Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Randomised Controlled Trials and Dose of DHA
3.2. Randomised Controlled Trials with Blood n-3 Assessments and Comparison to Omega-3 Index
3.3. Importance of the Assessment of Omega-3 Fatty Acid Status Pre- and Post-Supplementation
3.4. Evidence from Epidemiological Studies
3.5. Duration of Interventions
3.6. Evidence for the Beneficial Effect of High Dose n-3 LCPUFA Post-Myocardial Infarction
4. Summary
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Makrides, M.; Gibson, R.A. Long-chain polyunsaturated fatty acid requirements during pregnancy and lactation. Am. J. Clin. Nutr. 2000, 71, 307S–311S. [Google Scholar] [PubMed]
- Sattar, N.; Berry, C.; Greer, I.A. Essential fatty acids in relation to pregnancy complications and fetal development. Br. J. Obstet. Gynaecol. 1998, 105, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.J.; Onyiaodike, C.C.; Brown, E.A.; Jordan, F.; Murray, H.; Nibbs, R.J.; Sattar, N.; Lyall, H.; Nelson, S.M.; Freeman, D.J. Maternal plasma DHA levels increase prior to 29 days post-LH surge in women undergoing frozen embryo transfer: A prospective, observational study of human pregnancy. J. Clin. Endocrinol. Metab. 2016, 101, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Montgomery, P. Docosahexaenoic acid for reading, cognition and behavior in children aged 7–9 years: A randomized, controlled trial (the dolab study). PLoS ONE 2012, 7, e43909. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, P.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Richardson, A.J. Low blood long chain omega-3 fatty acids in UK children are associated with poor cognitive performance and behavior: A cross-sectional analysis from the DOLAB study. PLoS ONE 2013, 8, e66697. [Google Scholar] [CrossRef]
- Stonehouse, W. Does consumption of LC omega-3 PUFA enhance cognitive performance in healthy school-aged children and throughout adulthood? Evidence from clinical trials. Nutrients 2014, 6, 2730–2758. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Milte, C.M.; Street, S.J.; Buckley, J.D.; Coates, A.M.; Petkov, J.; Howe, P.R. Effects of n-3 fatty acids, EPA v. DHA, on depressive symptoms, quality of life, memory and executive function in older adults with mild cognitive impairment: A 6-month randomised controlled trial. Br. J. Nutr. 2012, 107, 1682–1693. [Google Scholar] [PubMed]
- GISSI Prevezione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin e after myocardial infarction: Results of the GISSI-prevenzione trial. Lancet 1999, 354, 447–455.
- Bang, H.O.; Dyerberg, J.; Sinclair, H.M. The composition of the Eskimo food in north western Greenland. Am. J. Clin. Nutr. 1980, 33, 2657–2661. [Google Scholar] [PubMed]
- Dyerberg, J.B.H.; Stoffersen, E.; Moncada, S.; Vane, J.R. Eicosapentaenoic acid and prevention of thrombosis and atherosclerosis? Lancet 1978, 312, 117–119. [Google Scholar] [CrossRef]
- Mozaffarian, D. Jelis, fish oil, and cardiac events. Lancet 2007, 369, 1062–1063. [Google Scholar] [CrossRef]
- Iso, H.; Kobayashi, M.; Ishihara, J.; Sasaki, S.; Okada, K.; Kita, Y.; Kokubo, Y.; Shoichiro, T.; JPHC Study Group. Intake of fish and n-3 fatty acids and risk of coronary heart disease among Japanese. Circulation 2006, 113, 195–202. [Google Scholar] [PubMed]
- Hibbeln, J.R.; Nieminen, L.R.G.; Blasbalg, T.L.; Riggs, J.A.; Lands, W.E.M. Healthy intakes of n-3 and n-6 fatty acids: Estimations considering worldwide diversity. Am. J. Clin. Nutr. 2006, 83 (Suppl. 6), 1483S–1493S. [Google Scholar] [PubMed]
- Nakamura, Y.; Ueshima, H.; Okuda, N.; Higashiyama, A.; Kita, Y.; Kadowaki, T.; Okamura, T.; Murakami, Y.; Okayama, A.; Choudhury, S.R.; et al. Relation of dietary and other lifestyle traits to difference in serum adiponection concentration of Japanese in Japan and Hawaii: The interlipid study. Am. J. Clin. Nutr. 2008, 88, 424–430. [Google Scholar] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed]
- Adkins, Y.; Kelley, D.S. Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. J. Nutr. Biochem. 2010, 21, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Fukuda, D.; Aihara, K.I.; Akaike, M.; Shimabukuro, M.; Sata, M. N-3 polyunsaturated fatty acids: Promising nutrients for preventing cardiovascular disease. J. Atheroscler. Thromb. 2017, 24, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- McLennan, P.L.; Howe, P.R.; Abeywardena, M.; Muggli, R.; Raederstorff, D.; Mano, M.; Rayner, T.; Head, R. The cardiovascular protective role of docosahexaenoic acid. Eur. J. Pharmacol. 1996, 300, 83–89. [Google Scholar] [CrossRef]
- McLennan, P.L. Cardiac physiology and clinical efficacy of dietary fish oil clarified through cellular mechanisms of omega-3 polyunsaturated fatty acids. Eur. J. Appl. Physiol. 2014, 114, 1333–1356. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.T.; Dai, J.H.; Gao, Q. Effects of omega-3 fatty acid on major cardiovascular events and mortality in patients with coronary heart disease: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.M.; Myung, S.K.; Lee, Y.J.; Seo, H.G.; Korean Meta-analysis Study Group. Efficacy of omega-3 fatty acid supplements (eicosapentaenoic acid and docosahexaenoic acid) in the secondary prevention of cardiovascular disease: A meta-analysis of randomized, double-blind, placebo-controlled trials. Arch. Intern. Med. 2012, 172, 686–694. [Google Scholar] [PubMed]
- Nestel, P.; Clifton, P.; Colquhoun, D.; Noakes, M.; Mori, T.A.; Sullivan, D.; Thomas, B. Indications for omega-3 long chain polyunsaturated fatty acid in the prevention and treatment of cardiovascular disease. Heart Lung Circ. 2015, 24, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Kimmig, L.M.; Karalis, D.G. Do omega-3 polyunsaturated fatty acids prevent cardiovascular disease? A review of the randomized clinical trials. Lipid Insights 2013, 6, 13. [Google Scholar] [PubMed]
- Svensson, M.; Schmidt, E.B.; Jorgensen, K.A.; Christensen, J.H.; Group, O.S. N-3 fatty acids as secondary prevention against cardiovascular events in patients who undergo chronic hemodialysis: A randomized, placebo-controlled intervention trial. Clin. J. Am. Soc. Nephrol. 2006, 1, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- GISSI-HF Investigators. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the gissi-hf trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M.; Alpha Omega Trial Group. N-3 fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [PubMed]
- Rauch, B.; Schiele, R.; Schneider, S.; Diller, F.; Victor, N.; Gohlke, H.; Gottwik, M.; Steinbeck, G.; Del Castillo, U.; Sack, R.; et al. Omega, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010, 122, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Einvik, G.; Klemsdal, T.O.; Sandvik, L.; Hjerkinn, E.M. A randomized clinical trial on n-3 polyunsaturated fatty acids supplementation and all-cause mortality in elderly men at high cardiovascular risk. Eur. J. Cardiovasc. Prev. Rehabil. 2010, 17, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S.; SU.FOL.OM3 Collaborative Group. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. Br. Med. J. 2010, 341, c6273. [Google Scholar]
- Moertl, D.; Hammer, A.; Steiner, S.; Hutuleac, R.; Vonbank, K.; Berger, R. Dose-dependent effects of omega-3-polyunsaturated fatty acids on systolic left ventricular function, endothelial function, and markers of inflammation in chronic heart failure of nonischemic origin: A double-blind, placebo-controlled, 3-arm study. Am. Heart J. 2011, 161. [Google Scholar] [CrossRef] [PubMed]
- The Origin Trial Investigators. N-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N. Engl. J. Med. 2012, 367, 309–318. [Google Scholar]
- Wu, J.H.; Marchioli, R.; Silletta, M.G.; Macchia, A.; Song, X.; Siscovick, D.S.; Harris, W.S.; Masson, S.; Latini, R.; Albert, C.; et al. Plasma phospholipid omega-3 fatty acids and incidence of postoperative atrial fibrillation in the opera trial. J. Am. Heart Assoc. 2013, 2, e000397. [Google Scholar] [CrossRef] [PubMed]
- The Risk and Prevention Study Collaborative Group. N-3 fatty acids in patients with multiple cardiovascular risk factors. N. Engl. J. Med. 2013, 368, 1800–1808. [Google Scholar]
- Writing Group for the AREDS2 Research Group. Effect of long-chain omega-3 fatty acids and lutein + zeaxanthin supplements on cardiovascular outcomes: Results of the age-related eye disease study 2 (AREDS2) randomized clinical trial. JAMA Intern. Med. 2014, 174, 763–771. [Google Scholar]
- Hara, M.; Sakata, Y.; Nakatani, D.; Suna, S.; Usami, M.; Matsumoto, S.; Hamasaki, T.; Doi, Y.; Nishino, M.; Sato, H.; et al. Low levels of serum n-3 polyunsaturated fatty acids are associated with worse heart failure-free survival in patients after acute myocardial infarction. Circ. J. 2013, 77, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Erkkila, A.T.; Matthan, N.R.; Herrington, D.M.; Lichtenstein, A.H. Higher plasma docosahexaenoic acid is associated with reduced progression of coronary atherosclerosis in women with cad. J. Lipid Res. 2006, 47, 2814–2819. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Von Schacky, C. The omega-3 index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [PubMed]
- Stark, K.D.; Aristizabal Henao, J.J.; Metherel, A.H.; Pilote, L. Translating plasma and whole blood fatty acid compositional data into the sum of eicosapentaenoic and docosahexaenoic acid in erythrocytes. Prostaglandins Leukot. Essent. Fatty Acids 2016, 104, 1–10. [Google Scholar] [PubMed]
- Farquharson, A.L.; Metcalf, R.G.; Sanders, P.; Stuklis, R.; Edwards, J.R.; Gibson, R.A.; Cleland, L.G.; Sullivan, T.R.; James, M.J.; Young, G.D. Effect of dietary fish oil on atrial fibrillation after cardiac surgery. Am. J. Cardiol. 2011, 108, 851–856. [Google Scholar] [PubMed]
- James, M.J.; Sullivan, T.R.; Metcalf, R.G.; Cleland, L.G. Pitfalls in the use of randomised controlled trials for fish oil studies with cardiac patients. Br. J. Nutr. 2014, 112, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Bell, G.A.; Kantor, E.D.; Lampe, J.W.; Kristal, A.R.; Heckbert, S.R.; White, E. Intake of long-chain omega-3 fatty acids from diet and supplements in relation to mortality. Am. J. Epidemiol. 2014, 179, 710–720. [Google Scholar] [CrossRef] [PubMed]
- Del Gobbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wennberg, M.; Yakoob, M.Y.; Chiuve, S.E.; Dela Cruz, L.; Frazier-Wood, A.C.; et al. Omega-3 polyunsaturated fatty acid biomarkers and coronary heart disease: Pooling project of 19 cohort studies. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Masson, S.; Barlera, S.; Milani, V.; Pileggi, S.; Franzosi, M.G.; Marchioli, R.; Tognoni, G.; Tavazzi, L.; Latini, R.; et al. Red blood cell oleic acid levels reflect olive oil intake while omega-3 levels reflect fish intake and the use of omega-3 acid ethyl esters: The Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico-Heart Failure trial. Nutr. Res. 2016, 36, 989–994. [Google Scholar]
- Heydari, B.; Abdullah, S.; Pottala, J.V.; Shah, R.; Abbasi, S.; Mandry, D.; Francis, S.A.; Lumish, H.; Ghoshhajra, B.B.; Hoffmann, U.; et al. Effect of omega-3 acid ethyl esters on left ventricular remodeling after acute myocardial infarction: The omega-remodel randomized clinical trial. Circulation 2016, 134, 378–391. [Google Scholar] [CrossRef] [PubMed]
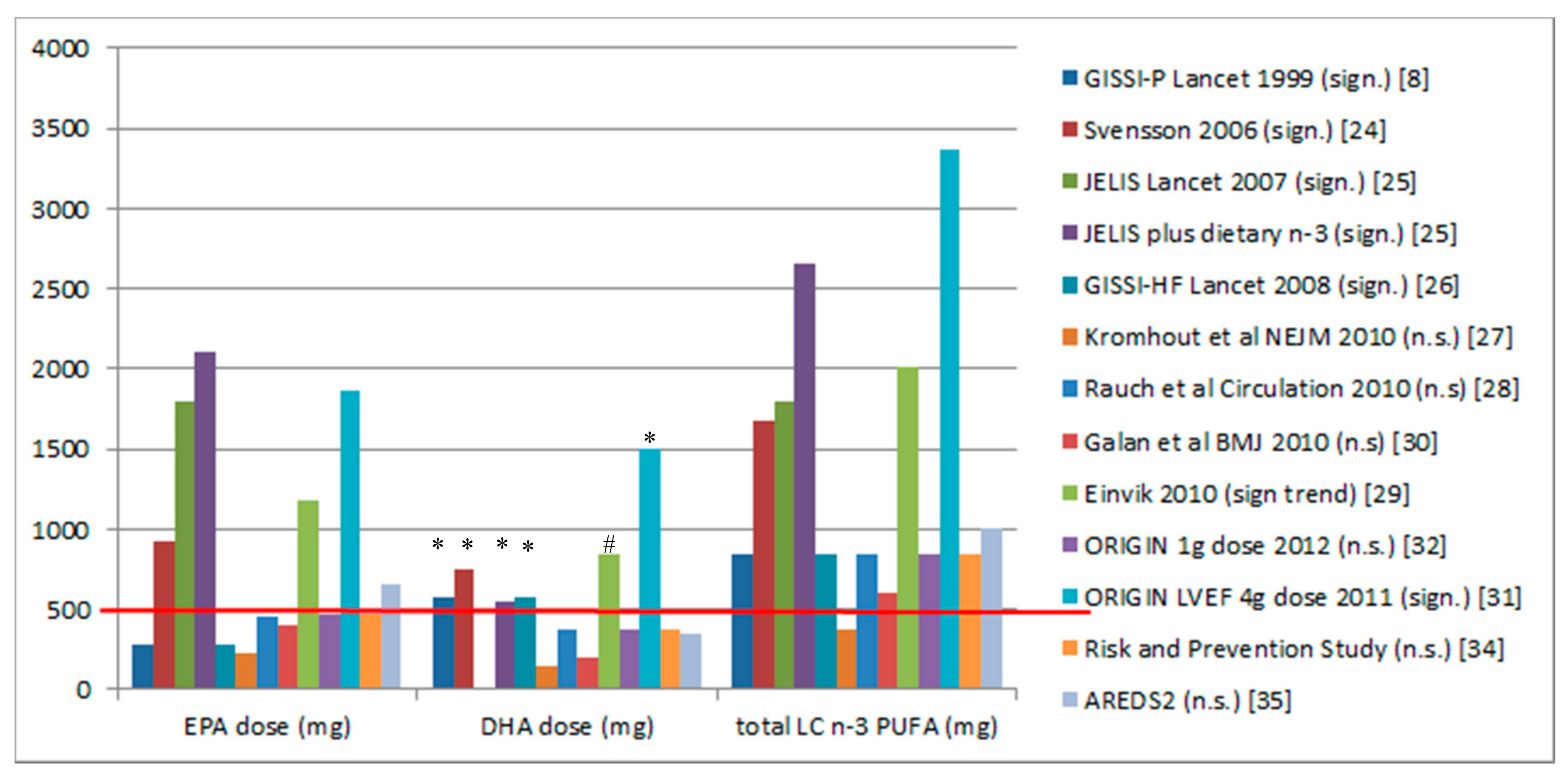
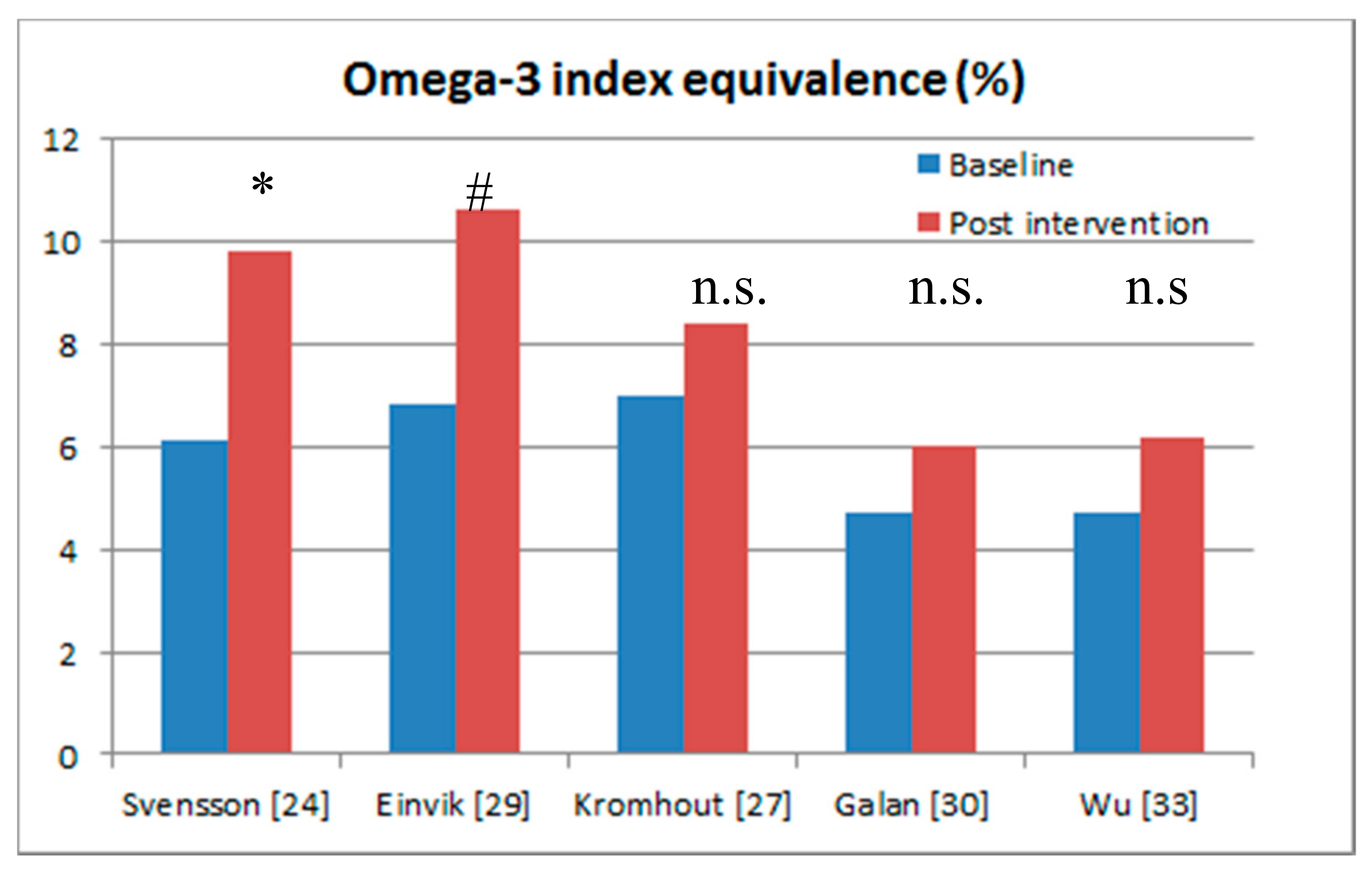
| Reference | Blood Sample Type | Equation and Pearson’s Correlation [39] | Omega-3 Index Equivalence at Baseline | Omega-3 Index Equivalence Post-Intervention | Outcomes |
|---|---|---|---|---|---|
| Svensson et al. 2006 [24] | Serum phospholipid fatty acids | Y = 0.93x + 0.55 R = 0.94 | 6.1% | 9.8% | Myocardial infarction 3.9% versus 12.6% (p = 0.036) Major coronary events 6.8% versus 16.5% (p = 0.043) Note the large increase in omega-3 fatty acid status (38%) achieved Omega-3 fatty acid status is in the cardio-protective zone. |
| Einvik et al. 2010 [29] | Serum fatty acids | Y = 0.94x + 1.17 R = 0.74 | 6.8% | 10.6% | All-cause mortality 5% versus 8.5% (p = 0.063) Note the large increase in omega-3 fatty acid status (36%) achieved Omega-3 fatty acid status is in the cardio-protective zone. |
| Kromhout et al. 2010 [27] | Plasma cholesteryl esters fatty acids | Y = 1.59x + 2.05 R = 0.85 | 7.0% | 8.4% | No significant outcomes. Note the small increase in omega-3 fatty acid status (17%) achieved. |
| Galan et al. 2010 [30] | Plasma fatty acids | Y = 0.94x + 1.17 R = 0.74 | 4.7% | 6.0% | No significant outcomes. Note the small increase in omega-3 fatty acid status (22%) achieved. Omega-3 fatty acid status of 6% is not in the cardio-protective zone |
| Wu et al. 2013 [33] | Serum phospholipid fatty acids | Y = 0.93x + 0.55 R = 0.94 | 4.7% | 6.2% | No significant outcomes. Note the small increase in omega-3 fatty acid status (24%) achieved. Omega-3 fatty acid status of 6% is not in the cardio-protective zone |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyer, B.J.; Groot, R.H.M.d. Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA. Nutrients 2017, 9, 1305. https://doi.org/10.3390/nu9121305
Meyer BJ, Groot RHMd. Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA. Nutrients. 2017; 9(12):1305. https://doi.org/10.3390/nu9121305
Chicago/Turabian StyleMeyer, Barbara J., and Renate H. M. de Groot. 2017. "Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA" Nutrients 9, no. 12: 1305. https://doi.org/10.3390/nu9121305
APA StyleMeyer, B. J., & Groot, R. H. M. d. (2017). Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA. Nutrients, 9(12), 1305. https://doi.org/10.3390/nu9121305






