Eicosapentaenoic and Docosahexaenoic Acid-Enriched High Fat Diet Delays Skeletal Muscle Degradation in Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Experiment
2.2. Diets
2.3. Muscle Composition
2.4. RNA Isolation and Quality Assurance
2.5. Western Blot Analysis
2.6. Microarray—Data Acquisition and Analysis
2.7. Fatty Acid Analysis
2.8. Statistical Analysis of Western Blot and Fatty Acid Analyses
3. Results
3.1. Effect of EPA and DHA vs Corn Oil on gSkM Weight and Transcriptome
3.2. gSkM Fatty Acid Composition
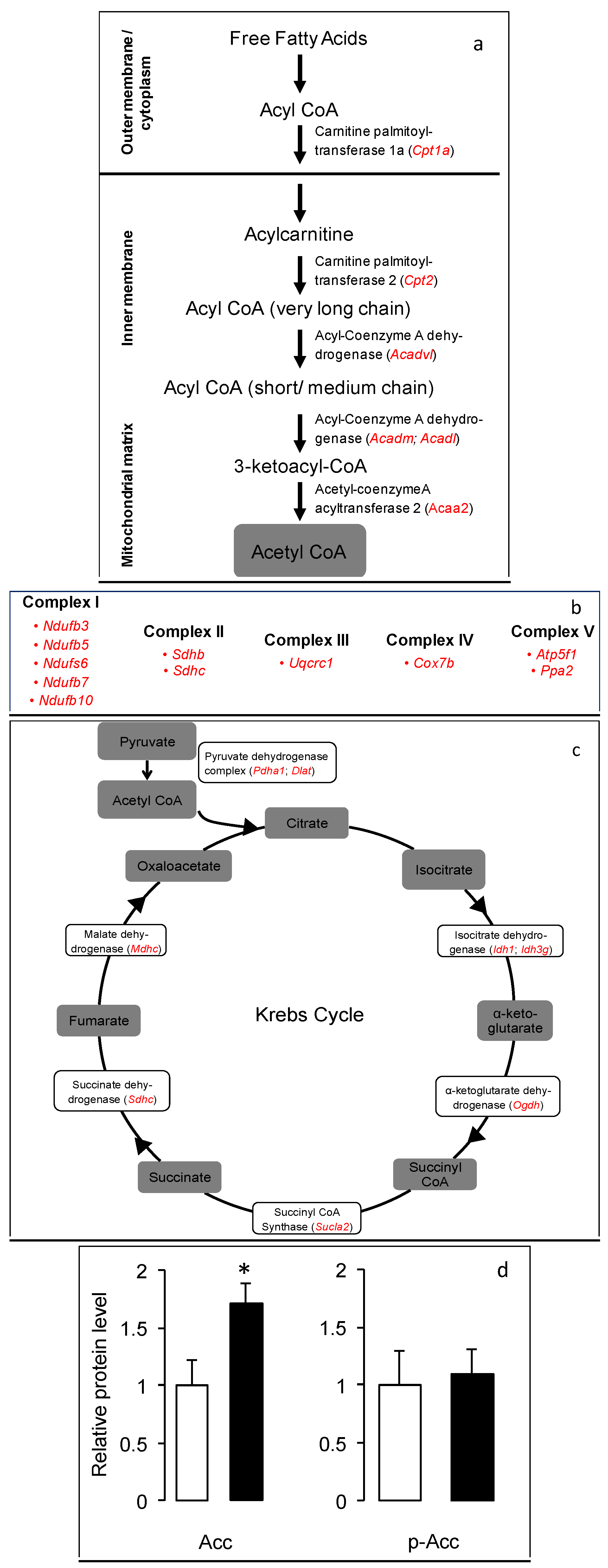
3.3. Increased Mitochondrial B—Oxidation and Oxidative Phosphorylation in the Gskm of Mice Fed HFD-ED
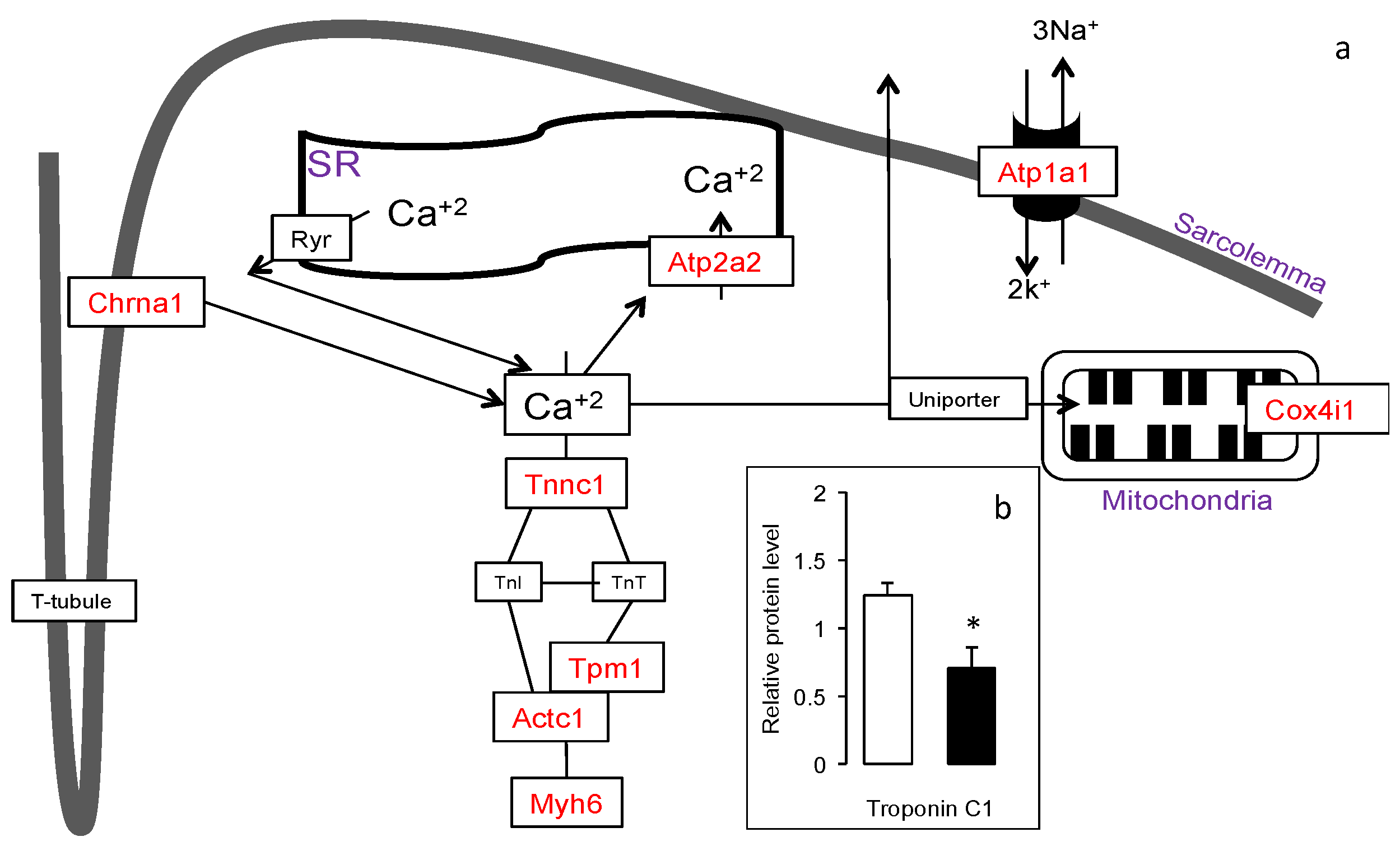
3.4. Increased Expression of Muscle Contraction Pathway Genes in the Gskm of Mice Fed HFD-ED
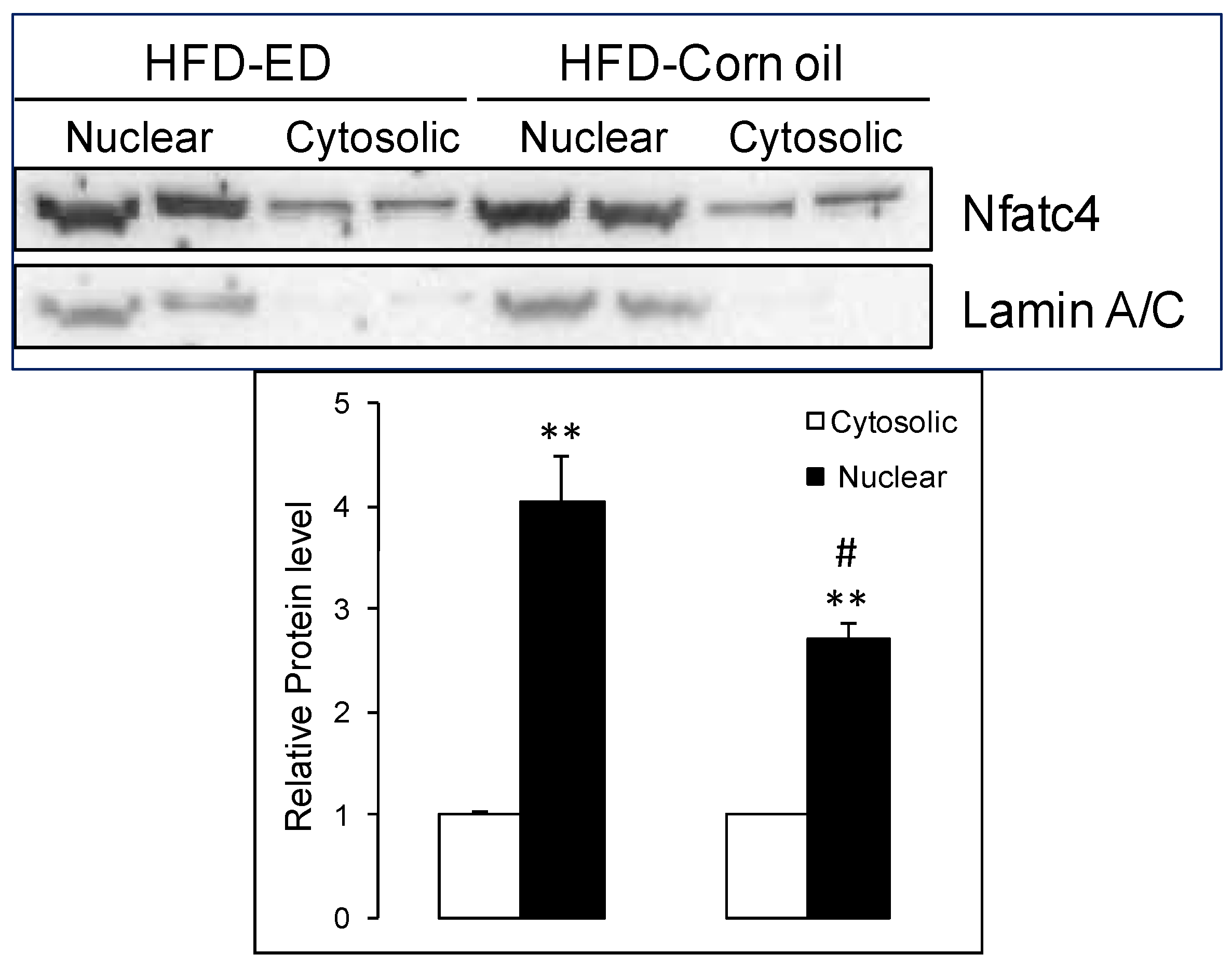
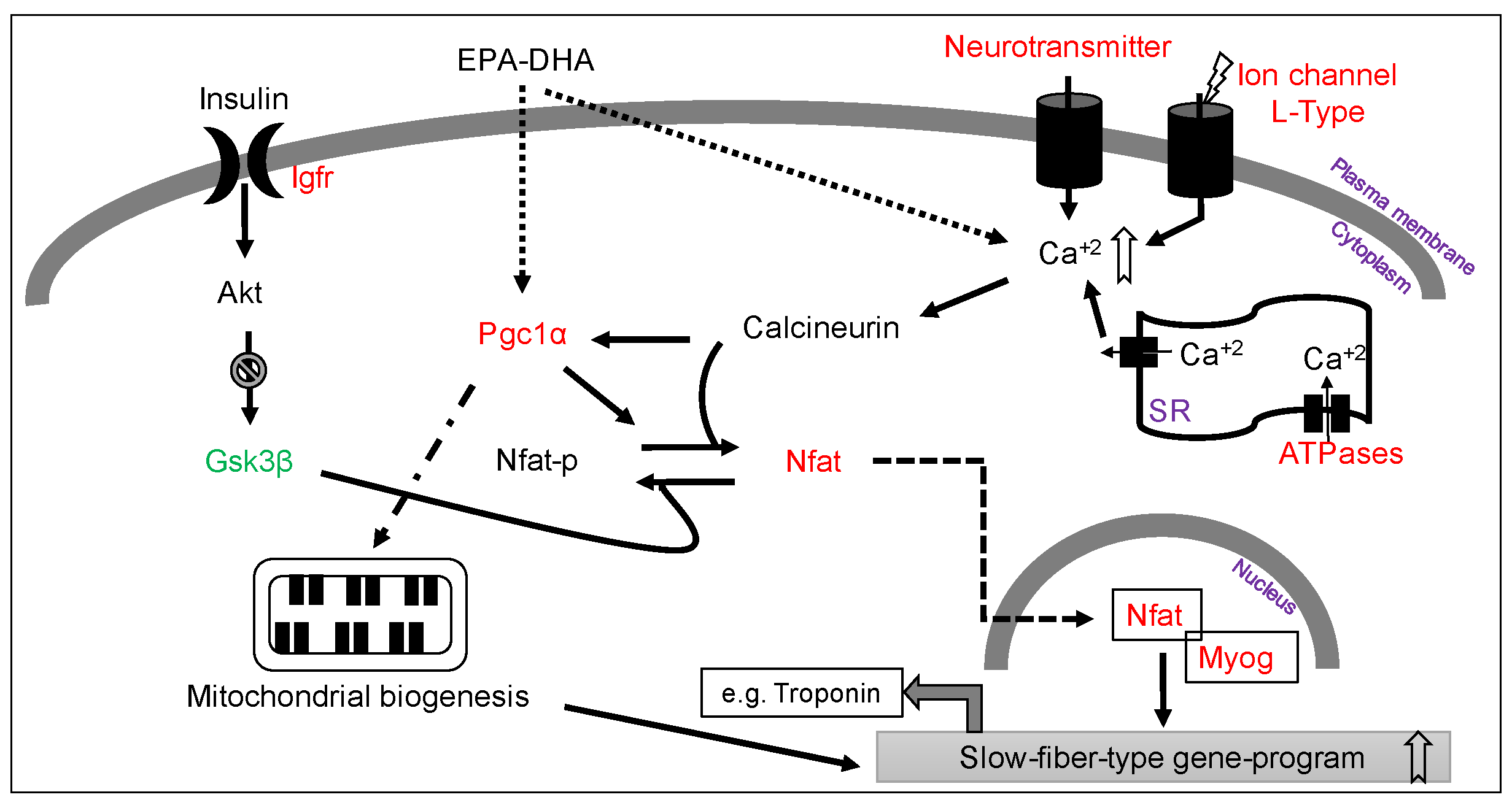
3.5. Increased Slow-Fiber-Specific Gene Expression Program in the Gskm of Mice Fed HFD-ED
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Authors Contributions
Conflicts of Interests
References and Notes
- World Health Organization. Obesity and Overweight Fact Sheet N°311; World Health Organization Media Centre: Geneva, Switzerland, 2015. [Google Scholar]
- Lauretani, F.; Russo, C.R.; Bandinelli, S.; Bartali, B.; Cavazzini, C.; Di Iorio, A.; Corsi, A.M.; Rantanen, T.; Guralnik, J.M.; Ferrucci, L. Age-associated changes in skeletal muscles and their effect on mobility: An operational diagnosis of sarcopenia. J. Appl. Physiol. 2003, 95, 1851–1860. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Chilibeck, P.D. Differences in size, strength, and power of upper and lower body muscle groups in young and older men. J. Gerontol. 2005, 60, 148–156. [Google Scholar] [CrossRef]
- Von Haehling, S.; Morley, J.E.; Anker, S.D. An overview of sarcopenia: Facts and numbers on prevalence and clinical impact. J. Cachexia Sarcopenia Muscle 2010, 1, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, I.H. Sarcopenia: Origins and clinical relevance. J. Nutr. 1997, 127, 990S–991S. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Johannesdottir, S.A.; Lemeshow, S.; Lash, T.L.; Ulrichsen, S.P.; Botker, H.E.; Sorensen, H.T. Obesity in young men, and individual and combined risks of type 2 diabetes, cardiovascular morbidity and death before 55 years of age: A danish 33-year follow-up study. BMJ Open 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Roubenoff, R. Sarcopenic obesity: The confluence of two epidemics. Obes. Res. 2004, 12, 887–888. [Google Scholar] [CrossRef] [PubMed]
- Schaap, L.A.; Pluijm, S.M.; Deeg, D.J.; Harris, T.B.; Kritchevsky, S.B.; Newman, A.B.; Colbert, L.H.; Pahor, M.; Rubin, S.M.; Tylavsky, F.A.; et al. Higher inflammatory marker levels in older persons: Associations with 5-year change in muscle mass and muscle strength. J. Gerontol. 2009, 64, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Pahor, M.; Taaffe, D.R.; Goodpaster, B.H.; Simonsick, E.M.; Newman, A.B.; Nevitt, M.; Harris, T.B. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: The health abc study. J. Gerontol. 2002, 57, M326–M332. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, Z.; Zhang, J.; Hua, J.; He, W.; Zhu, S. Estimation of total body skeletal muscle mass in chinese adults: Prediction model by dual-energy X-ray absorptiometry. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Wang, Z.; Heymsfield, S.B.; Baumgartner, R.N.; Gallagher, D. Total-body skeletal muscle mass: Estimation by a new dual-energy X-ray absorptiometry method. Am. J. Clin. Nutr. 2002, 76, 378–383. [Google Scholar]
- Meyer, C.; Dostou, J.M.; Welle, S.L.; Gerich, J.E. Role of human liver, kidney, and skeletal muscle in postprandial glucose homeostasis. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Julliand, S.; Reeds, D.N.; Sinacore, D.R.; Klein, S.; Mittendorfer, B. Fish oil-derived n-3 pufa therapy increases muscle mass and function in healthy older adults. Am. J. Clin. Nutr. 2015, 102, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.W.; Saito, H.; Trocki, O.; Ogle, C.K. The importance of lipid type in the diet after burn injury. Ann. Surg. 1986, 204, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gingras, A.A.; White, P.J.; Chouinard, P.Y.; Julien, P.; Davis, T.A.; Dombrowski, L.; Couture, Y.; Dubreuil, P.; Myre, A.; Bergeron, K.; et al. Long-chain omega-3 fatty acids regulate bovine whole-body protein metabolism by promoting muscle insulin signalling to the akt-mtor-s6k1 pathway and insulin sensitivity. J. Physiol. 2007, 579, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Fetterman, J.W., Jr.; Zdanowicz, M.M. Therapeutic potential of n-3 polyunsaturated fatty acids in disease. Am. J Health Syst. Pharm. 2009, 66, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Gabrielsson, B.G.; Wikstrom, J.; Jakubowicz, R.; Marmon, S.K.; Carlsson, N.G.; Jansson, N.; Gan, L.M.; Undeland, I.; Lonn, M.; Holmang, A.; et al. Dietary herring improves plasma lipid profiles and reduces atherosclerosis in obese low-density lipoprotein receptor-deficient mice. Int. J. Mol. Med. 2012, 29, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Nookaew, I.; Gabrielsson, B.G.; Holmang, A.; Sandberg, A.S.; Nielsen, J. Identifying molecular effects of diet through systems biology: Influence of herring diet on sterol metabolism and protein turnover in mice. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Rodacki, C.L.; Rodacki, A.L.; Pereira, G.; Naliwaiko, K.; Coelho, I.; Pequito, D.; Fernandes, L.C. Fish-oil supplementation enhances the effects of strength training in elderly women. Am. J. Clin. Nutr. 2012, 95, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Soni, N.K.; Nookaew, I.; Sandberg, A.S.; Gabrielsson, B.G. Eicosapentaenoic and docosahexaenoic acid-enriched high fat diet delays the development of fatty liver in mice. Lipids Health Dis. 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Amrutkar, M.; Cansby, E.; Nunez-Duran, E.; Piraz zi, C.; Stahlman, M.; Stenfeldt, E.; Smith, U.; Boren, J.; Mahlapuu, M. Protein kinase stk25 regulates hepatic lipid partitioning and progression of liver steatosis and nash. FASEB J. 2015, 29, 1564–1576. [Google Scholar] [CrossRef] [PubMed]
- For More Information about ncbi refseq, Please Go to http://ftp.Ncbi.Nih.Gov/refseq/release/.
- For More Information about meebo, Please Go to http://www.Arrays.Ucsf.Edu/archive/meebo.Html.
- Functional Annotation of Mouse. Available online: http://fantom2.Gsc.Riken.Jp/ (accessed on 25 August 2016).
- Du, P.; Kibbe, W.A.; Lin, S.M. Lumi: A pipeline for processing illumina microarray. Bioinformatics 2008, 24, 1547–1548. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucl. Acids Res. 2015, 43. [Google Scholar] [CrossRef]
- Luo, W.; Friedman, M.S.; Shedden, K.; Hankenson, K.D.; Woolf, P.J. Gage: Generally applicable gene set enrichment for pathway analysis. BMC Bioinformatics 2009, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Brouwer, C. Pathview: An R/Bioconductor package for pathway-based data integration and visualization. Bioinformatics 2013, 29. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Kaluzny, M.A.; Duncan, L.A.; Merritt, M.V.; Epps, D.E. Rapid separation of lipid classes in high yield and purity using bonded phase columns. J. Lipid Res. 1985, 26, 135–140. [Google Scholar] [PubMed]
- Lepage, G.; Roy, C.C. Improved recovery of fatty acid through direct transesterification without prior extraction or purification. J. Lipid. Res. 1984, 25, 1391–1396. [Google Scholar] [PubMed]
- Tuazon, M.A.; Henderson, G.C. Fatty acid profile of skeletal muscle phospholipid is altered in mdx mice and is predictive of disease markers. Metable 2012, 61, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Lowell, B.B.; Shulman, G.I. Mitochondrial dysfunction and type 2 diabetes. Science 2005, 307, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Dufour, S.; Befroy, D.; Garcia, R.; Shulman, G.I. Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N. Engl. J. Med. 2004, 350, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Patti, M.E.; Butte, A.J.; Crunkhorn, S.; Cusi, K.; Berria, R.; Kashyap, S.; Miyazaki, Y.; Kohane, I.; Costello, M.; Saccone, R.; et al. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of pgc1 and nrf1. Proc. Natl. Acad. Sci. USA 2003, 100, 8466–8471. [Google Scholar] [CrossRef] [PubMed]
- Sparks, L.M.; Xie, H.; Koza, R.A.; Mynatt, R.; Hulver, M.W.; Bray, G.A.; Smith, S.R. A high-fat diet coordinately downregulates genes required for mitochondrial oxidative phosphorylation in skeletal muscle. Diabetes 2005, 54, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Mootha, V.K.; Lindgren, C.M.; Eriksson, K.F.; Subramanian, A.; Sihag, S.; Lehar, J.; Puigserver, P.; Carlsson, E.; Ridderstrale, M.; Laurila, E.; et al. Pgc-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 2003, 34, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.S.; Savage, D.B.; Abu-Elheiga, L.; Liu, Z.X.; Kim, S.; Kulkarni, A.; Distefano, A.; Hwang, Y.J.; Reznick, R.M.; Codella, R.; et al. Continuous fat oxidation in acetyl-coa carboxylase 2 knockout mice increases total energy expenditure, reduces fat mass, and improves insulin sensitivity. Proc. Natl. Acad. Sci. USA 2007, 104, 16480–16485. [Google Scholar] [CrossRef] [PubMed]
- Bruce, C.R.; Hoy, A.J.; Turner, N.; Watt, M.J.; Allen, T.L.; Carpenter, K.; Cooney, G.J.; Febbraio, M.A.; Kraegen, E.W. Overexpression of carnitine palmitoyltransferase-1 in skeletal muscle is sufficient to enhance fatty acid oxidation and improve high-fat diet-induced insulin resistance. Diabetes 2009, 58, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Schwerzmann, K.; Hoppeler, H.; Kayar, S.R.; Weibel, E.R. Oxidative capacity of muscle and mitochondria: Correlation of physiological, biochemical, and morphometric characteristics. Proc. Natl. Acad. Sci. USA 1989, 86, 1583–1587. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Befroy, D.; Dufour, S.; Dziura, J.; Ariyan, C.; Rothman, D.L.; DiPietro, L.; Cline, G.W.; Shulman, G.I. Mitochondrial dysfunction in the elderly: Possible role in insulin resistance. Science 2003, 300, 1140–1142. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, M.; Kalyanasundaram, A. Serca pump isoforms: Their role in calcium transport and disease. Muscle Nerve 2007, 35, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Radzyukevich, T.L.; Neumann, J.C.; Rindler, T.N.; Oshiro, N.; Goldhamer, D.J.; Lingrel, J.B.; Heiny, J.A. Tissue-specific role of the na,k-atpase alpha2 isozyme in skeletal muscle. J. Biol. Chem. 2013, 288, 1226–1237. [Google Scholar] [CrossRef] [PubMed]
- Dunn, S.E.; Burns, J.L.; Michel, R.N. Calcineurin is required for skeletal muscle hypertrophy. J. Biol. Chem. 1999, 274, 21908–21912. [Google Scholar] [CrossRef] [PubMed]
- Dunn, S.E.; Chin, E.R.; Michel, R.N. Matching of calcineurin activity to upstream effectors is critical for skeletal muscle fiber growth. J. Cell. Biol. 2000, 151, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Dunn, S.E.; Simard, A.R.; Bassel-Duby, R.; Williams, R.S.; Michel, R.N. Nerve activity-dependent modulation of calcineurin signaling in adult fast and slow skeletal muscle fibers. J. Biol. Chem. 2001, 276, 45243–45254. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.M.; Taylor, J.M.; Tapscott, S.J.; Gurley, C.M.; Carter, W.J.; Peterson, C.A. Selective accumulation of myod and myogenin mrnas in fast and slow adult skeletal muscle is controlled by innervation and hormones. Development 1993, 118, 1137–1147. [Google Scholar]
- Lin, J.; Wu, H.; Tarr, P.T.; Zhang, C.Y.; Wu, Z.; Boss, O.; Michael, L.F.; Puigserver, P.; Isotani, E.; Olson, E.N.; et al. Transcriptional co-activator pgc-1 alpha drives the formation of slow-twitch muscle fibres. Nature 2002, 418, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Vega, R.B.; Huss, J.M.; Kelly, D.P. The coactivator pgc-1 cooperates with peroxisome proliferator-activated receptor alpha in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes. Mol. Cell Biol. 2000, 20, 1868–1876. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Puigserver, P.; Andersson, U.; Zhang, C.; Adelmant, G.; Mootha, V.; Troy, A.; Cinti, S.; Lowell, B.; Scarpulla, R.C.; et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999, 98, 115–124. [Google Scholar] [CrossRef]
- Puigserver, P.; Wu, Z.; Park, C.W.; Graves, R.; Wright, M.; Spiegelman, B.M. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 1998, 92, 829–839. [Google Scholar] [CrossRef]
- Noland, R.C.; Woodlief, T.L.; Whitfield, B.R.; Manning, S.M.; Evans, J.R.; Dudek, R.W.; Lust, R.M.; Cortright, R.N. Peroxisomal-mitochondrial oxidation in a rodent model of obesity-associated insulin resistance. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E986–E1001. [Google Scholar] [CrossRef] [PubMed]
- Neal, J.W.; Clipstone, N.A. Glycogen synthase kinase-3 inhibits the DNA binding activity of NFATc. J. Biol. Chem. 2001, 276, 3666–3673. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L.; Karan, S.; Kenny, A.M. Effect of dehydroepiandrosterone on muscle strength and physical function in older adults: A systematic review. J. Am. Geriatr. Soc. 2011, 59, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Emmelot-Vonk, M.H.; Verhaar, H.J.; Pour, H.R.N.; Aleman, A.; Lock, T.M.; Bosch, J.L.; Grobbee, D.E.; van der Schouw, Y.T. Effect of testosterone supplementation on functional mobility, cognition, and other parameters in older men: A randomized controlled trial. J. Am. Med. Assoc. 2008, 299, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Giannoulis, M.G.; Sonksen, P.H.; Umpleby, M.; Breen, L.; Pentecost, C.; Whyte, M.; McMillan, C.V.; Bradley, C.; Martin, F.C. The effects of growth hormone and/or testosterone in healthy elderly men: A randomized controlled trial. J. Clin. Endocrinol. Metab. 2006, 91, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.P.; Solomon, T.P.; Wojta, D.M.; Staten, M.A.; Holloszy, J.O. Effects of 7 days of exercise training on insulin sensitivity and responsiveness in type 2 diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E151–E156. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Control | HFD-ED | HFD-Corn oil |
|---|---|---|---|
| Total number of animals; n | 9 | 12 | 12 |
| Initial body weight (g) | 27.5 ± 0.8 | 28.6 ± 0.8 | 24.3 ± 0.5 |
| Final body weight (g) | 31.4 ± 1 | 33.8 ± 0.8 | 36.2 ± 0.9 |
| Change in body weight (g) | 3.9 ± 0.5 a | 5.2 ± 0.3 a | 8.6 ± 0.5 b |
| plasma cholesterol (mmol/L) | 5.6 ± 0.4 | 5.5 ± 0.4 | 5.2 ± 0.6 |
| plasma triglyceride (mmol/L) | 0.9 ± 0.1 a,b | 1.1 ± 0.1 a | 0.7 ± 0.1 b |
| Ingredient (g/100 g Diet) | Control | HFD-ED | HFD-CO | |
|---|---|---|---|---|
| Protein | Casein | 22.2 | 25.6 | 25.6 |
| Carbohydrates | Sucrose | 5.0 | 10.0 | 10.0 |
| Corn starch | 56.0 | 34.8 | 34.8 | |
| Cellulose | 5.0 | 5.8 | 5.8 | |
| Fat | Total | 5.0 | 15.0 | 15.0 |
| Corn oil | 2.5 | 3.0 | 5.0 | |
| Coconut oil | 2.5 | 10.0 | 10.0 | |
| EPAX oils a | 0 | 2.0 | 0 | |
| Minerals b | 2.0 | 2.5 | 2.5 | |
| Miconutrients c | 3.0 | 3.0 | 3.0 | |
| Choline bitartrate | 1.6 | 2.0 | 2.0 | |
| Cholesterol | 0 | 1.0 | 1.0 | |
| Methionine | 0.2 | 0.3 | 0.3 | |
| Energy content (kJ/100 g) | 1599 | 1752 | 1752 | |
| Protein E% | 24 | 25 | 25 | |
| Carbohydrate E% | 65 | 44 | 44 | |
| Fat E% | 12 | 32 | 32 | |
| Fatty acid composition d (mg/g diet) | C10:0 | 0.20 | 1.47 | 1.33 |
| C12:0 | 2.37 | 7.58 | 7.72 | |
| C14:0 | 1.54 | 4.58 | 4.78 | |
| C16:0 | 1.90 | 3.44 | 3.59 | |
| C18:0 | 0.68 | 2.26 | 2.49 | |
| SFA | 6.70 | 19.33 | 19.91 | |
| C18:1n9 | 2.82 | 4.80 | 5.26 | |
| MUFA | 2.82 | 4.80 | 5.26 | |
| C18:2n6 | 3.62 | 5.03 | 7.36 | |
| C18:3n6 | 0.12 | 0.22 | 0.26 | |
| n-6 PUFA | 3.74 | 5.26 | 7.62 | |
| C20:5n3 (EPA) | 0.00 | 2.03 | 0.01 | |
| C22:6n3 (DHA) | 0.00 | 4.58 | 0.01 | |
| n-3 PUFA | 0.00 | 6.61 | 0.02 | |
| Lipid Fraction | mg/g | mg/g | mg/g | % value | % value | % value |
|---|---|---|---|---|---|---|
| Neutral lipids | Control | HFD-ED | HFD-Corn oil | Control | HFD-ED | HFD-Corn oil |
| C12:0 | 0.12 ± 0.06 a | 1.51 ± 0.42 b | 2.87 ± 0.41 c | 0.44 ± 0.18 a | 3.93 ± 0.21 b | 4.02 ± 0.33 b |
| C14:0 | 1.18 ± 0.24 a | 3.15 ± 0.76 | 4.81 ± 0.49 b | 4.35 ± 0.48 a | 8.19 ± 0.52 b | 6.74 ± 0.23 c |
| C16:0 | 8.11 ± 2.31 a | 9.75 ± 2.28 a | 19.01 ± 2.11 b | 29.93 ± 1.78 | 25.35 ± 1.51 | 26.63 ± 2.34 |
| C18:0 | 0.55 ± 0.05 | 0.65 ± 0.05 | 0.68 ± 0.04 | 2.03 ± 0.48 a | 1.69 ± 0.4 a | 0.95 ± 0.09 b |
| C18:1 | 13.11 ± 3.05 a | 14.86 ± 3.45 a | 29.2 ± 2.19 b | 48.38 ± 0.81 a | 38.64 ± 1.06 b | 40.91 ± 1.6 b |
| C18:2 n6 | 3.75 ± 0.78 a | 7.29 ± 1.89 a | 14.55 ± 1.25 b | 13.84 ± 0.67 a | 18.95 ± 0.9 b | 20.38 ± 0.73 b |
| C18:3 n3 | 0.07 ± 0.04 | 0.07 ± 0.03 | 0.09 ± 0.02 | 0.26 ± 0.08 | 0.18 ± 0.04 | 0.13 ± 0.03 |
| C20:3 n6 | 0.02 ± 0.01 | 0.01 ± 0 a | 0.03 ± 0 b | 0.07 ± 0.06 | 0.03 ± 0 | 0.04 ± 0 |
| C22:5 n3 | 0.03 ± 0.01 | 0.06 ± 0.01 | 0 ± 0 | 0.11 ± 0.09 | 0.16 ± 0.01 a | 0 ± 0 b |
| C22:6 n3 | 0.14 ± 0.02 a | 1.1 ± 0.09 b | 0.14 ± 0.02 a | 0.52 ± 0.25 a | 2.86 ± 0.68 b | 0.2 ± 0.04 a |
| Total neutral lipids | 27.1 ± 6.48 a | 38.46 ± 8.89 a | 71.38 ± 6.13 b | |||
| Free fatty acids | Control | HFD-ED | HFD-Corn oil | Control | HFD-ED | HFD-Corn oil |
| C12:0 | 0.13 ± 0.04 | 0.16 ± 0.04 a | 0.04 ± 0.01 b | 6.6 ± 2.11 | 8.21 ± 2.26 a | 1.61 ± 0.43 b |
| C16:0 | 0.52 ± 0.07 a | 0.64 ± 0.02 | 0.78 ± 0.04 b | 26.4 ± 3.14 a | 32.82 ± 1.34 b | 31.33 ± 2.03 b |
| C16:1 | 0.12 ± 0.03 | 0.09 ± 0.03 | 0.15 ± 0.05 | 6.09 ± 1.17 | 4.62 ± 1.3 | 6.02 ± 1.58 |
| C18:1 | 1.07 ± 0.09 | 0.89 ± 0.07 a | 1.29 ± 0.12 b | 54.31 ± 4.03 a | 45.64 ± 1.96 b | 51.81 ± 1.68 |
| C18:2 n6 | 0 ± 0 a | 0 ± 0 a | 0.11 ± 0.02 b | 0 ± 0 a | 0 ± 0 a | 4.42 ± 0.46 b |
| C18:3 n3 | 0.09 ± 0 | 0.07 ± 0.01 | 0.07 ± 0.01 | 4.57 ± 0.64 a | 3.59 ± 0.51 | 2.81 ± 0.27 b |
| C20:3 n3 | 0.04 ± 0 a | 0 ± 0 b | 0.04 ± 0.01 a | 2.03 ± 0.27 a | 0 ± 0 b | 1.61 ± 0.24 c |
| C22:6 n3 | 0 ± 0 a | 0.09 ± 0.03 b | 0 ± 0 a | 0 ± 0 a | 4.62 ± 1.25 b | 0 ± 0.14 a |
| Total free fatty acids | 1.97 ± 0.17 | 1.95 ± 0.12 | 2.49 ± 0.21 | |||
| Phospholipids | Control | HFD-ED | HFD-Corn oil | Control | HFD-ED | HFD-Corn oil |
| C14:0 | 0.33 ± 0.02 a | 0.83 ± 0.04 b | 0.64 ± 0.07 | 2.17 ± 0.14 a | 6.14 ± 0.28 b | 4.7 ± 0.49 c |
| C16:0 | 5.06 ± 0.06 a | 4.93 ± 0.09 | 4.73 ± 0.06 b | 33.29 ± 0.76 a | 36.49 ± 0.55 b | 34.75 ± 1.37 |
| C18:0 | 1.97 ± 0.33 | 2.65 ± 0.18 | 2.01 ± 0.26 | 12.96 ± 2.38 | 19.62 ± 1.41 | 14.77 ± 2.02 |
| C18:1 | 3.67 ± 0.68 a | 0.39 ± 0.26 b | 2.07 ± 0.44 a | 24.14 ± 3.71 a | 2.89 ± 1.81 b | 15.21 ± 3.12 a |
| C18:2 n6 | 1.73 ± 0.06 a | 0.58 ± 0.09 b | 2.13 ± 0.05 c | 11.38 ± 0.28 a | 4.29 ± 0.61 b | 15.65 ± 0.6 c |
| C20:3 n6 | 0.05 ± 0 a | 0 ± 0 b | 0.06 ± 0.01 a | 0.33 ± 0 a | 0 ± 0 b | 0.44 ± 0.04 a |
| C20:4 n6 | 0.57 ± 0.01 a | 0.1 ± 0 b | 0.55 ± 0.01 a | 3.75 ± 0.12 a | 0.74 ± 0.01 b | 4.04 ± 0.28a |
| C20:5 n3 | 0 ± 0 | 0.04 ± 0 | 0.02 ± 0.02 | 0 ± 0 | 0.3 ± 0.01 | 0.15 ± 0.15 |
| C22:3 n6 | 0.54 ± 0.02 a | 0.07 ± 0 b | 0.26 ± 0.03 c | 3.55 ± 0.18 a | 0.52 ± 0.01 b | 1.91 ± 0.23 c |
| C22:5 n3 | 0.05 ± 0 a | 0.11 ± 0 b | 0.06 ± 0.01 a | 0.33 ± 0.02 a | 0.81 ± 0.02 b | 0.44 ± 0.05 c |
| C22:6 n3 | 1.23 ± 0.03 a | 3.45 ± 0.06 b | 1.07 ± 0.1 a | 8.09 ± 0.3 a | 25.54 ± 0.53 b | 7.86 ± 0.73 a |
| Total phospholipids | 15.2 ± 0.47 a | 13.15 ± 0.34 b | 13.61 ± 0.45 b | |||
| Total Fat content | 44.26 ± 6.99 | 53.56 ± 9 | 87.47 ± 6.45 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soni, N.K.; Ross, A.B.; Scheers, N.; Savolainen, O.I.; Nookaew, I.; Gabrielsson, B.G.; Sandberg, A.-S. Eicosapentaenoic and Docosahexaenoic Acid-Enriched High Fat Diet Delays Skeletal Muscle Degradation in Mice. Nutrients 2016, 8, 543. https://doi.org/10.3390/nu8090543
Soni NK, Ross AB, Scheers N, Savolainen OI, Nookaew I, Gabrielsson BG, Sandberg A-S. Eicosapentaenoic and Docosahexaenoic Acid-Enriched High Fat Diet Delays Skeletal Muscle Degradation in Mice. Nutrients. 2016; 8(9):543. https://doi.org/10.3390/nu8090543
Chicago/Turabian StyleSoni, Nikul K., Alastair B. Ross, Nathalie Scheers, Otto I Savolainen, Intawat Nookaew, Britt G. Gabrielsson, and Ann-Sofie Sandberg. 2016. "Eicosapentaenoic and Docosahexaenoic Acid-Enriched High Fat Diet Delays Skeletal Muscle Degradation in Mice" Nutrients 8, no. 9: 543. https://doi.org/10.3390/nu8090543
APA StyleSoni, N. K., Ross, A. B., Scheers, N., Savolainen, O. I., Nookaew, I., Gabrielsson, B. G., & Sandberg, A.-S. (2016). Eicosapentaenoic and Docosahexaenoic Acid-Enriched High Fat Diet Delays Skeletal Muscle Degradation in Mice. Nutrients, 8(9), 543. https://doi.org/10.3390/nu8090543





