Beneficial Effects of Small Molecule Oligopeptides Isolated from Panax ginseng Meyer on Pancreatic Beta-Cell Dysfunction and Death in Diabetic Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Animals and Gop Treatment
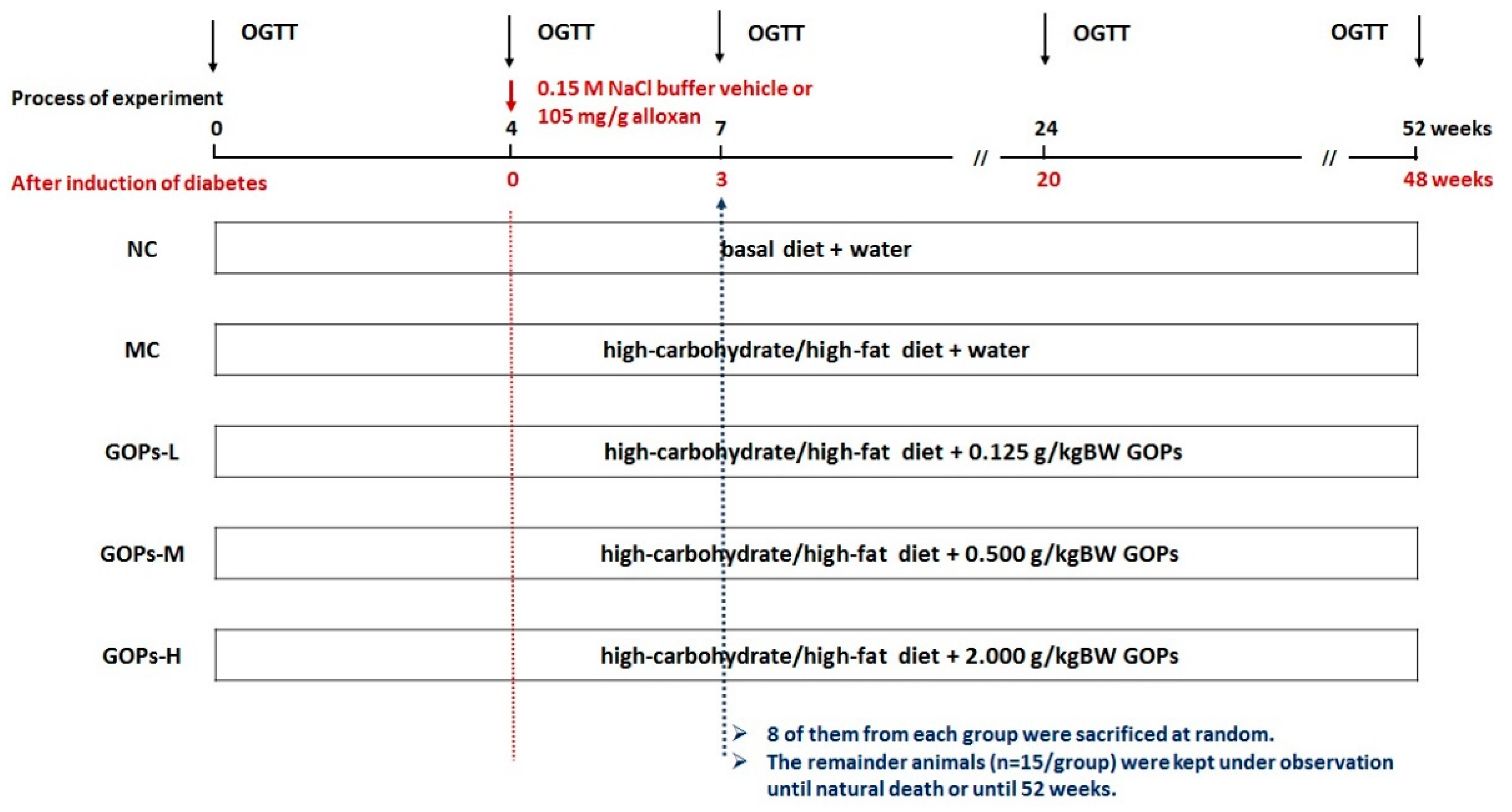
2.3. Experimental Protocols
2.4. Biochemical Assay and Oral Glucose Test Tolerance (OGTT)
2.5. Western-Blot Assay
2.6. Histopathology
2.7. Immunohistochemistry
2.8. Statistical Analysis
3. Results
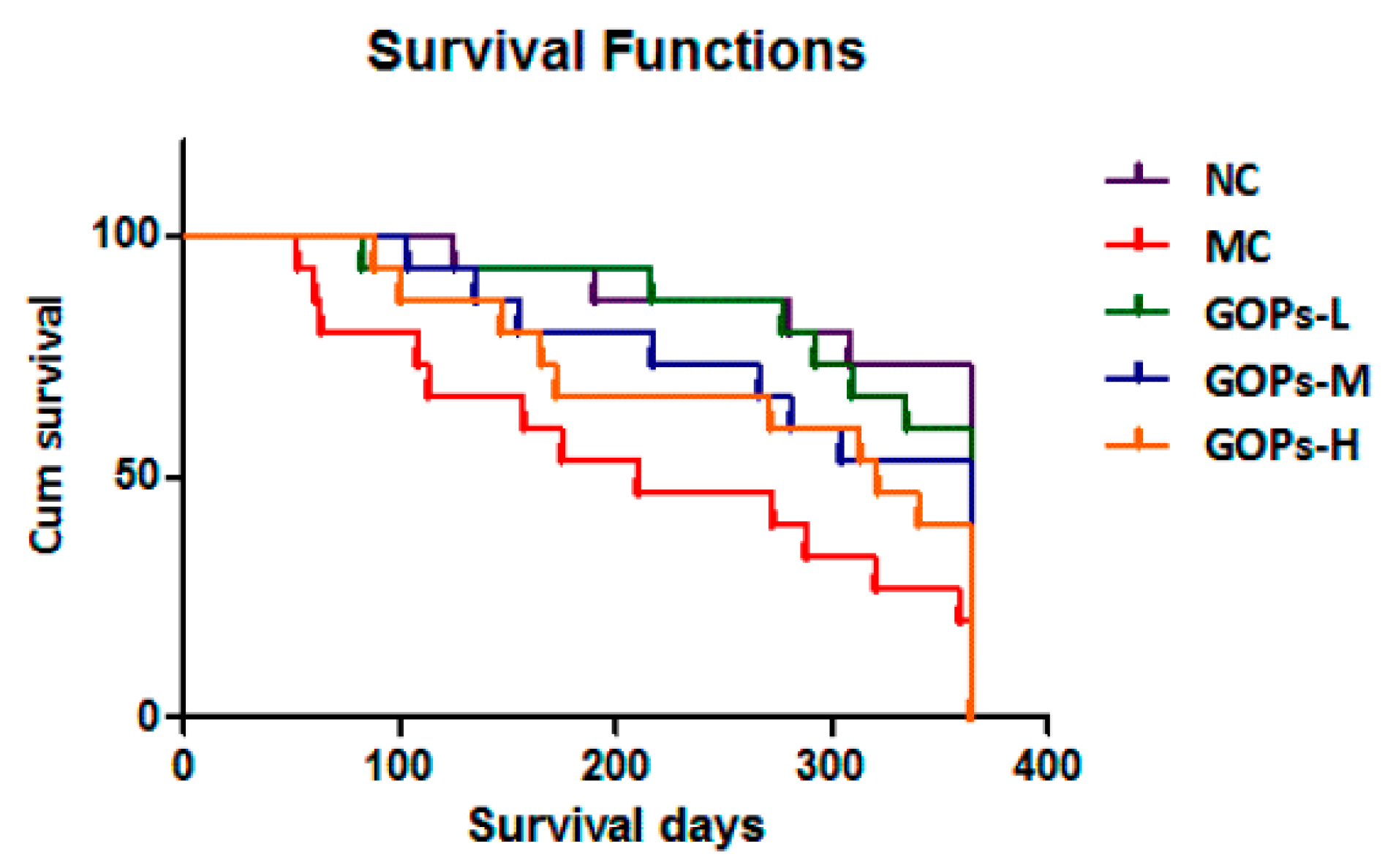
3.1. Effect of Long-Term GOPs Administration Could Bring a Survival
3.2. Effect of GOPs on Body Weight
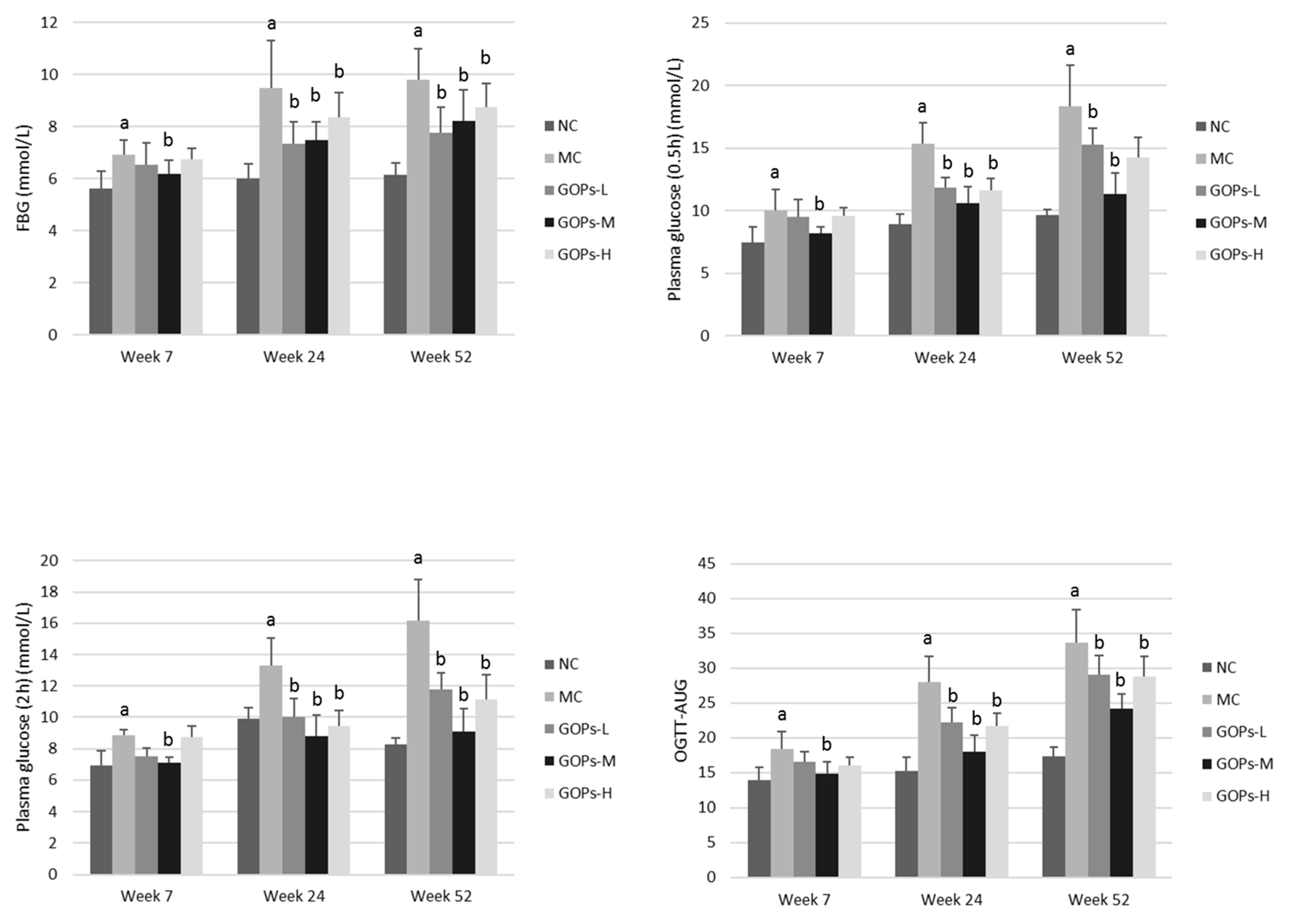
3.3. Effect of GOPs on OGTT and Insulin Metabolism
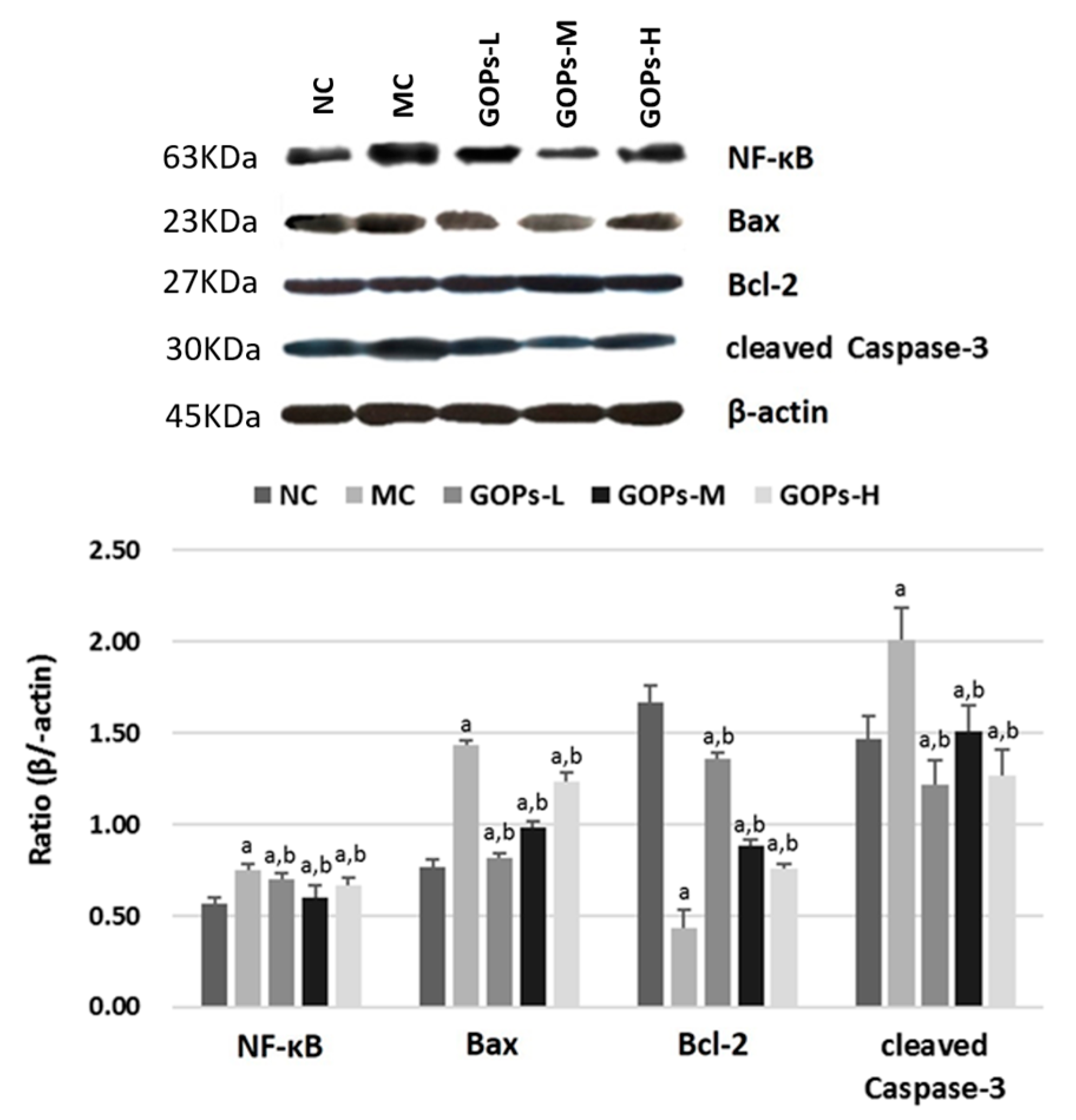
3.4. Effect of GOPs on NF-κB Activation in the Pancreas of Diabetic Rats
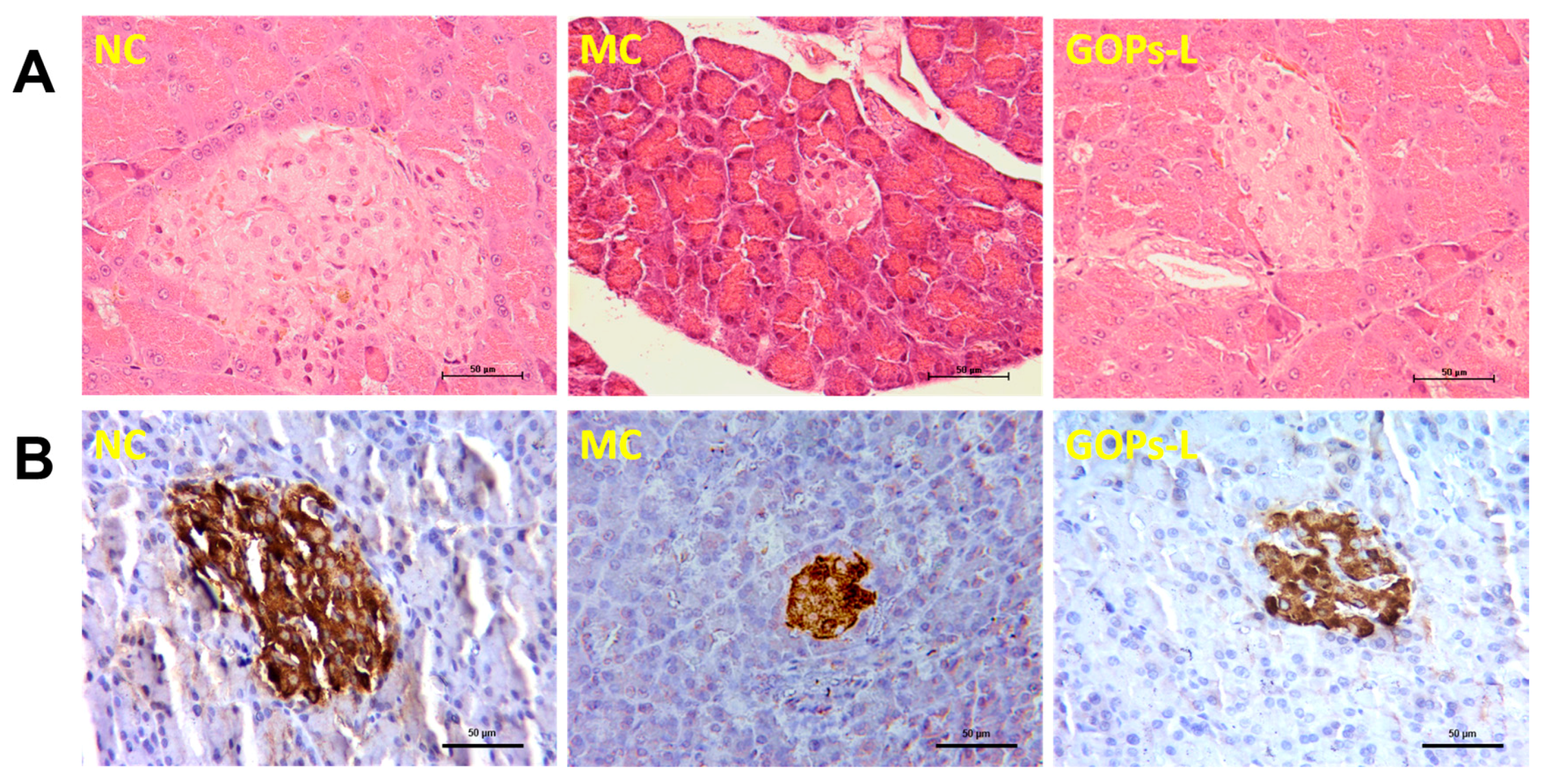
3.5. Effect of GOPs on Panceatic Histopathology and Insulin Expression in Pancreatic Islets
3.6. Effect of GOPs on SOD Activity and MDA Level
3.7. Effect of GOPs on Liver and Kidney Function
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Report on Diabetes; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Stumvoll, M.; Goldstein, B.J.; van Haeften, T.W. Type 2 diabetes: Principles of pathogenesis and therapy. Lancet 2005, 365, 1333–1346. [Google Scholar] [CrossRef]
- Morrison, J.A.; Glueck, C.J.; Horn, P.S.; Schreiber, G.B.; Wang, P. Pre-teen insulin resistance predicts weight gain, impaired fasting glucose, and type 2 diabetes at age 18–19 years: A 10-y prospective study of black and white girls. Am. J. Clin. Nutr. 2008, 88, 778–788. [Google Scholar] [PubMed]
- Tanabe, N.; Saito, K.; Yamada, Y.; Takasawa, T.; Seki, N.; Suzuki, H. Risk assessment by post-challenge plasma glucose, insulin response ratio, and other indices of insulin resistance and/or secretion for predicting the development of type 2 diabetes. Intern. Med. 2009, 48, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Prentki, M.; Nolan, C.J. Islet beta cell failure in type 2 diabetes. J. Clin. Investig. 2006, 116, 1802–1812. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, A.; Tatsumi, Y.; Deura, K.; Mizuno, S.; Ohno, Y.; Miyamatsu, N.; Watanabe, S. Impact of impaired insulin secretion and insulin resistance on the incidence of type 2 diabetes mellitus in a Japanese population: The Saku study. Diabetologia 2013, 56, 1671–1679. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.P.; Harmon, J.; Tran, P.O.; Poitout, V. Beta-cell glucose toxicity, lipotoxicity, and chronic oxidative stress in type 2 diabetes. Diabetes 2004, 53 (Suppl. 1), S119–S124. [Google Scholar] [CrossRef] [PubMed]
- Cnop, M.; Welsh, N.; Jonas, J.C.; Jörns, A.; Lenzen, S.; Eizirik, D.L. Mechanisms of pancreatic beta-cell death in type 1 and type 2 diabetes: Many differences, few similarities. Diabetes 2005, 54 (Suppl. 2), S97–S107. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Ma, H.; Gao, F.; Zhong, L.; Ren, J. Metallothionein alleviates oxidative stress-induced endoplasmic reticulum stress and myocardial dysfunction. J. Mol. Cell. Cardiol. 2009, 47, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.L.; Wang, Z.; Wang, H.; Zhang, C.; Zhang, Y.; Zhao, M.; Chen, Y.H.; Meng, X.H.; Xu, D.X. Ascorbic acid protects against cadmium-induced endoplasmic reticulum stress and germ cell apoptosis in testes. Reprod. Toxicol. 2012, 34, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.F.; Peng, H.B.; Liu, G.Q.; Zhang, F.; Li, Y. Beneficial effects of oligopeptides from marine salmon skin in a rat model of type 2 diabetes. Nutrition 2010, 26, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zhang, Z.; Dai, X.; Jiang, Y.; Bao, L.; Li, Y.; Li, Y. Grape seed proanthocyanidins ameliorate pancreatic beta-cell dysfunction and death in low-dose streptozotocin- and high-carbohydrate/high-fat diet-induced diabetic rats partially by regulating endoplasmic reticulum stress. Nutr. Metab. 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Senchina, D.S.; Shah, N.B.; Doty, D.M.; Sanderson, C.R.; Hallam, J.E. Herbal supplements and athlete immune function—What’s proven, disproven, and unproven? Exerc. Immunol. Rev. 2009, 15, 66–106. [Google Scholar] [PubMed]
- Song, J.Y.; Han, S.K.; Bae, K.G.; Lim, D.S.; Son, S.J.; Jung, I.S.; Yi, S.Y.; Yun, Y.S. Radioprotective effects of ginsan, an immunomodulator. Radiat. Res. 2003, 159, 768–774. [Google Scholar] [CrossRef]
- Lee, E.J.; Song, M.J.; Kwon, H.S.; Ji, G.E.; Sung, M.K. Oral administration of fermented red ginseng suppressed ovalbumininduced allergic responses in female BALB/c mice. Phytomedicine 2012, 19, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Seong, M.A.; Woo, J.K.; Kang, J.H.; Jang, Y.S.; Choi, S.; Jang, Y.S.; Lee, T.H.; Jung, K.H.; Kang, D.K.; Hurh, B.S.; et al. Oral administration of fermented wild ginseng ameliorates DSS-induced acute colitis by inhibiting NF-kappaB signaling and protects intestinal epithelial barrier. BMB Rep. 2015, 48, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Shishtar, E.; Sievenpiper, J.L.; Djedovic, V.; Cozma, A.I.; Ha, V.; Jayalath, V.H.; Jenkins, D.J.; Meija, S.B.; de Souza, R.J.; Jovanovski, E.; et al. The effect of ginseng (the genus Panax) on glycemic control: A systematic review and metaanalysis of randomized controlled clinical trials. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.Z.; Luo, L. Ginseng on hyperglycemia: Effects and mechanisms. Evid. Based Complement. Alternat. Med. 2009, 6, 423–427. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Wang, J.B.; Sun, B.; Zhao, J.; Li, L.; Xu, T.; Li, H.; Sun, J.Q.; Ren, J.; Liu, R.; et al. Suppression of TNF-α and free radicals reduces systematic inflammatory and metabolic disorders: Radioprotective effects of ginseng oligopeptides on intestinal barrier function and antioxidant defense. J. Nutr. Biochem. 2017, 40, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Cai, X.; Wang, J.; Zhang, Y.; Sun, B.; Li, Y. Anti-Fatigue Effects of Small Molecule Oligopeptides Isolated from Panax ginseng C.A. Meyer in Mice. Nutrients 2016, 8, 807. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Liang, R.; Guo, Q.; Wang, S.; Zhao, M.; Zhang, Z.; Wang, J.; Li, Y. Dietary nucleotides extend the life span in Sprague-Dawley rats. J. Nutr. Health Aging 2013, 17, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Decorde, K.; Teissedre, P.L.; Sutra, T.; Ventura, E.; Cristol, J.P.; Rouanet, J.M. Chardonnay grape seed procyanidin extract supplementation prevents high-fat diet-induced obesity in hamsters by improving adipokine imbalance and oxidative stress markers. Mol. Nutr. Food Res. 2009, 53, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.J.; Ji, B.P.; Chen, G.; Zhou, F.; Luo, Y.C.; Yu, H.Q.; Gao, F.Y.; Zhang, Z.P.; Li, H.Y. A combination of grape seed-derived procyanidins and gypenosides alleviates insulin resistance in mice and HepG2 cells. J. Food Sci. 2009, 74, H1–H7. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, S.; Li, J.; Ren, Z.; Chen, F.; Wang, X. Anti-hyperglycemic activity of polysaccharides from calyx of Physalis alkekengi var. franchetii Makino on alloxan-induced mice. Int. J. Biol. Macromol. 2017, 99, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Das, J.; Sil, P.C. Taurine ameliorates alloxan-induced diabetic renal injury, oxidative stress-related signaling pathways and apoptosis in rats. Amino Acids 2012, 43, 1509–1523. [Google Scholar] [CrossRef] [PubMed]
- Lapolla, A.; Poli, T.; Meneghini, F.; Zucchetto, M.; Franchin, A.; Barison, A.; Fedele, D. Glycated serum proteins and glucose tolerance. Acta Diabetol. Lat. 1988, 25, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Selvin, E.; Parrinello, C.M. Age-related differences in glycaemic control in diabetes. Diabetologia 2013, 56, 2549–2551. [Google Scholar] [CrossRef] [PubMed]
- Liener, U.C.; Knöferl, M.W.; Sträter, J.; Barth, T.F.; Pauser, E.M.; Nüssler, A.K.; Kinzl, L.; Brückner, U.B.; Gebhard, F. Induction of apoptosis following blunt chest trauma. Shock 2003, 20, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Eskes, R.; Desagher, S.; Antonsson, B.; Martinou, J.C. Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol. Cell. Biol. 2000, 20, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.; Cory, S. The Bcl-2 protein family: Arbiters of cell survival. Science 1998, 281, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- DiPenta, J.M.; Green-Johnson, J.M.; Murphy, R.J. Type 2 diabetes mellitus, resistance training, and innate immunity: Is there a common link? Appl. Physiol. Nutr. Metab. 2007, 32, 1025–1035. [Google Scholar] [CrossRef] [PubMed]
- Togashi, N.; Ura, N.; Higashiura, K.; Murakami, H.; Shimamoto, K. The contribution of skeletal muscle tumor necrosis factor-α to insulin resistance and hypertension in fructose-fed rats. J. Hypertens. 2000, 18, 1605–1610. [Google Scholar] [CrossRef] [PubMed]
- Tsiavou, A.; Hatziagelaki, E.; Chaidaroglou, A.; Koniavitou, K.; Degiannis, D.; Raptis, S.A. Correlation between intracellular interferon-gamma (IFN-gamma) production by CD4+ and CD8+ lymphocytes and IFN-gamma gene polymorphism in patients with type 2 diabetes mellitus and latent autoimmune diabetes of adults (LADA). Cytokine 2005, 31, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, Y. Cell death regulation by the Bcl-2 protein family in the mitochondria. J. Cell. Physiol. 2003, 195, 158–167. [Google Scholar] [CrossRef] [PubMed]

| Parameter | NC | MC | GOPs-L | GOPs-M | GOPs-H | |
|---|---|---|---|---|---|---|
| FINS | Week 7 | 14.92 ± 2.6 | 28.48 ± 5.27 a | 16.47 ± 4.18 b | 14.99 ± 3.77 b | 22.53 ± 7.48 |
| FINS | Week 52 | 12.34 ± 0.71 | 14.98 ± 0.98 | 13.67 ± 0.54 | 13.25 ± 0.69 | 13.21 ± 0.6 |
| GSP | Week 7 | 1.95 ± 0.09 | 2.68 ± 0.12 a | 2.33 ± 0.24 b | 2.45 ± 0.09 b | 2.64 ± 0.15 |
| GSP | Week 52 | 2.01 ± 0.08 | 4.03 ± 0.14 a | 3.38 ± 0.5 b | 3.55 ± 0.43 b | 3.47 ± 0.23 b |
| HOMA-IR | Week 7 | 1.25 ± 0.21 | 2.80 ± 0.55 a | 1.42 ± 0.79 b | 1.61 ± 0.53 b | 2.05 ± 0.82 |
| HOMA-IR | Week 52 | 3.31 ± 0.26 | 6.51 ± 0.45 a | 4.84 ± 0.44 b | 4.84 ± 0.44 b | 5.29 ± 0.28 b |
| HOMA-B | Week 7 | 98.03 ± 12.75 | 46.53 ± 4.83 a | 62.82 ± 12.16 b | 57.6 ± 10.87 | 48.85 ± 9.85 |
| HOMA-B | Week 52 | 69.79 ± 46.52 | 43.46 ± 15.51 a | 51.08 ± 8.7 b | 52.00 ± 10.82 b | 45.49 ± 11.18 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, M.; Sun, B.; Li, D.; Mao, R.; Li, H.; Li, Y.; Wang, J. Beneficial Effects of Small Molecule Oligopeptides Isolated from Panax ginseng Meyer on Pancreatic Beta-Cell Dysfunction and Death in Diabetic Rats. Nutrients 2017, 9, 1061. https://doi.org/10.3390/nu9101061
Xu M, Sun B, Li D, Mao R, Li H, Li Y, Wang J. Beneficial Effects of Small Molecule Oligopeptides Isolated from Panax ginseng Meyer on Pancreatic Beta-Cell Dysfunction and Death in Diabetic Rats. Nutrients. 2017; 9(10):1061. https://doi.org/10.3390/nu9101061
Chicago/Turabian StyleXu, Meihong, Bin Sun, Di Li, Ruixue Mao, Hui Li, Yong Li, and Junbo Wang. 2017. "Beneficial Effects of Small Molecule Oligopeptides Isolated from Panax ginseng Meyer on Pancreatic Beta-Cell Dysfunction and Death in Diabetic Rats" Nutrients 9, no. 10: 1061. https://doi.org/10.3390/nu9101061
APA StyleXu, M., Sun, B., Li, D., Mao, R., Li, H., Li, Y., & Wang, J. (2017). Beneficial Effects of Small Molecule Oligopeptides Isolated from Panax ginseng Meyer on Pancreatic Beta-Cell Dysfunction and Death in Diabetic Rats. Nutrients, 9(10), 1061. https://doi.org/10.3390/nu9101061






