Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Data Sources and Searches
2.2. Study Selection
2.3. Data Extraction
2.4. Definition: Adherence to Mediterranean Diet
2.5. Statistical Analysis
3. Results
3.1. Literature Search and Study Characteristics
3.2. Main Outcomes
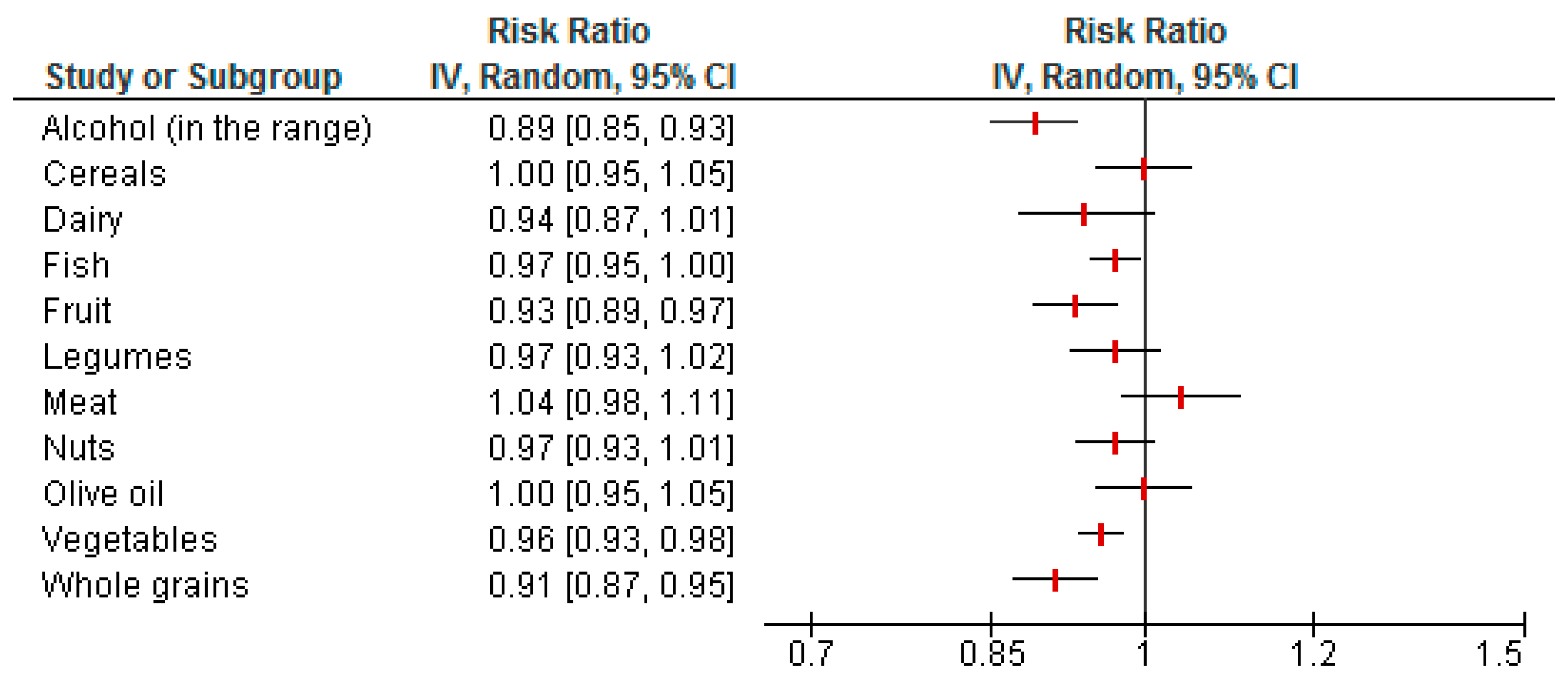
3.3. Food Group Components of the MedD and Risk of Cancer
3.4. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
Appendix A
References
- Stewart, B.W.; Bray, F.; Forman, D.; Ohgaki, H.; Straif, K.; Ullrich, A.; Wild, C.P. Cancer prevention as part of precision medicine: ‘Plenty to be done’. Carcinogenesis 2016, 37, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Tyrovolas, S.; Panagiotakos, D.B. The role of Mediterranean type of diet on the development of cancer and cardiovascular disease, in the elderly: A systematic review. Maturitas 2010, 65, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, Z.; Peckham, C.S.; Ades, A.E. Changing prevalence of juvenile-onset diabetes mellitus. Lancet 1988, 2, 88–90. [Google Scholar] [CrossRef]
- Ligibel, J.A.; Alfano, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Burger, R.A.; Chlebowski, R.T.; Fabian, C.J.; Gucalp, A.; Hershman, D.L.; Hudson, M.M.; et al. American Society of Clinical Oncology position statement on obesity and cancer. J. Clin. Oncol. 2014, 32, 3568–3574. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- WCRF. World Cancer Research Fund International. Continuous Update Project (Cup). 2017. Available online: http://www.wcrf.org/int/research-we-fund/continuous-update-project-cup (accessed on 20 July 2017).
- Heller, M.C.; Keoleian, G.A.; Willett, W.C. Toward a life cycle-based, diet-level framework for food environmental impact and nutritional quality assessment: A critical review. Environ. Sci. Technol. 2013, 47, 12632–12647. [Google Scholar] [CrossRef] [PubMed]
- Milner, J.A. Molecular targets for bioactive food components. J. Nutr. 2004, 134, 2492s–2498s. [Google Scholar] [PubMed]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K.; International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; et al. Alcohol consumption and site-specific cancer risk: A comprehensive dose-response meta-analysis. Br. J. Cancer 2015, 112, 580–593. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc. Nutr. Soc. 2008, 67, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Mullan, F. Seasons of survival: Reflections of a physician with cancer. N. Engl. J. Med. 1985, 313, 270–273. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.E.; Lin, C.C.; Mariotto, A.B.; Siegel, R.L.; Stein, K.D.; Kramer, J.L.; Alteri, R.; Robbins, A.S.; Jemal, A. Cancer treatment and survivorship statistics, 2014. CA Cancer J. Clin. 2014, 64, 252–271. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Rowland, J.H.; Kent, E.E.; Forsythe, L.P.; Loge, J.H.; Hjorth, L.; Glaser, A.; Mattioli, V.; Fossa, S.D. Cancer survivorship research in Europe and the United states: Where have we been, where are we going, and what can we learn from each other? Cancer 2013, 119 (Suppl. 11), 2094–2108. [Google Scholar] [CrossRef] [PubMed]
- Schwedhelm, C.; Boeing, H.; Hoffmann, G.; Aleksandrova, K.; Schwingshackl, L. Effect of diet on mortality and cancer recurrence among cancer survivors: A systematic review and meta-analysis of cohort studies. Nutr. Rev. 2016, 74, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Collins, C.E.; Young, A.F.; Hodge, A. Diet quality is associated with higher nutrient intake and self-rated health in mid-aged women. J. Am. Coll. Nutr. 2008, 27, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R.; Gross, M.D.; Tapsell, L.C. Food synergy: An operational concept for understanding nutrition. Am. J. Clin. Nutr. 2009, 89, 1543S–1548S. [Google Scholar] [CrossRef] [PubMed]
- Edefonti, V.; Randi, G.; La Vecchia, C.; Ferraroni, M.; Decarli, A. Dietary patterns and breast cancer: A review with focus on methodological issues. Nutr. Rev. 2009, 67, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Keys, A. Mediterranean diet and public health: Personal reflections. Am. J. Clin. Nutr. 1995, 61, 1321S–1323S. [Google Scholar] [PubMed]
- Keys, A.; Menotti, A.; Karvonen, M.J.; Aravanis, C.; Blackburn, H.; Buzina, R.; Djordjevic, B.S.; Dontas, A.S.; Fidanza, F.; Keys, M.H. The diet and 15-year death rate in the Seven countries study. Am. J. Epidemiol. 1986, 124, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Missbach, B.; Konig, J.; Hoffmann, G. Adherence to a Mediterranean diet and risk of diabetes: A systematic review and meta-analysis. Public Health Nutr. 2015, 18, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Mediterranean dietary pattern, inflammation and endothelial function: A systematic review and meta-analysis of intervention trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Fidanza, F.; Alberti, A.; Fruttini, D. The Nicotera diet: The reference Italian Mediterranean diet. World Rev. Nutr. Diet. 2005, 95, 115–121. [Google Scholar] [PubMed]
- Fidanza, F.; Alberti, A.; Lanti, M.; Menotti, A. Mediterranean Adequacy Index: Correlation with 25-year mortality from coronary heart disease in the Seven countries study. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 254–258. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Monounsaturated fatty acids, olive oil and health status: A systematic review and meta-analysis of cohort studies. Lipids Health Dis. 2014, 13, 154. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: A systematic review and meta-analysis of observational studies. Int. J. Cancer 2014, 135, 1884–1897. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Adherence to mediterranean diet and risk of cancer: An updated systematic review and meta-analysis of observational studies. Cancer Med. 2015, 4, 1933–1947. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014, 17, 2769–2782. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Does a Mediterranean-type diet reduce cancer risk? Curr. Nutr. Rep. 2016, 5, 9–17. [Google Scholar] [CrossRef] [PubMed]
- De Lorgeril, M.; Salen, P.; Martin, J.L.; Monjaud, I.; Boucher, P.; Mamelle, N. Mediterranean dietary pattern in a randomized trial: Prolonged survival and possible reduced cancer rate. Arch. Intern. Med. 1998, 158, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Toledo, E.; Salas-Salvado, J.; Donat-Vargas, C.; Buil-Cosiales, P.; Estruch, R.; Ros, E.; Corella, D.; Fito, M.; Hu, F.B.; Aros, F.; et al. Mediterranean diet and invasive breast cancer risk among women at high cardiovascular risk in the PREDIMED trial: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1752–1760. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvado, J. Primary prevention of cardiovascular disease with a mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- De Lorgeril, M.; Renaud, S.; Mamelle, N.; Salen, P.; Martin, J.L.; Monjaud, I.; Guidollet, J.; Touboul, P.; Delaye, J. Mediterranean alpha-linolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 1994, 343, 1454–1459. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Hu, F.B.; McCullough, M.L.; Newby, P.K.; Willett, W.C.; Holmes, M.D. Diet quality is associated with the risk of estrogen receptor-negative breast cancer in postmenopausal women. J. Nutr. 2006, 136, 466–472. [Google Scholar] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the Mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the MedDietscore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Menotti, A.; Alberti-Fidanza, A.; Fidanza, F.; Lanti, M.; Fruttini, D. Factor analysis in the identification of dietary patterns and their predictive role in morbid and fatal events. Public Health Nutr. 2012, 15, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Murtaugh, M.A.; Sweeney, C.; Giuliano, A.R.; Herrick, J.S.; Hines, L.; Byers, T.; Baumgartner, K.B.; Slattery, M.L. Diet patterns and breast cancer risk in hispanic and non-Hispanic white women: The Four-Corners Breast Cancer Study. Am. J. Clin. Nutr. 2008, 87, 978–984. [Google Scholar] [PubMed]
- Cottet, V.; Touvier, M.; Fournier, A.; Touillaud, M.S.; Lafay, L.; Clavel-Chapelon, F.; Boutron-Ruault, M.C. Postmenopausal breast cancer risk and dietary patterns in the E3N-EPIC prospective cohort study. Am. J. Epidemiol. 2009, 170, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Bessaoud, F.; Tretarre, B.; Daures, J.P.; Gerber, M. Identification of dietary patterns using two statistical approaches and their association with breast cancer risk: A case-control study in Southern France. Ann. Epidemiol. 2012, 22, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Boldo, E.; Pérez-Gómez, B.; Lope, V.; Altzibar, J.M.; Martín, V.; Castaño-Vinyals, G.; Guevara, M.; Dierssen-Sotos, T.; Tardón, A.; et al. Adherence to the Western, Prudent and Mediterranean dietary patterns and breast cancer risk: MCC-Spain study. Maturitas 2017, 103, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Boldo, E.; Amiano, P.; Castaño-Vinyals, G.; Aragonés, N.; Gómez-Acebo, I.; Peiró, R.; Jimenez-Moleón, J.J.; Alguacil, J.; Tardón, A.; et al. Mediterranean dietary pattern is associated to low risk of aggressive prostate cancer: MCC-Spain study. J. Urol. 2017. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey, S.G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Stovold, E.; Beecher, D.; Foxlee, R.; Noel-Storr, A. Study flow diagrams in cochrane systematic review updates: An adapted PRISMA flow diagram. Syst. Rev. 2014, 3, 54. [Google Scholar] [CrossRef] [PubMed]
- Anic, G.M.; Park, Y.; Subar, A.F.; Schap, T.E.; Reedy, J. Index-based dietary patterns and risk of lung cancer in the NIH-AARP Diet and Health Study. Eur. J. Clin. Nutr. 2016, 70, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Dugue, P.A.; Hodge, A.M.; Brinkman, M.T.; Bassett, J.K.; Shivappa, N.; Hebert, J.R.; Hopper, J.L.; English, D.R.; Milne, R.L.; Giles, G.G. Association between selected dietary scores and the risk of urothelial cell carcinoma: A prospective cohort study. Int. J. Cancer 2016, 139, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Haridass, V. Diet Quality Scores and Risk of Incident Breast Cancer in the California Teachers Study 2015. (Dissertation). Available online: http://escholarship.Org/uc/item/87t2942b (accessed on 25 July 2017).
- Hirko, K.A.; Willett, W.C.; Hankinson, S.E.; Rosner, B.A.; Beck, A.H.; Tamimi, R.M.; Eliassen, A.H. Healthy dietary patterns and risk of breast cancer by molecular subtype. Breast Cancer Res Treat. 2016, 155, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.M.; Bassett, J.K.; Shivappa, N.; Hebert, J.R.; English, D.R.; Giles, G.G.; Severi, G. Dietary Inflammatory Index, Mediterranean diet score, and lung cancer: A prospective study. Cancer Causes Control 2016, 27, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.; Harmon, B.E.; Ollberding, N.J.; Wilkens, L.R.; Monroe, K.R.; Kolonel, L.N.; Le Marchand, L.; Boushey, C.J.; Maskarinec, G. Among 4 diet quality indexes, only the Alternate Mediterranean diet score is associated with better colorectal cancer survival and only in african american women in the Multiethnic cohort. J. Nutr. 2016, 146, 1746–1755. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Hakansson, N.; Wolk, A. Healthy dietary patterns and incidence of biliary tract and gallbladder cancer in a prospective study of women and men. Eur. J. Cancer 2017, 70, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Lassale, C.; Gunter, M.J.; Romaguera, D.; Peelen, L.M.; Van der Schouw, Y.T.; Beulens, J.W.; Freisling, H.; Muller, D.C.; Ferrari, P.; Huybrechts, I.; et al. Diet quality scores and prediction of all-cause, cardiovascular and cancer mortality in a pan-european cohort study. PLoS ONE 2016, 11, e0159025. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Shivappa, N.; Hebert, J.R.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G.; Gnagnarella, P. Dietary inflammatory index and risk of lung cancer and other respiratory conditions among heavy smokers in the COMOS screening study. Eur. J. Nutr. 2016, 55, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Molina-Montes, E.; Sanchez, M.J.; Buckland, G.; Bueno-de-Mesquita, H.B.; Weiderpass, E.; Amiano, P.; Wark, P.A.; Kuhn, T.; Katzke, V.; Huerta, J.M.; et al. Mediterranean diet and risk of pancreatic cancer in the European Prospective Investigation into Cancer and Nutrition cohort. Br. J. Cancer 2017, 116, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Boushey, C.J.; Wilkens, L.R.; Haiman, C.A.; Le Marchand, L. High-quality diets associate with reduced risk of colorectal cancer: Analyses of diet quality indexes in the Multiethnic cohort. Gastroenterology 2017, 153, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Van den Brandt, P.A.; Schulpen, M. Mediterranean diet adherence and risk of postmenopausal breast cancer: Results of a cohort study and meta-analysis. Int. J. Cancer 2017, 140, 2220–2231. [Google Scholar] [CrossRef] [PubMed]
- Vargas, A.J.; Neuhouser, M.L.; George, S.M.; Thomson, C.A.; Ho, G.Y.; Rohan, T.E.; Kato, I.; Nassir, R.; Hou, L.; Manson, J.E. Diet quality and colorectal cancer risk in the Women’s Health Initiative observational study. Am. J. Epidemiol. 2016, 184, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Whalen, K.A.; Judd, S.; McCullough, M.L.; Flanders, W.D.; Hartman, T.J.; Bostick, R.M. Paleolithic and Mediterranean diet pattern scores are inversely associated with all-cause and cause-specific mortality in adults. J. Nutr. 2017, 147, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Butler, L.M.; Wu, A.H.; Wang, R.; Koh, W.P.; Yuan, J.M.; Yu, M.C. A vegetable-fruit-soy dietary pattern protects against breast cancer among postmenopausal Singapore Chinese women. Am. J. Clin. Nutr. 2010, 91, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.; Evans, C.; Cade, J.; Hancock, N.; Greenwood, D. The Mediterranean diet and risk of colorectal cancer in the UK Women’s cohort study. Int. J. Epidemiol. 2017. [Google Scholar] [CrossRef]
- Askari, F.; Beyzaei, B.; Tehrani, A.; Parizi, M.K.; Mirshekarlou, E.N.; Rashidkhani, B. Adherence to Mediterranean-style dietary pattern and risk of prostate cancer: A case-control study in Iran. Pak. J. Nutr. 2016, 15, 305–311. [Google Scholar]
- Campagna, M.; Cocco, P.; Zucca, M.; Angelucci, E.; Gabbas, A.; Latte, G.C.; Uras, A.; Rais, M.; Sanna, S.; Ennas, M.G. Risk of lymphoma subtypes and dietary habits in a Mediterranean area. Cancer Epidemiol. 2015, 39, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Giraldi, L.; Panic, N.; Cadoni, G.; Boccia, S.; Leoncini, E. Association between Mediterranean diet and head and neck cancer: Results of a large case-control study in Italy. Eur. J. Cancer Prev. 2017, 26, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Rosato, V.; Guercio, V.; Bosetti, C.; Negri, E.; Serraino, D.; Giacosa, A.; Montella, M.; La Vecchia, C.; Tavani, A. Mediterranean diet and colorectal cancer risk: A pooled analysis of three Italian case-control studies. Br. J. Cancer 2016, 115, 862–865. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, J.; Giraldi, L.; Arzani, D.; Pastorino, R.; Biondi, A.; Persiani, R.; Boccia, S.; Leoncini, E. Adherence to Mediterranean diet and risk of gastric cancer: Results of a case-control study in Italy. Eur. J. Cancer. Prev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Turati, F.; Bravi, F.; Polesel, J.; Bosetti, C.; Negri, E.; Garavello, W.; Taborelli, M.; Serraino, D.; Libra, M.; Montella, M.; et al. Adherence to the Mediterranean diet and nasopharyngeal cancer risk in Italy. Cancer Causes Control 2017, 28, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lin, X.L.; Fan, Y.Y.; Liu, Y.T.; Zhang, X.L.; Lu, Y.K.; Xu, C.H.; Chen, Y.M. Diet quality scores and risk of nasopharyngeal carcinoma in Chinese adults: A case-control study. Nutrients 2016, 8, 112. [Google Scholar] [CrossRef] [PubMed]
- Knoops, K.T.; de Groot, L.C.; Kromhout, D.; Perrin, A.E.; Moreiras-Varela, O.; Menotti, A.; van Staveren, W.A. Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: The HALE project. JAMA 2004, 292, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Tognon, G.; Nilsson, L.M.; Lissner, L.; Johansson, I.; Hallmans, G.; Lindahl, B.; Winkvist, A. The Mediterranean diet score and mortality are inversely associated in adults living in the subarctic region. J. Nutr. 2012, 142, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Nkondjock, A.; Ghadirian, P. Diet quality and BRCA-associated breast cancer risk. Breast Cancer Res. Treat. 2007, 103, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Ax, E.; Garmo, H.; Grundmark, B.; Bill-Axelson, A.; Holmberg, L.; Becker, W.; Zethelius, B.; Cederholm, T.; Sjogren, P. Dietary patterns and prostate cancer risk: Report from the population based ULSAM cohort study of Swedish men. Nutr. Cancer 2014, 66, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Bamia, C.; Lagiou, P.; Buckland, G.; Grioni, S.; Agnoli, C.; Taylor, A.J.; Dahm, C.C.; Overvad, K.; Olsen, A.; Tjonneland, A.; et al. Mediterranean diet and colorectal cancer risk: Results from a European cohort. Eur. J. Epidemiol. 2013, 28, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Couto, E.; Boffetta, P.; Lagiou, P.; Ferrari, P.; Buckland, G.; Overvad, K.; Dahm, C.C.; Tjonneland, A.; Olsen, A.; Clavel-Chapelon, F.; et al. Mediterranean dietary pattern and cancer risk in the EPIC cohort. Br. J. Cancer 2011, 104, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; Travier, N.; Cottet, V.; Gonzalez, C.A.; Lujan-Barroso, L.; Agudo, A.; Trichopoulou, A.; Lagiou, P.; Trichopoulos, D.; Peeters, P.H.; et al. Adherence to the Mediterranean diet and risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition cohort study. Int. J. Cancer 2013, 132, 2918–2927. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; Agudo, A.; Travier, N.; Huerta, J.M.; Cirera, L.; Tormo, M.J.; Navarro, C.; Chirlaque, M.D.; Moreno-Iribas, C.; Ardanaz, E.; et al. Adherence to the Mediterranean diet reduces mortality in the Spanish cohort of the European Prospective Investigation into Cancer and Nutrition (Epic-spain). Br. J. Nutr. 2011, 106, 1581–1591. [Google Scholar] [CrossRef] [PubMed]
- Couto, E.; Sandin, S.; Lof, M.; Ursin, G.; Adami, H.O.; Weiderpass, E. Mediterranean dietary pattern and risk of breast cancer. PLoS ONE 2013, 8, e55374. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Guillen-Grima, F.; De Irala, J.; Ruiz-Canela, M.; Bes-Rastrollo, M.; Beunza, J.J.; Lopez del Burgo, C.; Toledo, E.; Carlos, S.; Sanchez-Villegas, A. The Mediterranean diet is associated with a reduction in premature mortality among middle-aged adults. J. Nutr. 2012, 142, 1672–1678. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, C.; Turati, F.; Dal Pont, A.; Ferraroni, M.; Polesel, J.; Negri, E.; Serraino, D.; Talamini, R.; La Vecchia, C.; Zeegers, M.P. The role of Mediterranean diet on the risk of pancreatic cancer. Br. J. Cancer 2013, 109, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Moller, E.; Galeone, C.; Andersson, T.M.; Bellocco, R.; Adami, H.O.; Andren, O.; Gronberg, H.; La Vecchia, C.; Mucci, L.A.; Balter, K. Mediterranean diet score and prostate cancer risk in a Swedish population-based case-control study. J. Nutr. Sci. 2013, 2, e15. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; Agudo, A.; Lujan, L.; Jakszyn, P.; Bueno-de-Mesquita, H.B.; Palli, D.; Boeing, H.; Carneiro, F.; Krogh, V.; Sacerdote, C.; et al. Adherence to a Mediterranean diet and risk of gastric adenocarcinoma within the European Prospective Investigation into Cancer and Nutrition cohort study. Am. J. Clin. Nutr. 2010, 91, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Praud, D.; Bertuccio, P.; Bosetti, C.; Turati, F.; Ferraroni, M.; La Vecchia, C. Adherence to the Mediterranean diet and gastric cancer risk in Italy. Int. J. Cancer 2014, 134, 2935–2941. [Google Scholar] [CrossRef] [PubMed]
- Cade, J.E.; Taylor, E.F.; Burley, V.J.; Greenwood, D.C. Does the Mediterranean dietary pattern or the Healthy Diet Index influence the risk of breast cancer in a large British cohort of women? Eur. J Clin. Nutr. 2011, 65, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Lagiou, P.; Trichopoulos, D.; Sandin, S.; Lagiou, A.; Mucci, L.; Wolk, A.; Weiderpass, E.; Adami, H.O. Mediterranean dietary pattern and mortality among young women: A cohort study in Sweden. Br. J. Nutr. 2006, 96, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.B.; Subar, A.F.; Peters, U.; Weissfeld, J.L.; Bresalier, R.S.; Risch, A.; Schatzkin, A.; Hayes, R.B. Adherence to the USDA food guide, DASH eating plan, and Mediterranean dietary pattern reduces risk of colorectal adenoma. J. Nutr. 2007, 137, 2443–2450. [Google Scholar] [PubMed]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Stanczyk, F.Z.; Pike, M.C. Dietary patterns and breast cancer risk in Asian American women. Am. J. Clin. Nutr. 2009, 89, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Hu, F.B.; Wu, K.; Chiuve, S.E.; Fuchs, C.S.; Giovannucci, E. The Mediterranean and Dietary Approaches to Stop Hypertension (DASH) diets and colorectal cancer. Am. J. Clin. Nutr. 2010, 92, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Kenfield, S.A.; DuPre, N.; Richman, E.L.; Stampfer, M.J.; Chan, J.M.; Giovannucci, E.L. Mediterranean diet and prostate cancer risk and mortality in the Health Professionals Follow-up Study. Eur. Urol. 2014, 65, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Q.; Park, Y.; Wu, J.W.; Ren, J.S.; Goldstein, A.M.; Taylor, P.R.; Hollenbeck, A.R.; Freedman, N.D.; Abnet, C.C. Index-based dietary patterns and risk of esophageal and gastric cancer in a large cohort study. Clin. Gastroenterol. Hepatol. 2013, 11, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Reedy, J.; Mitrou, P.N.; Krebs-Smith, S.M.; Wirfalt, E.; Flood, A.; Kipnis, V.; Leitzmann, M.; Mouw, T.; Hollenbeck, A.; Schatzkin, A.; et al. Index-based dietary patterns and risk of colorectal cancer: The NIH-AARP Diet and Health Study. Am. J. Epidemiol. 2008, 168, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Mitrou, P.N.; Kipnis, V.; Thiebaut, A.C.; Reedy, J.; Subar, A.F.; Wirfalt, E.; Flood, A.; Mouw, T.; Hollenbeck, A.R.; Leitzmann, M.F.; et al. Mediterranean dietary pattern and prediction of all-cause mortality in a US population: Results from the NIH-AARP Diet and Health Study. Arch. Int. Med. 2007, 167, 2461–2468. [Google Scholar] [CrossRef] [PubMed]
- Samoli, E.; Lagiou, A.; Nikolopoulos, E.; Lagogiannis, G.; Barbouni, A.; Lefantzis, D.; Trichopoulos, D.; Brennan, P.; Lagiou, P. Mediterranean diet and upper aerodigestive tract cancer: The Greek segment of the Alcohol-Related cancers and Genetic Susceptibility in Europe study. Br. J. Nutr. 2010, 104, 1369–1374. [Google Scholar] [CrossRef] [PubMed]
- Bosire, C.; Stampfer, M.J.; Subar, A.F.; Park, Y.; Kirkpatrick, S.I.; Chiuve, S.E.; Hollenbeck, A.R.; Reedy, J. Index-based dietary patterns and the risk of prostate cancer in the NIH-AARP Diet and Health Study. Am. J. Epidemiol. 2013, 177, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Cottet, V.; Bonithon-Kopp, C.; Kronborg, O.; Santos, L.; Andreatta, R.; Boutron-Ruault, M.C.; Faivre, J. Dietary patterns and the risk of colorectal adenoma recurrence in a European intervention trial. Eur. J. Cancer Prev. 2005, 14, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Demetriou, C.A.; Hadjisavvas, A.; Loizidou, M.A.; Loucaides, G.; Neophytou, I.; Sieri, S.; Kakouri, E.; Middleton, N.; Vineis, P.; Kyriacou, K. The Mediterranean dietary pattern and breast cancer risk in Greek-cypriot women: A case-control study. BMC Cancer 2012, 12, 113. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, C.; Gallus, S.; Trichopoulou, A.; Talamini, R.; Franceschi, S.; Negri, E.; La Vecchia, C. Influence of the Mediterranean diet on the risk of cancers of the upper aerodigestive tract. Cancer Epidemiol. Biomark. Prev. 2003, 12, 1091–1094. [Google Scholar]
- Kontou, N.; Psaltopoulou, T.; Soupos, N.; Polychronopoulos, E.; Xinopoulos, D.; Linos, A.; Panagiotakos, D.B. Metabolic syndrome and colorectal cancer: The protective role of Mediterranean diet-a case-control study. Angiology 2012, 63, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; Ros, M.M.; Roswall, N.; Bueno-de-Mesquita, H.B.; Travier, N.; Tjonneland, A.; Kiemeney, L.A.; Sacerdote, C.; Tumino, R.; Ljungberg, B.; et al. Adherence to the Mediterranean diet and risk of bladder cancer in the EPIC cohort study. Int. J. Cancer 2014, 134, 2504–2511. [Google Scholar] [CrossRef] [PubMed]
- Cuenca-Garcia, M.; Artero, E.G.; Sui, X.; Lee, D.C.; Hebert, J.R.; Blair, S.N. Dietary indices, cardiovascular risk factors and mortality in middle-aged adults: Findings from the Aerobics Center Longitudinal Study. Ann. Epdemiol. 2014, 24, 297.e2–303.e2. [Google Scholar] [CrossRef] [PubMed]
- George, S.M.; Ballard-Barbash, R.; Manson, J.E.; Reedy, J.; Shikany, J.M.; Subar, A.F.; Tinker, L.F.; Vitolins, M.; Neuhouser, M.L. Comparing indices of diet quality with chronic disease mortality risk in postmenopausal women in the Women’s Health Initiative Observational Study: Evidence to inform national dietary guidance. Am. J. Epdemiol. 2014, 180, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Maisonneuve, P.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G. Red meat, Mediterranean diet and lung cancer risk among heavy smokers in the COSMOS screening study. Ann. Oncol. 2013, 24, 2606–2611. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Kashambwa, R.; Sato, K.; Chiuve, S.E.; Fuchs, C.S.; Wu, K.; Giovannucci, E.; Ogino, S.; Hu, F.B.; Meyerhardt, J.A. Post diagnosis diet quality and colorectal cancer survival in women. PLoS ONE 2014, 9, e115377. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Willett, W.C.; Fung, T.; Rosner, B.; Holmes, M.D. Diet quality indices and postmenopausal breast cancer survival. Nutr. Cancer 2011, 63, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Q.; Park, Y.; McGlynn, K.A.; Hollenbeck, A.R.; Taylor, P.R.; Goldstein, A.M.; Freedman, N.D. Index-based dietary patterns and risk of incident hepatocellular carcinoma and mortality from chronic liver disease in a prospective study. Hepatology 2014, 60, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Q.; Park, Y.; Wu, J.W.; Goldstein, A.M.; Taylor, P.R.; Hollenbeck, A.R.; Freedman, N.D.; Abnet, C.C. Index-based dietary patterns and risk of head and neck cancer in a large prospective study. Am. J. Clin. Nutr. 2014, 99, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, E.; Rodriguez-Artalejo, F.; Li, T.Y.; Fung, T.T.; Li, S.; Willett, W.C.; Rimm, E.B.; Hu, F.B. The Mediterranean-style dietary pattern and mortality among men and women with cardiovascular disease. Am. J. Clin. Nutr. 2014, 99, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Reedy, J.; Krebs-Smith, S.M.; Miller, P.E.; Liese, A.D.; Kahle, L.L.; Park, Y.; Subar, A.F. Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. J. Nutr. 2014, 144, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Harmon, B.E.; Boushey, C.J.; Shvetsov, Y.B.; Ettienne, R.; Reedy, J.; Wilkens, L.R.; Le Marchand, L.; Henderson, B.E.; Kolonel, L.N. Associations of key diet-quality indexes with mortality in the Multiethnic Cohort: The Dietary Patterns Methods Project. Am. J. Clin. Nutr. 2015, 101, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Vormund, K.; Braun, J.; Rohrmann, S.; Bopp, M.; Ballmer, P.; Faeh, D. Mediterranean diet and mortality in Switzerland: An alpine paradox? Eur. J. Nutr. 2015, 54, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Poole, E.M.; Terry, K.L.; Fung, T.T.; Rosner, B.A.; Willett, W.C.; Tworoger, S.S. A prospective cohort study of dietary indices and incidence of epithelial ovarian cancer. J. Ovarian Res. 2014, 7, 112. [Google Scholar] [CrossRef] [PubMed]
- Castello, A.; Pollan, M.; Buijsse, B.; Ruiz, A.; Casas, A.M.; Baena-Canada, J.M.; Lope, V.; Antolin, S.; Ramos, M.; Munoz, M.; et al. Spanish Mediterranean diet and other dietary patterns and breast cancer risk: Case-control EPIGEICAM study. Br. J. Cancer 2014, 111, 1454–1462. [Google Scholar] [CrossRef] [PubMed]
- Dalvi, T.B.; Canchola, A.J.; Horn-Ross, P.L. Dietary patterns, Mediterranean diet, and endometrial cancer risk. Cancer Causes Control 2007, 18, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Biondi, A.; Galvano, F.; Mistretta, A.; Marventano, S.; Buscemi, S.; Drago, F.; Basile, F. Factors associated with colorectal cancer in the context of the Mediterranean diet: A case-control study. Nutr. Cancer 2014, 66, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Filomeno, M.; Bosetti, C.; Garavello, W.; Levi, F.; Galeone, C.; Negri, E.; La Vecchia, C. The role of a Mediterranean diet on the risk of oral and pharyngeal cancer. Br. J. Cancer 2014, 111, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Mourouti, N.; Kontogianni, M.D.; Papavagelis, C.; Plytzanopoulou, P.; Vassilakou, T.; Malamos, N.; Linos, A.; Panagiotakos, D.B. Adherence to the Mediterranean diet is associated with lower likelihood of breast cancer: A case-control study. Nutr. Cancer 2014, 66, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Pot, G.K.; Stephen, A.M.; Dahm, C.C.; Key, T.J.; Cairns, B.J.; Burley, V.J.; Cade, J.E.; Greenwood, D.C.; Keogh, R.H.; Bhaniani, A.; et al. Dietary patterns derived with multiple methods from food diaries and breast cancer risk in the UK Dietary Cohort Consortium. Eur. J. Clin. Nutr. 2014, 68, 1353–1358. [Google Scholar] [CrossRef] [PubMed]
- Turati, F.; Trichopoulos, D.; Polesel, J.; Bravi, F.; Rossi, M.; Talamini, R.; Franceschi, S.; Montella, M.; Trichopoulou, A.; La Vecchia, C.; et al. Mediterranean diet and hepatocellular carcinoma. J. Hepatol. 2014, 60, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Whalen, K.A.; McCullough, M.; Flanders, W.D.; Hartman, T.J.; Judd, S.; Bostick, R.M. Paleolithic and Mediterranean diet pattern scores and risk of incident, sporadic colorectal adenomas. Am. J. Epidemiol. 2014, 180, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- George, S.M.; Ballard, R.; Shikany, J.M.; Crane, T.E.; Neuhouser, M.L. A prospective analysis of diet quality and endometrial cancer among 84,415 postmenopausal women in the Women’s Health Initiative. Ann. Epidemiol. 2015, 25, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Filomeno, M.; Bosetti, C.; Bidoli, E.; Levi, F.; Serraino, D.; Montella, M.; La Vecchia, C.; Tavani, A. Mediterranean diet and risk of endometrial cancer: A pooled analysis of three Italian case-control studies. Br. J. Cancer 2015, 112, 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef] [PubMed]
- Benetou, V.; Trichopoulou, A.; Orfanos, P.; Naska, A.; Lagiou, P.; Boffetta, P.; Trichopoulos, D.; Cohort, G.E. Conformity to traditional Mediterranean diet and cancer incidence: The Greek EPIC cohort. Br. J. Cancer 2008, 99, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Muller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384s–392s. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective; AICR: Washington, DC, USA, 2007. [Google Scholar]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Lau, R.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dietary fibre, whole grains, and risk of colorectal cancer: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2011, 343, d6617. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.R.; Abar, L.; Chan, D.; Vingeliene, S.; Polemiti, E.; Stevens, C.; Greenwood, D.; Norat, T. Foods and beverages and colorectal cancer risk: A systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR Continuous Update Project. Ann. Oncol. 2017, 28, 1788–1802. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Navarro Rosenblatt, D.A.; Chan, D.S.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy products, calcium, and prostate cancer risk: A systematic review and meta-analysis of cohort studies. Am. J. Clin. Nutr. 2015, 101, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Schwedhelm, C.; Kalle-Uhlmann, T.; Missbach, B.; Knuppel, S.; Boeing, H. Consumption of dairy products in relation to changes in anthropometric variables in adult populations: A systematic review and meta-analysis of cohort studies. PLoS ONE 2016, 11, e0157461. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.M.; Knuppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.G.; Sun, J.W.; Yang, Y.; Ma, X.; Wang, Y.Y.; Xiang, Y.B. Fish consumption and all-cause mortality: A meta-analysis of cohort studies. Eur. J. Clin. Nutr. 2016, 70, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Caygill, C.P.; Hill, M.J. Fish, n-3 fatty acids and human colorectal and breast cancer mortality. Eur. J. Cancer Prev. 1995, 4, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Szymanski, K.M.; Wheeler, D.C.; Mucci, L.A. Fish consumption and prostate cancer risk: A review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Lampousi, A.M.; Knuppel, S.; Iqbal, K.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of all-cause mortality: A systematic review and meta-analysis of prospective studies. Am. J. Clin. Nutr. 2017, 105, 1462–1473. [Google Scholar] [CrossRef] [PubMed]
- Tavani, A.; Pelucchi, C.; Parpinel, M.; Negri, E.; Franceschi, S.; Levi, F.; La Vecchia, C. N-3 polyunsaturated fatty acid intake and cancer risk in Italy and Switzerland. Int. J. Cancer 2003, 105, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Chapkin, R.S.; Davidson, L.A.; Ly, L.; Weeks, B.R.; Lupton, J.R.; McMurray, D.N. Immunomodulatory effects of (n-3) fatty acids: Putative link to inflammation and colon cancer. J. Nutr. 2007, 137, 200s–204s. [Google Scholar] [PubMed]
- Schwingshackl, L.; Hoffmann, G.; Missbach, B.; Stelmach-Mardas, M.; Boeing, H. An umbrella review of nuts intake and risk of cardiovascular disease. Curr. Pharm. Des. 2017, 23, 1016–1027. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Sun, Q.; Bernstein, A.M.; Schulze, M.B.; Manson, J.E.; Stampfer, M.J.; Willett, W.C.; Hu, F.B. Red meat consumption and mortality: Results from 2 prospective cohort studies. Arch. Intern. Med. 2012, 172, 555–563. [Google Scholar] [PubMed]
- Rohrmann, S.; Overvad, K.; Bueno-de-Mesquita, H.B.; Jakobsen, M.U.; Egeberg, R.; Tjønneland, A.; Nailler, L.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; Krogh, V.; et al. Meat consumption and mortality-results from the European Prospective Investigation into Cancer and Nutrition. BMC Med. 2013, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Alcohol Consumption and Ethyl Carbamate; International Agency for Research on Cancer: Lyon, France, 2010; p. 1440. [Google Scholar]
- Allen, N.E.; Beral, V.; Casabonne, D.; Kan, S.W.; Reeves, G.K.; Brown, A.; Green, J.; Collaborators, M.W.S. Moderate alcohol intake and cancer incidence in women. J. Natl. Cancer Inst. 2009, 101, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Lachenmeier, D.W.; Rehm, J. Moderate red wine drinking does not help cut women’s breast cancer risk. J. Womens Health. (Larchmt) 2012, 21, 469–470. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Hunter, D.J.; Spiegelman, D.; Adami, H.O.; Albanes, D.; Bernstein, L.; van den Brandt, P.A.; Buring, J.E.; Cho, E.; Folsom, A.R.; et al. Alcohol intake and renal cell cancer in a pooled analysis of 12 prospective studies. J. Natl. Cancer Inst. 2007, 99, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Mack, W.J.; Preston-Martin, S.; Dal Maso, L.; Galanti, R.; Xiang, M.; Franceschi, S.; Hallquist, A.; Jin, F.; Kolonel, L.; La Vecchia, C.; et al. A pooled analysis of case-control studies of thyroid cancer: Cigarette smoking and consumption of alcohol, coffee, and tea. Cancer Causes Control 2003, 14, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Morton, L.M.; Zheng, T.; Holford, T.R.; Holly, E.A.; Chiu, B.C.; Costantini, A.S.; Stagnaro, E.; Willett, E.V.; Dal Maso, L.; Serraino, D.; et al. Alcohol consumption and risk of non-Hodgkin lymphoma: A pooled analysis. Lancet Oncol. 2005, 6, 469–476. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The mediterranean diets: What is so special about the diet of Greece? The scientific evidence. J. Nutr. 2001, 131, 3065S–3073S. [Google Scholar] [PubMed]
- Trichopoulou, A.; Orfanos, P.; Norat, T.; Bueno-de-Mesquita, B.; Ocké, M.C.; Peeters, P.H.; van der Schouw, Y.T.; Boeing, H.; Hoffmann, K.; Boffetta, P.; et al. Modified Mediterranean diet and survival: EPIC-Elderly Prospective Cohort Study. BMJ 2005, 330, 991. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, A.; De Pergola, G. Mediterranean diet and cardiovascular disease: A critical evaluation of a priori dietary indexes. Nutrients 2015, 7, 7863–7888. [Google Scholar] [CrossRef] [PubMed]
- Verberne, L.; Bach-Faig, A.; Buckland, G.; Serra-Majem, L. Association between the Mediterranean diet and cancer risk: A review of observational studies. Nutr. Cancer 2010, 62, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Lampousi, A.M.; Portillo, M.P.; Romaguera, D.; Hoffmann, G.; Boeing, H. Olive oil in the prevention and management of type 2 diabetes mellitus: A systematic review and meta-analysis of cohort studies and intervention trials. Nutr. Diabetes 2017, 7, e262. [Google Scholar] [CrossRef] [PubMed]
- Cicerale, S.; Conlan, X.A.; Sinclair, A.J.; Keast, R.S. Chemistry and health of olive oil phenolics. Crit. Rev. Food Sci. Nutr. 2009, 49, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.; Forman, D.; Bray, F. Globocan 2012 v1.1, Cancer Incidence and Mortality Worldwide: IARC Cancerbase No. 11. Available online: http://www.wcrf.org/int/cancer-facts-figures/data-cancer-frequency-country (accessed on 25 July 2017).
- Imamura, F.; Micha, R.; Khatibzadeh, S.; Fahimi, S.; Shi, P.; Powles, J.; Mozaffarian, D.; Global Burden of Diseases Nutrition and Chronic Diseases Expert Group (NutriCoDE). Dietary quality among men and women in 187 countries in 1990 and 2010: A systematic assessment. Lancet Glob. Health 2015, 3, e132–e142. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Diet quality as assessed by the Healthy Eating Index, the Alternate Healthy Eating Index, the Dietary Approaches to Stop Hypertension score, and health outcomes: A systematic review and meta-analysis of cohort studies. J. Acad. Nutr. Diet. 2015, 115, 780–800. [Google Scholar] [CrossRef] [PubMed]
- Boniol, M.; Autier, P. Prevalence of main cancer lifestyle risk factors in europe in 2000. Eur. J. Cancer 2010, 46, 2534–2544. [Google Scholar] [CrossRef] [PubMed]
| Author | Country Study Name | Study Design | Outcome | Population Follow-Up (Years) | Age at Entry | Sex | Components of Score Score Range | Adjustment | RR/HR/OR (95% CI) Multivariable Adjusted |
|---|---|---|---|---|---|---|---|---|---|
| De Lorgeril et al. [32] | France Lyon Diet Heart Study | RCT | Cancer mortality Lung cancer Digestive tract cancer Urinary tract cancer Throat cancer | 605 4 | 54 | M/W | 1. MedD: More bread, more root vegetables and green vegetables, more fish, less meat, no day without fruit, and butter and cream to be replaced with margarine supplied by the study; supplemented with a rapeseed oil-based margarine 2. control diet | NA | Cancer mortality MedD with margarine RR: 0.75 (0.17, 3.33) versus control diet Lung cancer MedD with margarine RR: 2.01 (0.37, 10.87) versus control diet Throat cancer MedD with margarine RR: 0.14 (0.01, 2.76) versus control diet |
| Toledo et al. [33] | Spain PREDIMED | RCT | Breast cancer | 4282 4.8 | 60–80 | W | 1. MedD supplemented with extra-virgin olive oil; 2. MedD supplemented with mixed nuts; 3. control diet (advice to reduce dietary fat) | Age, study site, BMI, waist-to-height ratio, hormone therapy, PA, total energy intake, alcohol, age at menopause, baseline adherence to the MedD | MedD with olive oil RR: 0.32 (0.13, 0.79) versus control diet MedD with nuts RR: 0.59 (0.26, 1.35) versus control diet Both MedD RR: 0.43 (0.21, 0.88) versus control diet |
| Anic et al. [50] | United States NIH-AARP | Cohort | Lung cancer | 460,770 10.5 | 50–71 | M/W | 1.↑ whole grains; 2.↑ vegetables; 3.↑ fruits; 4.↑ nuts; 5.↑ legumes; 6.↑ fish; 7.↑ MUFA:SFA ratio; 8.↓ red and processed meats; 9.↔ alcohol MedD score range: 0–9 | Age, sex, race, education, BMI, PA, total energy, smoking status, cigarettes per day, time since quitting smoking, and regular use of cigars/pipes | HR: 0.85 (0.79, 0.91) for fifth versus first quintile |
| Butler et al. [64] | Singapore SCHS | Cohort | Breast cancer | 34,028 >5 | 45–74 | W | 1.↑ cereals; 2.↑ vegetables; 3.↑ fruits/nuts; 4.↑ legumes; 5.↑ fish; 6.↑ MUFA: SFA ratio; 7.↓ meat; 8.↓ dairy; 9.↓ carbohydrates; 10. ↓ alcohol MedD score range: 0–10 | NA | HR: 0.96 (0.76, 1.21) for third versus first tertile |
| Dugué et al. [51] | Australia MCCS | Cohort | Urothelial cell carcinoma | 37,442 21.3 | 40–69 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ cereals; 4.↑ legumes; 5.↑ fish; 6.↑ olive oil; 7.↓ dairy; 8.↓ red meat; 9.↔ alcohol MedD score range: 0–9 | Sex, country of birth, smoking, alcohol, BMI, PA, education, socioeconomic status | HR: 0.89 (0.62, 1.26) for fifth versus first quintile |
| Haridass et al. [52] | United States CTS | Cohort | Breast cancer | 90,244 16 | 22–104 | W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts/legumes; 4.↑ fish; 5.↑ whole grains; 6.↑ MUFA:SFA; 7.↓ red and processed meats; 8.↔ alcohol MedD score range: 0–8 | Age at baseline, race, menopausal status, age at menarche, breast cancer family history, smoking, BMI, energy intake | HR: 0.91 (0.83, 1.01) for fifth versus first quintile |
| Hirko et al. [53] | United States NHS | Cohort | Breast cancer (by molecular subtype) | 100,643 22 | 30–55 | W | 1.↑ fruits; 2.↑ vegetables; 3.↑ legumes and soy; 4.↑ nuts; 5.↑ fish and seafood; 6.↑ whole grains; 7.↑ MUFA:SFA; 8.↓ red and processed meat; 9.↔ alcohol MedD score range: 0–9 | BMI at age 18, weight change since age 18, PA, energy intake, parity/age at first birth, menopausal hormone use, oral contraceptive use, age at menarche, age at menopause, family history of breast cancer, benign breast cancer diagnosis | Luminal A HR: 1.09 (0.91, 1.30) for fifth quintile (5.5–9.0) versus first (0–2.6) quintile Luminal B HR: 1.02 (0.76, 1.37) for fifth quintile (5.5–9.0) versus first (0–2.6) quintile HER2 type HR: 0.74 (0.42, 1.29) for fifth quintile (5.5–9.0) versus first (0–2.6) quintile Basal-like HR: 0.78 (0.49, 1.26) for fifth quintile (5.5–9.0) versus first (0–2.6) quintile Unclassified HR: 0.89 (0.41, 1.89) for fifth quintile (5.5–9.0) versus first (0–2.6) quintile |
| Hodge et al. [54] | Australia MCCS | Cohort | Lung cancer | 35,303 18 | 40–69 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ cereals; 4.↑ legumes; 5.↑ fish; 6.↑ olive oil; 7.↓ dairy; 8.↓ red meat; 9.↔ alcohol MedD score range: 0–9 | Pack-years, years since quitting smoking, smoking status, country of birth, education, BMI, PA, sex, SEIFA quintile, energy | HR: 0.64 (0.45, 0.90) for highest category (7–9) versus lowest category (0–3) |
| Jacobs et al. [55] | United States MEC | Cohort | Colorectal cancer mortality among cancer survivors | 4204 6.0 | 45–75 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts; 4.↑ legumes; 5.↑ fish; 6.↑ whole grains; 7.↑ MUFA:SFA; 8.↓ red and processed meat; 9.↔ alcohol MedD score range: 0–9 | Age at diagnosis, ethnicity, stage at diagnosis, total energy intake, smoking status, pack-years, PA, education, radiation treatment, chemotherapy, NSAID use, family history of CRC, comorbidities | Men HR: 1.07 (0.81, 1.42) for fourth quartile (6–9) versus first quartile (0–2) Women HR: 0.74 (0.54, 1.01) for fourth quartile (6–9) versus first quartile (0–2) |
| Jones et al. [65] | United Kingdom WHS | Cohort | Colorectal cancer | 35,372 17.4 | 35–69 | W | 1.↑ vegetables; 2.↑ fruits & nuts; 3.↑ legumes; 4.↑ cereals; 5.↑ fish; 6.↑ MUFA+PUFA: SFA; 7.↓ dairy; 8.↓ meat; 9. ↓ poultry; 10.↔ alcohol MedD score range: 0–10 | Age, BMI, energy intake, physical activity, smoking status, socioeconomic status and family history of colorectal cancer | HR: 0.82 (0.57, 1.17) Fifth quintile (7–10) versus first quintile (0–2) |
| Larsson et al. [56] | Sweden Swedish Mammography Cohort Cohort of Swedish Men | Cohort | Biliary tract cancer Gallbladder cancer | 76,014 13.3 | 45–83 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ legumes and nuts; 4.↑ whole-grains; 5.↑ fish; 6.↓ full-fat dairy products; 7.↓ red meat and processed meat; 8.↑ olive oil; 9.↔ alcohol MedD score range: 9–45 | Age, sex, education, smoking status and pack-years of smoking, diabetes, BMI, total energy intake | Extrahepatic Biliary tract cancer HR: 0.41 (0.25, 0.67) for third tertile (29–45) to first tertile (9–24) Gallbladder cancer HR: 0.42 (0.23, 0.79) for third tertile (29–45) to first tertile (9–24) Intrahepatic BTC HR: 0.71 (0.25, 2.04) for third tertile (29–45) to first tertile (9–24) |
| Lassale et al. [57] | Europe EPIC | Cohort | Cancer mortality | 451,256 12.8 | 25–70 | M/W | MedD: 1. fruits; 2. vegetables; 3. legumes; 4. grains; 5. fish; 6. meat; 7. dairy products; 8. MUFA:SFA; 9. alcohol MedD score range: 0–9 rMedD: 1. fruits; 2. vegetables; 3. legumes; 4. grains; 5. fish; 6. meat; 7. dairy products; 8. olive oil; 9. alcohol rMED score range: 0–18 MSDPS: 1. whole grain cereals (8 servings/day); 2. fruits (3 servings/day); 3. vegetables (6 servings/day); 4. dairy (2 servings/day); 5. fish (6 servings/week); 6. wine (3 and 1.5 servings/day for men and women, respectively); 7. poultry (4 servings/week); 8. olives, legumes, and nuts (4 servings/week); 9. potatoes (3 servings/week); 10. eggs (3 servings/week); 11. sweets (3 servings/week); 12. meats (1 servings/week); 13. olive oil (exclusive use) MSDPS score range: 0–100 | Age at baseline, BMI, PA, smoking status, education, stratified by sex and study center | MedD HR: 0.82 (0.77, 0.88) for fourth versus first quartile rMED HR: 0.82 (0.77, 0.88) for fourth versus first quartile MSDPS HR: 0.84 (0.79, 0.90) for fourth versus first quartile |
| Maisonneuve et al. [58] | Italy COSMOS | Cohort | Lung cancer | 4336 8.5 | 50–84 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts; 4.↑ cereals; 5.↑ legumes; 6.↑ fish; 7.↓ red and processed meats; 8.↔ alcohol; 9.↑ MUFA:SFA MedD score range: 0–9 | Baseline risk probability (age, sex, smoking duration, smoking intensity, years of smoking cessation, asbestos exposure), total energy, dietary inflammatory index | HR: 0.20 (0.04, 0.91) for highest score category (8–9) versus lowest score category (0–1) |
| Molina-Montes et al. [59] | Europe EPIC | Cohort | Exocrine pancreatic cancer | 477,309 11.3 | 35–70 | M/W | 1.↑ fruits and nuts; 2.↑ vegetables; 3.↑ legumes; 4.↑ fish and seafood; 5.↑ olive oil; 6.↑ cereals; 7.↓ meat; 8.↓ dairy products MedD score range: 0–16 | Total energy intake, BMI, smoking status and intensity, alcohol intake, diabetes, stratified by age, sex and study centre | HR: 0.99 (0.77, 1.26) for highest score category (10–16) versus lowest score category (0–5) |
| Park et al. [60] | United States MEC | Cohort | Colorectal cancer | 190,949 16 | 45–75 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts; 4.↑ legumes; 5.↑ fish; 6.↑ whole grains; 7.↑ MUFA:SFA; 8.↓ red and processed meat; 9.↔ alcohol MedD score range: 0–9 | Age at cohort entry, family history of colorectal cancer, history of colorectal polyp, BMI, smoking, multivitamin, nonsteroidal anti-inflammatory drugs, physical activity, menopausal status, menopausal hormone therapy use for women only, and total energy intake | HR: ♂ 0.84 (0.72, 0.97) HR: ♀ 0.96 (0.82, 1.13) for highest score category (6–9) versus lowest score category (0–2) |
| Van den Brandt et al. [61] | The Netherlands NCS | Cohort | Breast cancer | 62,573 | 55–69 | W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts; 4.↑ whole grains; 5.↑ legumes; 6.↑ fish; 7.↑ MUFA:SFA; 8.↓ red and processed meats; 9.↔ alcohol MedD score range: 0–9 | Age, smoking, duration, body height, BMI, non-occupational physical activity, highest level of education, family history of breast cancer in mother or sisters, history of benign breast disease, age at menarche, parity, age at first birth, age at menopause, oral contraceptive use, postmenopausal HRT, energy intake and alcohol intake | HR: 0.87 (0.72, 1.06) for highest score category (6–8) vs. lowest score category (0–3) Without alcohol component |
| Vargas et al. [62] | United States WHIOS | Cohort | Colorectal cancer | 78,273 12.4 | 50–79 | W | 1.↑ vegetables; 2.↑ fruits; 3.↑ nuts; 4.↑ whole grains; 5.↑ legumes; 6.↑ fish; 7.↑ MUFA:SFA; 8.↓ red and processed meats; 9.↔ alcohol MedD score range: 0–9 | Age, race/ethnicity, PA, education, smoking, hormone replacement therapy | HR: 0.91 (0.74, 1.11) for fifth quintile (6–9) versus first quintile (0–2) |
| Whalen et al. [63] | United States REGARDS | Cohort | Cancer mortality | 21,423 6.25 | >45 | M/W | 1.↑ vegetables; 2.↑ fruits; 3.↑ lean meats; 4.↑ fish; 5.↑ nuts; 6.↑ MUFA:SFA; 7.↓ red and processed meats; 8.↓ sodium; 9. ↔ dairy; 10.↔ grains and starches; 11.↔ alcohol MedD score range: 11–55 | Sex, race, total energy intake, BMI, PA, smoking, annual income, hormone replacement therapy use (in women) at baseline | HR: 0.64 (0.48, 0.84) for fifth versus first quintile |
| Author | Country Study Name | Study Design | Outcome | Cases/Controls | Age at Entry | Sex | Components of Score Score Range | Adjustment | Multivariable Adjusted |
| Askari et al. [66] | Iran | Case-control | Prostate cancer | 52/104 | 40–78 | M | 1. whole grain cereals (8 servings/day); 2. fruits (3 servings/day); 3. vegetables (6 servings/day); 4. dairy products (2 servings/day); 5. fish and other seafood (6 servings/week); 6. poultry (4 servings/week); 7. olives/legumes/nuts (4 servings/week); 8. potatoes and other starchy roots (3 servings/week); 9. eggs (3 servings/week); 10. sweets (3 servings/week); 11. meat (1 servings/week); 12. olive oil (exclusive use) MedD score range: 0–100 | Age, BMI, smoking, energy intake, education, diabetes | OR: 0.28 (0.08, 0.91) for third versus first tertile |
| Campagna et al. [67] | Italy | Case-control | Lymphoma | 322/446 | n.d | M/W | 1.↑ fruits; 2.↑ vegetables; 3.↑ legumes; 4.↑ fresh fish and seafood; 5.↑ pasta, rice, and bread; 6.↓ red meat; 7.↔ wine MedD score range: n.d | Age, sex, education | OR: 0.9 (0.6, 1.5) for fifth versus first quintile |
| Castello et al. [43] | Spain | Case-control | Breast cancer | 1181/1682 | 20–85 | W | PCA: ↑ fish; ↑ vegetables; ↑ legumes; ↑ boiled potatoes; ↑ fruits; ↑ olives and vegetable oil; ↓ juices | menopausal status, age, education, BMI, age at first delivery, family history of breast cancer, physical activity, smoking status, caloric intake and alcohol intake as fixed effects and province of residence as a random effect term | OR: 0.90 (0.69, 1.17) for fourth vs. first quartile |
| Castello et al. [44] | Spain | Case-control | Prostate cancer | 754/1277 | 38–85 | M | PCA: ↑ fish; ↑ vegetables; ↑ legumes; ↑ boiled potatoes; ↑ fruits; ↑ olives and vegetable oil; ↓ juices | Age, education, BMI, age at first delivery, family history of prostate cancer and caloric intake as fixed effects and province of residence as a random effect | OR: 0.91 (0.66, 1.25) for fourth vs. first quartile |
| Giraldi et al. [68] | Italy | Case-control | Head and neck cancer | 500/433 | n.d | M/W | 1.↑ fruits; 2.↑ vegetables; 3.↑ legumes; 4.↑ fish; 5.↓ meat and meat products; 6.↔ alcohol MedD score range: 0–12 | Age, sex, smoking, alcohol, total energy intake | OR: 0.64 (0.58, 0.71) per 1-point increase |
| Rosato et al. [69] | Italy | Case-control | Colorectal cancer | 3745/6804 | 19–74 | M/W | 1.↑ vegetables; 2.↑ legumes; 3.↑ fruits and nuts; 4.↑ cereals; 5.↑ fish and seafood; 6.↑ MUFA:SFA; 7.↓ dairy; 8.↓ meat and meat products; 9.↔ alcohol MedD score range: 0–9 | Age, sex, calendar period, center, education, BMI, PA, family history of intestinal cancer, total energy intake | OR: 0.52 (0.43, 0.62) for highest score category (7–9) versus lowest score category (0–2) |
| Stojanovic 2017 [70] | Italy | Case-control | Gastric cancer | 223/223 | NA | NA | NA | NA | OR: 0.70 (0.61, 0.81) |
| Turati et al. [71] | Italy | Case-control | Nasopharyngeal cancer | 198/594 | 18–76 | M/W | 1.↑ vegetables; 2.↑ legumes; 3.↑ fruits and nuts; 4.↑ cereals; 5.↑ fish and seafood; 6.↑ MUFA:SFA; 7.↓ dairy products; 8.↓ meats; 9.↔ alcohol MedD score range: 0–9 | sex, age, place of residence, education, smoking, total energy intake | OR: 0.66 (0.44, 0.99) for highest score category (>6) versus lowest score category (0–4) |
| Wang et al. [72] | China | Case-control | Nasopharyngeal cancer | 600/600 | M/W | 1.↑ whole grains; 2.↑ vegetables; 3.↑ fruits; 4.↑ legumes; 5.↑ nuts; 6.↑ fish; 7.↑ MUFA:SFA; 8.↔ alcohol; 9.↓ red and processed meats MedD score range: 0–9 | Age, BMI, occupation, marital status, education, household income, smoking, drinking, exposure to potential toxic substances, multivitamin supplements, chronic rhinitis history, PA, energy intake, preserved vegetables and animal food | OR: 0.85 (0.59, 1.22) for fourth versus first quartile |
| Outcome | No. of Studies | Study Type | Risk Ratio/Odds Ratio | 95% CI | I2 (%) |
|---|---|---|---|---|---|
| Cancer mortality | 1 | RCT | 0.75 | 0.17, 3.33 | NA |
| 14 | Cohort | 0.86 | 0.81, 0.91 | 82 | |
| Colorectal cancer | 11 | Observational | 0.82 | 0.75, 0.88 | 73 |
| incidence | 6 | Cohort | 0.86 | 0.80, 0.92 | 28 |
| 5 | Case-control | 0.71 | 0.57, 0.88 | 88 | |
| Breast cancer | 1 | RCT | 0.43 | 0.21, 0.88 | NA |
| 16 | Observational | 0.92 | 0.89, 0.96 | 8 | |
| incidence | 7 | Cohort | 0.94 | 0.90, 0.99 | 11 |
| 9 | Case-control | 0.89 | 0.85, 0.94 | 0 | |
| Prostate cancer | 6 | Combined | 0.96 | 0.92, 1.00 | 0 |
| incidence | 3 | Cohort | 0.96 | 0.92, 1.00 | 0 |
| 3 | Case-control | 0.90 | 0.64, 1.26 | 52 | |
| Gastric cancer | 4 | Combined | 0.72 | 0.60, 0.86 | 55 |
| incidence | 2 | Cohort | 0.82 | 0.61, 1.10 | 49 |
| 2 | Case-control | 0.65 | 0.53, 0.79 | 53 | |
| Liver cancer | 2 | Combined | 0.58 | 0.46, 0.73 | 0 |
| incidence | 1 | Cohort | 0.62 | 0.47, 0.82 | NA |
| 1 | Case-control | 0.51 | 0.34, 0.77 | NA | |
| Esophageal cancer | 2 | Combined | 0.49 | 0.22, 1.09 | 83 |
| incidence | 1 | Cohort | 0.68 | 0.34, 1.36 | NA |
| 1 | Case-control | 0.26 | 0.13, 0.52 | NA | |
| Head and neck cancer | 1 | RCT | 0.14 | 0.01, 2.76 | 86 |
| 7 | Observational | 0.49 | 0.37, 0.66 | 87 | |
| incidence | 1 | Cohort | 0.61 | 0.33, 1.14 | 77 |
| 6 | Case-control | 0.46 | 0.32, 0.67 | 89 | |
| Endometrial cancer | 3 | Combined | 0.72 | 0.40, 1.31 | 94 |
| incidence | 1 | Cohort | 0.98 | 0.82, 1.17 | NA |
| 2 | Case-control | 0.61 | 0.29, 1.29 | 89 | |
| Respiratory cancer | 1 | RCT | 2.01 | 0.37, 10.87 | NA |
| incidence | 3 | Cohort | 0.71 | 0.49, 1.02 | 66 |
| Bladder cancer incidence | 2 | Cohort | 0.85 | 0.72, 1.01 | NA |
| Pancreatic cancer | 2 | Combined | 0.69 | 0.34, 1.41 | 92 |
| 1 | Cohort | 0.99 | 0.77, 1.27 | NA | |
| 1 | Case-control | 0.48 | 0.35, 0.66 | NA | |
| Gallbladder cancer | 1 | Cohort | 0.42 | 0.23, 0.77 | NA |
| Biliary tract cancer | 1 | Cohort | 0.44 | 0.29, 0.67 | NA |
| Ovarian cancer | 1 | Cohort | 0.91 | 0.71, 1.17 | NA |
| Lymphoma | 1 | Case-control | 0.90 | 0.60, 1.35 | NA |
| Cancer mortality among cancer survivors | 4 | Cohort | 0.95 | 0.82, 1.12 | 5 |
| Recurrence among cancer survivors | 1 | Cohort | 0.61 | 0.18, 2.07 | NA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1063. https://doi.org/10.3390/nu9101063
Schwingshackl L, Schwedhelm C, Galbete C, Hoffmann G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients. 2017; 9(10):1063. https://doi.org/10.3390/nu9101063
Chicago/Turabian StyleSchwingshackl, Lukas, Carolina Schwedhelm, Cecilia Galbete, and Georg Hoffmann. 2017. "Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis" Nutrients 9, no. 10: 1063. https://doi.org/10.3390/nu9101063
APA StyleSchwingshackl, L., Schwedhelm, C., Galbete, C., & Hoffmann, G. (2017). Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients, 9(10), 1063. https://doi.org/10.3390/nu9101063




