Dietary Anthocyanins against Obesity and Inflammation
Abstract
1. Introduction
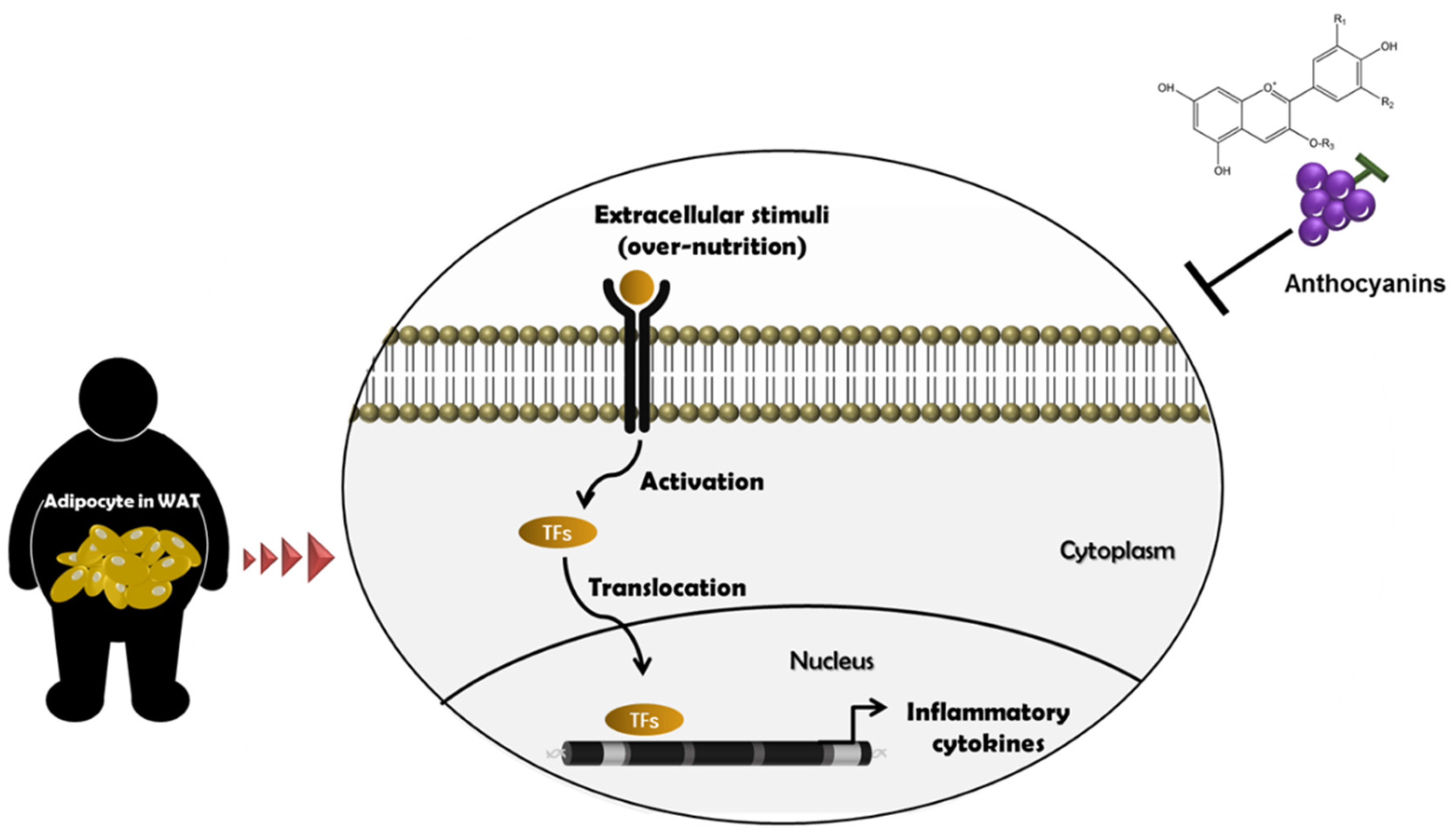
2. Metainflammation and Its Mechanism
3. Establishment of an Obesity Model
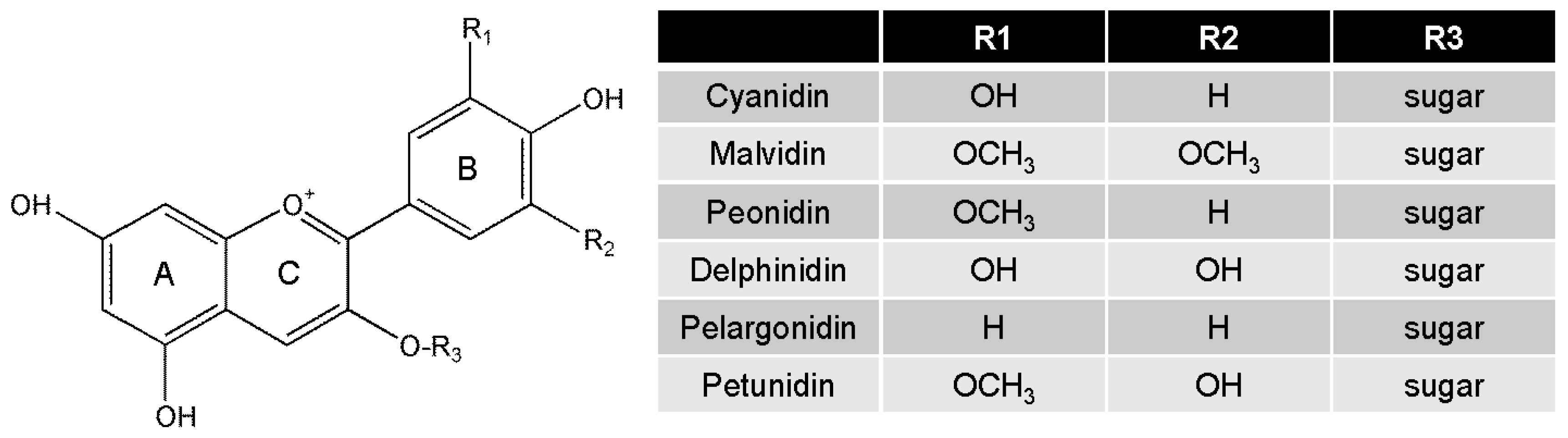
4. Biological Functions of Anthocyanins
5. Bioavailability of Anthocyanins
6. Studies of Dietary Anthocyanins on Regulation of Inflammation and Obesity in Various Models
7. Summary
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviation
| TG | triacylglycerol |
| LDL | low density lipoprotein |
| COX-2 | cyclooxygenase-2 |
| TNF-α | tumor necrosis factor-α |
| IL-6 | interleukin-6 |
| IL-1β | interleukin-1β |
| IL-10 | interleukin-10 |
| iNOS | inducible nitric synthase |
| MCP-1 | monocyte chemoattactant protein-1 |
| NF-κB | nuclear factor kappa B |
| BMI | body mass index |
| CRP | c-reactive protein |
| WC | waist circumstance |
| IRF-3 | interferon regulatory factor 3 |
| AP-1 | activated protein-1 |
| WAT | white adipose tissue |
References
- Adams, K.F.; Schatzkin, A.; Harris, T.B.; Kipnis, V.; Mouw, T.; Ballard-Barbash, R.; Hollenbeck, A.; Leitzmann, M.F. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N. Engl. J. Med. 2006, 355, 763–778. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 2011, 377, 557–567. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Obin, M.S. Obesity and the role of adipose tissue in inflammation and metabolism. Am. J. Clin. Nutr. 2006, 83, S461–S465. [Google Scholar]
- Osborn, O.; Olefsky, J.M. The cellular and signaling networks linking the immune system and metabolism in disease. Nat. Med. 2012, 18, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Gregor, M.F.; Hotamisligil, G.S. Inflammatory mechanisms in obesity. Annu. Rev. Immunol. 2011, 29, 415–445. [Google Scholar] [CrossRef] [PubMed]
- Mraz, M.; Haluzik, M. The role of adipose tissue immune cells in obesity and low-grade inflammation. J. Endocrinol. 2014, 222, R113–R127. [Google Scholar] [CrossRef] [PubMed]
- Brooks-Worrell, B.; Narla, R.; Palmer, J.P. Biomarkers and immune-modulating therapies for type 2 diabetes. Trends Immunol. 2012, 33, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Reilly, S.M.; Chiang, S.H.; Decker, S.J.; Chang, L.; Uhm, M.; Larsen, M.J.; Rubin, J.R.; Mowers, J.; White, N.M.; Hochberg, I.; et al. An inhibitor of the protein kinases tbk1 and ikk-varepsilon improves obesity-related metabolic dysfunctions in mice. Nat. Med. 2013, 19, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Garrow, J.S. Magic bullets now uncontrolled. BMJ 2010, 340, c1351. [Google Scholar] [CrossRef] [PubMed]
- Cefalu, W.T.; Ye, J.; Zuberi, A.; Ribnicky, D.M.; Raskin, I.; Liu, Z.; Wang, Z.Q.; Brantley, P.J.; Howard, L.; Lefevre, M. Botanicals and the metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, S481–S487. [Google Scholar]
- Czemplik, M.; Kulma, A.; Wang, Y.F.; Szopa, J. Therapeutic strategies of plant-derived compounds for diabetes via regulation of monocyte chemoattractant protein-1. Curr. Med. Chem. 2017, 24, 1453–1468. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Giusti, M.M. Anthocyanins: Natural colorants with health-promoting properties. Annu. Rev. Food Sci. Technol. 2010, 1, 163–187. [Google Scholar] [CrossRef] [PubMed]
- Barzilay, J.I.; Abraham, L.; Heckbert, S.R.; Cushman, M.; Kuller, L.H.; Resnick, H.E.; Tracy, R.P. The relation of markers of inflammation to the development of glucose disorders in the elderly: The cardiovascular health study. Diabetes 2001, 50, 2384–2389. [Google Scholar] [CrossRef] [PubMed]
- Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables and Grains, 1st ed.; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- De Pascual-Teresa, S.; Sanchez-Ballesta, M.T. Anthocyanins: From plant to health. Phytochem. Rev. 2008, 7, 281–299. [Google Scholar] [CrossRef]
- Valenti, L.; Riso, P.; Mazzocchi, A.; Porrini, M.; Fargion, S.; Agostoni, C. Dietary anthocyanins as nutritional therapy for nonalcoholic fatty liver disease. Oxid. Med. Cell. Longev. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Slavin, M.; Frankenfeld, C.L. Systematic review of anthocyanins and markers of cardiovascular disease. Nutrients 2016, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ling, W.; Guo, H.; Song, F.; Ye, Q.; Zou, T.; Li, D.; Zhang, Y.; Li, G.; Xiao, Y.; et al. Anti-inflammatory effect of purified dietary anthocyanin in adults with hypercholesterolemia: A randomized controlled trial. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Bastard, J.P.; Maachi, M.; Lagathu, C.; Kim, M.J.; Caron, M.; Vidal, H.; Capeau, J.; Feve, B. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur. Cytokine Netw. 2006, 17, 4–12. [Google Scholar] [PubMed]
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S. The metabolic syndrome and c-reactive protein, fibrinogen, and leukocyte count: Findings from the third national health and nutrition examination survey. Atherosclerosis 2003, 168, 351–358. [Google Scholar] [CrossRef]
- Odegaard, J.I.; Chawla, A. The immune system as a sensor of the metabolic state. Immunity 2013, 38, 644–654. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Wang, X.; Lantier, L.; Lyubetskaya, A.; Eguchi, J.; Kang, S.; Tenen, D.; Roh, H.C.; Kong, X.; Kazak, L.; et al. Irf3 promotes adipose inflammation and insulin resistance and represses browning. J. Clin. Investig. 2016, 126, 2839–2854. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Ismail, A.; Kersten, S. Molecular mechanisms underlying the potential antiobesity-related diseases effect of cocoa polyphenols. Mol. Nutr. Food Res. 2014, 58, 33–48. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A.; Hardie, D.G. Metabolism of inflammation limited by ampk and pseudo-starvation. Nature 2013, 493, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.A.; Scherer, P.E.; Gupta, R.K. Improved methodologies for the study of adipose biology: Insights gained and opportunities ahead. J. Lipid Res. 2014, 55, 605–624. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ojeda, F.J.; Ruperez, A.I.; Gomez-Llorente, C.; Gil, A.; Aguilera, C.M. Cell models and their application for studying adipogenic differentiation in relation to obesity: A review. Int. J. Mol. Sci. 2016, 17, 1040. [Google Scholar] [CrossRef] [PubMed]
- Moseti, D.; Regassa, A.; Kim, W.K. Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int. J. Mol. Sci. 2016, 17, 124. [Google Scholar] [CrossRef] [PubMed]
- Esteve Rafols, M. Adipose tissue: Cell heterogeneity and functional diversity. Endocrinol. Nutr. 2014, 61, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Cawthorn, W.P.; Scheller, E.L.; MacDougald, O.A. Adipose tissue stem cells meet preadipocyte commitment: Going back to the future. J. Lipid Res. 2012, 53, 227–246. [Google Scholar] [CrossRef] [PubMed]
- Armani, A.; Mammi, C.; Marzolla, V.; Calanchini, M.; Antelmi, A.; Rosano, G.M.; Fabbri, A.; Caprio, M. Cellular models for understanding adipogenesis, adipose dysfunction, and obesity. J. Cell. Biochem. 2010, 110, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Lutz, T.A.; Woods, S.C. Overview of animal models of obesity. Curr. Protoc. Pharmacol. 2012. [Google Scholar] [CrossRef]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Frederich, R.C.; Lollmann, B.; Hamann, A.; Napolitano-Rosen, A.; Kahn, B.B.; Lowell, B.B.; Flier, J.S. Expression of ob mrna and its encoded protein in rodents. Impact of nutrition and obesity. J. Clin. Investig. 1995, 96, 1658–1663. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.S.; Liu, Q.; Hammond, H.A.; Dugan, V.; Hey, P.J.; Caskey, C.J.; Hess, J.F. Leptin receptor missense mutation in the fatty zucker rat. Nat. Genet. 1996, 13, 18–19. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, C.; Raun, K.; Yan, F.F.; Larsen, M.O.; Tang-Christensen, M. Laboratory animals as surrogate models of human obesity. Acta Pharmacol. Sin. 2012, 33, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Surwit, R.S.; Feinglos, M.N.; Rodin, J.; Sutherland, A.; Petro, A.E.; Opara, E.C.; Kuhn, C.M.; Rebuffe-Scrive, M. Differential effects of fat and sucrose on the development of obesity and diabetes in c57bl/6j and a/j mice. Metabolism 1995, 44, 645–651. [Google Scholar] [CrossRef]
- Schemmel, R.; Mickelsen, O.; Motawi, K. Conversion of dietary to body energy in rats as affected by strain, sex and ration. J. Nutr. 1972, 102, 1187–1197. [Google Scholar] [PubMed]
- Levin, B.E.; Hogan, S.; Sullivan, A.C. Initiation and perpetuation of obesity and obesity resistance in rats. Am. J. Physiol. 1989, 256, R766–R771. [Google Scholar] [PubMed]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Tsuda, T.; Shiga, K.; Ohshima, K.; Kawakishi, S.; Osawa, T. Inhibition of lipid peroxidation and the active oxygen radical scavenging effect of anthocyanin pigments isolated from Phaseolus vulgaris L. Biochem. Pharmacol. 1996, 52, 1033–1039. [Google Scholar] [CrossRef]
- Tedesco, I.; Luigi Russo, G.; Nazzaro, F.; Russo, M.; Palumbo, R. Antioxidant effect of red wine anthocyanins in normal and catalase-inactive human erythrocytes. J. Nutr. Biochem. 2001, 12, 505–511. [Google Scholar] [CrossRef]
- Isaak, C.K.; Petkau, J.C.; Blewett, H.; Karmin, O.; Siow, Y.L. Lingonberry anthocyanins protect cardiac cells from oxidative-stress-induced apoptosis. Can. J. Physiol. Pharmacol. 2017, 95, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Heo, H.J.; Lee, C.Y. Strawberry and its anthocyanins reduce oxidative stress-induced apoptosis in pc12 cells. J. Agric. Food Chem. 2005, 53, 1984–1989. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Horio, F.; Osawa, T. The role of anthocyanins as an antioxidant under oxidative stress in rats. Biofactors 2000, 13, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Thoppil, R.J.; Bhatia, D.; Barnes, K.F.; Haznagy-Radnai, E.; Hohmann, J.; Darvesh, A.S.; Bishayee, A. Black currant anthocyanins abrogate oxidative stress through nrf2-mediated antioxidant mechanisms in a rat model of hepatocellular carcinoma. Curr. Cancer Drug Targets 2012, 12, 1244–1257. [Google Scholar] [PubMed]
- Afaq, F.; Saleem, M.; Krueger, C.G.; Reed, J.D.; Mukhtar, H. Anthocyanin- and hydrolyzable tannin-rich pomegranate fruit extract modulates mapk and nf-kappab pathways and inhibits skin tumorigenesis in cd-1 mice. Int. J. Cancer 2005, 113, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.W.; Lee, W.S.; Shin, S.C.; Kim, G.Y.; Choi, B.T.; Choi, Y.H. Anthocyanins downregulate lipopolysaccharide-induced inflammatory responses in bv2 microglial cells by suppressing the nf-kappab and akt/mapks signaling pathways. Int. J. Mol. Sci. 2013, 14, 1502–1515. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Klimis-Zacas, D. Anti-inflammatory effect of anthocyanins via modulation of nuclear factor-kappab and mitogen-activated protein kinase signaling cascades. Nutr. Rev. 2015, 73, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Nair, M.G.; Strasburg, G.M.; Chang, Y.C.; Booren, A.M.; Gray, J.I.; DeWitt, D.L. Antioxidant and antiinflammatory activities of anthocyanins and their aglycon, cyanidin, from tart cherries. J. Nat. Prod. 1999, 62, 294–296. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Serraino, I.; Dugo, P.; Di Paola, R.; Mondello, L.; Genovese, T.; Morabito, D.; Dugo, G.; Sautebin, L.; Caputi, A.P.; et al. Protective effects of anthocyanins from blackberry in a rat model of acute lung inflammation. Free Radic. Res. 2003, 37, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.W.; Gong, C.C.; Song, H.F.; Cui, Y.Y. Effects of anthocyanins on the prevention and treatment of cancer. Br. J. Pharmacol. 2017, 174, 1226–1243. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Del Bo, C.; Ciappellano, S.; Riso, P.; Klimis-Zacas, D. Berry fruit consumption and metabolic syndrome. Antioxidants 2016, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Overall, J.; Bonney, S.A.; Wilson, M.; Beermann, A.; Grace, M.H.; Esposito, D.; Lila, M.A.; Komarnytsky, S. Metabolic effects of berries with structurally diverse anthocyanins. Int. J. Mol. Sci. 2017, 18, 422. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Meckling, K.A.; Marcone, M.F.; Kakuda, Y.; Tsao, R. Synergistic, additive, and antagonistic effects of food mixtures on total antioxidant capacities. J. Agric. Food Chem. 2011, 59, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D.J.; Preston, T.; Kroon, P.A.; Botting, N.P.; Kay, C.D. Human metabolism and elimination of the anthocyanin, cyanidin-3-glucoside: A (13)c-tracer study. Am. J. Clin. Nutr. 2013, 97, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, S230–S242. [Google Scholar]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; da Silva Pinto, M. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Temelli, F.; Chen, L. Encapsulation of anthocyanin in liposomes using supercritical carbon dioxide: Effects of anthocyanin and sterol concentrations. J. Func. Food 2017, 34, 159–167. [Google Scholar] [CrossRef]
- Kanokpanont, S.; Yamdech, R.; Aramwit, P. Stability enhancement of mulberry-extracted anthocyanin using alginate/chitosan microencapsulation for food supplement application. Artif. Cells Nanomed. Biotechnol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ju, J.H.; Yoon, H.S.; Park, H.J.; Kim, M.Y.; Shin, H.K.; Park, K.Y.; Yang, J.O.; Sohn, M.S.; Do, M.S. Anti-obesity and antioxidative effects of purple sweet potato extract in 3t3-l1 adipocytes in vitro. J. Med. Food 2011, 14, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Jiang, X.; Xiao, Z.; Yu, L.; Pham, Q.; Sun, J.; Chen, P.; Yokoyama, W.; Yu, L.L.; Luo, Y.S.; et al. Red cabbage microgreens lower circulating low-density lipoprotein (ldl), liver cholesterol, and inflammatory cytokines in mice fed a high-fat diet. J. Agric. Food Chem. 2016, 64, 9161–9171. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Jiang, Z.; Yin, J.; Long, H.; Zheng, X. Anti-obesity effects of artificial planting blueberry (vaccinium ashei) anthocyanin in high-fat diet-treated mice. Int. J. Food Sci. Nutr. 2016, 67, 257–264. [Google Scholar] [CrossRef] [PubMed]
- DeFuria, J.; Bennett, G.; Strissel, K.J.; Perfield, J.W., 2nd; Milbury, P.E.; Greenberg, A.S.; Obin, M.S. Dietary blueberry attenuates whole-body insulin resistance in high fat-fed mice by reducing adipocyte death and its inflammatory sequelae. J. Nutr. 2009, 139, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Tang, Q.; Gao, Z.; Yu, Z.; Song, H.; Zheng, X.; Chen, W. Blueberry and mulberry juice prevent obesity development in c57bl/6 mice. PLoS ONE 2013, 8, e77585. [Google Scholar] [CrossRef] [PubMed]
- Benn, T.; Kim, B.; Park, Y.K.; Wegner, C.J.; Harness, E.; Nam, T.G.; Kim, D.O.; Lee, J.S.; Lee, J.Y. Polyphenol-rich blackcurrant extract prevents inflammation in diet-induced obese mice. J. Nutr. Biochem. 2014, 25, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Yin, J.; Zhang, G.; Long, H.; Zheng, X. Mulberry and cherry anthocyanin consumption prevents oxidative stress and inflammation in diet-induced obese mice. Mol. Nutr. Food Res. 2016, 60, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Farrell, N.J.; Norris, G.H.; Ryan, J.; Porter, C.M.; Jiang, C.; Blesso, C.N. Black elderberry extract attenuates inflammation and metabolic dysfunction in diet-induced obese mice. Br. J. Nutr. 2015, 114, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Wi, H.-R.; Choi, S.; Ha, T.J.; Lee, B.W.; Lee, M. Inhibitory effect of anthocyanin-rich black soybean testa (Glycine max (L.) merr.) on the inflammation-induced adipogenesis in a dio mouse model. J. Funct. Food 2015, 14, 623–633. [Google Scholar] [CrossRef]
- Dragano, N.R.; Marques, A.; Cintra, D.E.; Solon, C.; Morari, J.; Leite-Legatti, A.V.; Velloso, L.A.; Marostica-Junior, M.R. Freeze-dried jaboticaba peel powder improves insulin sensitivity in high-fat-fed mice. Br. J. Nutr. 2013, 110, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Anderson, R.A. An extract of chokeberry attenuates weight gain and modulates insulin, adipogenic and inflammatory signalling pathways in epididymal adipose tissue of rats fed a fructose-rich diet. Br. J. Nutr. 2012, 108, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Seymour, E.M.; Lewis, S.K.; Urcuyo-Llanes, D.E.; Tanone, I.I.; Kirakosyan, A.; Kaufman, P.B.; Bolling, S.F. Regular tart cherry intake alters abdominal adiposity, adipose gene transcription, and inflammation in obesity-prone rats fed a high fat diet. J. Med. Food 2009, 12, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Sorn, S.R.; Park, Y.; Park, H.K. Anthocyanin rich-black soybean testa improved visceral fat and plasma lipid profiles in overweight/obese korean adults: A randomized controlled trial. J. Med. Food 2016, 19, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Azzini, E.; Venneria, E.; Ciarapica, D.; Foddai, M.S.; Intorre, F.; Zaccaria, M.; Maiani, F.; Palomba, L.; Barnaba, L.; Tubili, C.; et al. Effect of red orange juice consumption on body composition and nutritional status in overweight/obese female: A pilot study. Oxid. Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Silveira, J.Q.; Dourado, G.K.; Cesar, T.B. Red-fleshed sweet orange juice improves the risk factors for metabolic syndrome. Int. J. Food Sci. Nutr. 2015, 66, 830–836. [Google Scholar] [CrossRef] [PubMed]
| Advantage | Disadvantage | |||
|---|---|---|---|---|
| In vitro | mouse | mesenchymal stem cells embryonic stem cells primary preadipocytes |
|
|
| human | preadipocytes adipose-derived stem cells |
|
| |
| In vivo | monogenic | ob/ob mice Zucker fatty rats |
|
|
| polygenic | High energy diets-fed mice Diet-induced obesity |
|
| |
| Food Sources | Identified Bioactive Dose of Anthocyanins | Mediators | Inducer | Metabolic Marker | Inflammatory Marker | Ref. |
|---|---|---|---|---|---|---|
| Purple sweet potato | cyanidnin | 3T3-L1 | Stimuli vs. undifferentiated cells | [61] | ||
| (3-caffeylferulysophoroside-5-glucoside) | leptin ↓ | COX-2 ↓ | ||||
| peonidin | adipogenic factors ↓ | MCP-1 ↓ | ||||
| (3-caffeylferulysophoroside-5-glucoside) | IL-6 ↓ | |||||
| Dose: 4.28 μg/mL to 12.84 μg/mL a | ||||||
| Red cabbage microgreen | cyanidin | mice (C57BL/6) | high fat-diet vs. normal-diet for 8 weeks | [62] | ||
| (3-diglucoside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-(sinapoyl)-diglucoside-5-glucosides) | ||||||
| cyanidin | ||||||
| (3-(glucosyl)(sinapoyl)(p-coumaroyl)sophorside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-(glucosyl)(sinapoyl)(feruloyl)sophorside-5-glucoside) | LDL ↓ | CRP ↓ | ||||
| cyanidin | cholesterol ↓ | TNF-α ↓ | ||||
| (3-diferuloylsophoroside-5-glucoside) | TG ↓ | |||||
| cyanidin | ||||||
| (3-(coumaroyl)sophoroside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-(feruloyl)sophoroside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-diferuloylsophoroside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-(sinapoyl)(feruloyl)sophoroside-5-glucoside) | ||||||
| cyanidin | ||||||
| (3-(sinapoyl)(sinapoyl)sophoroside-5-glucoside) | ||||||
| Dose: 139.596 nmol/g b | ||||||
| Blueberry | delphinidins | mice (C57BL/6) | high fat-diet vs. normal diet for 8 weeks | glucose ↓ | TNF-α ↓ IL-6 ↓ MCP-1 ↓ iNOS ↓ IL-10 ↑ CD11c+ | [64] |
| cyanidins | ||||||
| peonidins | ||||||
| malvidins | ||||||
| Dose: 1.29 mg/g b | ||||||
| Blueberry | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 8 weeks | [63] | ||
| (3-galactoside) | ||||||
| cyanidin | ||||||
| (3-arabinoside) | ||||||
| delphinidin | ||||||
| (3-arabinoside) | glucose ↓ | TNF-α ↓ | ||||
| delphinidin | TG ↓ | IL-6 ↓ | ||||
| (3-galactoside) | cholesterol ↓ | |||||
| petunidin | insulin ↓ | |||||
| (3-glucoside) | leptin ↓ | |||||
| petunidin | ||||||
| (3-arabinoside) | ||||||
| malvidin | ||||||
| (3-galactoside) | ||||||
| malvidin | ||||||
| (3-glucoside) | ||||||
| Dose: 50 to 200 μg/g b | ||||||
| Black elderberry | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 16 weeks | [68] | ||
| (3-glucoside) | TG ↓ | MCP-1 ↓ | ||||
| cyanidin | insulin ↓ | TNF-α ↓ | ||||
| (3-sambubioside) | cholesterol ↓ | |||||
| Dose: 3.334, 1.7 μg/g b | ||||||
| Blackcurrant | delphinidin | mice (C57BL/6) | high fat/cholesterol-diet vs. normal diet for 12 weeks | adipogenic genes ↓ | [66] | |
| (3-glucoside) | ||||||
| delphinidin | ||||||
| (3-o-rutinoside) | TNF-α ↓ | |||||
| cyanidin | IL-6 ↓ | |||||
| (3-glucoside) | IL-1β ↓ | |||||
| cyanidin | ||||||
| (3-rutinoside) | ||||||
| Dose: 298.1 μg/g b | ||||||
| Mulberry | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 16 weeks | [67] | ||
| (3-glucoside) | TNF-α ↓ | |||||
| cyanidin | glucose ↓ | IL-6 ↓ | ||||
| (3-rutinoside) | leptin ↓ | iNOS ↓ | ||||
| pelarginidin | NF-κB ↓ | |||||
| (3-glucose) | ||||||
| Dose: 200 μg/g b | ||||||
| Cherry | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 16 weeks | [67] | ||
| (3-2G-glucosylrutinoside) | ||||||
| cyanidin | TNF-α ↓ | |||||
| (3-rutinoside) | glucose ↓ | IL-6 ↓ | ||||
| pelarginidin | leptin ↓ | iNOS ↓ | ||||
| (3-rutinoside) | NF-κB ↓ | |||||
| Dose: 200 μg/g b | ||||||
| Blueberry juice | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 12 weeks | [65] | ||
| (3-galactoside) | ||||||
| cyanidin | ||||||
| (3-arabinoside) | ||||||
| delphinidin | ||||||
| (3-glucoside) | ||||||
| delphinidin | leptin ↓ | |||||
| (3-galactoside) | cholesterol ↓ | TNF-α ↓ | ||||
| delphinidin | adiponectin ↑ | IL-6 ↓ | ||||
| (3-arabinoside) | TG ↓ | |||||
| petunidin | ||||||
| (3-glucoside) | ||||||
| petunidin | ||||||
| (3-arabinoside) | ||||||
| malvidin | ||||||
| (3-galactoside) | ||||||
| malvidin | ||||||
| (3-glucoside) | ||||||
| Dose: 4.09 mg/mL c | ||||||
| Mulberry juice | cyanidin | mice (C57BL/6) | high fat-diet vs. normal diet for 12 weeks | [65] | ||
| (3-glucoside) | ||||||
| cyanidin | ||||||
| (3-rutinoside) | leptin ↓ | TNF-α ↓ | ||||
| pelargonidin | adiponectin ↑ | IL-6 ↓ | ||||
| (3-glucoside) | ||||||
| pelargonidin | ||||||
| (3-rutinoside) | ||||||
| Dose: 21.86 mg/mL c | ||||||
| Black soybean | delphinidin | mice (C57BL/6) | high fat-diet vs. normal diet for 12 weeks | [69] | ||
| (3-glucoside) | ||||||
| cyanidin | ||||||
| (3-glucoside) | ||||||
| petunidin | TG ↓ | TNF-α ↓ | ||||
| (3-glucoside) | cholesterol ↓ | IL-6 ↓ | ||||
| pelargonodin | IL-10↑ | |||||
| (3-glucoside) | ||||||
| peonidin | ||||||
| (3-glucoside) | ||||||
| Dose: 12.48 mg/g b | ||||||
| Jaboticaba peel | delphinidin | swiss inbred mice | high fat-diet vs. normal diet for 6 weeks | insulin ↓ | [70] | |
| (3- O-glycoside) | IL-6 ↓ | |||||
| cyanidin | IL-1β ↓ | |||||
| (3- O-glycoside) | ||||||
| Dose: 259.9, 519.8, 1039.6 μg/g b | ||||||
| Chokeberry | total anthocyanin | Wistar rat | fructose-rich diet vs. normal diet for 6 weeks | glucose ↓ insulin ↓ TG ↓ cholesterol ↓ | TNF-α ↓ | [71] |
| Dose: 10 or 20 mg/kg d | IL-6 ↓ | |||||
| Tart cherry | cyanidin | zucker fatty rats | Spontaneously obese for 90 days | [72] | ||
| (3-sophoroside) | ||||||
| cyanidin | ||||||
| (3-glucosylrutinoside) | ||||||
| cyanidin- | glucose ↓ | |||||
| (3-glucoside) | insulin ↓ | TNF-α ↓ | ||||
| cyanidin | cholesterol ↓ | IL-6 ↓ | ||||
| (3-rutinoside) | TG ↓ | |||||
| peonidin | ||||||
| (3-glucoside) | ||||||
| Pelargonidin | ||||||
| Dose: 0.6598 mg/g b |
| Food Sources | Bioactives Dose of Anthocyanins | Subject Duration | Metabolic Marker | Inflammatory Marker | Ref. |
|---|---|---|---|---|---|
| Black soybean | cyanidin | [73] | |||
| (3-glucosides) | BMI > 23 | ||||
| delphinidin | WC > 90 for male | TG ↓ | TNF-α ↓ | ||
| (3-glucoside) | WC > 85 for female | cholesterol ↓ | MCP-1 ↓ | ||
| petunidin | For 8 weeks | LDL ↓ | |||
| (3-glucoside) | |||||
| 31.48 mg/day | |||||
| Red orange juice | Anthocyanin mixture 250 mg/day | average BMI = 34.4 ± 4.8 for 12 weeks | Δ leptin ↓ | CRP ↓ | [74] |
| Δ adiponectin ↓ | TNF-α ↓ | ||||
| Red-fleshed sweet orange juice | anthocyanin mixture 750 mL/day | age 23–59 BMI 18.5–24.986.4/74.6 | cholesterol ↓ | CRP↓ | [75] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-M.; Yoon, Y.; Yoon, H.; Park, H.-M.; Song, S.; Yeum, K.-J. Dietary Anthocyanins against Obesity and Inflammation. Nutrients 2017, 9, 1089. https://doi.org/10.3390/nu9101089
Lee Y-M, Yoon Y, Yoon H, Park H-M, Song S, Yeum K-J. Dietary Anthocyanins against Obesity and Inflammation. Nutrients. 2017; 9(10):1089. https://doi.org/10.3390/nu9101089
Chicago/Turabian StyleLee, Yoon-Mi, Young Yoon, Haelim Yoon, Hyun-Min Park, Sooji Song, and Kyung-Jin Yeum. 2017. "Dietary Anthocyanins against Obesity and Inflammation" Nutrients 9, no. 10: 1089. https://doi.org/10.3390/nu9101089
APA StyleLee, Y.-M., Yoon, Y., Yoon, H., Park, H.-M., Song, S., & Yeum, K.-J. (2017). Dietary Anthocyanins against Obesity and Inflammation. Nutrients, 9(10), 1089. https://doi.org/10.3390/nu9101089




