Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
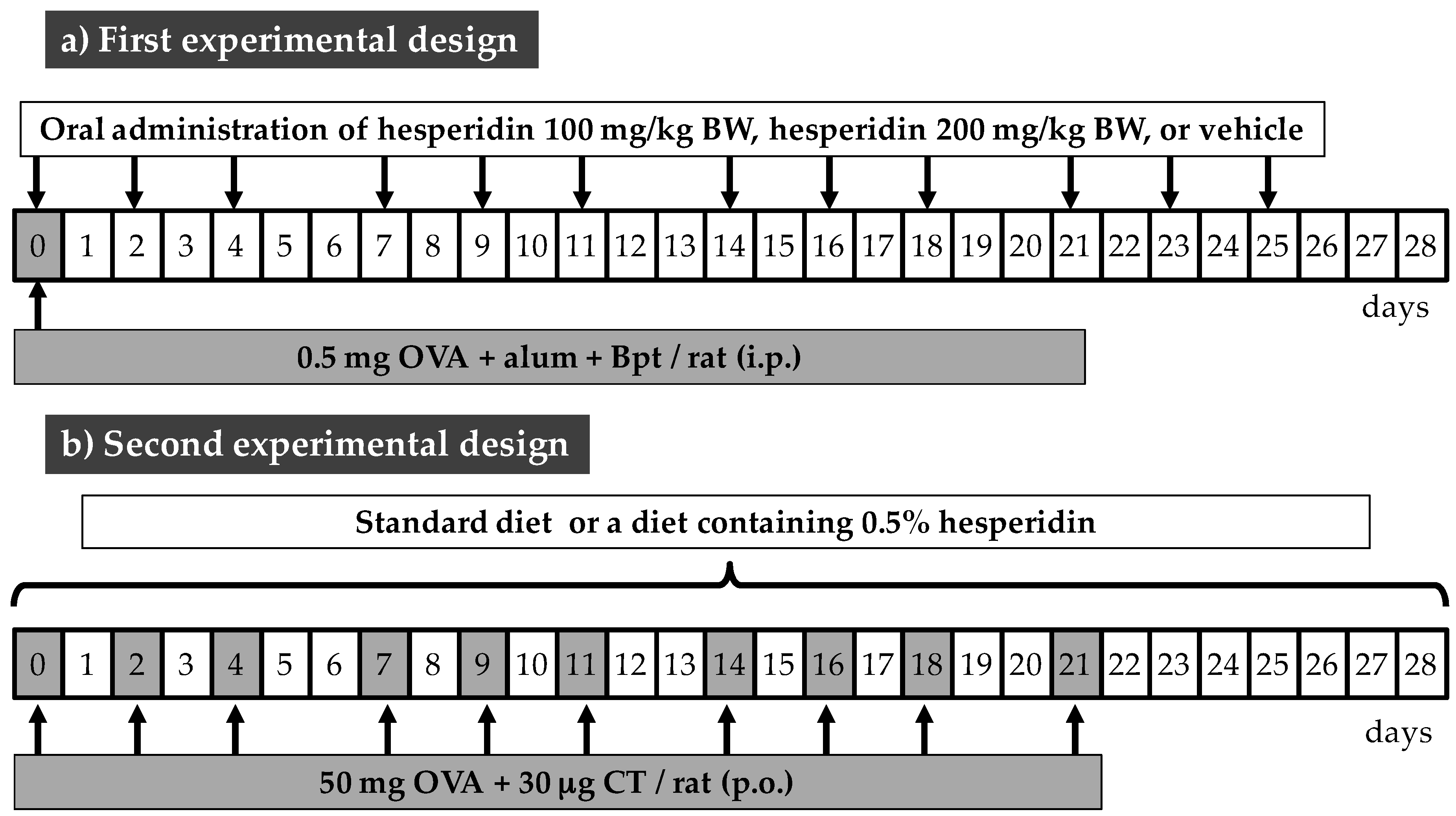
2.2. Animals and Experimental Designs
2.3. Sample Collection and Processing
2.4. Lymphocyte Phenotypic Analysis
2.5. Specific Anti-OVA Antibodies and Intestinal IgA Quantification
2.6. Cytokine Quantification
2.7. Statistical Analysis
3. Results
3.1. Effect of Hesperidin on Food and Water Intake and Body Weight
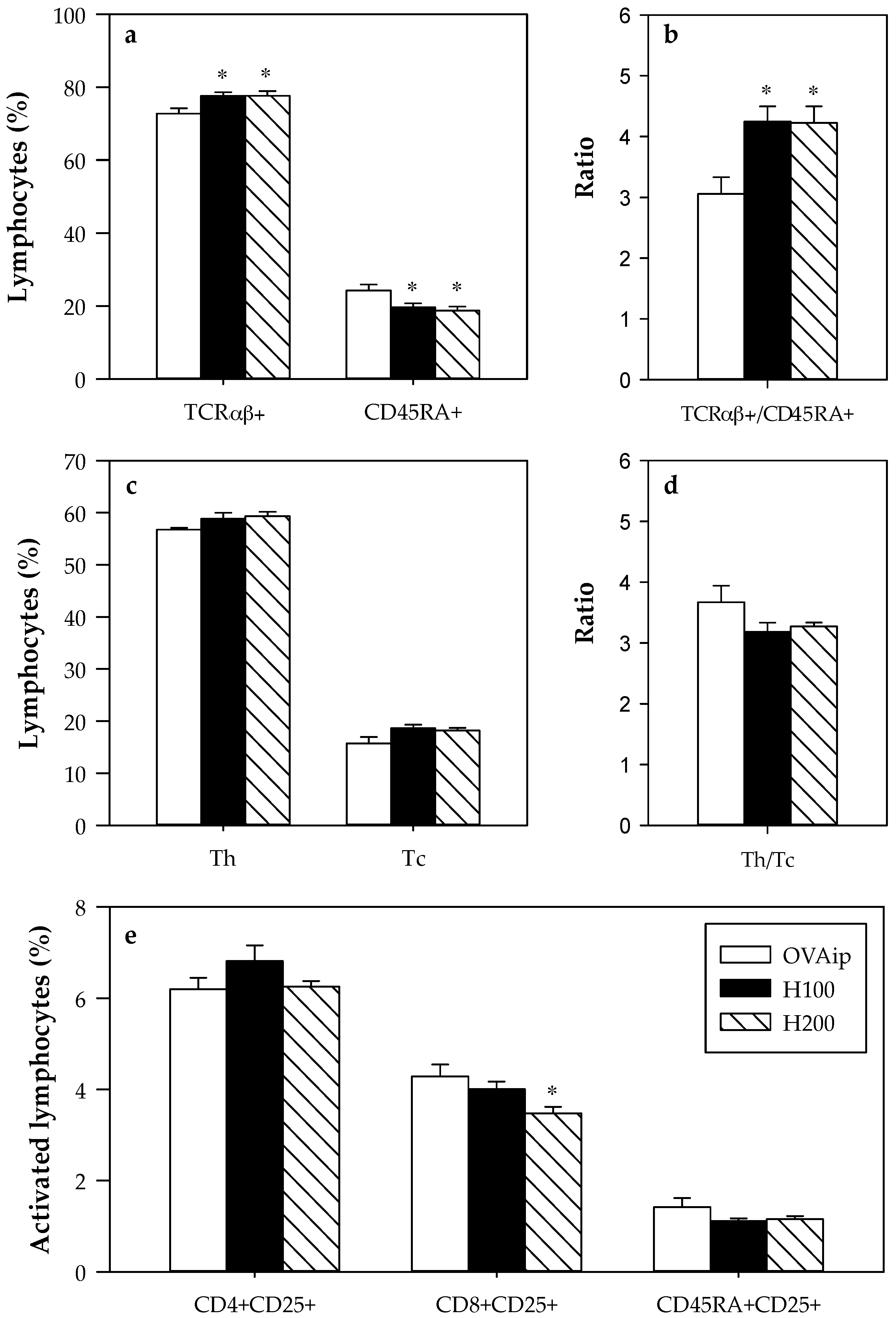
3.2. Effect of 100–200 mg/kg Hesperidin on Mesenteric Lymph Node Lymphocyte Composition and Functionality
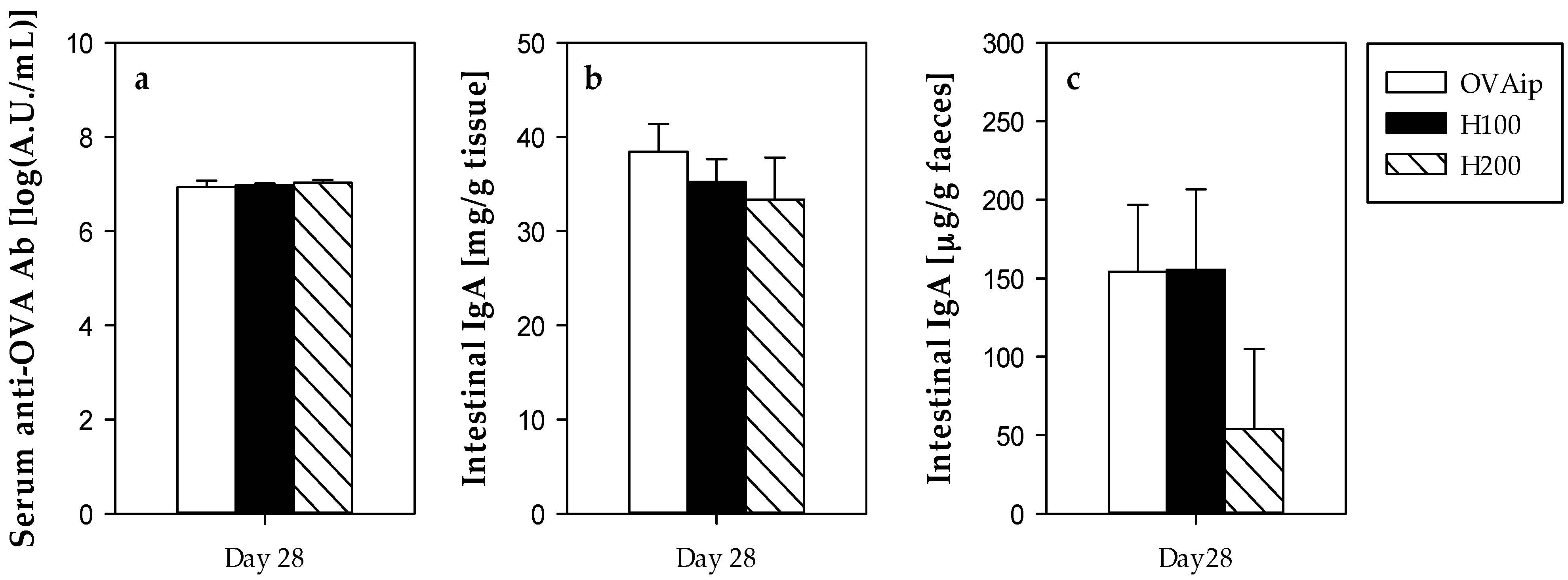
3.3. Effect of 100–200 mg/kg Hesperidin on Antibody Synthesis and Intestinal IgA
3.4. Effect of 0.5% Hesperidin on Intestinal Lymphocyte Composition
3.5. Effect of 0.5% Hesperidin on Antibody Synthesis and Total IgA
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garg, A.; Garg, S.; Zaneveld, L.J.D.; Singla, A.K. Chemistry and pharmacology of the citrus bioflavonoid hesperidin. Phyther. Res. 2001, 15, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, A.; Saavedra, N.; Salazar, L.A.; Abdalla, D.S.P. Modulation of immune function by polyphenols: possible contribution of epigenetic factors. Nutrients 2013, 5, 2314–2332. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Gabriele, M.; Frassinetti, S.; Caltavuturo, L.; Montero, L.; Dinelli, G.; Longo, V.; Di Gioia, D.; Pucci, L. Citrus bergamia powder: Antioxidant, antimicrobial and anti-inflammatory properties. J. Funct. Foods 2017, 31, 255–265. [Google Scholar] [CrossRef]
- Scalbert, A.; Manach, C.; Morand, C.; Rémésy, C. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [PubMed]
- Pérez-Cano, F.J.; Franch, À.; Pérez-Berezo, T.; Ramos-Romero, S.; Castellote, C.; Castell, M. The effects of flavonoids on the immune system. Bioact. Food Diet. Interv. Arthr. Relat. Inflamm. Dis. 2014, 12, 205–210. [Google Scholar]
- Castell, M.; Perez-Cano, F.; Abril-Gil, M.; Franch, À. Flavonoids on allergy. Curr. Pharm. Des. 2014, 20, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Kim, B.-K.; Lee, Y.-C. Antiasthmatic effects of hesperidin, a potential Th2 cytokine antagonist, in a mouse model of allergic asthma. Med. Inflamm. 2011, 2011, 485402. [Google Scholar] [CrossRef] [PubMed]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and Anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: an updated review of their molecular mechanisms and experimental models. Phyther. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, K.; Mizuno, T.; Aida, K.; Uchino, K. Hesperidin as an inhibitor of lipases from porcine pancreas and Pseudomonas. Biosci. Biotechnol. Biochem. 1997, 61, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Tanabe, S.; Sugiyama, M.; Konishi, Y. Transepithelial transport of hesperetin and hesperidin in intestinal Caco-2 cell monolayers. Biochim. Biophys. Acta Biomembr. 2008, 1778, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Hemanth Kumar, B.; Dinesh Kumar, B.; Diwan, P.V. Hesperidin, a citrus flavonoid, protects against l-methionine-induced hyperhomocysteinemia by abrogation of oxidative stress, endothelial dysfunction and neurotoxicity in Wistar rats. Pharm. Biol. 2017, 55, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Hasegawa, J.; Cai, X.; Yang, J.; Ishihara, Y.; Ping, B.; Tsuno, S.; Endo, Y.; Matsuda, A.; Miura, N. Effects of hesperidin on the progression of hypercholesterolemia and fatty liver induced by high-cholesterol diet in rats. Yonago Acta Med. 2016, 59, 67–80. [Google Scholar] [PubMed]
- Chiba, H.; Kim, H.; Matsumoto, A.; Akiyama, S.; Ishimi, Y.; Suzuki, K.; Uehara, M. Hesperidin prevents androgen deficiency-induced bone loss in male mice. Phyther. Res. 2014, 28, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Ikemura, M.; Sasaki, Y.; Giddings, J.C.; Yamamoto, J. Preventive effects of hesperidin, glucosyl hesperidin and naringin on hypertension and cerebral thrombosis in stroke-prone spontaneously hypertensive Rats. Phyther. Res. 2012, 26, 1272–1277. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Ci, X.; Chu, X.; Wei, M.; Hua, S.; Deng, X. Hesperidin suppresses ovalbumin-induced airway inflammation in a mouse allergic asthma model. Inflammation 2012, 35, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Dourado, G.K.Z.S.; Ribeiro, L.C.D.A.; Carlos, I.Z.; César, T.B. Orange juice and hesperidin promote differential innate immune response in macrophages ex vivo. Int. J. Vitam. Nutr. Res. 2014, 83, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Abril-Gil, M.; Massot-Cladera, M.; Pérez-Cano, F.J.; Castellote, C.; Franch, A.; Castell, M. A diet enriched with cocoa prevents IgE synthesis in a rat allergy model. Pharmacol. Res. 2012, 65, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Abril-Gil, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Induction of an oral sensitization model in rats. Clin. Immunol. Endocr. Metab. Drugs 2014, 1, 1–10. [Google Scholar] [CrossRef]
- Camps-Bossacoma, M.; Pérez-Cano, F.J.; Franch, À.; Untermayr, E.; Castell, M. Effect of a cocoa diet on the small intestine and gut-associated lymphoid tissue composition in a rat oral sensitization model. J. Nutr. Biochem. 2017, 42, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Abril-Gil, M.; Saldaña-Ruiz, S.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Cocoa diet prevents antibody synthesis and modifies lymph node composition and functionality in a rat oral sensitization model. Nutrients 2016, 8, 242. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Somani, R. Antioxidant potential of hesperidin protects gentamicin induced nephrotoxicity in experimental rats. Austin J. Pharmacol. Ther. 2015, 3, 1071. [Google Scholar]
- Eisenbarth, S.C. Use and limitations of alum-based models of allergy. Clin. Exp. Allergy 2008, 38, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Selgrade, M.J.K.; Gilmour, M.I. Systemic administration of Bordetella pertussis enhances pulmonary sensitization to house dust mite in juvenile rats. Toxicol. Sci. 2003, 72, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Pilegaard, K.; Madsen, C. An oral Brown Norway rat model for food allergy: Comparison of age, sex, dosing volume, and allergen preparation. Toxicology 2004, 196, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Hassanain, E.; Silverberg, J.I.; Norowitz, K.B.; Chice, S.; Bluth, M.H.; Brody, N.; Joks, R.; Durkin, H.G.; Smith-Norowitz, T.A. Green tea (Camelia sinensis) suppresses B cell production of IgE without inducing apoptosis. Ann. Clin. Lab. Sci. 2010, 40, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, T.; Ito, K.; Ikeda, Y.; Kizaki, M. Green tea component, catechin, induces apoptosis of human malignant B cells via production of reactive oxygen species. Clin. Cancer Res. 2005, 11, 6040–6049. [Google Scholar] [CrossRef] [PubMed]
- Domínguez Ortega, J.; León, F.; Martínez Alonso, J.C.; Alonso Llamazares, A.; Roldán, E.; Robledo, T.; Mesa, M.; Bootello, A.; Martínez-Cócera, C. Fluorocytometric analysis of induced sputum cells in an asthmatic population. J. Investig. Allergol. Clin. Immunol. 2004, 14, 108–113. [Google Scholar] [PubMed]
- Chung, F. Anti-inflammatory cytokines in asthma and allergy: Interleukin-10, interleukin-12, interferon-γ. Med. Inflamm. 2001, 10, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.K.; Fonseca, B.P.; Barboza, B.A.; Viola, J.P. The role of interferon-gamma on immune and allergic responses. Mem. Inst. Oswaldo Cruz 2005, 100, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, M.N.; Habauzit, V.; Trzeciakiewicz, A.; Morand, C.; Gil-Izquierdo, A.; Mardon, J.; Lebecque, P.; Davicco, M.J.; Chee, W.S.S.; Coxam, V.; et al. Hesperidin inhibits ovariectomized-induced osteopenia and shows differential effects on bone mass and strength in young and adult intact rats. J. Appl. Physiol. 2008, 104, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Habauzit, V.; Sacco, S.M.; Gil-Izquierdo, A.; Trzeciakiewicz, A.; Morand, C.; Barron, D.; Pinaud, S.; Offord, E.; Horcajada, M.N. Differential effects of two citrus flavanones on bone quality in senescent male rats in relation to their bioavailability and metabolism. Bone 2011, 49, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Sato, Y.; Watanabe, T.; Nagaoka, M.H.; Yoshioka, Y.; Shoji, T.; Kanda, T.; Yamada, K.; Totsuka, M.; Teshima, R.; et al. Dietary unripe apple polyphenol inhibits the development of food allergies in murine models. FEBS Lett. 2005, 579, 4485–4491. [Google Scholar] [CrossRef] [PubMed]
- Hänninen, A.; Harrison, L.C. Gamma delta T cells as mediators of mucosal tolerance: The autoimmune diabetes model. Immunol. Rev. 2000, 173, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Singh, A.K.; Shilpi Lal, G. Phenotypic and functional plasticity of gamma-delta (γδ) T cells in inflammation and tolerance. Int. Rev. Immunol. 2014, 33, 537–558. [Google Scholar] [CrossRef] [PubMed]
- Bol-Schoenmakers, M.; Marcondes Rezende, M.; Bleumink, R.; Boon, L.; Man, S.; Hassing, I.; Fiechter, D.; Pieters, R.H.H.; Smit, J.J. Regulation by intestinal γδ T cells during establishment of food allergic sensitization in mice. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Frossard, C.P.; Asigbetse, K.E.; Burger, D.; Eigenmann, P.A. Gut T cell receptor-γδ+ intraepithelial lymphocytes are activated selectively by cholera toxin to break oral tolerance in mice. Clin. Exp. Immunol. 2015, 180, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Agace, W.W.; Higgins, J.M.; Sadasivan, B.; Brenner, M.B.; Parker, C.M. T-lymphocyte-epithelial-cell interactions: Integrin αE(CD103)β7, LEEP-CAM and chemokines. Curr. Opin. Cell Biol. 2000, 12, 563–568. [Google Scholar] [CrossRef]
- Annacker, O.; Coombes, J.L.; Malmstrom, V.; Uhlig, H.H.; Bourne, T.; Johansson-Lindbom, B.; Agace, W.W.; Parker, C.M.; Powrie, F. Essential role for CD103 in the T cell-mediated regulation of experimental colitis. J. Exp. Med. 2005, 202, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Han, Y.; Kayahara, M.; Watanabe, T.; Arishige, H.; Kato, N. Consumption of curcumin elevates fecal immunoglobulin A, an index of intestinal immune function, in rats fed a high-fat diet. J. Nutr. Sci. Vitaminol. 2010, 56, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Taira, T.; Yamaguchi, S.; Takahashi, A.; Okazaki, Y.; Yamaguchi, A.; Sakaguchi, H.; Chiji, H. Dietary polyphenols increase fecal mucin and immunoglobulin A and ameliorate the disturbance in gut microbiota caused by a high fat diet. J. Clin. Biochem. Nutr. 2015, 57, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.H. Anti-inflammatory effects and its mechanisms of hesperidin in an asthmatic mouse model induced by ovalbumin. J. Exp. Biomed. Sci. 2010, 16, 83–90. [Google Scholar]

| Food Intake (g/100 g BW/Day) | Water Intake (mL/100 g BW/Day) | |||||
|---|---|---|---|---|---|---|
| OVAip | H100 | H200 | OVAip | H100 | H200 | |
| Day 4 | 13.06–13.97 | 12.80–13.07 | 13.59–13.92 | 11.73–11.74 | 11.52–15.78 | 11.95–12.11 |
| Day 11 | 13.51–13.57 | 7.75–8.64 | 10.20–10.47 | 11.31–12.66 | 11.26–12.39 | 12.15–12.74 |
| Day 18 | 11.60–12.03 | 11.94–12.02 | 11.82–12.24 | 10.39–11.03 | 11.07–13.83 | 11.42–12.11 |
| Day 25 | 9.32–9.34 | 9.35–9.35 | 9.07–9.08 | 11.01–11.20 | 12.68–14.55 | 13.19–14.85 |
| Day 28 | 9.58–10.16 | 9.38–10.03 | 9.73–9.85 | 11.77–12.87 | 13.95–15.61 | 14.05–15.05 |
| Food Intake (g/100 g BW/Day) | Water Intake (mL/100 g BW/Day) | |||
|---|---|---|---|---|
| OVAoral | H0.5 | OVAoral | H0.5 | |
| Day 7 | 10.93–11.37 | 10.74–10.97 | 16.84–24.85 | 14.93–20.67 |
| Day 14 | 11.65–11.70 | 11.28–11.52 | 11.39–14.45 | 11.05–16.23 |
| Day 21 | 10.25–10.60 | 10.76–10.77 | 9.01–10.57 | 9.95–14.33 |
| Day 28 | 8.33–8.55 | 7.90–8.57 | 9.57–10.52 | 8.24–12.44 |
| MLNL | PPL | IEL | LPL | |||||
|---|---|---|---|---|---|---|---|---|
| Lymphocytes (%) | OVAoral | H0.5 | OVAoral | H0.5 | OVAoral | H0.5 | OVAoral | H0.5 |
| CD45RA+ | 12.30 ± 0.38 | 12.20 ± 0.29 | 58.90 ± 4.29 | 59.81 ± 3.81 | 10.54 ± 2.88 | 10.89 ± 0.88 | 25.86 ± 6.25 | 46.10 ± 6.11 * |
| TCRαβ+ | 77.21 ± 0.66 | 77.15 ± 0.72 | 16.16 ± 0.61 | 17.74 ± 1.12 | 41.59 ± 2.27 | 37.72± 3.13 | 29.65 ± 4.12 | 21.00 ± 2.78 |
| TCRγδ+ | 1.50 ± 0.04 | 1.45 ± 0.03 | 0.67 ± 0.09 | 0.92 ± 0.10 | 11.60 ± 1.21 | 16.22 ± 1.63 * | 3.81 ± 0.60 | 1.32 ± 0.40 * |
| TCRγδ+CD8αα+ | 73.12 ± 4.60 | 80.50 ± 3.39 | ||||||
| TCRγδ+CD8αβ+ | 26.88 ± 4.60 | 19.49 ± 3.39 | ||||||
| NK | 0.32 ± 0.03 | 0.26 ± 0.03 | 3.31 ± 0.45 | 2.92 ± 0.27 | 24.33 ± 2.52 | 22.24 ± 1.82 | 13.81 ± 2.14 | 3.97 ± 1.06 * |
| TCRαβ+CD4+ | 77.03 ± 0.66 | 77.22 ± 0.62 | 65.28 ± 1.38 | 68.69 ± 1.76 | 22.10 ± 6.99 | 13.07 ± 3.60 | 60.45 ± 6.92 | 80.08 ± 5.55 * |
| TCRαβ+CD8+ | 22.76 ± 0.69 | 22.58 ± 0.49 | 24.73 ± 1.17 | 23.37 ± 1.32 | 71.88 ± 6.43 | 81.00 ± 3.45 | 34.63 ± 5.99 | 18.09 ± 1.45 * |
| TCRαβ+NK | 0.61 ± 0.02 | 0.58 ± 0.04 | 6.95 ± 0.98 | 5.33 ± 0.59 | 4.95 ± 0.63 | 4.47 ± 0.54 | 3.47 ± 0.76 | 1.46 ± 0.23 * |
| TLR4+ | 37.76 ± 2.33 | 33.04 ± 2.74 | 5.43 ± 0.65 | 7.83 ± 1.12 | 6.88 ± 1.60 | 7.39 ± 2.41 | ||
| CD45RA+TLR4+ | 41.41 ± 5.34 | 32.46 ± 4.10 | ||||||
| CD45RA+CD25+ | 3.96 ± 0.32 | 3.65 ± 0.61 | ||||||
| CD4+CD25+ | 5.58 ± 0.15 | 5.38 ± 0.21 | 10.79 ± 0.96 | 9.22 ± 0.86 | ||||
| CD4+CD62L+ | 66.99 ± 2.15 | 64.42 ± 4.29 | 5.99 ± 3.46 | 1.75 ± 0.81 | 1.77 ± 1.09 | 0.81 ± 0.21 | ||
| CD4+CD103+ | 91.09 ± 4.56 | 96.03 ± 0.98 | 19.02 ± 2.30 | 9.62 ± 1.59 * | ||||
| CD4+TLR4+ | 13.41 ± 2.12 | 11.15 ± 1.43 | ||||||
| CD8+CD25+ | 3.95 ± 0.13 | 4.21 ± 0.32 | 53.01 ± 0.98 | 54.87 ± 3.98 | ||||
| CD8+CD62L+ | 63.78 ± 2.99 | 60.35 ± 5.99 | 38.92 ± 8.70 | 19.18 ± 7.76 | 1.34 ± 0.49 | 1.77 ± 0.61 | ||
| CD8+CD103+ | 11.83 ± 2.74 | 15.36 ± 2.37 | 77.70 ± 5.86 | 46.69 ± 11.97 * | ||||
| CD8+TLR4+ | 36.48 ± 5.30 | 34.03 ± 3.89 | ||||||
| Group | ||
|---|---|---|
| OVAoral | H0.5 | |
| Serum total Ig anti-OVA [log(U.A./mL)] | 3.45 ± 0.32 | 3.16 ± 0.34 |
| Serum IgG1 anti-OVA [log(U.A./mL)] | 1.52 ± 0.26 | 1.86 ± 0.29 |
| Serum IgG2a anti-OVA [log(U.A./mL)] | 1.40 ± 0.34 | 1.21 ± 0.50 |
| Serum IgG2b anti-OVA [log(U.A./mL)] | 1.48 ± 0.05 | 1.65 ± 0.23 |
| Serum IgG2c anti-OVA | ND | ND |
| Serum IgE anti-OVA | ND | ND |
| Serum total IgA (µg/mL) | 5.72 ± 0.15 | 5.36 ± 0.24 |
| Intestinal total IgA (µg/g faeces) | 34.60 ± 6.79 | 58.07 ± 7.61 * |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camps-Bossacoma, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response. Nutrients 2017, 9, 580. https://doi.org/10.3390/nu9060580
Camps-Bossacoma M, Franch À, Pérez-Cano FJ, Castell M. Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response. Nutrients. 2017; 9(6):580. https://doi.org/10.3390/nu9060580
Chicago/Turabian StyleCamps-Bossacoma, Mariona, Àngels Franch, Francisco J. Pérez-Cano, and Margarida Castell. 2017. "Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response" Nutrients 9, no. 6: 580. https://doi.org/10.3390/nu9060580
APA StyleCamps-Bossacoma, M., Franch, À., Pérez-Cano, F. J., & Castell, M. (2017). Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response. Nutrients, 9(6), 580. https://doi.org/10.3390/nu9060580








