Dietary Natural Products for Prevention and Treatment of Breast Cancer
Abstract
:1. Introduction
2. Soy
2.1. Epidemiological Evidence
2.2. Experimental Evidence
3. Fruits
3.1. Pomegranate
3.2. Mangosteen
3.3. Citrus Fruits
3.4. Apple
3.5. Grape
3.6. Mango
3.7. Other Fruits
4. Vegetables
4.1. Cruciferous Vegetables
4.1.1. Isothiocyanates
4.1.2. Indole-3-Carbinol
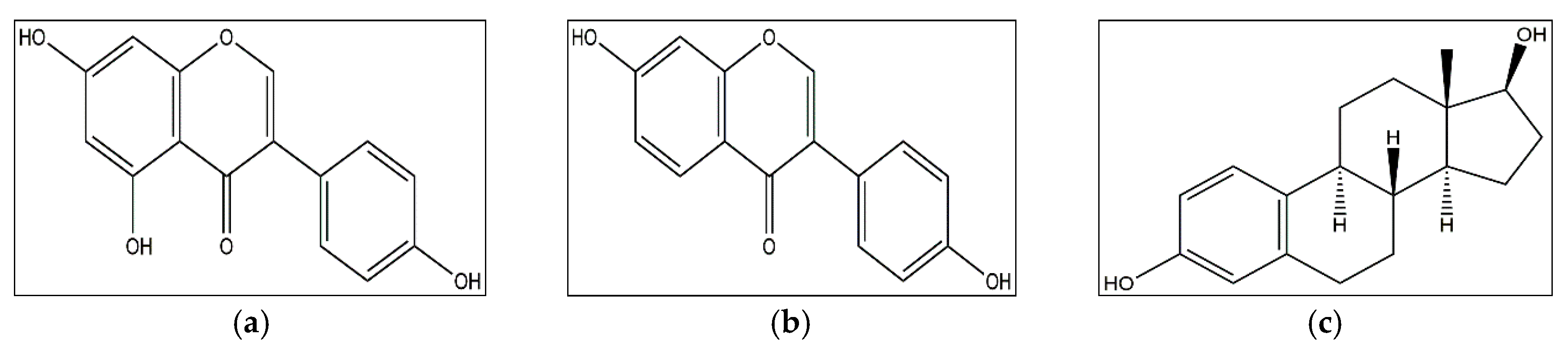
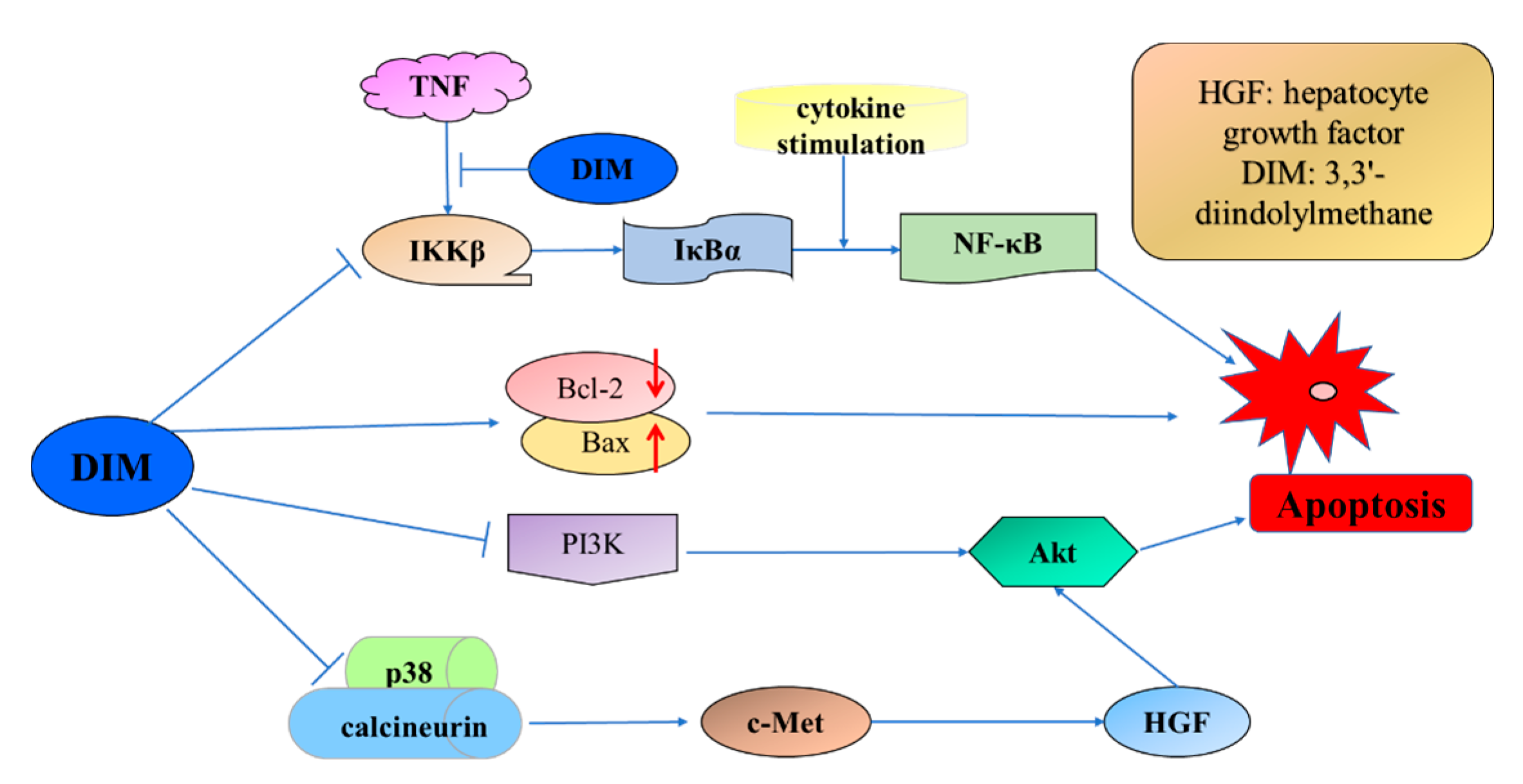
4.1.3. 3,3′-Diindolylmethane
4.2. Other Vegetables
5. Spices
5.1. Ginger
5.2. Garlic
5.3. Black Cumin
5.4. Other Spices
6. Edible Macro-Fungi
7. Cereals
8. Synergistic Effects of Dietary Natural Products with Anticancer Therapies
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.S.; Pusztai, L. Breast Cancer 2 Gene expression profiling in breast cancer: Classification, prognostication, and prediction. Lancet 2011, 378, 1812–1823. [Google Scholar]
- Perou, C.M.; Borresen-Dale, A.L. Systems biology and genomics of breast cancer. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef]
- Hudis, C.A.; Gianni, L. Triple-negative breast cancer: An unmet medical need. Oncologist 2011, 161, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moulder, S.; Hortobagyi, G.N. Advances in the treatment of breast cancer. Clin. Pharmacol. Ther. 2008, 83, 26–36. [Google Scholar] [PubMed]
- DeSantis, C.; Ma, J.M.; Bryan, L.; Jemal, A. Breast cancer statistics, 2013. CA Cancer J. Clin. 2014, 64, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Cazzaniga, M.; Bonanni, B. Breast cancer chemoprevention: Old and new approaches. J. Biomed. Biotechnol. 2012, 2012, 985620. [Google Scholar] [PubMed]
- Reddy, K.B. Triple-negative breast cancers: An updated review on treatment options. Curr. Oncol. 2011, 18, E173–E179. [Google Scholar] [CrossRef] [PubMed]
- Bhadoria, A.S.; Kapil, U.; Sareen, N.; Singh, P. Reproductive factors and breast cancer: A case-control study in tertiary care hospital of North India. Indian J. Cancer 2013, 50, 316–321. [Google Scholar] [PubMed]
- Rieder, V.; Salama, M.; Glockner, L.; Muhr, D.; Berger, A.; Tea, M.K.; Pfeiler, G.; Rappaport-Fuerhauser, C.; Gschwantler-Kaulich, D.; Weingartshofer, S. Effect of lifestyle and reproductive factors on the onset of breast cancer in female BRCA 1 and 2 mutation carriers. Mol. Genet. Genom. Med. 2016, 4, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Sisti, J.S.; Collins, L.C.; Beck, A.H.; Tamimi, R.M.; Rosner, B.A.; Eliassen, A.H. Reproductive risk factors in relation to molecular subtypes of breast cancer: Results from the nurses’ health studies. Int. J. Cancer 2016, 138, 2346–2356. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.Y.; Ursin, G.; Xu, X.X.; Lee, E.; Togawa, K.; Duan, L.; Lu, Y.N.; Malone, K.E.; Marchbanks, P.A.; McDonald, J.A. Reproductive factors and the risk of triple-negative breast cancer in white women and African-American women: A pooled analysis. Breast Cancer Res. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Narod, S.A. Hormone replacement therapy and the risk of breast cancer. Nat. Rev. Clin. Oncol. 2011, 8, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kolonel, L.N.; Lim, U.; White, K.K.; Henderson, B.E.; Wilkens, L.R. Alcohol consumption and breast cancer risk among women from five ethnic groups with light to moderate intakes: The Multiethnic Cohort Study. Int. J. Cancer 2014, 134, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; O’Brien, K.M.; Zhao, S.S.; Weinberg, C.R.; Baird, D.D.; Sandler, D.P. Gestational diabetes mellitus may be associated with increased risk of breast cancer. Br. J. Cancer 2017, 116, 960–963. [Google Scholar] [CrossRef] [PubMed]
- Charlot, M.; Castro-Webb, N.; Bethea, T.N.; Bertrand, K.; Boggs, D.A.; Denis, G.V.; Adams-Campbell, L.L.; Rosenberg, L.; Palmer, J.R. Diabetes and breast cancer mortality in Black women. Cancer Cause Control 2017, 28, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Pierobon, M.; Frankenfeld, C.L. Obesity as a risk factor for triple-negative breast cancers: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 137, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.J.; Lu, Y.S.; Wu, K.J.; Lin, Q.; Shen, W.; Zhu, M.J.; Huang, S.; Chen, J. Does night work increase the risk of breast cancer? A systematic review and meta-analysis of epidemiological studies. Cancer Epidemiol. 2013, 37, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Campeau, P.M.; Foulkes, W.D.; Tischkowitz, M.D. Hereditary breast cancer: New genetic developments, new therapeutic avenues. Hum. Genet. 2008, 124, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zhou, Y.; Li, Y.; Xu, D.P.; Li, S.; Li, H.B. Spices for prevention and treatment of cancers. Nutrients 2016, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.P.; Li, S.; Chen, Y.M.; Li, H.B. Natural polyphenols for prevention and treatment of cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Li, S.; Li, H.B. Dietary natural products for prevention and treatment of liver cancer. Nutrients 2016, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Li, Y.; Zhou, T.; Xu, D.P.; Zhang, P.; Li, S.; Li, H.B. Bioactivities and health benefits of mushrooms mainly from China. Molecules 2016, 21, 938. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, S.; Li, H.B.; Deng, G.F.; Ling, W.H.; Xu, X.R. Antiproliferative activities of tea and herbal infusions. Food Funct. 2013, 4, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Bella, F.; Godos, J.; Sciacca, S.; Del Rio, D.; Ray, S.; Galvano, F.; Giovannucci, E.L. Possible role of diet in cancer: Systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nutr. Rev. 2017, 75, 405–419. [Google Scholar] [CrossRef]
- Farvid, M.S.; Chen, W.Y.; Michels, K.B.; Cho, E.; Willett, W.C.; Eliassen, A.H. Fruit and vegetable consumption in adolescence and early adulthood and risk of breast cancer: Population based cohort study. BMJ Br. Med. J. 2016, 353. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Kim, J.H.; Nam, S.J.; Ryu, S.; Kong, G. Dietary intake of soy protein and tofu in association with breast cancer risk based on a case-control study. Nutr. Cancer 2008, 60, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Nechuta, S.J.; Caan, B.J.; Chen, W.Y.; Lu, W.; Chen, Z.; Kwan, M.L.; Flatt, S.W.; Zheng, Y.; Zheng, W.; Pierce, J.P. Soy food intake after diagnosis of breast cancer and survival: An in-depth analysis of combined evidence from cohort studies of US and Chinese women. Am. J. Clin. Nutr. 2012, 96, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A.; Rock, C.L.; Thompson, P.A.; Caan, B.J.; Cussler, E.; Flatt, S.W.; Pierce, J.P. Vegetable intake is associated with reduced breast cancer recurrence in tamoxifen users: A secondary analysis from the Women’s Healthy Eating and Living Study. Breast Cancer Res. Treat. 2011, 125, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Lv, Z.D.; Liu, X.P.; Zhao, W.J.; Dong, Q.; Li, F.N.; Wang, H.B.; Kong, B. Curcumin induces apoptosis in breast cancer cells and inhibits tumor growth in vitro and in vivo. Int. J. Clin. Exp. Pathol. 2014, 7, 2818–2824. [Google Scholar] [PubMed]
- Gallardo, M.; Calaf, G.M. Curcumin inhibits invasive capabilities through epithelial mesenchymal transition in breast cancer cell lines. Int. J. Oncol. 2016, 49, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Varinska, L.; Gal, P.; Mojzisova, G.; Mirossay, L.; Mojzis, J. Soy and breast cancer: Focus on angiogenesis. Int. J. Mol. Sci. 2015, 16, 11728–11749. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.J.; Xie, M.Y.; Kluxen, F.M.; Diel, P. Genistein modulates the anti-tumor activity of cisplatin in MCF-7 breast and HT-29 colon cancer cells. Arch. Toxicol. 2014, 88, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Bonofiglio, D.; Giordano, C.; de Amicis, F.; Lanzino, M.; Ando, S. Natural products as promising antitumoral agents in breast cancer: Mechanisms of action and molecular targets. Mini Rev. Med. Chem. 2016, 16, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Shu, X.O.; Gao, Y.T.; Yang, G.; Li, Q.; Li, H.L.; Jin, F.; Zheng, W. Soy food consumption is associated with lower risk of coronary heart disease in Chinese women. J. Nutr. 2003, 133, 2874–2878. [Google Scholar] [PubMed]
- Mueller, N.T.; Odegaard, A.O.; Gross, M.D.; Koh, W.P.; Yu, M.C.; Yuan, J.M.; Pereira, M.A. Soy intake and risk of type 2 diabetes mellitus in Chinese Singaporeans. Eur. J. Nutr. 2012, 51, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; Qin, L.Q. Soy isoflavones consumption and risk of breast cancer incidence or recurrence: A meta-analysis of prospective studies. Breast Cancer Res. Treat. 2011, 125, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; Del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Chi, F.; Wu, R.; Zeng, Y.C.; Xing, R.; Liu, Y.; Xu, Z.G. Post-diagnosis soy food intake and breast cancer survival: A meta-analysis of cohort studies. Asian Pac. J. Cancer Prev. 2013, 14, 2407–2412. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.Y.; Zhou, L.; Jiao, S.C.; Xu, L.Z. Relationship between soy food intake and breast cancer in China. Asian Pac. J. Cancer Prev. 2011, 12, 2837–2840. [Google Scholar] [PubMed]
- Trock, B.J.; Hilakivi-Clarke, L.; Clarke, R. Meta-analysis of soy intake and breast cancer risk. J. Natl. Cancer Inst. 2006, 98, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C. Factors to consider in the association between soy Iisoflavone intake and breast cancer risk. J. Epidemiol. 2010, 20, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Baglia, M.L.; Zheng, W.; Li, H.L.; Yang, G.; Gao, J.; Gao, Y.T.; Shu, X.O. The association of soy food consumption with the risk of subtype of breast cancers defined by hormone receptor and HER2 status. Int. J. Cancer 2016, 139, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Wada, K.; Nakamura, K.; Tamai, Y.; Tsuji, M.; Kawachi, T.; Hori, A.; Takeyama, N.; Tanabashi, S.; Matsushita, S.; Tokimitsu, N. Soy isoflavone intake and breast cancer risk in Japan: From the Takayama study. Int. J. Cancer 2013, 133, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.F.; Kang, H.B.; Li, B.L.; Zhang, R.M. Positive effects of Ssoy isoflavone food on survival of breast cancer patients in China. Asian Pac. J. Cancer Prev. 2012, 13, 479–482. [Google Scholar] [CrossRef] [PubMed]
- Ko, K.P.; Kim, S.W.; Ma, S.H.; Park, B.; Ahn, Y.; Lee, J.W.; Lee, M.H.; Kang, E.; Kim, L.S.; Jung, Y. Dietary intake and breast cancer among carriers and noncarriers of BRCA mutations in the Korean Hereditary Breast Cancer Study. Am. J. Clin. Nutr. 2013, 98, 1493–1501. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.Y.; Cai, Q.Y.; Bao, P.P.; Wu, J.; Wen, W.Q.; Ye, F.; Zheng, W.; Zheng, Y.; Shu, X.O. Long-term soy consumption and tumor tissue microRNA and gene expression in triple-negative breast cancer. Cancer Am. Cancer Soc. 2016, 122, 2544–2551. [Google Scholar] [CrossRef] [PubMed]
- Conroy, S.M.; Maskarinec, G.; Park, S.Y.; Wilkens, L.R.; Henderson, B.E.; Kolonel, L.N. The effects of soy consumption before diagnosis on breast cancer survival: The multiethnic cohort study. Nutr. Cancer 2013, 65, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Maskarinec, G.; Park, S.Y.; Ettienne, R.; Matsuno, R.K.; Long, C.; Steffen, A.D.; Henderson, B.E.; Kolonel, L.N.; Le Marchand, L. Dietary isoflavone intake is not statistically significantly associated with breast cancer risk in the Multiethnic Cohort. Br. J. Nutr. 2014, 112, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.D.; Park, K.S.; Ro, J.; Kim, J. Differential influence of dietary soy intake on the risk of breast cancer recurrence related to HER2 status. Nutr. Cancer 2012, 64, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Chatterton, R.T.; Michel, N.; Bryk, M.; Lee, O.; Ivancic, D.; Heinz, R.; Zalles, C.M.; Helenowski, I.B.; Jovanovic, B.D. Soy isoflavone supplementation for breast cancer risk reduction: A randomized phase II trial. Cancer Prev. Res. 2012, 5, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Sobue, T.; Sasaki, S.; Kobayashi, M.; Arai, Y.; Uehara, M.; Adlercreutz, H.; Watanabe, S.; Takahashi, T.; Iitoi, Y. Validity and reproducibility of a self-administered food-frequency questionnaire to assess isoflavone intake in a Japanese population in comparison with dietary records and blood and urine isoflavones. J. Nutr. 2001, 131, 2741–2747. [Google Scholar] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.; Carlsson, B.; Grandien, K.; Enmark, E.; Häggblad, J.; Nilsson, S.; Gustafsson, J.A. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta. Endocrinology 1997, 138, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Pons, D.G.; Nadal-Serrano, M.; Blanquer-Rossello, M.M.; Sastre-Serra, J.; Oliver, J.; Roca, P. Genistein modulates proliferation and mitochondrial functionality in breast cancer cells depending on ERalpha/ERbeta ratio. J. Cell. Biochem. 2014, 115, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Nadal-Serrano, M.; Pons, D.G.; Sastre-Serra, J.; Blanquer-Rossello, M.D.; Roca, P.; Oliver, J. Genistein modulates oxidative stress in breast cancer cell lines according to ER alpha/ER beta ratio: Effects on mitochondrial functionality, sirtuins, uncoupling protein 2 and antioxidant enzymes. Int. J. Biochem. Cell Biol. 2013, 45, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Pons, D.G.; Nadal-Serrano, M.; Torrens-Mas, M.; Oliver, J.; Roca, P. The phytoestrogen genistein affects breast cancer cells treatment depending on the ER alpha/ER beta ratio. J. Cell. Biochem. 2016, 117, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.P.; Han, D.; Liu, G.; Gao, S.G.; Cai, X.Q.; Duan, R.H.; Feng, X.S. Effects of soy isoflavone and endogenous oestrogen on breast cancer in MMTV-erbB2 transgenic mice. J. Int. Med. Res. 2012, 40, 2073–2082. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, J.; Mo, B.Q.; Hu, C.Y.; Liu, H.Q.; Qi, H.; Wang, X.R.; Xu, J.D. Genistein induces cell apoptosis in MDA-MB-231 breast cancer cells via the mitogen-activated protein kinase pathway. Toxicol. In Vitro 2008, 22, 1749–1753. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.J.; Allsopp, P.; Samaletdin, A.; Rowland, I.R. Daidzein, R-(+)equol and S-(−)equol inhibit the invasion of MDA-MB-231 breast cancer cells potentially via the down-regulation of matrix metalloproteinase-2. Eur. J. Nutr. 2014, 53, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Seol, J.Y.; Youn, Y.N.; Koo, M.; Kim, H.J.; Choi, S.Y. Influence of water-soluble extracts of long-term fermented doenjang on bone metabolism bioactivity and breast cancer suppression. Food Sci. Biotechnol. 2016, 25, 517–524. [Google Scholar] [CrossRef]
- Zhao, Q.X.; Zhao, M.; Parris, A.B.; Xing, Y.; Yang, X.H. Genistein targets the cancerous inhibitor of PP2A to induce growth inhibition and apoptosis in breast cancer cells. Int. J. Oncol. 2016, 49, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Duan, Y.X.; Zhang, X.; Ye, Y.; Ge, B.; Chen, J. Genistein induces apoptosis by the inactivation of the IGF-1R/p-Akt signaling pathway in MCF-7 human breast cancer cells. Food Funct. 2015, 6, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, H.J. A daidzein metabolite, 6,7,4′-trihydroxyisoflavone inhibits cellular proliferation through cell cycle arrest and apoptosis induction in MCF10CA1a human breast cancer cells. J. Korean Soc. Appl. Biol. Chem. 2013, 56, 695–700. [Google Scholar] [CrossRef]
- Stocco, B.; Toledo, K.A.; Fumagalli, H.F.; Bianchini, F.J.; Fortes, V.S.; Fonseca, M.; Toloi, M. Biotransformed soybean extract induces cell death of estrogen-dependent breast cancer cells by modulation of apoptotic proteins. Nutr. Cancer 2015, 67, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Bai, Q.; Zou, L.Y.; Zhang, Q.Y.; Zhou, Y.; Chang, H.; Yi, L.; Zhu, J.D.; Mi, M.T. Genistein inhibits DNA methylation and increases expression of tumor suppressor genes in human breast cancer cells. Genes Chromosomes Cancer 2014, 53, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Zhang, Q.; Wang, X.; Yang, X.; Wang, X.Y.; Huang, Z.; Jiao, Y.C.; Wang, J. Quantitative phosphoproteomics reveals genistein as a modulator of cell cycle and DNA damage response pathways in triple-negative breast cancer cells. Int. J. Oncol. 2016, 48, 1016–1028. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Zhou, W.B.; He, W.; Liu, X.A.; Ding, Q.; Ling, L.J.; Zha, X.M.; Wang, S. Genistein inhibits MDA-MB-231 triple-negative breast cancer cell growth by inhibiting NF-kappa B activity via the Notch-1 pathway. Int. J. Mol. Med. 2012, 30, 337–343. [Google Scholar] [PubMed]
- Fan, P.H.; Fan, S.J.; Wang, H.; Mao, J.; Shi, Y.; Ibrahim, M.M.; Ma, W.; Yu, X.T.; Hou, Z.H.; Wang, B. Genistein decreases the breast cancer stem-like cell population through Hedgehog pathway. Stem Cell Res. Ther. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Vemuri, S.; Alsahafi, S.; Castillo, R.; Cheriyath, V. Glycone-rich soy isoflavone extracts promote estrogen receptor positive breast cancer cell growth. Nutr. Cancer 2016, 68, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Gamra, I.; Davenport, A.; Lester, R.; Zhao, L.J.; Wei, Y.D. Genistein induces cytochrome P450 1B1 gene expression and cell proliferation in human breast cancer MCF-7 Ccells. J. Environ. Pathol. Toxicol. Pathol. 2015, 34, 153–159. [Google Scholar] [CrossRef]
- Andrade, J.E.; Ju, Y.H.; Baker, C.; Doerge, D.R.; Helferich, W.G. Long-term exposure to dietary sources of genistein induces estrogen-independence in the human breast cancer (MCF-7) xenograft model. Mol. Nutr. Food Res. 2015, 59, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.J.; Belosay, A.; Hartman, J.A.; Song, H.X.; Zhang, Y.K.; Wang, W.D.; Doerge, D.R.; Helferich, W.G. Dietary soy isoflavones increase metastasis to lungs in an experimental model of breast cancer with bone micro-tumors. Clin. Exp. Metastasis 2015, 32, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Gaete, L.; Tchernitchin, A.N.; Bustamante, R.; Villena, J.; Lemus, I.; Gidekel, M.; Cabrera, G.; Astorga, P. Daidzein-estrogen interaction in the rat uterus and its effect on human breast cancer cell growth. J. Med. Food 2012, 15, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Koo, J.; Cabarcas-Petroski, S.; Petrie, J.L.; Diette, N.; White, R.J.; Schramm, L. Induction of proto-oncogene BRF2 in breast cancer cells by the dietary soybean isoflavone daidzein. BMC Cancer 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Rigalli, J.P.; Tocchetti, G.N.; Arana, M.R.; Villanueva, S.; Catania, V.A.; Theile, D.; Ruiz, M.L.; Weiss, J. The phytoestrogen genistein enhances multidrug resistance in breast cancer cell lines by translational regulation of ABC transporters. Cancer Lett. 2016, 376, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Chen, M.L.; Qin, Y.; Zhang, Q.Y.; Xu, H.X.; Zhou, Y.; Mi, M.T.; Zhu, J.D. Isoflavone consumption and risk of breast cancer: A dose-response meta-analysis of observational studies. Asia Pac. J. Clin. Nutr. 2013, 22, 118–127. [Google Scholar] [PubMed]
- Li, Y.; Zhang, J.J.; Xu, D.P.; Zhou, T.; Zhou, Y.; Li, S.; Li, H.B. Bioactivities and health benefits of wild fruits. Int. J. Mol. Sci. 2016, 17, 1258. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, S.; Li, H.B.; Deng, G.F.; Ling, W.H.; Wu, S.; Xu, X.R.; Chen, F. Antiproliferative activity of peels, pulps and seeds of 61 fruits. J. Funct. Foods 2013, 5, 1298–1309. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Qin, X.S.; Gan, R.Y.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 wild fruits from South China. Molecules 2010, 15, 8602–8617. [Google Scholar] [CrossRef] [PubMed]
- Xia, E.Q.; Deng, G.F.; Guo, Y.J.; Li, H.B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.; Vieira, A.R.; Rosenblatt, D.; Vieira, R.; Greenwood, D.C.; Norat, T. Fruits, vegetables and breast cancer risk: A systematic review and meta-analysis of prospective studies. Breast Cancer Res. Treat. 2012, 134, 479–493. [Google Scholar] [CrossRef] [PubMed]
- He, J.J.; Gu, Y.T.; Zhang, S.J. Consumption of vegetables and fruits and breast cancer survival: A systematic review and meta-analysis. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Martins-Green, M. The potential of pomegranate and its components for prevention and treatment of breast cancer. Agro Food Ind. Hi-Tech 2013, 24, 58–61. [Google Scholar]
- Adams, L.S.; Zhang, Y.; Navindra, P.; Seeram, N.P.; Heber, D; Chen, S. Pomegranate juice, total pomegranate ellagitannins, and punicalagin suppress inflammatory cell signaling in colon cancer cells. J. Agric. Food Chem. 2006, 54, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Legua, P.; Forner-Giner, M.A.; Nuncio-Jauregui, N.; Hernandez, F. Polyphenolic compounds, anthocyanins and antioxidant activity of nineteen pomegranate fruits: A rich source of bioactive compounds. J. Funct. Foods 2016, 23, 628–636. [Google Scholar] [CrossRef]
- Shirode, A.B.; Kovvuru, P.; Chittur, S.V.; Henning, S.M.; Heber, D.; Reliene, R. Antiproliferative effects of pomegranate extract in MCF-7 breast cancer cells are associated with reduced DNA repair gene expression and induction of double strand breaks. Mol. Carcinog. 2014, 53, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Bishayee, A. Mechanism of breast cancer preventive action of pomegranate: Disruption of estrogen receptor and Wnt/beta-catenin signaling pathways. Molecules 2015, 20, 22315–22328. [Google Scholar] [CrossRef] [PubMed]
- Costantini, S.; Rusolo, F.; De Vito, V.; Moccia, S.; Picariello, G.; Capone, F.; Guerriero, E.; Castello, G.; Volpe, M.G. Potential anti-inflammatory effects of the hydrophilic fraction of pomegranate (Punica granatum L.) seed oil on breast cancer cell lines. Molecules 2014, 19, 8644–8660. [Google Scholar] [CrossRef] [PubMed]
- Rocha, A.; Wang, L.; Penichet, M.; Martins-Green, M. Pomegranate juice and specific components inhibit cell and molecular processes critical for metastasis of breast cancer. Breast Cancer Res. Treat. 2012, 136, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, N.; Talcott, S.; Safe, S.; Mertens-Talcott, S.U. Cytotoxicity of pomegranate polyphenolics in breast cancer cells in vitro and vivo: Potential role of miRNA-27a and miRNA-155 in cell survival and inflammation. Breast Cancer Res. Treat. 2012, 136, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Dikmen, M.; Ozturk, N.; Ozturk, Y. The antioxidant potency of Punica granatum L. fruit peel reduces cell proliferation and induces apoptosis on breast cancer. J. Med. Food 2011, 14, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Khan, G.N.; Gorin, M.A.; Rosenthal, D.; Pan, Q.T.; Bao, L.W.; Wu, Z.F.; Newman, R.A.; Pawlus, A.D.; Yang, P.Y.; Lansky, E.P. Pomegranate fruit extract impairs invasion and motility in human breast cancer. Integr. Cancer Ther. 2009, 8, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.S.; Zhang, Y.J.; Seeram, N.P.; Heber, D.; Chen, S.A. Pomegranate ellagitannin-derived compounds exhibit antiproliferative and antiaromatase activity in breast cancer cells in vitro. Cancer Prev. Res. 2010, 3, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.; Lansky, E.P. Breast cancer chemopreventive properties of pomegranate (Punica granatum) fruit extracts in a mouse mammary organ culture. Eur. J. Cancer Prev. 2004, 13, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.D.; Mehta, R.; Yu, W.P.; Neeman, I.; Livney, T.; Amichay, A.; Poirier, D.; Nicholls, P.; Kirby, A.; Jiang, W.G. Chemopreventive and adjuvant therapeutic potential of pomegranate (Punica granatum) for human breast cancer. Breast Cancer Res. Treat. 2002, 71, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Moongkarndi, P.; Kosem, N.; Kaslungka, S.; Luanratana, O.; Pongpan, N.; Neungton, N. Antiproliferation, antioxidation and induction of apoptosis by Garcinia mangostana (mangosteen) on SKBR3 human breast cancer cell line. J. Ethnopharmacol. 2004, 90, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.M.; Zhao, M.M.; Yang, B.; Bai, W.D. Immunomodulatory and anticancer activities of phenolics from Garcinia mangostana fruit pericarp. Food Chem. 2009, 116, 969–973. [Google Scholar] [CrossRef]
- Gutierrez-Orozco, F.; Failla, M. Biological activities and bioavailability of mangosteen xanthones: A critical review of the current evidence. Nutrients 2013, 5, 3163–3183. [Google Scholar] [CrossRef] [PubMed]
- Balunas, M.J.; Su, B.; Brueggemeier, R.W.; Kinghorn, A.D. Xanthones from the botanical dietary supplement mangosteen (Garcinia mangostana) with aromatase inhibitory activity. J. Nat. Prod. 2008, 71, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Kritsanawong, S.; Innajak, S.; Imoto, M.; Watanapokasin, R. Antiproliferative and apoptosis induction of alpha-mangostin in T47D breast cancer cells. Int. J. Oncol. 2016, 48, 2155–2165. [Google Scholar] [PubMed]
- Kurose, H.; Shibata, M.A.; Iinuma, M.; Otsuki, Y. Alterations in cell cycle and induction of apoptotic cell death in breast cancer cells treated with alpha-mangostin extracted from mangosteen pericarp. J. Biomed. Biotechnol. 2012, 2012, 9p. [Google Scholar] [CrossRef] [PubMed]
- Moongkarndi, P.; Jaisupa, N.; Samer, J.; Kosem, N.; Konlata, J.; Rodpai, E.; Pongpan, N. Comparison of the biological activity of two different isolates from mangosteen. J. Pharm. Pharmacol. 2014, 66, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Tian, W.X.; Ma, X.F. Alpha-mangostin inhibits intracellular fatty acid synthase and induces apoptosis in breast cancer cells. Mol. Cancer 2014, 13, 138. [Google Scholar] [CrossRef] [PubMed]
- Won, Y.S.; Lee, J.H.; Kwon, S.J.; Kim, J.Y.; Park, K.H.; Lee, M.K.; Seo, K.I. Alpha-Mangostin-induced apoptosis is mediated by estrogen receptor alpha in human breast cancer cells. Food Chem. Toxicol. 2014, 66, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Shibata, M.A.; Iinuma, M.; Morimoto, J.; Kurose, H.; Akamatsu, K.; Okuno, Y.; Akao, Y.; Otsuki, Y. α-Mangostin extracted from the pericarp of the mangosteen (Garcinia mangostana Linn) reduces tumor growth and lymph node metastasis in an immunocompetent xenograft model of metastatic mammary cancer carrying a p53 mutation. BMC Med. 2011, 9, 69. [Google Scholar] [CrossRef] [PubMed]
- Doi, H.; Shibata, M.A.; Shibata, E.; Morimoto, J.; Akao, Y.; Iinuma, M.; Tanigawa, N.; Otsuki, Y. Panaxanthone isolated from pericarp of Garcinia mangostana L. suppresses tumor growth and metastasis of a mouse model of mammary cancer. Anticancer Res. 2009, 29, 2485–2495. [Google Scholar] [PubMed]
- Song, J.K.; Bae, J.M. Citrus fruit intake and breast cancer risk: A quantitative systematic review. J. Breast Cancer 2013, 16, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Shin, M.S.; Kim, S.N.; Kim, H.Y.; Kim, K.H.; Shin, K.S.; Kang, K.S. Polysaccharides from Korean Citrus hallabong peels inhibit angiogenesis and breast cancer cell migration. Int. J. Biol. Macromol. 2016, 85, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.T.; Song, Y.W.; Tran, T.A.; Kim, K.S.; Cho, S.K. Induction of apoptosis in anoikis-resistant breast cancer stem cells by supercritical CO2 extracts from Citrus hassaku Hort ex Tanaka. J. Korean Soc. Appl. Biol. 2014, 57, 469–472. [Google Scholar] [CrossRef]
- Alshatwi, A.A.; Shafi, G.; Hasan, T.N.; Al-Hazzani, A.A.; Alsaif, M.A.; Alfawaz, M.A.; Lei, K.Y.; Munshi, A. Apoptosis-mediated inhibition of human breast cancer cell proliferation by lemon citrus extract. Asian Pac. J. Cancer Prev. 2011, 12, 1555–1559. [Google Scholar] [PubMed]
- Li, H.Z.; Yang, B.; Huang, J.; Xiang, T.X.; Yin, X.D.; Wan, J.Y.; Luo, F.; Zhang, L.; Li, H.Y.; Ren, G.S. Naringin inhibits growth potential of human triple-negative breast cancer cells by targeting beta-catenin signaling pathway. Toxicol. Lett. 2013, 220, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.J.; Wilson, L.; Jordan, M.A.; Nguyen, V.; Tang, J.; Smiyun, G. Hesperidin suppressed proliferations of both human breast cancer and androgen-dependent prostate cancer cells. Phytother. Res. 2010, 241, S15–S19. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.F.; Zhang, H.S.; Yang, X.B.; Zhu, Y.L.; Zhang, M. Evaluation of antioxidative and antitumor activities of extracted flavonoids from Pink Lady apples in human colon and breast cancer cell lines. Food Funct. 2015, 6, 3789–3798. [Google Scholar] [CrossRef] [PubMed]
- Schiavano, G.F.; De Santi, M.; Brandi, G.; Fanelli, M.; Bucchini, A.; Giamperi, L.; Giomaro, G. Inhibition of breast cancer cell proliferation and in vitro tumorigenesis by a new red apple cultivar. PLoS ONE 2015, 10, e135840. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, R.H. Apple phytochemical extracts inhibit proliferation of estrogen-dependent and estrogen-independent human breast cancer cells through cell cycle modulation. J. Agric. Food Chem. 2008, 56, 11661–11667. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Liu, R.H. Effect of selected phytochemicals and apple extracts on NF-kappa B activation in human breast cancer MCF-7 cells. J. Agric. Food Chem. 2007, 55, 3167–3173. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Liu, R.H. Effect of 2 alpha-hydroxyursolic acid on NF-kappa B activation induced by TNF-alpha in human breast cancer MCF-7 cells. J. Agric. Food Chem. 2008, 56, 8412–8417. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Li, T.; Liu, R.H. 2 alpha-Hydroxyursolic acid inhibited cell proliferation and induced apoptosis in MDA-MB-231 human breast cancer cells through the p38/MAPK signal transduction pathway. J. Agric. Food Chem. 2016, 64, 1806–1816. [Google Scholar] [CrossRef] [PubMed]
- Delphi, L.; Sepehri, H. Apple pectin: A natural source for cancer suppression in 4T1 breast cancer cells in vitro and express p53 in mouse bearing 4T1 cancer tumors, in vivo. Biomed. Pharmacother. 2016, 84, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, R.H. Synergistic effect of apple extracts and quercetin 3-beta-d-glucoside combination on antiproliferative activity in MCF-7 human breast cancer cells in vitro. J. Agric. Food Chem. 2009, 57, 8581–8586. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Chen, Q.Y.; Wu, L.J.; Yao, X.M.; Sun, X.J. Antitumor and antimetastatic activities of grape skin polyphenols in a murine model of breast cancer. Food Chem. Toxicol. 2012, 50, 3462–3467. [Google Scholar] [CrossRef] [PubMed]
- Dinicola, S.; Pasqualato, A.; Cucina, A.; Coluccia, P.; Ferranti, F.; Canipari, R.; Catizone, A.; Proietti, S.; D’Anselmi, F.; Ricci, G. Grape seed extract suppresses MDA-MB231 breast cancer cell migration and invasion. Eur. J. Nutr. 2014, 53, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Hakimuddin, F.; Paliyath, G.; Meckling, K. Treatment of MCF-7 breast cancer cells with a red grape wine polyphenol fraction results in disruption of calcium homeostasis and cell cycle arrest causing selective cytotoxicity. J. Agric. Food Chem. 2006, 54, 7912–7923. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Kim, M.R.; Kim, O.; Phuong, N.; Yoon, J.; Oh, W.K.; Bae, K.; Kang, K.W. Amurensin G inhibits angiogenesis and tumor growth of tamoxifen-resistant breast cancer via Pin1 inhibition. Food Chem. Toxicol. 2012, 50, 3625–3634. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.J.; Smith, B.A.; Smith, B.N.; Loyd, Q.; Nagappan, P.; McKeithen, D.; Wilder, C.L.; Platt, M.O.; Hudson, T.; Odero-Marah, V.A. Muscadine grape skin extract can antagonize Snail-cathepsin L-mediated invasion, migration and osteoclastogenesis in prostate and breast cancer cells. Carcinogenesis 2015, 36, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, N.; Kim, H.; Krenek, K.; Talcott, S.T.; Mertens-Talcott, S.U. Mango polyphenolics suppressed tumor growth in breast cancer xenografts in mice: Role of the PI3K/AKT pathway and associated microRNAs. Nutr. Res. 2015, 35, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Hoang, V.; Pierson, J.T.; Curry, M.C.; Shaw, P.N.; Dietzgen, R.G.; Gidley, M.J.; Roberts-Thomson, S.J.; Monteith, G.R. Polyphenolic contents and the effects of methanol extracts from mango varieties on breast cancer cells. Food Sci. Biotechnol. 2015, 24, 265–271. [Google Scholar] [CrossRef]
- Abdullah, A.H.; Mohammed, A.S.; Rasedee, A.; Mirghani, M. Oxidative stress-mediated apoptosis induced by ethanolic mango seed extract in cultured estrogen receptor positive breast cancer MCF-7 cells. Int. J. Mol. Sci. 2015, 16, 3528–3536. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.; Mohammed, A.S.; Rasedee, A.; Mirghani, M.; Al-Qubaisi, M.S. Induction of apoptosis and oxidative stress in estrogen receptor-negative breast cancer, MDA-MB231 cells, by ethanolic mango seed extract. BMC Complement. Altern. Med. 2015, 15, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nemec, M.J.; Kim, H.; Marciante, A.B.; Barnes, R.C.; Talcott, S.T.; Mertens-Talcott, S.U. Pyrogallol, an absorbable microbial gallotannins-metabolite and mango polyphenols (Mangifera Indica, L.) suppress breast cancer ductal carcinoma in situ proliferation in vitro. Food Funct. 2016, 7, 3825–3833. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.H.; Wu, C.S.; Wang, M. The jujube (Ziziphus jujuba Mill.) fruit: A review of current knowledge of fruit composition and health benefits. J. Agric. Food Chem. 2013, 61, 3351–3363. [Google Scholar] [CrossRef] [PubMed]
- Plastina, P.; Bonofiglio, D.; Vizza, D.; Fazio, A.; Rovito, D.; Giordano, C.; Barone, I.; Catalano, S.; Gabriele, B. Identification of bioactive constituents of Ziziphus jujube fruit extracts exerting antiproliferative and apoptotic effects in human breast cancer cells. J. Ethnopharmacol. 2012, 140, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.F.; Song, C.K.; Viemstein, H.; Unger, F.; Liang, Z.S. Apoptosis of human breast cancer cells induced by microencapsulated betulinic acid from sour jujube fruits through the mitochondria transduction pathway. Food Chem. 2013, 138, 1998–2007. [Google Scholar] [CrossRef] [PubMed]
- Abedini, M.R.; Erfanian, N.; Nazem, H.; Jamali, S.; Hoshyar, R. Anti-proliferative and apoptotic effects of Ziziphus jujube on cervical and breast cancer cells. Avicenna J. Phytomed. 2016, 6, 142–148. [Google Scholar] [PubMed]
- Somasagara, R.R.; Hegde, M.; Chiruvella, K.K.; Musini, A.; Choudhary, B.; Raghavan, S.C. Extracts of strawberry fruits induce intrinsic pathway of apoptosis in breast cancer cells and inhibits tumor progression in mice. PLoS ONE 2012, 7, e47021. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.; Tang, J.; Oroudjev, E.; Lee, C.J.; Marasigan, C.; Wilson, L.; Ayoub, G. Cytotoxic effects of bilberry extract on MCF7-GFP-Tubulin breast cancer cells. J. Med. Food 2010, 13, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Li, L.Y.; Adams, L.S.; Chen, S.; Killian, C.; Ahmed, A.; Seeram, N.P. Eugenia Jambolana Lam. berry extract inhibits growth and induces apoptosis of human breast cancer but not non-tumorigenic breast cells. J. Agric. Food Chem. 2009, 57, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, R.H. Cranberry phytochemical extracts induce cell cycle arrest and apoptosis in human MCF-7 breast cancer cells. Cancer Lett. 2006, 241, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Noratto, G.; Porter, W.; Byrne, D.; Cisneros-Zevallos, L. Polyphenolics from peach (Prunus persica var. Rich Lady) inhibit tumor growth and metastasis of MDA-MB-435 breast cancer cells in vivo. J. Nutr. Biochem. 2014, 25, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.H.; Im, H.G.; Lee, S.O.; Sung, C.; Park, D.C.; Lee, I.S. Induction of apoptosis by immature fruits of Prunus salicina Lindl. cv. Soldam in MDA-MB-231 human breast cancer cells. Int. J. Food Sci. Nutr. 2007, 58, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska, U.; Szewczyk, K.; Owczarek, K.; Hrabec, Z.; Podsedek, A.; Koziolkiewicz, M.; Hrabec, E. Flavanols from Japanese quince (Chaenomeles japonica) fruit inhibit human prostate and breast cancer cell line invasiveness and cause favorable changes in Bax/Bcl-2 mRNA ratio. Nutr. Cancer 2013, 65, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.M.; Hogan, S.; Schmelz, E.M.; Ju, Y.H.; Canning, C.; Zhou, K.Q. Selective growth inhibition of human breast cancer cells by Graviola fruit extract in vitro and in vivo involving downregulation of EGFR expression. Nutr. Cancer 2011, 63, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Yuan, S.L.; Wang, J.; Lin, P.; Liu, G.J.; Lu, Y.R.; Zhang, J.; Wang, W.D.; Wei, Y.Q. Anticancer activity of litchi fruit pericarp extract against human breast cancer in vitro and in vivo. Toxicol. Appl. Pharm. 2006, 215, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, S.; Perez, H.D.; Paroulek, A.; Chinnakkannu, P.; Kandalam, U.; Jaffe, M.; Rathinavelu, A. Bromelain-induced apoptosis in GI-101A breast cancer cells. J. Med. Food 2012, 15, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.J.; Lv, K.Z. Cruciferous vegetables intake is inversely associated with risk of breast cancer: A meta-analysis. Breast J. 2013, 22, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.G.; Ding, L.; Wang, Z.Y. Isothiocyanates repress estrogen receptor a expression in breast cancer cells. Oncol. Rep. 2009, 21, 185–192. [Google Scholar] [PubMed]
- Tseng, E.; Scott-Ramsay, E.A.; Morris, M.E. Dietary organic isothiocyanates are cytotoxic in human breast cancer MCF-7 and mammary epithelial MCF-12A cell lines. Exp. Biol. Med. 2004, 229, 835–842. [Google Scholar]
- Bradlow, H.L. Indole-3-carbinol as a chemoprotective agent in breast and prostate cancer. In Vivo 2008, 22, 441–445. [Google Scholar] [PubMed]
- Thomson, C.A.; Ho, E.; Strom, M.B. Chemopreventive properties of 3,3′-diindolylmethane in breast cancer: Evidence from experimental and human studies. Nutr. Rev. 2016, 74, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Sehrawat, A.; St Croix, C.; Baty, C.J.; Watkins, S.; Tailor, D.; Singh, R.P.; Singh, S.V. Inhibition of mitochondrial fusion is an early and critical event in breast cancer cell apoptosis by dietary chemopreventative benzyl isothiocyanate. Mitochondrion 2016, 30, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Singh, S.V. p53-Independent apoptosis by benzyl isothiocyanate in human breast cancer cells is mediated by suppression of XIAP expression. Cancer Prev. Res. 2010, 3, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Vogel, V.; Singh, S.V. Benzyl isothiocyanate-induced apoptosis in human breast cancer cells is initiated by reactive oxygen species and regulated by Bax and Bak. Mol. Cancer Ther. 2006, 5, 2931–2945. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Eom, S.J.; Hong, J.E.; Lee, J.Y.; Choi, M.S.; Park, J. Benzyl isothiocyanate inhibits basal and hepatocyte growth factor-stimulated migration of breast cancer cells. Mol. Cell. Biochem. 2012, 359, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Hugo, H.; Ackland, M.L.; Blick, T.; Lawrence, M.G.; Clements, J.A.; Williams, E.D.; Thompson, E.W. Epithelial-mesenchymal and mesenchymal-Epithelial transitions in carcinoma progression. J. Cell. Physiol. 2007, 213, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Sehrawat, A.; Singh, S.V. Benzyl isothiocyanate inhibits epithelial-mesenchymal transition in cultured and vxenografted human breast cancer cells. Cancer Prev. Res. 2011, 4, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Bommareddy, A.; Kim, S.H.; Sehrawat, A.; Hahm, E.R.; Singh, S.V. Benzyl isothiocyanate causes FoxO1-mediated autophagic death in human breast cancer cells. PLoS ONE 2012, 7, e32597. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Nagalingam, A.; Saxena, N.K.; Singh, S.V.; Sharma, D. Benzyl isothiocyanate inhibits oncogenic actions of leptin in human breast cancer cells by suppressing activation of signal transducer and activator of transcription 3. Carcinogenesis 2011, 32, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Warin, R.; Xiao, D.; Arlotti, J.A.; Bommareddy, A.; Singh, S.V. Inhibition of human breast cancer xenograft growth by cruciferous vegetable constituent benzyl isothiocyanate. Mol. Carcinogen. 2010, 49, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Sehrawat, A.; Singh, S.V. Dietary chemopreventative benzyl isothiocyanate inhibits breast cancer stem cells in vitro and in vivo. Cancer Prev. Res. 2013, 6, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Hahm, E.R.; Singh, S.V. Bim contributes to phenethyl isothiocyanate-induced apoptosis in breast cancer cells. Mol. Carcinog. 2012, 51, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Mukherjee, S.; Biswas, J.; Roy, M. Phenethyl isothiocyanate, by virtue of its antioxidant activity, inhibits invasiveness and metastatic potential of breast cancer cells: HIF-1 alpha as a putative target. Free Radic. Res. 2016, 50, 84–100. [Google Scholar] [CrossRef] [PubMed]
- Aras, U.; Gandhi, Y.A.; Masso-Welch, P.A.; Morris, M.E. Chemopreventive and anti-angiogenic effects of dietary phenethyl isothiocyanate in an N-methyl nitrosourea-induced breast cancer animal model. Biopharm. Drug Dispos. 2013, 34, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.J.; Brazeau, D.A.; Morris, M.E. Dietary phenethyl isothiocyanate alters gene expression in human breast cancer cells. Evid.-Based Complement. Altern. Med. 2011, 2011, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.C.; Singletary, K. Regulation of estrogen receptor alpha expression in human breast cancer cells by sulforaphane. J. Nutr. Biochem. 2009, 20, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Pledgie-Tracy, A.; Sobolewski, M.D.; Davidson, N.E. Sulforaphane induces cell type-specific apoptosis in human breast cancer cell lines. Mol. Cancer Ther. 2007, 6, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Kanematsu, S.; Yoshizawa, K.; Uehara, N.; Miki, H.; Sasaki, T.; Kuro, M.; Lai, Y.C.; Kimura, A.; Yuri, T.; Tsubura, A. Sulforaphane inhibits the growth of KPL-1 human breast cancer cells in vitro and suppresses the growth and metastasis of orthotopically transplanted KPL-1 cells in female athymic mice. Oncol. Rep. 2011, 26, 603–608. [Google Scholar] [PubMed]
- Lee, Y.R.; Noh, E.M.; Han, J.H.; Kim, J.M.; Hwang, B.M.; Kim, B.S.; Lee, S.H.; Jung, S.H.; Youn, H.J.; Chung, E.Y. Sulforaphane controls TPA-induced MMP-9 expression through the NF-kappa B signaling pathway, but not AP-1, in MCF-7 breast cancer cells. BMB Rep. 2013, 46, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Teng, W.D.; Qu, Y.; Wang, H.Y.; Yuan, Q.P. Sulforaphene inhibits triple negative breast cancer through activating tumor suppressor Egr1. Breast Cancer Res. Treat. 2016, 158, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Meeran, S.M.; Patel, S.N.; Tollefsbol, T.O. Sulforaphane causes epigenetic repression of hTERT expression in human breast cancer cell lines. PLoS ONE 2010, 5, e11457. [Google Scholar] [CrossRef] [PubMed]
- Jo, E.H.; Kim, S.H.; Ahn, N.S.; Park, J.S.; Hwang, J.W.; Lee, Y.S.; Kang, K.S. Efficacy of sulforaphane is mediated by p38 MAP kinase and caspase-7 activations in ER-positive and COX-2-expressed human breast cancer cells. Eur. J. Cancer Prev. 2007, 16, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.H.; Qi, M.; Chen, D.Z.; Yuan, R.Q.; Goldberg, I.D.; Rosen, E.M.; Auborn, K.; Fan, S.J. Suppression of breast cancer invasion and migration by indole-3-carbinol: Associated with up-regulation of BRCA1 and E-cadherin/catenin complexes. J. Mol. Med. 2000, 78, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.N.; Jun, W.; Choue, R.; Lee, J. I3C and ICZ inhibit migration by suppressing the EMT process and FAK expression in breast cancer cells. Mol. Med. Rep. 2013, 7, 384–388. [Google Scholar] [PubMed]
- Hung, W.C.; Chang, H.C. Indole-3-carbinol inhibits Sp1-induced matrix metalloproteinase-2 expression to attenuate migration and invasion of breast cancer cells. J. Agric. Food Chem. 2009, 57, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K.; Sarkar, F.H.; Banerjee, S.; Wang, Z.W.; Liao, D.; Hong, X.; Sarkar, N.H. Therapeutic intervention of experimental breast cancer bone metastasis by indole-3-carbinol in SCID-human mouse model. Mol. Cancer Ther. 2006, 5, 2747–2756. [Google Scholar] [CrossRef] [PubMed]
- Garcia, H.H.; Brar, G.A.; Nguyen, D.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol (I3C) inhibits cyclin-dependent kinase-2 function in human breast cancer cells by regulating the size distribution, associated cyclin E forms, and subcellular localization of the CDK2 protein complex. J. Biol. Chem. 2005, 280, 8756–8764. [Google Scholar] [CrossRef] [PubMed]
- Cram, E.J.; Liu, B.D.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol inhibits CDK6 expression in human MCF-7 breast cancer cells by disrupting Sp1 transcription factor interactions with a composite element in the CDK6 gene promoter. J. Biol. Chem. 2001, 276, 22332–22340. [Google Scholar] [CrossRef] [PubMed]
- Marconett, C.N.; Sundar, S.N.; Tseng, M.; Tin, A.S.; Tran, K.Q.; Mahuron, K.M.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol downregulation of telomerase gene expression requires the inhibition of estrogen receptor-alpha and Sp1 transcription factor interactions within the hTERT promoter and mediates the G1 cell cycle arrest of human breast cancer cells. Carcinogenesis 2011, 32, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K.; Aranha, O.; Glazyrin, A.; Chinni, S.R.; Sarkar, F.H. Translocation of Bax to mitochondria induces apoptotic cell death in Indole-3-carbinol (I3C) treated breast cancer cells. Oncogene 2000, 19, 5764–5771. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, F.H.; Rahman, K.; Li, Y.W. Bax translocation to mitochondria is an important event in inducing apoptotic cell death by indole-3-carbinol (I3C) treatment of breast cancer cells. J. Nutr. 2003, 133S, 2434S–2439S. [Google Scholar]
- Marconett, C.N.; Singhal, A.K.; Sundar, S.N.; Firestone, G.L. Indole-3-carbinol disrupts estrogen receptor-alpha dependent expression of insulin-like growth factor-1 receptor and insulin receptor substrate-1 and proliferation of human breast cancer cells. Mol. Cell. Endocrinol. 2012, 363, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Brew, C.T.; Aronchik, I.; Kosco, K.; McCammon, J.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol inhibits MDA-MB-231 breast cancer cell motility and induces stress fibers and focal adhesion formation by activation of Rho kinase activity. Int. J. Cancer 2009, 124, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Old, L.J.; Schreiber, R.D. The roles of IFN gamma in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev. 2002, 13, 95–109. [Google Scholar] [CrossRef]
- Chatterji, U.; Riby, J.E.; Taniguchi, T.; Bjeldanes, E.L.; Bjeldanes, L.F.; Firestone, G.L. Indole-3-carbinol stimulates transcription of the interferon gamma receptor 1 gene and augments interferon responsiveness in human breast cancer cells. Carcinogenesis 2004, 25, 1119–1128. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.; Firestone, G.L.; Bjeldanes, L.F. Bcl-2 family-mediated apoptotic effects of 3,3′-diindolylmethane (DIM) in human breast cancer cells. Biochem. Pharmacol. 2002, 63, 1085–1097. [Google Scholar] [CrossRef]
- Rahman, K.; Sarkar, F.H. Inhibition of nuclear translocation of nuclear factor-kappa B contributes to 3,3′-diindolylmethane-induced apoptosis in breast cancer cells. Cancer Res. 2005, 65, 364–371. [Google Scholar] [PubMed]
- Nicastro, H.L.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-Diindolylmethane rapidly and selectively inhibits hepatocyte growth factor/c-Met signaling in breast cancer cells. J. Nutr. Biochem. 2013, 24, 1882–1888. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.C. 3,3′-Diindolylmethane inhibits breast cancer cell growth via miR-21-mediated Cdc25A degradation. Mol. Cell. Biochem. 2011, 358, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.B.; Kim, H.A.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-Diindolylmethane (DIM) induces a G1 cell cycle arrest in human breast cancer cells that is accompanied by Sp1-mediated activation of p21(WAF1/CIP1) expression. Carcinogenesis 2002, 23, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.X.; Sohn, H.; Xue, L.; Firestone, G.L.; Bjeldanes, L.F. 3,3′-diindolylmethane is a novel mitochondrial H+-ATP synthase inhibitor that can induce p21(Cip1/Waf1) expression by induction of oxidative stress in human breast cancer cells. Cancer Res. 2006, 66, 4880–4887. [Google Scholar] [CrossRef] [PubMed]
- Hsu, E.L.; Chen, N.; Westbrook, A.; Wang, F.; Zhang, R.X.; Taylor, R.T.; Hankinson, O. CXCR4 and CXCL12 down-regulation: A novel mechanism for the chemoprotection of 3,3′-diindolylmethane for breast and ovarian cancers. Cancer Lett. 2008, 265, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K.; Li, Y.W.; Wang, Z.W.; Sarkar, S.H.; Sarkar, F.H. Gene expression profiling revealed survivin as a target of 3,3′-diindolylmethane-induced cell growth inhibition and apoptosis in breast cancer cells. Cancer Res. 2006, 66, 4952–4960. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Firestone, G.L.; Bjeldanes, L.F. DIM stimulates IFN gamma gene expression in human breast cancer cells via the specific activation of JNK and p38 pathways. Oncogene 2005, 24, 2343–2353. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, G.J.; Azuine, M.A.; Rao, G.S.; Arai, T.; Iida, A.; Tokuda, H. Cytotoxic effect of the red beetroot (Beta vulgaris L.) extract compared to doxorubicin (Adriamycin) in the human prostate (PC-3) and breast (MCF-7) cancer cell lines. Anti-Cancer Agents Med. Chem. 2011, 11, 280–284. [Google Scholar] [CrossRef]
- Butalla, A.C.; Crane, T.E.; Patil, B.; Wertheim, B.C.; Thompson, P.; Thomson, C.A. Effects of a carrot juice intervention on plasma carotenoids, oxidative stress, and inflammation in overweight breast cancer survivors. Nutr. Cancer 2012, 64, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Ansari, J.A.; Ahmad, M.K.; Khan, A.R.; Fatima, N.; Khan, H.J.; Rastogi, N.; Mishra, D.P.; Mahdi, A.A. Anticancer and antioxidant activity of Zingiber officinale Roscoe rhizome. Indian J. Exp. Biol. 2016, 54, 767–773. [Google Scholar]
- Elkady, A.I.; Abuzinadah, O.A.; Baeshen, N.; Rahmy, T.R. Differential control of growth, apoptotic activity, and gene expression in human breast cancer cells by extracts derived from medicinal herbs Zingiber officinale. J. Biomed. Biotechnol. 2012, 2012, 614356. [Google Scholar] [CrossRef] [PubMed]
- Joo, J.H.; Hong, S.S.; Cho, Y.R.; Seo, D.W. 10-Gingerol inhibits proliferation and invasion of MDA-MB-231 breast cancer cells through suppression of Akt and p38(MAPK) activity. Oncol. Rep. 2016, 35, 779–784. [Google Scholar] [PubMed]
- Lee, H.S.; Seo, E.Y.; Kang, N.E.; Kim, W.K. (6)-Gingerol inhibits metastasis of MDA-MB-231 human breast cancer cells. J. Nutr. Biochem. 2008, 19, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Yang, H.; Tan, S.H.; Chui, W.K.; Chew, E.H. 6-Shogaol, an active constituent of ginger, inhibits breast cancer cell invasion by reducing matrix metalloproteinase-9 expression via blockade of nuclear factor-kappa B activation. Br. J. Pharmacol. 2010, 161, 1763–1777. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.H.; Wu, C.H.; Yeh, C.T.; Yen, G.C. Invadopodia-associated proteins blockade as a novel mechanism for 6-shogaol and pterostilbene to reduce breast cancer cell motility and invasion. Mol. Nutr. Food Res. 2013, 57, 886–895. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.; Vasudevan, S.; Sengupta, S. 6-Shogaol inhibits breast cancer cells and stem cell-like spheroids by modulation of Notch signaling pathway and induction of autophagic cell death. PLoS ONE 2015, 10, e1376149. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.L.; Chen, C.Y.; Hou, M.F.; Tsai, E.M.; Jong, Y.J.; Hung, C.H.; Kuo, P.L. 6-Dehydrogingerdione, an active constituent of dietary ginger, induces cell cycle arrest and apoptosis through reactive oxygen species/c-Jun N-terminal kinase pathways in human breast cancer cells. Mol. Nutr. Food Res. 2010, 54, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Arslan, M.; Ozdemir, L. Oral intake of ginger for chemotherapy-induced nausea and vomiting among women with breast cancer. Clin. J. Oncol. Nurs. 2015, 19, E92–E97. [Google Scholar] [CrossRef] [PubMed]
- Lua, P.L.; Salihah, N.; Mazlan, N. Effects of inhaled ginger aromatherapy on chemotherapy-induced nausea and vomiting and health-related quality of life in women with breast cancer. Complement. Ther. Med. 2015, 23, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.M.; Ban, J.O.; Park, K.R.; Lee, C.K.; Jeong, H.S.; Han, S.B.; Hong, J.T. Potential therapeutic effects of functionally active compounds isolated from garlic. Pharmacol. Ther. 2014, 142, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Pourzand, A.; Tajaddini, A.; Pirouzpanah, S.; Asghari-Jafarabadi, M.; Samadi, N.; Ostadrahimi, A.R.; Sanaat, Z. Associations between dietary Allium vegetables and risk of breast cancer: A hospital-based matched case-control study. J. Breast Cancer 2016, 19, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, H.; Tsuta, K.; Kiuchi, K.; Senzaki, H.; Tanaka, K.; Hioki, K.; Tsubura, A. Growth inhibitory effects of diallyl disulfide on human breast cancer cell lines. Carcinogenesis 2001, 22, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Hahm, E.R.; Singh, S.V. Diallyl trisulfide inhibits estrogen receptor-alpha activity in human breast cancer cells. Breast Cancer Res. Treat. 2014, 144, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, K.M.; Lin, G.M.; Zhao, Z.X. Antitumor mechanisms of S-allyl mercaptocysteine for breast cancer therapy. BMC Complement. Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Lee, H.W.; Kim, B.O.; Rhee, D.K.; Pyo, S. Allicin inhibits invasion and migration of breast cancer cells through the suppression of VCAM-1: Regulation of association between p65 and ER-alpha. J. Funct. Foods 2015, 15, 172–185. [Google Scholar] [CrossRef]
- Altonsy, M.O.; Habib, T.N.; Andrews, S.C. Diallyl disulfide-induced apoptosis in a breast-cancer cell line (MCF-7) may be caused by inhibition of histone deacetylation. Nutr. Cancer 2012, 64, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.Y.; Yao, S.Q.; Zu, X.Y.; Huang, Z.X.; Liu, L.J.; Zhong, M.; Zhu, B.Y.; Tang, S.S.; Liao, D.F. Apoptosis induced by diallyl disulfide in human breast cancer cell line MCF-7. Acta Pharmacol. Sin. 2008, 29, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.S.; Chen, B.; Liu, X.P.; Liu, P.; Zheng, G.P.; Ye, F.; Tang, H.L.; Xie, X.M. Diallyl disulfide suppresses SRC/Ras/ERK signaling-mediated proliferation and metastasis in human breast cancer by up-regulating miR-34a. PLoS ONE 2014, 9, e11272011. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Yang, B.; Xiang, T.X.; Peng, W.Y.; Qiu, Z.; Wan, J.Y.; Zhang, L.; Li, H.Y.; Li, H.Z.; Ren, G.S. Diallyl disulfide inhibits growth and metastatic potential of human triple-negative breast cancer cells through inactivation of the β-catenin signaling pathway. Mol. Nutr. Food Res. 2015, 59, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Na, H.K.; Kim, E.H.; Choi, M.A.; Park, J.M.; Kim, D.H.; Surh, Y.J. Diallyl trisulfide induces apoptosis in human breast cancer cells through ROS-mediated activation of JNK and AP-1. Biochem. Pharmacol. 2012, 84, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Malki, A.; El-Saadani, M.; Sultan, A.S. Garlic constituent diallyl trisulfide induced apoptosis in MCF7 human breast cancer cells. Cancer Biol. Ther. 2009, 8, 2174–2184. [Google Scholar] [CrossRef]
- Liu, Y.P.; Zhu, P.T.; Wang, Y.Y.; Wei, Z.H.; Tao, L.; Zhu, Z.J.; Sheng, X.B.; Wang, S.L.; Ruan, J.S.; Liu, Z.G. Antimetastatic therapies of the polysulfide diallyl Ttrisulfide against triple-negative breast cancer (TNBC) via suppressing MMP2/9 by blocking NF-kappa B and ERK/MAPK signaling pathways. PLoS ONE 2015, 10, e1237814. [Google Scholar]
- Kim, S.H.; Kaschula, C.H.; Priedigkeit, N.; Lee, A.V.; Singh, S.V. Forkhead Box Q1 is a novel target of breast cancer stem cell inhibition by diallyl trisulfide. J. Biol. Chem. 2016, 291, 13495–13508. [Google Scholar] [CrossRef] [PubMed]
- Baharetha, H.M.; Nassar, Z.D.; Aisha, A.F.; Ahamed, M.; Al-Suede, F.; Abd Kadir, M.O.; Ismail, Z.; Majid, A. Proapoptotic and antimetastatic properties of supercritical CO2 extract of Nigella sativa Linn. against breast cancer cells. J. Med. Food 2013, 16, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Alhazmi, M.I.; Hasan, T.N.; Shafi, G.; Al-Assaf, A.H.; Alfawaz, M.A.; Alshatwi, A.A. Roles of p53 and caspases in induction of apoptosis in MCF-7 breast cancer cells treated with a methanolic extract of Nigella sativa seeds. Asian Pac. J. Cancer Prev. 2014, 15, 9655–9660. [Google Scholar] [CrossRef] [PubMed]
- Schneider-Stock, R.; Fakhoury, I.H.; Zaki, A.M.; El-Baba, C.O.; Gali-Muhtasib, H.U. Thymoquinone: Fifty years of success in the battle against cancer models. Drug Discov. Today 2014, 19, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Franke, T.F.; Cantley, L.C. Apoptosis. A bad kinase makes good. Nature 1997, 390, 116–117. [Google Scholar] [CrossRef] [PubMed]
- Arafa, E.; Zhu, Q.Z.; Shah, Z.I.; Wani, G.; Barakat, B.M.; Racoma, I.; El-Mandy, M.A.; Wani, A.A. Thymoquinone up-regulates PTEN expression and induces apoptosis in doxorubicin-resistant human breast cancer cells. Mutat. Res. 2011, 706, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.; Kumar, B.; Dey, K.K.; Pal, I.; Parekh, A.; Mandal, M. Molecular targeting of Akt by thymoquinone promotes G(1) arrest through translation inhibition of cyclin D1 and induces apoptosis in breast cancer cells. Life Sci. 2013, 93, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Woo, C.C.; Hsu, A.; Kumar, A.P.; Sethi, G.; Tan, K. Thymoquinone inhibits tumor growth and induces apoptosis in a breast cancer xenograft mouse model: The role of p38 MAPK and ROS. PLoS ONE 2013, 8, e75356. [Google Scholar] [CrossRef] [PubMed]
- Woo, C.C.; Loo, S.Y.; Gee, V.; Yap, C.W.; Sethi, G.; Kumar, A.P.; Tan, K. Anticancer activity of thymoquinone in breast cancer cells: Possible involvement of PPAR-gamma pathway. Biochem. Pharmacol. 2011, 82, 464–475. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.M.; Kim, S.J. Thymoquinone (TQ) regulates cyclooxygenase-2 expression and prostaglandin E2 production through PI3kinase (PI3K)/p38 kinase pathway in human breast cancer cell line, MDA-MB-231. Anim. Cells Syst. 2012, 16, 274–279. [Google Scholar] [CrossRef]
- Chou, C.C.; Wu, Y.C.; Wang, Y.F.; Chou, M.J.; Kuo, S.J.; Chen, D.R. Capsaicin-induced apoptosis in human breast cancer MCF-7 cells through caspase-independent pathway. Oncol. Rep. 2009, 21, 665–671. [Google Scholar] [PubMed]
- Chang, H.C.; Chen, S.T.; Chien, S.Y.; Kuo, S.J.; Tsai, H.T.; Chen, D.R. Capsaicin may induce breast cancer cell death through apoptosis-inducing factor involving mitochondrial dysfunction. Hum. Exp. Toxicol. 2011, 30, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Thoennissen, N.H.; O’Kelly, J.; Lu, D.; Iwanski, G.B.; La, D.T.; Abbassi, S.; Leiter, A.; Karlan, B.; Mehta, R.; Koeffler, H.P. Capsaicin causes cell-cycle arrest and apoptosis in ER-positive and -negative breast cancer cells by modulating the EGFR/HER-2 pathway. Oncogene 2010, 29, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.G.; Zhu, P.T.; Tao, Y.; Shen, C.S.; Wang, S.L.; Zhao, L.G.; Wu, H.Y.; Fan, F.T.; Lin, C.; Chen, C. Cancer-promoting effect of capsaicin on DMBA/TPA-induced skin tumorigenesis by modulating inflammation, Erk and p38 in mice. Food Chem. Toxicol. 2015, 81, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Li, T.Z.; Xu, G.H.; Luo, B.B.; Chen, Y.X.; Zhang, T. Low-concentration capsaicin promotes colorectal cancer metastasis by triggering ROS production and modulating Akt/mTOR and STAT-3 pathways. Neoplasma 2013, 60, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Caprodossi, S.; Amantini, C.; Nabissi, M.; Morelli, M.B.; Farfariello, V.; Santoni, M.; Gismondi, A.; Santoni, G. Capsaicin promotes a more aggressive gene expression phenotype and invasiveness in null-TRPV1 urothelial cancer cells. Carcinogenesis 2011, 32, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Greenshields, A.L.; Doucette, C.D.; Sutton, K.M.; Madera, L.; Annan, H.; Yaffe, P.B.; Knickle, A.F.; Dong, Z.M.; Hoskin, D.W. Piperine inhibits the growth and motility of triple-negative breast cancer cells. Cancer Lett. 2015, 357, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Do, M.T.; Kim, H.G.; Choi, J.H.; Khanal, T.; Park, B.H.; Tran, T.P.; Jeong, T.C.; Jeong, H.G. Antitumor efficacy of piperine in the treatment of human HER2-overexpressing breast cancer cells. Food Chem. 2013, 141, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.H.; Fu, Q.H.; Liu, Y.; Jiang, K.; Guo, Q.M.; Chen, Q.Y.; Yan, B.; Wang, Q.Q.; Shen, J.G. Piperine suppresses tumor growth and metastasis in vitro and in vivo in a 4T1 murine breast cancer model. Acta Pharmacol. Sin. 2012, 33, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Chryssanthi, D.G.; Lamari, F.N.; Iatrou, G.; Pylara, A.; Karamanos, N.K.; Cordopatis, P. Inhibition of breast cancer cell proliferation by style constituents of different Crocus species. Anticancer Res. 2007, 27, 357–362. [Google Scholar] [PubMed]
- Chryssanthi, D.G.; Dedes, P.G.; Karamanos, N.K.; Cordopatis, P.; Lamari, F.N. Crocetin inhibits invasiveness of MDA-MB-231 breast cancer cells via downregulation of matrix metalloproteinases. Planta Med. 2011, 77, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Sajjadi, M.; Bathaie, Z. Comparative study on the preventive effect of saffron carotenoids, crocin and crocetin, in NMU-induced breast cancer in rats. Cell J. 2017, 19, 94–101. [Google Scholar] [PubMed]
- Lin, C.; Lin, S.H.; Lin, C.; Liu, Y.; Chen, C.; Chu, C.; Huang, H.; Lin, M. Inhibitory effect of clove methanolic extract and eugenol on dendritic cell functions. J. Funct. Foods 2016, 27, 439–447. [Google Scholar] [CrossRef]
- Vidhya, N.; Devaraj, S.N. Induction of apoptosis by eugenol in human breast cancer cells. Indian J. Exp. Biol. 2011, 49, 871–878. [Google Scholar] [PubMed]
- Al-Sharif, I.; Remmal, A.; Aboussekhra, A. Eugenol triggers apoptosis in breast cancer cells through E2F1/survivin down-regulation. BMC Cancer 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Vallinas, M.; Molina, S.; Vicente, G.; Sanchez-Martinez, R.; Vargas, T.; Garcia-Risco, M.R.; Fornari, T.; Reglero, G.; de Molina, A.R. Modulation of estrogen and epidermal growth factor receptors by rosemary extract in breast cancer cells. Electrophoresis 2014, 35, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Fuke, Y.; Hishinuma, M.; Namikawa, M.; Oishi, Y.; Matsuzaki, T. Wasabi-derived 6-(methylsulfinyl)hexyl isothiocyanate induces apoptosis in human breast cancer by possible involvement of the NF-kappa B pathways. Nutr. Cancer 2014, 66, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Tang, E.; Rajarajeswaran, J.; Fung, S.Y.; Kanthimathi, M.S. Antioxidant activity of Coriandrum sativum and protection against DNA damage and cancer cell migration. BMC Complement. Altern. Med. 2013, 13, 347. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Verron, E.; Rohanizadeh, R. Molecular mechanisms of anti-metastatic activity of curcumin. Anticancer Res. 2016, 36, 5639–5647. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Yu, J.Y.; Cui, R.; Lin, J.J.; Ding, X.T. Curcumin in treating breast cancer: A review. JALA J. Lab. Autom. 2016, 21, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kadakol, A.; Shasthrula, P.K.; Mundhe, N.A.; Jamdade, V.S.; Barua, C.C.; Gaikwad, A.B. Curcumin as an adjuvant to breast cancer treatment. Anti-Cancer Agents Med. Chem. 2015, 15, 647–656. [Google Scholar] [CrossRef]
- Liu, D.W.; Chen, Z.W. The effect of curcumin on breast cancer cells. J. Breast Cancer 2013, 16, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.A.; Kim, K.; Nam, S.J.; Kong, G.; Kim, M.K. A case-control study on the dietary intake of mushrooms and breast cancer risk among Korean women. Int. J. Cancer 2008, 122, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Zou, L.; Chen, W.; Zhu, B.B.; Shen, N.; Ke, J.T.; Lou, J.; Song, R.R.; Zhong, R.; Miao, X.P. Dietary mushroom intake may reduce the risk of breast cancer: Evidence from a meta-analysis of observational studies. PLoS ONE 2014, 9, e93437. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.Y.; Fong, P.C.; Yu, C.C.; Tsai, W.C.; Tzeng, Y.M.; Chang, W.W. Methyl antcinate A suppresses the population of cancer stem-like cells in MCF7 human breast cancer cell line. Molecules 2013, 18, 2539–2548. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.Y.; Hu, X.P.; Xiong, H.; Qiu, H.; Yuan, X.L.; Zhu, F.; Wang, Y.H.; Zou, Y.M. A polysaccharide from Huaier induced apoptosis in MCF-7 breast cancer cells via down-regulation of MTDH protein. Carbohydr. Polym. 2016, 151, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.L.; Zhao, Y.; Jiao, Y.D.; Shi, T.R.; Yang, X.B. ROS-dependent mitochondria molecular mechanisms underlying antitumor activity of Pleurotus abalonus acidic polysaccharides in human breast cancer MCF-7 cells. PLoS ONE 2013, 8, e64266. [Google Scholar] [CrossRef] [PubMed]
- Sliva, D.; Labarrere, C.; Slivova, V.; Sedlak, M.; Lloyd, F.P.; Ho, N. Ganoderma lucidum suppresses motility of highly invasive breast and prostate cancer cells. Biochem. Biophys. Res. Commun. 2002, 298, 603–612. [Google Scholar] [CrossRef]
- Choong, Y.K.; Noordin, M.M.; Mohamed, S.; Ali, A.M.; Umar, N.; Tong, C.C. The nature of apoptosis of human breast cancer cells induced by three species of genus Ganoderma, P. Karst. (aphyllophoromycetideae) crude extracts. Int. J. Med. Mushrooms 2008, 10, 115–125. [Google Scholar] [CrossRef]
- Grube, S.J.; Eng, E.T.; Kao, Y.C.; Kwon, A.; Chen, S. White button mushroom phytochemicals inhibit aromatase activity and breast cancer cell proliferation. J. Nutr. 2001, 131, 3288–3293. [Google Scholar] [PubMed]
- Xu, J.K.; Yuan, Q.G.; Luo, P.; Sun, X.L.; Ma, J.C. Pleurotus eous polysaccharides suppress angiogenesis and induce apoptosis via ROS-dependent JNK activation and mitochondrial mediated mechanisms in MCF-7 human breast cancer cells. Bangladesh J. Pharmacol. 2015, 10, 78–86. [Google Scholar] [CrossRef]
- Aune, D.; Chan, D.; Greenwood, D.C.; Vieira, A.R.; Rosenblatt, D.; Vieira, R.; Norat, T. Dietary fiber and breast cancer risk: A systematic review and meta-analysis of prospective studies. Ann. Oncol. 2012, 23, 1394–1402. [Google Scholar] [CrossRef] [PubMed]
- Awika, J.M.; Rooney, L.W. Sorghum phytochemicals and their potential impact on human health. Phytochemistry 2004, 65, 1199–1221. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Darvin, P.; Lim, E.J.; Joung, Y.H.; Hong, D.Y.; Park, E.U.; Park, S.H.; Choi, S.K.; Moon, E.S.; Cho, B.W. Hwanggeumchal sorghum induces cell cycle arrest, and suppresses tumor growth and metastasis through Jak2/STAT pathways in breast cancer xenografts. PLoS ONE 2012, 7, e40531. [Google Scholar] [CrossRef] [PubMed]
- Suganyadevi, P.; Saravanakumar, K.M.; Mohandas, S. The antiproliferative activity of 3-deoxyanthocyanins extracted from red sorghum (Sorghum bicolor) bran through P-53-dependent and Bcl-2 gene expression in breast cancer cell line. Life Sci. 2013, 92, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Kubatka, P.; Kello, M.; Kajo, K.; Kruzliak, P.; Vybohova, D.; Smejkal, K.; Marsik, P.; Zulli, A.; Gonciova, G.; Mojzis, J. Young barley indicates antitumor effects in experimental breast cancer in vivo and in vitro. Nutr. Cancer 2016, 68, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Lee, C.W.; Ohm, J.B. In vitro study on effect of germinated wheat on human breast cancer cells. Cereal Chem. 2016, 93, 647–649. [Google Scholar] [CrossRef]
- Xue, J.P.; Wang, G.; Zhao, Z.B.; Wang, Q.; Shi, Y. Synergistic cytotoxic effect of genistein and doxorubicin on drug-resistant human breast cancer MCF-7/Adr cells. Oncol. Rep. 2014, 32, 1647–1653. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, S.; Shyam, H.; Sharma, R.; Balapure, A.K. Genistein synergizes centchroman action in human breast cancer cells. Indian J. Pharmacol. 2016, 48, 637–642. [Google Scholar] [PubMed]
- Charalambous, C.; Pitta, C.A.; Constantinou, A.I. Equol enhances tamoxifen’s anti-tumor activity by induction of caspase-mediated apoptosis in MCF-7 breast cancer cells. BMC Cancer 2013, 13, 238. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, B.; Ghavami, L.; Nikoofar, A.; Goliaei, B. Equol as a potent radiosensitizer in estrogen receptor-positive and -negative human breast cancer cell lines. Breast Cancer 2015, 22, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Kambhampati, S.; Banerjee, S.K.; Haque, I. Pomegranate sensitizes tamoxifen action in ER-alpha positive breast cancer cells. J. Cell Commun. Signal. 2011, 5, 317–324. [Google Scholar] [CrossRef] [PubMed]
- McGuire, K.P.; Ngoubilly, N.; Neavyn, M.; Lanza-Jacoby, S. 3,3′-diindolylmethane and paclitaxel act synergistically to promote apoptosis in HER2/Neu human breast cancer cells. J. Surg. Res. 2006, 132, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.J.; Lv, M.M.; Wang, Y.L.; Zhang, J.G. Development of novel application of 3,3′-diindolylmethane: Sensitizing multidrug resistance human breast cancer cells to gamma-irradiation. Pharm. Biol. 2016, 54, 3164–3168. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K.W.; Ali, S.; Aboukameel, A.; Sarkar, S.H.; Wang, Z.; Philip, P.A.; Sakr, W.A.; Raz, A. Inactivation of NF-κB by 3,3′-diindolylmethane contributes to increased apoptosis induced by chemotherapeutic agent in breast cancer cells. Mol. Cancer Ther. 2007, 6, 2757–2765. [Google Scholar] [CrossRef] [PubMed]
- Ganji-Harsini, S.; Khazaei, M.; Rashidi, Z.; Ghanbari, A. Thymoquinone could increase the efficacy of tamoxifen induced apoptosis in human breast cancer cells: An in vitro study. Cell J. 2016, 18, 245–254. [Google Scholar] [PubMed]
- Sakalar, C.; Izgi, K.; Iskender, B.; Sezen, S.; Aksu, H.; Cakir, M.; Kurt, B.; Turan, A.; Canatan, H. The combination of thymoquinone and paclitaxel shows anti-tumor activity through the interplay with apoptosis network in triple-negative breast cancer. Tumor Biol. 2016, 37, 4467–4477. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamed, S.; Yokoyama, S.; Refaat, A.; Ogura, K.; Yagita, H.; Awale, S.; Saiki, I. Piperine enhances the efficacy of TRAIL-based therapy for triple-negative breast cancer cells. Anticancer Res. 2014, 34, 1893–1899. [Google Scholar] [PubMed]
- Gollapudi, S.; Ghoneum, M. MGN-3/Biobran, modified arabinoxylan from rice bran, sensitizes human breast cancer cells to chemotherapeutic agent, daunorubicin. Cancer Detect. Prev. 2008, 32, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ghoneum, M.; El-Din, N.; Ali, D.A.; El-Dein, M.A. Modified arabinoxylan from rice bran, MGN-3/Biobran, sensitizes metastatic breast cancer cells to paclitaxel in vitro. Anticancer Res. 2014, 34, 81–87. [Google Scholar] [PubMed]
- Bar-Sela, G.; Tsalic, M.; Fried, G.; Goldberg, H. Wheat grass juice may improve hematological toxicity related to chemotherapy in breast cancer patients: A pilot study. Nutr. Cancer 2007, 58, 43–48. [Google Scholar] [CrossRef] [PubMed]

| Natural Product | Study Type | Subject | Outcome | Association | Ref. |
|---|---|---|---|---|---|
| soy | cohort study | 70,578 Chinese women aged 40–70 years | BC risk | overall: HR = 0.78, 95% CI = 0.63–0.97, premenopausal women: HR = 0.46; 95% CI: 0.29–0.74), | [45] |
| ER+/PR+ postmenopausal women: HR = 0.72; 95% CI = 0.53–0.96 | |||||
| ER-/PR− premenopausal women: HR = 0.46; 95% CI = 0.22–0.97 | |||||
| soy | cohort study | 15,607 Japanese women aged 35 or above | BC risk | postmenopausal women: | [46] |
| trend p = 0.023 for soy consumption; | |||||
| trend p = 0.046 for isoflavone consumption | |||||
| soy | prospective study | 649 Chinese women with BC | BC death | HR = 0.71, 95% CI = 0.52–0.98 | [47] |
| BC prognosis | ER+ patients: HR = 0.59, 95% CI = 0.40–0.93 | ||||
| soy | cohort study | affected BC patients and unaffected high risk family members in Korea | BC risk | BRCA2 mutation carriers: | [48] |
| HR = 0.39; 95% CI = 0.19– 0.79 | |||||
| soy | cohort study | 9514 BC survivors | risk of recurrence | HR = 0.75; 95% Cl = 0.61–0.92 | [30] |
| soy | cohort study | 3842 multiethnic women | all-cause mortality | no significant association | [50] |
| BC specific mortality | |||||
| soy | cohort study | 84,450 multiethnic women with BC | BC risk | no significant association | [51] |
| soy | cohort study | 339 Korean women with BC | risk of recurrence | no significant association | [52] |
| Citrus fruits | meta-analysis | 8393 participants: 3789 cases and 4,705 controls | BC risk | OR = 0.90; 95% CI = 0.85–0.96; p < 0.001 | [112] |
| cruciferous vegetables | meta-analysis | 18,673 BC cases | BC risk | RR = 0.85, 95% CI = 0.77–0.94 | [150] |
| garlic | case-control study | 285 Iranian women aged 25–65 years with BC | BC risk | ORs = 0.41, 95% CI = 0.20–0.83 | [211] |
| mushroom | case-control study | 362 women aged 30–65 years with BC | BC risk | postmenopausal women: | [255] |
| for daily intake, OR=0.16, 95% CI = 0.04-0.54, p= 0.0058; | |||||
| for average frequency, OR = 0.17, 95% CI = 0.05–0.54, p = 0.0037 | |||||
| mushroom | meta-analysis | 6890 BC cases | BC risk | premenopausal women: | [256] |
| RR = 0.96, 95% CI = 0.91–1.00; | |||||
| postmenopausal women: | |||||
| RR = 0.94, 95% CI = 0.91–0.97 |
| Natural Product | Constituents | Study Type | Main Effect and Possible Mechanism | Ref. |
|---|---|---|---|---|
| Soy | ||||
| soy | genistein | in vitro | - inducing cell cycle arrest, - improving mitochondrial functionality, - regulating oxidative stress, uncoupling proteins, antioxidant enzymes and sirtuin, - enhancing effects of anticancer drugs | [58,59,60] |
| soy | genistein | in vivo | reducing breast cancer incidence in a high-oestrogen environment | [61] |
| fermented doenjang | NA | in vitro | inducing cell cycle arrest, proliferation inhibition, and apoptosis | [64] |
| soy | genistein | in vitro | inducing apoptosis through: - downregulation of the cancerous inhibitor of protein phosphatase 2A - the inactivation of the IGF-1R/p-Akt signaling pathway | [65,66] |
| soy | 6,7,4′-trihydroxyisoflavone | in vitro | inducing apoptosis and cell cycle arrest at S- and G2/M phases | [67] |
| soybean | NA | in vitro | inducing cell death via activation of caspase-3 and upregulation of proapoptotic molecule expression | [68] |
| soy | genistein | in vitro | inhibiting DNA methylation and increasing expression of tumor suppressor genes | [69] |
| soy | genistein | in vitro | inhibiting cancer cell growth through modulating the DNA damage response and cell cycle | [70] |
| soy | genistein | in vitro | inhibiting cancer cell growth through inhibiting activity of NF-κB via the Nocth-1 signaling pathway | [71] |
| soy | genistein | in vitro and in vivo | decreasing breast cancer stem-like cell population through Hedgehog pathway | [72] |
| soy | daidzein, equol | in vitro | inhibiting the invasion through the down-regulation of MMP-2 expression | [63] |
| Fruits | ||||
| pomegranate | NA | in vitro | inhibiting growth by inducing cell cycle arrest in G2/M and inducing apoptosis | [91] |
| pomegranate | NA | in vivo | preventing mammary tumorigenesis via concurrent disruption of ER and Wnt/-catenin signaling pathways | [92] |
| pomegranate | luteolin, ellagic acid, punicic acid | in vitro | inhibiting growth, increasing adhesion and decreasing migration of breast cancer cells | [94] |
| pomegranate | NA | in vitro and in vivo | showing cytotoxicities by targeting microRNAs155 and 27a, reducing cell proliferation and inducing apoptosis | [95,96] |
| pomegranate | ellagitannins, phenolic acids, conjugated octadecatrienoic acids | in vitro | inhibiting invasion and motility of cancer cells by inhibiting RhoC and RhoA protein expression | [97] |
| pomegranate | ellagitannin-derived compounds | in vitro | inhibiting aromatase activity and cell proliferation | [98] |
| pomegranate | NA | in vitro | inhibiting the cancerous lesion formation | [99,100] |
| mangosteen | NA | in vitro | inhibiting proliferation and inducing apoptosis | [101] |
| mangosteen | phenolics | in vitro | showing cytotoxicities | [102] |
| mangosteen | garcinone D, garcinone E, α-mangostin γ-mangostin | in vitro | dose-dependent anti-aromatase activity | [104] |
| mangosteen | α-mangostin | in vitro | inducing apoptosis through modulating HER2/PI3K/Akt and MAPK signaling pathways | [105] |
| mangosteen | α-mangostin | in vitro | inducing mitochondria-mediated apoptosis and cell cycle alterations | [106] |
| mangosteen | α-mangostin | in vitro | showing cytotoxicities | [107] |
| mangosteen | α-mangostin | in vitro | inhibiting FAS expression and activity, and inducing apoptosis | [108] |
| mangosteen | α-mangostin | in vitro | inducing apoptosis and decreasing the expression of ER alpha and pS2 | [109] |
| mangosteen | α-mangostin | in vivo | increasing survival rates and suppressing tumor volume and the multiplicity of lymph node metastases | [110] |
| in vitro | inducing apoptosis and cell cycle arrest | |||
| mangosteen | panaxanthone | in vivo | suppressing tumor volumes and decreasing the multiplicity of lung metastasis and lymph node metastasis | [111] |
| in vitro | inducing apoptosis | |||
| Citrus fruit | polysaccharides | in vitro | inhibiting angiogenesis and cell migration | [113] |
| Citrus fruit | NA | in vitro | inducing apoptosis | [114] |
| Citrus fruit | NA | in vitro | inducing apoptosis via upregulating the expression of bax and caspase-3 genes and downregulating the expression of bcl-2 gene | [115] |
| Citrus fruit | naringin | in vitro | inhibiting growth potential by targeting β-catenin pathway | [116] |
| in vivo | inhibiting cell proliferation and promoting cell apoptosis and G1 cycle arrest through modulating β-catenin pathway | |||
| Citrus fruit | hesperidin | in vitro | anti-proliferative effect | [117] |
| apple | flavonoids | in vitro | inhibiting growth and inducing apoptosis | [118] |
| apple | polyphenol | in vitro | inhibiting tumorigenesis of pre-neoplastic cells by suppressing colony formation and ERK1/2 phosphorylation | [119] |
| apple | NA | in vitro | inhibiting proliferation and inducing cell cycle arrest at G1 phase | [120] |
| apple | 2α-hydroxyursolic acid | in vitro | inhibit NF-κB activation through suppressing the proteasomal activities | [121,122] |
| apple | 2α-hydroxyursolic acid | in vitro | antiproliferative and pro-apoptotic effect by regulating the p38/MAPK signal transduction pathway | [123] |
| apple | pectic acid | in vitro | inducing apoptosis and inhibiting cell growth | [124] |
| in vivo | preventing tumor metastasis mice via over-expression of P53 | |||
| apple | NA | in vitro | enhancing the anti-proliferative effect of quercetin 3-beta-d-glucoside | [125] |
| grape | polyphenols | in vivo | inhibiting the lungs metastasis | [126] |
| in vitro | inhibiting migration by blocking the PI3k/Akt and MAPK pathways | |||
| grape | NA | in vitro | suppressing migration and invasion | [127] |
| grape | polyphenols | in vitro | inducing membrane damage, disrupting mitochondrial function and inducing G2/M cell cycle arrest | [128] |
| grape | amurensin G | in vitro | inhibiting VEGF production | [129] |
| grape | anthocyanin | in vitro | decreasing invasion, migration and bone turnover, via inhibiting expression of Snail and phosphorylated STAT3 and abrogating Snail-mediated CatL activity | [130] |
| mango | polyphenolics | in vitro | showing cytotoxic effects | [131] |
| in vivo | reducing the tumor volume by regulating the PI3K/AKT pathway and miR-126 | |||
| mango | polyphenols | in vitro | inhibiting cell viability | [132] |
| mango | NA | in vitro | inducing apoptosis via the activation of oxidative stress | [133,134] |
| mango | pyrogallol | in vitro | inhibiting proliferation through mediating the AKT/mTOR signaling pathway | [135] |
| jujube | triterpenic acids | in vitro | inducing apoptotic cell death | [137] |
| jujube | betulinic acid | in vitro | inducing apoptosis through the mitochondria transduction pathway | [138] |
| jujube | NA | in vitro | inhibiting proliferation and inducing apoptosis | [139] |
| strawberry | NA | in vitro | showing cytotoxic effects | [140] |
| in vivo | inhibiting the proliferation of tumor cells by activating apoptosis | |||
| bilberry | NA | in vitro | inhibiting proliferation and inducing apoptosis | [141] |
| jamun fruit | NA | in vitro | inhibiting proliferation and inducing apoptosis | [142] |
| cranberry | NA | in vitro | inducing apoptosis and G1 phase arrest | [143] |
| peach | polyphenolics | in vivo | suppressing tumor growth and lung metastasis by inhibition of metalloproteinases gene expression | [144] |
| plum | phenolics and condensed tannins | in vitro | inducing apoptosis | [145] |
| quince fruit | NA | in vitro | inhibiting proliferation and invasiveness | [146] |
| graviola fruit | NA | in vitro | inhibiting the growth of cancer cells | [147] |
| in vivo | inhibiting tumor growth by 32% (p < 0.01) through the EGFR/ERK signaling pathway | |||
| litchi fruit | NA | in vitro | inhibited cell growth | [148] |
| in vivo | reducing tumor mass volume | |||
| pineapple | bromelain | in vitro | inducing apoptosis | [149] |
| Vegetables | ||||
| Cruciferous vegetables | benzyl isothiocyanate | in vitro | inducing apoptosis which was associated with: - inhibition of mitochondrial fusion - suppression of XIAP expression - generation of ROS | [155,156,157] |
| in vitro and in vivo | suppressing the invasion and migration involving: - suppression of uPA activity and of Akt signaling - suppression on EMT process | [158,160] | ||
| in vitro and in vivo | - inducing FoxO1-mediated autophagic death, - acting against the oncogenic effects of leptin, - suppressing proliferation and neovascularization, - inhibiting breast cancer stem cells | [161,162,163,164] | ||
| Cruciferous vegetables | phenethyl isothiocyanate | in vitro and in vivo | - inducing apoptosis, - suppressing adhesion, aggregation, migration and invasion, - prolonging the tumor-free survival and reducing the tumor incidence, - causing alterations in some genes | [165,166,167,168] |
| Cruciferous vegetables | sulforaphane | in vitro and in vivo | - downregulating ER-α expression, - inducing apoptosis, - inhibiting metastasis, - activating tumor suppressor Egr1, - downregulating telomerase, - regulating p38 MAPK and caspase-7 activations | [169,170,171,172,173,174,175] |
| Cruciferous vegetables | indole-3-carbinol | in vitro and in vivo | suppressing metastasis through - up-regulation of BRCA1 and E-cadherin/catenin complexes - suppressing EMT process and downregulating FAK expression - inhibition on MMP-2 expression - inhibiting CXCR4 and MMP-9 expression by downregulation of the NF-κB signaling pathway | [176,177,178,179] |
| in vitro and in vivo | - regulating the cell cycle progression - downregulating expression of telomerase gene - inducing apoptotic cell death - suppressing the expression of IGF1R and IRS1 - inducing stress fibers and focal adhesion formation - stimulating expression of IFNγR1 | [180,181,182,183,184,185,186,188] | ||
| Cruciferous vegetables | 3,3′-diindolylmethane | in vitro | inducing apoptosis through: - inactivation of Akt and NF-κB activity - downregulating Bcl-2 and upregulating Bax | [189,190,191] |
| in vitro | - inducing cell cycle arrest - lowering the invasive and metastatic potential - stimulating the expression and secretion of IFNγ | [192,193,194,195,196,197] | ||
| red beetroot | betanin | in vitro | showing a dose-dependent cytotoxic effect | [198] |
| Spices | ||||
| ginger | NA | in vitro | inhibiting the proliferation and colony formation | [200] |
| ginger | NA | in vitro | inducing apoptosis and inhibiting expression of c-Myc and hTERT | [201] |
| ginger | 10-gingerol | in vitro | inhibiting proliferation and metastasis, inducing cell cycle arrest | [202] |
| ginger | 6-gingerol | in vitro | inhibiting metastasis by suppressing MMP-2 and -9. | [203] |
| ginger | 6-shogaol | in vitro | inhibiting invasion by reducing MMP-9 expression via blockade of NF-κB activation | [204] |
| inhibiting invasion by suppressing invadopodium formation and MMP activity | [205] | |||
| inhibiting growth and sustainability of spheroid generated from adherent breast cancer cells | [206] | |||
| ginger | 6-dehydrogingerdione | in vitro | inducing apoptosis and cell cycle arrest in G2/M phase | [207] |
| garlic | diallyl disulfide | in vitro | inducing apoptosis though: - inhibition of histone deacetylation - inhibition of ERK and the activation of the SAPK/JNK and p38 pathways | [216,217] |
| in vitro and in vivo | inhibit proliferation and metastasis via: - suppression of the SRC/Ras/ERK pathway - inactivation of the β-catenin signaling pathway | [218,219] | ||
| garlic | diallyl trisulfide | in vitro and in vivo | inducing apoptosis though: - overproduction of ROS and subsequent activation of JNK and AP-1 - upregulating FAS, Bax and p53, and down-regulating Akt and Bcl-2 | [220,221] |
| in vitro | - inhibiting migration and invasion; - inhibiting ER-α - decreasing protein level of FoxQ1 | [213,222,223] | ||
| garlic | S-allyl mercaptocysteine | in vitro | inducing mitochondrial apoptosis and cell cycle arrest | [214] |
| garlic | allicin | in vitro | inhibiting invasion and metastasis | [215] |
| black cumin | extracts | in vitro | inducing apoptosis and inhibiting metastasis | [224,225] |
| black cumin | thymoquinone | in vitro and in vivo | inducing apoptosis through: - inhibiting Akt phosphorylation - inducing p38 phosphorylation via ROS generation | [228,229,230] |
| in vitro | inhibiting proliferation by modulation of the PPAR-γ activation pathway | [231] | ||
| in vitro | regulating COX-2 and E2 | [232] | ||
| red chili pepper | capsaicin | in vitro and in vivo | - inducing apoptosis - inhibiting growth and migration | [233,234,235] |
| black pepper | piperine | in vitro and in vivo | - inhibiting growth, motility and metastasis - inducing apoptosis - suppressing the lung metastasis | [239,240,241] |
| saffron | crocetin | in vitro | inhibiting proliferation and invasion, through decreasing MMP expression | [243] |
| clove | eugenol | in vitro and in vivo | inhibiting growth and proliferation, inducing apoptosis through targeting the E2F1/survivin pathway | [246,247] |
| rosemary | extracts | in vitro | exerting antitumor activity through mediation of ER-α and HER2 signalings | [248] |
| wasabi | 6-(methylsulfinyl)hexyl isothiocyanate | in vivo | inducing apoptosis by inhibiting NF-κB and regulating the PI3K/AKT pathway | [249] |
| coriander | root extract | in vitro | affecting antioxidant enzymes, inducing G2/M phase arrest and apoptosis | [250] |
| Edible Macro-Fungi | ||||
| Antrodia camphorate | methyl antcinate A | in vitro | suppressing the population of cancer stem-like cells | [257] |
| Trametes robiniophila Murr. | polysaccharides | in vitro | induced apoptosis through down-regulation of metadherin | [258] |
| Pleurotus abalonus | polysaccharides | in vitro | inhibiting antiproliferation and inducing apoptosis via ROS-mediated mitochondrial apoptotic pathway | [259] |
| Ganoderma lucidum | extracts | in vitro | inhibiting invasion via inhibiting the expression of uPA and uPA receptor | [260] |
| causing both apoptosis and necrosis | [261] | |||
| Agaricus bisporus | extracts | in vitro | suppressing the aromatase activity dose-dependently | [262] |
| Pleutorus eous | polysaccharides | in vitro | inhibiting angiogenesis and inducing apoptosis | [263] |
| Cereals | ||||
| Sorghum | extracts | in vivo | suppressing tumor growth, inducing cell cycle arrest, and inhibiting metastasis | [266] |
| 3-deoxyanthocyanin | in vitro | inducing apoptosis by upregulating the p53 gene and downregulating the Bcl-2 gene | [267] | |
| barley | extracts | in vitro and in vivo | exerting antiproliferative and pro-apoptotic activities | [268] |
| wheat | germinated wheat flour | in vitro | inhibiting growth and inducing apoptosis | [269] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Li, S.; Meng, X.; Gan, R.-Y.; Zhang, J.-J.; Li, H.-B. Dietary Natural Products for Prevention and Treatment of Breast Cancer. Nutrients 2017, 9, 728. https://doi.org/10.3390/nu9070728
Li Y, Li S, Meng X, Gan R-Y, Zhang J-J, Li H-B. Dietary Natural Products for Prevention and Treatment of Breast Cancer. Nutrients. 2017; 9(7):728. https://doi.org/10.3390/nu9070728
Chicago/Turabian StyleLi, Ya, Sha Li, Xiao Meng, Ren-You Gan, Jiao-Jiao Zhang, and Hua-Bin Li. 2017. "Dietary Natural Products for Prevention and Treatment of Breast Cancer" Nutrients 9, no. 7: 728. https://doi.org/10.3390/nu9070728
APA StyleLi, Y., Li, S., Meng, X., Gan, R.-Y., Zhang, J.-J., & Li, H.-B. (2017). Dietary Natural Products for Prevention and Treatment of Breast Cancer. Nutrients, 9(7), 728. https://doi.org/10.3390/nu9070728







