Omega-3 Fatty Acids and Cardiovascular Disease: Summary of the 2016 Agency of Healthcare Research and Quality Evidence Review
Abstract
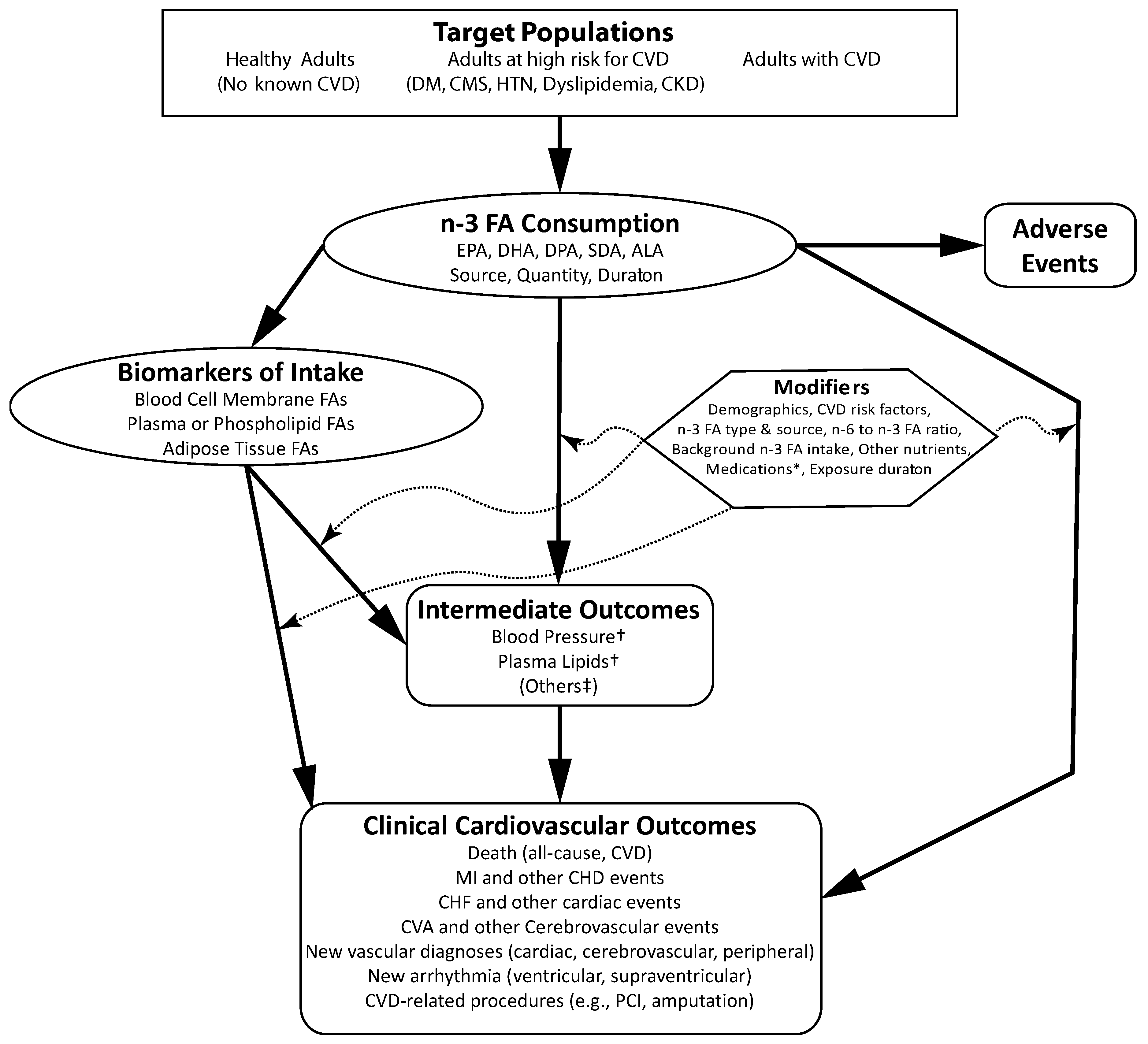
:1. Introduction
2. Materials and Methods
3. Results
3.1. Key Question 1: Efficacy or Association of n-3 FA and CVD Outcomes or Risk Factors
3.1.1. Total n-3 FA
3.1.2. Marine Oils
3.1.3. Specific Marine Oils
3.1.4. Sub-Questions
1. People with No Known CVD, At Increased Risk for CVD, and With Known CVD.
2. Relative Effect of Different n-3 FAs.
3. Ordering of n-3 FAs by Strength of Effect.
3.2. Key Question 2: n-3 FA Variables and Modifiers
Sub-Questions
1. Subpopulations
2. Confounders or Interacting Factors
3–6. Different Ratios of n-3 FA Components, Different n-3 FA Sources, and n-6 FA to n-3 FA Ratio
7. Threshold or Dose–Response Relationship
8. Duration of Intervention or Exposure
9. Effect of Baseline n-FA Status
3.3. Adverse Events
Sub-Questions
1. Adverse Events across All Studies
2. Adverse Events among People with CVD or Diabetes
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- Scientific Report of the 2015 Dietary Guidelines Advisory Committee. 2015. Available online: http://health.gov/dietaryguidelines/2015-scientific-report/pdfs/scientific-report-of-the-2015-dietary-guidelines-advisory-committee.pdf (accessed on 7 August 2017).
- Institute of Medicine. Dietary Reference Intakes. Energy, Carbohydrate, Fiber, Fat Fatty Acids, Cholesterol, Protein and Amino Acids; National Academy of Sciences: Washington, DC, USA, 2005. [Google Scholar]
- Dietary Guidelines for Americans, 2010. Health. gov. Available online: https://health.gov/dietaryguidelines/dga2010/DietaryGuidelines2010.pdf (accessed on 7 August 2017).
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; Van Hubbard, S.; de Jesus, J.M.; Lee, I.-M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; Miller, N.H.; et al. 2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 3027–3028. [Google Scholar] [CrossRef] [PubMed]
- Rehm, C.D.; Peñalvo, J.L.; Afshin, A.; Mozaffarian, D. Dietary Intake among US Adults, 1999–2012. JAMA 2016, 315, 2542–2553. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Chung, M.; Balk, E.; Kupelnick, B.; DeVine, D.; Lawrence, A.; Lichtenstein, A.; Lau, J. Effects of Omega-3 Fatty Acids on Cardiovascular Disease; Evidence Report/Technology Assessment No. 94 AHRQ Publication No. 04-E009-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Balk, E.; Chung, M.; Lichtenstein, A.; Chew, P.; Kupelnick, B.; Lawrence, A.; DeVine, D.; Lau, J. Effects of Omega-3 Fatty Acids on Cardiovascular Risk Factors and Intermediate Markers of Cardiovascular Disease; Evidence Report/Technology Assessment No. 93 AHRQ Publication No. 04-E010-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. n-3 Fatty acids from fish or fish-oil supplements, but not alpha-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar] [PubMed]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: A systematic review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on coronary restenosis, intima-media thickness, and exercise tolerance: A systematic review. Atherosclerosis 2006, 184, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Cigarette Smoking Among Adults—United States, 2005–2015. Morb. Mortal. Wkly. Rep. 2016, 65, 1205–1211. [Google Scholar]
- Flegal, K.M.; Carroll, M.D.; Kit, B.K.; Ogen, C.L. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA 2012, 307, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Adam, G.P.; Langberg, V.; Halladay, C.; Chung, M.; Lin, L.; Robertson, S.; Yip, A.; Steele, D.; Smith, B.T.; Lau, J.; et al. Omega-3 Fatty Acids and Cardiovascular Disease: An Updated Systematic Review; Evidence Report/Technology Assessment No. 223 AHRQ Publication No. 16-E002-EF; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2016.
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Nissensohn, M.; Øverby, N.C.; Fekete, K. Methods of assessment of n-3 long-chain polyunsaturated fatty acid status in humans: A systematic review. Am. J. Clin. Nutr. 2009, 89, 2070S–2084S. [Google Scholar]
- Serra-Majem, L.; Nissensohn, M.; Øverby, N.C.; Fekete, K. Dietary methods and biomarkers of omega 3 fatty acids: A systematic review. Br. J. Nutr. 2012, 107 (Suppl. 2), S64–S76. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M. Triglycerides and Triglyceride-Rich Lipoproteins in the Causal Pathway of Cardiovascular Disease. Am. J. Cardiol. 2016, 118, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G. Triglyceride-Rich Lipoproteins and Atherosclerotic Cardiovascular Disease: New Insights from Epidemiology, Genetics, and Biology. Circ. Res. 2016, 118, 547–563. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Heath and Care Excellence. Cardiovascular Disease: Risk Assessment and Reduction, Including Lipid Modification, 2014. National Institute for Health and Care Excellence. Available online: https://www.nice.org.uk/guidance/CG181 (accessed on 27 July 2017).
| Key Question | Question Text |
|---|---|
| 1 | What is the efficacy or association of n-3 FA (EPA, DHA, EPA+DHA, DPA, SDA, ALA, or total n-3 FA) exposures in reducing CVD outcomes (incident CVD events, including all-cause death, CVD death, nonfatal CVD events, new diagnosis of CVD, peripheral vascular disease, CHF, major arrhythmias, and hypertension diagnosis) and specific CVD risk factors (BP, key plasma lipids)? |
| 1.1 | What is the efficacy or association of
n-3 FA in preventing CVD outcomes in people • Without known CVD (primary prevention) • At high risk for CVD (primary prevention), and • With known CVD (secondary prevention)? |
| 1.2 | What is the relative efficacy of different n-3 FA on CVD outcomes and risk factors? |
| 1.3 | Can the CVD outcomes be ordered by strength of intervention effect of n-3 FA? |
| 2 | n-3 FA variables and modifiers: |
| 2.1 | How does the efficacy or association of n-3 FA in preventing CVD outcomes and with CVD risk factors differ in subpopulations, including men, premenopausal women, postmenopausal women, and different age or race/ethnicity groups? |
| 2.2 | What are the effects of potential confounders or interacting factors—such as plasma lipids, body mass index, BP, diabetes, kidney disease, other nutrients or supplements, and drugs (e.g., statins, aspirin, diabetes drugs, hormone replacement therapy)? |
| 2.3 | What is the efficacy or association of different ratios of n-3 FA components in dietary supplements or biomarkers on CVD outcomes and risk factors? |
| 2.4 | How does the efficacy or association of n-3 FA on CVD outcomes and risk factors differ by ratios of different n-3 FA—DHA, EPA, and ALA, or other n-3 FA? |
| 2.5 | How does the efficacy or association of n-3 FA on CVD outcomes and risk factors differ by source (e.g., fish and seafood, common plant oils (e.g., soybean, canola), fish oil supplements, fungal-algal supplements, flaxseed oil supplements)? |
| 2.6 | How does the ratio of n-6 FA to n-3 FA intakes or biomarker concentrations affect the efficacy or association of n-3 FA on CVD outcomes and risk factors? |
| 2.7 | Is there a threshold or dose–response relationship between n-3 FA exposures and CVD outcomes and risk factors? Does the study type affect these relationships? |
| 2.8 | How does the duration of intervention or exposure influence the effect of n-3 FA on CVD outcomes and risk factors? |
| 2.9 | What is the effect of baseline n-3 FA status (intake or biomarkers) on the efficacy of n-3 FA intake or supplementation on CVD outcomes and risk factors? |
| 3 | Adverse events: |
| 3.1 | What adverse effects are related to n-3 FA intake (in studies of CVD outcomes and risk factors)? |
| 3.2 | What adverse events are reported specifically among people with CVD or diabetes (in studies of CVD outcomes and risk factors)? |
| Effect or Association | Strength of Evidence | Finding | Study Types | Effect Sizes |
|---|---|---|---|---|
| Higher n-3 FA intake or biomarker levels with lower CVD risks or events | High | Marine Oil * Supplementation (Or Increased Intake) Raises HDL-C | RCTs (of mostly supplements) | Summary net change in HDL-C: 0.9 mg/dL (95% CI 0.2, 1.6) |
| High | Marine oil supplementation (or increased intake) lowers Tg | RCTs (of mostly supplements) | Summary net change in Tg: −24 mg/dL (95% CI −31, −18) | |
| High | Marine oil supplementation (or increased intake) lowers TC/HDL-C ratio | RCTs (of mostly supplements) | Summary net change in TC/ HDL-C ratio: −0.17 (95% CI −0.26, −0.09) | |
| Low | Marine oil increased intake lowers risk of ischemic stroke | Observational studies (of total dietary intake) | By metaregression: 0.51 (95% CI 0.29, 0.89) per g/day | |
| Higher n-3 FA intake or biomarker levels with higher CVD risk | High | Marine Oil Supplementation (Or Increased Intake) Raises LDL-C | RCTs (of mostly supplements) | Summary net change in LDL-C: 2.0 mg/dL (95% CI 0.4, 3.6) |
| Strength of Evidence | Omega-3 Fatty Acid and Outcome | Study Types | Summary Effect Sizes |
|---|---|---|---|
| High | Marine oil* supplementation (or increased intake) and MACE | RCTs (of mostly supplements), supported by observational studies (of total dietary intake) | RCTs: 0.96 (95% CI 0.91, 1.02) |
| High | Marine oil intake and all-cause death | RCTs (of mostly supplements) and observational studies (of total dietary intake) | RCTs: 0.97 (95% CI 0.92, 1.03). Observational studies: 0.62 (95% CI 0.31, 1.25) per g/day |
| High | Marine oil intake and SCD | RCTs (of mostly supplements), supported by observational studies (of total dietary intake) | RCTs: 1.04 (95% CI 0.92, 1.17) |
| High | Marine oil intake and coronary revascularization | RCTs (of mostly supplements), supported by observational studies (of total dietary intake) | Not significant, not meta-analyzed |
| High | Marine oil intake and systolic or diastolic blood pressure | RCTs (of mostly supplements) | RCTs: summary net change in systolic blood pressure: 0.1 mg/dL (95% CI −0.2, 0.4); summary net change in diastolic blood pressure: −0.2 mg/dL (95% CI −0.4, 0.5) |
| Moderate | Marine oil intake and atrial fibrillation | RCTs (of mostly supplements) and observational studies (of total dietary intake) | Not significant, not meta-analyzed. Observational studies were inconsistent. |
| Moderate | Purified DHA supplementation and systolic or diastolic blood pressure | RCTs (of supplements) | Not significant, not meta-analyzed |
| Moderate | Purified DHA supplementation and LDL-C | RCTs (of supplements) | Not significant, not meta-analyzed |
| Moderate | ALA intake and systolic or diastolic blood pressure | RCTs (of mostly supplements) | Not significant, not meta-analyzed |
| Moderate | ALA intake and LDL-C, HDL-C, and Tg | RCTs (of mostly supplements) | Not significant, not meta-analyzed |
| Low | Total n-3 FA intake and stroke death | Observational studies (of total dietary intake and biomarkers) | Not significant, not meta-analyzed |
| Low | Total n-3 FA intake and myocardial infarction | Observational studies (of total dietary intake) | Not significant, not meta-analyzed |
| Low | Marine oil intake and CVD death | RCTs (of mostly supplements) and observational studies (of total dietary intake) | RCTs: 0.92 (95% CI 0.82, 1.02). Observational studies: 0.88 (95% CI 0.82, 0.95) per g/day |
| Low | Marine oil intake and CHD death | RCTs (of mostly supplements) and observational studies (of total dietary intake) | RCTs imprecise. Observational studies: 1.09 (95% CI 0.76, 1.57) per g/day |
| Low | Marine oil intake and CHD | Observational studies (of total dietary intake and biomarkers) | Observational studies: 0.94 (95% CI 0.81, 1.10) per g/day |
| Low | Marine oil intake and myocardial infarction | RCTs (of mostly supplements) | RCTs: 0.88 (95% CI 0.77, 1.02) |
| Low | Marine oil intake and angina pectoris | RCTs (of mostly supplements) | Not significant, not meta-analyzed |
| Low | Marine oil intake and CHF | RCTs (of mostly supplements) and observational studies (of total dietary intake) | RCTs not significant, not meta-analyzed. Observational studies: 0.76 (95% CI 0.58, 1.00) per g/day |
| Low | Marine oil intake and total stroke (fatal and nonfatal ischemic and hemorrhagic stroke) | RCTs (of mostly supplements) and observational studies (of total dietary intake) | RCTs: 0.97 (95% CI 0.83, 1.13). Observational studies: 0.68 (95% CI 0.53, 0.87) per g/day |
| Low | Marine oil intake and hemorrhagic stroke | Observational studies (of total dietary intake) | Observational studies: 0.61 (95% CI 0.34, 1.11) per g/day |
| Low | EPA intake and CHD | Observational studies (of total dietary intake) | Not significant, not meta-analyzed |
| Low | EPA biomarkers and atrial fibrillation | Observational studies (of biomarkers) | Not significant, not meta-analyzed |
| Low | DHA intake and CHD | Observational studies (of total dietary intake and biomarkers) | Not significant, not meta-analyzed |
| Low | DPA biomarkers and atrial fibrillation | Observational studies (of biomarkers) | Not significant, not meta-analyzed |
| Low | ALA intake and CHD death | Observational studies (of total dietary intake), supported by RCT (of supplementation) and observational study (of biomarkers) | Observational studies: 0.94 (95% CI 0.85, 1.03) per g/day |
| Low | ALA intake and CHD | Observational studies (of total dietary intake) | Observational studies: 0.97 (95% CI 0.92, 1.03) per g/day |
| Low | ALA intake and atrial fibrillation | Observational studies (of total dietary intake and biomarkers) | Not significant, not meta-analyzed |
| Low | ALA intake and CHF | Observational studies (of total dietary intake and biomarkers), supported by RCT (of supplementation) | Not significant, not meta-analyzed |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balk, E.M.; Lichtenstein, A.H. Omega-3 Fatty Acids and Cardiovascular Disease: Summary of the 2016 Agency of Healthcare Research and Quality Evidence Review. Nutrients 2017, 9, 865. https://doi.org/10.3390/nu9080865
Balk EM, Lichtenstein AH. Omega-3 Fatty Acids and Cardiovascular Disease: Summary of the 2016 Agency of Healthcare Research and Quality Evidence Review. Nutrients. 2017; 9(8):865. https://doi.org/10.3390/nu9080865
Chicago/Turabian StyleBalk, Ethan M., and Alice H. Lichtenstein. 2017. "Omega-3 Fatty Acids and Cardiovascular Disease: Summary of the 2016 Agency of Healthcare Research and Quality Evidence Review" Nutrients 9, no. 8: 865. https://doi.org/10.3390/nu9080865
APA StyleBalk, E. M., & Lichtenstein, A. H. (2017). Omega-3 Fatty Acids and Cardiovascular Disease: Summary of the 2016 Agency of Healthcare Research and Quality Evidence Review. Nutrients, 9(8), 865. https://doi.org/10.3390/nu9080865





