Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of Test Meals
2.2. Chemicals and Enzymes
2.3. Simulated Human Digestion
2.4. Gastric Digestion
2.5. Duodenal Digestion
2.6. Poliphenols Extraction from Samples before and after Dynamic in Vitro Digestion
2.7. Polyphenols Release and Radical Scavenging Activity
2.8. Statistical Analysis
3. Results
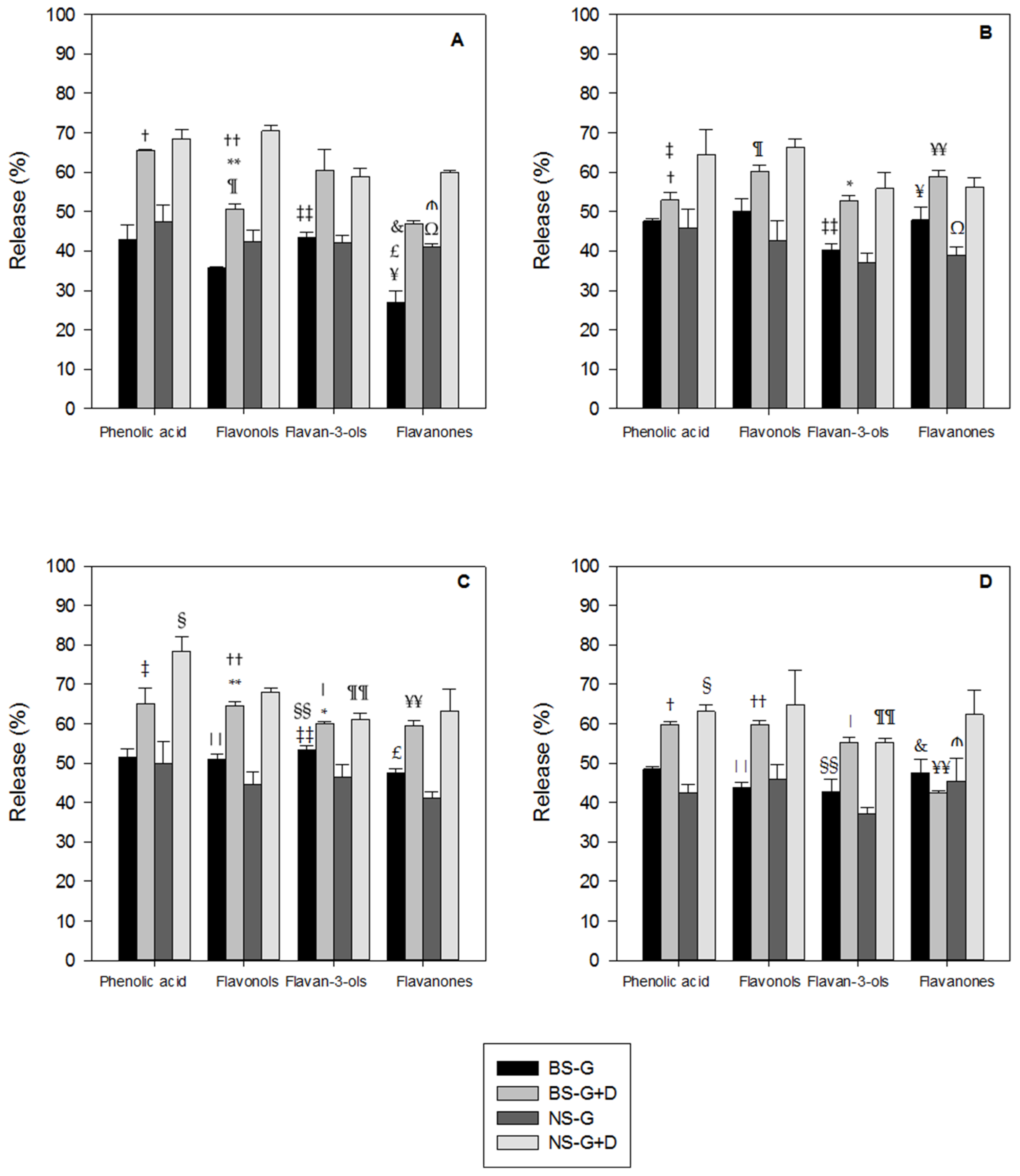
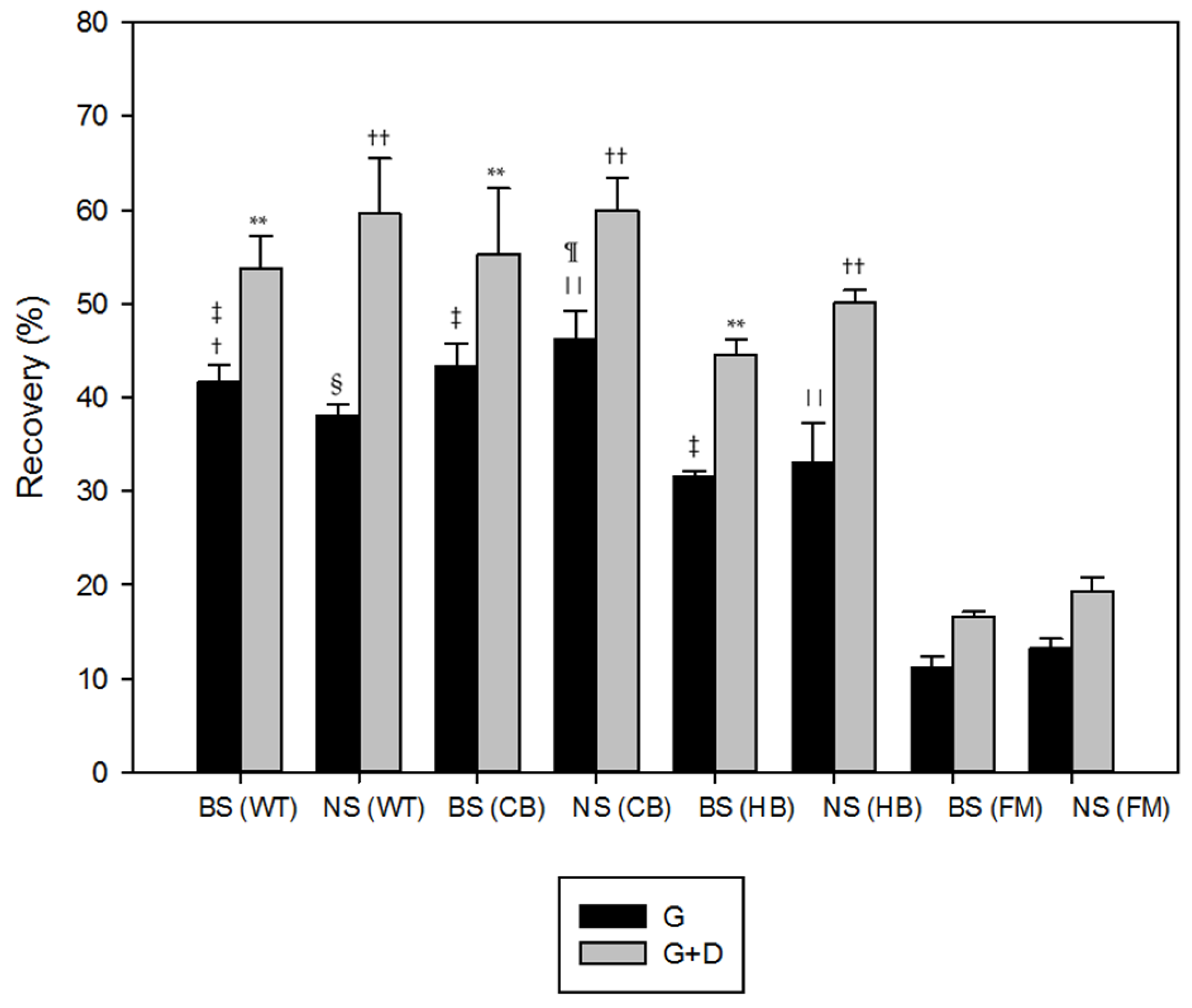
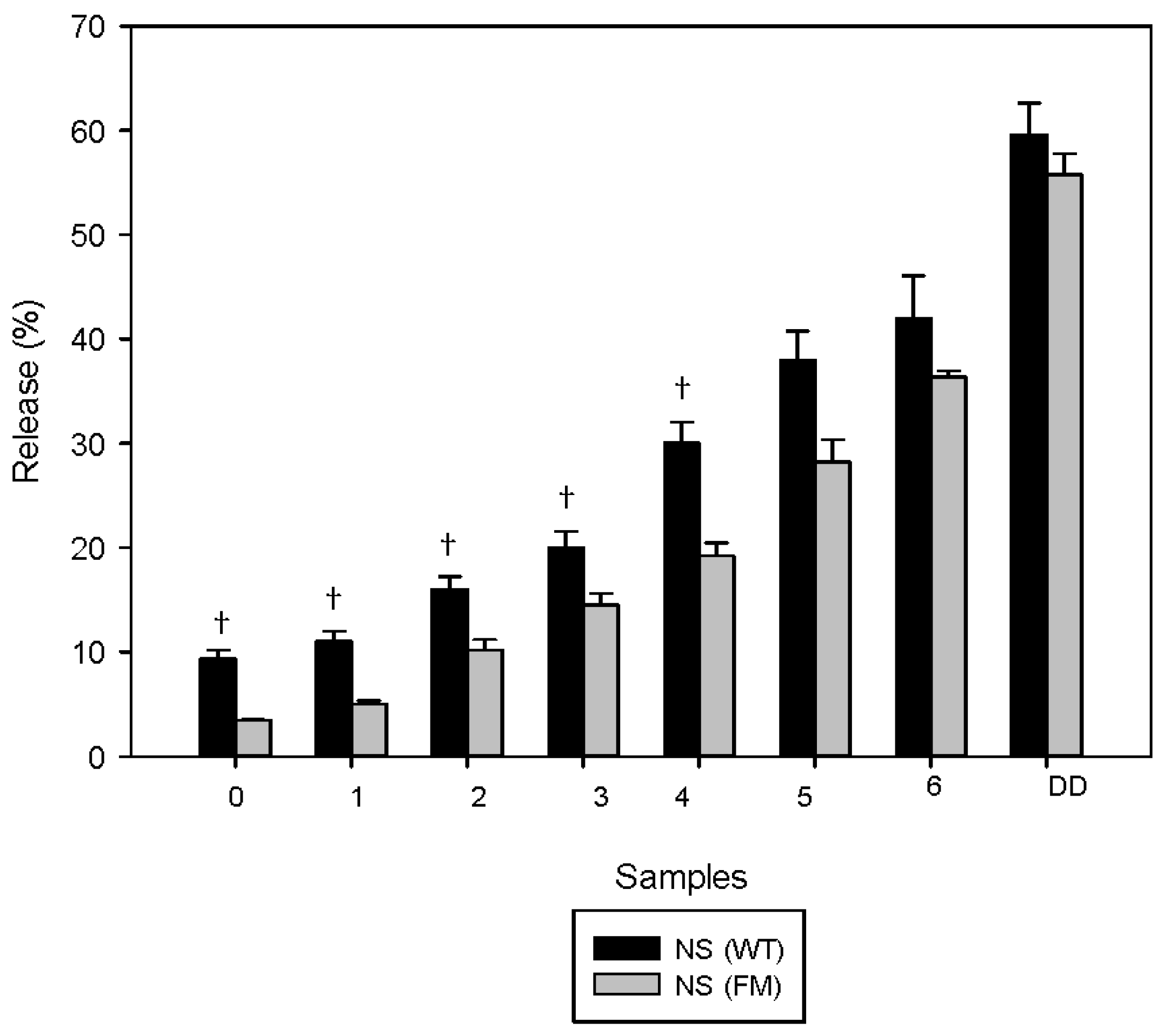
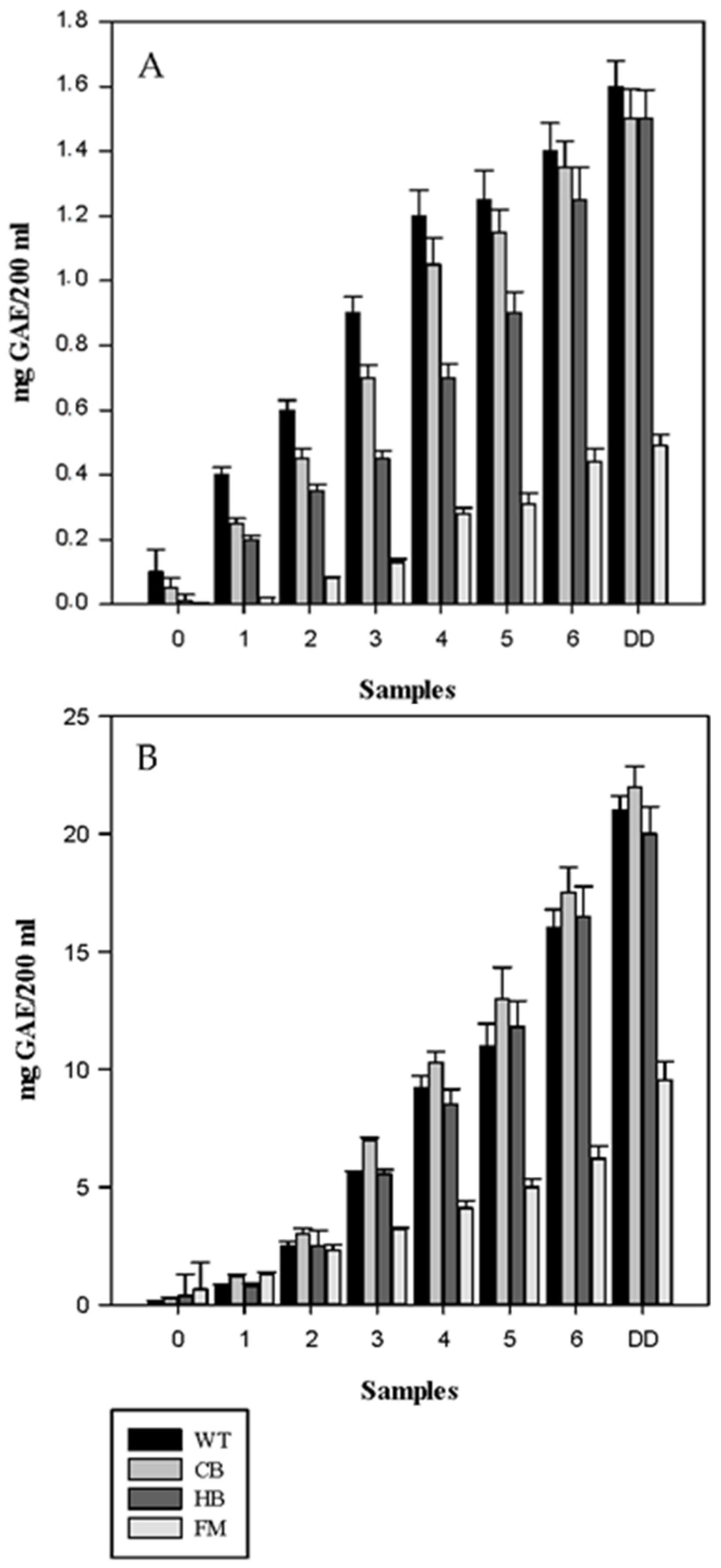
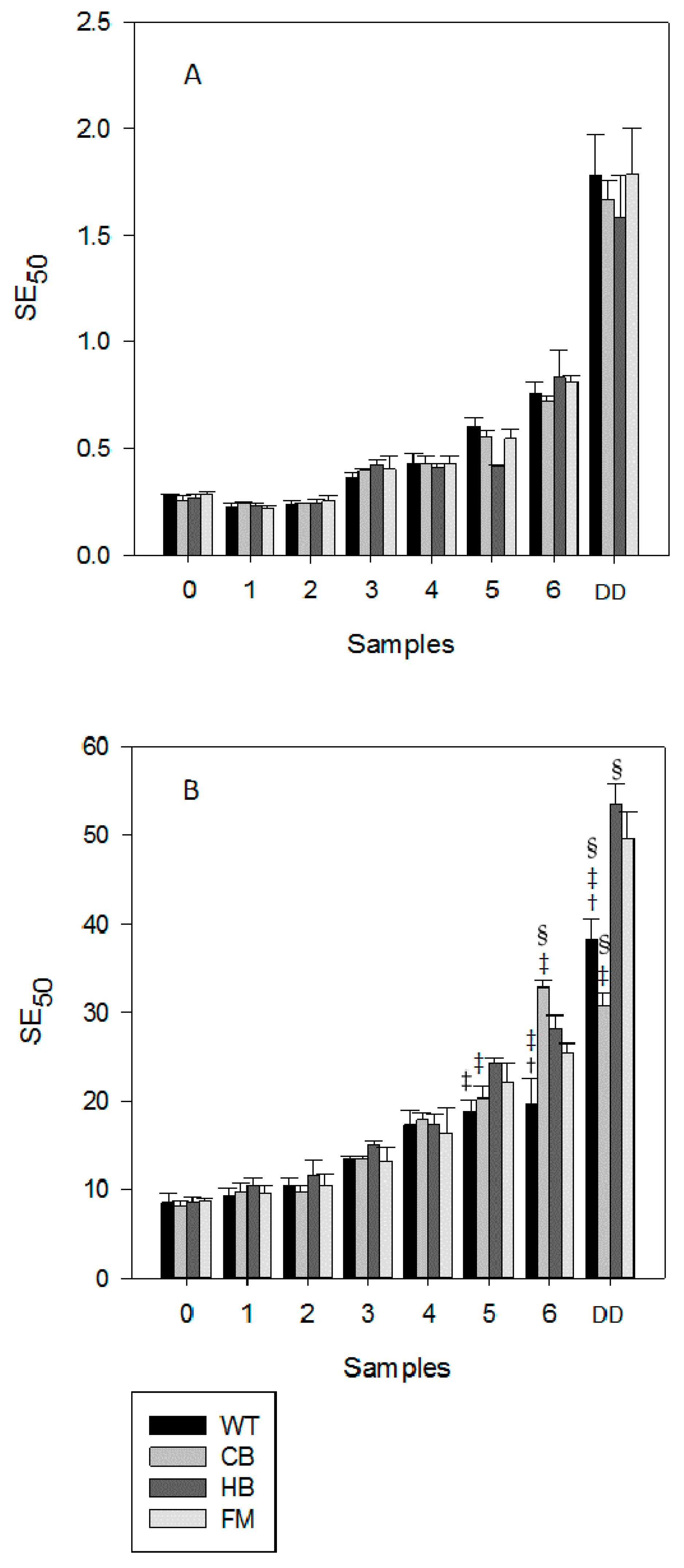
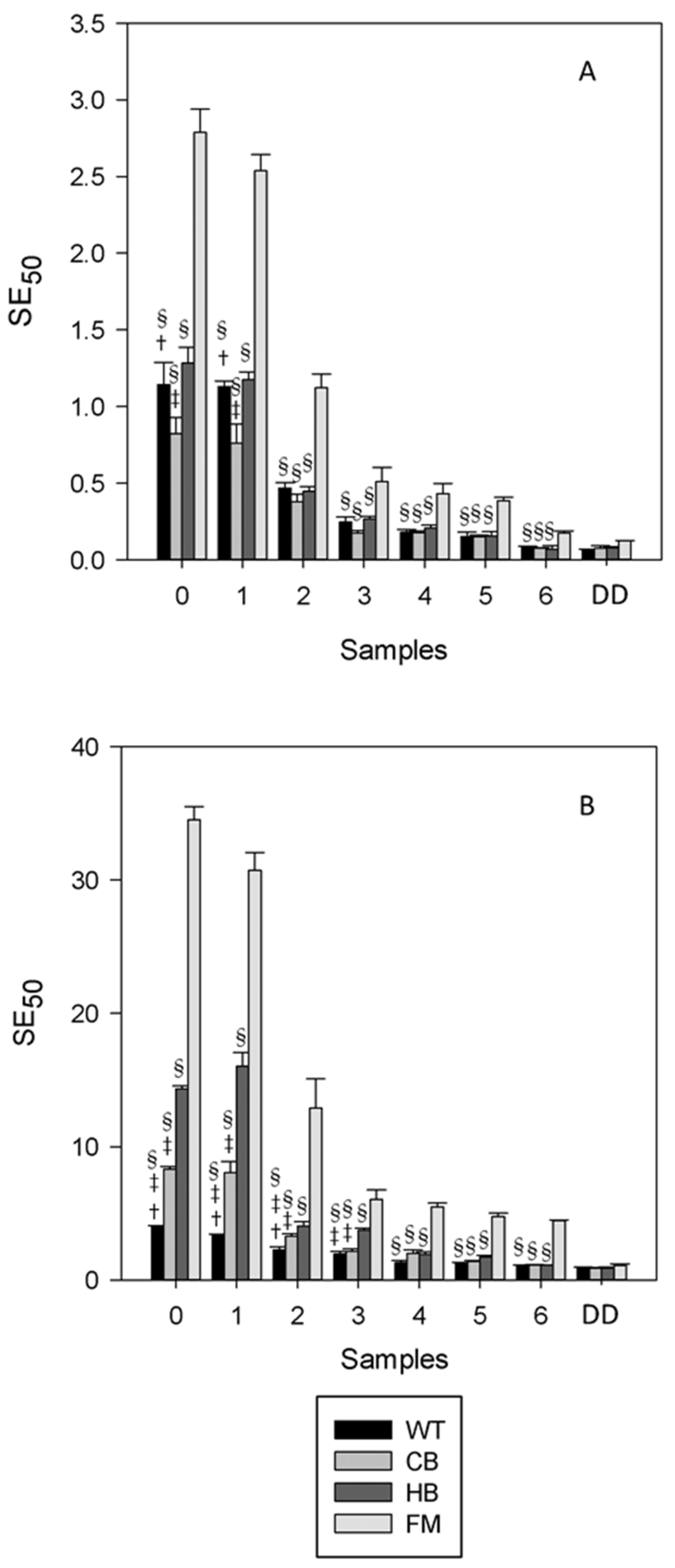
3.1. Polyphenols Release during Simulated Digestion
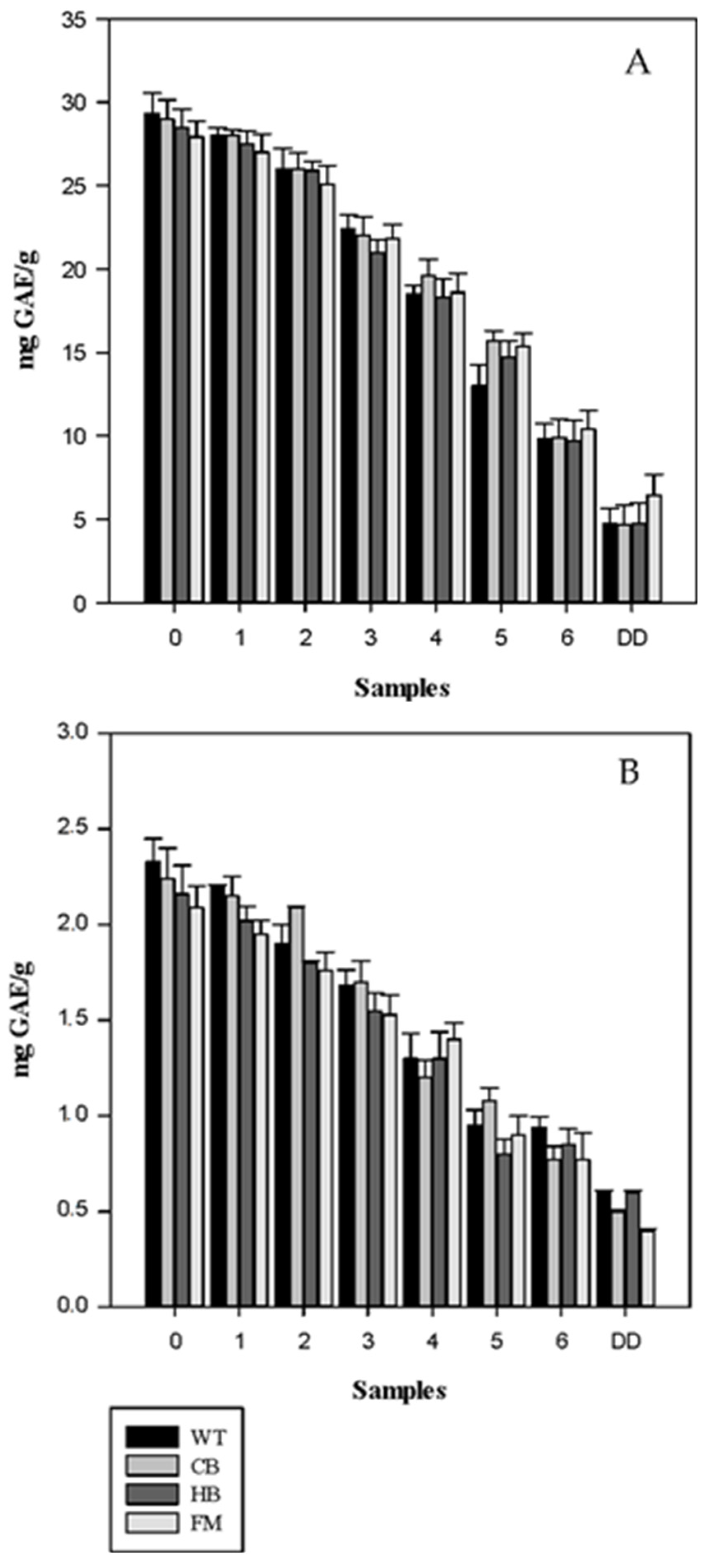
3.2. Antioxidant Profile during Digestion
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| NS | natural almond skin |
| BS | blanched almond skin |
| CB | crisp bread water |
| WT | water |
| FM | full-fat milk |
| HB | home-made biscuits |
| NSWT G | natural almond skin in water post in vitro gastric digestion |
| NSHB G | natural almond skin in home-made biscuits post in vitro gastric digestion |
| NSCB G | natural almond skin in crisp-bread post in vitro gastric digestion |
| NSFM G | natural almond skin in full-fat milk post in vitro gastric digestion |
| NSWT G + D | natural almond skin in water post in vitro gastric plus duodenal digestion |
| NSHB G + D | natural almond skin in home-made biscuits post in vitro gastric plus duodenal digestion |
| NSCB G + D | natural almond skin in crisp-bread post in vitro gastric plus duodenal digestion |
| NSFM G + D | natural almond skin in full-fat milk post in vitro gastric plus duodenal digestion |
| BSWT G | blanched almond skin in water post in vitro gastric digestion |
| BSHB G | blanched almond skin in home-made biscuits post in vitro gastric digestion |
| BSCB G | blanched almond skin in crisp-bread post in vitro gastric digestion |
| BSFM G | blanched almond skin in full-fat milk post in vitro gastric digestion |
| BSWT G + D | blanched almond skin in water post in vitro gastric plus duodenal digestion |
| BSHB G + D | blanched almond skin in home-made biscuits post in vitro gastric plus duodenal digestion |
| BSCB G + D | blanched almond skin in crisp-bread post in vitro gastric plus duodenal digestion |
| BSFM G + D | blanched almond skin in full-fat milk post in vitro gastric plus duodenal digestion |
References
- Mandalari, G.; Tomaino, A.; Rich, G.T.; Lo Curto, R.B.; Arcoraci, T.; Martorana, M.; Martorana, M.; Bisignano, C.; Saija, A.; Parkerd, M.L.; et al. Polyphenol and nutrient release from skin of almonds during simulated human digestion. Food Chem. 2010, 122, 1083–1088. [Google Scholar] [CrossRef]
- Mandalari, G.; Bisignano, C.; Genovese, T.; Mazzon, E.; Wickham, M.S.J.; Paterniti, I.; Cuzzocrea, S. Natural almond skin reduced oxidative stress and inflammation in an experimental model of inflammatory bowel disease. Int. Immunopharm. 2011, 11, 915–924. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Genovese, T.; Bisignano, C.; Mazzon, E.; Wickham, M.S.J.; di Paola, R.; Bisignano, G.; Cuzzocrea, S. Neuroprotective effects of almond skins in experimental spinal cord injury. Clin. Nutr. 2011, 30, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.O.; Blumberg, J.B. In vitro activity of almond skin polyphenols for scavenging free radicals and inducing quinine reductase. J. Agric. Food Chem. 2008, 56, 4427–4434. [Google Scholar] [CrossRef] [PubMed]
- Milbury, P.E.; Chen, C.Y.; Dolnikowski, G.G.; Blumberg, J.B. Determination of flavonoids and phenolics and their distribution in almonds. J. Agric. Food Chem. 2006, 54, 5027–5033. [Google Scholar] [CrossRef] [PubMed]
- Garrido, I.; Monagas, M.; Gómez-Cordovés, C.; Bartolomé, B. Polyphenols and antioxidant properties of almond skins: Influence of industrial processing. J. Food Sci. 2008, 73, C106–C115. [Google Scholar] [CrossRef] [PubMed]
- Benavente-García, O.; Castillo, J. Update on uses and properties of citrus flavonoids: New findings in anticancer, cardiovascular, and anti-inflammatory activity. J. Agric. Food Chem. 2008, 56, 6185–6205. [Google Scholar] [CrossRef] [PubMed]
- Vuorela, S.; Kreander, K.; Karonen, M.; Nieminen, R.; Hämäläinen, M.; Galkin, A.; Laitinen, L.; Salminen, J.P.; Moilanen, E.; Pihlaja, K.; et al. Preclinical evaluation of rapeseed, raspberry, and pine bark phenolics for health related effects. J. Agric. Food Chem. 2005, 53, 5922–5931. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Maier, C.S. The chemistry of gut microbial metabolism of polyphenols. Phytochem. Rev. 2016, 15, 425–444. [Google Scholar] [CrossRef] [PubMed]
- Tagliazucchi, D.; Verzelloni, E.; Bertolini, D.; Conte, A. In vitro bioaccessibility and antioxidant activity of grape polyphenols. Food Chem. 2010, 120, 599–606. [Google Scholar] [CrossRef]
- Stanisavljevic, N.; Samardzic, J.; Jankovic, T.; Šavikin, K.; Mojsin, M.; Topalovic, V. Antioxidant and antiproliferative activity of chokeberry juice phenolics during in vitro simulated digestion in the presence of food matrix. Food Chem. 2015, 175, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Bisignano, C.; Filocamo, A.; Chessa, S.; Saro, M.; Torre, G.; Faulks, R.M.; Dugo, P. Bioaccessibility of pistachio polyphenols, xanthophylls, and tocopherols during simulated human digestion. Nutrition 2013, 29, 338–344. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef] [PubMed]
- D’Antuono, I.; Garbetta, A.; Linsalata, V.; Minervini, F.; Cardinali, A. Polyphenols from artichoke heads (Cynara cardunculus L. subsp. scolymus Hayek): In vitro bio-accessibility, intestinal uptake and bioavailability. Food Funct. 2015, 6, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vicente, A.; Gil-Izquierdo, A.; Garcia-Viguera, C. In vitro gastrointestinal digestion study of pomegranate juice phenolic compounds, anthocyanins, and vitamin C. J. Agric. Food Chem. 2002, 50, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Roura, E.; Andres-Lacueva, C.; Estruch, R.; Mata-Bilbao, M.L.; Izquierdo-Pulido, M.; Waterhouse, A.L.; Lamuela-Raventós, R.M. Milk does not affect the bioavailability of cocoa powder flavonoid in healthy human. Ann. Nutr. Met. 2007, 51, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Tomaino, A.; Arcoraci, T.; Martorana, M.; Lo Turco, V.; Cacciola, F.; Richa, G.T.; Bisignano, C.; Saija, A.; Dugo, P.; et al. Characterization of polyphenols, lipids and dietary fibre from skins of almonds (Amygdalus communis L.). J. Food Comp. Anal. 2010, 23, 166–174. [Google Scholar] [CrossRef]
- Pitino, I.; Randazzo, C.L.; Mandalari, G.; Lo Curto, A.; Faulks, R.M.; le Marc, Y.; Bisignano, C.; Caggia, C.; Wickham, M.S. Survival of Lactobacillus rhamnosus strains in the upper gastrointestinal tract. Food Microbiol. 2010, 27, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagents. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Bonina, F.; Saija, A.; Tomaino, A.; Lo Cascio, R.; Rapisarda, P.; Dederen, J.C. In vitro antioxidant activity and in vivo photoprotective effect of a red orange extract. Int. J. Cosmet. Sci. 1998, 20, 331–342. [Google Scholar]
- Xu, L.; Diosady, L.L. Removal of phenolic compounds in the production of high-quality canola protein isolates. Food Res. Int. 2002, 35, 23–30. [Google Scholar] [CrossRef]
- Mandalari, G.; Grundy, M.M.; Grassby, T.; Parker, M.L.; Cross, K.L.; Chessa, S.; Bisignano, C.; Barreca, D.; Bellocco, E.; Laganà, G.; et al. The effects of processing and mastication on almond lipid bioaccessibility using novel methods of in vitro digestion modelling and micro-structural analysis. Br. J. Nutr. 2014, 112, 1521–1529. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Hoffmann, L.; Bohn, T. Total phenolics, flavonoids, anthocyanins and antioxidant activity following simulated gastro-intestinal digestion and dialysis of apple varieties: Bioaccessibility and potential uptake. Food Chem. 2011, 128, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Fogliano, V.; Corollaro, M.L.; Vitaglione, P.; Napolitano, A.; Ferracane, R.; Travaglia, F.; Arlorio, M.; Costabile, A.; Klinder, A.; Gibson, G. In vitro bioaccessibility and gut biotransformation of polyphenols present in the water-insoluble cocoa fraction. Mol. Nutr. Food Res. 2011, 55, S44–S55. [Google Scholar] [CrossRef] [PubMed]
- Grassby, T.; Picout, D.R.; Mandalari, G.; Faulks, R.M.; Kendall, C.W.; Rich, G.T.; Wickham, M.S.; Lapsley, K.; Ellis, P.R. Modelling of nutrient bioaccessibility in almond seeds based on the fracture properties of their cell walls. Food Funct. 2014, 5, 3096–3106. [Google Scholar] [CrossRef] [PubMed]
- Faulks, R.M.; Southon, S. Challenges to understanding and measuring carotenoid bioavailability. Biochim. Biophys. Acta 2005, 1740, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Costa, L.G.; Lean, M.E.; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis. 2010, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Swendseid, M.E. Plasma beta-carotene response in humans after meals supplemented with dietary pectin. Am. J. Clin. Nutr. 1992, 55, 96–99. [Google Scholar] [PubMed]
- Palafox-Carlos, H.; Ayala-Zavala, J.F.; González-Aguilar, G.A. The role of dietary fiber in the bioaccessibility and bioavailability of fruit and vegetable antioxidants. J. Food Sci. 2011, 76, R6–R15. [Google Scholar] [CrossRef] [PubMed]
- Lamothe, S.; Langlois, A.; Bazinet, L.; Couillard, C.; Britten, M. Antioxidant activity and nutrient release from polyphenol-enriched cheese in a simulated gastrointestinal environment. Food Funct. 2016, 7, 1634–1644. [Google Scholar] [CrossRef] [PubMed]
- Lamothe, S.; Azimy, N.; Bazinet, L.; Couillard, C.; Britten, M. Interaction of green tea polyphenols with dairy matrices in a simulated gastrointestinal environment. Food Funct. 2014, 5, 2621–2631. [Google Scholar] [CrossRef] [PubMed]
- Ortega, N.; Reguant, J.; Romero, M.P.; Macià, A.; Motilva, M.J. Effect of fat content on the digestibility and bioaccessibility of cocoa polyphenol by an in vitro digestion model. J. Agric. Food Chem. 2009, 57, 5743–5749. [Google Scholar] [CrossRef] [PubMed]
- Lesser, S.; Cermak, R. Wolffram S. Bioavailability of quercetin in pigs is influenced by the dietary fat content. J. Nutr. 2004, 134, 1508–1511. [Google Scholar] [PubMed]
- Ozdal, T.; Capanoglu, E.; Altay, F. A review on protein–phenolic interactions and associated changes. Food Res. Int. 2013, 51, 954–970. [Google Scholar] [CrossRef]
- Cebeci, F.; Şahin-Yeşilçubuk, N. The matrix effect of blueberry, oat meal and milk on polyphenols, antioxidant activity and potential bioavailability. Int. J. Food Sci. Nutr. 2014, 65, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Dietary factors affecting polyphenol bioavailability. Nutr. Rev. 2014, 72, 429–452. [Google Scholar] [CrossRef] [PubMed]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Chemistry, pharmacology and health benefits of anthocyanins. Phytother. Res. 2016, 30, 1265–1286. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rubio, A.; Gavara, R.; Lagaron, J.M. Bioactive packaging: Turning foods into healthier foods through biomaterials. Trends Food Sci. Technol. 2006, 17, 567–575. [Google Scholar] [CrossRef]
- Koga, C.C.; Lee, S.Y.; Lee, Y. Consumer acceptance of bars and gummies with unencapsulated and encapsulated resveratrol. J. Food Sci. 2016, 81, S1222–S1229. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.R.; Martins, S.; Segundo, M.A.; Reis, S. Nanoscale delivery of resveratrol towards enhancement of supplements and nutraceuticals. Nutrients 2016, 8, 131. [Google Scholar] [CrossRef] [PubMed]
| Matrix | DGM 1 | DGM 2 | DGM 3 | DGM 4 | DGM 5 | DGM 6 | DD | TDT |
|---|---|---|---|---|---|---|---|---|
| Sampling Time (min) | ||||||||
| Water | 4 | 8 | 12 | 16 | 20 | 24 | 120 | 144 |
| Home-made biscuit | 4 | 8 | 12 | 16 | 20 | 24 | 120 | 144 |
| Crisp bread | 5 | 10 | 15 | 20 | 25 | 30 | 120 | 150 |
| Full-fat milk | 6 | 12 | 18 | 24 | 30 | 36 | 120 | 156 |
| Natural Skin | |||||
| Sample | Phenolic acids | Flavonols | Flavan-3-ols | Flavanones | Total phenols |
| W | 2.15 ± 0.11 | 14.31 ± 1.05 | 2.37 ± 0.18 | 3.43 ± 0.22 | 22.26 |
| HB | 17.85 ± 1.02 | 115.30 ± 8.32 | 20.82 ± 1.65 | 30.44 ± 2.21 | 184.41 |
| CB | 12.53 ± 0.89 | 83.89 ± 4.22 | 14.72 ± 1.12 | 21.78 ± 1.44 | 132.92 |
| FM | 2.07 ± 0.12 | 13.95 ± 1.22 | 2.15 ± 0.12 | 3.22 ± 0.17 | 21.39 |
| Blanched Skin | |||||
| Sample | Phenolic acids | Flavonols | Flavan-3-ols | Flavanones | Total phenols |
| W | 0.31 ± 0.02 | 1.28 ± 0.05 | 0.62 ± 0.03 | 0.22 ± 0.02 | 2.43 |
| HB | 2.25 ± 0.14 | 9.82 ± 0.59 | 5.18 ± 0.21 | 1.73 ± 0.102 | 18.98 |
| CB | 1.78 ± 0.12 | 7.24 ± 0.35 | 3.65 ± 0.25 | 1.31 ± 0.09 | 13.98 |
| FM | 0.29 ± 0.01 | 1.19 ± 0.08 | 0.60 ± 0.02 | 0.19 ± 0.01 | 2. 27 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mandalari, G.; Vardakou, M.; Faulks, R.; Bisignano, C.; Martorana, M.; Smeriglio, A.; Trombetta, D. Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion. Nutrients 2016, 8, 568. https://doi.org/10.3390/nu8090568
Mandalari G, Vardakou M, Faulks R, Bisignano C, Martorana M, Smeriglio A, Trombetta D. Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion. Nutrients. 2016; 8(9):568. https://doi.org/10.3390/nu8090568
Chicago/Turabian StyleMandalari, Giuseppina, Maria Vardakou, Richard Faulks, Carlo Bisignano, Maria Martorana, Antonella Smeriglio, and Domenico Trombetta. 2016. "Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion" Nutrients 8, no. 9: 568. https://doi.org/10.3390/nu8090568
APA StyleMandalari, G., Vardakou, M., Faulks, R., Bisignano, C., Martorana, M., Smeriglio, A., & Trombetta, D. (2016). Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion. Nutrients, 8(9), 568. https://doi.org/10.3390/nu8090568








