The Effect of Polyphenol-Rich Interventions on Cardiovascular Risk Factors in Haemodialysis: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search
2.2. Data Extraction
2.3. Assessment of Study and Evidence Quality
2.4. Data Analysis
3. Results
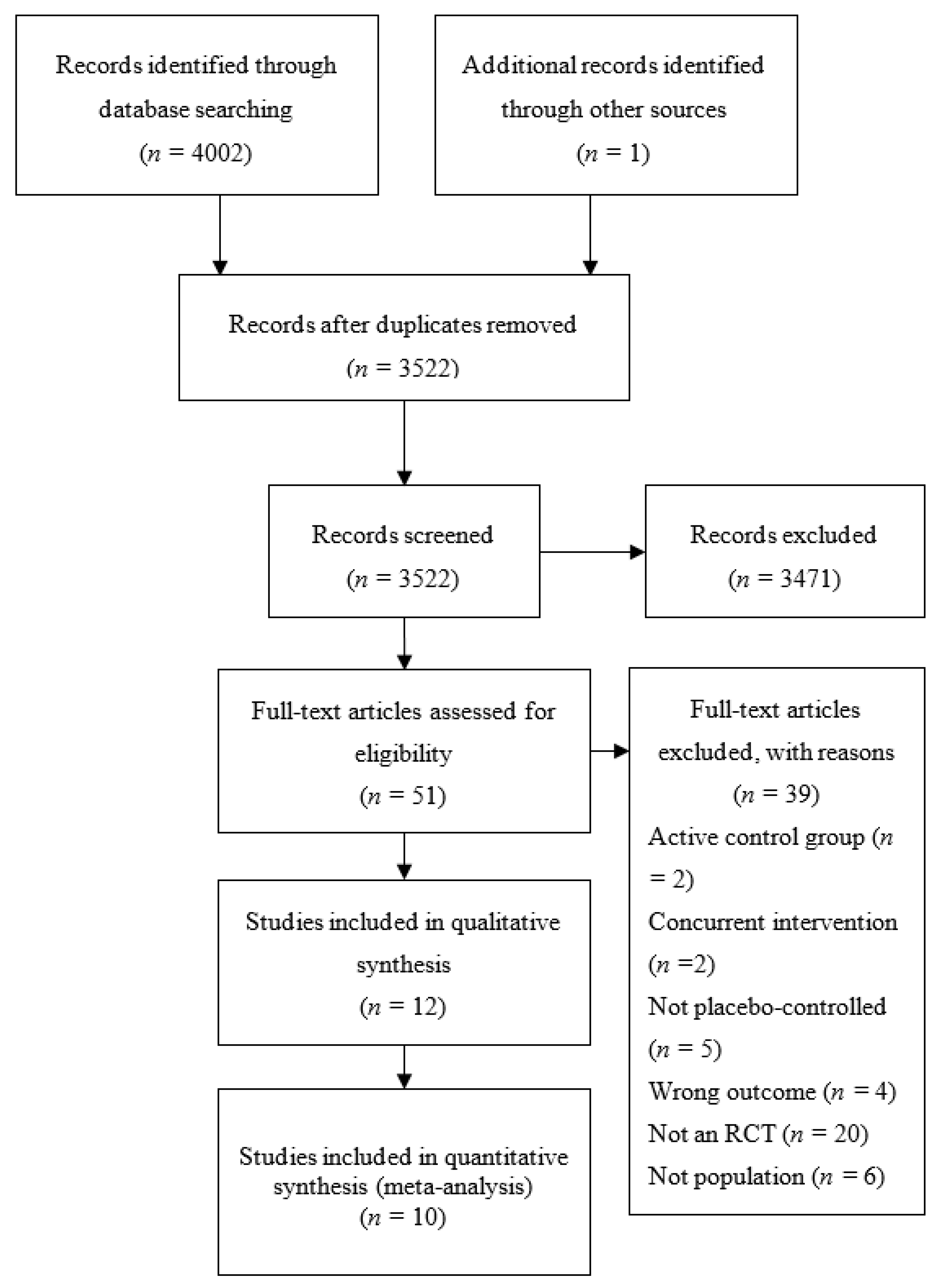
3.1. Study Selection
3.2. Study Characteristics
3.3. Interventions
3.4. Study Results
3.4.1. Oxidative Stress
3.4.2. Inflammation
3.4.3. Hemodynamic Measures
3.4.4. Lipid Profiles
3.5. Adverse Events
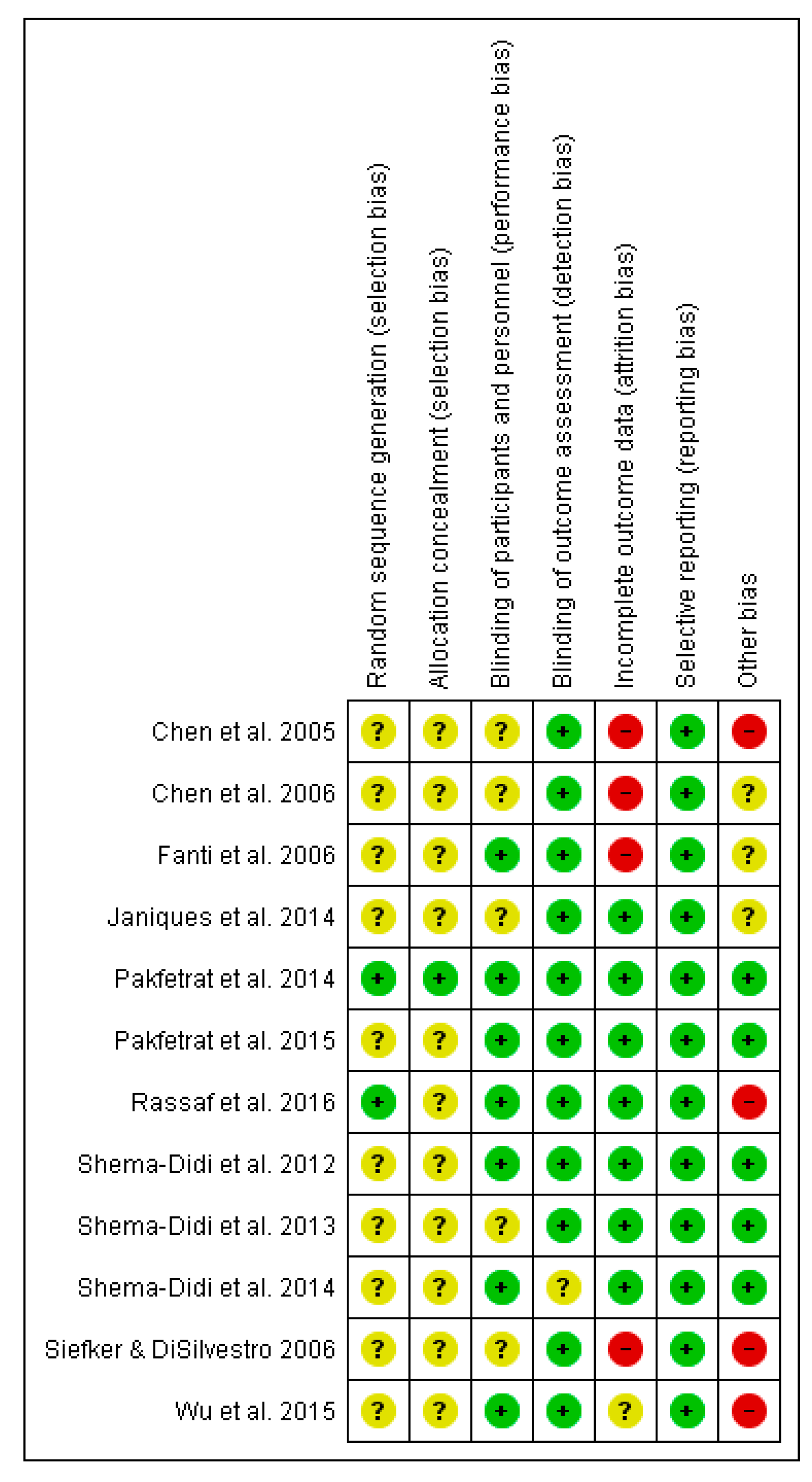
3.6. Risk of Bias
3.7. Quality of Evidence
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Couser, W.G.; Remuzzi, G.; Mendis, S.; Tonelli, M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 2011, 80, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- De Jager, D.J.; Grootendorst, D.C.; Jager, K.J.; van Dijk, P.C.; Tomas, L.M.; Ansell, D.; Collart, F.; Finne, P.; Heaf, J.G.; De Meester, J.; et al. Cardiovascular and noncardiovascular mortality among patients starting dialysis. JAMA 2009, 302, 1782–1789. [Google Scholar] [CrossRef] [PubMed]
- Stenvinkel, P.; Carrero, J.J.; Axelsson, J.; Lindholm, B.; Heimbürger, O.; Massy, Z. Emerging biomarkers for evaluating cardiovascular risk in the chronic kidney disease patient: How do new pieces fit into the uremic puzzle? Clin. J. Am. Soc. Nephrol. 2008, 3, 505–521. [Google Scholar] [CrossRef] [PubMed]
- Stenvinkel, P.; Larsson, T.E. Chronic kidney disease: A clinical model of premature aging. Am. J. Kidney Dis. 2013, 62, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Mancini, E. Cardiac effects of chronic inflammation in dialysis patients. Nephrol. Dial. Transplant. 2002, 17, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; Stenvinkel, P. Inflammation in End-Stage Renal Disease—What Have We Learned in 10 Years? Semin. Dial. 2010, 23, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Morena, M.; Delbosc, S.; Dupuy, A.M.; Canaud, B.; Cristol, J.P. Overproduction of reactive oxygen species in end-stage renal disease patients: A potential component of hemodialysis-associated inflammation. Hemodial. Int. 2005, 9, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.T.; Palmer, S.C.; Wai, S.N.; Ruospo, M.; Carrero, J.J.; Campbell, K.L.; Strippoli, G.F. Healthy Dietary Patterns and Risk of Mortality and ESRD in CKD: A Meta-Analysis of Cohort Studies. Clin. J. Am. Soc. Nephrol. 2017, 12, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Luis, D.; Zlatkis, K.; Comenge, B.; Garcia, Z.; Navarro, J.F.; Lorenzo, V.; Carrero, J.J. Dietary Quality and Adherence to Dietary Recommendations in Patients Undergoing Hemodialysis. J. Ren. Nutr. 2016, 26, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.T.; Rossi, M.; Johnson, D.W.; Campbell, K.L. Beyond Sodium, Phosphate and Potassium: Potential Dietary Interventions in Kidney Disease. Semin. Dial. 2017, 30, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.; Kelly, J.; Tapsell, L. Dietary Modeling of Foods for Advanced CKD Based on General Healthy Eating Guidelines: What Should Be on the Plate? Am. J. Kidney Dis. 2017, 69, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Ferri, C. Flavonoids: Antioxidants Against Atherosclerosis. Nutrients 2010, 2, 889. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2014, 349. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.J.; Dwyer, J.T.; Jacques, P.F.; McCullough, M.L. Associations between flavonoids and cardiovascular disease incidence or mortality in European and US populations. Nutr. Rev. 2012, 70, 491–508. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Rimm, E.B.; Medina-Remón, A.; Martínez-González, M.A.; López-Sabater, M.C.; Covas, M.I.; Corella, D.; Salas-Salvadó, J.; Gómez-Gracia, E.; Lapetra, J.; et al. Polyphenol intake and mortality risk: A re-analysis of the PREDIMED trial. BMC Med. 2014, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Rimm, E.B.; Medina-Remón, A.; Martínez-González, M.A.; de la Torre, R.; Corella, D.; Salas-Salvadó, J.; Gómez-Gracia, E.; Lapetra, J.; Arós, F.; et al. Inverse association between habitual polyphenol intake and incidence of cardiovascular events in the PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Rienks, J.; Barbaresko, J.; Nöthlings, U. Association of Polyphenol Biomarkers with Cardiovascular Disease and Mortality Risk: A Systematic Review and Meta-Analysis of Observational Studies. Nutrients 2017, 9, 415. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Fruit polyphenols: A review of anti-inflammatory effects in humans. Crit. Rev. Food Sci. Nutr. 2016, 56, 419–444. [Google Scholar] [CrossRef] [PubMed]
- Zern, T.L.; West, K.L.; Fernandez, M.L. Grape polyphenols decrease plasma triglycerides and cholesterol accumulation in the aorta of ovariectomized guinea pigs. J. Nutr. 2003, 133, 2268–2272. [Google Scholar] [PubMed]
- Diebolt, M.; Bucher, B.; Andriantsitohaina, R. Wine polyphenols decrease blood pressure, improve NO vasodilatation, and induce gene expression. Hypertension 2001, 38, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Abdollahzad, H.; Eghtesadi, S.; Nourmohammadi, I.; Khadem-Ansari, M.; Nejad-Gashti, H.; Esmaillzadeh, A. Effect of vitamin C supplementation on oxidative stress and lipid profiles in hemodialysis patients. Int. J. Vitam. Nutr. Res. 2009, 79, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Bhogade, R.; Suryakar, A.; Joshi, N.; Patil, R. Effect of vitamin E supplementation on oxidative stress in hemodialysis patients. Indian J. Clin. Biochem. 2008, 23, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Baldi, S.; Innocenti, M.; Frascerra, S.; Nannipieri, M.; Lippi, A.; Rindi, P.; Ferrannini, E. Effects of hemodialysis and vitamin E supplementation on low-density lipoprotein oxidizability in end-stage renal failure. J. Nephrol. 2013, 26, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Janiques, A.G.; Leal Vde, O.; Stockler-Pinto, M.B.; Moreira, N.X.; Mafra, D. Effects of grape powder supplementation on inflammatory and antioxidant markers in hemodialysis patients: A randomized double-blind study. J. Bras. Nefrol. 2014, 36, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Shema-Didi, L.; Kristal, B.; Ore, L.; Shapiro, G.; Geron, R.; Sela, S. Pomegranate juice intake attenuates the increase in oxidative stress induced by intravenous iron during hemodialysis. Nutr. Res. 2013, 33, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Pakfetrat, M.; Akmali, M.; Malekmakan, L.; Dabaghimanesh, M.; Khorsand, M. Role of turmeric in oxidative modulation in end-stage renal disease patients. Hemodial. Int. 2015, 19, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Rassaf, T.; Rammos, C.; Hendgen-Cotta, U.B.; Heiss, C.; Kleophas, W.; Dellanna, F.; Floege, J.; Hetzel, G.R.; Kelm, M. Vasculoprotective Effects of Dietary Cocoa Flavanols in Patients on Hemodialysis: A Double–Blind, Randomized, Placebo–Controlled Trial. Clin. J. Am. Soc. Nephrol. 2016, 11, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLOS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Perez-Jimenez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remón, A.; M’Hiri, N.; García-Lobato, P.; Manach, C.; Knox, C.; Eisner, R.; Wishart, D.S.; et al. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Wiley Online Library: Hoboken, NJ, USA, 2008; Volume 5. [Google Scholar]
- Guyatt, G.H.; Oxman, A.D.; Schünemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles in the Journal of Clinical Epidemiology. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Handbook for Grading the Quality of Evidence and the Strength of Recommendations Using the GRADE Approach. Updated October 2013. Available online: http://gdt.guidelinedevelopment.org (accessed on 12 April 2017).
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Controll. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Shema-Didi, L.; Kristal, B.; Sela, S.; Geron, R.; Ore, L. Does Pomegranate intake attenuate cardiovascular risk factors in hemodialysis patients? Nutr. J. 2014, 13, 18. [Google Scholar] [CrossRef] [PubMed]
- Shema-Didi, L.; Sela, S.; Ore, L.; Shapiro, G.; Geron, R.; Moshe, G.; Kristal, B. One year of pomegranate juice intake decreases oxidative stress, inflammation, and incidence of infections in hemodialysis patients: A randomized placebo-controlled trial. Free Radic. Biol. Med. 2012, 53, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Pakfetrat, M.; Basiri, F.; Malekmakan, L.; Roozbeh, J. Effects of turmeric on uremic pruritus in end stage renal disease patients: A double-blind randomized clinical trial. J. Nephrol. 2014, 27, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.P.; Wu, M.S.; Yang, C.C.; Huang, K.C.; Liou, S.Y.; Hsu, S.M.; Chien, C.T. Chronic green tea extract supplementation reduces hemodialysis-enhanced production of hydrogen peroxide and hypochlorous acid, atherosclerotic factors, and proinflammatory cytokines. Am. J. Clin. Nutr. 2007, 86, 1539–1547. [Google Scholar] [PubMed]
- Fanti, P.; Asmis, R.; Stephenson, T.J.; Sawaya, B.P.; Franke, A.A. Positive effect of dietary soy in ESRD patients with systemic inflammation—Correlation between blood levels of the soy isoflavones and the acute-phase reactants. Nephrol. Dial. Transplant. 2006, 21, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, J.; Yang, C.; Peng, S.; Ferng, S. Effect of soya protein on serum lipid profile and lipoprotein concentrations in patients undergoing hypercholesterolaemic haemodialysis. Br. J. Nutr. 2006, 95, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ferng, S.; Yang, C.; Peng, S.; Lee, H.; Chen, J. Variable effects of soy protein on plasma lipids in hyperlipidemic and normolipidemic hemodialysis patients. Am. J. Kidney Dis. 2005, 46, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Siefker, K.; DiSilvestro, R.A. Safety and antioxidant effects of a modest soy protein intervention in hemodialysis patients. J. Med. Food 2006, 9, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.T.; Fitschen, P.J.; Kistler, B.M.; Jeong, J.H.; Chung, H.R.; Aviram, M.; Phillips, S.A.; Fernhall, B.; Wilund, K.R. Effects of Pomegranate Extract Supplementation on Cardiovascular Risk Factors and Physical Function in Hemodialysis Patients. J. Med. Food 2015, 18, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E., Jr.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.J.; Nihal, M.; Siddiqui, I.A.; Scarlett, C.O.; Bailey, H.H.; Mukhtar, H.; Ahmad, N. Enhancing the bioavailability of resveratrol by combining it with piperine. Mol. Nutr. Food Res. 2011, 55, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Milenkovic, D.; Van de Wiele, T.; Rodriguez-Mateos, A.; de Roos, B.; Garcia-Conesa, M.T.; Landberg, R.; Gibney, E.R.; Heinonen, M.; Tomás-Barberán, F.; et al. Addressing the inter-individual variation in response to consumption of plant food bioactives: Towards a better understanding of their role in healthy aging and cardiometabolic risk reduction. Mol. Nutr. Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Setchell, K.D.R.; Clerici, C. Equol: History, Chemistry, and Formation. J. Nutr. 2010, 140, 1355S–1362S. [Google Scholar] [CrossRef] [PubMed]
- Marín, L.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Bioavailability of Dietary Polyphenols and Gut Microbiota Metabolism: Antimicrobial Properties. BioMed Res. Int. 2015, 2015, 18. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; DeSantis, T.Z.; Ni, Z.; Nguyen, T.H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Ramezani, A.; Massy, Z.A.; Meijers, B.; Evenepoel, P.; Vanholder, R.; Raj, D.S. Role of the Gut Microbiome in Uremia: A Potential Therapeutic Target. Am. J. Kidney Dis. 2016, 67, 483–498. [Google Scholar] [CrossRef] [PubMed]
| Study | Setting | Study Design | Population |
|---|---|---|---|
| Soy protein (contains isoflavones) | |||
| Fanti et al. 2006 [40] |
|
|
|
| Chen et al. 2005 [42] |
|
|
|
| Chen et al. 2006 [41] |
|
|
|
| Siefker & DiSilvestro 2006 [43] |
|
|
|
| Grape (contains various polyphenols) | |||
| Janiques et al. 2014 [25] |
|
|
|
| Turmeric (contains curcuminoids) | |||
| Pakfetrat et al. 2014 [38] |
|
|
|
| Pakfetrat et al. 2015 a [27] |
|
|
|
| Cocoa (contains flavanols) | |||
| Rassaf et al. 2016 [28] |
|
|
|
| Pomegranate (Phenolic acids & Flavanoids) | |||
| Shema-Didi et al. 2012 [37] |
|
|
|
| Shema-Didi et al. 2014 [36] | As per Shema-Didi et al. 2012 [37] | Shema-Didi et al. 2012 [37] | Shema-Didi et al. 2012 [37] |
| Shema-Didi et al. 2013 [26] |
|
|
|
| Wu et al. 2015 [44] |
|
|
|
| Study | Intervention | Results |
|---|---|---|
| Soy protein (contains isoflavones) | ||
| Fanti et al. 2006 [40] | Intervention: Isoflavone-containing soy-based a protein powder mixed into a drink (54 mg isoflavones), a protein bar (26 mg isoflavones) or cereal product (26 mg isoflavones). Comparator: Isoflavone-free milk-based protein powder mixed into a drink, a protein bar or a cereal product. Energy, protein, CHO, fat, Na and K intake equal to intervention product. Dose: one supplement item daily (powder 3 times per week; bar or cereal 4 times per week). Duration: 8-weeks. | Pro-inflammatory markers: 8-weeks post-baseline: Serum CRP
|
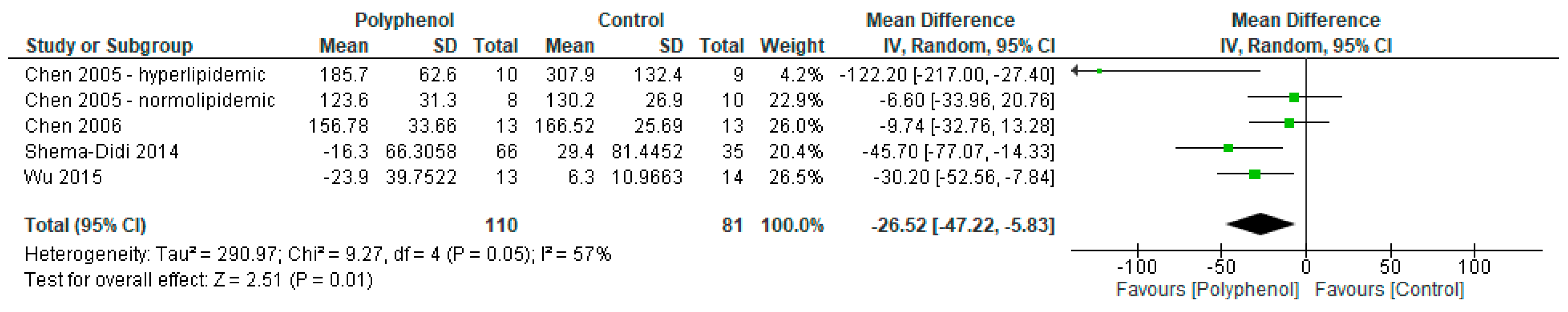
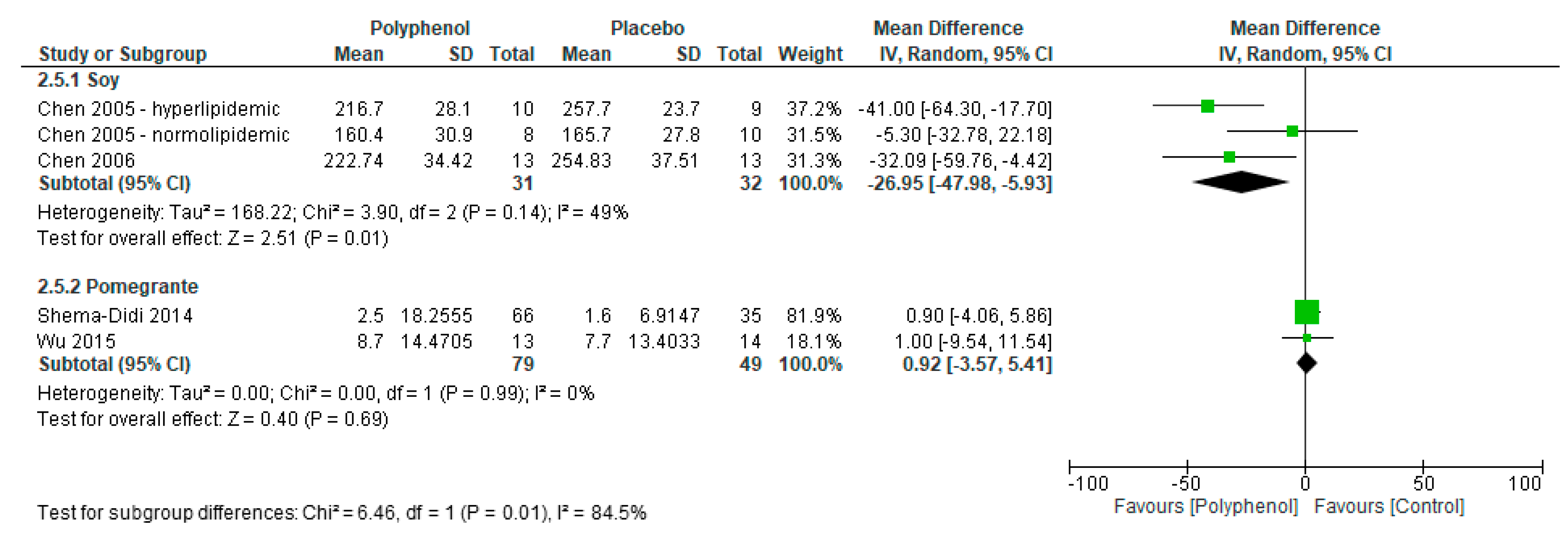
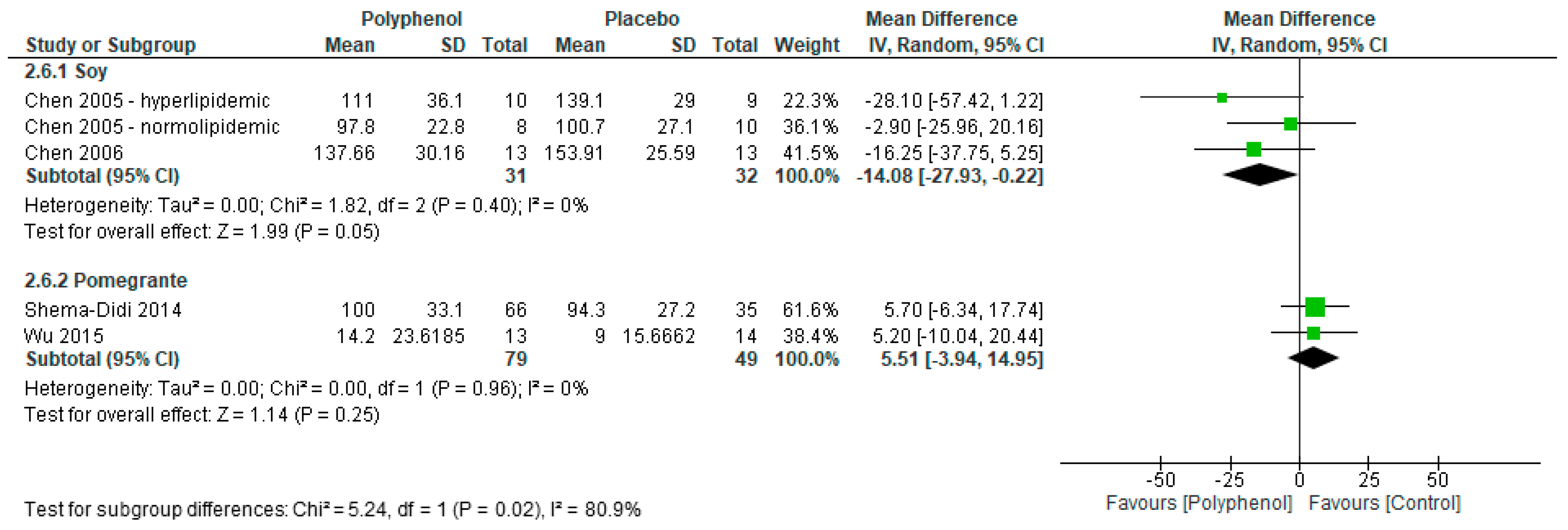
| Chen et al. 2005 [42] | Intervention: 30 g isolated soy protein (36.3 mg isoflavone content; as reported in Chen et al. 2006 [41]) fortified with calcium, mixed with 200 mL fluid. Comparator: 30 g milk protein, with equivalent amounts of calcium, energy, carbohydrate and fat; mixed with 200 mL fluid. Dose: one supplement daily Duration: 12-weeks. | Normolipidemic Subjects—Lipid profile: 12-weeks post-baseline: Fasting serum TG:
Fasting serum TG:
|
| Chen et al. 2006 [41] | Supplement: 30 g isolated soy protein (36.3 mg isoflavone content) fortified with calcium, mixed with 200 mL fluid. Comparator: 30 g milk protein, with equivalent amounts of protein, calcium, energy, carbohydrate and fat; mixed with 200 mL fluid. Dose: one supplement daily Duration: 12-weeks. | Lipid profile: 12-weeks post-baseline: Fasting serum TG:
|
| Siefker & DiSilvestro 2006 [43] | Intervention: soy protein powder (52 mg isoflavone content) mixed with fluid, artificial chocolate flavoured. Comparator: whey protein powder mixed with fluid, artificial chocolate flavoured. Dose: one supplement 4 times per week Duration: 4-weeks. | Oxidative stress markers: 4-weeks post-baseline: Oxidized LDL-C:
TNF-α:
|
| Grape (contains various polyphenols) | ||
| Janiques et al. 2014 [25] | Intervention: 12 g (500 mg total polyphenols) grape powder mixed into grape jelly. Comparator: grape jelly/placebo. Dose: 1 tablespoon of jelly daily consumed in the afternoon. Duration: 5-weeks. | Anti-inflammatory markers: Not clear when measured; assumed to be measured at end of treatment (5-weeks post-baseline): Plasma GSH-Px:
Plasma CRP:
|
| Turmeric (contains curcuminoids) | ||
| Pakfetrat et al. 2014 [38] | Intervention: capsule of 500 mg (22.1 mg active ingredient curcumin, a polyphenol) turmeric powder Comparator: capsule of starch/placebo. Dose: 3 capsules per day, one with each main meal. Duration: 8-weeks | Pro-inflammatory markers: Not clear when measured; assumed to be measured at end of treatment (8-weeks post-baseline): High-sensitivity CRP:
|
| Pakfetrat et al. 2015 [27] | Intervention: capsule of 500 mg (22.1 mg active ingredient curcumin, a polyphenol) turmeric powder Comparator: capsule of starch/placebo. Dose: 3 capsules per day, one with each main meal. Duration: 8-weeks | Oxidative stress: 8-weeks post-baseline: Red blood cell catalase (CAT):
|
| Cocoa (contains flavanols) | ||
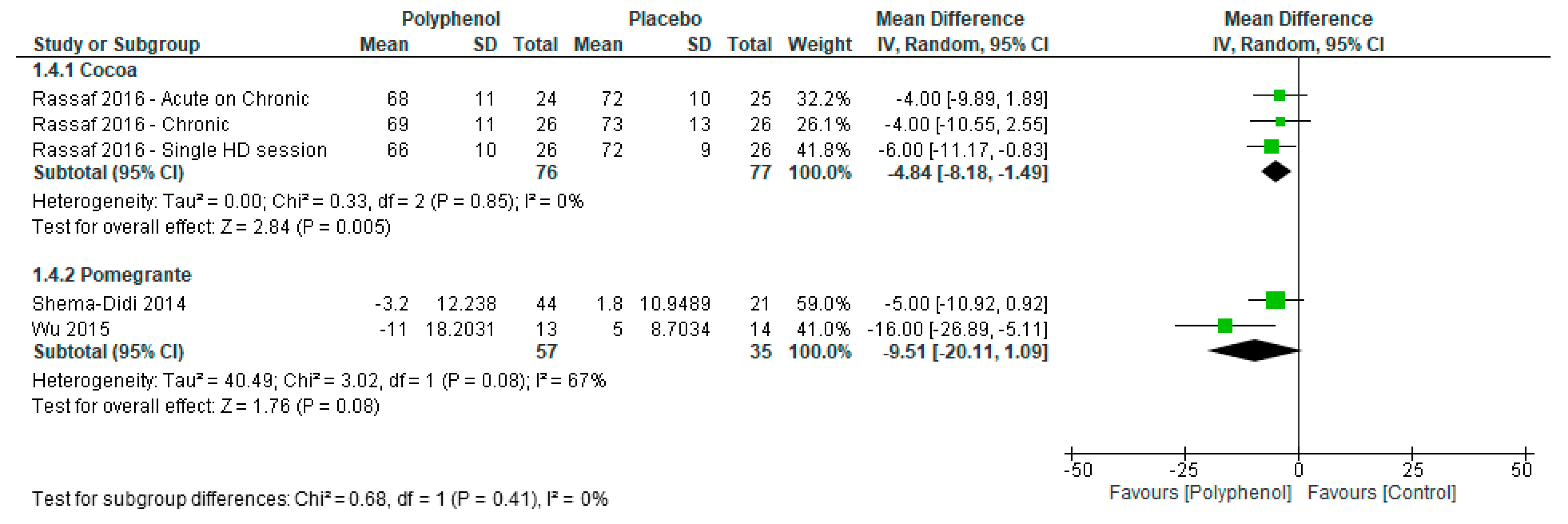
| Rassaf et al. 2016 [28] | Intervention: 450 mg cocoa-flavanol containing low-energy fruit-flavoured powder to be mixed as a drink. Comparator: low-energy fruit-flavoured powder to be mixed as a drink, matched for energy, protein, CHO, micronutrients, theobromine and caffeine. Dose: Two sachets (900 mg cocoa-flavanols) daily Duration: 30 days | Haemodynamics markers: 30-days post-baseline: Flow-mediated vasodilation of the brachial artery:
Systolic blood pressure: Acute
Diastolic blood pressure:
Systolic blood pressure:
IL-6:
Oxidized LDL-C:
|
| Pomegranate (Phenolic acids & Flavonoids) | ||
| Shema-Didi et al. 2012 [37] | Intervention: Commercial pomegranate juice (0.7 mmol polyphenols/100 mL juice). Comparator: Placebo juice containing artificial pomegranate extract, confirmed no polyphenol content. Dose: 100 mL, three times per week. Duration: 12-months. | Oxidative stress: 12-months post-baseline: CD11b:
Serum IL-6:
Common carotid intima-media thickness:
|
| Shema-Didi et al. 2014 [36] | Shema-Didi et al. 2012 [37] | Haemodynamics markers: 12-months post-baseline: Systolic blood pressure:
LDL-C:
|
| Shema-Didi et al. 2013 [26] | Intervention: Commercial pomegranate juice (0.7 mmol polyphenols/100 mL juice). Comparator: Placebo juice containing artificial pomegranate extract, confirmed no polyphenol content. Dose: 100 mL Duration: Once during the first hour of a haemodialysis session. | Oxidative stress markers: At the end of the dialysis session: Serum myeloperoxidase (MPO):
Serum polymorphonuclear leukocytes (PMNLs):
|
| Wu et al. 2015 [44] | Intervention: 1000 mg capsule of purified pomegranate polyphenol extract with 600–755 mg gallic acid equivalents. Comparator: Noncaloric placebo capsule not further described. Dose: 1 capsule daily Duration:6-months | Haemodynamics markers: 6-months post-baseline: Systolic blood pressure:
Oxygen radical absorbance capacity (ORAC)
CRP:
Total cholesterol:
|
| Quality Assessment | Number of Patients | Effect | Quality | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Number of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | Polyphenol | Placebo | Absolute (95% CI) | |
| Oxidative stress (myeloperoxidase) | ||||||||||
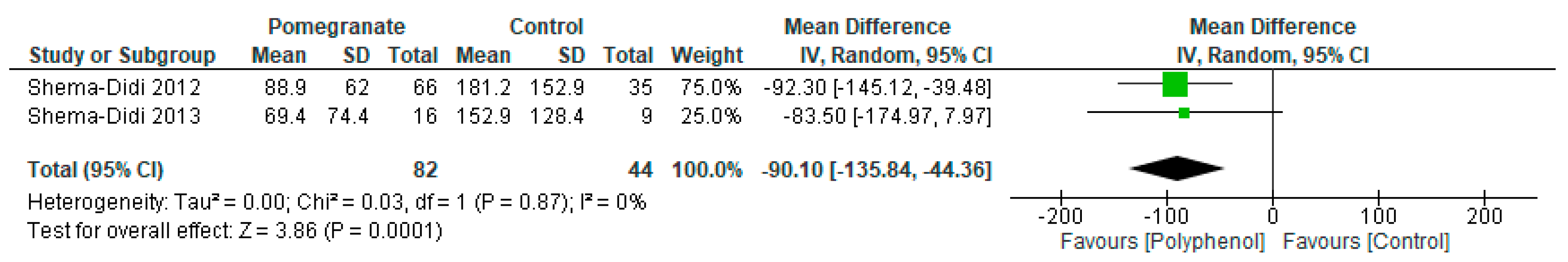
| 2 | Randomised trials | Not serious | Not serious | Not serious | Serious a | Strong effect | 82 | 44 | MD 90.1 SD lower (135.8 lower to 44.4 lower) | MODERATE |
| Oxidative stress (other markers—not pooled) | ||||||||||
| 5 | Randomised trials | Not serious | serious b | Not serious | Serious c | None | - | - | See comment | MODERATE |
| Inflammation (CRP) | ||||||||||
| 5 | Randomised trials | Not serious | very serious d | Not serious | Serious e | None | 96 | 99 | MD 1.9 mg/dL higher (2.2 lower to 6.1 higher) | VERY LOW |
| Inflammation (IL-6) | ||||||||||
| 2 | Randomised trials | Not serious | very serious d | Not serious | Serious e | Both pomegranate studies | 79 | 49 | MD 1.6 mg/dL lower (5.1 lower to 1.96 higher) | VERY LOW |
| Inflammation (AOPP) | ||||||||||
| 3 | Randomised trials | Not serious | very serious d | Not serious | Serious e | All pomegranate studies | 96 | 57 | MD 17.7 mg/dL lower (46.5 lower to 11.1 higher) | VERY LOW |
| Diastolic blood pressure | ||||||||||
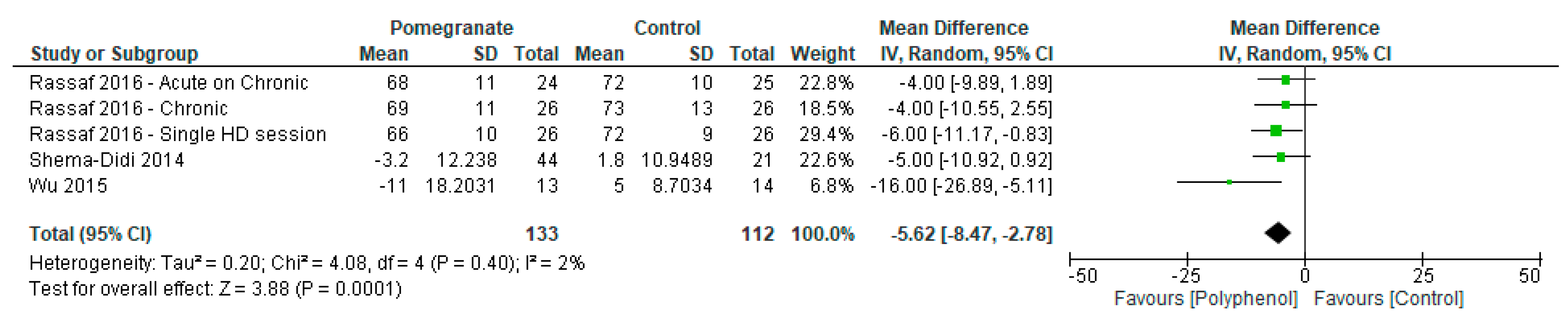
| 4 | Randomised trials | Not serious | Not serious | Not serious | Not serious | None | 133 | 112 | MD 5.6 mmHg lower (8.47 lower to 2.78 lower) | HIGH |
| Systolic blood pressure | ||||||||||
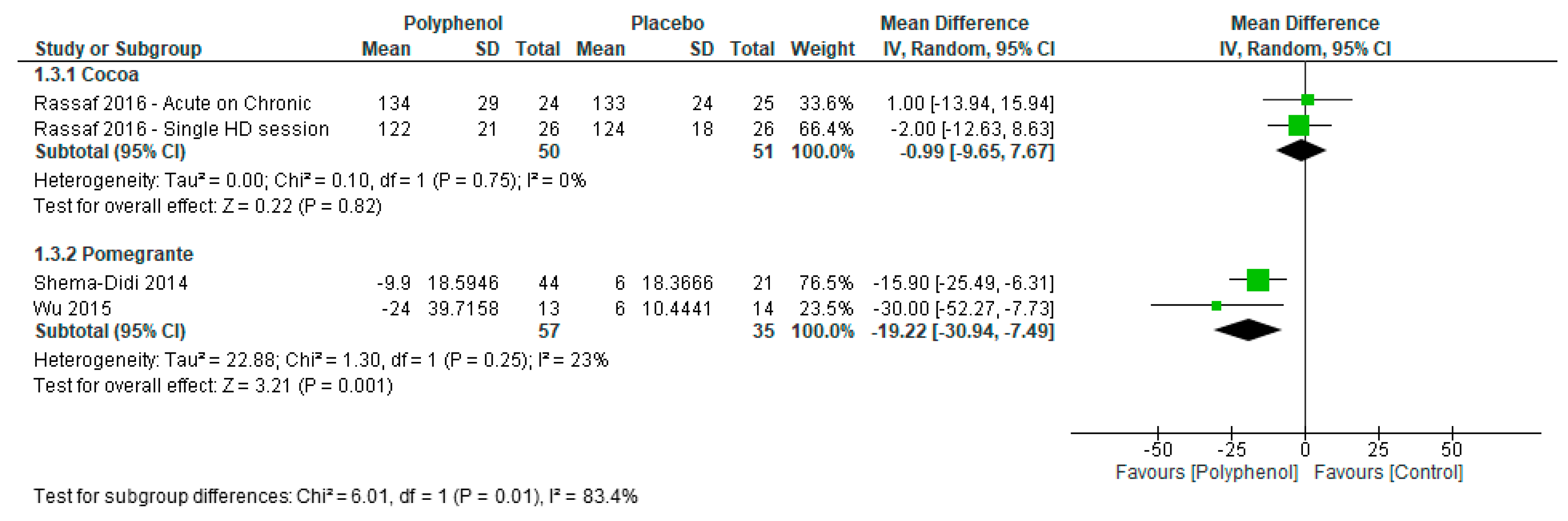
| 4 | Randomised trials | Not serious | Serious f | Not serious | Serious e | None | 107 | 86 | MD 10 mmHg lower (21.4 lower to 1.4 higher) | LOW |
| Hemodynamic measures (other markers—not pooled) | ||||||||||
| 4 | Randomised trials | Not serious | Serious g | Not serious | Serious h | None | - | - | Not pooled. See Table 2 | LOW |
| Lipid profile (TG) | ||||||||||
| 4 | Randomised trials | Not serious | Serious f | Not serious | Very serious e | None | 110 | 81 | MD 26.5 mg/dL lower (47.2 lower to 5.8 lower) | VERY LOW |
| Lipid profile (Total-C) | ||||||||||
| 4 | Randomised trials | Not serious | Very serious d | Not serious | Very serious e | None | 110 | 81 | MD 11.2 mg/dL lower (24.8 lower to 2.3 higher) | VERY LOW |
| Lipid profile (HDL-C) | ||||||||||
| 4 | Randomised trials | Not serious | Not serious | Not serious | Serious e | None | 110 | 81 | MD 2.4 mg/dL higher (0.1 lower to 4.8 higher) | MODERATE |
| Lipid profile (LDL-C) | ||||||||||
| 4 | Randomised trials | Not serious | Not serious | Not serious | Serious e | None | 110 | 81 | MD 3.3 mg/dL lower (14.5 lower to 7.8 higher) | MODERATE |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marx, W.; Kelly, J.; Marshall, S.; Nakos, S.; Campbell, K.; Itsiopoulos, C. The Effect of Polyphenol-Rich Interventions on Cardiovascular Risk Factors in Haemodialysis: A Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1345. https://doi.org/10.3390/nu9121345
Marx W, Kelly J, Marshall S, Nakos S, Campbell K, Itsiopoulos C. The Effect of Polyphenol-Rich Interventions on Cardiovascular Risk Factors in Haemodialysis: A Systematic Review and Meta-Analysis. Nutrients. 2017; 9(12):1345. https://doi.org/10.3390/nu9121345
Chicago/Turabian StyleMarx, Wolfgang, Jaimon Kelly, Skye Marshall, Stacey Nakos, Katrina Campbell, and Catherine Itsiopoulos. 2017. "The Effect of Polyphenol-Rich Interventions on Cardiovascular Risk Factors in Haemodialysis: A Systematic Review and Meta-Analysis" Nutrients 9, no. 12: 1345. https://doi.org/10.3390/nu9121345
APA StyleMarx, W., Kelly, J., Marshall, S., Nakos, S., Campbell, K., & Itsiopoulos, C. (2017). The Effect of Polyphenol-Rich Interventions on Cardiovascular Risk Factors in Haemodialysis: A Systematic Review and Meta-Analysis. Nutrients, 9(12), 1345. https://doi.org/10.3390/nu9121345





