Citrulline Malate Does Not Improve Muscle Recovery after Resistance Exercise in Untrained Young Adult Men
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
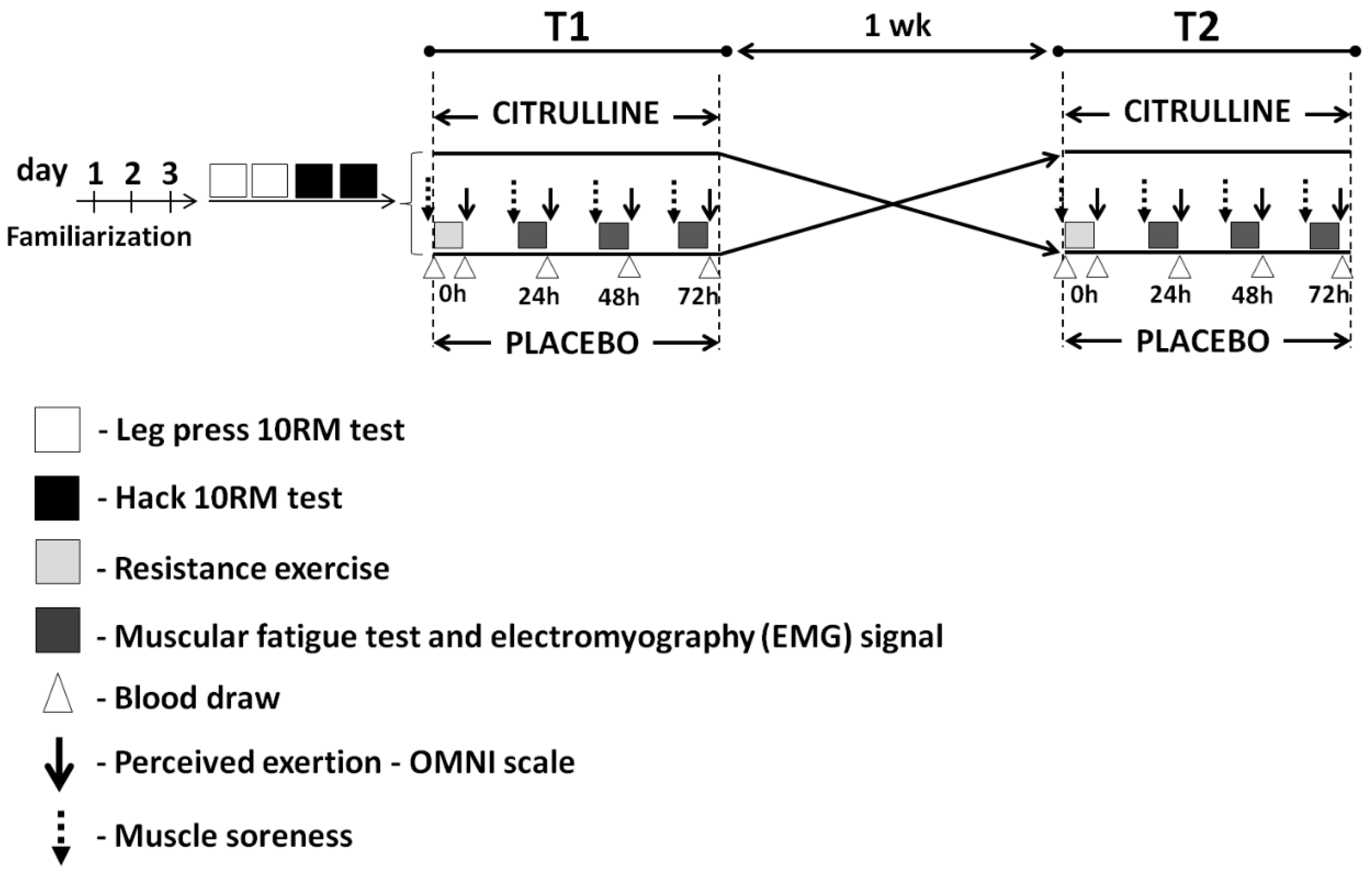
2.2. Experimental Design
2.3. Familiarization Protocol
2.4. Determination of 10 Repetition Maximum Load
2.5. Resistance Exercise
2.6. Supplementation Protocol
2.7. Dietary Intake
2.8. Perceived Exertion
2.9. Delayed-Onset Muscle Soreness
2.10. Muscular Endurance Tests and Electromyographic (EMG) Signal Recordings
2.11. Blood Collection and Analysis
2.12. Statistical Analyses
3. Results
3.1. Dietary Intake
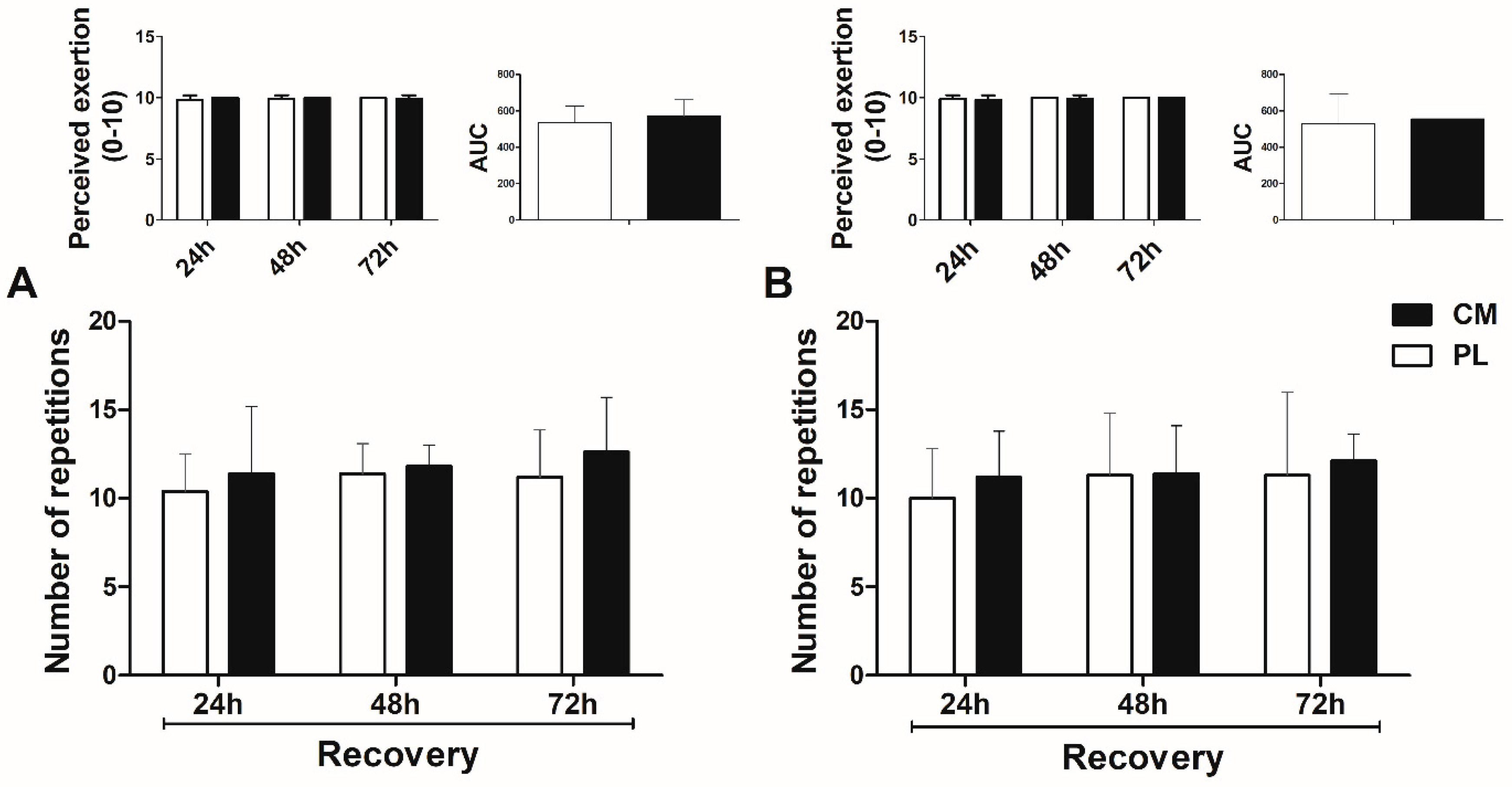
3.2. Total Repetitions and Perceived Exertion in the Fatigue Tests during Recovery
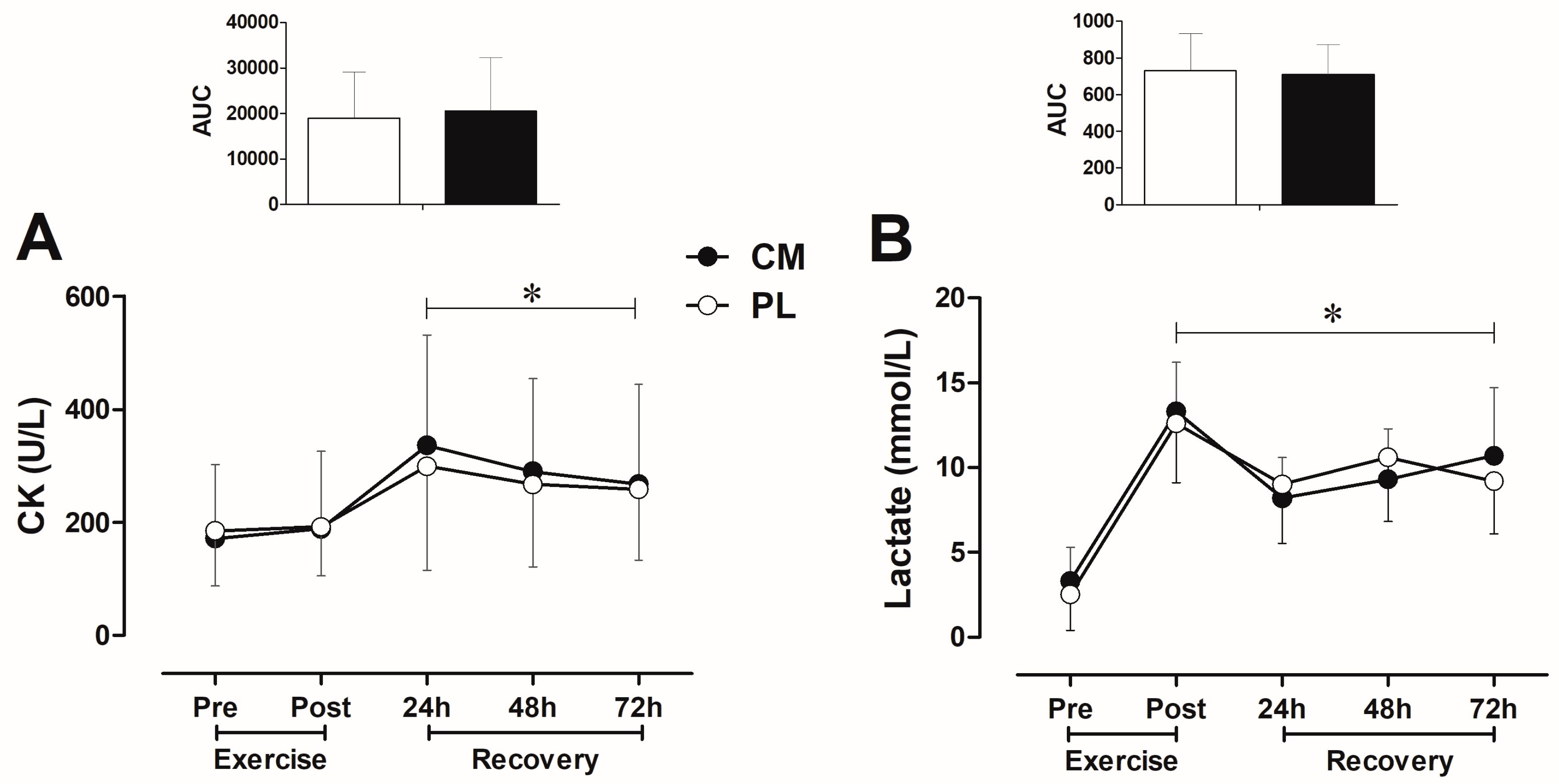
3.3. CK and Lactate Blood Levels after Fatigue Test during Recovery
3.4. Muscle Soreness during Recovery
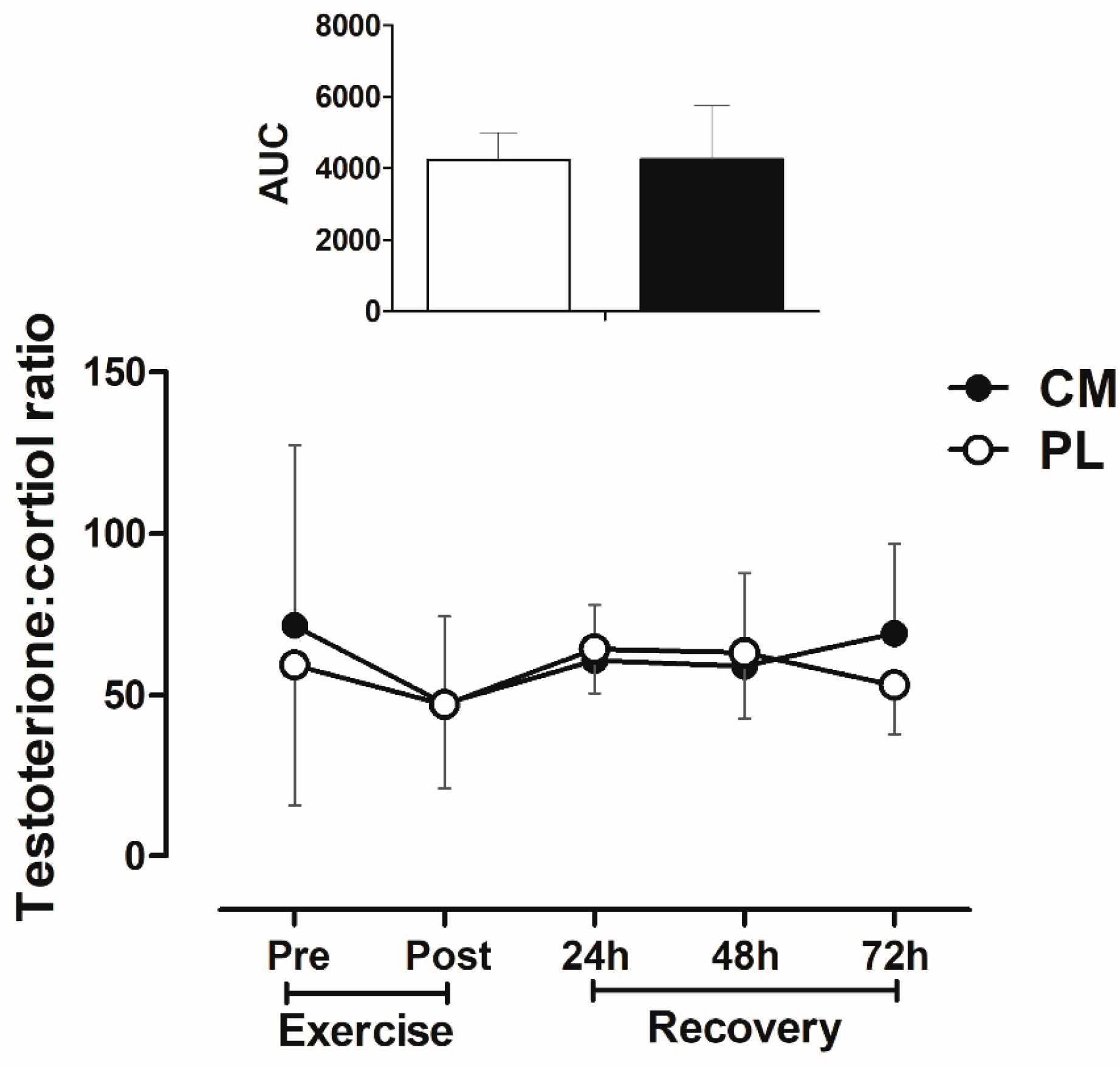
3.5. Testosterone: Cortisol Ratio after Fatigue Test during Recovery
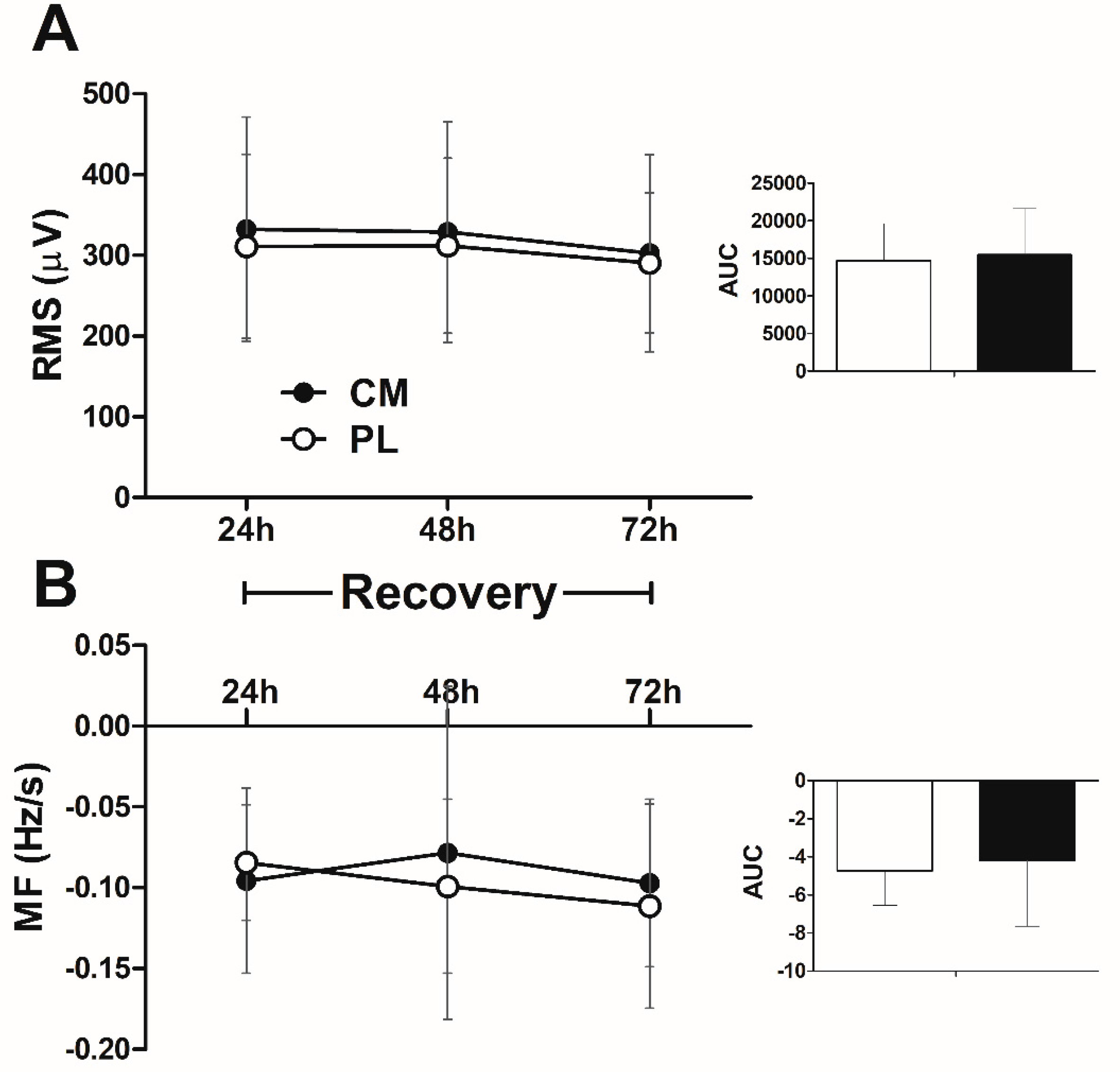
3.6. EMG Signal in the Fatigue Test during Recovery
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alvares, T.S.; Meirelles, C.I.T.; Bhambhani, Y.N.; Paschoalin, V.M.; Gomes, P.S. l-Arginine as a potential ergogenic aid in healthy subjects. Sports Med. 2011, 41, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Bendahan, D.; Mattei, J.P.; Ghattas, B.; Confort-Gouny, S.; Le Guern, M.E.; Cozzone, P.J. Citrulline/malate promotes aerobic energy production in human exercising muscle. Br. J. Sports Med. 2002, 36, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Bescos, R.; Sureda, A.; Tur, J.A.; Pons, A. The effect of nitric-oxide-related supplements on human performance. Sports Med. 2012, 42, 99–117. [Google Scholar] [CrossRef] [PubMed]
- Vanuxem, D.; Duflot, J.C.; Prevot, H.; Bernasconi, P.; Blehaut, H.; Fornaris, E.; Vanuxem, P. Influence of an anti- asthenia agent, citrulline malate, on serum lactate and ammonia kinetics during a maximum exercise test in sedentary subjects. Semin. Hop Paris 1990, 66, 477–481. [Google Scholar]
- Sureda, A.; Pons, A. Arginine and cittruline supplementation in sports and exercise: Ergogenic nutrients. Med. Sport Sci. 2012, 59, 18–28. [Google Scholar] [PubMed]
- Perez-Guisado, J.; Jakeman, P.M. Citrulline malate enhances athletic anaerobic performance and relieves muscle soreness. J. Strength Cond. Res. 2010, 24, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Wax, B.; Kavazis, A.N.; Luckett, W. Effects of supplemental citrulline-malate ingestion on blood lactate, cardiovascular dynamics, and resistance exercise performance in trained males. J. Diet. Suppl. 2015, 13, 269–282. [Google Scholar] [CrossRef] [PubMed]
- Glenn, J.; Gray, M.; Wethington, L.; Stone, M.; Stewart, R.; Moyen, N. Acute citrulline-malate supplementation improves upper- and lower-body submaximal weightlifting exercise performance in resistance-trained females. Eur. J. Nutr. 2017, 56, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Rougé, C.; Des Robert, C.; Robins, A.; Le Bacquer, O.; Volteau, C.; De La Cochetière, M.F.; Darmaun, D. Manipulation of citrulline availability in humans. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G1061–G1067. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Cordova, A.; Ferrer, M.D.; Perez, G.; Tur, J.A.; Pons, A. l-Citrulline malate influence over branched chain amino acid utilization during exercise. Eur. J. Appl. Physiol. 2010, 110, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.; Flet, L.; Vavasseur, F.; Lemerle, M.; Ferchaud-Roucher, V.; Picot, D.; Darmaun, D. Oral citrulline does not affect whole body protein metabolism in healthy human volunteers: Results of a prospective, randomized, double-blind, cross-over study. Clin. Nutr. 2011, 30, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Bloomer, R.J. Nitric oxide supplements for sports. J. Strength Cond. Res. 2010, 32, 14–20. [Google Scholar] [CrossRef]
- Little, J.P.; Forbes, S.C.; Candow, D.G.; Cornish, S.M.; Chilibeck, P.D. Creatine, arginine alphaketoglutarate, amino acids, and medium-chain triglycerides and endurance and performance. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 493–508. [Google Scholar] [CrossRef] [PubMed]
- Wilcock, I.M.; Cronin, J.B.; Hing, W.A. Physiological response to water immersion: A method for sport recovery? Sports Med. 2006, 36, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Briand, J.; Blehaut, H.; Calvayrac, R.; Laval-Martin, D. Use of a microbial model for the determination of drug effects on cell metabolism and energetics: Study of citrulline-malate. Biopharm. Drug Dispos. 1992, 13, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Curis, E.; Nicolis, I.; Moinard, C.; Osowska, S.; Zerrouk, N.; Benazeth, S.; Cynober, L.A. Almost all about citrulline in mammals. Amino Acids 2005, 29, 177–205. [Google Scholar] [CrossRef] [PubMed]
- Cutrufello, P.T.; Gadomski, S.J.; Zavorsky, G.S. The effect of l-citrulline and watermelon juice supplementation on anaerobic and aerobic exercise performance. J. Sports Sci. 2015, 33, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Breuillard, C.; Cynober, L.; Moinard, C. Citrulline and nitrogen homeostasis: An overview. Amino Acids 2015, 47, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Machida, M.; Kohara, A.; Omi, N.; Takemasa, T. Effects of citrulline supplementation on fatigue and exercise performance in mice. J. Nutr. Sci. Vitaminol. 2011, 57, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. American College of Sports Medicine Joint Position Statement. Nutrition and Athletic Performance. Med. Sci. Sports Exerc. 2016, 48, 543–568. [Google Scholar] [PubMed]
- American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar]
- Sureda, A.; Cordova, A.; Ferrer, M.D.; Tauler, P.; Perez, G.; Tur, J.A.; Pons, A. Effects of L-citrulline oral supplementation on polymorphonuclear neutrophils oxidative burst and nitric oxide production after exercise. Free Radic. Res. 2009, 43, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.J.; Goss, F.L.; Rutkowski, J.; Lenz, B.; Dixon, C.; Timmer, J.; Frazee, K.; Dube, J.; Andreacci, J. Concurrent validation of the OMNI perceived exertion scale for resistance exercise. Med. Sci. Sport Exerc. 2003, 35, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Day, M.L.; McGuigan, M.R.; Brice, G.; Foster, C. Monitoring exercise intensity during resistance training using the session RPE scale. J. Strength Cond Res. 2004, 18, 353–358. [Google Scholar] [PubMed]
- Mattacola, C.G.; Perrin, D.H.; Gansneder, B.M.; Allen, J.D.; Mickey, C.A. A comparison of visual analog and graphic rating scales for assessing pain following delayed onset muscle soreness. J. Sport Rehabil. 1997, 6, 38–46. [Google Scholar] [CrossRef]
- Aguiar, A.F.; Buzzachera, C.F.; Pereira, R.M.; Sanches, V.C.; Januário, R.B.; da Silva, R.A.; Rabelo, L.M.; de Oliveira Gil, A.W. A single set of exhaustive exercise before resistance training improves muscular performance in young men. Eur. J. Appl. Physiol. 2015, 115, 1589–1599. [Google Scholar] [CrossRef] [PubMed]
- Larivière, C.; Arsenault, A.B.; Gravel, D.; Gagnon, D.; Loisel, P. Evaluation of measurement strategies to increase the reliability of EMG spectral indices to assess back muscle fatigue and recovery. J. Electromyogr. Kinesiol. 2002, 12, 91–102. [Google Scholar] [CrossRef]
- Kienbacher, T.; Habenicht, R.; Starek, C.; Mair, P.; Wolf, M.; Paul, B.; Riegler, S.; Kollmitzer, J.; Ebenbichler, G. The potential use of spectral electromyographic fatigue as a screening and outcome monitoring tool of sarcopenic back muscle alterations. J. NeuroEng. Rehabil. 2014, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.A.; Vieira, E.R.; Cabrera, M.; Altimari, L.R.; Aguiar, A.F.; Nowotny, A.H.; Carvalho, A.F.; Oliveira, M. Back muscle fatigue of younger and older adults with and without chronic low back pain using two protocols: A case-control study. J. Electromyogr. Kinesiol. 2015, 25, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, S.; Cuthill, I.C. Effect size, confidence interval and statistical significance: A practical guide for biologists. Biol. Rev. Camb. Philos. Soc. 2007, 82, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Page, P. Beyond statistical significance: Clinical interpretation of rehabilitation research literature. Int. J. Sports Phys. Ther. 2014, 9, 726–736. [Google Scholar] [PubMed]
- Batterham, A.M.; Hopkins, W.G. Making meaningful inferences about magnitudes. Int. J. Sports Physiol. Perform. 2006, 1, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, M. Distinguishing between statistical significance and practical/clinical meaningfulness using statistical inference. Sports Med. 2014, 44, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for Behavioral Sciences, 1st ed.; Academic Press: New York, NY, USA, 1977; p. 490. [Google Scholar]
- Levillain, O.; Parvy, P.; Hassler, C. Amino acid handling in uremic rats: Citrulline, a reliable marker of renal insufficiency and proximal tubular dysfunction. Metabolism 1997, 46, 611–618. [Google Scholar] [CrossRef]
- Byrne, C.; Twist, C.; Eston, R. Neuromuscular function after exercise-induced muscle damage: Theoretical and applied implications. Sports Med. 2004, 34, 49–69. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Cotie, L.M.; MacDonald, M.J.; Mitchell, C.J.; Prior, T.; Baker, S.K.; Phillips, S.M. Citrulline does not enhance blood flow, microvascular circulation, or myofibrillar protein synthesis in elderly men at rest or following exercise. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E71–E83. [Google Scholar] [CrossRef] [PubMed]
- De Luca, C.J. Use of the surface EMG signal for performance evaluation of back muscles. Muscle Nerve 1993, 16, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Arab, A.M.; Salavati, M. Sensitivity, specificity and predictive value of the clinical trunk muscle endurance tests in low back pain. Clin. Rehabil. 2007, 21, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.A.; Lariviere, C.; Arsenault, A.B.; Nadeau, S.; Plamondon, A. The comparison of wavelet- and Fourier-based electromyographic indices of back muscle fatigue during dynamic contractions: Validity and reliability results. Electromyogr. Clin. Neurophysiol. 2008, 48, 147–162. [Google Scholar] [PubMed]
- Adam, A.; De Luca, C.J. Recruitment order of motor units in human vastus lateralis muscle is maintained during fatiguing contraction. J. Neurophysiol. 2003, 90, 2919–2927. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Newton, M.; Sacco, P. Delayed-onset muscle soreness does not reflect the magnitude of eccentric exercise-induced muscle damage. Scan J. Med. Sci. Sports 2002, 12, 337–346. [Google Scholar] [CrossRef]
- Moinard, C.; Nicolis, I.; Neveux, N.; Darquy, S.; Bénazeth, S.; Cynober, L. Dose-ranging effects of citrulline administration on plasma amino acids and hormonal patterns in healthy subjects: The Citrudose pharmacokinetic study. Br. J. Nutr. 2008, 99, 855–862. [Google Scholar] [CrossRef] [PubMed]
| Age, year | 24.0 ± 3.3 |
| Height, cm | 175.1 ± 4.8 |
| Weight, kg | 77.4 ± 9.6 |
| BMI, kg/m2 | 25.3 ± 2.9 |
| Insulin, µIU/mL | 9.3 ± 6.4 |
| Glucose, mg/dL | 71.9 ± 19.0 |
| Cholesterol, mg/dL | 155.2 ± 31.0 |
| T1 | T2 | p Value | |
|---|---|---|---|
| Protein, g/kg/day | 1.8 ± 1.0 | 1.7 ± 0.8 | 0.51 |
| Carbohydrate, g/kg/day | 4.6 ± 1.6 | 4.9 ± 1.4 | 0.31 |
| Fat, g/kg/day | 1.1 ± 0.6 | 1.2 ± 0.8 | 0.67 |
| Total energy, kcal | 2681 ± 806 | 2874 ± 1058 | 0.47 |
| Total energy, kJ | 11,218 ± 3373 | 12,027 ± 4429 | 0.47 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, D.K.; Jacinto, J.L.; De Andrade, W.B.; Roveratti, M.C.; Estoche, J.M.; Balvedi, M.C.W.; De Oliveira, D.B.; Da Silva, R.A.; Aguiar, A.F. Citrulline Malate Does Not Improve Muscle Recovery after Resistance Exercise in Untrained Young Adult Men. Nutrients 2017, 9, 1132. https://doi.org/10.3390/nu9101132
Da Silva DK, Jacinto JL, De Andrade WB, Roveratti MC, Estoche JM, Balvedi MCW, De Oliveira DB, Da Silva RA, Aguiar AF. Citrulline Malate Does Not Improve Muscle Recovery after Resistance Exercise in Untrained Young Adult Men. Nutrients. 2017; 9(10):1132. https://doi.org/10.3390/nu9101132
Chicago/Turabian StyleDa Silva, Douglas K., Jeferson L. Jacinto, Walquiria B. De Andrade, Mirela C. Roveratti, José M. Estoche, Mario C. W. Balvedi, Douglas B. De Oliveira, Rubens A. Da Silva, and Andreo F. Aguiar. 2017. "Citrulline Malate Does Not Improve Muscle Recovery after Resistance Exercise in Untrained Young Adult Men" Nutrients 9, no. 10: 1132. https://doi.org/10.3390/nu9101132
APA StyleDa Silva, D. K., Jacinto, J. L., De Andrade, W. B., Roveratti, M. C., Estoche, J. M., Balvedi, M. C. W., De Oliveira, D. B., Da Silva, R. A., & Aguiar, A. F. (2017). Citrulline Malate Does Not Improve Muscle Recovery after Resistance Exercise in Untrained Young Adult Men. Nutrients, 9(10), 1132. https://doi.org/10.3390/nu9101132





