The Role of AOPP in Age-Related Bone Loss and the Potential Benefits of Berry Anthocyanins
Abstract
:1. Introduction
2. Aging-Associated Osteoporosis
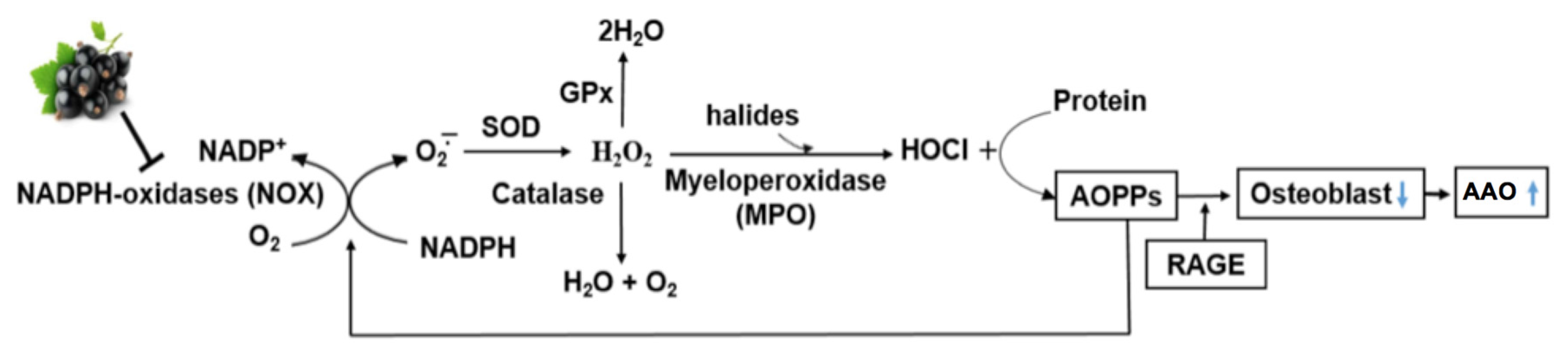
3. AOPP as a Marker of Oxidative Stress in Bone
4. Mechanisms of Action of AOPP in Bone
5. Antioxidant Intake and Bone Health: Potential Benefits of Berry Anthocyanins
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Yamagishi, S. Role of Advanced Glycation End Products (AGEs) in Osteoporosis in Diabetes. Curr. Drug Targets 2011, 12, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, P.; Constantinescu, F. A Review of Osteoporosis in the Older Adult. Clin. Geriatr. Med. 2017, 33, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Raisz, L.G. Science in medicine Pathogenesis of osteoporosis: Concepts, conflicts, and prospects. J. Clin. Investig. 2005, 115, 3318–3325. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.H.; Zhong, Z.M.; Li, X.D.; Wu, Q.; Zheng, S.; Zhou, J.; Ye, W.B.; Xie, F.; Wu, X.H.; Huang, Z.P.; et al. Advanced oxidation protein products accelerate bone deterioration in aged rats. Exp. Gerontol. 2014, 50, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Duque, G.; Troen, B.R. Understanding the mechanisms of senile osteoporosis: New facts for a major geriatric syndrome. J. Am. Geriatr. Soc. 2008, 56, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.H.; Lee, H.C. Oxidative Stress, Mitochondrial DNA Mutation, and Impairment of Antioxidant Enzymes in Aging. Exp. Biol. Med. 2002, 227, 671–682. [Google Scholar] [CrossRef]
- Wauquier, F.; Leotoing, L.; Coxam, V.; Guicheux, J.; Wittrant, Y. Oxidative stress in bone remodelling and disease. Trends Mol. Med. 2009, 15, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, A.M.; Chir, B. Bone remodeling and bone loss: Understanding the pathophysiology of osteoporosis. Clin. Obstet. Gynecol. 1987, 30, 789–811. [Google Scholar] [CrossRef] [PubMed]
- Atashi, F.; Modarressi, A.; Pepper, M.S. The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: A review. Stem Cells Dev. 2015, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- Garrett, I.R.; Boyce, B.F.; Oreffo, R.O.C.; Bonewald, L.; Poser, J.; Mundy, G.R. Oxygen-derived free radicals stimulate osteoclastic bone resorption in rodent bone in vitro and in vivo. J. Clin. Investig. 1990, 85, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Katsumata, S.; Matsuxaki, H.; Suzuki, K. Dietary zinc deficiency induces oxidative stress and promotes tumor necrosis factor-α- and interleukin-1β-induced RANKL expressio in rat bone. J. Clin. Biochem. Nutr. 2016, 58, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Han, L.; Martin-Millan, M.; Plotkin, L.I.; Stewart, S.A.; Roberson, P.K.; Kousteni, S.; O’Brien, C.A.; Bellido, T.; Parfitt, A.M.; et al. Skeletal Involution by Age-associated Oxidative Stress and Its Acceleration by Loss of Sex Steroids. J. Biol. Chem. 2007, 282, 27285–27297. [Google Scholar] [CrossRef] [PubMed]
- Hazen, S.L.; Hsu, F.F.; Heinecke, J.W. p-Hydroxyphenylacetaldehyde Is the Major Product of l-Tyrosine Oxidation by Activated Human Phagocytes. J. Biol. Chem. 1996, 271, 1861–1867. [Google Scholar] [CrossRef] [PubMed]
- Heinecke, J.W.; Li, W.; Daehnke, H.L.; Goldstein, J.A. Dityrosine, a Specific Marker of Oxidation. J. Biol. Chem. 1993, 268, 4069–4077. [Google Scholar] [PubMed]
- Witko-Sarsat, V.; Friedlander, M.; Capeillère-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Iwao, Y.; Anraku, M.; Hiraike, M.; Kawai, K.; Nakajou, K.; Kai, T.; Suenaga, A.; Otagiri, M. The structural and pharmacokinetic properties of oxidized human serum albumin, advanced oxidation protein products (AOPP). Drug Metab. Pharmacokinet. 2006, 21, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.J.; Niu, H.X.; Hou, F.F.; Zhang, L.; Fu, N.; Nagai, R.; Lu, X.; Chen, B.H.; Shan, Y.X.; Tian, J.W.; et al. Advanced oxidation protein products activate vascular endothelial cells via a RAGE-mediated signaling pathway. Antioxid. Redox Signal. 2008, 10, 1699–1712. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.S.; Tobin, J.D.; Hollis, B.W.; Gundberg, C.M.; Roy, T.A.; Plato, C.C. Biochemical parameters associated with low bone density in healthy men and women. J Bone Min Res. 1992, 7, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.B.; Zhong, Z.M.; Hou, G.; Jiang, H.; Chen, J.T. Involvement of oxidative stress in age-related bone loss. J. Surg. Res. 2011, 169, e37–e42. [Google Scholar] [CrossRef] [PubMed]
- Boskey, A.L.; Coleman, R. Aging and Bone. J. Dent. Res. 2010, 89, 1333–1348. [Google Scholar] [CrossRef] [PubMed]
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Cervellati, C.; Bonaccorsi, G.; Cremonini, E.; Romani, A.; Fila, E.; Castaldini, M.C.; Ferrazzini, S.; Fifanti, M.; Massari, L. Oxidative stress and bone resorption interplay as a possible trigger for postmenopausal osteoporosis. Biomed. Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rodríguez, M.A.; Ruiz-Ramos, M.; Correa-Muñoz, E.; Mendoza-Núñez, V.M. Oxidative stress as a risk factor for osteoporosis in elderly Mexicans as characterized by antioxidant enzymes. BMC Musculoskelet. Disord. 2007, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Östman, B.; Michaëlsson, K.; Helmersson, J.; Gedeborg, R.; Melhus, H. Oxidative stress and bone mineral density in elderly men: Antioxidant activity of alpha-tocopherol. Free Radic. Biol. Med. 2009, 47, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Lv, B.; Wang, Y. ProteinpPhosphatase 2A mediates oxidative stress induced apoptosis in osteoblasts. Mediat. Inflamm. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Almeida, M.; Han, L.; Martin-Millan, M.; O’Brien, C.A.; Manolagas, S.C. Oxidative stress antagonizes Wnt signaling in osteoblast precursors by diverting β-catenin from T cell factor- to forkhead box O-mediated transcription. J. Biol. Chem. 2007, 282, 27298–27305. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Zhong, Z.M.; Pan, Y.; Zeng, J.H.; Zheng, S.; Zhu, S.Y.; Chen, J.T. Advanced Oxidation Protein Products as a Novel Marker of Oxidative Stress in Postmenopausal Osteoporosis. Med. Sci. Monit. 2015, 21, 2428–2432. [Google Scholar] [CrossRef] [PubMed]
- Cervellati, C.; Bonaccorsi, G.; Cremonini, E.; Bergamini, C.M.; Patella, A.; Castaldini, C.; Ferrazzini, S.; Capatti, A.; Picarelli, V.; Pansini, F.S.; et al. Bone mass density selectively correlates with serum markers of oxidative damage in post-menopausal women. Clin. Chem. Lab. Med. 2013, 51, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Yildirimturk, S.; Batu, S.; Alatli, C.; Olgac, V.; Firat, D.; Sirin, Y. The effects of supplemental melatonin administration on the healing of bone defects in streptozotocin-induced diabetic rats. J. Appl. Oral Sci. 2016, 24, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Huang, D.; Wang, K.; Lin, B.; Liu, Y.; Liu, S.; Wu, W.; Zhang, H. Advanced oxidation protein products induce apoptosis, and upregulate sclerostin and RANKL expression, in osteocytic MLO-Y4 cells via JNK/p38 MAPK activation. Mol. Med. Rep. 2016, 15, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Baron, R.; Kneissel, M. WNT signaling in bone homeostasis and disease: From human mutations to treatments. Nat. Med. 2013, 19, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Xing, L. Biology of RANK, RANKL, and osteoprotegerin. Arthritis Res. Ther. 2007, 9, S1. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Yang, L.; Li, Y.; Zhang, H.; Chen, H.; Liu, D.; Li, Q.; Cai, D. Effect of advanced oxidation protein products on the proliferation and osteogenic differentiation of rat mesenchymal stem cells. Int. J. Mol. Med. 2013, 32, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.M.; Bai, L.; Chen, J.T. Advanced oxidation protein products inhibit proliferation and differentiation of rat osteoblast-like cells via NF-κB pathway. Cell. Physiol. Biochem. 2009, 24, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Naito, A.; Azuma, S.; Tanaka, S.; Miyazaki, T.; Takaki, S.; Takatsu, K.; Nakao, K.; Nakamura, K.; Katsuki, M.; Yamamoto, T.; et al. Severe osteopetrosis, defective interleukin-1 signalling and lymph node organogenesis in TRAF6-deficient mice. Genes Cells 1999, 4, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. The role of NFAT in osteoclast formation. Ann. N. Y. Acad. Sci. 2007, 1116, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Wautier, M.; Chappey, O.; Corda, S.; Stern, D.M.; Schmidt, A.M.; Wautier, J. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E685–E694. [Google Scholar] [PubMed]
- Li, G.; Xu, J.; Li, Z. Receptor for advanced glycation end products inhibits proliferation in osteoblast through suppression of Wnt, PI3K and ERK signaling. Biochem. Biophys. Res. Commun. 2012, 423, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Immel, D.; Xi, C.X.; Bierhaus, A.; Feng, X.; Mei, L.; Nawroth, P.; Stern, D.M.; Xiong, W.C. Regulation of osteoclast function and bone mass by RAGE. J. Exp. Med. 2006, 203, 1067–1080. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Xiong, F.; Hong, Y.; Jung, J.U.; Li, X.S.; Liu, J.Z.; Yan, R.; Mei, L.; Feng, X.; Xiong, W.C. APPswe/AB regulation of osteoclast activation and RAGE expression in an age-dependent manner. J. Bone Miner. Res. 2011, 26, 1084–1098. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.M.; Kim, Y.H. Hesperetin attenuates the highly reducing sugar-triggered inhibition of osteoblast differentiation. Cell Biol. Toxicol. 2008, 24, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Choi, E.M. Myricetin, a naturally occurring flavonoid, prevents 2-deoxy-d-ribose induced dysfunction and oxidative damage in osteoblastic MC3T3-E1 cells. Eur. J. Pharmacol. 2008, 591, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Mayo, J.C.; Sainz, R.M.; Leon, J.; Czarnocki, Z. Melatonin as an antioxidant: Biochemical mechanisms and pathophysiological implications in humans. Acta Biochim. Pol. 2003, 50, 1129–1146. [Google Scholar] [PubMed]
- Wang, F.W.; Wang, Z.; Zhang, Y.M.; Du, Z.X.; Zhang, X.L.; Liu, Q.; Guo, Y.J.; Li, X.G.; Hao, A.J. Protective effect of melatonin on bone marrow mesenchymal stem cells against hydrogen peroxide-induced apoptosis in vitro. J. Cell. Biochem. 2013, 114, 2346–2355. [Google Scholar] [CrossRef] [PubMed]
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; López, G.; Lorenzo, L.; Ocaña-Peinado, F.M.; Olea-Serrano, F. Association between dietary antioxidant quality score (DAQs) and bone mineral density in Spanish women. Nutr. Hosp. 2012, 27, 1886–1893. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Munger, R.G.; West, N.A.; Cutler, D.R.; Wengreen, H.J.; Corcoran, C.D. Antioxidant intake and risk of osteoporotic hip fracture in Utah: An effect modified by smoking status. Am. J. Epidemiol. 2006, 163, 9–17. [Google Scholar] [CrossRef] [PubMed]
- De Franca, N.A.; Camargo, M.B.; Lazaretti-Castro, M.; Martini, L.A. Antioxidant intake and bone status in a cross-sectional study of Brazilian women with osteoporosis. Nutr. Health 2013, 22, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.L.; von Bergen, V.; Chyu, M.C.; Jenkins, M.R.; Mo, H.; Chen, C.H.; Kwun, I.S. Fruits and dietary phytochemicals in bone protection. Nutr. Res. 2012, 32, 897–910. [Google Scholar] [CrossRef] [PubMed]
- McGhie, T.K.; Walton, M.C.; Barnett, L.E.; Vather, R.; Martin, H.; Au, J.; Alspach, P.A.; Booth, C.L.; Kruger, M.C. Boysenberry and blackcurrant drinks increased the plasma antioxidant capacity in an elderly population but had little effect on other markers of oxidative stress. J. Sci. Food Agric. 2007, 87, 2519–2527. [Google Scholar] [CrossRef]
- Kumazawa, Y.; Kawaguchi, K.; Takimoto, H. Immunomodulating Effects of Flavonoids on Acute and Chronic Inflammatory Responses Caused by Tumor Necrosis Factor α. Curr. Pharm. Des. 2006, 12, 4271–4279. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Kim, B.; Yang, Y.; Pham, T.X.; Park, Y.K.; Manatou, J.; Koo, S.I.; Chun, O.K.; Lee, J.Y. Berry anthocyanins suppress the expression and secretion of proinflammatory mediators in macrophages by inhibiting nuclear translocation of NF-κB independent of NRF2-mediated mechanism. J. Nutr. Biochem. 2014, 25, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wu, S.M.; Xu, Z.Y.; Ou-Yang, S. Rabbiteye blueberry prevents osteoporosis in ovariectomized rats. J. Orthop. Surg. Res. 2014, 9, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Mun, S.; Lee, S.G.; Vance, T.M.; Hubert, P.; Koo, S.I.; Lee, S.K.; Chun, O.K. Anthocyanin-Rich Blackcurrant Extract Attenuates Ovariectomy-Induced Bone Loss in Mice. J. Med. Food 2016, 19, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Devareddy, L.; Hooshmand, S.; Collins, J.K.; Lucas, E.A.; Chai, S.C.; Arjmandi, B.H. Blueberry prevents bone loss in ovariectomized rat model of postmenopausal osteoporosis. J. Nutr. Biochem. 2008, 19, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Komine, M.; Kukita, A.; Kukita, T.; Ogata, Y.; Hotokebuchi, T.; Kohashi, O. Tumor necrosis factor-alpha cooperates with receptor activator of nuclear factor kappaB ligand in generation of osteoclasts in stromal cell-depleted rat bone marrow cell culture. Bone 2001, 28, 474–483. [Google Scholar] [CrossRef]
- Fox, S.W.; Evans, K.E.; Lovibond, A.C. Transforming growth factor-β enables NFATc1 expression during osteoclastogenesis. Biochem. Biophys. Res. Commun. 2008, 366, 123–128. [Google Scholar] [CrossRef] [PubMed]
| Study | Population | Observations | Limitations |
|---|---|---|---|
| Zhang (2011) [19] | Young, adult, and old (n = 26 each) male Wistar rats | AOPP in plasma and femur increased with aging and were negatively associated with femur BMD | Sample size; potentially limited translatability to humans |
| Wu (2015) [27] | 60 postmenopausal women with osteoporosis, 60 without osteoporosis | AOPP was associated with reduced BMD and increased bone turnover markers | Sample size; no adjustment for factors such as BMI, diet, or smoking; BMD assessed only at lumbar spine |
| Cervellati (2013) [28] | 98 pre- and 93 post-menopausal women | No significant association between AOPP and bone status | Potential for residual confounding; AOPP assessed only in serum |
| Cervellati (2014) [22] | 167 postmenopausal women | No significant association between AOPP and bone status | Potential for residual confounding; AOPP assessed only in serum |
| Study | Cell Type | Treatments | Outcome |
|---|---|---|---|
| Yu (2016) [30] | Osteocytic MLO-Y4 cells | Cultured with AOPP-MSA (25, 50, 100, or 200 μg/mL for 24 h or 200 μg/mL for 3, 6, 12, or 24 h) | AOPP triggered apoptosis and upregulated expression of sclerostin and RANKL in a JNK/p38 MAPK-dependent manner |
| Sun (2013) [33] | Rat MSC | Cultured with AOPP-BSA (50, 100, 200, or 400 μg/mL for 3 days or 200 μg/mL for 24, 48, or 72 h) | AOPP inhibited proliferation, reduced ALP activity and ALP and collagen I mRNA, increased ROS generation, upregulated RAGE expression |
| Zhong (2009) [34] | Rat osteoblast-like cells | Cultured with AOPP-RSA (50, 100, or 200 μg/mL for 24 h or 100 μg/mL for 24, 48, or 72 h) | AOPP inhibited proliferation, reduced ALP activity, downregulated expression of osteocalcin, induced ROS generation and NFκB phosphorylation |
| Study | Population | Treatments | Duration | Outcome |
|---|---|---|---|---|
| Li (2014) [52] | Female Sprague Dawley rats (total n = 30) | Randomized to sham operation, OVX control, and OVX blueberry treatment (10% w/w freeze-dried blueberry powder) | 12 weeks | Blueberry inhibited bone resorption, bone loss, and the reduction of bone strength of OVX rats |
| Zheng (2016) [53] | Female C57BL/6J mice (total n = 54) | Randomized to sham operation or OVX, then further divided into control diet or diet containing 1% blackcurrant extract | 4, 8, or 12 weeks | Blackcurrant attenuated OVX-induced bone loss as measured by BMD and trabecular volume; blackcurrant reduced bone resorption activity |
| Devareddy (2008) [54] | Female Sprague Dawley rats (n = 30) | Randomized to sham operation, OVX control, and OVX blueberry treatment (5% w/w dried blueberry powder) | 100 days | Blueberry prevented OVX-induced loss of whole-body BMD; blueberry treatment group had lower serum osteocalcin |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melough, M.M.; Sun, X.; Chun, O.K. The Role of AOPP in Age-Related Bone Loss and the Potential Benefits of Berry Anthocyanins. Nutrients 2017, 9, 789. https://doi.org/10.3390/nu9070789
Melough MM, Sun X, Chun OK. The Role of AOPP in Age-Related Bone Loss and the Potential Benefits of Berry Anthocyanins. Nutrients. 2017; 9(7):789. https://doi.org/10.3390/nu9070789
Chicago/Turabian StyleMelough, Melissa M., Xin Sun, and Ock K. Chun. 2017. "The Role of AOPP in Age-Related Bone Loss and the Potential Benefits of Berry Anthocyanins" Nutrients 9, no. 7: 789. https://doi.org/10.3390/nu9070789
APA StyleMelough, M. M., Sun, X., & Chun, O. K. (2017). The Role of AOPP in Age-Related Bone Loss and the Potential Benefits of Berry Anthocyanins. Nutrients, 9(7), 789. https://doi.org/10.3390/nu9070789






