The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review
Abstract
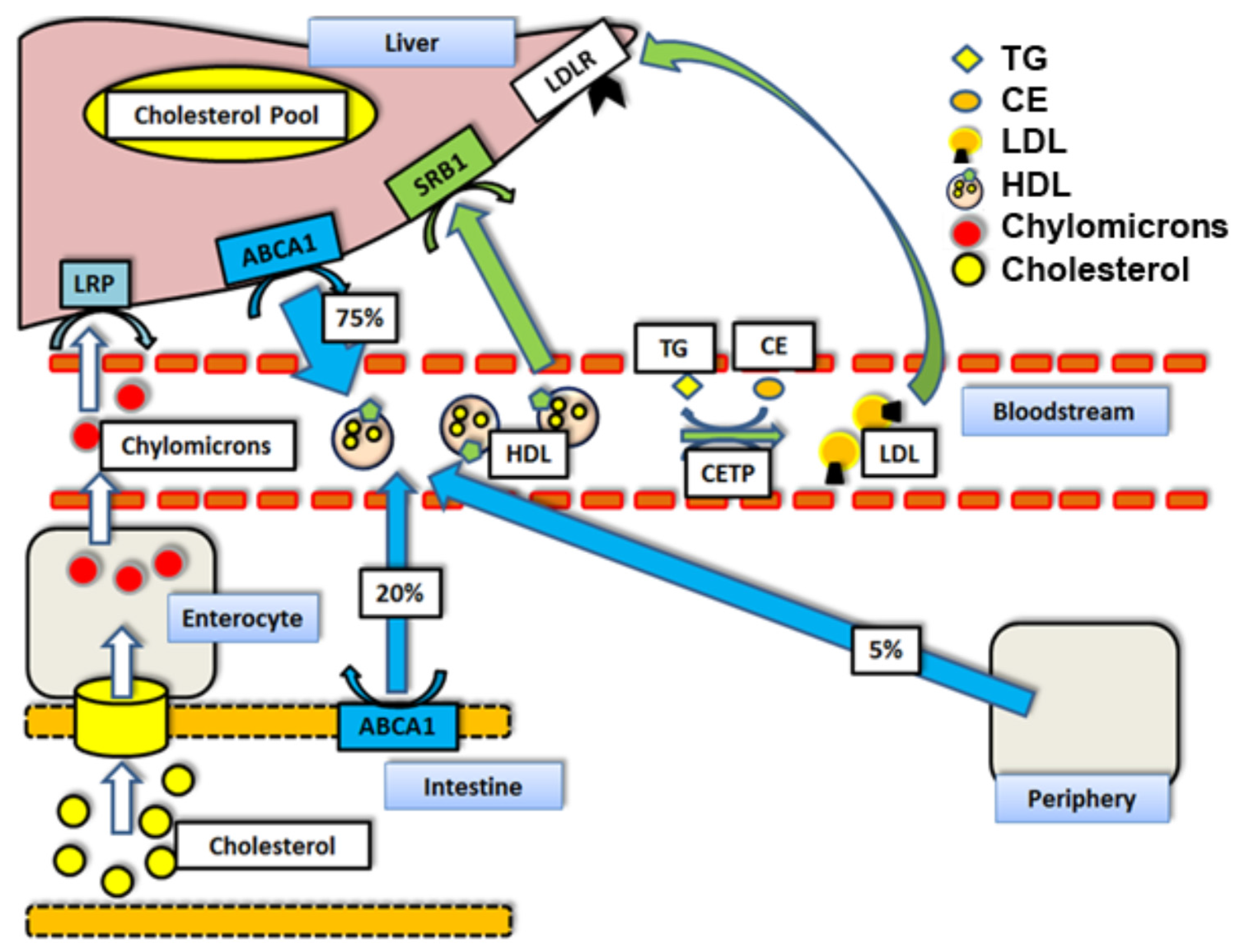
1. Introduction
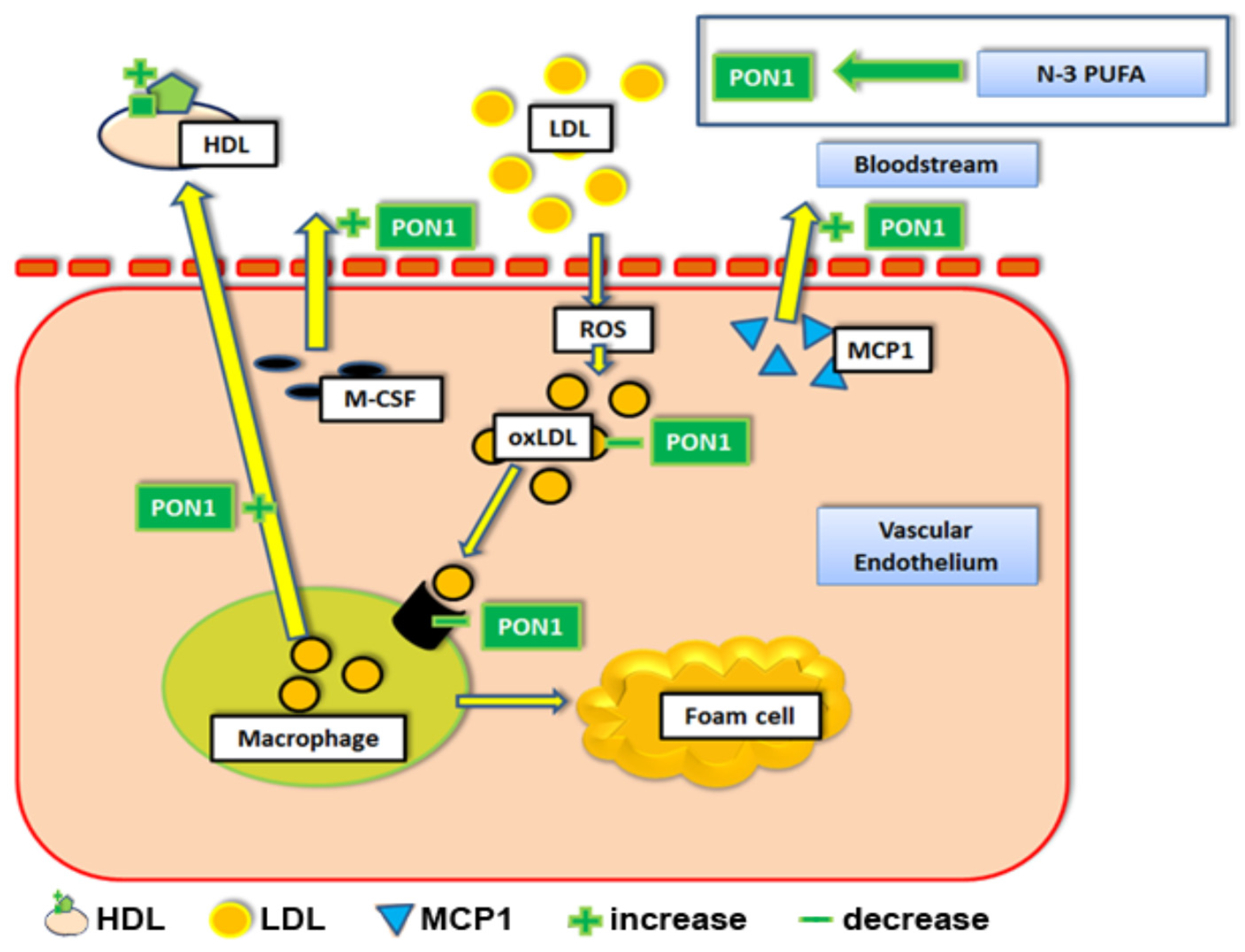
1.1. Apolipoprotein AI (apoA-I), Paraoxonase-1 (PON1), and n-3 PUFAs
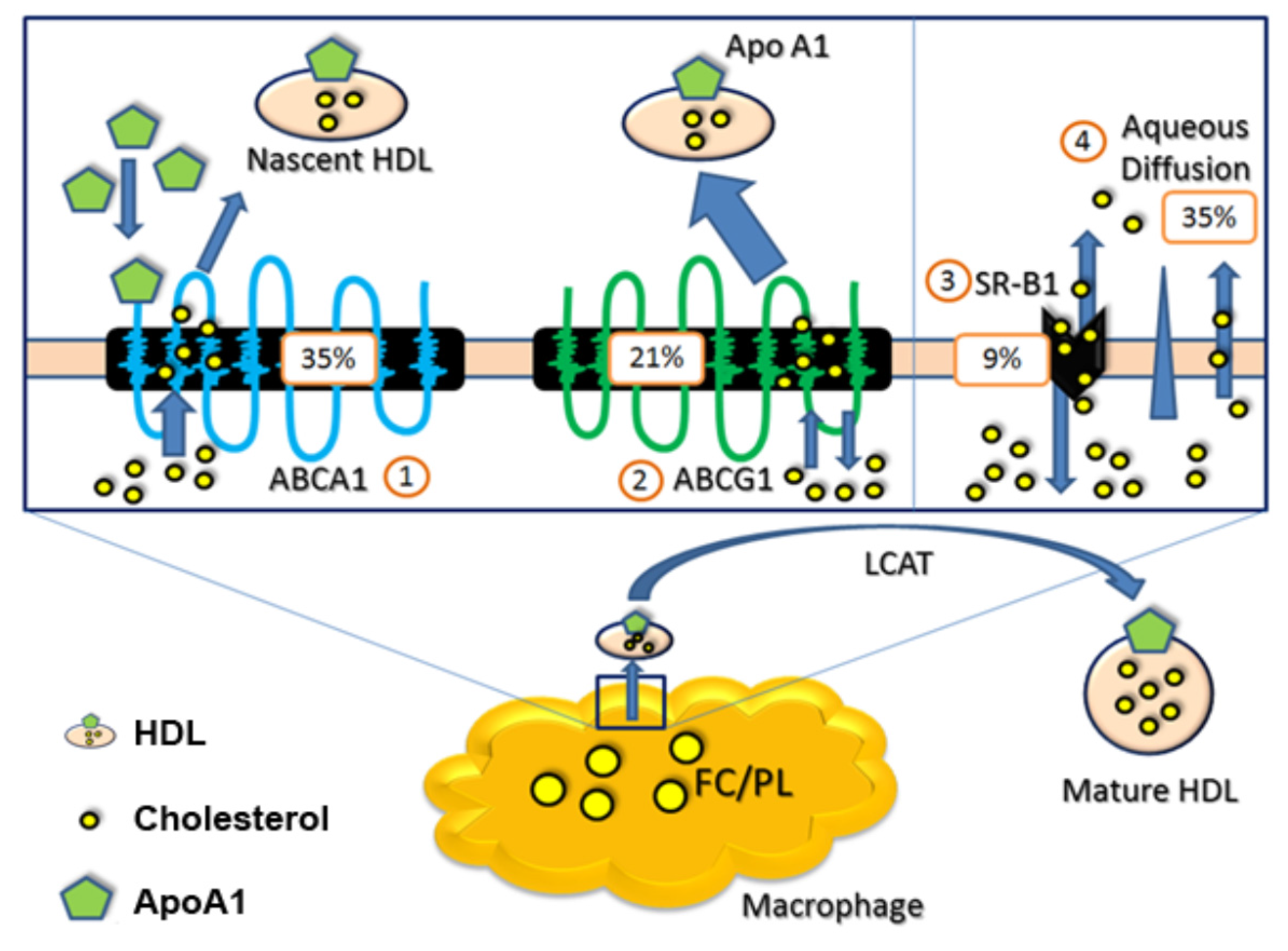
1.2. ATP-Binding Cassette Transporter A1 (ABCA1) and n-3 PUFAs
1.3. ATP-Binding Cassette Transporter G1 (ABCG1) and n-3 PUFAs
1.4. Apolipoprotein E (apoE) and n-3 PUFAs
1.5. Scavenger Receptor Class B Type I (SR-BI) and n-3 PUFAs
1.6. Cholesterol Ester Transfer Protein (CETP) and n-3 PUFAs
1.7. LDL-Receptor (LDLr) and n-3 PUFAs
1.8. Cholesterol 7alpha-Hydroxylase (CYP7A1), ABCG5/G8, NPC1L1 and n-3 PUFAs
2. Conclusions
Acknowledgments
Author Contributions
Conflicts of interest
References
- World Health Organization (WHO). The Top 10 Causes of Death; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Rader, D.J. Regulation of reverse cholesterol transport and clinical implications. Am. J. Cardiol. 2003, 92, 42–49. [Google Scholar] [CrossRef]
- Kannel, W.B.; Dawber, T.R.; Friedman, G.D.; Glennon, W.E.; Mcnamara, P.M. Risk Factors in Coronary Heart Disease: An Evaluation of Several Serum Lipids as Predictors of Coronary Heart Disease: The Framingham Study. Ann. Intern. Med. 1964, 61 (5 Pt 1), 888–899. [Google Scholar] [CrossRef]
- Assmann, G.; Cullen, P.; Schulte, H. Simple Scoring Scheme for Calculating the Risk of Acute Coronary Events Based on the 10-Year Follow-Up of the Prospective Cardiovascular Münster (PROCAM) Study. Circulation 2002, 105, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, H.K.; Kiss, D.; Rader, D. HDL-cholesterol and cardiovascular disease: rethinking our approach. Curr. Opin. Cardiol. 2015, 30, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Badimon, J.J.; Badimon, L.; Fuster, V. Regression of atherosclerotic lesions by high density lipoprotein plasma fraction in the cholesterol-fed rabbit. J. Clin. Investig. 1990, 85, 1234–1241. [Google Scholar] [CrossRef] [PubMed]
- Rubin, E.M.; Krauss, R.M.; Spangler, E.A.; Verstuyft, J.G.; Clift, S.M. Inhibition of early atherogenesis in transgenic mice by human apolipoprotein AI. Nature 1991, 353, 265–267. [Google Scholar] [CrossRef] [PubMed]
- Tangirala, R.K.; Tsukamoto, K.; Chun, S.H.; Usher, D.; Puré, E.; Rader, D.J. Regression of Atherosclerosis Induced by Liver-Directed Gene Transfer of Apolipoprotein A-I in Mice. Circulation 1999, 100, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Mahdy Ali, K.; Wonnerth, A.; Huber, K.; Wojta, J. Cardiovascular disease risk reduction by raising HDL cholesterol—Current therapies and future opportunities. Br. J. Pharmacol. 2012, 167, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R.S.; Brewer, H.B.; Davidson, W.S.; Fayad, Z.A.; Fuster, V.; Goldstein, J.; Hellerstein, M.; Jiang, X.C.; Phillips, M.C.; Rader, D.J.; et al. Cholesterol efflux and atheroprotection: Advancing the concept of reverse cholesterol transport. Circulation 2012, 125, 1905–1919. [Google Scholar] [CrossRef] [PubMed]
- Siscovick, D.S.; Barringer, T.A.; Fretts, A.M.; Wu, J.H.Y.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; Alger, H.M.; et al. Omega-3 Polyunsaturated Fatty Acid (Fish Oil) Supplementation and the Prevention of Clinical Cardiovascular Disease. Circulation 2017, 135, e867–e884. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Wu, J.H.Y. Omega-3 Fatty Acids and Cardiovascular Disease. J. Am. Coll. Cardiol. 2011, 58, 2047–2067. [Google Scholar] [CrossRef] [PubMed]
- Ramprasath, V.R.; Jones, P.J.H.; Buckley, D.D.; Woollett, L.A.; Heubi, J.E. Decreased plasma cholesterol concentrations after PUFA-rich diets are not due to reduced cholesterol absorption/synthesis. Lipids 2012, 47, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Vecka, M.; Dušejovská, M.; Stankova, B.; Zeman, M.; Vavrova, L.; Kodydkova, J.; Slabý, A.; Žák, A. N-3 polyunsaturated fatty acids in the treatment of atherogenic dyslipidemia. Neuro Endocrinol. Lett. 2012, 33 (Suppl. S2), 87–92. [Google Scholar] [PubMed]
- Wang, F.; Wang, Y.; Zhu, Y.; Liu, X.; Xia, H.; Yang, X.; Sun, G. Treatment for 6 months with fish oil-derived n-3 polyunsaturated fatty acids has neutral effects on glycemic control but improves dyslipidemia in type 2 diabetic patients with abdominal obesity: A randomized, double-blind, placebo-controlled trial. Eur. J. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, J. The role of the exogenous pathway in hypercholesterolaemia. Eur. Hear. J. Suppl. 2001, 3, 2–5. [Google Scholar] [CrossRef]
- Phillips, M.C. Molecular mechanisms of cellular cholesterol efflux. J. Biol. Chem. 2014, 289, 24020–24029. [Google Scholar] [CrossRef] [PubMed]
- Cuchel, M.; Rader, D.J. Macrophage reverse cholesterol transport: Key to the regression of atherosclerosis? Circulation 2006, 113, 2548–2555. [Google Scholar] [CrossRef] [PubMed]
- Rader, D.J.; Alexander, E.T.; Weibel, G.L.; Billheimer, J.; Rothblat, G.H. The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis. J. Lipid Res. 2009, 50 (Suppl. S1), 89–94. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Van Berkel, T.J.; Van Eck, M. Relative roles of various efflux pathways in net cholesterol efflux from macrophage foam cells in atherosclerotic lesions. Curr. Opin. Lipidol. 2010, 21, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.V.; Cuchel, M.; de la Llera-Moya, M.; Rodrigues, A.; Burke, M.F.; Jafri, K.; French, B.C.; Phillips, J.A.; Mucksavage, M.L.; Wilensky, R.L.; et al. Cholesterol efflux capacity, high-density lipoprotein function, and Atherosclerosis. N. Engl. J. Med. 2011, 364, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.; Watts, G.F.; Mori, T.A.; Barrett, P.H.R.; Redgrave, T.G.; Beilin, L.J. Randomized controlled trial of the effect of n-3 fatty acid supplementation on the metabolism of apolipoprotein B-100 and chylomicron remnants in men with visceral obesity. Am. J. Clin. Nutr. 2003, 77, 300–307. [Google Scholar] [PubMed]
- Park, Y.; Harris, W.S. Omega-3 fatty acid supplementation accelerates chylomicron triglyceride clearance. J. Lipid Res. 2003, 44, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.; Watts, G.F.; Barrett, P.H.R.; Beilin, L.J.; Redgrave, T.G.; Mori, T.A. Regulatory Effects of HMG CoA Reductase Inhibitor and Fish Oils on Apolipoprotein B-100 Kinetics in Insulin-Resistant Obese Male Subjects With Dyslipidemia. Diabetes 2002, 51, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.G.; Stone, N.J. Antiatherosclerotic and antithrombotic effects of omega-3 fatty acids. Am. J. Cardiol. 2006, 98, 39i–49i. [Google Scholar] [CrossRef] [PubMed]
- Zuliani, G.; Galvani, M.; Leitersdorf, E.; Volpato, S.; Cavalieri, M.; Fellin, R. The Role of Polyunsaturated Fatty Acids (PUFA) in the Treatment of Dyslipidemias. Curr. Pharm. Des. 2009, 15, 4087–4093. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, T.; Pellizzon, M.A.; Aihara, M.; Stylianou, I.M.; Billheimer, J.T.; Rothblat, G.; Rader, D.J. Fish Oil Promotes Macrophage Reverse Cholesterol Transport in Mice. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1502–1508. [Google Scholar] [CrossRef] [PubMed]
- Balogun, K.A.; Randunu, R.S.; Cheema, S.K. The effect of dietary omega-3 polyunsaturated fatty acids on plasma lipids and lipoproteins of C57BL/6 mice is age and sex specific. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Kasbi Chadli, F.; Nazih, H.; Krempf, M.; Nguyen, P.; Ouguerram, K. Omega 3 Fatty Acids Promote Macrophage Reverse Cholesterol Transport in Hamster Fed High Fat Diet. PLoS ONE 2013, 8, e61109. [Google Scholar] [CrossRef] [PubMed]
- Moss, J.W.E.; Davies, T.S.; Garaiova, I.; Plummer, S.F.; Michael, D.R.; Ramji, D.P. A Unique Combination of Nutritionally Active Ingredients Can Prevent Several Key Processes Associated with Atherosclerosis In Vitro. PLoS ONE 2016, 11, e0151057. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, Y.; Koo, S.I. ATP-binding cassette transporter A1 and HDL metabolism: Effects of fatty acids. J. Nutr. Biochem. 2012, 23, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zhang, T.; Zhao, S.; Li, W.; Ma, L.; Ding, M.; Liu, Y. Effects of n-3 polyunsaturated fatty acids high fat diet intervention on the synthesis of hepatic high-density lipoprotein cholesterol in obesity-insulin resistance rats. Lipids Health Dis. 2016, 15. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Irino, Y.; Shinohara, M.; Tsuda, S.; Mori, T.; Nagao, M.; Oshita, T.; Mori, K.; Hara, T.; Toh, R.; et al. Eicosapentaenoic Acid-Enriched High-Density Lipoproteins Exhibit Anti-Atherogenic Properties. Circ. J. 2017. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Ishida, T.; Nagao, M.; Mori, T.; Monguchi, T.; Sasaki, M.; Mori, K.; Kondo, K.; Nakajima, H.; Honjo, T.; et al. Administration of high dose eicosapentaenoic acid enhances anti-inflammatory properties of high-density lipoprotein in Japanese patients with dyslipidemia. Atherosclerosis 2014, 237, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Gaidukov, L.; Viji, R.I.; Yacobson, S.; Rosenblat, M.; Aviram, M.; Tawfik, D.S. ApoE induces serum paraoxonase PON1 activity and stability similar to ApoA-I. Biochemistry 2010, 49, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Litvinov, D.; Mahini, H.; Garelnabi, M. Antioxidant and anti-inflammatory role of paraoxonase 1: Implication in arteriosclerosis diseases. N. Am. J. Med. Sci. 2012, 4, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Fülöp, P.; Harangi, M.; Seres, I.; Paragh, G. Paraoxonase-1 and adipokines: Potential links between obesity and Atherosclerosis. Chem. Biol. Interact. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mackness, M.; Mackness, B. Human paraoxonase-1 (PON1): Gene structure and expression, promiscuous activities and multiple physiological roles. Gene 2015, 567, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Burillo, E.; Mateo-Gallego, R.; Cenarro, A.; Fiddyment, S.; Bea, A.M.; Jorge, I.; Vázquez, J.; Civeira, F. Beneficial effects of omega-3 fatty acids in the proteome of high-density lipoprotein proteome. Lipids Health Dis. 2012, 11, 116. [Google Scholar] [CrossRef] [PubMed]
- Berrougui, H.; Loued, S.; Khalil, A. Purified human paraoxonase-1 interacts with plasma membrane lipid rafts and mediates cholesterol efflux from macrophages. Free Radic. Biol. Med. 2012, 52, 1372–1381. [Google Scholar] [CrossRef] [PubMed]
- Landry, Y.D.; Denis, M.; Nandi, S.; Bell, S.; Vaughan, A.M.; Zha, X. ATP-binding cassette transporter A1 expression disrupts raft membrane microdomains through its ATPase-related functions. J. Biol. Chem. 2006, 281, 36091–36101. [Google Scholar] [CrossRef] [PubMed]
- Vedhachalam, C.; Duong, P.T.; Nickel, M.; Nguyen, D.; Dhanasekaran, P.; Saito, H.; Rothblat, G.H.; Lund-Katz, S.; Phillips, M.C. Mechanism of ATP-binding cassette transporter A1-mediated cellular lipid efflux to apolipoprotein A-I and formation of high density lipoprotein particles. J. Biol. Chem. 2007, 282, 25123–25130. [Google Scholar] [CrossRef] [PubMed]
- Daniels, T.F.; Killinger, K.M.; Michal, J.J.; Wright, R.W.; Jiang, Z. Lipoproteins, cholesterol homeostasis and cardiac health. Int. J. Biol. Sci. 2009, 5, 474–488. [Google Scholar] [CrossRef] [PubMed]
- Rye, K.A.; Barter, P.J. Cardioprotective functions of HDLs. J. Lipid Res. 2014, 55, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Mulya, A.; Lee, J.Y.; Gebre, A.K.; Thomas, M.J.; Colvin, P.L.; Parks, J.S. Minimal lipidation of pre-beta HDL by ABCA1 results in reduced ability to interact with ABCA1. Arter. Thromb. Vasc. Biol. 2007, 27, 1828–1836. [Google Scholar] [CrossRef] [PubMed]
- Fournier, N.; Tardivel, S.; Benoist, J.-F.; Vedie, B.; Rousseau-Ralliard, D.; Nowak, M.; Allaoui, F.; Paul, J.L. Eicosapentaenoic acid membrane incorporation impairs ABCA1-dependent cholesterol efflux via a protein kinase A signaling pathway in primary human macrophages. Biochim. Biophys. Acta 2016, 1861, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Uehara, Y.; Miura, S.; von Eckardstein, A.; Abe, S.; Fujii, A.; Matsuo, Y.; Rust, S.; Lorkowski, S.; Assmann, G.; Yamada, T.; et al. Unsaturated fatty acids suppress the expression of the ATP-binding cassette transporter G1 (ABCG1) and ABCA1 genes via an LXR/RXR responsive element. Atherosclerosis 2007, 191, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, A.M.; Oram, J.F. ABCG1 Redistributes Cell Cholesterol to Domains Removable by High Density Lipoprotein but Not by Lipid-depleted Apolipoproteins. J. Biol. Chem. 2005, 280, 30150–30157. [Google Scholar] [CrossRef] [PubMed]
- Sankaranarayanan, S.; Oram, J.F.; Asztalos, B.F.; Vaughan, A.M.; Lund-Katz, S.; Adorni, M.P.; Phillips, M.C.; Rothblat, G.H. Effects of acceptor composition and mechanism of ABCG1-mediated cellular free cholesterol efflux. J. Lipid Res. 2009, 50, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Moradi Sarabi, M.; Doosti, M.; Einollahi, N.; Hesami, S.S.; Dashti, N. Effect of eicosapentaenoic acid on the expression of ABCG1 gene in the human monocyte THP-1 cells. Acta Med. Iran. 2014, 52, 176–181. [Google Scholar] [PubMed]
- Barberger-Gateau, P.; Samieri, C.; Féart, C.; Plourde, M. Dietary Omega 3 polyunsaturated fatty acids and Alzheimer’s disease: Interaction with apolipoprotein E genotype. Curr. Alzheimer Res. 2011, 8, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Evans, D.A.; Bienias, J.L.; Tangney, C.C.; Bennett, D.A.; Wilson, R.S.; Aggarwal, N.; Schneider, J. Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch. Neurol. 2003, 60, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Kalmijn, S.; Launer, L.J.; Ott, A.; Witteman, J.C.; Hofman, A.; Breteler, M.M. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann. Neurol. 1997, 42, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Kolovou, G.; Anagnostopoulou, K.; Mikhailidis, D.P.; Cokkinos, D.V. Apolipoprotein E knockout models. Curr. Pharm. Des. 2008, 14, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Sheril, A.; Jeyakumar, S.M.; Jayashree, T.; Giridharan, N.V.; Vajreswari, A. Impact of feeding polyunsaturated fatty acids on cholesterol metabolism of dyslipidemic obese rats of WNIN/GR-Ob strain. Atherosclerosis 2009, 204, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Lambert, C.; Cubedo, J.; Padró, T.; Sánchez-Hernández, J.; Antonijoan, R.M.; Perez, A.; Badimon, L. Phytosterols and Omega 3 Supplementation Exert Novel Regulatory Effects on Metabolic and Inflammatory Pathways: A Proteomic Study. Nutrients 2017, 9, 599. [Google Scholar] [CrossRef] [PubMed]
- Minihane, A.M. Impact of Genotype on EPA and DHA Status and Responsiveness to Increased Intakes. Nutrients 2016, 8, 123. [Google Scholar] [CrossRef] [PubMed]
- Chouinard-Watkins, R.; Conway, V.; Minihane, A.M.; Jackson, K.G.; Lovegrove, J.A.; Plourde, M. Interaction between BMI and APOE genotype is associated with changes in the plasma long-chain-PUFA response to a fish-oil supplement in healthy participants. Am. J. Clin. Nutr. 2015, 102, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Thifault, E.; Cormier, H.; Bouchard-Mercier, A.; Rudkowska, I.; Paradis, A.-M.; Garneau, V.; Ouellette, C.; Lemieux, S.; Couture, P.; Vohl, M.C. Effects of age, sex, body mass index and APOE genotype on cardiovascular biomarker response to an n-3 polyunsaturated fatty acid supplementation. J. Nutrigenet. Nutrigenom. 2013, 6, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Van Eck, M.; Bos, I.S.T.; Hildebrand, R.B.; Van Rij, B.T.; Van Berkel, T.J.C. Dual role for scavenger receptor class B, type I on bone marrow-derived cells in atherosclerotic lesion development. Am. J. Pathol. 2004, 165, 785–794. [Google Scholar] [CrossRef]
- Ji, Y.; Jian, B.; Wang, N.; Sun, Y.; Moya, M.L.; Phillips, M.C.; Rothblat, G.H.; Swaney, J.B.; Tall, A.R. Scavenger receptor BI promotes high density lipoprotein-mediated cellular cholesterol efflux. J. Biol. Chem. 1997, 272, 20982–20985. [Google Scholar] [CrossRef] [PubMed]
- Yancey, P.G.; de la Llera-Moya, M.; Swarnakar, S.; Monzo, P.; Klein, S.M.; Connelly, M.A.; Johnson, W.J.; Williams, D.L.; Rothblat, G.H. High density lipoprotein phospholipid composition is a major determinant of the bi-directional flux and net movement of cellular free cholesterol mediated by scavenger receptor BI. J. Biol. Chem. 2000, 275, 36596–36604. [Google Scholar] [CrossRef] [PubMed]
- Yvan-Charvet, L.; Pagler, T.A.; Wang, N.; Senokuchi, T.; Brundert, M.; Li, H.; Rinninger, F.; Tall, A.R. SR-BI inhibits ABCG1-stimulated net cholesterol efflux from cells to plasma HDL. J. Lipid Res. 2008, 49, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Collins, H.L.; Ranalletta, M.; Fuki, I.V.; Billheimer, J.T.; Rothblat, G.H.; Tall, A.R.; Rader, D.J. Macrophage ABCA1 and ABCG1, but not SR-BI, promote macrophage reverse cholesterol transport in vivo. J. Clin. Investig. 2007, 117, 2216–2224. [Google Scholar] [CrossRef] [PubMed]
- Brundert, M.; Heeren, J.; Bahar-Bayansar, M.; Ewert, A.; Moore, K.J.; Rinninger, F. Selective uptake of HDL cholesteryl esters and cholesterol efflux from mouse peritoneal macrophages independent of SR-BI. J. Lipid Res. 2006, 47, 2408–2421. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Da Silva, J.R.; Reilly, M.; Billheimer, J.T.; Rothblat, G.H.; Rader, D.J. Hepatic expression of scavenger receptor class B type I (SR-BI) is a positive regulator of macrophage reverse cholesterol transport in vivo. J. Clin. Investig. 2005, 115, 2870–2874. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Geng, Y.; Xiao, N.; Yin, M.; Mao, L.; Ren, G.; Zhang, C.; Liu, P.; Lu, N.; An, L.; Pan, J. High dietary n-6/n-3 PUFA ratio promotes HDL cholesterol level, but does not suppress atherogenesis in apolipoprotein E-null mice 1. J. Atheroscler. Thromb. 2009, 16, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Mashurabad, P.C.; Kondaiah, P.; Palika, R.; Ghosh, S.; Nair, M.K.; Raghu, P. Eicosapentaenoic acid inhibits intestinal β-carotene absorption by downregulation of lipid transporter expression via PPAR-α dependent mechanism. Arch. Biochem. Biophys. 2016, 590, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Raposo, H.F.; Patrício, P.R.; Simões, M.C.; Oliveira, H.C.F. Fibrates and fish oil, but not corn oil, up-regulate the expression of the cholesteryl ester transfer protein (CETP) gene. J. Nutr. Biochem. 2014, 25, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Cheema, S.K.; Agarwal-Mawal, A.; Murray, C.M.; Tucker, S. Lack of stimulation of cholesteryl ester transfer protein by cholesterol in the presence of a high-fat diet. J. Lipid Res. 2005, 46, 2356–2366. [Google Scholar] [CrossRef] [PubMed]
- Michalik, L.; Wahli, W. Peroxisome proliferator-activated receptors: Three isotypes for a multitude of functions. Curr. Opin. Biotechnol. 1999, 10, 564–570. [Google Scholar] [CrossRef]
- Ishida, T.; Ohta, M.; Nakakuki, M.; Kami, H.; Uchiyama, R.; Kawano, H.; Notsu, T.; Imada, K.; Shimano, H. Distinct regulation of plasma LDL cholesterol by eicosapentaenoic acid and docosahexaenoic acid in high fat diet-fed hamsters: Participation of cholesterol ester transfer protein and LDL receptor. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Leigh-Firbank, E.C.; Minihane, A.M.; Leake, D.S.; Wright, J.W.; Murphy, M.C.; Griffin, B.A.; Williams, C.M. Eicosapentaenoic acid and docosahexaenoic acid from fish oils: Differential associations with lipid responses. Br. J. Nutr. 2002, 87, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Amanda Pepper-Yowell, M.S.; Ballou, M.; Trojan, S.; Thompson, L.; Moustaid-Moussa, N.; Wang, S.; Sheridan, M. The Role Docosapentaenoic Acid Omega-3 Plays in Controlling Lipid Metabolism and Inflammation Associated with Obesity, Atherosclerosis, and Neurodegenerative Disease. Ph.D. Thesis, Texas Tech University, Lubbock, TX, USA, 2014. [Google Scholar]
- Vasandani, C.; Kafrouni, A.I.; Caronna, A.; Bashmakov, Y.; Gotthardt, M.; Horton, J.D.; Spady, D.K. Upregulation of hepatic LDL transport by n-3 fatty acids in LDL receptor knockout mice. J. Lipid Res. 2002, 43, 772–784. [Google Scholar] [PubMed]
- Pan, M.; Cederbaum, A.I.; Zhang, Y.-L.; Ginsberg, H.N.; Williams, K.J.; Fisher, E.A. Lipid peroxidation and oxidant stress regulate hepatic apolipoprotein B degradation and VLDL production. J. Clin. Investig. 2004, 113, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Maitin, V.; Parathath, S.; Andreo, U.; Lin, S.X.; St. Germain, C.; Yao, Z.; Maxfield, F.R.; Williams, K.J.; Fisher, E.A. Presecretory oxidation, aggregation, and autophagic destruction of apoprotein-B: A pathway for late-stage quality control. Proc. Natl. Acad. Sci. USA 2008, 105, 5862–5867. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, D.; Witztum, J.L. Is the Oxidative Modification Hypothesis Relevant to Human Atherosclerosis? Circulation 2002, 105, 2107–2111. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.G.; Zhao, X.-Q.; Chait, A.; Fisher, L.D.; Cheung, M.C.; Morse, J.S.; Dowdy, A.A.; Marino, E.K.; Bolson, E.L.; Alaupovic, P.; et al. Simvastatin and Niacin, Antioxidant Vitamins, or the Combination for the Prevention of Coronary Disease. N. Engl. J. Med. 2001, 345, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.G.; Cheung, M.C.; Lee, A.C.; Zhao, X.-Q.; Chait, A. Antioxidant Vitamins and Lipid Therapy. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Bérard, A.M.; Dumon, M.-F.; Darmon, M. Dietary fish oil up-regulates cholesterol 7α-hydroxylase mRNA in mouse liver leading to an increase in bile acid and cholesterol excretion. FEBS Lett. 2004, 559, 125–128. [Google Scholar] [CrossRef]
- Jakulj, L.; Besseling, J.; Stroes, E.S.G.; Groen, A.K. Intestinal cholesterol secretion: Future clinical implications. Neth. J. Med. 2013, 71, 459–465. [Google Scholar] [PubMed]
- Alvaro, A.; Rosales, R.; Masana, L.; Vallvé, J.-C. Polyunsaturated fatty acids down-regulate in vitro expression of the key intestinal cholesterol absorption protein NPC1L1: No effect of monounsaturated nor saturated fatty acids. J. Nutr. Biochem. 2010, 21, 518–525. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pizzini, A.; Lunger, L.; Demetz, E.; Hilbe, R.; Weiss, G.; Ebenbichler, C.; Tancevski, I. The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review. Nutrients 2017, 9, 1099. https://doi.org/10.3390/nu9101099
Pizzini A, Lunger L, Demetz E, Hilbe R, Weiss G, Ebenbichler C, Tancevski I. The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review. Nutrients. 2017; 9(10):1099. https://doi.org/10.3390/nu9101099
Chicago/Turabian StylePizzini, Alex, Lukas Lunger, Egon Demetz, Richard Hilbe, Guenter Weiss, Christoph Ebenbichler, and Ivan Tancevski. 2017. "The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review" Nutrients 9, no. 10: 1099. https://doi.org/10.3390/nu9101099
APA StylePizzini, A., Lunger, L., Demetz, E., Hilbe, R., Weiss, G., Ebenbichler, C., & Tancevski, I. (2017). The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review. Nutrients, 9(10), 1099. https://doi.org/10.3390/nu9101099



