Glycemic Response to Black Beans and Chickpeas as Part of a Rice Meal: A Randomized Cross-Over Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Design
2.3. Test Meals
2.4. Data and Statistical Analysis
3. Results
3.1. Participant Characteristics
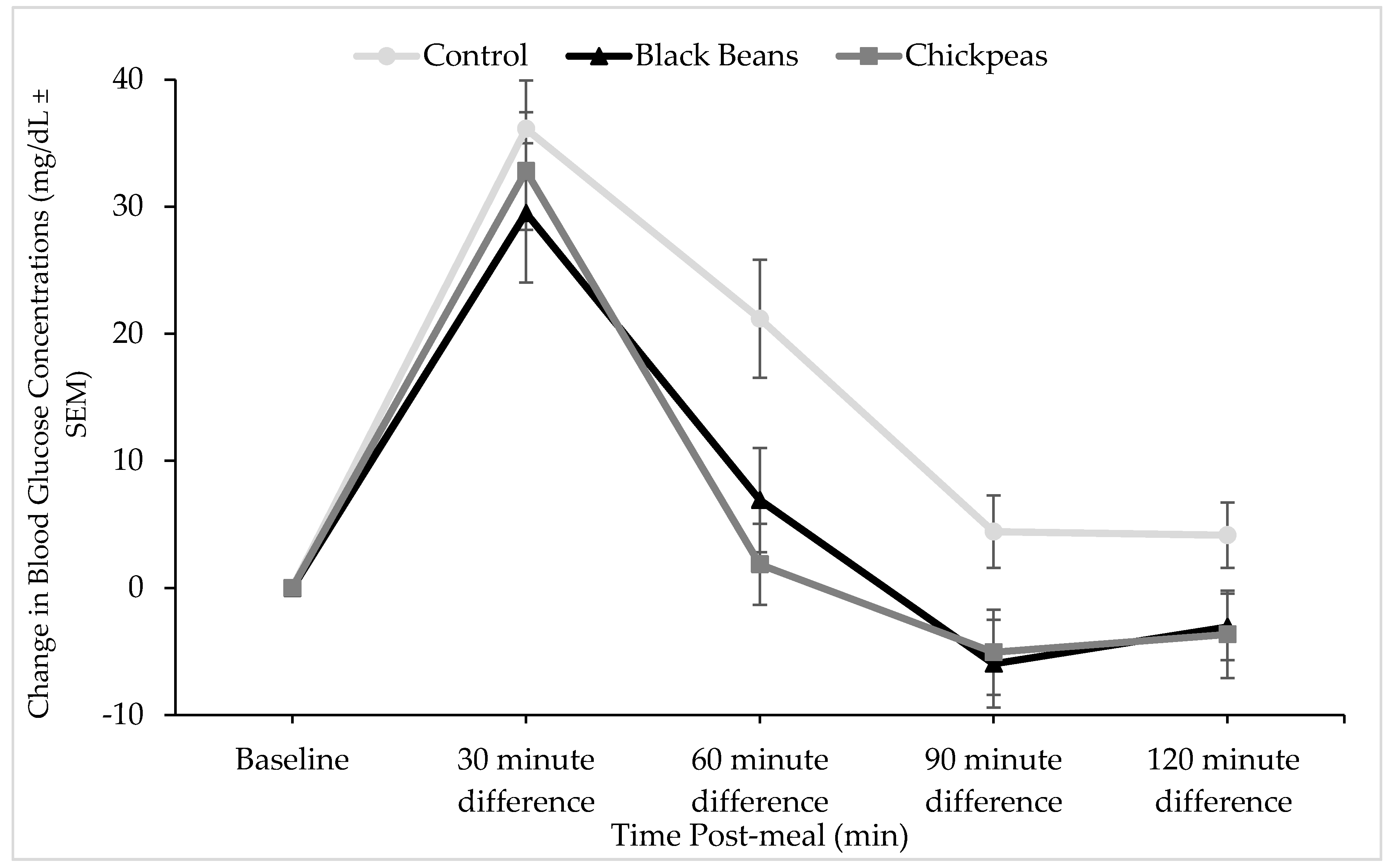
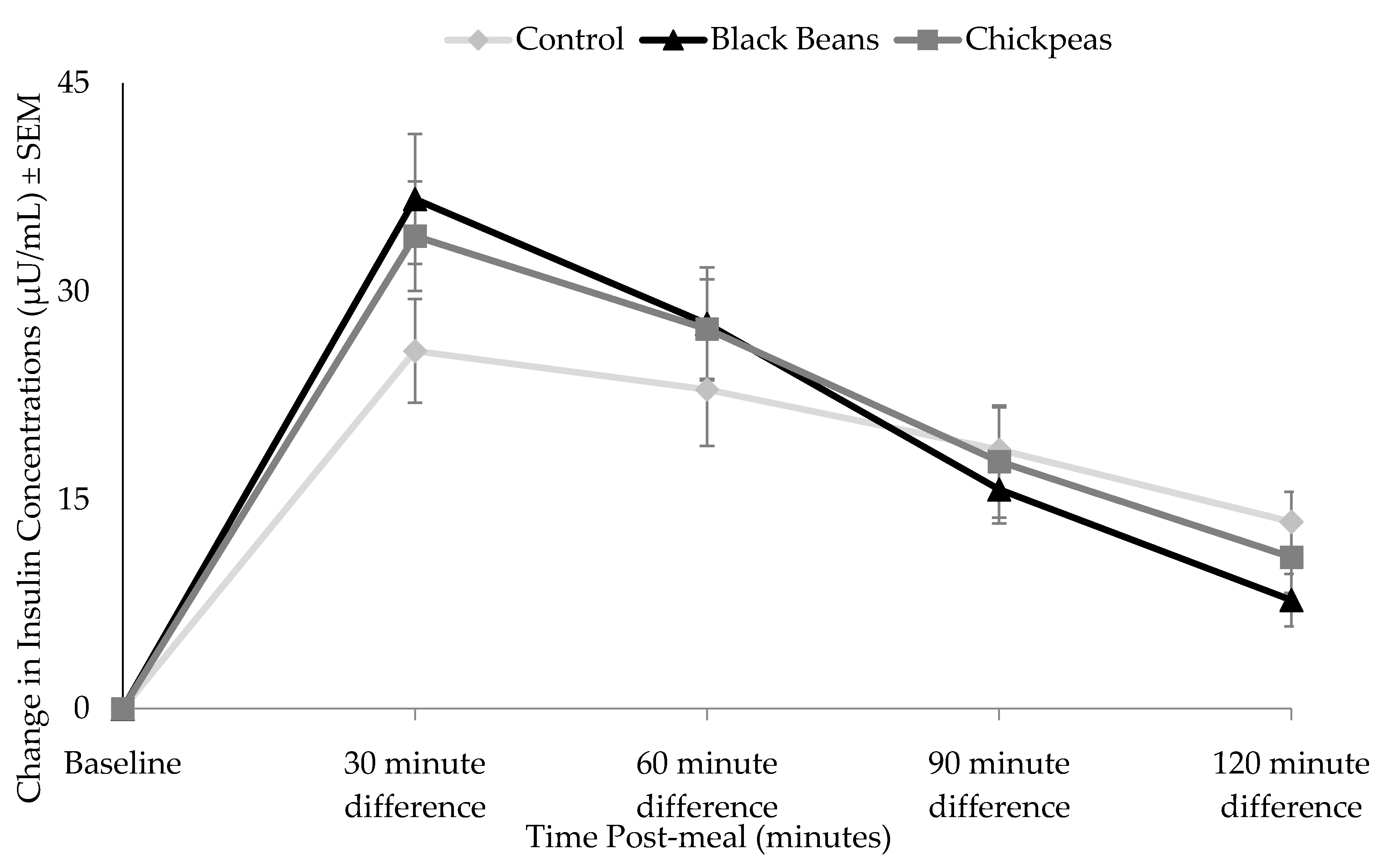
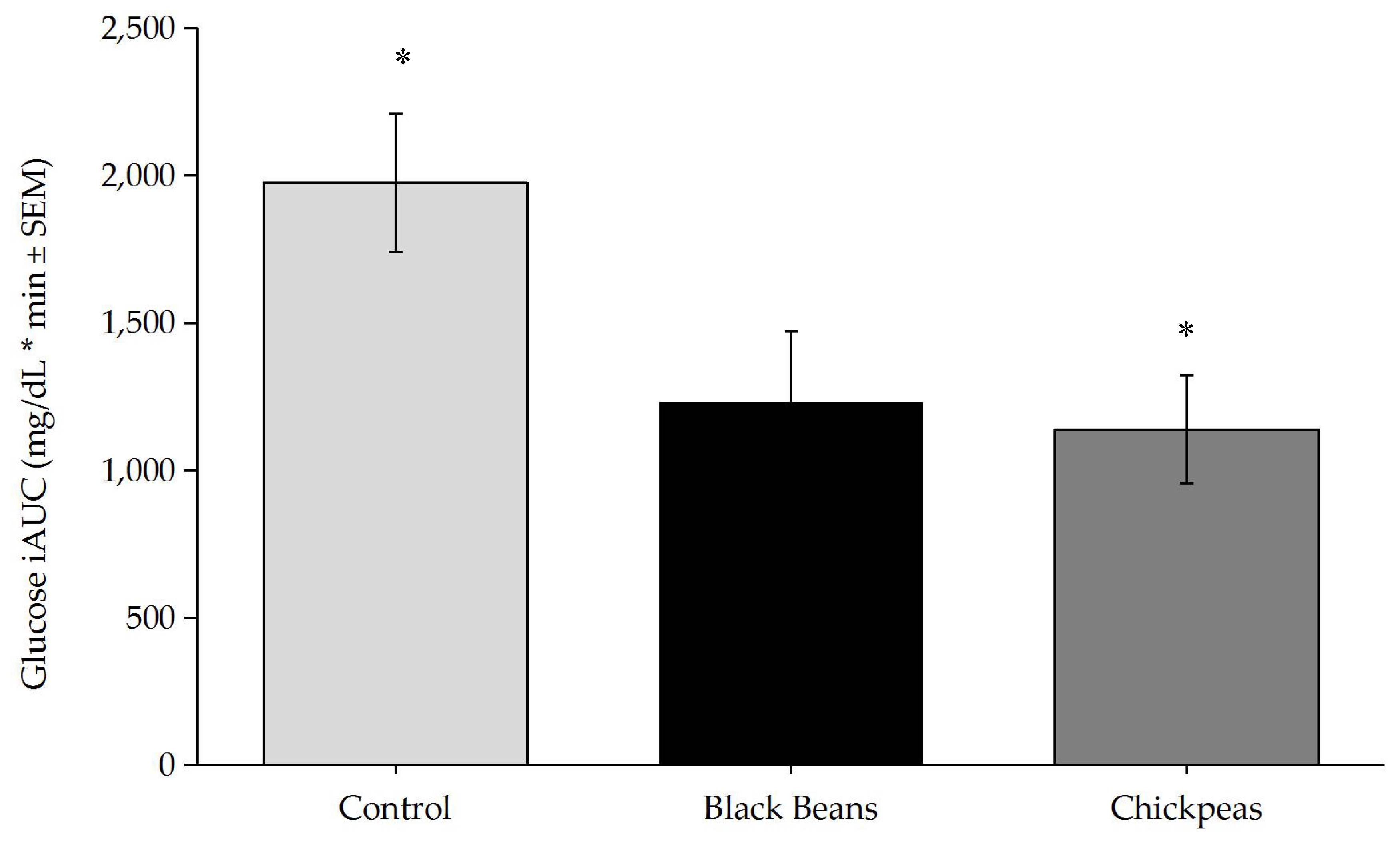
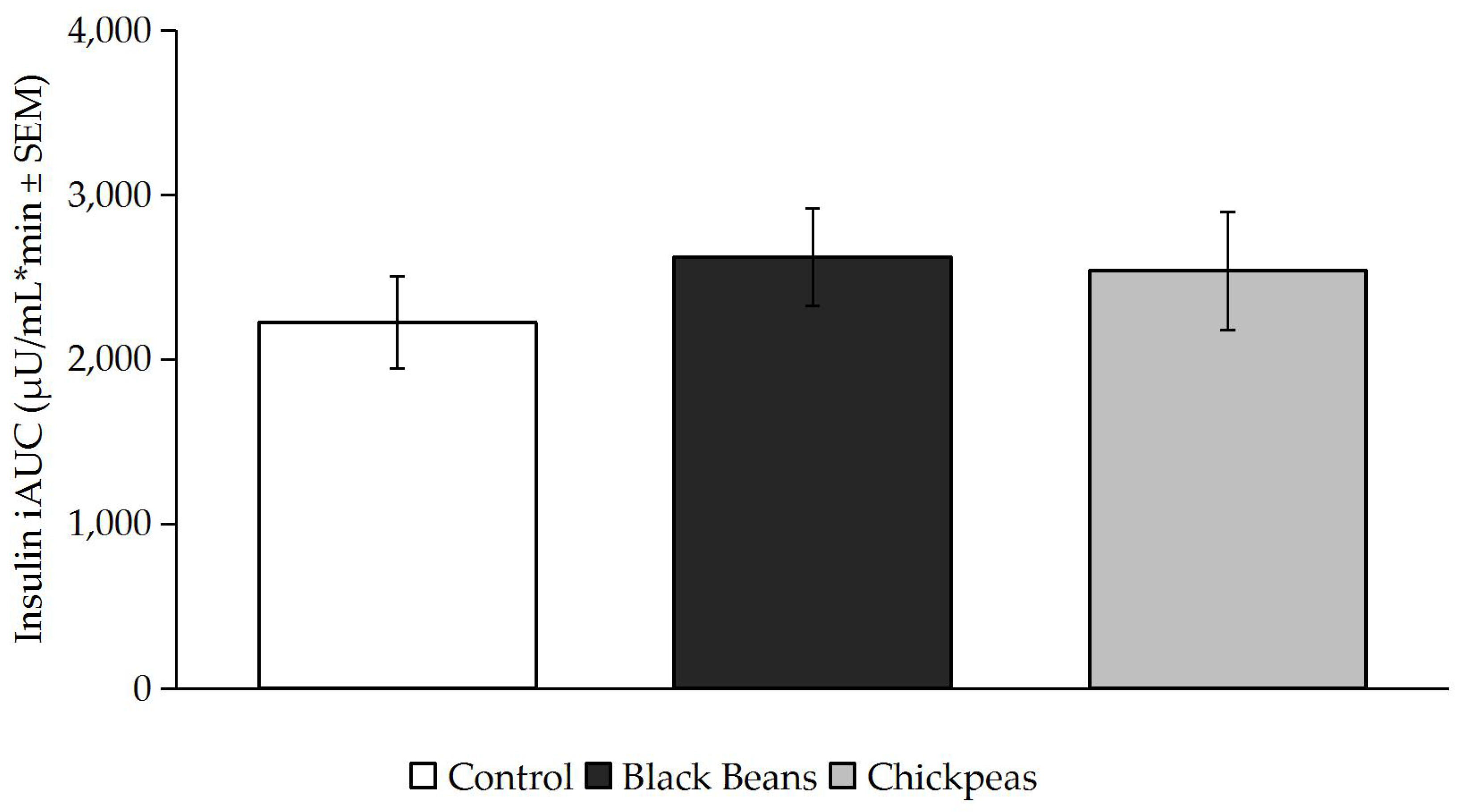
3.2. Glucose and Insulin Responses
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wilk, R.; Barbosa, L. Rice and Beans: A Unique Dish in a Hundred Places; Berg: New York, NY, USA, 2012; ISBN 978-1847889041 (pbk.). [Google Scholar]
- McCrory, M.A.; Hamaker, B.R.; Lovejoy, J.C.; Eichelsdoerfer, P.E. Pulse Consumption, Satiety, and Weight Management. Adv. Nutr. Int. Rev. J. 2010, 1, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Lam, H.-M.; Nguyen, H.T.; Siddique, K.H.M.; Varshney, R.K.; Colmer, T.D.; Cowling, W.; Bramley, H.; Mori, T.A.; Hodgson, J.M.; et al. Neglecting legumes has compromised human health and sustainable food production. Nat. Plants 2016, 2, 16112. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, A.M.; Winham, D.M.; Thompson, S.V. Phaseolus beans: Impact on glycaemic response and chronic disease risk in human subjects. Br. J. Nutr. 2012, 108, S52–S65. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; Thompson, A.M.; Tees, M.T.; Nguyen, C.H.; Winham, D.M. Non-soy legume consumption lowers cholesterol levels: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Hartman, T.J.; Albert, P.S.; Zhang, Z.; Bagshaw, D.; Kris-Etherton, P.M.; Ulbrecht, J.; Miller, C.K.; Bobe, G.; Colburn, N.H.; Lanza, E. Consumption of a legume-enriched, low-glycemic index diet is associated with biomarkers of insulin resistance and inflammation among men at risk for colorectal cancer. J. Nutr. 2010, 140, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Ha, V.; Sievenpiper, J.L.; de Souza, R.J.; Jayalath, V.H.; Mirrahimi, A.; Agarwal, A.; Chiavaroli, L.; Mejia, S.B.; Sacks, F.M.; Di Buono, M.; et al. Effect of dietary pulse intake on established therapeutic lipid targets for cardiovascular risk reduction: A systematic review and meta-analysis of randomized controlled trials. CMAJ 2014, 186, E252–E262. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; de Souza, R.J.; Choo, V.L.; Ha, V.; Cozma, A.I.; Chiavaroli, L.; Mirrahimi, A.; Blanco Mejia, S.; Di Buono, M.; Bernstein, A.M.; et al. Effects of dietary pulse consumption on body weight: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Thorne, M.J.; Thompson, L.U.; Jenkins, D.J. Factors affecting starch digestibility and the glycemic response with special reference to legumes. Am. J. Clin. Nutr. 1983, 38, 481–488. [Google Scholar] [PubMed]
- Park, O.J.; Kang, N.E.; Chang, M.J.; Kim, W.K. Resistant starch supplementation influences blood lipid concentrations and glucose control in overweight subjects. J. Nutr. Sci. Vitaminol. Tokyo 2004, 50, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Hosoya, S.; Nishimura, S.; Tanaka, T.; Kajimoto, Y.; Nishimura, A.; Kajimoto, O. Effect of bread containing resistant starch on postprandial blood glucose levels in humans. Biosci. Biotechnol. Biochem. 2005, 69, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Kendall, C.W.C.; de Souza, R.J.; Jayalath, V.H.; Cozma, A.I.; Ha, V.; Mirrahimi, A.; Chiavaroli, L.; Augustin, L.S.A.; Blanco Mejia, S.; et al. Dietary pulses, satiety and food intake: A systematic review and meta-analysis of acute feeding trials. Obesity 2014, 22, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Mattei, J.; Malik, V.; Wedick, N.M.; Hu, F.B.; Spiegelman, D.; Willett, W.C.; Campos, H. Reducing the global burden of type 2 diabetes by improving the quality of staple foods: The Global Nutrition and Epidemiologic Transition Initiative. Glob. Health 2015, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Doak, C.M.; Popkin, B.M. Overweight and Obesity. In Nutrition and Health in a Developing World; Springer International Publishing: Cham, Switzerland, 2017; pp. 143–158. [Google Scholar]
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4·4 million participants. Lancet 2016, 387, 1513–1530. [Google Scholar] [CrossRef]
- Madsbad, S. Impact of postprandial glucose control on diabetes-related complications: How is the evidence evolving? J. Diabetes Complicat. 2016, 30, 374–385. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care 2013, 39, 1033–1046. [Google Scholar] [CrossRef]
- Abdullah, M.; Marinangeli, C.; Jones, P.; Carlberg, J. Canadian Potential Healthcare and Societal Cost Savings from Consumption of Pulses: A Cost-Of-Illness Analysis. Nutrients 2017, 9, 793. [Google Scholar] [CrossRef] [PubMed]
- Heron, M. Deaths: Leading Causes for 2014. Natl. Vital Stat. Rep. 2016, 65, 1–96. [Google Scholar] [PubMed]
- Winham, D.M.; Armstrong Florian, T.L.; Thompson, S.V. Low-income US women under-informed of the specific health benefits of consuming beans. PLoS ONE 2016, 11, e0147592. [Google Scholar] [CrossRef] [PubMed]
- Akibode, S.; Maredia, M. Global and Regional Trends in Production, Trade and Consumption of Food Legume Crops; Department of Agricultural, Food and Resource Economics, Michigan State University: East Lansing, MI, USA, 2011. [Google Scholar]
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference; Release 28; US Department of Agriculture, Agricultural Research Service: Beltsville, MD, USA, 2016.
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. Dietary Guidelines for Americans 2015–2020; U.S. Department of Health and Human Services; U.S. Department of Agriculture: Washington, DC, USA, 2015.
- Lucier, G.; BiingHwan, L.; Allshouse, J.; Scott Kantor, L. Factors affecting dry bean consumption in the United States. Veg. Spec. Situat. Outlook 2000, 280, 26–34. [Google Scholar]
- Mitchell, D.C.; Lawrence, F.R.; Hartman, T.J.; Curran, J.M. Consumption of dry beans, peas, and lentils could improve diet quality in the US population. J. Am. Diet. Assoc. 2009, 109, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Winham, D.M.; Armstrong Florian, T.L. Hispanic women in EFNEP have low adherence with dietary guidelines regardless of acculturation level. J. Hunger Environ. Nutr. 2010, 5, 498–509. [Google Scholar] [CrossRef]
- Martínez, A.D. Reconsidering acculturation in dietary change research among Latino immigrants: Challenging the preconditions of US migration. Ethn. Health 2013, 18, 115–135. [Google Scholar] [CrossRef] [PubMed]
- Batres-Marquez, S.P.; Jensen, H.H.; Upton, J. Rice Consumption in the United States: Recent Evidence from Food Consumption Surveys. J. Am. Diet. Assoc. 2009, 109, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Muthayya, S.; Sugimoto, J.D.; Montgomery, S.; Maberly, G.F. An overview of global rice production, supply, trade, and consumption. Ann. N. Y. Acad. Sci. 2014, 1324, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Foster-Powell, K.; Holt, S.H.A.; Brand-Miller, J.C. International table of glycemic index and glycemic load values: 2002. Am. J. Clin. Nutr. 2002, 76, 5–56. [Google Scholar] [PubMed]
- Araya, H.; Pak, N.; Vera, G.; Alviña, M. Digestion rate of legume carbohydrates and glycemic index of legume-based meals. Int. J. Food Sci. Nutr. 2003, 54, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Bornet, F.R.; Costagliola, D.; Rizkalla, S.W.; Blayo, A.; Fontvieille, A.M.; Haardt, M.J.; Letanoux, M.; Tchobroutsky, G.; Slama, G. Insulinemic and glycemic indexes of six starch-rich foods taken alone and in a mixed meal by type 2 diabetics. Am. J. Clin. Nutr. 1987, 45, 588–595. [Google Scholar] [PubMed]
- Sáyago-Ayerdi, S.G.; Tovar, J.; Osorio-Díaz, P.; Paredes-López, O.; Bello-Pérez, L.A. In Vitro Starch Digestibility and predicted glycemic index of corn tortilla, black beans, and tortilla−bean mixture: effect of cold storage. J. Agric. Food Chem. 2005, 53, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Udani, J.K.; Singh, B.B.; Barrett, M.L.; Preuss, H.G. Lowering the glycemic index of white bread using a white bean extract. Nutr. J. 2009, 8, 52. [Google Scholar] [CrossRef] [PubMed]
- Winham, D.M.; Hutchins, A.M.; Melde, C.L. Pinto bean, navy bean, and black-eyed pea consumption do not significantly lower the glycemic response to a high glycemic index treatment in normoglycemic adults. Nutr. Res. 2007, 27, 535–541. [Google Scholar] [CrossRef]
- Greffeuille, V.; Marsset-Baglieri, A.; Molinari, N.; Cassan, D.; Sutra, T.; Avignon, A.; Micard, V. Enrichment of pasta with faba bean does not impact glycemic or insulin response but can enhance satiety feeling and digestive comfort when dried at very high temperature. Food Funct. 2015, 6, 2996–3005. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.V.; Winham, D.M.; Hutchins, A.M. Bean and rice meals reduce postprandial glycemic response in adults with type 2 diabetes: A cross-over study. Nutr. J. 2012, 11. [Google Scholar] [CrossRef] [PubMed]
- Mohan, V.; Spiegelman, D.; Sudha, V.; Gayathri, R.; Hong, B.; Praseena, K.; Anjana, R.M.; Wedick, N.M.; Arumugam, K.; Malik, V.; et al. Effect of brown rice, white rice, and brown rice with legumes on blood glucose and insulin responses in overweight Asian Indians: A randomized controlled trial. Diabetes Technol. Ther. 2014, 16, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Mbanya, J.-C.N.; Mfopou, J.K.; Sobngwi, E.; Mbanya, D.N.S.; Ngogang, J.Y. Metabolic and hormonal effects of five common African diets eaten as mixed meals: The Cameroon Study. Eur. J. Clin. Nutr. 2003, 57, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Tucker, A.J.; Vandermey, J.S.; Robinson, L.E.; Graham, T.E.; Bakovic, M.; Duncan, A.M. Effects of breads of varying carbohydrate quality on postprandial glycaemic, incretin and lipidaemic response after first and second meals in adults with diet-controlled type 2 diabetes. J. Funct. Foods 2014, 6, 116–125. [Google Scholar] [CrossRef]
- Sugiyama, M.; Tang, A.C.; Wakaki, Y.; Koyama, W. Glycemic index of single and mixed meal foods among common Japanese foods with white rice as a reference food. Eur. J. Clin. Nutr. 2003, 57, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Asp, N.G. Classification and methodology of food carbohydrates as related to nutritional effects. Am. J. Clin. Nutr. 1995, 61, 930S–937S. [Google Scholar] [PubMed]
- Schakel, S.; Schauer, R.; Himes, J.; Harnack, L.; Van Heel, N. Development of a glycemic index database for dietary assessment. J. Food Compos. Anal. 2008, 21, S50–S55. [Google Scholar] [CrossRef]
- Venn, B.J.; Mann, J.I. Cereal grains, legumes and diabetes. Eur. J. Clin. Nutr. 2004, 58, 1443–1461. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- Josse, A.R.; Kendall, C.W.C.; Augustin, L.S.A.; Ellis, P.R.; Jenkins, D.J.A. Almonds and postprandial glycemia—A dose-response study. Metabolism 2007, 56, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Panlasigui, L.N.; Panlilio, L.M.; Madrid, J.C. Glycaemic response in normal subjects to five different legumes commonly used in the Philippines. Int. J. Food Sci. Nutr. 1995, 46, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Duh, M.S.; Lefebvre, P.; Fastenau, J.; Piech, C.T.; Waltzman, R.J. Assessing the clinical benefits of erythropoietic agents using area under the hemoglobin change curve. Oncologist 2005, 10, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.J.; Merz, C.N.B.; ScM, F.; Blum, F.C.B.; McBride, F.P.; Eckel, F.R.H.; Schwartz, F.J.S.; Goldberg, A.C.; Shero, F.S.T. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults. Circulation 2013. [Google Scholar] [CrossRef]
- Beutler, E.; Waalen, J. The definition of anemia: What is the lower limit of normal of the blood hemoglobin concentration? Blood 2006, 107, 1747–1750. [Google Scholar] [CrossRef] [PubMed]
- Mollard, R.C.; Luhovyy, B.L.; Panahi, S.; Nunez, M.; Hanley, A.; Anderson, G.H. Regular consumption of pulses for 8 weeks reduces metabolic syndrome risk factors in overweight and obese adults. Br. J. Nutr. 2012, 108, S111–S122. [Google Scholar] [CrossRef] [PubMed]
- Havemeier, S.; Erickson, J.; Slavin, J. Dietary guidance for pulses: The challenge and opportunity to be part of both the vegetable and protein food groups. Ann. N. Y. Acad. Sci. 2017, 1392, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.P.; Simonson, D.C.; DeFronzo, R.A. Menstrual cyclicity has a profound effect on glucose homeostasis. Fertil. Steril. 1989, 52, 204–208. [Google Scholar] [CrossRef]
- Bennal, A.; Kerure, S. Glucose handling during menstrual cycle. Int. J. Reprod. Contracept. Obstet. Gynecol. 2013, 2, 284–287. [Google Scholar] [CrossRef]
| Characteristic | Rice Only Control | Black Beans and White Rice | Chickpeas and White Rice |
|---|---|---|---|
| Total weight (g) | 180.0 | 248.5 | 248.5 |
| Rice (g) | 180.0 | 118.5 | 118.5 |
| Beans (g) | --- | 130.0 | 130.0 |
| Energy (kcal) | 232.0 | 263.0 | 258.0 |
| Carbohydrate (g) | 49.5 | 56.1 | 53.1 |
| Available CHO (g) | 49.5 | 48.6 | 47.6 |
| Fiber (g) | 0.7 | 7.5 | 5.5 |
| Protein (g) | 4.8 | 11.2 | 9.2 |
| Fat (g) | 0.5 | 0.8 | 2.3 |
| Characteristic | Mean ± SEM | Range of Values |
|---|---|---|
| Age (yrs) | 36 ± 4 | 21–58 |
| Weight (kg) | 67.7 ± 2.8 | 55.9–82.0 |
| Height (cm) | 166.8 ± 1.6 | 160.0–180.3 |
| BMI (kg/m2) | 23.3 ± 0.9 | 19.2–28.7 |
| Triglycerides (mg/dL) | 100.8 ± 16.6 | 38–198 |
| Total cholesterol (mg/dL) | 180.2 ± 13.6 | 112–267 |
| LDL (mg/dL) | 108.6 ± 13.2 | 54–183 |
| HDL (mg/dL) | 58.2 ± 3.75 | 41–83 |
| VLDL (mg/dL) | 17.9 ± 2.9 | 7–33 |
| Hemoglobin (g/dL) | 14.4 ± 0.5 | 12.3–16.3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Winham, D.M.; Hutchins, A.M.; Thompson, S.V. Glycemic Response to Black Beans and Chickpeas as Part of a Rice Meal: A Randomized Cross-Over Trial. Nutrients 2017, 9, 1095. https://doi.org/10.3390/nu9101095
Winham DM, Hutchins AM, Thompson SV. Glycemic Response to Black Beans and Chickpeas as Part of a Rice Meal: A Randomized Cross-Over Trial. Nutrients. 2017; 9(10):1095. https://doi.org/10.3390/nu9101095
Chicago/Turabian StyleWinham, Donna M., Andrea M. Hutchins, and Sharon V. Thompson. 2017. "Glycemic Response to Black Beans and Chickpeas as Part of a Rice Meal: A Randomized Cross-Over Trial" Nutrients 9, no. 10: 1095. https://doi.org/10.3390/nu9101095
APA StyleWinham, D. M., Hutchins, A. M., & Thompson, S. V. (2017). Glycemic Response to Black Beans and Chickpeas as Part of a Rice Meal: A Randomized Cross-Over Trial. Nutrients, 9(10), 1095. https://doi.org/10.3390/nu9101095







