Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design and Procedures
2.3. Analytical Measures
2.3.1. Complete Blood Count
2.3.2. Plasma Cytokines
2.3.3. Granulocyte and Monocyte Phagocytosis, Oxidative Burst Activity
2.3.4. Plasma Antioxidant Capacity
2.3.5. Plasma Amino Acid Analysis
2.3.6. Total Nitrate
2.4. Statistical Analysis
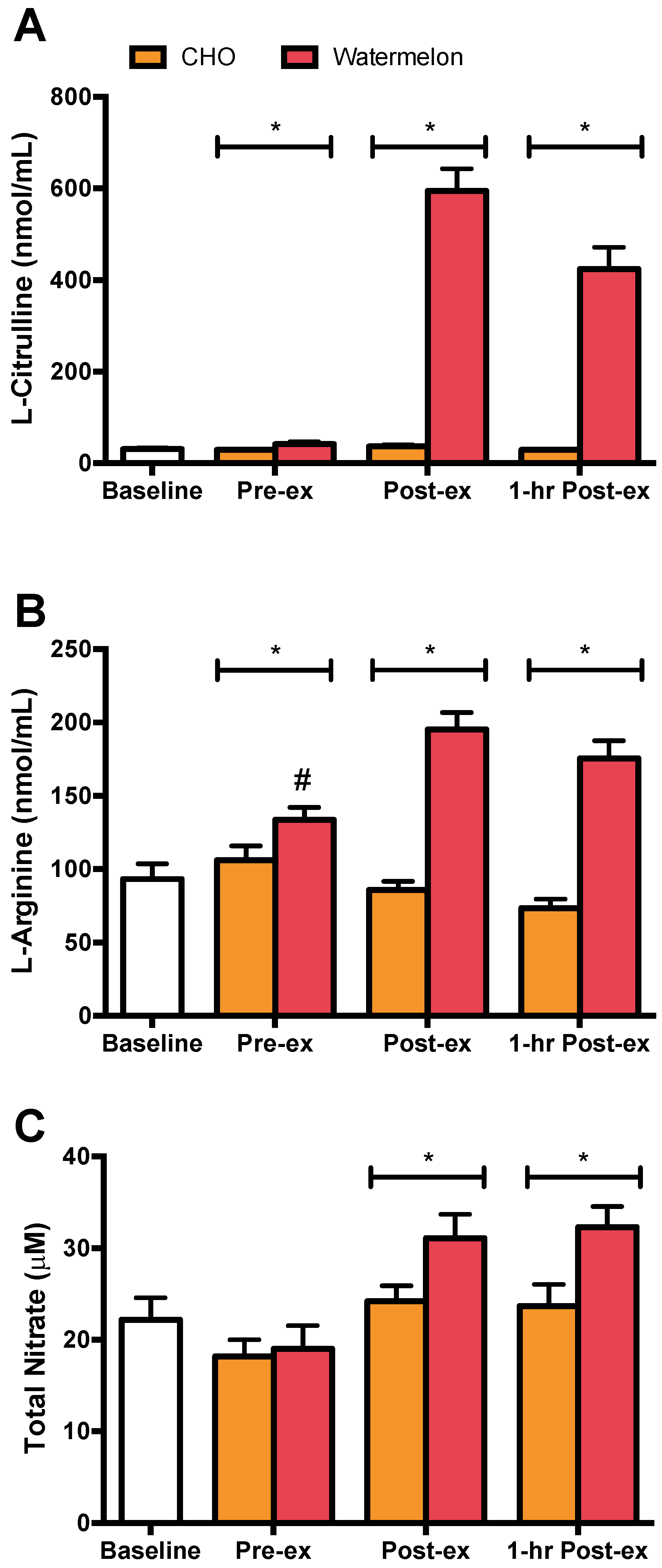
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Levine, S.A.; Gordon, B.; Derick, C.L. Some changes in the chemical constituents of the blood following a marathon race: With special reference to the development of hypoglycemia. J. Am. Med. Assoc. 1924, 82, 1778–1779. [Google Scholar] [CrossRef]
- Gordon, B.; Kohn, L.A.; Levine, S.A.; Matton, M.; Scriver, W.M.; Whiting, W.B. Sugar content of the blood in runners following a marathon race: With especial reference to the prevention of hypoglycemia: Further observations. J. Am. Med. Assoc. 1925, 85, 508–509. [Google Scholar] [CrossRef]
- Coyle, E.F.; Hagberg, J.M.; Hurley, B.F.; Martin, W.H.; Ehsani, A.A.; Holloszy, J.O. Carbohydrate feeding during prolonged strenuous exercise can delay fatigue. J. Appl. Physiol. 1983, 55, 230–235. [Google Scholar] [PubMed]
- Carter, J.; Jeukendrup, A.E.; Mundel, T.; Jones, D.A. Carbohydrate supplementation improves moderate and high-intensity exercise in the heat. Pflug. Arch. 2003, 446, 211–219. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, M.V.; Madsen, K.; Fukui, R.; Santos, A.; da Silva, M.E. Carbohydrate supplementation delays DNA damage in elite runners during intensive microcycle training. Eur. J. Appl. Physiol. 2012, 112, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Scharhag, J.; Meyer, T.; Auracher, M.; Gabriel, H.H.; Kindermann, W. Effects of graded carbohydrate supplementation on the immune response in cycling. Med. Sci. Sports Exerc. 2006, 38, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Jentjens, R.L.; Moseley, L.; Waring, R.H.; Harding, L.K.; Jeukendrup, A.E. Oxidation of combined ingestion of glucose and fructose during exercise. J. Appl. Physiol. 2004, 96, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Summers, R.W.; Schedl, H.P.; Flanagan, S.W.; Chang, R.; Gisolfi, C.V. Effects of carbohydrate type and concentration and solution osmolality on water absorption. Med. Sci. Sports Exerc. 1995, 27, 1607–1615. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, W.J.; Stannard, S.R.; Clarke, J.A.; Rowlands, D.S. Fructose-maltodextrin ratio governs exogenous and other cho oxidation and performance. Med. Sci. Sports Exerc. 2013, 45, 1814–1824. [Google Scholar] [CrossRef] [PubMed]
- Too, B.W.; Cicai, S.; Hockett, K.R.; Applegate, E.; Davis, B.A.; Casazza, G.A. Natural versus commercial carbohydrate supplementation and endurance running performance. J. Int. Soc. Sports Nutr. 2012, 9, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, S.D.; Bazzarre, T.L.; Snider, I.P.; Goldfarb, A.H. Differences in the effects of carbohydrate food form on endurance performance to exhaustion. Int. J. Sport Nutr. 1993, 3, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Gillitt, N.D.; Henson, D.A.; Sha, W.; Shanely, R.A.; Knab, A.M.; Cialdella-Kam, L.; Jin, F. Bananas as an energy source during exercise: A metabolomics approach. PLoS ONE 2012, 7, e37479. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Gillitt, N.D.; Sha, W.; Meaney, M.P.; John, C.; Pappan, K.L.; Kinchen, J.M. Metabolomics-based analysis of banana and pear ingestion on exercise performance and recovery. J. Proteome Res. 2015, 14, 5367–5377. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture. USDA National Nutrient Database for Standard Reference, Release 27. Available online: http://www.ars.usda.gov/ba/bhnrc/ndl (accessed on 10 July 2014).
- Rimando, A.M.; Perkins-Veazie, P.M. Determination of citrulline in watermelon rind. J. Chromatogr. A 2005, 1078, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Ghavipour, M.; Saedisomeolia, A.; Djalali, M.; Sotoudeh, G.; Eshraghyan, M.R.; Moghadam, A.M.; Wood, L.G. Tomato juice consumption reduces systemic inflammation in overweight and obese females. Br. J. Nutr. 2013, 109, 2031–2035. [Google Scholar] [CrossRef] [PubMed]
- Gann, P.H.; Ma, J.; Giovannucci, E.; Willett, W.; Sacks, F.M.; Hennekens, C.H.; Stampfer, M.J. Lower prostate cancer risk in men with elevated plasma lycopene levels: Results of a prospective analysis. Cancer Res. 1999, 59, 1225–1230. [Google Scholar] [PubMed]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Aspects Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Moran, N.E.; Cichon, M.J.; Riedl, K.M.; Grainger, E.M.; Schwartz, S.J.; Novotny, J.A.; Erdman, J.W., Jr.; Clinton, S.K. Compartmental and noncompartmental modeling of (1)(3)c-lycopene absorption, isomerization, and distribution kinetics in healthy adults. Am. J. Clin. Nutr. 2015, 102, 1436–1449. [Google Scholar] [CrossRef] [PubMed]
- Tsitsimpikou, C.; Kioukia-Fougia, N.; Tsarouhas, K.; Stamatopoulos, P.; Rentoukas, E.; Koudounakos, A.; Papalexis, P.; Liesivuori, J.; Jamurtas, A. Administration of tomato juice ameliorates lactate dehydrogenase and creatinine kinase responses to anaerobic training. Food Chem. Toxicol. 2013, 61, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Harms-Ringdahl, M.; Jenssen, D.; Haghdoost, S. Tomato juice intake suppressed serum concentration of 8-oxodg after extensive physical activity. Nutr. J. 2012, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Cordova, A.; Ferrer, M.D.; Tauler, P.; Perez, G.; Tur, J.A.; Pons, A. Effects of l-citrulline oral supplementation on polymorphonuclear neutrophils oxidative burst and nitric oxide production after exercise. Free Radic. Res. 2009, 43, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Bradley, S.J.; Kingwell, B.A.; McConell, G.K. Nitric oxide synthase inhibition reduces leg glucose uptake but not blood flow during dynamic exercise in humans. Diabetes 1999, 48, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
- Mandel, H.; Levy, N.; Izkovitch, S.; Korman, S.H. Elevated plasma citrulline and arginine due to consumption of citrullus vulgaris (watermelon). J. Inherit. Metab. Dis. 2005, 28, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.K.; Wu, G.; Perkins-Veazie, P.; Spears, K.; Claypool, P.L.; Baker, R.A.; Clevidence, B.A. Watermelon consumption increases plasma arginine concentrations in adults. Nutrition 2007, 23, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Root, M.M.; McGinn, M.C.; Nieman, D.C.; Henson, D.A.; Heinz, S.A.; Shanely, R.A.; Knab, A.M.; Jin, F. Combined fruit and vegetable intake is correlated with improved inflammatory and oxidant status from a cross-sectional study in a community setting. Nutrients 2012, 4, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Akashi, K.; Miyake, C.; Yokota, A. Citrulline, a novel compatible solute in drought-tolerant wild watermelon leaves, is an efficient hydroxyl radical scavenger. FEBS Lett. 2001, 508, 438–442. [Google Scholar] [CrossRef]
- Müller, L.; Fröhlich, K.; Böhm, V. Comparative antioxidant activities of carotenoids measured by ferric reducing antioxidant power (FRAP), abts bleaching assay (αTEAC), DPPH assay and peroxyl radical scavenging assay. Food Chem. 2011, 129, 139–148. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The frap assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.J.; Vinyard, B.T.; Wiley, E.R.; Brown, E.D.; Collins, J.K.; Perkins-Veazie, P.; Baker, R.A.; Clevidence, B.A. Consumption of watermelon juice increases plasma concentrations of lycopene and beta-carotene in humans. J. Nutr. 2003, 133, 1043–1050. [Google Scholar] [PubMed]
- Tarazona-Diaz, M.P.; Alacid, F.; Carrasco, M.; Martinez, I.; Aguayo, E. Watermelon juice: Potential functional drink for sore muscle relief in athletes. J. Agric. Food Chem. 2013, 61, 7522–7528. [Google Scholar] [CrossRef] [PubMed]
- Cutrufello, P.T.; Gadomski, S.J.; Zavorsky, G.S. The effect of l-citrulline and watermelon juice supplementation on anaerobic and aerobic exercise performance. J. Sports Sci. 2015, 33, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture. USDA National Nutrient Database for Standard Reference, Release 27. 2015. Available online: http://www.ars.usda.gov/main/site_main.htm?modecode=80-40-05-25 (accessed on 17 July 2015). [Google Scholar]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [PubMed]
- Knab, A.M.; Nieman, D.C.; Gillitt, N.D.; Shanely, R.A.; Cialdella-Kam, L.; Henson, D.; Sha, W.; Meaney, M.P. Effects of a freeze-dried juice blend powder on exercise-induced inflammation, oxidative stress, and immune function in cyclists. Appl. Physiol. Nutr. Metab. 2014, 39, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Meaney, M.P.; Nieman, D.C.; Henson, D.A.; Jiang, Q.; Wang, F.Z. Measuring granulocyte and monocyte phagocytosis and oxidative burst activity in human blood. J. Vis. Exp. 2016, in press. [Google Scholar]
- Shanely, R.A.; Knab, A.M.; Nieman, D.C.; Jin, F.; McAnulty, S.R.; Landram, M.J. Quercetin supplementation does not alter antioxidant status in humans. Free Radic. Res. 2010, 44, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Meininger, C.J. Analysis of citrulline, arginine, and methylarginines using high-performance liquid chromatography. Methods Enzymol. 2008, 440, 177–189. [Google Scholar] [PubMed]
- Nieman, D.C.; Nehlsen-Cannarella, S.L.; Fagoaga, O.R.; Henson, D.A.; Utter, A.; Davis, J.M.; Williams, F.; Butterworth, D.E. Influence of mode and carbohydrate on the cytokine response to heavy exertion. Med. Sci. Sports Exerc. 1998, 30, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Perkins-Veazie, P.; Collins, J.K.; Davis, A.R.; Roberts, W. Carotenoid content of 50 watermelon cultivars. J. Agric. Food Chem. 2006, 54, 2593–2597. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, N.R.; Di Marco, N.M.; Langley, S. American College of Sports Medicine position stand: Nutrition and athletic performance. Med. Sci. Sports Exerc. 2009, 41, 709–731. [Google Scholar] [PubMed]
- Fujisawa, T.; Mulligan, K.; Wada, L.; Schumacher, L.; Riby, J.; Kretchmer, N. The effect of exercise on fructose absorption. Am. J. Clin. Nutr. 1993, 58, 75–79. [Google Scholar] [PubMed]
- Jeukendrup, A.E.; Jentjens, R. Oxidation of carbohydrate feedings during prolonged exercise: Current thoughts, guidelines and directions for future research. Sports Med. 2000, 29, 407–424. [Google Scholar] [CrossRef] [PubMed]
- Sawka, M.N.; Burke, L.M.; Eichner, E.R.; Maughan, R.J.; Montain, S.J.; Stachenfeld, N.S. American College of Sports Medicine position stand. Exercise and fluid replacement. Med. Sci. Sports Exerc. 2007, 39, 377–390. [Google Scholar] [PubMed]
- Cao, G.; Prior, R.L. Comparison of different analytical methods for assessing total antioxidant capacity of human serum. Clin. Chem. 1998, 44, 1309–1315. [Google Scholar] [PubMed]
- Duthie, G.; Robertson, J.; Maughan, R.; Morrice, P. Blood antioxidant status and erythrocyte lipid peroxidation following distance running. Arch. Biochem. Biophys. 1990, 282, 78–83. [Google Scholar] [CrossRef]
- Perheentupa, J.; Raivio, K. Fructose-induced hyperuricaemia. Lancet 1967, 2, 528–531. [Google Scholar] [CrossRef]
- McAnulty, S.R.; Hosick, P.A.; McAnulty, L.S.; Quindry, J.C.; Still, L.; Hudson, M.B.; Dibarnardi, A.N.; Milne, G.L.; Morrow, J.D.; Austin, M.D. Effect of pharmacological lowering of plasma urate on exercise-induced oxidative stress. Appl. Physiol. Nutr. Metab. 2007, 32, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- Quindry, J.C.; Kavazis, A.N.; Powers, S.K. Exercise-induced oxidative stress: Are supplemental antioxidants warranted? In The Encyclopaedia of Sports Medicine; John Wiley & Sons Ltd.: Chichester, UK, 2013; pp. 263–276. [Google Scholar]
- Kruger, K.; Pilat, C.; Schild, M.; Lindner, N.; Frech, T.; Muders, K.; Mooren, F.C. Progenitor cell mobilization after exercise is related to systemic levels of G-CSF and muscle damage. Scand. J. Med. Sci. Sports 2015, 25, e283–e291. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C.; Volker, K.; Klocke, R.; Nikol, S.; Waltenberger, J.; Kruger, K. Exercise delays neutrophil apoptosis by a G-CSF-dependent mechanism. J. Appl. Physiol. 2012, 113, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Yamada, M.; Suzuki, K.; Kudo, S.; Totsuka, M.; Nakaji, S.; Sugawara, K. Raised plasma G-CSF and il-6 after exercise may play a role in neutrophil mobilization into the circulation. J. Appl. Physiol. 2002, 92, 1789–1794. [Google Scholar] [CrossRef] [PubMed]
- Moinard, C.; Nicolis, I.; Neveux, N.; Darquy, S.; Benazeth, S.; Cynober, L. Dose-ranging effects of citrulline administration on plasma amino acids and hormonal patterns in healthy subjects: The citrudose pharmacokinetic study. Br. J. Nutr. 2008, 99, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Hickner, R.C.; Tanner, C.J.; Evans, C.A.; Clark, P.D.; Haddock, A.; Fortune, C.; Geddis, H.; Waugh, W.; McCammon, M. l-citrulline reduces time to exhaustion and insulin response to a graded exercise test. Med. Sci. Sports Exerc. 2006, 38, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Morita, M.; Kobayashi, Y.; Kamimura, A. Oral l-citrulline supplementation enhances cycling time trial performance in healthy trained men: Double-blind randomized placebo-controlled 2-way crossover study. J. Int. Soc. Sports Nutr. 2016, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Blackwell, J.R.; Lord, T.; Vanhatalo, A.; Winyard, P.G.; Jones, A.M. l-citrulline supplementation improves O2 uptake kinetics and high-intensity exercise performance in humans. J. Appl. Physiol. 2015, 119, 385–395. [Google Scholar] [CrossRef] [PubMed]

| Variable | Mean ± SEM |
|---|---|
| Age (year) | 48.5 ± 2.3 |
| Body mass (kg) | 81.04 ± 2.2 |
| % Body fat | 19.6 ± 1.5 |
| BMI (kg/m2) | 25.1 ± 0.7 |
| Years cycling | 10.95 ± 2.3 |
| Wattmax | 314 ± 9.8 |
| Peak oxygen consumption (VO2peak, mL·kg−1·min−1) | 51.5 ± 1.9 |
| Variable | Baseline | Pre-Exercise | Post-Exercise | 1-h Post-Exercise | Time; Interaction p Values | |
|---|---|---|---|---|---|---|
| Inflammatory markers | ||||||
| WBC (109/L) | 6.2 ± 0.34 | |||||
| CHO | 5.74 ± 0.31 | 12.2 ± 0.96 | 10.9 ± 0.83 | <0.001; 0.125 | ||
| WM | 5.58 ± 0.39 | 14.7 ± 1.01 | 12.4 ± 0.85 | |||
| TNF-α (pg/mL) | 10.5 ± 1.03 | |||||
| CHO | 10.2 ± 0.92 | 12.8 ± 1.11 | 11.9 ± 1.14 | 0.005; 0.936 | ||
| WM | 10.2 ± 0.99 | 12.3 ± 1.29 | 12.1 ± 1.19 | |||
| IL-6 (pg/mL) | 1.02 ± 0.28 | |||||
| CHO | 0.80 ± 0.13 | 10.2 ± 1.83 | 7.92 ± 1.58 | <0.001; 0.921 | ||
| WM | 0.71 ± 0.14 | 9.98 ± 1.63 | 7.68 ± 1.56 | |||
| IL-8 (pg/mL) | 3.21 ± 0.33 | |||||
| CHO | 3.26 ± 0.37 | 10.9 ± 1.01 | 12.2 ± 1.55 | <0.001; 0.506 | ||
| WM | 3.59 ± 0.33 | 12.1 ± 1.52 | 11.4 ± 1.55 | |||
| IL-10 (pg/mL) | 2.31 ± 0.44 | |||||
| CHO | 2.46 ± 0.48 | 10.5 ± 3.83 | 8.44 ± 2.82 | 0.002; 0.292 | ||
| WM | 2.93 ± 0.82 | 15.5 ± 5.68 | 16.0 ± 6.64 | |||
| MCP-1 (pg/mL) | 194 ± 7.80 | |||||
| CHO | 188 ± 8.99 | 344 ± 23.8 | 333 ± 27.3 | <0.001; 0.206 | ||
| WM | 188 ± 9.44 | 375 ± 21.1 | 338 ± 16.9 | |||
| G-CSF (pg/mL) | 9.47 ± 0.56 | |||||
| CHO | 9.94 ± 0.97 | 16.2 ± 1.55 | 16.8 ± 1.53 | <0.001; 0.041 | ||
| WM | 10.7 ± 0.96 | 18.7 ± 2.12 | 19.9 ± 1.94 | |||
| GR-PHAG (MFI) | 49.7 ± 4.20 | |||||
| CHO | 32.7 ± 2.81 | 63.2 ± 8.83 | 75.3 ± 11.9 | 0.001; 0.635 | ||
| WM | 40.8 ± 6.75 | 72.2 ± 12.0 | 83.6 ± 16.3 | |||
| MO-PHAG (MFI) | 26.5 ± 1.64 | |||||
| CHO | 20.1 ± 1.50 | 36.0 ± 3.46 | 44.4 ± 5.18 | <0.001; 0.612 | ||
| WM | 23.9 ± 3.89 | 39.2 ± 4.87 | 44.4 ± 6.88 | |||
| GR-OBA (MFI) | 23.3 ± 1.11 | |||||
| CHO | 16.3 ± 1.35 | 24.6 ± 2.08 | 26.3 ± 2.78 | <0.001; 0.612 | ||
| WM | 18.5 ± 2.54 | 26.2 ± 3.28 | 23.8 ± 3.47 | |||
| MO-OBA (MFI) | 11.8 ± 0.44 | |||||
| CHO | 9.70 ± 0.59 | 13.5 ± 0.73 | 14.7 ± 0.98 | <0.001; 0.173 | ||
| WM | 10.6 ± 1.15 | 13.1 ± 1.09 | 13.0 ± 1.09 | |||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shanely, R.A.; Nieman, D.C.; Perkins-Veazie, P.; Henson, D.A.; Meaney, M.P.; Knab, A.M.; Cialdell-Kam, L. Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity. Nutrients 2016, 8, 518. https://doi.org/10.3390/nu8080518
Shanely RA, Nieman DC, Perkins-Veazie P, Henson DA, Meaney MP, Knab AM, Cialdell-Kam L. Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity. Nutrients. 2016; 8(8):518. https://doi.org/10.3390/nu8080518
Chicago/Turabian StyleShanely, R. Andrew, David C. Nieman, Penelope Perkins-Veazie, Dru A. Henson, Mary P. Meaney, Amy M. Knab, and Lynn Cialdell-Kam. 2016. "Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity" Nutrients 8, no. 8: 518. https://doi.org/10.3390/nu8080518
APA StyleShanely, R. A., Nieman, D. C., Perkins-Veazie, P., Henson, D. A., Meaney, M. P., Knab, A. M., & Cialdell-Kam, L. (2016). Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity. Nutrients, 8(8), 518. https://doi.org/10.3390/nu8080518







