Indications and Use of the Gluten Contamination Elimination Diet for Patients with Non-Responsive Celiac Disease
Abstract
:1. Introduction
2. Treatment with the GFD
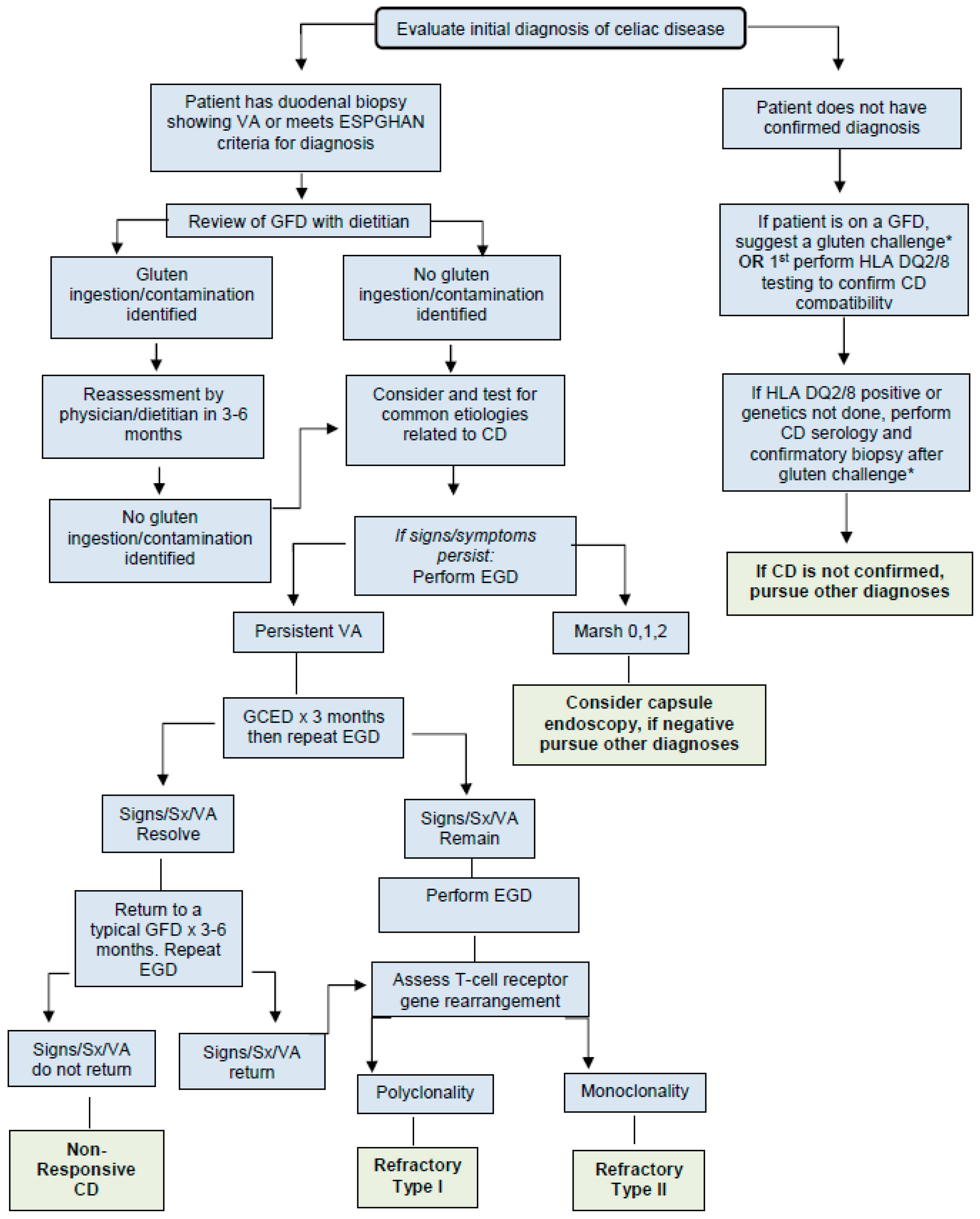
3. Evaluation of a Patient with Celiac Disease and Persistent Symptoms, Elevated Serology, and/or Persistent Enteropathy (Figure 1)
3.1. Evaluate Diagnosis of Celiac Disease
3.2. Evaluate for Gluten Contamination
- Recheck labels of favorite everyday foods, as ingredients can change without notice;
- Contact manufacturers of products that contain the statement “manufactured in a plant that also produces or used on a machine that also processes wheat” to ask about the procedures they use to avoid cross-contamination;
- Recheck all over-the-counter and prescription medications with the manufacturers to be sure they do not contain gluten;
- Evaluate religious ceremony/holiday foods or communion hosts to be sure they contain less than 20 parts per million of gluten;
- Evaluate frequency and strategies used when dining away from home;
- Ensure any ingested oats are certified gluten free;
- Evaluate the tolerance of gluten-free oats in the diet. The addition of uncontaminated oats to the GFD has been tolerated by the majority of CD patients; however, a few people with CD may be clinically intolerant to oats [25];
- Look for sources of cross-contamination at home, and ensure the following are implemented in the home:
- ○
- Use a separate toaster
- ○
- Thoroughly clean kitchen counters
- ○
- Use clean or separate cooking and serving utensils
- ○
- Avoid “double dipping” in common condiment jars.
3.3. Consideration of Other Etiologies of Symptoms
3.4. Evaluation of Celiac Disease Status
4. Assessment and Treatment of Patients with Non-Responsive and Refractory Celiac Disease
4.1. Immunosupressant Agents
4.2. Gluten Contamination Elimination Diet
4.2.1. Initiation of the Gluten Contamination Elimination Diet
4.2.2. Duration of the Gluten Contamination Elimination Diet
4.2.3. The Process of Returning to a Typical Gluten-free Diet
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Fasano, A.; Berti, I.; Gerarduzzi, T.; Not, T.; Colletti, R.B.; Drago, S.; Elitsur, Y.; Green, P.H.; Guandalini, S.; Hill, I.D.; et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: A large multicenter study. Arch. Intern. Med. 2003, 163, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Kryszak, D.; Bhatti, B.; Sturgeon, C.; Helzlsouer, K.; Clipp, S.L.; Gelfond, D.; Puppa, E.; Sferruzza, A.; Fasano, A. Natural history of celiac disease autoimmunity in a USA cohort followed since 1974. Ann. Med. 2010, 42, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Hill, I.D.; Dirks, M.H.; Liptak, G.S.; Colletti, R.B.; Fasano, A.; Guandalini, S.; Hoffenberg, E.J.; Horvath, K.; Murray, J.A.; Pivor, M.; et al. Guideline for the diagnosis and treatment of celiac disease in children: Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 136–160. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. ACG clinical guidelines: Diagnosis and management of celiac disease. Am. J. Gastroenterol. 2013, 108, 656–676. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.A.; Watson, T.; Clearman, B.; Mitros, F. Effect of a gluten-free diet on gastrointestinal symptoms in celiac disease. Am. J. Clin. Nutr. 2004, 79, 669–673. [Google Scholar] [PubMed]
- Hollon, J.R.; Pamela, A.C.; Margaret, L.M.; Elaine, L.L.P.; Alessio, F. Trace gluten contamination may play a role in mucosal and clinical recovery in a subgroup of diet-adherent non-responsive celiac disease patients. BMC Gastroenterol. 2013, 13, 40. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Rahim, M.W.; See, J.A.; Lahr, B.D.; Wu, T.T.; Murray, J.A. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am. J. Gastroenterol. 2010, 105, 1412–1420. [Google Scholar] [CrossRef] [PubMed]
- Ghazzawi, Y.; Rubio-Tapia, A.; Murray, J.A.; Absah, I. Mucosal healing in children with treated celiac disease. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 229–231. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.M.; Weir, D.C.; DeGroote, M.; Mitchell, P.D.; Singh, P.; Silvester, J.; Leichtner, A.; Fasano, A. Value of IgA tTG in Predicting Mucosal Recovery in Children with Celiac Disease on a Gluten-Free Diet. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Laurikka, P.; Salmi, T.; Collin, P.; Huhtala, H.; Mäki, M.; Kaukinen, K.; Kurppa, K. Gastrointestinal Symptoms in Celiac Disease Patients on a Long-Term Gluten-Free Diet. Nutrients 2016, 8, 429. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Cirillo, M.; Cavallaro, R.; Mazzacca, G. Long-term follow-up of celiac adults on gluten-free diet: Prevalence and correlates of intestinal damage. Digestion 2002, 66, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Mahadev, S.; Murray, J.A.; Wu, T.T.; Chandan, V.S.; Torbenson, M.S.; Kelly, C.P.; Maki, M.; Green, P.H.; Adelman, D.; Lebwohl, B. Factors associated with villus atrophy in symptomatic coeliac disease patients on a gluten-free diet. Aliment. Pharmacol. Ther. 2017, 45, 1084–1093. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Murray, J.A.; Rubio-Tapia, A.; Green, P.H.; Ludvigsson, J.F. Predictors of persistent villous atrophy in coeliac disease: A population-based study. Aliment. Pharmacol. Ther. 2014, 39, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Bannister, E.G.; Cameron, D.J.S.; Jessica, N. Can celiac serology alone be used as a marker of duodenal mucosal recovery in children with celiac disease on a gluten-free diet? Am. J. Gastroenterol. 2014, 109, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Vecsei, E.; Steinwendner, S.; Kogler, H.; Innerhofer, A.; Hammer, K.; Haas, O.A.; Amann, G.; Chott, A.; Vogelsang, H.; Schoenlechner, R.; et al. Follow-up of pediatric celiac disease: Value of antibodies in predicting mucosal healing, a prospective cohort study. BMC Gastroenterol. 2014, 14, 28. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Murray, J.A. Classification and management of refractory coeliac disease. Gut 2010, 59, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.K.; Lo, W.; Memeo, L.; Rotterdam, H.; Green, P.H. Duodenal histology in patients with celiac disease after treatment with a gluten-free diet. Gastrointest. Endosc. 2003, 57, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Vahedi, K.; Mascart, F.; Mary, J.Y.; Laberenne, J.E.; Bouhnik, Y.; Morin, M.C.; Ocmant, A.; Velly, C.; Colombel, J.F.; Matuchansky, C. Reliability of antitransglutaminase antibodies as predictors of gluten-free diet compliance in adult celiac disease. Am. J. Gastroenterol. 2003, 98, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Fabiani, E.; Iacono, G.; D’Agate, C.; Francavilla, R.; Biagi, F.; Volta, U.; Accomando, S.; Picarelli, A.; De Vitis, I.; et al. A prospective, double-blind, placebo-controlled trial to establish a safe gluten threshold for patients with celiac disease. Am. J. Clin. Nutr. 2007, 85, 160–166. [Google Scholar] [PubMed]
- Leffler, D.A.; Jessica, E.-G.; Melinda, D.; Detlef, S.; Francis, C.; Debra, L.F.; Jessica, B.-H.; Ciaran, P.K. Factors that influence adherence to a gluten-free diet in adults with celiac disease. Dig. Dis. Sci. 2008, 53, 1573–1581. [Google Scholar] [CrossRef] [PubMed]
- Gibert, A.; Espadaler, M.; Angel, C.M.; Sánchez, A.; Vaqué, C.; Rafecas, M. Consumption of gluten-free products: Should the threshold value for trace amounts of gluten be at 20, 100 or 200 p.p.m.? Eur. J. Gastroenterol. Hepatol. 2006, 18, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Schuppan, D.; Junker, Y.; Barisani, D. Celiac disease: From pathogenesis to novel therapies. Gastroenterology 2009, 137, 1912–1933. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.; Detlef, S.; Kumar, P.; Robert, N.; Jeffery, D.G.; Joshua, H.; Toufic, K.; Melinda, D.; Ciarán, P.K. Kinetics of the histological, serological and symptomatic responses to gluten challenge in adults with coeliac disease. Gut 2013, 62, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- La Vieille, S.; Pulido, O.M.; Abbott, M.; Koerner, T.B.; Celiac, S.G. Disease and Gluten-Free Oats: A Canadian Position Based on a Literature Review. Can. J. Gastroenterol. Hepatol. 2016, 2016, 1870305. [Google Scholar] [CrossRef] [PubMed]
- Revised criteria for diagnosis of coeliac disease. Report of Working Group of European Society of Paediatric Gastroenterology and Nutrition. Arch. Dis. Child. 1990, 65, 909–911. [Google Scholar]
- De Chaisemartin, L.; Tchao, M.; Georgia, M.; Fahima, F.; Frédérique, H.; Dorothée, R.; Fabienne, B.; Christophe, C.; Marie-Agnès, D. Application of Deamidated Gliadin Antibodies in the Follow-Up of Treated Celiac Disease. PLoS ONE 2015, 10, e0136745. [Google Scholar] [CrossRef] [PubMed]
- Dickey, W.; Hughes, D.F.; McMillan, S.A. Disappearance of endomysial antibodies in treated celiac disease does not indicate histological recovery. Am. J. Gastroenterol. 2000, 95, 712–714. [Google Scholar] [CrossRef] [PubMed]
- Silvester, J.A.; Kurada, S.; Szwajcer, A.; Kelly, C.P.; Leffler, D.A.; Duerksen, D.R. Tests for Serum Transglutaminase and Endomysial Antibodies Do Not Detect Most Patients with Celiac Disease and Persistent Villous Atrophy on Gluten-free Diets: A Meta-analysis. Gastroenterology 2017, 153, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Kelly, D.G.; Lahr, B.D.; Dogan, A.; Wu, T.T.; Murray, J.A. Clinical staging and survival in refractory celiac disease: A single center experience. Gastroenterology 2009, 136, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Maiuri, L.; Russo, I.; Tortora, R.; Bucci, C.; Cappello, C.; Santonicola, A.; Luciani, A.; Passananti, V.; Iovino, P. Efficacy of budesonide therapy in the early phase of treatment of adult coeliac disease patients with malabsorption: An in vivo/in vitro pilot study. Clin. Exp. Pharmacol. Physiol. 2009, 36, 1170–1176. [Google Scholar] [CrossRef] [PubMed]
- Wahab, P.J.; Crusius, J.B.; Meijer, J.W.; Uil, J.J.; Mulder, C.J. Cyclosporin in the treatment of adults with refractory coeliac disease--an open pilot study. Aliment. Pharmacol. Ther. 2000, 14, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Goerres, M.S.; Meijer, J.W.; Wahab, P.J.; Kerckhaert, J.A.; Groenen, P.J.; Van Krieken, J.H.; Mulder, C.J. Azathioprine and prednisone combination therapy in refractory coeliac disease. Aliment. Pharmacol. Ther. 2003, 18, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Jamma, S.; Leffler, D.A.; Dennis, M.; Najarian, R.M.; Schuppan, D.B.; Sheth, S.; Kelly, C.P. Small intestinal release mesalamine for the treatment of refractory celiac disease type I. J. Clin. Gastroenterol. 2011, 45, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Costantino, G.; della Torre, A.; Lo Presti, M.A.; Caruso, R.; Mazzon, E.; Fries, W. Treatment of life-threatening type I refractory coeliac disease with long-term infliximab. Dig. Liver Dis. 2008, 40, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.M.; Moorghen, M; Probert, C.S. Refractory coeliac disease: Remission with infliximab and immunomodulators. Eur. J. Gastroenterol. Hepatol. 2005, 17, 667–669. [Google Scholar] [CrossRef] [PubMed]
- Vivas, S.; Ruiz de Morales, J.M.; Fernando, R.; Suárez-Vilela, D. Alemtuzumab for refractory celiac disease in a patient at risk for enteropathy-associated T-cell lymphoma. N. Engl. J. Med. 2006, 354, 2514–2515. [Google Scholar] [CrossRef] [PubMed]
- Malamut, G.; El Machhour, R.; Montcuquet, N.; Martin-Lannerée, S.; Dusanter-Fourt, I.; Verkarre, V.; Mention, J.J.; Rahmi, G.; Kiyono, H.; Butz, E.A.; et al. IL-15 triggers an antiapoptotic pathway in human intraepithelial lymphocytes that is a potential new target in celiac disease-associated inflammation and lymphomagenesis. J. Clin. Investig. 2010, 120, 2131–2343. [Google Scholar] [CrossRef] [PubMed]
- Zanini, B.; Marullo, M.; Villanacci, V.; Salemme, M.; Lanzarotto, F.; Ricci, C.; Lanzini, A. Persistent Intraepithelial Lymphocytosis in Celiac Patients Adhering to Gluten-Free Diet Is Not Abolished Despite a Gluten Contamination Elimination Diet. Nutrients 2016, 8, 525. [Google Scholar] [CrossRef] [PubMed]
| Gluten Contamination |
|---|
| Change in fiber intake |
| Lactose Intolerance |
| Autoimmune enteropathy |
| Irritable bowel syndrome |
| Functional gastrointestinal disorders |
| Small-bowel bacterial overgrowth |
| Microscopic colitis |
| Pancreatic insufficiency |
| Refractory celiac disease |
| Indications for Use | Not for Use |
|---|---|
| Diagnosis of celiac disease is confirmed. | Diagnosis of celiac disease is not confirmed. |
| Patient has been on a gluten-free diet for 12 months. | Patient has non-celiac gluten sensitivity. |
| Patient has been seen by a dietician to review the diet for possible gluten exposure. | Patient has not been on a gluten-free diet for 12 months. |
| Patient has Marsh 3 damage on repeat small intestinal biopsy with or without elevated celiac antibodies. | Patients has Marsh 2 damage on repeat endoscopy in the presence of normal serology and no signs or symptoms associated with CD. |
| Proper education, support, and follow-up can be provided over the next 3 months. | Patient has Marsh 0-1 damage on repeat small intestinal biopsy with or without elevated celiac antibodies. |
| May consider use on a case by case basis for patients with persistent symptoms, elevated serology, and Marsh 2 damage on repeat small intestinal biopsy. |
| Phase 1: Week 1–2 | |
|---|---|
| Foods Allowed | |
| Fruits/Vegetables | All Fresh Fruits and Vegetables (no frozen or canned fruits/vegetables) |
| Grains | Rice (brown and white) (preferably labeled gluten free) |
| Proteins | Chicken Turkey (not self-basting) Fresh Fish/seafood Eggs |
| Beverages/Nutritional Supplements | 100% Fruit/Vegetable Juices Gatorade Fresh ground coffee 100% black or green tea (no herbal teas) Boost, Ensure (no malt flavor) |
| Seasoning/Condiments/Misc. | Fresh herbs (no dried herbs) Salt Fresh ground pepper Plain honey Olive oil Vinegar (excluding malt vinegar and flavored vinegars) |
| Phase 2: Week 3–12 | |
|---|---|
| Gradually Introduce Any of the Following Items over the Next Few Weeks. One New Food Can Be Introduced at a Time, With a New Food Added Every 2–3 Days. If Symptoms Develop or Worsen, Consult with Your Dietitian/Physician | |
| Foods Allowed | |
| Dairy (if tolerated) | Butter Yogurt (plain, unflavored) Cream (plain, unflavored) Cheeses Only ingredients: pasteurized milk, cheese cultures, salt, enzymes Cottage cheese Only ingredients: cultured milk, cream, and salt |
| Fruits/Vegetables | All Fresh Fruits and Vegetables (no frozen or canned fruits/vegetables) |
| Grains | Dried beans Carefully clean and sort for foreign particles Rice (brown and white) (preferably labeled gluten free) |
| Proteins (fresh only) | Chicken Turkey (not self-basting) Fresh fish/seafood Beef Pork and Lamb (no ham or bacon) Nuts in the shell or raw Eggs |
| Beverages/Nutritional Supplements | 100% Fruit/Vegetable Juices Gatorade Fresh ground coffee 100% black or green tea (no herbal teas) Boost, Ensure (no malt flavor) |
| Seasoning/Condiments/Misc. | Fresh herbs (no dried herbs) Salt and fresh ground pepper Plain honey and sugar Lemon Olive oil Vinegar (excluding malt vinegar and flavored vinegars) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonard, M.M.; Cureton, P.; Fasano, A. Indications and Use of the Gluten Contamination Elimination Diet for Patients with Non-Responsive Celiac Disease. Nutrients 2017, 9, 1129. https://doi.org/10.3390/nu9101129
Leonard MM, Cureton P, Fasano A. Indications and Use of the Gluten Contamination Elimination Diet for Patients with Non-Responsive Celiac Disease. Nutrients. 2017; 9(10):1129. https://doi.org/10.3390/nu9101129
Chicago/Turabian StyleLeonard, Maureen M., Pamela Cureton, and Alessio Fasano. 2017. "Indications and Use of the Gluten Contamination Elimination Diet for Patients with Non-Responsive Celiac Disease" Nutrients 9, no. 10: 1129. https://doi.org/10.3390/nu9101129
APA StyleLeonard, M. M., Cureton, P., & Fasano, A. (2017). Indications and Use of the Gluten Contamination Elimination Diet for Patients with Non-Responsive Celiac Disease. Nutrients, 9(10), 1129. https://doi.org/10.3390/nu9101129





