Inhibitory Effect of Arachis hypogaea (Peanut) and Its Phenolics against Methylglyoxal-Derived Advanced Glycation End Product Toxicity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Processing of Peanut Samples
2.3. Extraction
2.4. UPLC-MS/MS Analysis of Individual Phenolic Compounds
2.5. Preparation of Glycated Bovine Serum Albumin
2.6. Cell Culture
2.7. Cell Viability
2.8. Western Blotting Analysis
2.9. Intracellular ROS Detection
2.10. Inhibitory Effects of Peanut Extracts and Constituent Phenolic Compounds on AGE Formation
2.11. AGE Breaking Activity of Peanut Extracts and Constituent Phenolic Compounds
2.12. Statistical Analyses
3. Results
3.1. Method Development for UHPLC-MS/MS Analysis
3.2. Determination of Polyphenols in Peanut Extracts
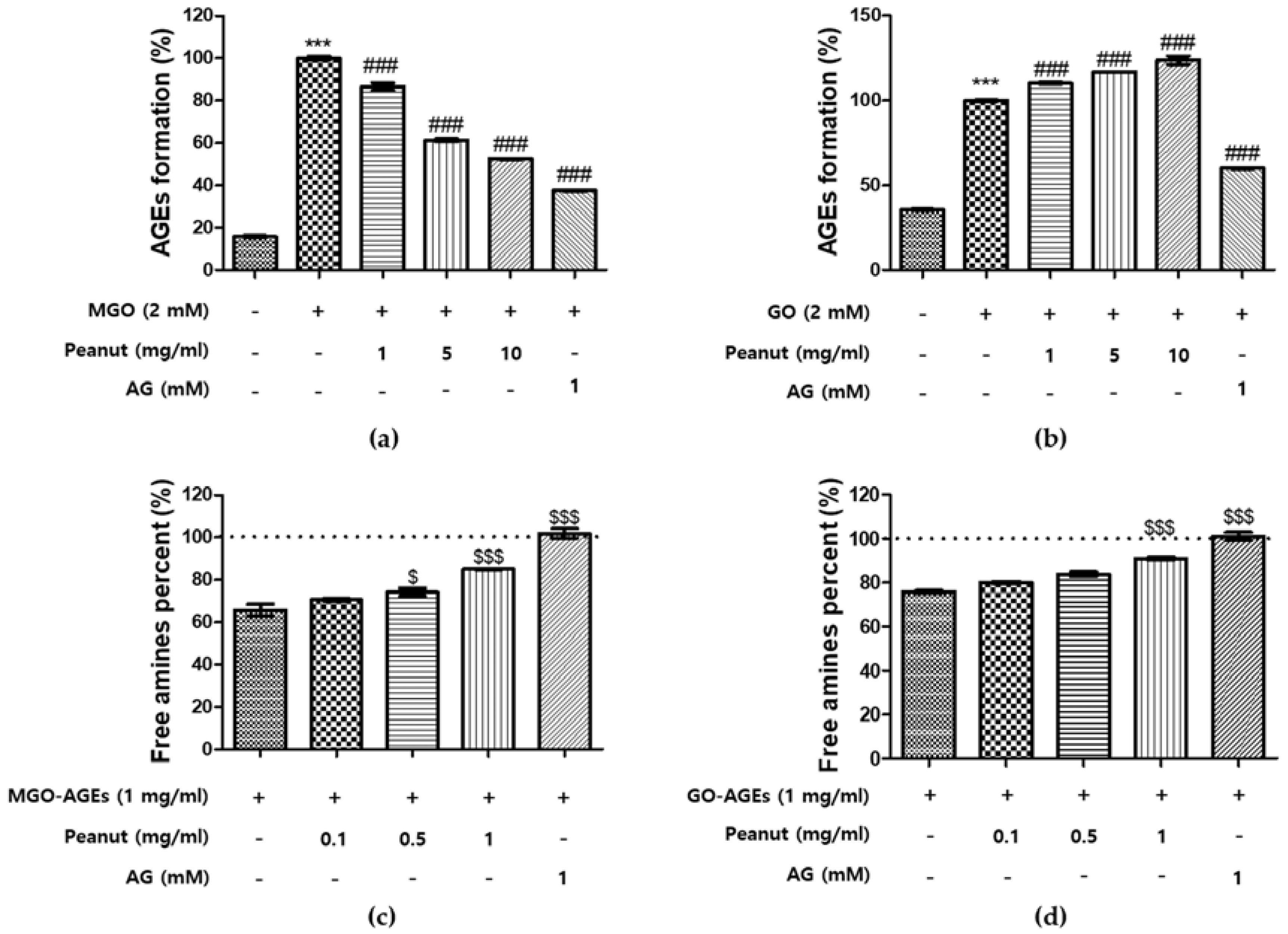
3.3. Effect of Peanut Extracts on the Formation of AGEs
3.4. Breaking Ability of Peanut Extracts on AGEs
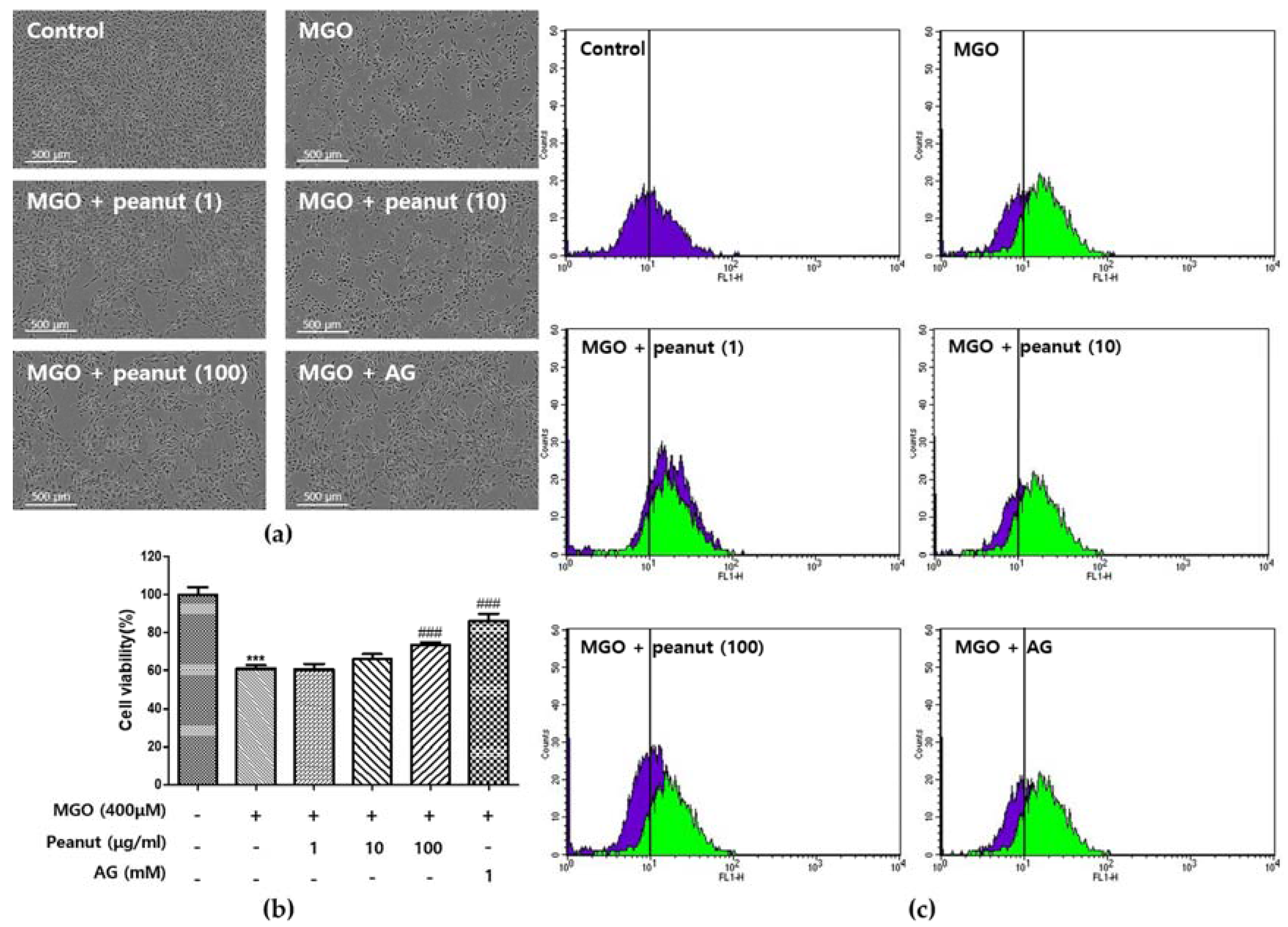
3.5. Effect of Peanut Extracts on MGO-Induced Cytotoxicity
3.6. Effect of Peanut Extracts on Cellular ROS Generation
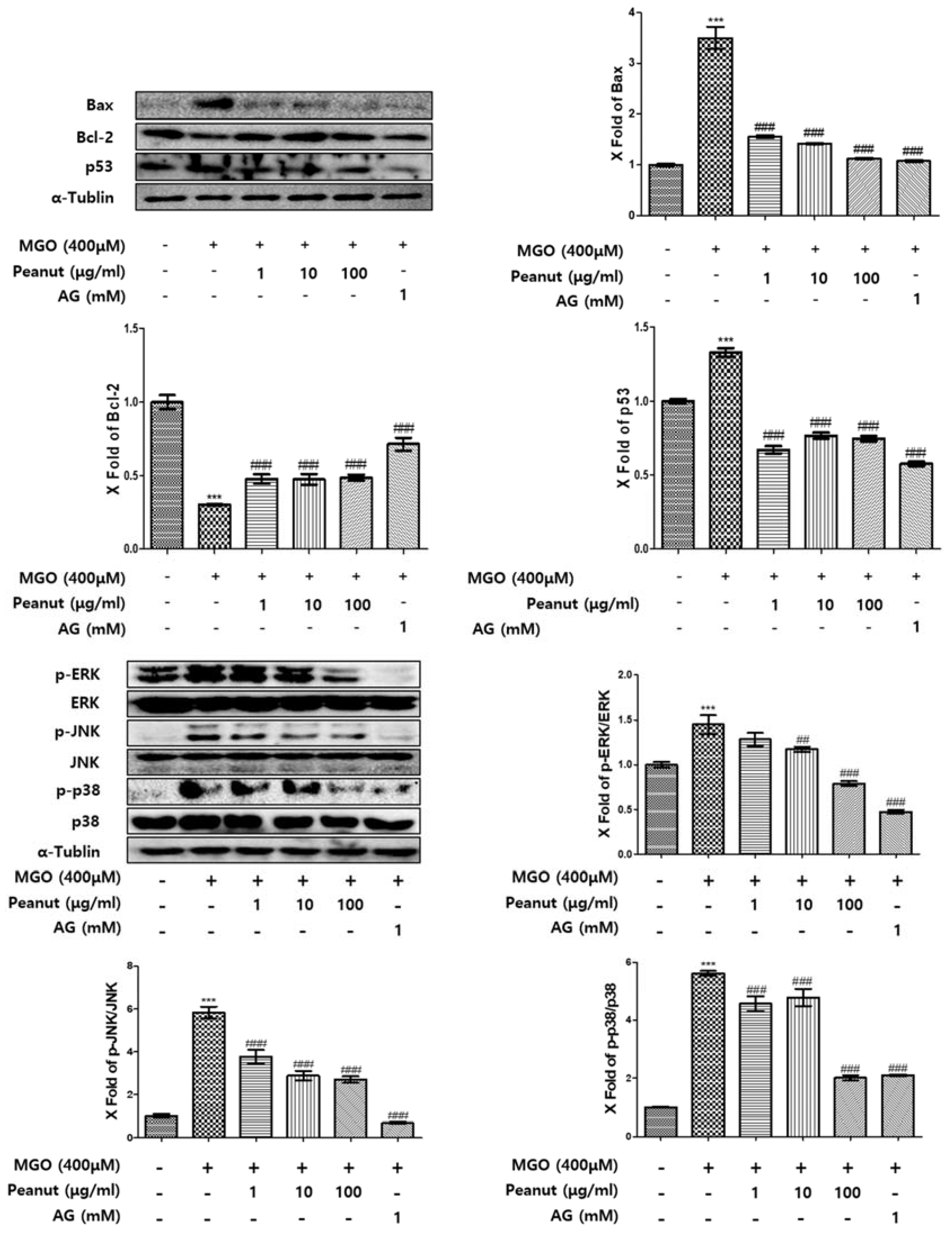
3.7. Effect of Peanut Extracts on MGO-Induced Apoptosis
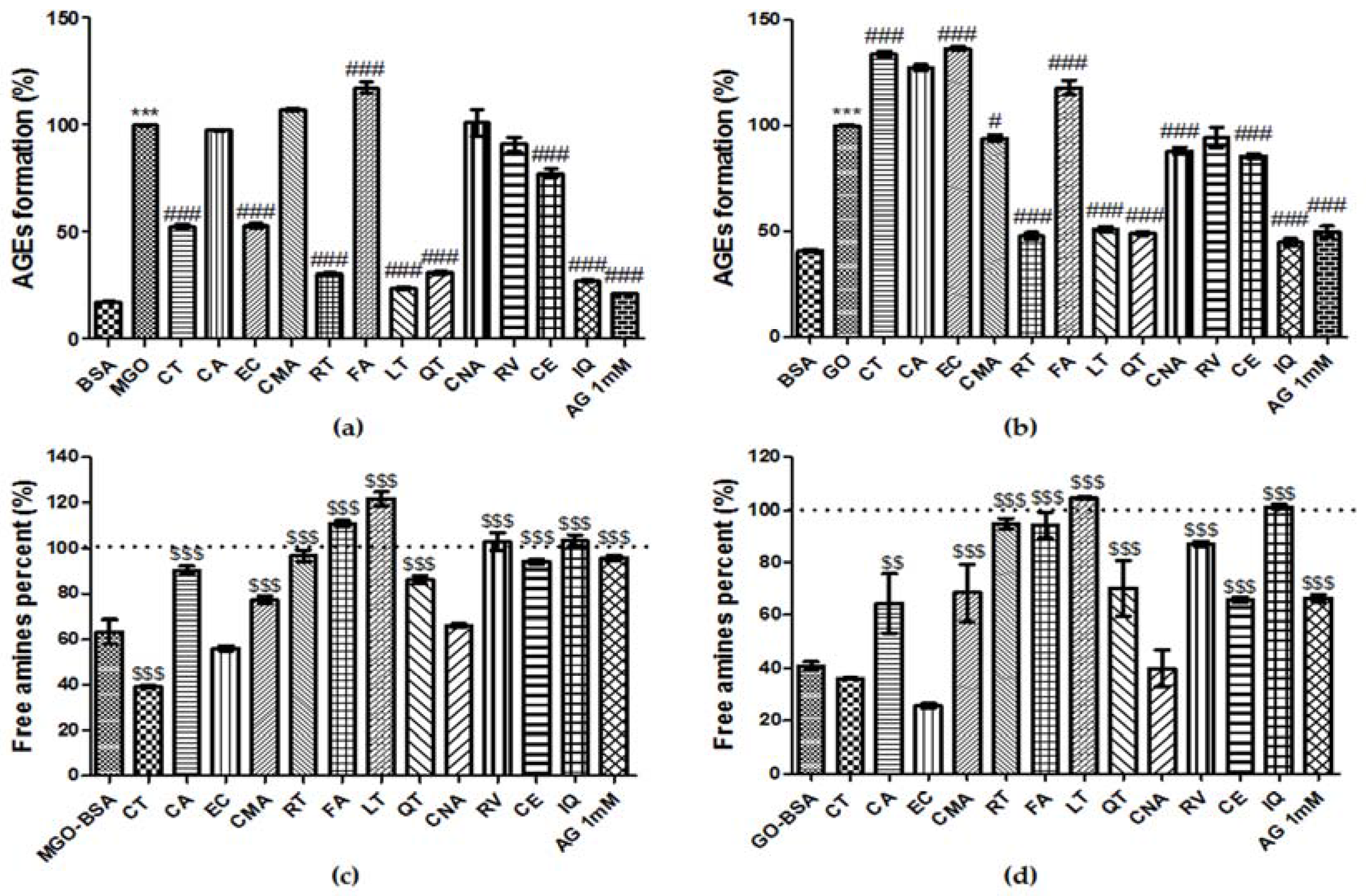
3.8. Effect of Phenolic Compounds on the Formation of AGEs
3.9. Breaking Ability of Phenolic Compounds on AGEs
3.10. Effect of Phenolic Compounds on MGO-Induced Cytotoxicity
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Singh, V.P.; Bali, A.; Singh, N.; Jaggi, A.S. Advanced glycation end products and diabetic complications. Korean J. Physiol. Pharmacol. 2014, 18, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, S.R.; Baynes, J.W. Role of the maillard reaction in diabetes mellitus and diseases of aging. Drugs Aging 1996, 9, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Dyer, D.; Blackledge, J.; Katz, B.; Hull, C.; Adkisson, H.; Thorpe, S.; Lyons, T.; Baynes, J. The maillard reaction in vivo. Z. Für Ernährungswissenschaft 1991, 30, 29–45. [Google Scholar] [CrossRef]
- Beisswenger, P.J.; Moore, L.L.; Brinck-Johnsen, T.; Curphey, T.J. Increased collagen-linked pentosidine levels and advanced glycosylation end products in early diabetic nephropathy. J. Clin. Investig. 1993, 92, 212. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.E.; Yang, H.; Jeong, S.I.; Jin, Y.-H.; Park, C.-S.; Park, Y.S. Methylglyoxal-mediated alteration of gene expression in human endothelial cells. BioChip J. 2011, 5, 220–228. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Saibabu, V.; Fatima, Z.; Khan, L.A.; Hameed, S. Therapeutic potential of dietary phenolic acids. Adv. Pharmacol. Sci. 2015, 2015, 823539. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, H.; Frost, L.; Yuan, T.; Dain, J.A.; Seeram, N.P. Pomegranate phenolics inhibit formation of advanced glycation endproducts by scavenging reactive carbonyl species. Food Funct. 2014, 5, 2996–3004. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Zheng, Z.; Cheng, K.-W.; Shan, F.; Ren, G.-X.; Chen, F.; Wang, M. Inhibitory effect of mung bean extract and its constituents vitexin and isovitexin on the formation of advanced glycation endproducts. Food Chem. 2008, 106, 475–481. [Google Scholar] [CrossRef]
- Lunceford, N.; Gugliucci, A. Ilex paraguariensis extracts inhibit age formation more efficiently than green tea. Fitoterapia 2005, 76, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Hu, F.B.; Ros, E.; Sabaté, J. The role of tree nuts and peanuts in the prevention of coronary heart disease: Multiple potential mechanisms. J. Nutr. 2008, 138, 1746S–1751S. [Google Scholar] [PubMed]
- Ros, E. Dietary cis-monounsaturated fatty acids and metabolic control in type 2 diabetes. Am. J. Clin. Nutr. 2003, 78, 617S–625S. [Google Scholar] [PubMed]
- Akhtar, S.; Khalid, N.; Ahmed, I.; Shahzad, A. Suleria HAR: Physicochemical characteristics, functional properties, and nutritional benefits of peanut oil: A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1562–1575. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.M.; Agostini-Costa, T.N.D.S.; Gimenes, M.A.; Silveira, D. Chemical composition and biological activities of arachis species. J. Agric. Food Chem. 2011, 59, 4321–4330. [Google Scholar] [CrossRef] [PubMed]
- Khangholi, S.; Majid, F.A.A.; Berwary, N.J.A.; Ahmad, F.; Aziz, R.B.A. The mechanisms of inhibition of advanced glycation end products formation through polyphenols in hyperglycemic condition. Planta Med. 2016, 82, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Gültekin-Özgüven, M.; Davarcı, F.; Paslı, A.A.; Demir, N.; Özçelik, B. Determination of phenolic compounds by ultra high liquid chromatography-tandem mass spectrometry: Applications in nuts. LWT Food Sci. Technol. 2015, 64, 42–49. [Google Scholar] [CrossRef]
- Flores, M.I.A.; Romero-González, R.; Frenich, A.G.; Vidal, J.L.M. Analysis of phenolic compounds in olive oil by solid-phase extraction and ultra high performance liquid chromatography–tandem mass spectrometry. Food Chem. 2012, 134, 2465–2472. [Google Scholar] [CrossRef] [PubMed]
- Andersen, O.M.; Markham, K.R. Flavonoids: Chemistry, Biochemistry and Applications; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Kiho, T.; Kato, M.; Usui, S.; Hirano, K. Effect of buformin and metformin on formation of advanced glycation end products by methylglyoxal. Clin. Chim. Acta 2005, 358, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Habeeb, A.S.A. Determination of free amino groups in proteins by trinitrobenzenesulfonic acid. Anal. Biochem. 1966, 14, 328–336. [Google Scholar] [CrossRef]
- Motilva, M.-J.; Serra, A.; Macià, A. Analysis of food polyphenols by ultra high-performance liquid chromatography coupled to mass spectrometry: An overview. J. Chromatogr. A 2013, 1292, 66–82. [Google Scholar] [CrossRef] [PubMed]
- ICH Topic Q2 (R1) Validation of Analytical Procedures: Text and Methodology. International Conference on Harmonization, 1994 (November 1996), 17.
- SANTE, E. Guidance document on analytical quality control and method validation procedures for pesticides residues analysis in food and feed European Commission. 2015.
- Guideline, I.H.T. Validation of analytical procedures: Text and methodology. Q2 (R1) 2005, 1.
- Figarola, J.L.; Singhal, J.; Rahbar, S.; Awasthi, S.; Singhal, S.S. Lr-90 prevents methylglyoxal-induced oxidative stress and apoptosis in human endothelial cells. Apoptosis 2014, 19, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.M.; Huang, D.Y.; Huang, Y.P.; Hsu, S.H.; Kang, L.Y.; Shen, C.M.; Lin, W.W. Methylglyoxal induces cell death through endoplasmic reticulum stress-associated ros production and mitochondrial dysfunction. J. Cell. Mol. Med. 2016, 20, 1749–1760. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, Y.; Yang, X.; Lu, P.; Yan, X.; Xiao, F.; Zhou, H.; Wen, C.; Shi, M.; Lu, J. Effects of methylglyoxal and glyoxalase i inhibition on breast cancer cells proliferation, invasion, and apoptosis through modulation of mapks, mmp9, and bcl-2. Cancer Boil. Ther. 2016, 17, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Chukwumah, Y.C.; Walker, L.T.; Verghese, M.; Bokanga, M.; Ogutu, S.; Alphonse, K. Comparison of extraction methods for the quantification of selected phytochemicals in peanuts (Arachis hypogaea). J. Agric. Food Chem. 2007, 55, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.P.N.; Thi, N.V.; Van, P.T.; Diem, P.Q.N.; Thuy, D.N.T.; That, Q.T.; Phi, P.N.K. Phytochemical constituents and determination of resveratrol from the roots of Arachis hypogea L. Am. J. Plant Sci. 2013, 2013. [Google Scholar] [CrossRef]
- Kornsteiner, M.; Wagner, K.-H.; Elmadfa, I. Tocopherols and total phenolics in 10 different nut types. Food Chem. 2006, 98, 381–387. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Chukwumah, Y.; Walker, L.T.; Verghese, M. Peanut skin color: A biomarker for total polyphenolic content and antioxidative capacities of peanut cultivars. Int. J. Mol. Sci. 2009, 10, 4941–4952. [Google Scholar] [CrossRef] [PubMed]
- Downey, M.O.; Dokoozlian, N.K.; Krstic, M.P. Cultural practice and environmental impacts on the flavonoid composition of grapes and wine: A review of recent research. Am. J. Enol. Vitic. 2006, 57, 257–268. [Google Scholar]
- Malheiro, R.; Sousa, A.; Casal, S.; Bento, A.; Pereira, J.A. Cultivar effect on the phenolic composition and antioxidant potential of stoned table olives. Food Chem. Toxicol. 2011, 49, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Kang, O.-J.; Gweon, O.-C. Comparison of phenolic acids and flavonoids in black garlic at different thermal processing steps. J. Funct. Foods 2013, 5, 80–86. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chang, S.K. Total phenolics, phenolic acids, isoflavones, and anthocyanins and antioxidant properties of yellow and black soybeans as affected by thermal processing. J. Agric. Food Chem. 2008, 56, 7165–7175. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekara, N.; Shahidi, F. Effect of roasting on phenolic content and antioxidant activities of whole cashew nuts, kernels, and testa. J. Agric. Food Chem. 2011, 59, 5006–5014. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ahmedna, M.; Goktepe, I. Effects of processing methods and extraction solvents on concentration and antioxidant activity of peanut skin phenolics. Food Chem. 2005, 90, 199–206. [Google Scholar] [CrossRef]
- Xu, B.; Chang, S.K. Total phenolic, phenolic acid, anthocyanin, flavan-3-ol, and flavonol profiles and antioxidant properties of pinto and black beans (Phaseolus vulgaris L.) as affected by thermal processing. J. Agric. Food Chem. 2009, 57, 4754–4764. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Dailey, A.; Vuong, Q.V. Effect of extraction solvents on recovery of bioactive compounds and antioxidant properties from macadamia (Macadamia tetraphylla) skin waste. Cog. Food Agric. 2015, 1, 1115646. [Google Scholar] [CrossRef]
- Janić, M.; Lunder, M.; Šabovič, M. Arterial stiffness and cardiovascular therapy. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Manigrasso, M.B.; Juranek, J.; Ramasamy, R.; Schmidt, A.M. Unlocking the biology of rage in diabetic microvascular complications. Trends Endocrinol. Metab. 2014, 25, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Wang, L.; Zhou, Q.; Yan, S.; Li, Z.; Sheng, J.; Zhang, W. (+)-catechin ameliorates diabetic nephropathy by trapping methylglyoxal in type 2 diabetic mice. Mol. Nutr. Food Res. 2014, 58, 2249–2260. [Google Scholar] [CrossRef] [PubMed]
- Al Maruf, A.; Lip, H.; Wong, H.; O’Brien, P.J. Protective effects of ferulic acid and related polyphenols against glyoxal-or methylglyoxal-induced cytotoxicity and oxidative stress in isolated rat hepatocytes. Chem.-Biol. Int. 2015, 234, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Kancheva, V.D.; Boranova, P.V.; Nechev, J.T.; Manolov, I.I. Structure–activity relationships of new 4-hydroxy bis-coumarins as radical scavengers and chain-breaking antioxidants. Biochimie 2010, 92, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, H.; Nakamura, S.; Morioka, M.; Tanaka, J.; Matsuda, H.; Yoshikawa, M. Effect of cinnamoyl and flavonol glucosides derived from cherry blossom flowers on the production of advanced glycation end products (ages) and age-induced fibroblast apoptosis. Phytother. Res. 2011, 25, 1328–1335. [Google Scholar] [CrossRef] [PubMed]
- Kyselova, Z.; Stefek, M.; Bauer, V. Pharmacological prevention of diabetic cataract. J. Diabetes Complicat. 2004, 18, 129–140. [Google Scholar] [CrossRef]
- Rasoanaivo, P.; Wright, C.W.; Willcox, M.L.; Gilbert, B. Whole plant extracts versus single compounds for the treatment of malaria: Synergy and positive interactions. Malar. J. 2011, 10, S4. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, J.; Gupta, N.; Agrawal, M.; Bhaskar, A.B.; Rao, P.L. Modulation of ros/mapk signaling pathways by okadaic acid leads to cell death via, mitochondrial mediated caspase-dependent mechanism. Apoptosis 2011, 16, 145–161. [Google Scholar] [CrossRef] [PubMed]
- Do, M.H.; Kim, S.N.; Seo, S.-Y.; Yeo, E.-J.; Kim, S.Y. Δ-tocopherol prevents methylglyoxal-induced apoptosis by reducing ros generation and inhibiting apoptotic signaling cascades in human umbilical vein endothelial cells. Food Funct. 2015, 6, 1568–1577. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.K.; Winocour, P.; Farrington, K. Oxidative stress in early diabetic nephropathy: Fueling the fire. Nat. Rev. Endocrinol. 2011, 7, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, A.; Fairbrother, W.J.; Leverson, J.D.; Souers, A.J. From basic apoptosis discoveries to advanced selective bcl-2 family inhibitors. Nat. Rev. Drug Discov. 2017, 16, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Emekli-Alturfan, E.; Kasikci, E.; Yarat, A. Peanut (Arachis hypogaea) consumption improves glutathione and hdl-cholesterol levels in experimental diabetes. Phytother. Res. 2008, 22, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Hu, F.B.; Tapsell, L.C.; Josse, A.R.; Kendall, C.W. Possible benefit of nuts in type 2 diabetes. J. Nutr. 2008, 138, 1752s–1756s. [Google Scholar] [PubMed]
- Jiang, R.; Jacobs, D.R., Jr.; Mayer-Davis, E.; Szklo, M.; Herrington, D.; Jenny, N.S.; Kronmal, R.; Barr, R.G. Nut and seed consumption and inflammatory markers in the multi-ethnic study of atherosclerosis. Am. J. Epidemiol. 2006, 163, 222–231. [Google Scholar] [CrossRef] [PubMed]

| Compound | Retention Time (min) | Cone Voltage (V) | Quantification Transition (m/z) * | Confirmation Transition (m/z) * | Ion Ratio (%) |
|---|---|---|---|---|---|
| (+)Catechin (CT) | 4.35 | 35 | 289 > 245 (15) | 289 > 109 (25) | 75.3 |
| Caffeic acid (CA) | 4.97 | 25 | 179 > 135 (15) | ||
| (−)Epicatechin (EC) | 5.25 | 35 | 289 > 109 (20) | 289 > 245 (15) | 63.4 |
| p-Coumaric acid (CMA) | 6.31 | 25 | 163 > 119 (15) | 163 > 93 (25) | 5 |
| Rutin (RT) | 6.68 | 50 | 609 > 300 (35) | 609 > 271 (55) | 43 |
| trans-Ferulic acid (FA) | 6.89 | 25 | 193 > 134 (15) | 193 > 149 (10) | 50.5 |
| Isoquercitrin (IQ) | 6.99 | 45 | 463 > 300 (25) | 463 > 271 (40) | 51.4 |
| Resveratrol (RV) | 8.95 | 40 | 227 > 143 (25) | 227 > 185 (18) | 76.5 |
| Luteolin (LT) | 9.89 | 45 | 285 > 133 (35) | 285 > 107 (35) | 13 |
| Quercetin (QT) | 9.98 | 35 | 301 > 151 (22) | 301 > 179 (20) | 37.7 |
| trans-Cinnamic acid (CNA) | 10.54 | 30 | 147 > 104 (10) | ||
| Chrysoeriol (CE) | 11.53 | 40 | 299 > 284 (20) | 29 > 256 (30) | 23.1 |
| Compound | Regression | R2 | Linear Range (mg/L) | LOQ (mg/L) | LOD (mg/L) | Recovery (%) | STD Solution Concentration (1 mg/L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ethanol | Ethanol 70% | Methanol 80% | Acetone 80% | Intra-Day (n = 6) | Inter-Day (Between 3 Days (n = 6)) | |||||||||||
| Mean | RSD (%) | Day 1 | Day 2 | Day 3 | Mean | RSD (%) | ||||||||||
| CNA | 15189.5X + 588.1 | 0.9922 | 0.0625–2.0 | 0.011 | 0.004 | 109.8 | 103.6 | 107.2 | 102.5 | 1.05 | 2.37 | 1.05 | 1.05 | 1.01 | 1.04 | 2.23 |
| EC | 10442.3X - 23.9 | 0.9905 | 0.0625–2.0 | 0.097 | 0.032 | 99.2 | 97.5 | 104.1 | 99.6 | 1.03 | 2.38 | 1.03 | 1.01 | 1.00 | 1.01 | 1.53 |
| CT | 8964.13X + 320.6 | 0.9988 | 0.0625–4.0 | 0.027 | 0.009 | 100.6 | 107.4 | 106.9 | 109.0 | 1.02 | 3.83 | 1.02 | 1.04 | 1.00 | 1.02 | 1.73 |
| CA | 81988.3X + 2639.0 | 0.9977 | 0.0625–4.0 | 0.005 | 0.002 | 112.5 | 102.6 | 100.5 | 101.9 | 1.03 | 2.84 | 1.03 | 1.05 | 1.04 | 1.04 | 0.98 |
| CMA | 54760.3X + 2819.4 | 0.9924 | 0.0625–2.0 | 0.008 | 0.003 | 103.5 | 96.5 | 112.6 | 109.5 | 1.02 | 1.48 | 1.02 | 1.06 | 0.99 | 1.02 | 3.29 |
| RT | 31479.4X + 318.3 | 0.9933 | 0.0625–2.0 | 0.004 | 0.001 | 101.6 | 102.5 | 103.5 | 99.8 | 0.91 | 2.48 | 0.91 | 0.87 | 0.84 | 0.87 | 3.64 |
| IQ | 57551.8X + 668.4 | 0.9999 | 0.0625–4.0 | 0.003 | 0.001 | 108.3 | 114.9 | 102.3 | 95.9 | 1.06 | 3.09 | 1.06 | 1.04 | 0.96 | 1.02 | 5.07 |
| FA | 37480.2X + 2359.4 | 0.9917 | 0.0625–2.0 | 0.004 | 0.001 | 90.2 | 92.6 | 88.9 | 83.9 | 1.01 | 2.42 | 1.01 | 1.06 | 1.03 | 1.03 | 2.76 |
| RV | 21170.8X + 1208.5 | 0.9936 | 0.0625–4.0 | 0.005 | 0.002 | 111.0 | 105.2 | 93.5 | 95.6 | 1.04 | 2.62 | 1.04 | 1.02 | 1.01 | 1.03 | 1.63 |
| LT | 112624X + 4641.9 | 0.9959 | 0.0625–2.0 | 0.015 | 0.005 | 106.2 | 102.4 | 103.1 | 98.8 | 1.10 | 2.21 | 1.10 | 1.09 | 1.04 | 1.07 | 3.12 |
| QT | 53101X + 1908.0 | 0.9945 | 0.0625–2.0 | 0.101 | 0.034 | 102.2 | 95.6 | 95.4 | 102.1 | 1.02 | 2.61 | 1.02 | 1.06 | 1.06 | 1.04 | 2.27 |
| CE | 145257X + 9713.4 | 0.9887 | 0.0625–2.0 | 0.005 | 0.002 | 116.5 | 109.2 | 102.4 | 106.6 | 1.08 | 2.78 | 1.08 | 1.06 | 0.89 | 1.01 | 10.47 |
| Compound | Raw | Roasted | Steamed | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 100% Ethanol | 70% Ethanol | 80% Methanol | 80% Acetone | 100% Ethanol | 70% Ethanol | 80% Methanol | 80% Acetone | 100% Ethanol | 70% Ethanol | 80% Methanol | 80% Acetone | |
| CNA | N.D. | 0.87 ± 0.03 | 3.64 ± 0.02 | 0.13 ± 0.02 | N.D. | 2.39 ± 0.08 a | 3.87 ± 0.06 a | 0.77 ± 0.04 a | N.D. | 1.32 ± 0.05 a | 3.71 ± 0.02 | 0.28 ± 0.03 |
| EC | 22.39 ± 0.39 | 31.22 ± 0.44 | 32.07 ± 0.83 | 27.71 ± 0.30 | 20.38 ± 0.08 | 24.34 ± 0.69 b | 25.49 ± 0.75 b | 22.62 ± 0.10 b | 16.98 ± 0.57 b | 21.07 ± 0.42 b | 20.45 ± 0.78 b | 22.27 ± 1.57 b |
| CT | 40.83 ± 0.21 | 41.03 ± 0.45 | 40.35 ± 0.26 | 40.67 ± 0.19 | 31.96 ± 0.25 | 32.58 ± 0.16 | 32.43 ± 0.33 | 32.99 ± 0.28 | 22.04 ± 0.41 | 28.07 ± 0.32 | 30.05 ± 0.25 | 30.92 ± 0.50 |
| CA | N.D. | 0.46 ± 0.01 | 0.55 ± 0.01 | 0.47 ± 0.01 | 0.16 ± 0.01 a | 1.16 ± 0.01 a | 1.22 ± 0.02 a | 1.09 ± 0.01 a | 0.09 ± 0.01 a | 0.87 ± 0.01 a | 1.04 ± 0.01 a | 1.01 ± 0.00 a |
| CMA | 0.64 ± 0.02 | 10.43 ± 0.16 | 18.92 ± 0.19 | 8.14 ± 0.10 | 7.15 ± 0.07 a | 50.86 ± 0.38 a | 57.85 ± 0.61 a | 46.50 ± 0.62 a | 3.38 ± 0.04 a | 20.62 ± 0.62 a | 29.38 ± 0.59 a | 15.84 ± 0.29 a |
| RT | 3.85 ± 0.01 | 3.61 ± 0.05 | 3.72 ± 0.03 | 3.84 ± 0.03 | 3.41 ± 0.06 b | 5.36 ± 0.07 a | 5.38 ± 0.05 a | 4.99 ± 0.01 a | 2.34 ± 0.24 b | 4.15 ± 0.02 a | 4.41 ± 0.04 a | 4.24 ± 0.05 a |
| IQ | 0.30 ± 0.00 | 0.30 ± 0.00 | 0.28 ± 0.00 | 0.31 ± 0.00 | 0.37 ± 0.00 a | 0.33 ± 0.00 a | 0.36 ± 0.01 a | 0.33 ± 0.00 a | 0.02 ± 0.00 a | 0.21 ± 0.01 a | 0.26 ± 0.00 | 0.24 ± 0.00 a |
| FA | N.D. | 0.24 ± 0.02 | 0.32 ± 0.02 | 0.22 ± 0.01 | N.D. | 1.29 ± 0.03 a | 1.37 ± 0.01 a | 1.13 ± 0.02 a | 0.12 ± 0.01 a | 1.14 ± 0.02 a | 1.16 ± 0.03 a | 0.89 ± 0.01 a |
| RV | N.D. | 0.28 ± 0.01 | 0.33 ± 0.01 | 0.27 ± 0.01 | N.D. | 0.64 ± 0.02 a | 0.79 ± 0.03 a | 0.40 ± 0.01 a | 0.05 ± 0.01 b | 0.21 ± 0.01 b | 0.24 ± 0.01 b | 0.32 ± 0.01 b |
| LT | N.D. | 0.71 ± 0.03 | 0.68 ± 0.01 | 0.81 ± 0.02 | N.D. | 0.63 ± 0.02 b | 0.48 ± 0.01 b | 0.64 ± 0.02b | N.D. | 0.41 ± 0.01 b | 0.43 ± 0.01 b | 0.61 ± 0.01 b |
| QT | 1.20 ± 0.01 | 2.53 ± 0.04 | 2.52 ± 0.05 | 3.49 ± 0.00 | 4.75 ± 0.26 a | 4.99 ± 0.04 a | 5.06 ± 0.05 a | 6.47 ± 0.03 a | N.D. | 1.92 ± 0.03 b | 1.90 ± 0.04 b | 2.63 ± 0.03 b |
| CE | 0.72 ± 0.03 | 0.85 ± 0.02 | 0.82 ± 0.02 | 0.91 ± 0.02 | 0.31 ± 0.01 b | 0.69 ± 0.03 b | 0.82 ± 0.00 | 0.72 ± 0.02 b | N.D. | 0.33 ± 0.01 b | 0.44 ± 0.01 b | 0.63 ± 0.02 b |
| Udo Island | Yecheon | Kimcheon | Hongcheon | ||
|---|---|---|---|---|---|
| Raw | Ethanol | 98.52 ± 1.82 | 62.87 ± 1.16 | 64.04 ± 0.66 | 63.96 ± 1.54 |
| 70% Ethanol | 107.43 ± 3.24 | 87.13 ± 1.85 | 80.70 ± 0.24 | 103.46 ± 2.25 | |
| 80% Methanol | 105.14± 0.54 | 97.29 ± 0.83 b | 87.36 ± 0.40 b | 114.64 ± 1.28 b | |
| 80% Acetone | 97.44 ± 1.51 | 80.54 ± 1.84 | 78.24 ± 0.22 | 91.43 ± 0.41 | |
| Roasted | Ethanol | 90.29 ± 0.91 | 63.64 ± 1.71 | 68.63 ± 0.61 | 51.37 ± 1.19 |
| 70% Ethanol | 142.26 ± 2.71 a | 108.10 ± 1.61 a | 121.53 ± 0.55 a | 118.30 ± 2.09 a | |
| 80% Methanol | 147.49 ± 2.19 a,b | 116.32 ± 1.26 a,b | 128.67 ± 4.38 a,b | 129.11 ± 0.99 a,b | |
| 80% Acetone | 126.84 ± 1.69 a | 102.97 ± 1.01 a | 114.10 ± 2.12 a | 108.66 ± 0.65 a | |
| Steamed | Ethanol | 64.94 ± 0.90 | 36.90 ± 1.30 | 50.54 ±1.87 | 21.96 ± 0.25 |
| 70% Ethanol | 92.49 ± 2.42 | 65.35 ± 1.84 | 78.80 ± 1.12 | 82.86 ± 1.56 | |
| 80% Methanol | 104.21 ± 0.81 b | 86.96 ± 0.74 | 86.39 ± 1.68 b | 94.33 ± 1.59 b | |
| 80% Acetone | 87.03 ± 0.83 | 82.40 ± 1.56 | 73.28 ± 3.64 | 76.36 ± 2.87 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.H.; Do, M.H.; Lee, J.H.; Jeong, M.; Lim, O.K.; Kim, S.Y. Inhibitory Effect of Arachis hypogaea (Peanut) and Its Phenolics against Methylglyoxal-Derived Advanced Glycation End Product Toxicity. Nutrients 2017, 9, 1214. https://doi.org/10.3390/nu9111214
Park SH, Do MH, Lee JH, Jeong M, Lim OK, Kim SY. Inhibitory Effect of Arachis hypogaea (Peanut) and Its Phenolics against Methylglyoxal-Derived Advanced Glycation End Product Toxicity. Nutrients. 2017; 9(11):1214. https://doi.org/10.3390/nu9111214
Chicago/Turabian StylePark, Sin Hee, Moon Ho Do, Jae Hyuk Lee, Minsun Jeong, Oh Kyung Lim, and Sun Yeou Kim. 2017. "Inhibitory Effect of Arachis hypogaea (Peanut) and Its Phenolics against Methylglyoxal-Derived Advanced Glycation End Product Toxicity" Nutrients 9, no. 11: 1214. https://doi.org/10.3390/nu9111214
APA StylePark, S. H., Do, M. H., Lee, J. H., Jeong, M., Lim, O. K., & Kim, S. Y. (2017). Inhibitory Effect of Arachis hypogaea (Peanut) and Its Phenolics against Methylglyoxal-Derived Advanced Glycation End Product Toxicity. Nutrients, 9(11), 1214. https://doi.org/10.3390/nu9111214





