Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice
Abstract
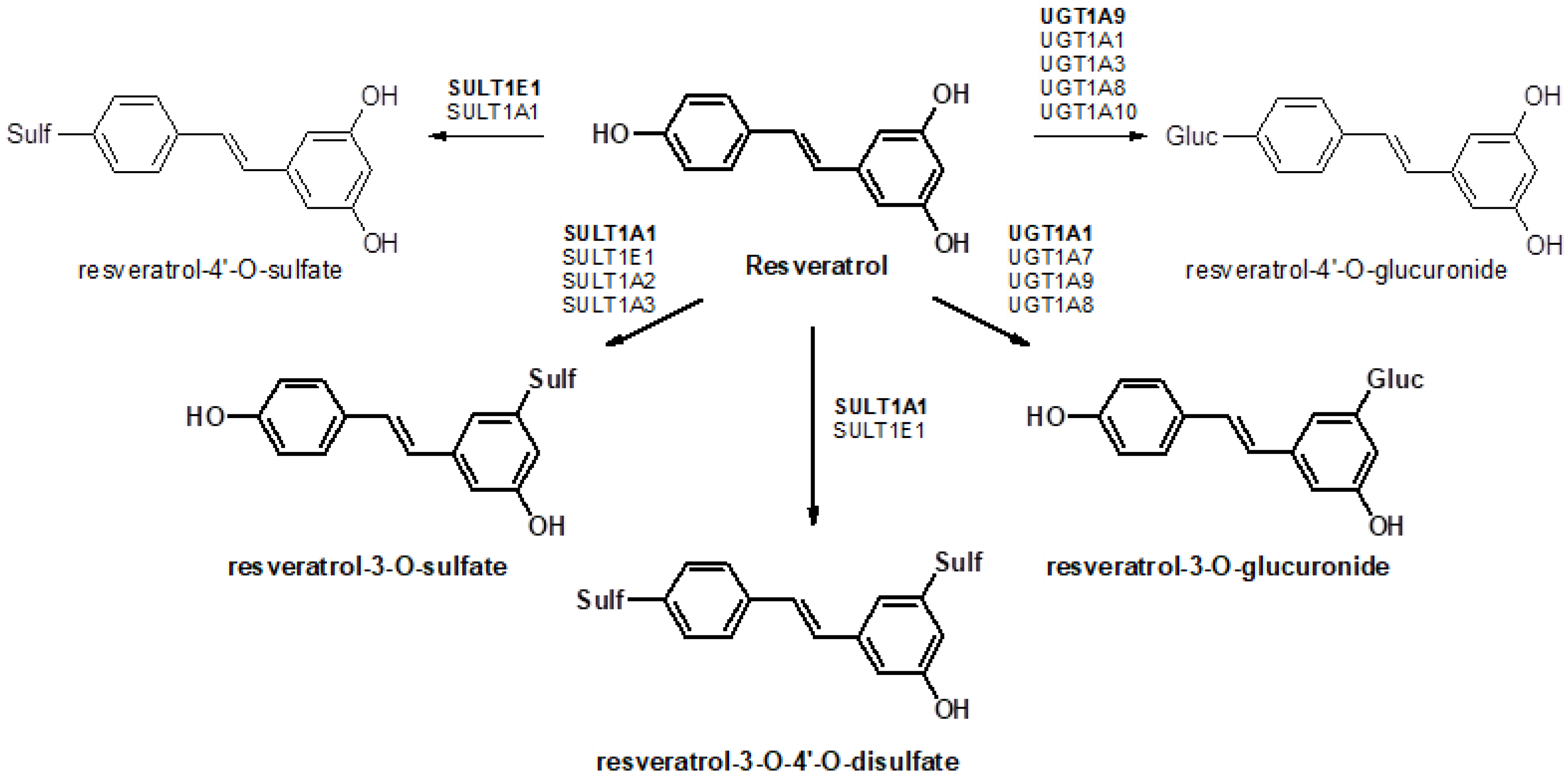
:1. Introduction
2. Material and Methods
2.1. Chemicals
2.2. Animal Experiments
2.3. Sample Preparation
2.4. High-Performance Liquid Chromatography (HPLC) Analysis
2.5. Reverse Transcription Quantitative PCR (RT-qPCR)
2.6. Data Analysis
3. Results
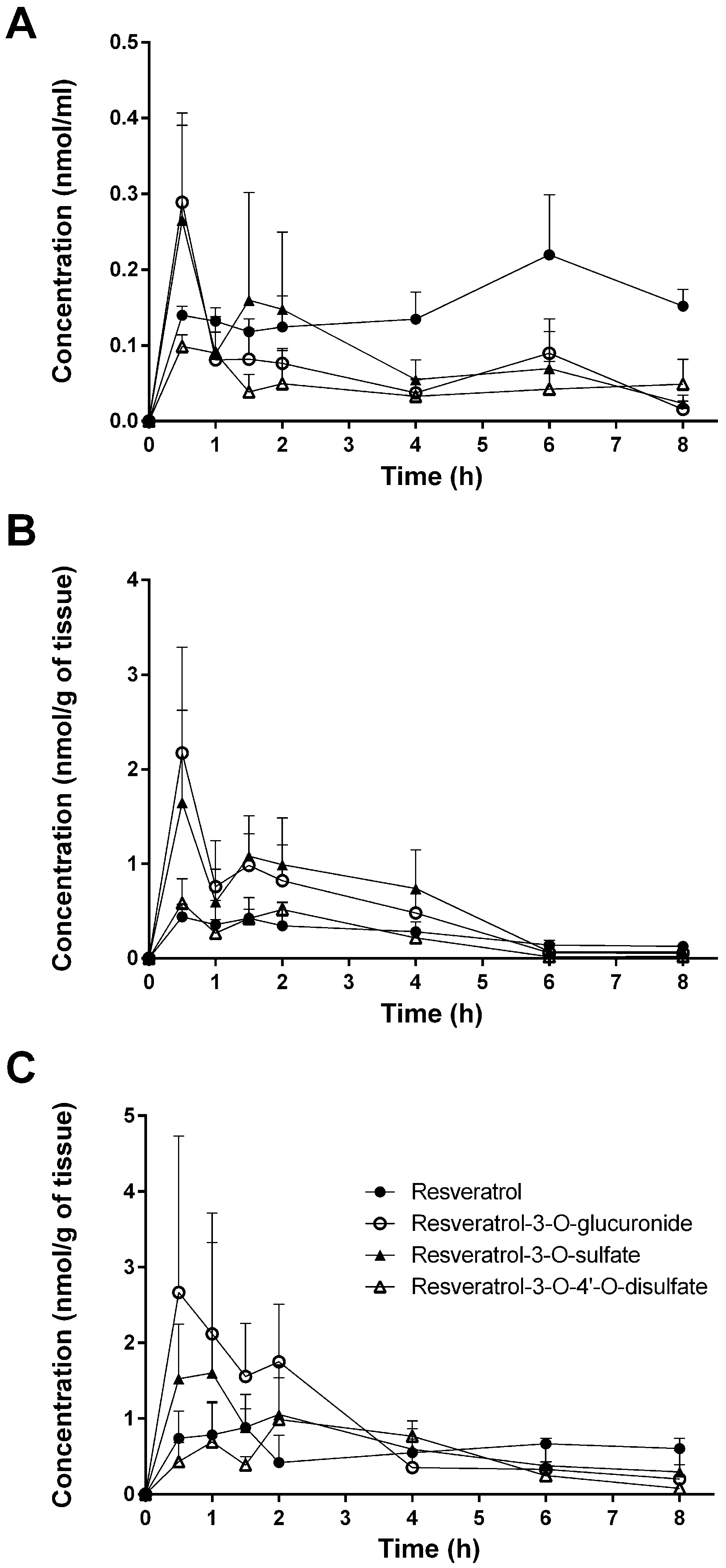
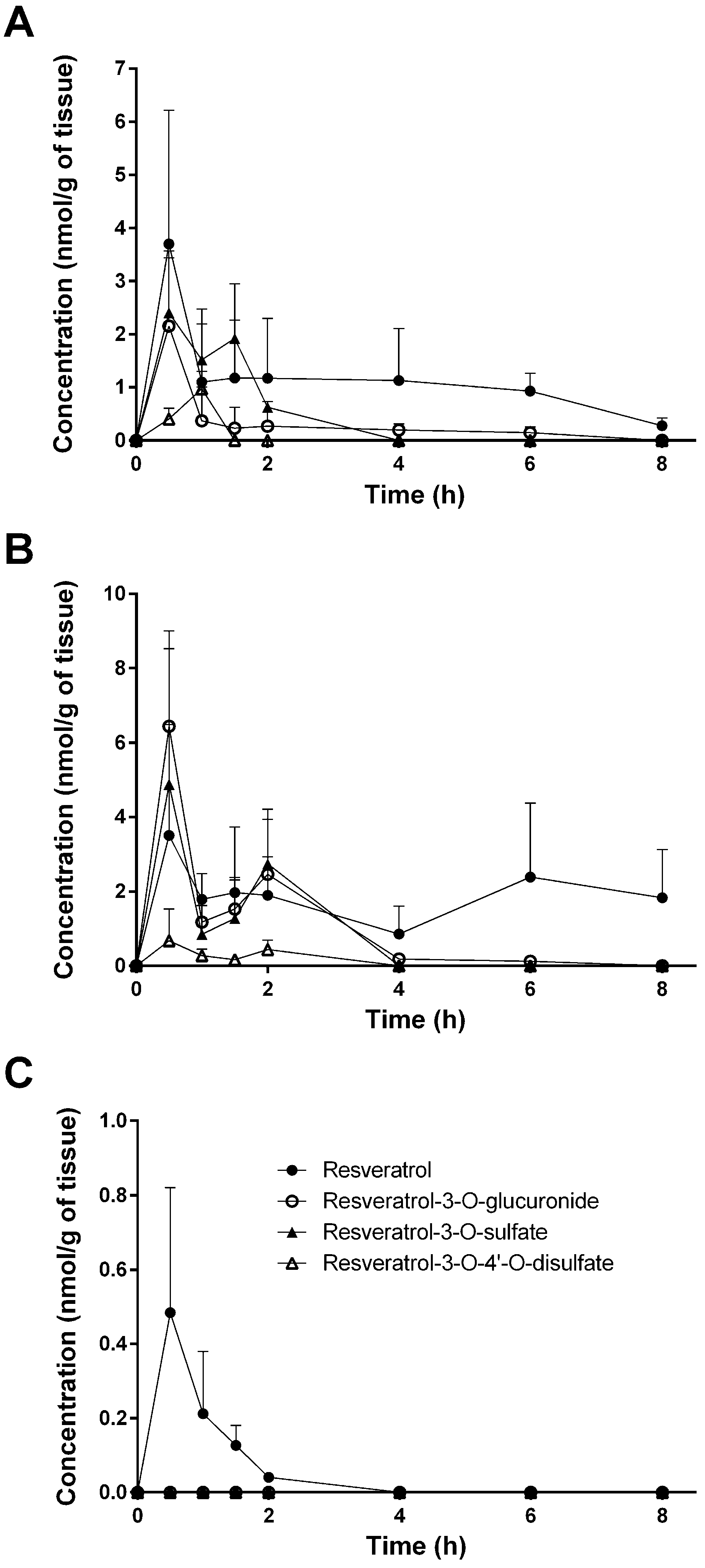
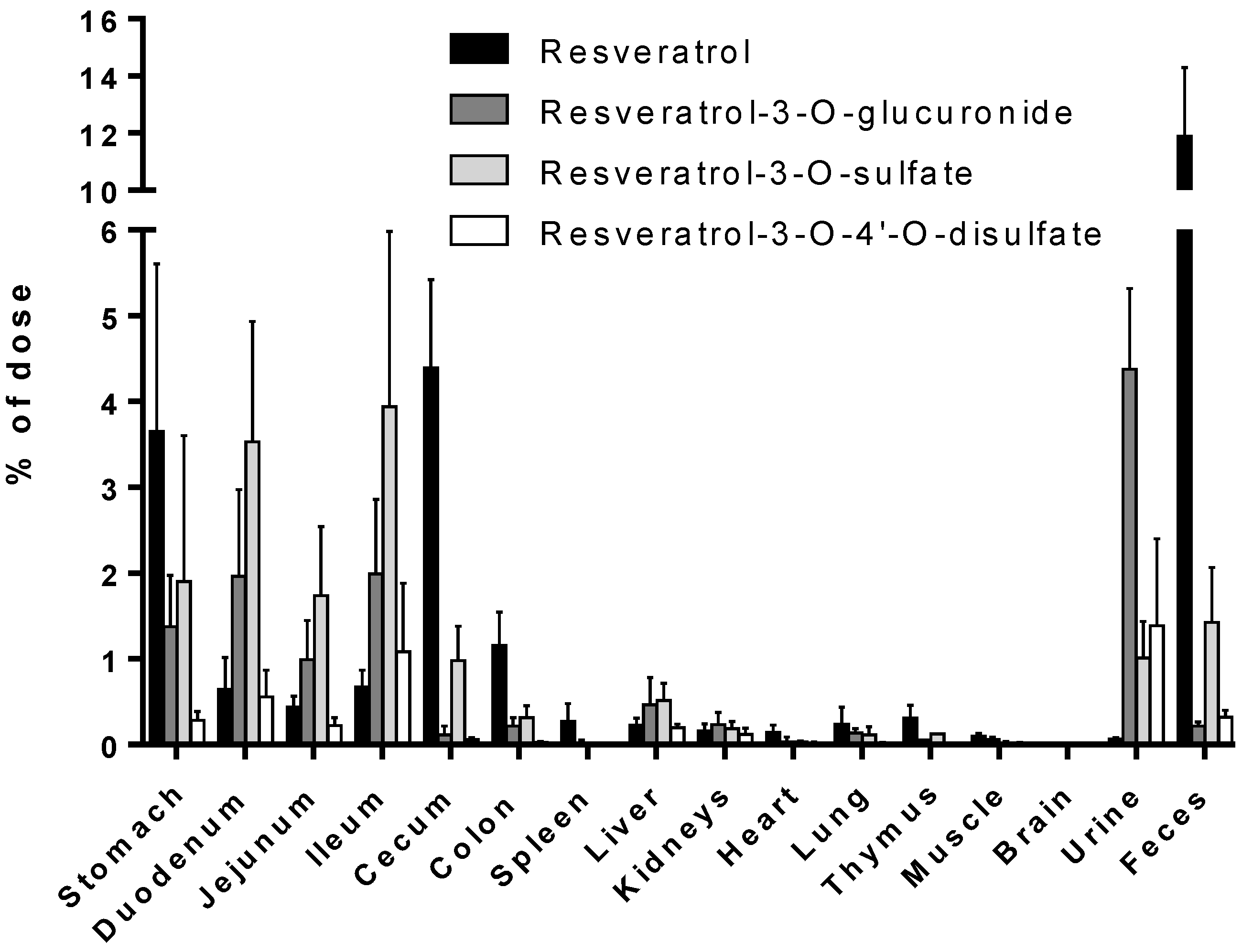
3.1. Tissue Levels of Resveratrol and Its Conjugated Metabolites
3.2. Expression of Sult and Ugt Genes in Mouse Tissues and Organs
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RT-qPCR | reverse transcription-quantitative PCR |
| Sult | sulfotransferase |
| Ugt | UDP-glucuronosyltransferase |
References
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 6, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Porro, C.; Cianciulli, A.; Calvello, R.; Panaro, M.A. Reviewing the role of resveratrol as a natural modulator of microglial activities. Curr. Pharm. Des. 2015, 21, 5277–5291. [Google Scholar] [CrossRef] [PubMed]
- Pallàs, M.; Porquet, D.; Vicente, A.; Sanfeliu, C. Resveratrol: New avenues for a natural compound in neuroprotection. Curr. Pharm. Des. 2013, 19, 6726–6731. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Marier, J.F.; Vachon, P.; Gritsas, A.; Zhang, J.; Moreau, J.P.; Ducharme, M.P. Metabolism and disposition of resveratrol in rats: Extent of absorption, glucuronidation, and enterohepatic recirculation evidenced by a linked-rat model. J. Pharmacol. Exp. Ther. 2002, 302, 369–373. [Google Scholar] [PubMed]
- Burkon, A.; Somoza, V. Quantification of free and protein-bound trans-resveratrol metabolites and identification of trans-resveratrol-C/O-conjugated diglucuronides-two novel resveratrol metabolites in human plasma. Mol. Nutr. Food Res. 2008, 52, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Brown, V.A.; Jones, D.J.; Britton, R.G.; Hemingway, D.; Miller, A.S.; West, K.P.; Booth, T.D.; Perloff, M.; Crowell, J.A.; et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010, 70, 7392–7399. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Maliakal, P.; Lu, H.; Lee, M.J.; Yang, C.S. Urinary and plasma levels of resveratrol and quercetin in humans, mice, and rats after ingestion of pure compounds and grape juice. J. Agric. Food Chem. 2004, 52, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Azorín-Ortuño, M.; Yáñez-Gascón, M.J.; Vallejo, F.; Pallarés, F.J.; Larrosa, M.; Lucas, R.; Morales, J.C.; Tomás-Barberán, F.A.; García-Conesa, M.T.; Espín, J.C. Metabolites and tissue distribution of resveratrol in the pig. Mol. Nutr. Food Res. 2011, 55, 1154–1168. [Google Scholar] [CrossRef] [PubMed]
- Maier-Salamon, A.; Hagenauer, B.; Reznicek, G.; Szekeres, T.; Thalhammer, T.; Jäger, W. Metabolism and disposition of resveratrol in the isolated perfused rat liver: Role of Mrp2 in the biliary excretion of glucuronides. J. Pharm. Sci. 2008, 97, 1615–1628. [Google Scholar] [CrossRef] [PubMed]
- Cottart, C.H.; Nivet-Antoine, V.; Beaudeux, J.L. Review of recent data on the metabolism, biological effects, and toxicity of resveratrol in humans. Mol. Nutr. Food Res. 2014, 58, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Rotches-Ribalta, M.; Andres-Lacueva, C.; Estruch, R.; Escribano, E.; Urpi-Sarda, M. Pharmacokinetics of resveratrol metabolic profile in healthy humans after moderate consumption of red wine and grape extract tablets. Pharmacol. Res. 2012, 66, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.P.; Chu, P.M.; Tsai, S.Y.; Wu, M.H.; Hou, Y.C. Pharmacokinetics and tissue distribution of resveratrol, emodin and their metabolites after intake of Polygonum cuspidatum in rats. J. Ethnopharmacol. 2012, 144, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Liu, X.; Wang, Q.; Cheng, S.; Zhang, S.; Zhang, M. Pharmacokinetics, tissue distribution and excretion study of resveratrol and its Prodrug 3,5,4′-tri-O-acetylresveratrol in rats. Phytomedicine 2013, 20, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Miksits, M.; Wlcek, K.; Svoboda, M.; Thalhammer, T.; Ellinger, I.; Stefanzl, G.; Falany, C.N.; Szekeres, T.; Jaeger, W. Expression of sulfotransferases and sulfatases in human breast cancer: Impact on resveratrol metabolism. Cancer Lett. 2010, 289, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Miksits, M.; Maier-Salamon, A.; Aust, S.; Thalhammer, T.; Reznicek, G.; Kunert, O.; Haslinger, E.; Szekeres, T.; Jaeger, W. Sulfation of resveratrol in human liver: Evidence of a major role for the sulfotransferases SULT1A1 and SULT1E1. Xenobiotica 2005, 35, 1101–1119. [Google Scholar] [CrossRef] [PubMed]
- Brill, S.S.; Furimsky, A.M.; Ho, M.N.; Furniss, M.J.; Li, Y.; Green, A.G.; Bradford, W.W.; Green, C.E.; Kapetanovic, I.M.; Iyer, L.V. Glucuronidation of trans-resveratrol by human liver and intestinal microsomes and UGT isoforms. J. Pharm. Pharmacol. 2006, 58, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Aumont, V.; Krisa, S.; Battaglia, E.; Netter, P.; Richard, T.; Mérillon, J.M.; Magdalou, J.; Sabolovic, N. Regioselective and stereospecific glucuronidation of trans- and cis-resveratrol in human. Arch. Biochem. Biophys. 2001, 393, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Riha, J.; Brenner, S.; Böhmdorfer, M.; Giessrigl, B.; Pignitter, M.; Schueller, K.; Thalhammer, T.; Stieger, B.; Somoza, V.; Szekeres, T.; Jäger, W. The effect of organic anion-transporting polypeptides 1B1, 1B3 and 2B1 on the antitumor activity of flavopiridol in breast cancer cells. Mol. Nutr. Food Res. 2014, 58, 324–332. [Google Scholar]
- Buckley, D.B.; Klaassen, C.D. Tissue- and gender-specific mRNA expression of UDP-glucuronosyltransferases (UGTs) in mice. Drug Metab. Dispos. 2007, 35, 121–227. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Gunewardena, S.; Cui, J.Y.; Yoo, B.; Zhong, X.B.; Klaassen, C.D. RNA-sequencing quantification of hepatic ontogeny and tissue distribution of mRNAs of phase II enzymes in mice. Drug Metab. Dispos. 2013, 41, 844–857. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zheng, H.; Zeng, S.; Xie, C.; Li, X.; Yan, T.; Gong, X.; Lu, L.; Qi, X.; Wang, Y.; et al. Profiles and Gender-Specifics of UDP-Glucuronosyltransferases and Sulfotransferases Expressions in the Major Metabolic Organs of Wild-Type and Efflux Transporter Knockout FVB Mice. Mol. Pharm. 2017, 14, 2967–2976. [Google Scholar] [CrossRef] [PubMed]
- Saldanha, A.J. Java Treeview-extensible visualization of microarray data. Bioinformatics 2004, 20, 3246–3248. [Google Scholar] [CrossRef] [PubMed]
- Maier-Salamon, A.; Hagenauer, B.; Wirth, M.; Gabor, F.; Szekeres, T.; Jäger, W. Increased transport of resveratrol across monolayers of the human intestinal Caco-2 cells is mediated by inhibition and saturation of metabolites. Pharm. Res. 2006, 9, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Mukai, M.; Isobe, T.; Okada, K.; Murata, M.; Shigeyama, M.; Hanioka, N. Species and sex differences in propofol glucuronidation in liver microsomes of humans, monkeys, rats and mice. Pharmazie 2015, 70, 466–470. [Google Scholar] [PubMed]
- Dai, P.; Luo, F.; Wang, Y.; Jiang, H.; Wang, L.; Zhang, G.; Zhu, L.; Hu, M.; Wang, X.; Lu, L.; et al. Species- and gender-dependent differences in the glucuronidation of a flavonoid glucoside and its aglycone determined using expressed UGT enzymes and microsomes. Biopharm. Drug Dispos. 2015, 36, 622–635. [Google Scholar] [CrossRef] [PubMed]
- Saghir, S.A.; Khan, S.A.; McCoy, A.T. Ontogeny of mammalian metabolizing enzymes in humans and animals used in toxicological studies. Crit. Rev. Toxicol. 2012, 42, 323–357. [Google Scholar] [CrossRef] [PubMed]
- Qosa, H.; Miller, D.S.; Pasinelli, P.; Trotti, D. Regulation of ABC efflux transporters at blood-brain barrier in health and neurological disorders. Brain Res. 2015, 1628, 298–316. [Google Scholar] [CrossRef] [PubMed]
- Van de Wetering, K.; Burkon, A.; Feddema, W.; Bot, A.; de Jonge, H.; Somoza, V.; Borst, P. Intestinal breast cancer resistance protein (BCRP)/Bcrp1 and multidrug resistance protein 3 (MRP3)/Mrp3 are involved in the pharmacokinetics of resveratrol. Mol. Pharmacol. 2009, 75, 876–885. [Google Scholar] [CrossRef] [PubMed]
- MacLean, C.; Moenning, U.; Reichel, A.; Fricker, G. Closing the gaps: A full scan of the intestinal expression of p-glycoprotein, breast cancer resistance protein, and multidrug resistance-associated protein 2 in male and female rats. Drug Metab. Dispos. 2008, 36, 1249–1254. [Google Scholar] [CrossRef] [PubMed]
- Menet, M.C.; Baron, S.; Taghi, M.; Diestra, R.; Dargère, D.; Laprévote, O.; Nivet-Antoine, V.; Beaudeux, J.L.; Bédarida, T.; Cottart, C.H. Distribution of trans-resveratrol and its metabolites after acute or sustained administration in mouse heart, brain, and liver. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Juan, M.E.; Maijó, M.; Planas, J.M. Quantification of trans-resveratrol and its metabolites in rat plasma and tissues by HPLC. J. Pharm. Biomed. Anal. 2010, 51, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Lou, B.S.; Wu, P.S.; Hou, C.W.; Cheng, F.Y.; Chen, J.K. Simultaneous quantification of trans-resveratrol and its sulfate and glucuronide metabolites in rat tissues by stable isotope-dilution UPLC-MS/MS analysis. J. Pharm. Biomed. Anal. 2014, 94, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Maier-Salamon, A.; Böhmdorfer, M.; Thalhammer, T.; Szekeres, T.; Jaeger, W. Hepatic glucuronidation of resveratrol: Interspecies comparison of enzyme kinetic profiles in human, mouse, rat, and dog. Drug Metab. Pharmacokinet. 2011, 26, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Kurita, A.; Miyauchi, Y.; Ikushiro, S.; Mackenzie, P.I.; Yamada, H.; Ishii, Y. Comprehensive characterization of mouse UDP-glucuronosyltransferase (Ugt) belonging to the Ugt2b subfamily: Identification of Ugt2b36 as the predominant isoform involved in morphine glucuronidation. J. Pharmacol. Exp. Ther. 2017, 361, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Gene Cards®: The Human Gene Database. Available online: www.genecards.org.
- Saeki, Y.; Sakakibara, Y.; Araki, Y.; Yanagisawa, K.; Suiko, M.; Nakajima, H.; Liu, M.C. Molecular cloning, expression, and characterization of a novel mouse liver SULT1B1 sulfotransferase. J. Biochem. 1998, 124, 55–64. [Google Scholar] [CrossRef]
- Johnson, J.J.; Nihal, M.; Siddiqui, I.A.; Scarlett, C.O.; Bailey, H.H.; Mukhtar, H.; Ahmad, N. Enhancing the bioavailability of resveratrol by combining it with piperine. Mol. Nutr. Food Res. 2011, 55, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhou, M.; Yang, F.F.; Liu, C.Y.; Pan, R.L.; Chang, Q.; Liu, X.M.; Liao, Y.H. Involvement of the inhibition of intestinal glucuronidation in enhancing the oral bioavailability of resveratrol by labrasol containing nanoemulsions. Mol. Pharm. 2015, 12, 1084–1095. [Google Scholar] [CrossRef] [PubMed]

| Sample | Resveratrol Concentration | ||||||
|---|---|---|---|---|---|---|---|
| 0.5 h | 1 h | 1.5 h | 2 h | 4 h | 6 h | 8 h | |
| Plasma | 0.140 ± 0.012 | 0.132 ± 0.017 | 0.118 ± 0.017 | 0.124 ± 0.041 | 0.135 ± 0.036 | 0.220 ± 0.079 | 0.152 ± 0.023 |
| Spleen | 4.347 ± 3.686 | 3.434 ± 1.946 | 3.305 ± 3.473 | 7.232 ± 1.050 | 3.218 ± 2.983 | 3.134 ± 1.824 | 2.229 ± 2.045 |
| Liver | 0.439 ± 0.133 | 0.358 ± 0.255 | 0.424 ± 0.217 | 0.345 ± 0.252 | 0.281 ± 0.105 | 0.141 ± 0.023 | 0.128 ± 0.033 |
| Kidney | 0.740 ± 0.361 | 0.786 ± 0.441 | 0.881 ± 0.436 | 0.418 ± 0.365 | 0.548 ± 0.140 | 0.667 ± 0.039 | 0.602 ± 0.137 |
| Heart | 3.698 ± 2.519 | 1.097 ± 1.094 | 1.176 ± 1.091 | 1.169 ± 1.126 | 1.129 ± 0.978 | 0.931 ± 0.339 | 0.276 ± 0.147 |
| Lung | 3.505 ± 2.985 | 1.791 ± 0.699 | 1.968 ± 1.769 | 1.899 ± 2.056 | 0.857 ± 0.749 | 2.388 ± 1.989 | 1.827 ± 1.300 |
| Stomach | 128.9 ± 74.38 | 111.8 ± 52.42 | 71.18 ± 60.49 | 40.93 ± 35.12 | 5.065 ± 2.220 | 1.709 ± 1.490 | 2.600 ± 1.072 |
| Duodenum | 10.01 ± 4.763 | 4.505 ± 2.787 | 6.551 ± 4.581 | 0.984 ± 0.081 | 0.752 ± 0.210 | 1.651 ± 0.673 | 1.100 ± 0.452 |
| Jejunum | 3.590 ± 1.692 | 1.693 ± 0.854 | 2.386 ± 2.288 | 0.889 ± 0.261 | 0.485 ± 0.133 | 0.715 ± 0.044 | 0.985 ± 0.515 |
| Ileum | 8.435 ± 2.611 | 26.17 ± 19.12 | 6.411 ± 2.166 | 2.074 ± 1.421 | 1.513 ± 0.512 | 1.289 ± 1.120 | 2.428 ± 1.751 |
| Cecum | 22.66 ± 15.47 | 11.52 ± 4.843 | 163.5 ± 138.5 | 30.72 ± 16.22 | 29.05 ± 19.92 | 1.761 ± 1.569 | 2.587 ± 2.235 |
| Colon | 7.422 ± 2.339 | 2.292 ± 1.390 | 22.04 ± 10.64 | 10.34 ± 6.731 | 9.754 ± 5.745 | 0.862 ± 1.087 | 2.655 ± 0.496 |
| Thymus | 15.01 ± 8.971 | 15.21 ± 6.934 | 4.784 ± 1.286 | 2.857 ± 0.192 | 0.818 ± 0.314 | 2.351 ± 1.051 | 11.01 ± 4.994 |
| Muscle | 1.530 ± 0.824 | 0.680 ± 0.619 | 0.557 ± 0.489 | 0.695 ± 1.203 | 0.555 ± 0.483 | 0.163 ± 0.128 | 0.333 ± 0.177 |
| Brain | 0.484 ± 0.337 | 0.212 ± 0.167 | 0.127 ± 0.054 | 0.041 ± 0.014 | ND | ND | ND |
| Urine | 4.716 ± 0.057 | 3.510 ± 0.513 | 15.70 ± 11.85 | 0.779 ± 0.106 | 5.091 ± 3.238 | 5.455 ± 2.562 | 4.472 ± 2.398 |
| Feces | 49.62 ± 29.79 | 186.5 ± 135.1 | 171.8 ± 79.23 | 292.5 ± 156.6 | 336.9 ± 219.2 | 22.24 ± 14.37 | 15.58 ± 13.93 |
| Sample | Resveratrol-3-O-Glucuronide Concentration | ||||||
|---|---|---|---|---|---|---|---|
| 0.5 h | 1 h | 1.5 h | 2 h | 4 h | 6 h | 8 h | |
| Plasma | 0.289 ± 0.102 | 0.081 ± 0.055 | 0.082 ± 0.040 | 0.076 ± 0.021 | 0.037 ± 0.019 | 0.090 ± 0.045 | 0.016 ± 0.011 |
| Spleen | 0.785 ± 1.360 | 1.908 ± 1.305 | 1.767 ± 1.061 | ND | ND | ND | ND |
| Liver | 2.173 ± 1.120 | 0.758 ± 0.488 | 0.984 ± 0.333 | 0.821 ± 0.380 | 0.480 ± 0.201 | 0.059 ± 0.101 | 0.057 ± 0.056 |
| Kidney | 2.665 ± 2.066 | 2.119 ± 1.598 | 1.556 ± 0.702 | 1.749 ± 0.759 | 0.351 ± 0.377 | 0.329 ± 0.048 | 0.202 ± 0.190 |
| Heart | 2.155 ± 1.284 | 0.370 ± 0.640 | 0.230 ± 0.398 | 0.269 ± 0.467 | 0.198 ± 0.114 | 0.145 ± 0.112 | ND |
| Lung | 6.440 ± 2.566 | 1.179 ± 0.658 | 1.527 ± 0.781 | 2.459 ± 0.471 | 0.185 ± 0.120 | 0.124 ± 0.095 | ND |
| Stomach | 72.69 ± 41.68 | 35.83 ± 10.97 | 21.93 ± 7.397 | 8.483 ± 3.440 | 2.754 ± 1.625 | 0.898 ± 0.786 | 1.140 ± 1.396 |
| Duodenum | 46.03 ± 36.63 | 15.96 ± 11.51 | 26.49 ± 13.38 | 3.761 ± 0.734 | 1.553 ± 1.101 | 0.672 ± 0.599 | 0.445 ± 0.241 |
| Jejunum | 11.30 ± 7.849 | 3.658 ± 2.922 | 10.74 ± 2.962 | 2.610 ± 2.396 | 0.946 ± 0.508 | 0.291 ± 0.234 | 0.183 ± 0.060 |
| Ileum | 35.07 ± 7.835 | 118.6 ± 49.70 | 12.26 ± 0.822 | 3.282 ± 2.030 | 1.716 ± 0.814 | 0.492 ± 0.440 | 0.594 ± 0.328 |
| Cecum | 1.528 ± 1.299 | 0.702 ± 0.653 | 0.627 ± 1.085 | 3.167 ± 2.962 | ND | ND | ND |
| Colon | 1.845 ± 0.530 | 0.707 ± 0.540 | 4.385 ± 5.582 | 3.830 ± 3.202 | 0.805 ± 0.394 | ND | ND |
| Thymus | 12.39 ± 2.539 | ND | ND | ND | ND | ND | ND |
| Muscle | 0.683 ± 0.673 | 0.642 ± 0.090 | 0.622 ± 0.299 | 0.342 ± 0.309 | 0.232 ± 0.101 | ND | ND |
| Brain | ND | ND | ND | ND | ND | ND | ND |
| Urine | 362.7 ± 86.98 | 522.5 ± 289.7 | 440.1 ± 146.9 | 147.5 ± 78.32 | 168.1 ± 118.9 | 120.5 ± 51.62 | 96.92 ± 36.83 |
| Feces | 22.60 ± 9.050 | ND | ND | ND | ND | 1.524 ± 1.321 | 9.743 ± 1.216 |
| Sample | Resveratrol-3-O-Sulfate Concentration | ||||||
|---|---|---|---|---|---|---|---|
| 0.5 h | 1 h | 1.5 h | 2 h | 4 h | 6 h | 8 h | |
| Plasma | 0.265 ± 0.141 | 0.089 ± 0.049 | 0.160 ± 0.142 | 0.148 ± 0.102 | 0.055 ± 0.026 | 0.069 ± 0.049 | 0.023 ± 0.011 |
| Spleen | 0.558 ± 0.966 | ND | ND | ND | ND | ND | ND |
| Liver | 1.649 ± 0.975 | 0.596 ± 0.347 | 1.081 ± 0.428 | 0.991 ± 0.493 | 0.739 ± 0.409 | 0.070 ± 0.122 | 0.070 ± 0.068 |
| Kidney | 1.525 ± 0.724 | 1.602 ± 1.726 | 0.885 ± 0.244 | 1.055 ± 0.489 | 0.594 ± 0.374 | 0.378 ± 0.366 | 0.295 ± 0.282 |
| Heart | 2.405 ± 1.166 | 1.517 ± 0.954 | 1.922 ± 1.031 | 0.627 ± 0.085 | ND | ND | ND |
| Lung | 4.877 ± 3.653 | 0.848 ± 0.771 | 1.271 ± 1.105 | 2.741 ± 1.465 | ND | ND | ND |
| Stomach | 96.16 ± 38.23 | 45.50 ± 21.81 | 36.51 ± 30.13 | 7.719 ± 4.434 | 3.605 ± 1.059 | 3.043 ± 1.057 | 4.332 ± 0.968 |
| Duodenum | 67.25 ± 27.95 | 27.08 ± 11.75 | 59.21 ± 34.83 | 8.202 ± 2.905 | 3.141 ± 1.562 | 1.358 ± 1.179 | 0.910 ± 0.868 |
| Jejunum | 13.24 ± 7.097 | 6.847 ± 4.393 | 16.05 ± 12.51 | 6.451 ± 3.212 | 2.370 ± 1.228 | 0.828 ± 0.637 | 0.497 ± 0.275 |
| Ileum | 73.23 ± 19.10 | 178.2 ± 125.5 | 41.65 ± 15.54 | 13.48 ± 8.766 | 6.577 ± 4.057 | 2.198 ± 0.852 | 1.583 ± 1.495 |
| Cecum | 12.67 ± 3.839 | 2.883 ± 2.691 | 24.94 ± 13.31 | 12.58 ± 8.254 | 3.829 ± 1.946 | 0.590 ± 0.122 | 0.123 ± 0.213 |
| Colon | 2.189 ± 0.749 | 0.929 ± 0.603 | 6.117 ± 2.114 | 5.133 ± 3.381 | 1.646 ± 1.982 | ND | ND |
| Thymus | 28.31 ± 14.53 | ND | ND | ND | ND | ND | ND |
| Muscle | 0.209 ± 0.363 | 0.217 ± 0.162 | 0.196 ± 0.339 | ND | ND | ND | ND |
| Brain | ND | ND | ND | ND | ND | ND | ND |
| Urine | 128.3 ± 33.15 | 108.5 ± 51.50 | 62.23 ± 15.03 | 54.72 ± 25.74 | 58.73 ± 3.598 | 52.40 ± 12.57 | 22.15 ± 7.149 |
| Feces | 41.64 ± 13.96 | 32.85 ± 5.838 | 6.733 ± 1.380 | 52.64 ± 12.72 | 11.21 ± 9.740 | 2.796 ± 0.435 | 21.55 ± 8.708 |
| Sample | Resveratrol-3-O-4′-O-Disulfate Concentration | ||||||
|---|---|---|---|---|---|---|---|
| 0.5 h | 1 h | 1.5 h | 2 h | 4 h | 6 h | 8 h | |
| Plasma | 0.099 ± 0.016 | 0.090 ± 0.028 | 0.039 ± 0.023 | 0.050 ± 0.044 | 0.033 ± 0.022 | 0.042 ± 0.037 | 0.049 ± 0.033 |
| Spleen | ND | ND | ND | ND | ND | ND | ND |
| Liver | 0.585 ± 0.258 | 0.267 ± 0.143 | 0.422 ± 0.100 | 0.517 ± 0.073 | 0.218 ± 0.036 | 0.019 ± 0.032 | 0.021 ± 0.037 |
| Kidney | 0.430 ± 0.252 | 0.692 ± 0.516 | 0.387 ± 0.113 | 0.986 ± 0.720 | 0.767 ± 0.101 | 0.247 ± 0.182 | 0.078 ± 0.046 |
| Heart | 0.406 ± 0.203 | 0.973 ± 0.332 | ND | ND | ND | ND | ND |
| Lung | 0.673 ± 0.867 | 0.275 ± 0.177 | 0.164 ± 0.102 | 0.438 ± 0.258 | ND | ND | ND |
| Stomach | 15.08 ± 9.413 | 4.088 ± 3.812 | 4.899 ± 3.492 | 1.448 ± 0.834 | 1.389 ± 0.556 | 0.251 ± 0.234 | 0.350 ± 0.076 |
| Duodenum | 12.54 ± 6.672 | 5.273 ± 4.943 | 5.080 ± 3.785 | 1.092 ± 0.104 | 0.956 ± 0.306 | 0.203 ± 0.176 | 0.123 ± 0.113 |
| Jejunum | 1.666 ± 1.280 | 1.210 ± 0.841 | 1.233 ± 1.175 | 0.665 ± 0.191 | 0.527 ± 0.315 | 0.166 ± 0.098 | ND |
| Ileum | 2.264 ± 0.441 | 2.594 ± 0.577 | 2.749 ± 1.006 | 1.474 ± 0.938 | 1.219 ± 0.518 | 0.260 ± 0.150 | 0.270 ± 0.168 |
| Cecum | 0.363 ± 0.319 | 0.280 ± 0.485 | 0.807 ± 0.706 | 1.123 ± 0.991 | 0.305 ± 0.328 | ND | ND |
| Colon | 0.751 ± 0.287 | 0.377 ± 0.136 | 0.843 ± 0.505 | 0.632 ± 0.433 | ND | ND | ND |
| Thymus | ND | ND | ND | ND | ND | ND | ND |
| Muscle | 0.167 ± 0.089 | 0.327 ± 0.167 | 0.255 ± 0.214 | ND | ND | ND | ND |
| Brain | ND | ND | ND | ND | ND | ND | ND |
| Urine | 7.382 ± 0.362 | 53.17 ± 23.97 | 10.19 ± 3.056 | 50.06 ± 24.88 | 67.57 ± 33.96 | 106.0 ± 48.06 | 58.26 ± 23.76 |
| Feces | 11.92 ± 2.654 | 6.930 ± 2.455 | 1.767 ± 0.060 | 1.821 ± 0.155 | 1.989 ± 0.446 | 7.778 ± 3.473 | 3.155 ± 5.465 |
| Sample | AUC (nmol/mL min or nmol/g min) | ||||||
|---|---|---|---|---|---|---|---|
| Res | Res3G | Res3S | ResDiS | AUCRes/AUCRes3G | AUCRes/AUCRes3S | AUCRes/AUCResDiS | |
| Plasma | 1.21 ± 0.72 | 0.59 ± 0.22 | 0.71 ± 0.28 | 0.37 ± 0.15 | 2.05 | 1.70 | 3.27 |
| Spleen | 27.8 ± 4.35 | 2.23 ± 1.91 | 0.28 ± 0.16 | n.a. | 12.5 | 99.7 | n.a. |
| Liver | 2.02 ± 0.44 | 4.12 ± 2.17 | 4.59 ± 1.65 | 1.78 ± 0.56 | 0.50 | 0.44 | 1.13 |
| Kidney | 4.76 ± 0.88 | 6.92 ± 2.67 | 5.56 ± 1.60 | 3.60 ± 0.99 | 0.69 | 0.86 | 1.32 |
| Heart | 8.84 ± 3.70 | 2.00 ± 1.16 | 1.99 ± 1.41 | 0.83± 0.37 | 4.41 | 4.45 | 10.6 |
| Lung | 14.3 ± 3.51 | 8.27 ± 3.44 | 6.92 ± 2.88 | 1.02 ± 0.67 | 1.73 | 2.07 | 14.0 |
| Stomach | 223 ± 118 | 84.3 ± 32.7 | 116 ± 46.2 | 17.5 ± 5.08 | 2.65 | 1.92 | 12.8 |
| Duodenum | 17.7 ± 10.1 | 53.8 ± 26.0 | 96.9 ± 37.3 | 15.3 ± 6.54 | 0.33 | 0.18 | 1.16 |
| Jejunum | 8.33 ± 3.59 | 18.8 ± 11.3 | 33.0 ± 16.1 | 4.27 ± 1.67 | 0.44 | 0.25 | 1.95 |
| Ileum | 31.1 ± 16.2 | 92.1 ± 38.6 | 182 ± 98.2 | 8.43 ± 3.96 | 0.34 | 0.17 | 0.62 |
| Cecum | 201 ± 63.5 | 5.39 ± 3.17 | 44.9 ± 24.9 | 2.74 ± 1.12 | 37.4 | 4.48 | 73.5 |
| Colon | 52.7 ± 22.0 | 9.87 ± 4.39 | 14.3 ± 6.12 | 1.21 ± 0.84 | 5.34 | 3.68 | 43.5 |
| Thymus | 34.9 ± 12.7 | 6.19 ± 2.26 | 14.2 ± 8.35 | n.a. | 5.63 | 2.46 | 0.0 |
| Muscle | 4.02 ± 1.53 | 2.37 ± 0.68 | 0.76 ± 0.21 | 0.74 ± 0.17 | 1.70 | 5.31 | 5.45 |
| Brain | 0.46 ± 0.18 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Urine | 38.8 ± 13.6 | 1521 ± 411 | 462 ± 103 | 503 ± 141 | 0.03 | 0.08 | 0.08 |
| Feces | 1304 ± 337 | 24.1 ± 12.6 | 156 ± 52.6 | 35.3 ± 11.9 | 54.1 | 8.36 | 37.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Böhmdorfer, M.; Szakmary, A.; Schiestl, R.H.; Vaquero, J.; Riha, J.; Brenner, S.; Thalhammer, T.; Szekeres, T.; Jäger, W. Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice. Nutrients 2017, 9, 1347. https://doi.org/10.3390/nu9121347
Böhmdorfer M, Szakmary A, Schiestl RH, Vaquero J, Riha J, Brenner S, Thalhammer T, Szekeres T, Jäger W. Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice. Nutrients. 2017; 9(12):1347. https://doi.org/10.3390/nu9121347
Chicago/Turabian StyleBöhmdorfer, Michaela, Akos Szakmary, Robert H. Schiestl, Javier Vaquero, Juliane Riha, Stefan Brenner, Theresia Thalhammer, Thomas Szekeres, and Walter Jäger. 2017. "Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice" Nutrients 9, no. 12: 1347. https://doi.org/10.3390/nu9121347
APA StyleBöhmdorfer, M., Szakmary, A., Schiestl, R. H., Vaquero, J., Riha, J., Brenner, S., Thalhammer, T., Szekeres, T., & Jäger, W. (2017). Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice. Nutrients, 9(12), 1347. https://doi.org/10.3390/nu9121347





