Impact of High-Dose Multi-Strain Probiotic Supplementation on Neurocognitive Performance and Central Nervous System Immune Activation of HIV-1 Infected Individuals
Abstract
:1. Introduction
2. Materials and Methods
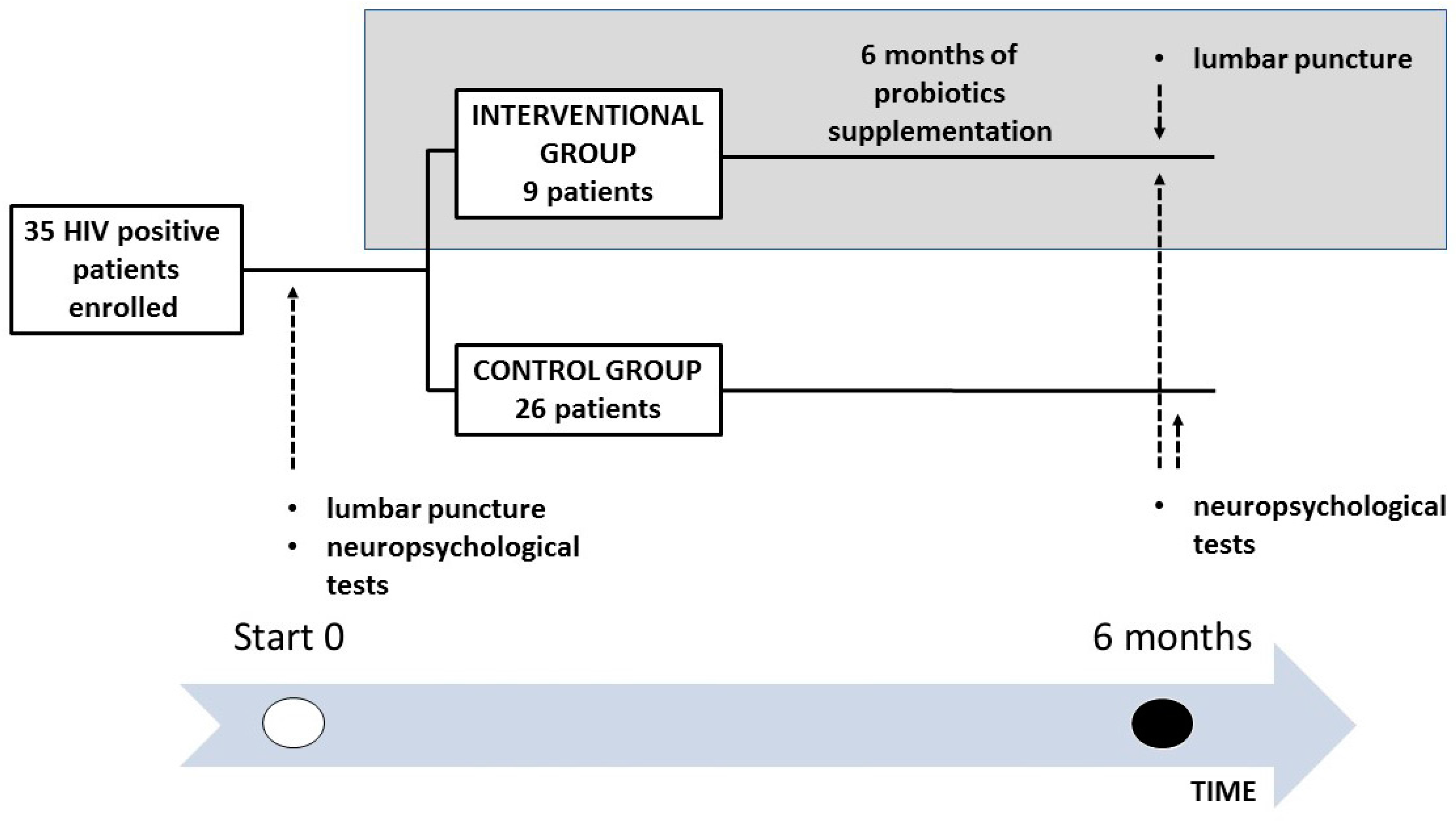
2.1. Study Design, Recruitment, Study Eligibility Criteria and Ethics Statement
2.2. Neuropsychological Test Battery
2.3. Evaluation of Neopterin Levels by ELISA Assay
2.4. Bacterial DNA Isolation from Fecal Samples
2.5. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics of HIV-1-Positive Patients
3.2. Correlations between Neuroinflammation and Neuropsychological Impairment
3.3. Results of Probiotic Supplementation: Reduction of Neuroinflammation and Recovery of Neuropsychological Impairment
3.4. Adherence to Probiotic Supplementation and Safety of the Treatment
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heaton, R.K.; Franklin, D.R.; Ellis, R.J.; McCutchan, J.A.; Letendre, S.L.; LeBlanc, S.; Corkran, S.H.; Duarte, N.A.; Clifford, D.B.; Woods, S.P.; et al. HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: Differences in rates, nature, and predictors. J. Neurovirol. 2011, 17, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Stilling, R.M.; Stanton, C.; Cryan, J.F. Collective unconscious: How gut microbes shape human behavior. J. Psychiatr. Res. 2015, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci. 2016, 8, 256. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Wang, K.; Hu, J. Effect of Probiotics on Depression: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2016, 8, 483. [Google Scholar] [CrossRef] [PubMed]
- Zilberman-Schapira, G.; Zmora, N.; Itav, S.; Bashiardes, S.; Elinav, H.; Elinav, E. The gut microbiome in human immunodeficiency virus infection. BMC Med. 2016, 14, 83. [Google Scholar] [CrossRef] [PubMed]
- Hagberg, L.; Cinque, P.; Gisslen, M.; Brew, B.J.; Spudich, S.; Bestetti, A.; Price, R.W.; Fuchs, D. Cerebrospinal fluid neopterin: An informative biomarker of central nervous system immune activation in HIV-1 infection. AIDS Res. Ther. 2010, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.E.; McArthur, J.C.; Cornblath, D.R. Neopterin and interferon-gamma in serum and cerebrospinal fluid of patients with HIV-associated neurologic disease. Neurology 1991, 41, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Sönnerborg, A.B.; von Stedingk, L.V.; Hansson, L.O.; Strannegård, O.O. Elevated neopterin and β2-microglobulin levels in blood and cerebrospinal fluid occur early in HIV-1 infection. AIDS 1989, 3, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, D.; Chiodi, F.; Albert, J.; Asjö, B.; Hagberg, L.; Hausen, A.; Norkrans, G.; Reibnegger, G.; Werner, E.R.; Wachter, H. Neopterin concentrations in cerebrospinal fluid and serum of individuals infected with HIV-1. AIDS 1989, 3, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Eden, A.; Price, R.W.; Spudich, S.; Fuchs, D.; Hagberg, L.; Gisslén, M. Immune activation of the central nervous system is still present after >4 years of effective HAART. J. Infect. Dis. 2007, 196, 1779–1783. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.; Yiannoutsos, C.T.; Fuchs, D.; Price, R.W.; Crozier, K.; Hagberg, L.; Spudich, S.; Gisslén, M. Cerebrospinal fluid neopterin decay characteristics after initiation of antiretroviral therapy. J. Neuroinflamm. 2013, 10, 10–62. [Google Scholar] [CrossRef] [PubMed]
- Viaccoz, A.; Ducray, F.; Tholance, Y.; Barcelos, G.K.; Thomas-Maisonneuve, L.; Ghesquières, H.; Meyronet, D.; Quadrio, I.; Cartalat-Carel, S.; Louis-Tisserand, G.; et al. CSF neopterin level as a diagnostic marker in primary central nervous system lymphoma. Neuro Oncol. 2015, 17, 1497–1503. [Google Scholar] [CrossRef] [PubMed]
- Tincati, C.; Douek, D.; Marchetti, G. Gut barrier structure, mucosal immunity and intestinal microbiota in the pathogenesis and treatment of HIV infection. AIDS Res. Ther. 2016, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- D’ettorre, G.; Rossi, G.; Scagnolari, C.; Andreotti, M.; Giustini, N.; Serafino, S.; Schietroma, I.; Scheri, G.C.; Fard, S.N.; Trinchieri, V. Probiotic supplementation promotes a reduction in T-cell activation, an increase in Th17 frequencies, and a recovery of intestinal epithelium integrity and mitochondrial morphology in ART-treated HIV-1-positive patients. Immun. Inflamm. Dis. 2017, 20. [Google Scholar] [CrossRef] [PubMed]
- Scagnolari, C.; Corano Scheri, G.; Selvaggi, C.; Schietroma, I.; Fard, S.N.; Mastrangelo, A.; Giustini, N.; Serafino, S.; Pinacchio, C.; Pavone, P. Probiotics Differently Affect Gut-Associated Lymphoid Tissue Indolamine-2,3-Dioxygenase mRNA and Cerebrospinal Fluid Neopterin Levels in Antiretroviral-Treated HIV-1 Infected Patients: A Pilot Study. Int. J. Mol. Sci. 2016, 17, 1639. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Gray, A.M.; Erickson, M.A.; Salameh, T.S.; Damodarasamy, M.; Sheibani, N.; Meabon, J.S.; Wing, E.E.; Morofuji, Y.; Cook, D.G. Lipopolysaccharide-induced blood-brain barrier disruption: Roles of cyclooxygenase, oxidative stress, neuroinflammation, and elements of the neurovascular unit. J. Neuroinflamm. 2015, 12, 223. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, G.; Fratino, M.; Selvaggi, C.; Giustini, N.; Serafino, S.; Schietroma, I.; Corano Scheri, G.; Pavone, P.; Passavanti, G.; Alunni Fegatelli, D.; et al. A pilot study on the effects of probiotic supplementation on neuropsychological performance and microRNA-29a-c levels in antiretroviral-treated HIV-1-infected patients. Brain Behav. 2017, 7, e00756. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, G.; Vullo, V.; d’Ettorre, G. Single-strain vs. multi-strain probiotic supplementation strategy against rheumatoid arthritis. Arthritis Rheumatol. 2017. [Google Scholar] [CrossRef] [PubMed]
- D’ettorre, G.; Ceccarelli, G.; Giustini, N.; Serafino, S.; Calantone, N.; de Girolamo, G.; Bianchi, L.; Bellelli, V.; Ascoli-Bartoli, T.; Marcellini, S. Probiotics Reduce Inflammation in Antiretroviral Treated, HIV-Infected Individuals: Results of the “Probio-HIV” Clinical Trial. PLoS ONE 2015, 10, e0137200. [Google Scholar] [CrossRef] [PubMed]
- Scheri, G.C.; Fard, S.N.; Schietroma, I.; Mastrangelo, A.; Pinacchio, C.; Giustini, N.; Serafino, S.; de Girolamo, G.; Cavallari, E.N.; Statzu, M. Modulation of Tryptophan/Serotonin Pathway by Probiotic Supplementation in Human Immunodeficiency Virus-Positive Patients: Preliminary Results of a New Study Approach. Int. J. Tryptophan Res. 2017. [Google Scholar] [CrossRef]
- O’Hara, A.M.; Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.V.; Suzuki, K.; Zhao, J. Probiotics: A proactive approach to health. A symposium report. Br. J. Nutr. 2015, 114 (Suppl. 1), S1–S15. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Probiotics Supplementation Group | Control Group | p-Value |
|---|---|---|---|
| N of subjects | 9 | 26 | |
| Males | 9 | 24 | |
| Females | 0 | 2 | |
| Age | 45 (35–52.5) | 43 (38.2–53) | 0.097 |
| Years from diagnosis | 14 (5–19.5) | 12.5 (7–23) | 0.593 |
| Years on ARV treatment | 14 (6.5–16) | 12.5 (7–20) | 0.373 |
| T CD4 nadir | 180 cell/μL (40–438) | 288 cell/μL (57–407) | 0.678 |
| T CD4 at enrollment | 651 cell/μL (563–883) | 526 cell/μL (340–663) | 0.515 |
| CPE score | 7 (7–7.25) | 7 (7–8) | 0.527 |
| HIV-RNA in CSF | <37 copies/mL | <37 copies/mL | - |
| Neopterin in CSF | 34.14 nmol/L (22.5–65.2) | 12.3 nmol/L (10.1–14.8) | 0.008 |
| Neurocognitive Tests | Probiotics Supplementation Group (T0) | Control Group (T0) | p-Value |
|---|---|---|---|
| Rey–Osterrieth Complex Figure immediate recall (more is better) | 16.6 (15.9–17.8) | 13.1 (8.5–22.0) | 0.860 |
| Rey–Osterrieth Complex Figure delayed recall (more is better) | 15.5 (14.3–17.8) | 11.6 (6.3–19.9) | 0.280 |
| Rey Auditory Verbal Learning Test immediate recall (more is better) | 46.0 (29.4–47.4) | 30.6 (27.9–40.0) | 0.214 |
| Rey Auditory Verbal Learning Test delayed recall (more is better) | 9.2 (5.6–10.9) | 5.2 (3.5–8.0) | 0.360 |
| Rey Auditory Verbal Learning Test recognition (more is better) | 98.0 (90.0–100.0) | 96.0 (92.0–98.0) | 0.400 |
| Verbal Fluency (more is better) | 15.0 (13.7–16.0) | 15.9 (13.9–18.0) | 0.314 |
| Phonological Verbal Fluency (more is better) | 30.0 (23.6–39.2) | 26.7 (21.6–35.1) | 0.906 |
| Semantic Verbal Fluency (more is better) | 47.0 (33.5–57.5) | 39.0 (33.0–42.0) | 0.173 |
| Visual Search Test (more is better) | 46.2 (45.1–60.0) | 46.7 (40.2–50.6) | 0.374 |
| Test of Weights and Measures Estimation—Time (more is better) | 19.0 (13.0–23.5) | 22.0 (19.0–24.0) | 0.074 |
| Test of Weights and Measures Estimation—Weight (more is better) | 19.0 (14.0–20.5) | 19.0 (16.5–21.0) | 0.933 |
| Test of Weights and Measures Estimation—Total (more is better) | 38.0 (30.5–46.5) | 40.0 (36.5–43.5) | 0.594 |
| Raven’s Standard Progressive Matrices (more is better) | 27.5 (22.6–31.6) | 28.3 (25.7–31.8) | 0.327 |
| Verbal Span forward (more is better) | 5.0 (3.5–5.7) | 5.2 (4.9–6.0) | 0.065 |
| Verbal Span backward (more is better) | 5.0 (4.0–5.0) | 4.0 (3.0–4.5) | 0.161 |
| Corsi Block Tapping Test forward (more is better) | 4.7 (4.0–5.2) | 5.5 (4.7–6.0) | 0.078 |
| Corsi Block Tapping Test backward (more is better) | 4.0 (3.0–4.0) | 5.0 (4.0–6.0) | 0.196 |
| Aachener Aphasia Test (more is better) | 9.0 (9.0–9.0) | 9.0 (9.0–9.0) | 1.000 |
| Trail Making Test A (s) (less is better) | 50.0 (44.0–62.0) | 50.0 (41.0–67.0) | 0.575 |
| Trail Making Test B (s) (less is better) | 115.0 (93.0–142.0) | 97.0 (77.5–144.5) | 0.086 |
| Performed Neurocognitive Tests | Probiotics Supplementation Group (T0 vs. T6) | Control Group (T0 vs. T6) | Probiotics Supplementation Group vs. Control Group (T6 vs.T6) |
|---|---|---|---|
| Rey–Osterrieth Complex Figure immediate recall (more is better) | 16.6 vs. 22.0 (p = 0.007) | 13.1 vs. 14.7 (p = 0.603) | 22.0 (19.0–23.7) vs. 14.7 (8.8–20.3) (p = 0.011) |
| Rey-Osterrieth Complex Figure delayed recall (more is better) | 15.5 vs. 22.4 (p = 0.008) | 11.6 vs. 12.6 (p = 0.369) | 22.4 (22.0–25.5) vs. 12.6 (5.7–19.1) (p = 0.011) |
| Rey Auditory Verbal Learning Test immediate recall (more is better) | 46.0 vs. 53.0 (p = 0.028) | 30.6 vs. 32.5 (p = 0.619) | 53.0 (49.3–55.6) vs. 32.5 (28.7–37.5) (p = 0.008) |
| Rey Auditory Verbal Learning Test delayed recall (more is better) | 9.2 vs. 12.0 (p = 0.034) | 5.2 vs. 5.3 (p = 0.241) | 12 .0 (10.7–13.8) vs. 5.3 (4.3–8.0) (p = 0.008) |
| Rey Auditory Verbal Learning Test recognition (more is better) | 98.0 vs. 99.0 (p = 0.176) | 96.0 vs. 96.0 (p = 0.575) | 99.0 (97.0–100.0) vs. 96.0 (92.0–98.0) (p = 0.013) |
| Verbal Fluency (more is better) | 15.0 vs. 15.9 (p = 0.233) | 15.9 vs. 15.3 (p = 0.152) | 15.9 (14.1–18.0) vs. 15.3 (13.7–16.8) (p = 0.594) |
| Phonological Verbal Fluency (more is better) | 30.0 vs. 44.0 (p = 0.028) | 26.7 vs. 25.9 (p = 0.271) | 44.0 (42.5–45.0) vs. 25.9 (21.6–36.1) (p = 0.021) |
| Semantic Verbal Fluency (more is better) | 47.0 vs. 49.0 (p = 0.373) | 39 vs. 38.0 (p = 0.396) | 49.0 (46.0–49.0) vs. 38.0 (33.5–43.5) (p = 0.123) |
| Visual Search Test (more is better) | 46.2 vs. 49.0 (p = 0.859) | 46.7 vs. 46.7 (p = 1.000) | 49.0 (45.6–50.0) vs. 46.7 (40.2–50.6) (p = 0.722) |
| Test of Weights and Measures Estimation—Time (more is better) | 19.0 vs. 23.0 (p = 0.038) | 22.0 vs. 22.0 (p = 0.776) | 23.0 (21.0–23.5) vs. 22.0 (18.0–25.0) (p = 0.512) |
| Test of Weights and Measures Estimation—Weight (more is better) | 19.0 vs. 21.0 (p = 0.027) | 19 vs. 20.0 (p = 0.843) | 21.0 (20.5–23.5) vs. 20.0 (15.5–21.5) (p = 0.08) |
| Test of Weights and Measures Estimation—Total (more is better) | 38.0 vs. 45.0 (p = 0.138) | 40.0 vs. 40.0 (p = 0.776) | 45.0 (41.5–46.0) vs. 40.0 (35.5–44.0) (p = 0.02) |
| Raven’s Standard Progressive Matrices (more is better) | 25.7 vs. 30.0 (p = 0.208) | 28.3 vs. 28.3 (p = 0.939) | 30.0 (28.5–33.5) vs. 28.3 (25.2–31.6) (p = 0.374) |
| Verbal Span forward (more is better) | 5.0 vs. 5.0 (p = 0.121) | 5.2 vs. 5.2 (p = 0.632) | 5.0 (5.0–6.0) vs. 5.2 (4.6–6.0) (p = 0.551) |
| Verbal Span backward (more is better) | 5.0 vs. 5.0 (p = 1.000) | 4.0 vs. 4.0 (p = 0.344) | 5.0 (4.0–5.0) vs. 4.0 (3.75–5.0) (p = 0.206) |
| Corsi Block Tapping Test forward (more is better) | 4.7 vs. 5.2 (p = 0.049) | 5.5 vs. 5.2 (p = 0.980) | 5.2 (5.0–5.5) vs. 5.2 (5.0–6.0) (p = 0.888) |
| Corsi Block Tapping Test backward (more is better) | 4.0 vs. 4.0 (p = 0.180) | 5.0 vs. 5.0 (p = 0.317) | 4.0 (4.0–4.5) vs. 5.0 (4.0–5.0) (p = 0.102) |
| Aachener Aphasia Test (more is better) | 9.0 vs. 9.0 (p = 1.000) | 9.0 vs. 9.0 (p = 1.000) | 9.0 (9.0–9.0) vs. 9.0 (9.0–9.0) (p = 1.000) |
| Trail Making Test A (s) (less is better) | 50.0 vs. 43.0 (p = 0.041) | 50.0 vs. 51.0 (p = 0.747) | 43.0 (39.0–53.0) vs. 51.0 (40.5–65.5) (p = 0.674) |
| Trail Making Test B (s) (less is better) | 115.0 vs. 120.0 (p = 0.726) | 97.0 vs. 98.0 (p = 0.279) | 120.0 (76.0–138.0) vs. 98.0 (78.5–146.0) (p = 0.138) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceccarelli, G.; Brenchley, J.M.; Cavallari, E.N.; Corano Scheri, G.; Fratino, M.; Pinacchio, C.; Schietroma, I.; Fard, S.N.; Scagnolari, C.; Mezzaroma, I.; et al. Impact of High-Dose Multi-Strain Probiotic Supplementation on Neurocognitive Performance and Central Nervous System Immune Activation of HIV-1 Infected Individuals. Nutrients 2017, 9, 1269. https://doi.org/10.3390/nu9111269
Ceccarelli G, Brenchley JM, Cavallari EN, Corano Scheri G, Fratino M, Pinacchio C, Schietroma I, Fard SN, Scagnolari C, Mezzaroma I, et al. Impact of High-Dose Multi-Strain Probiotic Supplementation on Neurocognitive Performance and Central Nervous System Immune Activation of HIV-1 Infected Individuals. Nutrients. 2017; 9(11):1269. https://doi.org/10.3390/nu9111269
Chicago/Turabian StyleCeccarelli, Giancarlo, Jason M. Brenchley, Eugenio Nelson Cavallari, Giuseppe Corano Scheri, Mariangela Fratino, Claudia Pinacchio, Ivan Schietroma, Saeid Najafi Fard, Carolina Scagnolari, Ivano Mezzaroma, and et al. 2017. "Impact of High-Dose Multi-Strain Probiotic Supplementation on Neurocognitive Performance and Central Nervous System Immune Activation of HIV-1 Infected Individuals" Nutrients 9, no. 11: 1269. https://doi.org/10.3390/nu9111269
APA StyleCeccarelli, G., Brenchley, J. M., Cavallari, E. N., Corano Scheri, G., Fratino, M., Pinacchio, C., Schietroma, I., Fard, S. N., Scagnolari, C., Mezzaroma, I., Vullo, V., & D’Ettorre, G. (2017). Impact of High-Dose Multi-Strain Probiotic Supplementation on Neurocognitive Performance and Central Nervous System Immune Activation of HIV-1 Infected Individuals. Nutrients, 9(11), 1269. https://doi.org/10.3390/nu9111269







