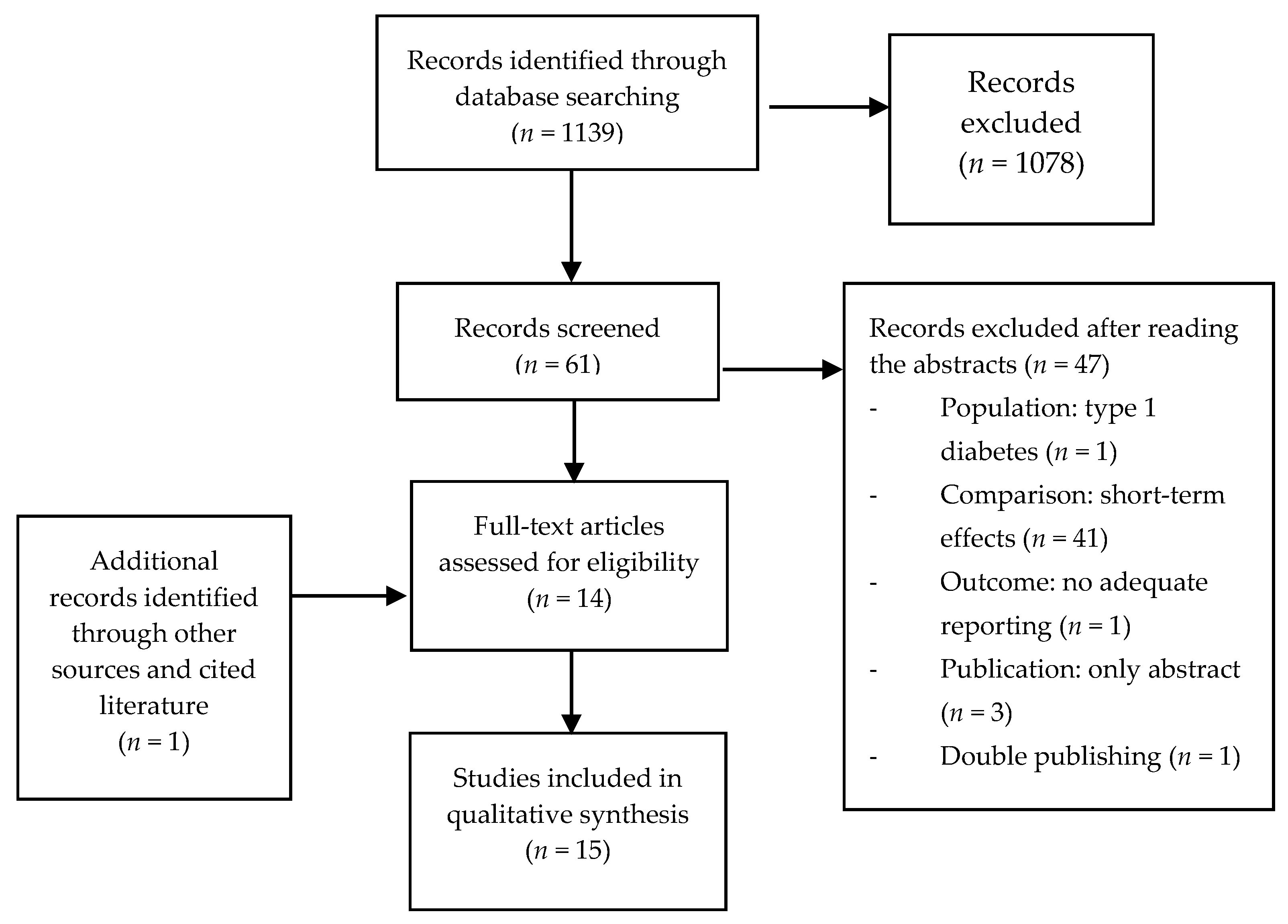
3.1. Literature Search
- (a)
- Population: 1 (type 1 diabetes)
- (b)
- Comparison: 41 trials only investigated short-term effects (<4-weeks treatment)
- (c)
- Outcome: 1 (glycemic parameter measured, but not reported)
- (d)
- Publication: 3 only abstracts (1 Russian, 1 Japanese, 1 Italian)
- (e)
- Double publishing: 1
3.2. Description of Included Studies
4. Discussion
4.1. Results of RCTs Comparing Water with Other Drinks or No Drink (Table 3)
4.2. Results of RCTs Comparing Low Mineralized Water with Bicarbonate-Rich Water with Low Magnesium (Table 4)
4.3. Results of RCT Comparing Low Mineralized Water with Bicarbonate-Rich Waters with High Magnesium (Table 5)
4.3.1. Effect of Bicarbonate as a Supplement
4.3.2. Effect of Mineral Rich Water in an Animal Model
4.4. Clinical Trials Considering Magnesium
4.4.1. Magnesium Blood Concentration
4.4.2. Dietary Magnesium
4.4.3. Relevance of Magnesium for Glucose Metabolism
4.5. Summary
5. Limitations and Generalizability
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. Diabetes. Available online: http://www.who.int/mediacentre/factsheets/fs312/en/ (accessed on 3 August 2017).
- Gesundheitssurvey Prävalenz Häufiger Erkrankungen. Available online: http://www.gbe-bund.de/gbe10/ergebnisse.prc_tab?fid=24742&suchstring=Diabetes&query_id=&sprache=D&fund_typ=TAB&methode=3&vt=1&verwandte=1&page_ret=0&seite=1&p_lfd_nr=75&p_news=&p_sprachkz=D&p_uid=gast&p_aid=75552896&hlp_nr=3&p_janein=J#SEARCH=%2522Diabetes%2522 (accessed on 13 April 2017).
- American Diabetes Association. 5. Prevention or Delay of Type 2 Diabetes. Diabetes Care 2017, 40, S44–S47. [Google Scholar] [CrossRef]
- Li, G.; Zhang, P.; Wang, J.; An, Y.; Gong, Q.; Gregg, E.W.; Yang, W.; Zhang, B.; Shuai, Y.; Hong, J.; et al. Cardiovascular mortality, all-cause mortality, and diabetes incidence after lifestyle intervention for people with impaired glucose tolerance in the Da Qing Diabetes Prevention Study: A 23-year follow-up study. Lancet Diabetes Endocrinol. 2014, 2, 474–480. [Google Scholar] [CrossRef]
- O’Connor, L.; Imamura, F.; Lentjes, M.A.H.; Khaw, K.-T.; Wareham, N.J.; Forouhi, N.G. Prospective associations and population impact of sweet beverage intake and type 2 diabetes, and effects of substitutions with alternative beverages. Diabetologia 2015, 58, 1474–1483. [Google Scholar] [CrossRef] [PubMed]
- Imamura, F.; O’Connor, L.; Ye, Z.; Mursu, J.; Hayashino, Y.; Bhupathiraju, S.N.; Forouhi, N.G. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: Systematic review, meta-analysis, and estimation of population attributable fraction. BMJ 2015, 351, h3576. [Google Scholar] [CrossRef] [PubMed]
- Gibson, S.A.; Horgan, G.W.; Francis, L.E.; Gibson, A.A.; Stephen, A.M. Low calorie beverage consumption is associated with energy and nutrient intakes and diet quality in British adults. Nutrients 2016, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Carroll, H.A.; Betts, J.A.; Johnson, L. An investigation into the relationship between plain water intake and glycated Hb (HbA1c): A sex-stratified, cross-sectional analysis of the UK National Diet and Nutrition Survey (2008–2012). Br. J. Nutr. 2016, 116, 1770–1780. [Google Scholar] [CrossRef] [PubMed]
- Stookey, J.J.D. Negative, null and beneficial effects of drinking water on energy intake, energy expenditure, fat oxidation and weight change in randomized trials: A qualitative review. Nutrients 2016, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Mandel, E.I.; Curhan, G.C.; Hu, F.B.; Taylor, E.N. Plasma bicarbonate and risk of type 2 diabetes mellitus. CMAJ Can. Med. Assoc. J. 2012, 184, E719–E725. [Google Scholar] [CrossRef] [PubMed]
- Simental-Mendía, L.E.; Sahebkar, A.; Rodríguez-Morán, M.; Guerrero-Romero, F. A systematic review and meta-analysis of randomized controlled trials on the effects of magnesium supplementation on insulin sensitivity and glucose control. Pharmacol. Res. 2016, 111, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.S.; Heilbronn, L.K.; Chen, D.L.; Coster, A.C.F.; Greenfield, J.R.; Samocha-Bonet, D. Dietary acid load, metabolic acidosis and insulin resistance—Lessons from cross-sectional and overfeeding studies in humans. Clin. Nutr. 2016, 35, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Souto, G.; Donapetry, C.; Calviño, J.; Adeva, M.M. Metabolic acidosis-induced insulin resistance and cardiovascular risk. Metab. Syndr. Relat. Disord. 2011, 9, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Rydén, L.; Grant, P.J.; Anker, S.D.; Berne, C.; Cosentino, F.; Danchin, N.; Deaton, C.; Escaned, J.; Hammes, H.P.; Huikuri, H.; et al. ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD—Summary. Diabetes Vasc. Dis. Res. 2014, 11, 133–173. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Gutenbrunner, C. Kontrollierte Studie über die Wirkung einer Haustrinkkur mit einem Natrium-Hydrogencarbonat-Säuerling auf die Blutzuckerregulation bei gesunden Versuchspersonen. Phys. Med. Rehabil. Kurortmed. 1993, 3, 108–110. [Google Scholar] [CrossRef]
- Tate, D.F.; Turner-McGrievy, G.; Lyons, E.; Stevens, J.; Erickson, K.; Polzien, K.; Diamond, M.; Wang, X.; Popkin, B. Replacing caloric beverages with water or diet beverages for weight loss in adults: Main results of the Choose Healthy Options Consciously Everyday (CHOICE) randomized clinical trial. Am. J. Clin. Nutr. 2012, 95, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Madjd, A.; Taylor, M.A.; Delavari, A.; Malekzadeh, R.; Macdonald, I.A.; Farshchi, H.R. Effects on weight loss in adults of replacing diet beverages with water during a hypoenergetic diet: A randomized, 24-wk clinical trial. Am. J. Clin. Nutr. 2015, 102, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Madjd, A.; Taylor, M.A.; Delavari, A.; Malekzadeh, R.; Macdonald, I.A.; Farshchi, H.R. Beneficial effects of replacing diet beverages with water on type 2 diabetic obese women following a hypo-energetic diet: A randomized, 24-week clinical trial. Diabetes Obes. Metab. 2017, 19, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Klemsdal, T.O.; Landaas, S.; Høieggen, A. No effect of increased water intake on blood viscosity and cardiovascular risk factors. Br. J. Nutr. 2006, 96, 993–996. [Google Scholar] [CrossRef] [PubMed]
- Maersk, M.; Belza, A.; Stødkilde-Jørgensen, H.; Ringgaard, S.; Chabanova, E.; Thomsen, H.; Pedersen, S.B.; Astrup, A.; Richelsen, B. Sucrose-sweetened beverages increase fat storage in the liver, muscle, and visceral fat depot: A 6-mo randomized intervention study. Am. J. Clin. Nutr. 2012, 95, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Burke, V.; Zilkens, R.R.; Hodgson, J.M.; Beilin, L.J.; Puddey, I.B. The effects of alcohol on ambulatory blood pressure and other cardiovascular risk factors in type 2 diabetes: A randomized intervention. J. Hypertens. 2016, 34, 421–428; discussion 428. [Google Scholar] [CrossRef] [PubMed]
- Gepner, Y.; Golan, R.; Harman-Boehm, I.; Henkin, Y.; Schwarzfuchs, D.; Shelef, I.; Durst, R.; Kovsan, J.; Bolotin, A.; Leitersdorf, E.; et al. Effects of initiating moderate alcohol intake on cardiometabolic risk in adults with type 2 diabetes: A 2-year randomized, controlled trial. Ann. Intern. Med. 2015, 163, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Rafraf, M.; Zemestani, M.; Asghari-Jafarabadi, M. Effectiveness of chamomile tea on glycemic control and serum lipid profile in patients with type 2 diabetes. J. Endocrinol. Invesig. 2015, 38, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Schoppen, S.; Pérez-Granados, A.M.; Carbajal, Á.; Oubiña, P.; Sánchez-Muniz, F.J.; Gómez-Gerique, J.A.; Vaquero, M.P. A sodium-rich carbonated mineral water reduces cardiovascular risk in postmenopausal women. J. Nutr. 2004, 134, 1058–1063. [Google Scholar] [PubMed]
- Toxqui, L.; Vaquero, M.P. An intervention with mineral water decreases cardiometabolic risk biomarkers. A crossover, randomised, controlled trial with two mineral waters in moderately hypercholesterolaemic adults. Nutrients 2016, 8, 400. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Granados, A.M.; Navas-Carretero, S.; Schoppen, S.; Vaquero, M.P. Reduction in cardiovascular risk by sodium-bicarbonated mineral water in moderately hypercholesterolemic young adults. J. Nutr. Biochem. 2010, 21, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Schorr, U.; Distler, A.; Sharma, A.M. Effect of sodium chloride- and sodium bicarbonate-rich mineral water on blood pressure and metabolic parameters in elderly normotensive individuals: A randomized double-blind crossover trial. J. Hypertens. 1996, 14, 131–135. [Google Scholar] [PubMed]
- Zair, Y.; Kasbi-Chadli, F.; Housez, B.; Pichelin, M.; Cazaubiel, M.; Raoux, F.; Ouguerram, K. Effect of a high bicarbonate mineral water on fasting and postprandial lipemia in moderately hypercholesterolemic subjects: A pilot study. Lipids Health Dis. 2013, 12, 105. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Goto, Y.; Ito, K.; Hayasaka, S.; Kurihara, S.; Soga, T.; Tomita, M.; Fukuda, S. The consumption of bicarbonate-rich mineral water improves glycemic control. Evid.-Based Complement. Altern. Med. 2015, 2015, 824395. [Google Scholar] [CrossRef] [PubMed]
- Zemestani, M.; Rafraf, M.; Asghari-Jafarabadi, M. Chamomile tea improves glycemic indices and antioxidants status in patients with type 2 diabetes mellitus. Nutrition 2016, 32, 66–72. [Google Scholar] [CrossRef] [PubMed]
- An, R.; McCaffrey, J. Plain water consumption in relation to energy intake and diet quality among US adults, 2005–2012. J. Hum. Nutr. Diet. 2016, 29, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, D.C.; Threapleton, D.E.; Evans, C.E.L.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.; Burley, V.J. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: Systematic review and dose–response meta-analysis of prospective studies. Br. J. Nutr. 2014, 112, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; McKee, M.; Galea, G.; Stuckler, D. Relationship of soft drink consumption to global overweight, obesity, and diabetes: A cross-national analysis of 75 countries. Am. J. Public Health 2013, 103, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Roussel, R.; Fezeu, L.; Bouby, N.; Balkau, B.; Lantieri, O.; Alhenc-Gelas, F.; Marre, M.; Bankir, L.; DESIR Study Group. Low water intake and risk for new-onset hyperglycemia. Diabetes Care 2011, 34, 2551–2554. [Google Scholar] [CrossRef] [PubMed]
- Carroll, H.A.; Davis, M.G.; Papadaki, A. Higher plain water intake is associated with lower type 2 diabetes risk: A cross-sectional study in humans. Nutr. Res. 2015, 35, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Consortium, T.I. Consumption of sweet beverages and type 2 diabetes incidence in European adults: Results from EPIC-InterAct. Diabetologia 2013, 56, 1520–1530. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Popkin, B.M.; Bray, G.A.; Després, J.-P.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes: A meta-analysis. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed]
- Gijsbers, L.; Ding, E.L.; Malik, V.S.; Goede, J.D.; Geleijnse, J.M.; Soedamah-Muthu, S.S. Consumption of dairy foods and diabetes incidence: A dose-response meta-analysis of observational studies. Am. J. Clin. Nutr. 2016, 103, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Mattei, J.; Malik, V.; Hu, F.B.; Campos, H. Substituting homemade fruit juice for sugar-sweetened beverages is associated with lower odds of metabolic syndrome among Hispanic adults. J. Nutr. 2012, 142, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Baliunas, D.O.; Taylor, B.J.; Irving, H.; Roerecke, M.; Patra, J.; Mohapatra, S.; Rehm, J. Alcohol as a risk factor for type 2 diabetes: A systematic review and meta-analysis. Diabetes Care 2009, 32, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.M.; Cobb, P.; Saydah, S.; Zhang, X.; de Jesus, J.M.; Cogswell, M.E. Dietary sodium reduction does not affect circulating glucose concentrations in fasting children or adults: Findings from a systematic review and meta-analysis. J. Nutr. 2015, 145, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Bellasi, A.; Di Micco, L.; Santoro, D.; Marzocco, S.; De Simone, E.; Cozzolino, M.; Di Lullo, L.; Guastaferro, P.; Di Iorio, B.; UBI Study Investigators. Correction of metabolic acidosis improves insulin resistance in chronic kidney disease. BMC Nephrol. 2016, 17, 158. [Google Scholar] [CrossRef] [PubMed]
- Farwell, W.R.; Taylor, E.N. Serum bicarbonate, anion gap and insulin resistance in the National Health and Nutrition Examination Survey. Diabet. Med. 2008, 25, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.D.; Severo, M.; Araújo, J.R.; Guimarães, J.T.; Pestana, D.; Santos, A.; Ferreira, R.; Ascensão, A.; Magalhães, J.; Azevedo, I.; et al. Relevance of a hypersaline sodium-rich naturally sparkling mineral water to the protection against metabolic syndrome induction in fructose-fed sprague-dawley rats: A biochemical, metabolic, and redox approach. Int. J. Endocrinol. 2014, 2014, 384583. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Renin-angiotensin-aldosterone system in insulin resistance and metabolic syndrome. J. Transl. Intern. Med. 2016, 4, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Guclu, A.; Erdur, F.M.; Turkmen, K. The emerging role of sirtuin 1 in cellular metabolism, diabetes mellitus, diabetic kidney disease and hypertension. Exp. Clin. Endocrinol. Diabetes 2016, 124, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.T.; Lerin, C.; Haas, W.; Gygi, S.P.; Spiegelman, B.M.; Puigserver, P. Nutrient control of glucose homeostasis through a complex of PGC-1α and SIRT1. Nature 2005, 434, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.D.; Severo, M.; Neves, D.; Ascensão, A.; Magalhães, J.; Guimarães, J.T.; Monteiro, R.; Martins, M.J. Natural mineral-rich water ingestion improves hepatic and fat glucocorticoid-signaling and increases sirtuin 1 in an animal model of metabolic syndrome. Horm. Mol. Biol. Clin. Investig. 2015, 21, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.D.; Passos, E.; Severo, M.; Vitó, I.; Wen, X.; Carneiro, F.; Gomes, P.; Monteiro, R.; Martins, M.J. Ingestion of a natural mineral-rich water in an animal model of metabolic syndrome: Effects in insulin signaling and endoplasmic reticulum stress. Horm. Mol. Biol. Clin. Investig. 2016, 26, 135–150. [Google Scholar] [CrossRef] [PubMed]
- Gommers, L.M.M.; Hoenderop, J.G.J.; Bindels, R.J.M.; de Baaij, J.H.F. Hypomagnesemia in type 2 diabetes: A vicious circle? Diabetes 2016, 65, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Simental-Mendía, L.E.; Hernández-Ronquillo, G.; Rodriguez-Morán, M. Oral magnesium supplementation improves glycaemic status in subjects with prediabetes and hypomagnesaemia: A double-blind placebo-controlled randomized trial. Diabetes Metab. 2015, 41, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C.; Krüger, K.; Völker, K.; Golf, S.W.; Wadepuhl, M.; Kraus, A. Oral magnesium supplementation reduces insulin resistance in non-diabetic subjects—A double-blind, placebo-controlled, randomized trial. Diabetes Obes. Metab. 2011, 13, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Lecube, A.; Baena-Fustegueras, J.A.; Fort, J.M.; Pelegri, D.; Hernandez, C.; Simo, R. Diabetes is the main factor accounting for hypomagnesemia in obese subjects. PLoS ONE 2012, 7, e30599. [Google Scholar] [CrossRef] [PubMed]
- Kieboom, B.C.T.; Ligthart, S.; Dehghan, A.; Kurstjens, S.; de Baaij, J.H.F.; Franco, O.H.; Hofman, A.; Zietse, R.; Stricker, B.H.; Hoorn, E.J. Serum magnesium and the risk of prediabetes: A population-based cohort study. Diabetologia 2017, 60, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Xun, P.; He, K.; Qin, L.-Q. Magnesium intake and risk of type 2 diabetes: Meta-analysis of prospective cohort studies. Diabetes Care 2011, 34, 2116–2122. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Han, H.; Li, M.; Liang, C.; Fan, Z.; Aaseth, J.; He, J.; Montgomery, S.; Cao, Y. Dose-response relationship between dietary magnesium intake and risk of type 2 diabetes mellitus: A systematic review and meta-regression analysis of prospective cohort studies. Nutrients 2016, 8, 739. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; Fly, A.D.; Yokota, K.; He, K. Dietary magnesium intake and risk of metabolic syndrome: A meta-analysis. Diabet. Med. 2014, 31, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C. Magnesium and disturbances in carbohydrate metabolism. Diabetes Obes. Metab. 2015, 17, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Ha, B.G.; Park, J.-E.; Shin, E.J.; Shon, Y.H. Modulation of glucose metabolism by balanced deep-sea water ameliorates hyperglycemia and pancreatic function in streptozotocin-induced diabetic mice. PLoS ONE 2014, 9, e102095. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; He, K. Dietary magnesium intake is inversely associated with serum C-reactive protein levels: Meta-analysis and systematic review. Eur. J. Clin. Nutr. 2014, 68, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, T.A.; Xiang, A.H.; Peters, R.K.; Kjos, S.L.; Marroquin, A.; Goico, J.; Ochoa, C.; Tan, S.; Berkowitz, K.; Hodis, H.N.; et al. Preservation of pancreatic beta-cell function and prevention of type 2 diabetes by pharmacological treatment of insulin resistance in high-risk hispanic women. Diabetes 2002, 51, 2796–2803. [Google Scholar] [CrossRef] [PubMed]
- Defronzo, R.A.; Tripathy, D.; Schwenke, D.C.; Banerji, M.; Bray, G.A.; Buchanan, T.A.; Clement, S.C.; Henry, R.R.; Kitabchi, A.E.; Mudaliar, S.; et al. Prediction of diabetes based on baseline metabolic characteristics in individuals at high risk. Diabetes Care 2013, 36, 3607–3612. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Magnesium improves the beta-cell function to compensate variation of insulin sensitivity: Double-blind, randomized clinical trial. Eur. J. Clin. Investig. 2011, 41, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.V.; Hocher, B.; Verkaart, S.; van Zeeland, F.; Pfab, T.; Slowinski, T.; Chen, Y.-P.; Schlingmann, K.P.; Schaller, A.; Gallati, S.; et al. Loss of insulin-induced activation of TRPM6 magnesium channels results in impaired glucose tolerance during pregnancy. Proc. Natl. Acad. Sci. USA 2012, 109, 11324–11329. [Google Scholar] [CrossRef] [PubMed]
| Search No. | Search Terms | Results | Search Fields |
|---|---|---|---|
| 1 | water | 819,017 | All Fields |
| 2 | glucose or glycemic or glycaemic or cholesterol | 733,923 | All Fields |
| 3 | drink* or consumption or beverage* | 188,224 | All Fields |
| 4 | study or trial* or review | 10,225,821 | All Fields |
| 5 | humans or women or men | 16,761,909 | All Fields |
| 6 | ((((water) AND ((glucose OR glycemic OR glycaemic OR cholesterol))) AND ((drink* OR consumption OR beverage*))) AND ((study OR trial* OR review))) AND (humans OR women OR men) | 1139 | All Fields |
| Parameters (with Normal Values) for Glycemic Control Reported in the Studies |
|---|
| Fasting plasma glucose (FPG < 110 mg/dL) |
| Glucose tolerance test (level 2 h after glucose loading < 140 mg/dL) |
| Fasting insulin (6–25 mU/L) |
| HOMA-IR (<2) (Homeostatic Model Assessment for Insulin Resistance) |
| (Product of fasting insulin (µU/mL) × fasting glucose (mg/dL))/405) |
| Glycated hemoglobin, HbA1c (<6%) |
| (Measure for blood glucose levels during the last 1 to 3 months) |
| Glycoalbumin (11–16%) |
| (Measure for blood glucose levels in the last 2 to 4 weeks) |
| Fructosamin (200–290 µmol/L) |
| (Measure for blood glucose levels in the last 1 to 3 weeks) |
| Author/y | Study Type | Population | Intervention | Comparison | Outcome | Results | Risk of Bias |
|---|---|---|---|---|---|---|---|
| Tate et al., 2012 [17] | RCT parallel 3 groups | 318 obese, BMI 36.3 kg/m2, 84% female, medium age 42 y, USA | Advice to replace more than 2 servings (each 350–500 mL) of caloric beverages per day with water for six months | CG1: Advice to replace more than 2 servings of caloric beverages per day with diet beverages CG2: no advice to change beverages, but general nutritional weight loss advices | FPG (SO) Weight loss (PO) | Water group had a sign. Increase of water consumption of 1 L after 3 months and 0.8 L after 6 months and a sign. Decrease of FPG compared to control group with no change of beverages after 3 and 6 months. | ITT, drop-outs 33/318 after 3 months and 46/318 after 6 months, no blinding |
| Madjd et al., 2015 [18] | RCT parallel 2 groups single-blind | 62 healthy overweight and obese women, BMI 27–40 kg/m2, 18–50 y, non-smokers, participating in a weight loss program, Iran | 250 mL tap water (not specified) per day after lunch for 24 weeks | 250 mL diet beverage | FPG Fasting insulin HOMA-IR GTT Body weight (all PO) | Water group sign. more weight loss (8.8 kg vs. 7.6 kg p = 0.015) lower FPG (2.8 mU/L vs. 1.7 mU/L p < 0.001) better insulin sensitivity (HOMA-IR 1 vs. 0.8 p < 0.001) better GTT (1 mmol/L vs. 0.7 mmol/L, p < 0.001) compared to control group after 24 weeks. | No ITT, 9/71 drop-outs, measures of blinding not reported |
| Madjd et al., 2017 [19] | RCT parallel 2 groups single-blind | 81 overweight and obese women, BMI 27–35 kg/m2 with type 2 diabetes HbA1c 6.5–7.2%, age 18–50 y, non-smokers, participating in a weight loss program with only metformin, Iran | 250 mL tap water (not specified) per day after lunch for 24 weeks | 250 mL diet beverage | FPG Fasting insulin HOMA-IR GTT Body weight (all PO) | Water group sign. more weight loss (−6.40 vs. −5.25 kg p = 0.006); lower FPG (−1.63 vs. −1.29 mmol/L p = 0.06) lower fasting insulin (−5.71 vs. −4.16 mU/L p < 0.011) better insulin sensitivity (HOMA-IR −3.2 vs. −2.48 p < 0.003) better GTT (−1.67 vs. −1.35 mmol/L p = 0.027) Compared to the control group after 24 weeks. | ITT, 16/81 drop-outs, measures of blinding not reported |
| Tonstad et al., 2006 [20] | RCT parallel 3 groups | 67 men and 27 post-menopausal women (total n = 94) with at least 1 CVRF, Norway | 1 L tap water (not specified) per day for 4 weeks | CG1: 1 L blueberry juice CG2: no change of habits | FPG Fasting insulin C-peptide (all SO) Blood viscosity (PO) | No sign. difference between groups after 4 weeks (exact data not presented) | No ITT, 5/99 drop-outs, no blinding, |
| Maersk et al., 2012 [21] | RCT parallel 2 groups | 47 healthy overweight adults, 20–50 y, BMI 26–40 kg/m2, RR < 160/100, Denmark | 1 L uncarbonated water per day for 6 months Aqua d’or HCO3− 71 mg/L Cl− 16.5 mg/L Na+ 14.9 mg/L Mg2+ 6 mg/L | 1 L regular Cola (sucrose-sweetened) 1 L diet Cola, aspartame-sweetened 1 L semi-skim milk | FPG Fasting insulin HOMA-IR (all SO) | No sign. difference for FPG Insulin and HOMA-IR between groups | No ITT, 13/60 drop-outs, no blinding, sign. difference for sex at baseline (adjusted in analysis), randomization and allocation not reported |
| Mori et al., 2016 [22] | RCT crossover 3 groups | 24 well controlled type 2 diabetes HbA1c < 8.5%, 19 men, 5 women, 40–70 y, regular alcohol intake women 20–30 g/day, men 30–40 g/day, Australia | Tap water (not specified) women 230 mL per day men 300 mL per day for 4 weeks | CG1: red wine women 230 mL/day (24 g alcohol/day) Men 300 mL/day (31 g alcohol/day), CG2: dealcoholized red wine women 230 mL/day Men 300 mL/day | FPG Fasting Insulin HOMA-IR (all SO) CVRF (PO) | No sign. difference for FPG Insulin and HOMA-IR between groups | No ITT, 4/28 drop-outs, no blinding |
| Gepner et al., 2015 [23] | RCT parallel 3 groups | 224 alcohol-abstaining adults type 2 diabetes, HbA1c 6.4–10%, Israel | 150 mL mineral water) per day in the evening for 2 years Mey Eden, different European sources, all low mineralized | CG1: 150 mL red wine CG2: 150 mL white wine | FPG HOMA-IR (all PO) Fasting insulin HbA1c (all SO) HDL-Chol, Apolip-a (PO) | Water group sign. Increase of FPG and HOMA-IR compared to white wine. No sign. difference for HbA1c, fasting insulin and outcomes compared to red wine | ITT, no blinding |
| Rafraf et al., 2015 [24] & Zemestani et al., 2016 [31] | RCT parallel 2 groups single-blind | 64 type 2 diabetes (males and females) aged 30–60 y Iran | 150 mL hot water three times per day immediately after meals for 8 weeks. | 150 mL chamomile tea (3 g/150 mL hot water) | FPG Fasting Insulin HOMA-IR HbA1c | Chamomile tea group sign. Decrease for HbA1c (p = 0.023), serum insulin levels (p < 0.001), HOMA-IR (p < 0.001), and no sign. difference for FPG compared to hot water | ITT, no drop-outs, single-blind, sign. difference for FPG, insulin and HOMA-IR at baseline, no PO specified |
| Author/y | Study Type | Population | Intervention | Comparison | Outcome | Results | Risk of Bias |
|---|---|---|---|---|---|---|---|
| Schoppen et al., 2004 [25] | RCT crossover 2 groups | 18 healthy women, >1 postmenopausal, BMI < 30 kg/m2, Spain | 1 L per day for 2 months Bicarbonate-rich carbonated water: HCO3− 2094 mg/L Cl− 583 mg/L Na+ 1116 mg/L Mg2+ 6 mg/L | Uncarbonated water: HCO3− 71 mg/L Cl− 6 mg/L Na+ 9 mg/L Mg2+ 3 mg/L | FPG (SO) CVRF (PO) | Bicarbonate rich water sign. (p < 0.001) decreased FPG compared to control water (5.54 vs. 5.17 mmol/L) | ITT, 0/18 drop-outs, no blinding, randomization and allocation not reported |
| Toxqui & Vaquero, 2016 [26] | RCT crossover 2 groups single-blind | 64 healthy adults 18–45 y, with moderately elevated cholesterol (5.2–7.8 mmol/L), Spain | 1 L of the test water per day for 8 weeks followed by an 8-week washout period Bicarbonate-rich carbonated water: HCO3− 2050 mg/L Cl− 622 mg/L Na+ 1090 mg/L Mg2+ 5.8 mg/L | Uncarbonated water: HCO3− 75 mg/L Cl− 4.8 mg/L Na+ 7.6 mg/L Mg2+ 2.8 mg/L | FPG Fasting insulin (all SO) Cholesterol (PO) | No sign. difference for FPG and fasting insulin between groups Sign. reduction of fasting glucose and LDL—cholesterol compared to baseline in both groups. | No ITT, 8/72 drop-outs, no blinding, |
| Pérez-Granados et al., 2010 [27] | RCT crossover 2 groups single-blind | 18 healthy adults 18–40 y, with moderately elevated cholesterol (>5.2 mmol/L), Spain | 1 L of the test water per day for 8 weeks followed by an 8-week washout period Bicarbonate-rich carbonated water: HCO3− 2120 mg/L Cl− 597 mg/L Na+ 1102 mg/L Mg2+ 9.4 mg/L | Carbonated water: HCO3− 104 mg/L Cl− 11 mg/L Na+ 8.7 mg/L Mg2+ 5.0 mg/L | FPG Fasting insulin (all SO) Cholesterol (PO) | Bicarbonate-rich water decreased FPG not sign. (p = 0.056) and no sign. difference for fasting insulin compared to control water. | No ITT, 10/28 drop-outs, single blind, randomization and allocation not reported |
| Schorr et al., 1996 [28] | RCT crossover 3 groups double-blind | 16 healthy adults 60–72 y, salt reduced diet (<2.3 g/day) Germany | 1.5 L of the test water per day for 4 weeks with a 2-week washout period before the control water Bicarbonate-rich water: (1.) HCO3− 879 mg/L Cl− 1507 mg/L Na+ 1295 mg/L Mg2+ 9.7 mg/L (2.) HCO3− 1983 mg/L Cl− 152 mg/L Na+ 602 mg/L Mg2+ 53 mg/L | Uncarbonated water: HCO3− < 5 mg/L Cl− < 5 mg/L Na+ < 5 mg/L Mg2+ < 1 mg/L | FPG Fasting insulin GTT (all PO) | No sign. difference between the groups. | No ITT, 5/21 drop-outs |
| Zair et al., 2013 [29] | RCT crossover 2 groups double-blind | 12 healthy men, 20–60 y, BMI 18.5–25 kg/m2, Cholesterol 2.2–3 g/L, France | 1.25 L per day for 8 weeks Bicarbonate-rich carbonated water: HCO3− 4168 mg/L Cl− 329 mg/L Na+ 1626 mg/L Mg2+ 11 mg/L | Carbonated water: HCO3− 183 mg/L Cl− 48 mg/L Na+ 31 mg/L Mg2+ 12 mg/L | FPG (SO) Blood lipids (PO) | No sign. difference between the groups for FPG. | ITT, 0/12 drop-outs, randomization and allocation not reported, blinding difficult due to taste of water |
| Author/y | Study Type | Population | Intervention | Comparison | Outcome | Results | Risk of Bias |
|---|---|---|---|---|---|---|---|
| Gutenbrunner, 1993 [16] | RCT parallel 2 groups single-blind | 23 healthy men, 19–31 y, Germany | 1.4 L per day of the test waters for 28 days Bicarbonate and magnesium-rich carbonated water: HCO3− 2946 mg/L Cl− 128 mg/L Na+ 967 mg/L Mg2+ 102 mg/L | Uncarbonated water: HCO3− 150 mg/L Na+ 19.8 mg/L Mg not mentioned | FPG Fasting insulin GTT Fructosamin (all PO) | Bicarbonate and magnesium-rich water sign. decreased glucose tolerance and fructosamin compared to control water, but not FPG and fasting insulin. | No ITT, 1/24 drop-outs, single blind, randomization and allocation not reported |
| Murakami et al., 2015 [30] | RCT parallel 2 groups | 19 healthy (7 men, 12 women), 47 y (26–59 y), Japan | 500 mL per day premeal, mineral or control water for one week in two cycles. The intervention lasted 4 weeks Bicarbonate and magnesium-rich uncarbonated water: HCO3− 2485 mg/L Cl− 182 mg/L Na+ 412 mg/L Mg2+ 291 mg/L | Uncarbonated water: HCO3− 28 mg/L Cl− 11 mg/L Na+ 10 mg/L Mg2+ 1.9 mg/L | FPG Glycoalbumin, Fasting insulin HOMA-IR (all PO) Microbiome | Bicarbonate and magnesium-rich water sign. decreased glycoalbumin compared to control water. No sign. differences for FPG and fasting insulin. Lean-inducible bacteria was increased | No ITT, 7/26 drop-outs, no blinding |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naumann, J.; Biehler, D.; Lüty, T.; Sadaghiani, C. Prevention and Therapy of Type 2 Diabetes—What Is the Potential of Daily Water Intake and Its Mineral Nutrients? Nutrients 2017, 9, 914. https://doi.org/10.3390/nu9080914
Naumann J, Biehler D, Lüty T, Sadaghiani C. Prevention and Therapy of Type 2 Diabetes—What Is the Potential of Daily Water Intake and Its Mineral Nutrients? Nutrients. 2017; 9(8):914. https://doi.org/10.3390/nu9080914
Chicago/Turabian StyleNaumann, Johannes, Diana Biehler, Tania Lüty, and Catharina Sadaghiani. 2017. "Prevention and Therapy of Type 2 Diabetes—What Is the Potential of Daily Water Intake and Its Mineral Nutrients?" Nutrients 9, no. 8: 914. https://doi.org/10.3390/nu9080914
APA StyleNaumann, J., Biehler, D., Lüty, T., & Sadaghiani, C. (2017). Prevention and Therapy of Type 2 Diabetes—What Is the Potential of Daily Water Intake and Its Mineral Nutrients? Nutrients, 9(8), 914. https://doi.org/10.3390/nu9080914





