Impact of Cocoa Consumption on Inflammation Processes—A Critical Review of Randomized Controlled Trials
Abstract
:1. Introduction
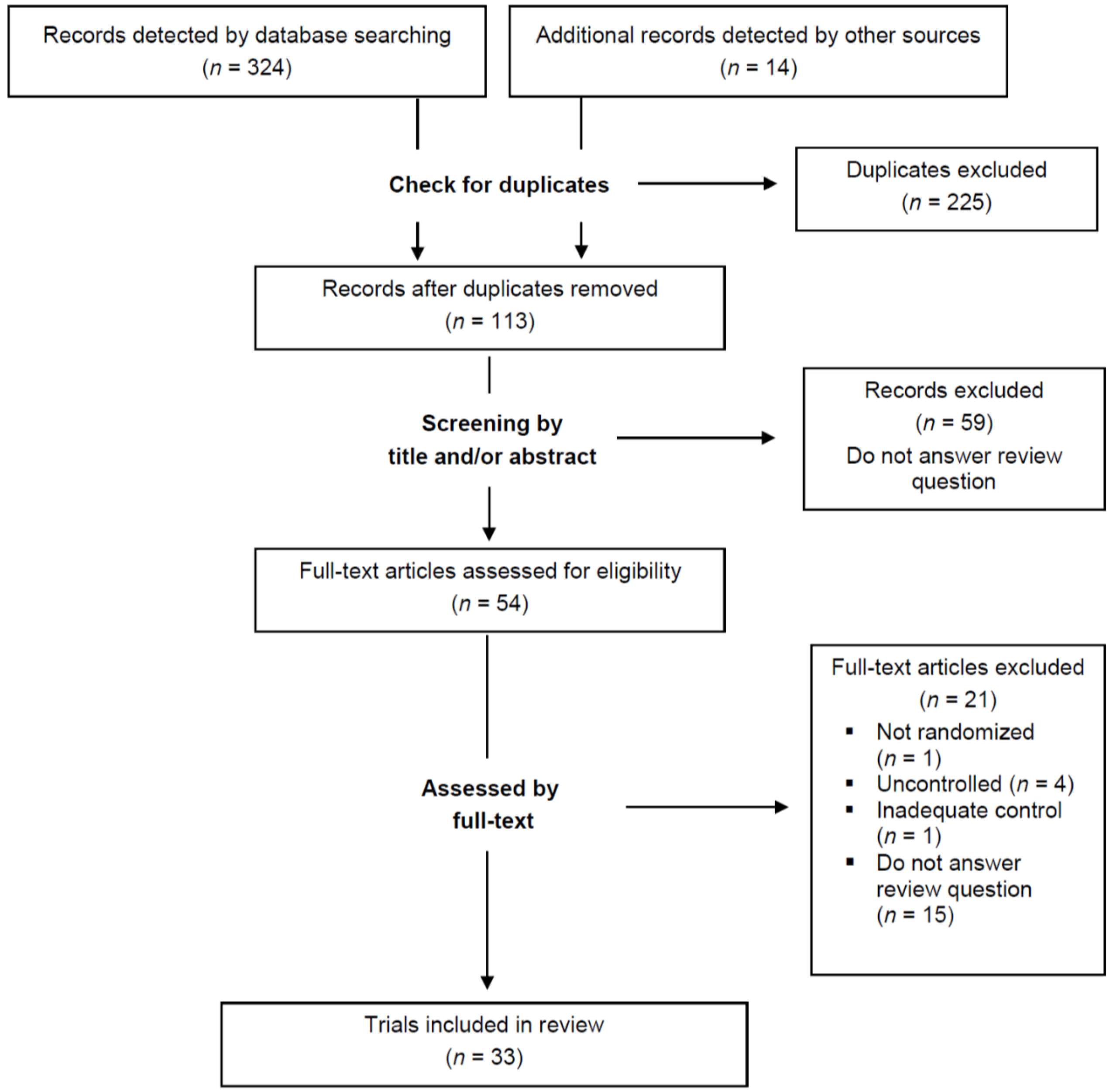
2. Materials and Methods
2.1. Literature Search
2.2. Selection of Trials
- Type of studies: randomized controlled trials
- Type of interventions: supplementing foods rich in cocoa without restrictions to the kind of food used (e.g., dark chocolate, cocoa-rich drinks, cocoa-enriched foods) or the frequency of supplementation (e.g., single or repeated, daily consumption)
- Type of controls: supplementing comparable foods lacking cocoa (e.g., white chocolate, milk, placebo drink, foods not enriched with cocoa), being low in cocoa or not providing cocoa-rich foods
- Type of inflammatory biomarkers: humoral parameters determined in serum or plasma (e.g., CRP, soluble adhesion molecules, eicosanoids, cytokines) and cellular parameters determined in leukocytes obtained from whole blood (e.g., cellular adhesion molecules, immunological functions)
2.3. Criteria for Judging the Quality of the Selected Trials
3. Results
3.1. Bolus Studies
3.2. Studies on Regular Cocoa Consumption
3.2.1. Healthy Subjects
3.2.2. Pre-/Hypertensive Subjects
3.2.3. Patients with Type 2 Diabetes or Impaired Glucose Tolerance
3.2.4. Patients with Coronary Heart Disease
3.3. Quality of Selected Studies
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Castellon, X.; Bogdanova, V. Chronic inflammatory diseases and endothelial dysfunction. Aging Dis. 2016, 7, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Hameed, I.; Masoodi, S.R.; Mir, S.A.; Nabi, M.; Ghazanfar, K.; Ganai, B.A. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J. Diabetes 2015, 6, 598–612. [Google Scholar] [PubMed]
- Colak, E.; Majkic-Singh, N.; Zoric, L.; Radosavljevic, A.; Kosanovic-Jakovic, N. The role of CRP and inflammation in the pathogenesis of age-related macular degeneration. Biochem. Med. 2012, 22, 39–48. [Google Scholar] [CrossRef]
- Spencer, J.P.; Vafeiadou, K.; Williams, R.J.; Vauzour, D. Neuroinflammation: Modulation by flavonoids and mechanisms of action. Mol. Aspects Med. 2012, 33, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Poredos, P.; Kaja Jezovnik, M. Markers of preclinical atherosclerosis and their clinical relevance. VASA. Z. Gefasskrankh. 2015, 44, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Tousoulis, D.; Papageorgiou, N.; Androulakis, E.; Siasos, G.; Latsios, G.; Tentolouris, K.; Stefanadis, C. Diabetes mellitus-associated vascular impairment: Novel circulating biomarkers and therapeutic approaches. J. Am. Coll. Cardiol. 2013, 62, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Kenny, T.P.; Keen, C.L.; Schmitz, H.H.; Gershwin, M.E. Immune effects of cocoa procyanidin oligomers on peripheral blood mononuclear cells. Exp. Biol. Med. 2007, 232, 293–300. [Google Scholar]
- Mao, T.K.; van de Water, J.; Keen, C.L.; Schmitz, H.H.; Gershwin, M.E. Modulation of TNF-α secretion in peripheral blood mononuclear cells by cocoa flavanols and procyanidins. Dev. Immunol. 2002, 9, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Ramiro, E.; Franch, A.; Castellote, C.; Perez-Cano, F.; Permanyer, J.; Izquierdo-Pulido, M.; Castell, M. Flavonoids from Theobroma cacao down-regulate inflammatory mediators. J. Agric. Food Chem. 2005, 53, 8506–8511. [Google Scholar] [CrossRef] [PubMed]
- Kenny, T.P.; Shu, S.A.; Moritoki, Y.; Keen, C.L.; Gershwin, M.E. Cocoa flavanols and procyanidins can modulate the lipopolysaccharide activation of polymorphonuclear cells in vitro. J. Med. Food 2009, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ramiro, E.; Franch, A.; Castellote, C.; Andres-Lacueva, C.; Izquierdo-Pulido, M.; Castell, M. Effect of Theobroma cacao flavonoids on immune activation of a lymphoid cell line. Br. J. Nutr. 2005, 93, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, G.G.; Carrasquedo, F.; Delfino, J.M.; Keen, C.L.; Fraga, C.G.; Oteiza, P.I. Epicatechin, catechin, and dimeric procyanidins inhibit PMA-induced NF-κB activation at multiple steps in Jurkat T cells. FASEB J. 2004, 18, 167–169. [Google Scholar] [PubMed]
- Mackenzie, G.G.; Oteiza, P.I. Modulation of transcription factor NF-κB in Hodgkin's lymphoma cell lines: Effect of (−)-epicatechin. Free Radic. Res. 2006, 40, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Selmi, C.; Mao, T.K.; Keen, C.L.; Schmitz, H.H.; Eric Gershwin, M. The anti-inflammatory properties of cocoa flavanols. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. S2), 163–171. [Google Scholar] [CrossRef]
- Ramiro-Puig, E.; Castell, M. Cocoa: Antioxidant and immunomodulator. Br. J. Nutr. 2009, 101, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Mehrinfar, R.; Frishman, W.H. Flavanol-rich cocoa: A cardioprotective nutraceutical. Cardiol. Rev. 2008, 16, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Schewe, T.; Kuhn, H.; Sies, H. Flavonoids of cocoa inhibit recombinant human 5-lipoxygenase. J. Nutr. 2002, 132, 1825–1829. [Google Scholar] [PubMed]
- Sies, H.; Schewe, T.; Heiss, C.; Kelm, M. Cocoa polyphenols and inflammatory mediators. Am. J. Clin. Nutr. 2005, 81, 304S–312S. [Google Scholar] [PubMed]
- Lee, K.W.; Kang, N.J.; Oak, M.H.; Hwang, M.K.; Kim, J.H.; Schini-Kerth, V.B.; Lee, H.J. Cocoa procyanidins inhibit expression and activation of MMP-2 in vascular smooth muscle cells by direct inhibition of MEK and MT1-MMP activities. Cardiovasc. Res. 2008, 79, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Murga, L.; Tarin, J.J.; Garcia-Perez, M.A.; Cano, A. The impact of chocolate on cardiovascular health. Maturitas 2011, 69, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Ding, E.L.; Hutfless, S.M.; Ding, X.; Girotra, S. Chocolate and prevention of cardiovascular disease: A systematic review. Nutr. Metab. 2006, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzoglou, A.; Mulligan, A.A.; Luben, R.N.; Lentjes, M.A.; Heiss, C.; Kelm, M.; Merx, M.W.; Spencer, J.P.; Schroeter, H.; Kuhnle, G.G. Assessment of the dietary intake of total flavan-3-ols, monomeric flavan-3-ols, proanthocyanidins and theaflavins in the European Union. Br. J. Nutr. 2014, 111, 1463–1473. [Google Scholar] [CrossRef] [PubMed]
- Cifuentes-Gomez, T.; Rodriguez-Mateos, A.; Gonzalez-Salvador, I.; Alanon, M.E.; Spencer, J.P. Factors affecting the absorption, metabolism, and excretion of cocoa flavanols in humans. J. Agric. Food Chem. 2015, 63, 7615–7623. [Google Scholar] [CrossRef] [PubMed]
- Desch, S.; Schmidt, J.; Kobler, D.; Sonnabend, M.; Eitel, I.; Sareban, M.; Rahimi, K.; Schuler, G.; Thiele, H. Effect of cocoa products on blood pressure: Systematic review and meta-analysis. Am. J. Hypertens. 2010, 23, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Kay, C.; Abdelhamid, A.; Kroon, P.A.; Cohn, J.S.; Rimm, E.B.; Cassidy, A. Effects of chocolate, cocoa, and flavan-3-ols on cardiovascular health: A systematic review and meta-analysis of randomized trials. Am. J. Clin. Nutr. 2012, 95, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Sullivan, T.; Fakler, P.; Frank, O.R.; Stocks, N.P. Does chocolate reduce blood pressure? A meta-analysis. BMC Med. 2010, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Shrime, M.G.; Bauer, S.R.; McDonald, A.C.; Chowdhury, N.H.; Coltart, C.E.; Ding, E.L. Flavonoid-rich cocoa consumption affects multiple cardiovascular risk factors in a meta-analysis of short-term studies. J. Nutr. 2011, 141, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; Le Cornu, K.A.; Ryder, J.J.; Hall, W.L.; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [PubMed]
- Tokede, O.A.; Gaziano, J.M.; Djousse, L. Effects of cocoa products/dark chocolate on serum lipids: A meta-analysis. Eur. J. Clin. Nutr. 2011, 65, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Liu, X.; Bai, Y.Y.; Li, S.H.; Sun, K.; He, C.; Hui, R. Short-term effect of cocoa product consumption on lipid profile: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2010, 92, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Corti, R.; Flammer, A.J.; Hollenberg, N.K.; Luscher, T.F. Cocoa and cardiovascular health. Circulation 2009, 119, 1433–1441. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Croce, G.; Tiberti, S.; Aggio, A.; Ferri, C. Flavonoids, vascular function and cardiovascular protection. Curr. Pharm. Des. 2009, 15, 1072–1084. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; di Giosia, P.; Barnabei, R.; Allegaert, L.; Bernaert, H.; Ferri, C. Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals. J. Hypertens. 2015, 33, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Kirch, N.; Ellinger, S. Cocoa flavanols and cardioprotective effects. Which flavanols may contribute to vascular health? Ernahrungs Umsch. Int. 2014, 61, 144–151. [Google Scholar]
- Ellinger, S.; Reusch, A.; Stehle, P.; Helfrich, H.P. Epicatechin ingested via cocoa products reduces blood pressure in humans: A nonlinear regression model with a bayesian approach. Am. J. Clin. Nutr. 2012, 95, 1365–1377. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (−)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Chun, O.K.; Chung, S.J.; Song, W.O. Estimated dietary flavonoid intake and major food sources of US adults. J. Nutr. 2007, 137, 1244–1252. [Google Scholar] [PubMed]
- Knaze, V.; Zamora-Ros, R.; Lujan-Barroso, L.; Romieu, I.; Scalbert, A.; Slimani, N.; Riboli, E.; van Rossum, C.T.; Bueno-de-Mesquita, H.B.; Trichopoulou, A.; et al. Intake estimation of total and individual flavan-3-ols, proanthocyanidins and theaflavins, their food sources and determinants in the European Prospective investigation into cancer and nutrition (EPIC) study. Br. J. Nutr. 2012, 108, 1095–1108. [Google Scholar] [CrossRef] [PubMed]
- di Giuseppe, R.; Di Castelnuovo, A.; Centritto, F.; Zito, F.; De Curtis, A.; Costanzo, S.; Vohnout, B.; Sieri, S.; Krogh, V.; Donati, M.B.; et al. Regular consumption of dark chocolate is associated with low serum concentrations of C-reactive protein in a healthy Italian population. J. Nutr. 2008, 138, 1939–1945. [Google Scholar] [PubMed]
- Ferri, N.; Paoletti, R.; Corsini, A. Biomarkers for atherosclerosis: Pathophysiological role and pharmacological modulation. Curr. Opin. Lipidol. 2006, 17, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Balshem, H.; Helfand, M.; Schunemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S.; et al. Grade guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Montori, V.; Akl, E.A.; Djulbegovic, B.; Falck-Ytter, Y.; et al. Grade guidelines: 4. Rating the quality of evidence—Study limitations (risk of bias). J. Clin. Epidemiol. 2011, 64, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Schramm, D.D.; Wang, J.F.; Holt, R.R.; Ensunsa, J.L.; Gonsalves, J.L.; Lazarus, S.A.; Schmitz, H.H.; German, J.B.; Keen, C.L. Chocolate procyanidins decrease the leukotriene-prostacyclin ratio in humans and human aortic endothelial cells. Am. J. Clin. Nutr. 2001, 73, 36–40. [Google Scholar] [PubMed]
- Heptinstall, S.; May, J.; Fox, S.; Kwik-Uribe, C.; Zhao, L. Cocoa flavanols and platelet and leukocyte function: Recent in vitro and ex vivo studies in healthy adults. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. S2), 197–205. [Google Scholar] [CrossRef]
- Flammer, A.J.; Hermann, F.; Sudano, I.; Spieker, L.; Hermann, M.; Cooper, K.A.; Serafini, M.; Luscher, T.F.; Ruschitzka, F.; Noll, G.; et al. Dark chocolate improves coronary vasomotion and reduces platelet reactivity. Circulation 2007, 116, 2376–2382. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Agell, M.; Urpi-Sarda, M.; Sacanella, E.; Camino-Lopez, S.; Chiva-Blanch, G.; Llorente-Cortes, V.; Tobias, E.; Roura, E.; Andres-Lacueva, C.; Lamuela-Raventos, R.M.; et al. Cocoa consumption reduces nf-kb activation in peripheral blood mononuclear cells in humans. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Davison, G.; Callister, R.; Williamson, G.; Cooper, K.A.; Gleeson, M. The effect of acute pre-exercise dark chocolate consumption on plasma antioxidant status, oxidative stress and immunoendocrine responses to prolonged exercise. Eur. J. Nutr. 2012, 51, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Flammer, A.J.; Sudano, I.; Wolfrum, M.; Thomas, R.; Enseleit, F.; Periat, D.; Kaiser, P.; Hirt, A.; Hermann, M.; Serafini, M.; et al. Cardiovascular effects of flavanol-rich chocolate in patients with heart failure. Eur. Heart J. 2012, 33, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.D.; Madden, L.A.; Smith, K.A.; Kilpatrick, E.S.; Atkin, S.L. High-polyphenol chocolate reduces endothelial dysfunction and oxidative stress during acute transient hyperglycaemia in type 2 diabetes: A pilot randomized controlled trial. Diabet. Med. 2013, 30, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Betts, N.M.; Leyva, M.J.; Fu, D.; Aston, C.E.; Lyons, T.J. Acute cocoa supplementation increases postprandial hdl cholesterol and insulin in obese adults with type 2 diabetes after consumption of a high-fat breakfast. J. Nutr. 2015, 145, 2325–2332. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa reduces blood pressure and insulin resistance and improves endothelium-dependent vasodilation in hypertensives. Hypertension 2005, 46, 398–405. [Google Scholar] [PubMed]
- Kurlandsky, S.B.; Stote, K.S. Cardioprotective effects of chocolate and almond consumption in healthy women. Nutr. Res. 2006, 26, 509–516. [Google Scholar] [CrossRef]
- Crews, W.D., Jr.; Harrison, D.W.; Wright, J.W. A double-blind, placebo-controlled, randomized trial of the effects of dark chocolate and cocoa on variables associated with neuropsychological functioning and cardiovascular health: Clinical findings from a sample of healthy, cognitively intact older adults. Am. J. Clin. Nutr. 2008, 87, 872–880. [Google Scholar] [PubMed]
- Njike, V.Y.; Faridi, Z.; Shuval, K.; Dutta, S.; Kay, C.D.; West, S.G.; Kris-Etherton, P.M.; Katz, D.L. Effects of sugar-sweetened and sugar-free cocoa on endothelial function in overweight adults. Int. J. Cardiol. 2011, 149, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Ibero-Baraibar, I.; Abete, I.; Navas-Carretero, S.; Massis-Zaid, A.; Martinez, J.A.; Zulet, M.A. Oxidised ldl levels decreases after the consumption of ready-to-eat meals supplemented with cocoa extract within a hypocaloric diet. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Sarria, B.; Martinez-Lopez, S.; Sierra-Cinos, J.L.; Garcia-Diz, L.; Mateos, R.; Bravo, L. Regular consumption of a cocoa product improves the cardiometabolic profile in healthy and moderately hypercholesterolaemic adults. Br. J. Nutr. 2014, 111, 122–134. [Google Scholar] [CrossRef] [PubMed]
- West, S.G.; McIntyre, M.D.; Piotrowski, M.J.; Poupin, N.; Miller, D.L.; Preston, A.G.; Wagner, P.; Groves, L.F.; Skulas-Ray, A.C. Effects of dark chocolate and cocoa consumption on endothelial function and arterial stiffness in overweight adults. Br. J. Nutr. 2014, 111, 653–661. [Google Scholar] [CrossRef] [PubMed]
- McFarlin, B.K.; Venable, A.S.; Henning, A.L.; Prado, E.A.; Best Sampson, J.N.; Vingren, J.L.; Hill, D.W. Natural cocoa consumption: Potential to reduce atherogenic factors? J. Nutr. Biochem. 2015, 26, 626–632. [Google Scholar] [CrossRef]
- Wang-Polagruto, J.F.; Villablanca, A.C.; Polagruto, J.A.; Lee, L.; Holt, R.R.; Schrader, H.R.; Ensunsa, J.L.; Steinberg, F.M.; Schmitz, H.H.; Keen, C.L. Chronic consumption of flavanol-rich cocoa improves endothelial function and decreases vascular cell adhesion molecule in hypercholesterolemic postmenopausal women. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. S2), 177–186. [Google Scholar] [CrossRef]
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr. 2008, 138, 1671–1676. [Google Scholar] [PubMed]
- Muniyappa, R.; Hall, G.; Kolodziej, T.L.; Karne, R.J.; Crandon, S.K.; Quon, M.J. Cocoa consumption for 2 wk enhances insulin-mediated vasodilatation without improving blood pressure or insulin resistance in essential hypertension. Am. J. Clin. Nutr. 2008, 88, 1685–1696. [Google Scholar] [CrossRef] [PubMed]
- Balzer, J.; Rassaf, T.; Heiss, C.; Kleinbongard, P.; Lauer, T.; Merx, M.; Heussen, N.; Gross, H.B.; Keen, C.L.; Schroeter, H.; et al. Sustained benefits in vascular function through flavanol-containing cocoa in medicated diabetic patients a double-masked, randomized, controlled trial. J. Am. Coll. Cardiol. 2008, 51, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Monagas, M.; Khan, N.; Andres-Lacueva, C.; Casas, R.; Urpi-Sarda, M.; Llorach, R.; Lamuela-Raventos, R.M.; Estruch, R. Effect of cocoa powder on the modulation of inflammatory biomarkers in patients at high risk of cardiovascular disease. Am. J. Clin. Nutr. 2009, 90, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.D.; Sathyapalan, T.; Kilpatrick, E.S.; Beckett, S.; Atkin, S.L. High-cocoa polyphenol-rich chocolate improves HDL cholesterol in Type 2 diabetes patients. Diabet. Med. 2010, 27, 1318–1321. [Google Scholar] [CrossRef] [PubMed]
- Stote, K.S.; Clevidence, B.A.; Novotny, J.A.; Henderson, T.; Radecki, S.V.; Baer, D.J. Effect of cocoa and green tea on biomarkers of glucose regulation, oxidative stress, inflammation and hemostasis in obese adults at risk for insulin resistance. Eur. J. Clin. Nutr. 2012, 66, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Parsaeyan, N.; Mozaffari-Khosravi, H.; Absalan, A.; Mozayan, M.R. Beneficial effects of cocoa on lipid peroxidation and inflammatory markers in type 2 diabetic patients and investigation of probable interactions of cocoa active ingredients with prostaglandin synthase-2 (PTGS-2/COX-2) using virtual analysis. J. Diabetes Metab. Disord. 2014, 13, 30. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Khalili, M.; Haghighat, N.; Eghtesadi, S.; Shidfar, F.; Heidari, I.; Ebrahimpour-Koujan, S.; Eghtesadi, M. High-cocoa polyphenol-rich chocolate improves blood pressure in patients with diabetes and hypertension. ARYA Atheroscler. 2015, 11, 21–29. [Google Scholar] [PubMed]
- Farouque, H.M.; Leung, M.; Hope, S.A.; Baldi, M.; Schechter, C.; Cameron, J.D.; Meredith, I.T. Acute and chronic effects of flavanol-rich cocoa on vascular function in subjects with coronary artery disease: A randomized double-blind placebo-controlled study. Clin. Sci. 2006, 111, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Jahn, S.; Taylor, M.; Real, W.M.; Angeli, F.S.; Wong, M.L.; Amabile, N.; Prasad, M.; Rassaf, T.; Ottaviani, J.I.; et al. Improvement of endothelial function with dietary flavanols is associated with mobilization of circulating angiogenic cells in patients with coronary artery disease. J. Am. Coll. Cardiol. 2010, 56, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, L.; Perri, L.; Catasca, E.; Pignatelli, P.; Brancorsini, M.; Nocella, C.; De Falco, E.; Bartimoccia, S.; Frati, G.; Carnevale, R.; et al. Dark chocolate acutely improves walking autonomy in patients with peripheral artery disease. J. Am. Heart Assoc. 2014, 3, e001072. [Google Scholar] [CrossRef] [PubMed]
- Horn, P.; Amabile, N.; Angeli, F.S.; Sansone, R.; Stegemann, B.; Kelm, M.; Springer, M.L.; Yeghiazarians, Y.; Schroeter, H.; Heiss, C. Dietary flavanol intervention lowers the levels of endothelial microparticles in coronary artery disease patients. Br. J. Nutr. 2014, 111, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Brasier, A.R. The nuclear factor-κB -interleukin-6 signalling pathway mediating vascular inflammation. Cardiovasc. Res. 2010, 86, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Lippi, C.; Necozione, S.; Desideri, G.; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr. 2005, 81, 611–614. [Google Scholar] [PubMed]
- Richelle, M.; Tavazzi, I.; Enslen, M.; Offord, E.A. Plasma kinetics in man of epicatechin from black chocolate. Eur. J. Clin. Nutr. 1999, 53, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Urpi-Sarda, M.; Ramiro-Puig, E.; Khan, N.; Ramos-Romero, S.; Llorach, R.; Castell, M.; Gonzalez-Manzano, S.; Santos-Buelga, C.; Andres-Lacueva, C. Distribution of epicatechin metabolites in lymphoid tissues and testes of young rats with a cocoa-enriched diet. Br. J. Nutr. 2010, 103, 1393–1397. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Orekhov, A.N.; Bobryshev, Y.V. Endothelial barrier and its abnormalities in cardiovascular disease. Front. Physiol. 2015, 6, 365. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Xu, A. Interplay between adipose tissue and blood vessels in obesity and vascular dysfunction. Rev. Endocr. Metab. Disord. 2013, 14, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.C.; Porter, K.E. Cellular and molecular mechanisms of endothelial dysfunction in diabetes. Diabetes Vasc. Dis. Res. 2013, 10, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.A.; Campos-Gimenez, E.; Jimenez Alvarez, D.; Nagy, K.; Donovan, J.L.; Williamson, G. Rapid reversed phase ultra-performance liquid chromatography analysis of the major cocoa polyphenols and inter-relationships of their concentrations in chocolate. J. Agric. Food Chem. 2007, 55, 2841–2847. [Google Scholar] [CrossRef] [PubMed]
- Esser, D.; Mars, M.; Oosterink, E.; Stalmach, A.; Muller, M.; Afman, L.A. Dark chocolate consumption improves leukocyte adhesion factors and vascular function in overweight men. FASEB J. 2014, 28, 1464–1473. [Google Scholar] [CrossRef] [PubMed]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O., III; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the centers for disease control and prevention and the american heart association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L. C-reaktives protein. In Labor und Diagnose, 8th ed.; Thomas, L., Ed.; TH-Books Verlagsgesellschaft: Frankfurt, Germany, 2012; Volume 2, pp. 1308–1328. (In German) [Google Scholar]
- Thomas, L. Zytokine und zytokinrezeptoren. In Labor und Diagnose, 8th ed.; Thomas, L., Ed.; TH-Books-Verlagsgesellschaft: Frankfurt, Germany, 2012; Volume 2, pp. 1278–1291. (In German) [Google Scholar]
- Lebranchu, Y.; Valentin, J.F.; Büchler, M.F. Measurement of soluble adhesion molecules in biological fluids. In Adhesion Molecules in Health and Disease; Paul, L.C., Issekutz, T.B., Eds.; Marcel Dekker: New York, NY, USA, 1997; pp. 87–122. [Google Scholar]
- Janeway, C.A.; Travers, P. Immunologie, 2nd ed.; Spektrum Akademischer Verlag: Heidelberg, Germany, 1997. (In German) [Google Scholar]
- Paul, L.C.; Issekutz, T.B. Adhesion Molecules in hEalth and Disease; Marcel Dekker: New York, NY, USA, 1997. [Google Scholar]
- Gratchev, A.; Sobenin, I.; Orekhov, A.; Kzhyshkowska, J. Monocytes as a diagnostic marker of cardiovascular diseases. Immunobiology 2012, 217, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Scheid, L.; Reusch, A.; Stehle, P.; Ellinger, S. Antioxidant effects of cocoa and cocoa products ex vivo and in vivo: Is there evidence from controlled intervention studies? Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Ketelhuth, D.F.; Hansson, G.K. Cellular immunity, low-density lipoprotein and atherosclerosis: Break of tolerance in the artery wall. Thromb. Haemost. 2011, 106, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Lovren, F.; Verma, S. Evolving role of microparticles in the pathophysiology of endothelial dysfunction. Clin. Chem. 2013, 59, 1166–1174. [Google Scholar] [CrossRef] [PubMed]
| Study (Reference) | n | Participants | Intervention | EC (mg) | Study Design | Sample | Parameter | Results | Annotations |
|---|---|---|---|---|---|---|---|---|---|
| Schramm et al., 2001 [43] | 10 | Healthy NS Age: 39 ± 1 BMI: 24.0 ± 0.7 CRP: n.d. | I: Flavanol-rich chocolate (Dove dark chocolate, Mars), 37 g, consumed with 45 g bagel | I: 41 | Double-blind, crossover | Plasma (0 h, 2 h) | Leukotrienes C4, D4, E4 (sum) | I < C (2 h) | Polyphenol-free diet, 1 week washout |
| C: Flavanol-low chocolate (Mars), 37 g, consumed with 45 g bagel | C: 2 | ||||||||
| Heptinstall et al., 2006 [44] | 12 | Healthy subjects Age: n.d. (adults) BMI: n.d. CRP: n.d. | I: Cocoa-rich beverages (CocoaPro, Mars) with different flavanol content: I1: 980 mg I2: 680 mg I3: 380 mg | n.d. | Double-blind, crossover | Monocytes from whole blood | CD62P (P-selectin) | I: ↓ C: o | 48 h before low-flavonoid diet, ≥10 days washout |
| CD11b b | I: ↓ C: ↓ | ||||||||
| C: Cocoa beverage, low in flavanols (80 mg) | n.d. | Neutrophils from whole blood (0 h, 2 h, 4 h, 6 h) | CD62P (P-selectin) | I: ↓ C: o | |||||
| CD11b b | I: ↓ C: ↓ | ||||||||
| Flammer et al., 2007 [45] | 22 | Patients with heart transplantation a Age: 54 ± 3 BMI: 25.7 ± 0.9 CRP: 5.7 ± 1.9 | I: Flavanol-rich dark chocolate (Nestlé Noir Intense, Nestlé), 40 g | I: 36 | Double-blind, parallel group | Serum (0 h, 2 h) | CRP c | I: o C: o | |
| C: Flavanol-free chocolate (Nestlé), 40 g | C: 0 | ||||||||
| Davison et al., 2012 [47] | 14 | Healthy NS Age: 22 ± 1 BMI: n.d. CRP: n.d. | I: Flavanol-rich dark chocolate (Nestlé Noir Intense, Nestlé), 100 g | I: 97 | Crossover | Plasma | IL-6 | I: o C1: o C2: o | Cycling for 2.5 h at 60% maximum O2 uptake |
| Neutrophils from whole blood (0 h up to 1 h post-exercise) | Respiratory burst d | I: o C1: o C2: o | |||||||
| Degranulation e | I: o C1: o C2: o | ||||||||
| C1: Cocoa-free chocolate, 71 g | C1: 0 | ||||||||
| C2: No cocoa product | C2: 0 | ||||||||
| Flammer et al., 2012 [48] | 20 | Congestive heart failure, NS Age: 59 ± 3 BMI: 25.8 ± 1.0 CRP: 2.9 ± 0.7 | I: Flavanol-rich dark chocolate (Nestlé Noir Intense, Nestlé), 40 g | I: 36 | Double-blind, parallel group | Plasma (0 h, 2 h) | CRP c | I: o C: o | 24 h before low-flavonoid diet |
| C: Cocoa-liquor-free chocolate (Nestlé), 28.4 g, weight-matched for fat and sugar content | C: 0 | ||||||||
| Mellor et al., 2013 [49] | 10 | Type 2 diabetes, stable, treated with metformin or lifestyle Age: 61 (42–68) f BMI: 32.5 ± 6.0 CRP: n.d. | I: Flavanol-rich chocolate (Acticoa, Barry Callebaut), 13.5 g, 1 h before an oral glucose load | n.d. | Placebo-controlled, double-blind, crossover | Serum (0 h and 2 h after an oral 75-g-glucose load) | ICAM-1 | I: o C: ↑ ΔI < ΔC | 2 weeks run-in without polyphenol-rich foods |
| n.d. | E-selectin | I: o C: o ΔI < ΔC | |||||||
| C: Low-flavanol chocolate (Barry Callebaut), 13.5 g, 1 h before an oral glucose load | P-selectin | I: o C: o ΔI = ΔC | |||||||
| P-selectin-glyco-protein ligand 1 | I: o C: o ΔI < ΔC | ||||||||
| Vázquez-Agell et al., 2013 [46] | 18 | Healthy NS Age: 26 ± 7 BMI: n.d. CRP: n.d. | I: Cocoa-rich beverages (Nutrexpa) I1: with 250 mL milk I2: with 250 mL water | I1: 28 I2: 28 | Crossover | Serum (0 h, 6 h) | E-selectin | I1: o I2: ↓ C: o | 7 days cocoa-free run-in, 48 h before low-polyphenol-diet |
| ICAM-1 | I1: ↓ I2: ↓ C: o I2 < I1 (6 h) | ||||||||
| C: Pure milk, 250 mL | C: 0 | VCAM-1 | I1: o I2: o C: o | ||||||
| Peripheral blood mononuclear cells (0 h, 6 h) | P-p65/β-actin | I1: o I2: ↓ C: ↑ I2 < I1 (6 h) | |||||||
| Loffredo et al., 2014 [71] | 20 | Peripheral artery disease g Age: 69 ± 2 BMI: 27 ± 1 CRP: n.d. | I: Dark chocolate (≥85% cocoa), 40 g/day C: Milk chocolate (≤35% cocoa), 40 g/day | unclear | Crossover, single-blind | Serum (0 h, 2 h) | NOX2-derived peptide | I: ↓ C: o | |
| Basu et al., 2015 [50] | 18 | Type 2 diabetes, stable ≥5 years, no insulin, obese, NS Age: 56 ± 3 BMI: 35.3 ± 2.0 CRP: 5.3 ± 1.2 | I: Cocoa drink, provided in closed lip cups, consumed with a high-fat-fast-food-style breakfast | I: 40 | Double-blind, crossover | Serum (0 h, 0.5 h, 1 h, 2 h, 4 h, 6 h) | CRP c | I: o C: o ΔI = ΔC | 24 h before no polyphenol-rich food, 3 days food records |
| C: Cocoa-free placebo drink, provided in closed lip cups, consumed with a high-fat-fast-food-style breakfast | C: 0 |
| Study (Reference) | n | Participants | Intervention | EC (mg) | IP (d) | Study Design | Sample | Parameter | Results | Annotations |
|---|---|---|---|---|---|---|---|---|---|---|
| Grassi et al., 2005 [51] | 20 | Healthy NS Age: 34 ± 2 BMI: 22.6 ± 0.6 CRP: 0.3 ± 0.1 | I: Dark chocolate (Ritter Sport, Halbbitter, Ritter), 100 g/day | I: 66 | 15 | Crossover | Serum | CRP a | I: o C: o | No flavonoid-rich food, food diaries, 1 week run-in and 1 week washout without chocolate |
| C: White chocolate (Milka, Kraft Foods), 90 g/day | C: 0 | ICAM-1 | I: o C: o | |||||||
| Kurlandsky and Stote, 2006 [52] | 24 | Healthy NS, serum cholesterol 4.1–7.8 mmol/L, no lipid lowering medication Age: 43 ± 2 BMI: 25.0 ± 0.8 CRP: 4.7 ± 1.5 | I: Dark chocolate (Dove Silky Dark Chocolate, Mars), 41 g/day | n.d. | 42 | Parallel group | Serum | CRP a | I: o C: o | Flavonoid-rich food limited, 3 days food records before and in week 2, 4, and 6 ofintervention |
| C: No chocolate | ICAM-1 | I: ↓ C: o | ||||||||
| VCAM-1 | I: o C: o | |||||||||
| Crews et al., 2008 [53] | 88 | Healthy subjects with prehypertension Age: 69 ± 1 BMI: 25.3 ± 0.5 CRP: 1.6 ± 0.2 | I: Dark chocolate, 37 g/day, + cocoa beverage, 237 mL/day (Hershey) | n.d. | 42 | Placebo-controlled, double-blind, parallel group | Serum | CRP a | ΔI = ΔC | No flavonoid-rich food, 1 week run-in without flavonoid-rich food, compliance: daily records |
| C: Cocoa-free placebo products (Hershey) | n.d. | |||||||||
| Njike et al., 2011 [54] | 44 | Healthy NS, overweight/obese Age: 52 ± 2 BMI: 30.2 ± 3.4 CRP: 0.9 (median) | I: Cocoa-rich beverage with water (Hershey), 454 g/day I1: sugared I2: sugar- free (I2) | I1: 48 I2: 48 | 42 | Double-blind, crossover | Serum | CRP a | I1: o I2: o C: o | No flavonoid-rich food 24 h before, 3 days food diary during each treatment, 4 weeks washout |
| C: Cocoa-free sugared placebo beverage (Hershey), 454 g/day | C: 0 | |||||||||
| Tzounis et al., 2011 [55] | 22 | Healthy subjects Age: 30 ± 3 BMI: 23.2 ± 0.4 CRP: 0.3 ± 0.1 | I: Flavanol-rich cocoa drink (Mars), 150 mL/day | I: 89 | 28 | Placebo-controlled, double-blind, crossover | Plasma | CRP a | I: ↓ C: o | 2 weeks run-in, 4 weeks washout, no flavonoid-rich food, 4 days food diaries during run-in, 3rd week of both treatments, and during 4th week washout compliance: % used cocoa sachets, self-reported intake >95% |
| C: Low-flavanol cocoa drink (Mars), 150 mL/day | C: 3 | |||||||||
| Ibero-Baraibar et al., 2014 [56] | 50 | Healthy NS, overweight/obese Age: 57 ± 1 BMI: 30.6 ± 0.3 CRP: n.d. | I: Cocoa extract (1.4 g/day; Nutrafur) in ready-to-eat meals, within a hypocaloric diet (energy restriction of 15%) | I: 153 | 28 | Double-blind, parallel group | Plasma | ICAM-1 | I: ↓ C: ↓ | 1 week run-in without cocoa, 3 days before low-polyphenol-diet; exclude polyphenol-rich foods; compliance >98% |
| C: Ready-to-eat meals not enriched with cocoa extract, within a hypocaloric diet (energy restriction of 15%) | C: 0 | VCAM-1 | I: o C: o | |||||||
| Sarriá et al., 2014 [57] | 44 | Healthy NS, normocholesterolemic (n = 24; N) or moderately hypercholesterolemic (n = 20; H) Age: 29 ± 1 BMI: 23.6 ± 0.5 CRP: n.d. | I: Cocoa drink with cocoa, 30 g/day, rich in dietary fiber (Nutrexpa), with 400 mL semi-skimmed milk | I: 9.3 | 28 | Crossover | Plasma | CRP | n.d. | 2 weeks run-in and during intervention, polyphenol-rich foods were restricted, 3-day food records, compliance measured |
| IL-1β | I: o C: o (N, H) | |||||||||
| IL-6 | I: o C: o (N, H) | |||||||||
| C: Cocoa-free drink with semi-skimmed milk, 400 mL/day | C: 0 | IL-8 | I: o C: o (N, H) | |||||||
| IL-10 | I: ↓ C: ↓ Ip < Cp (N, H) | |||||||||
| TNF-α | I: o C: o (N, H) | |||||||||
| MCP-1 | I: o C: o (N, H) | |||||||||
| VCAM-1 | I: o C: o (N, H) | |||||||||
| ICAM-1 | I: o C: o (N, H) | |||||||||
| West et al., 2014 [58] | 13 | Healthy NS, Overweight/obese Age: 52 ± 0.3 BMI: 27.8 ± 0.2 CRP: n.d. | I: Dark chocolate, 37 g/day + cocoa drink with 22 g cocoa/day | I: 73.6 | 42 | Placebo-controlled, crossover | Plasma | CRP a | n.d. | 2 days before no flavonoid-rich foods, 2 weeks washout |
| C: Low-flavanol chocolate + cocoa-free drink as color matched control | IL-1β | I: o C: o | ||||||||
| C: 0.9 | IL-6 | I: o C: o | ||||||||
| TNF-α | I: o C: o | |||||||||
| McFarlin et al., 2015 [59] | 24 | Healthy subjects with normal weight (n = 10; N), overweight (n = 7; O), or obesity (n = 7; B), Age: 22 ± 2 BMI: 21.6 ±1.9 (N), 27.0 ± 1.4 (O), 34.9 ± 9.9 (B) CRP: n.d. | I: Cocoa bars with 12.7 g natural cocoa (Hershey) C: Cocoa-free placebo bar (Hershey), matched for energy content and macronutrient composition | I: 48.0 C: 0 | 42 | Placebo-controlled, crossover | Serum/Plasma | CRP a | n.d. | no chocolate during study, 2 weeks washout |
| AGP | n.d. | |||||||||
| AMG | n.d. | |||||||||
| Adipsin | n.d. | |||||||||
| Haptoglobin | I: o (N,O), ↓ (B) C: o (N, O, B) t0: B > O > N | |||||||||
| E-selectin | I: o (N, O, B) C: o (N, O, B) t0: B > O, N | |||||||||
| SAP | n.d. | |||||||||
| Endocan-1 | I: o C: o (N, O, B) | |||||||||
| IL-1β | n.d. | |||||||||
| IL-6 | n.d. | |||||||||
| IL-8 | n.d. | |||||||||
| TNF-α | n.d. | |||||||||
| Non-inflammatory monocytes (CD16-) | CD11b | n.d. | ||||||||
| Proinflammatory monocytes (CD16+) | CD11b | n.d. | ||||||||
| All Monocytes | CD62L | I: ↑ (N) o (O), ↓ (B) C: o (N, O, B) t0: B > O > N | ||||||||
| Proinflammatory monocytes (CD16+) | CD62L | I: ↑ (N, O), ↓ (B) C: o (N, O, B) t0: B > O > N | ||||||||
| EMP in whole blood | EMP concentration | I: o (N, O), ↓ (B) C: o (N, O, B) t0: B > O > N |
| Study (Reference) | n | Participants | Intervention | EC (mg) | IP (d) | Study Design | Sample | Parameter | Results | Annotations |
|---|---|---|---|---|---|---|---|---|---|---|
| Grassi et al., 2005 [51] | 20 | Untreated grade I hypertension, NS Age: 44 ± 2 BMI: 25.4 ± 0.3 CRP: 0.4 ± 0.1 | I: Flavanol-rich dark chocolate (Ritter Sport Halbbitter, Ritter), 100 g/day | I: 66 | 15 | Crossover | Serum | CRP a | I: o C: o | No flavonoid-rich food, Food diaries, 1 week chocolate free run-in and washout |
| ICAM-1 | I: o C: o | |||||||||
| C: White chocolate (Milka, Kraft Foods), 90 g/day, matched for energy, macro-, micronutrients | C: 0 | |||||||||
| Wang-Polagruto et al., 2006 [60] | 32 | Hypertension stage 1 or prehypertension, hypercholesterolemia, NS Age: 57 ± 1 BMI: 25.1 ± 0.6 CRP: n.d. | I: Flavanol-rich cocoa drink with 18.8 g cocoa powder (Mars), sucrose, 240 mL/day | n.d. | 42 | Double-blind, parallel group | Plasma | P-selectin | I: o C: o | No flavonoid-rich food 24 h before study, 2 weeks run-in with flavanol-poor cocoa drink, 3 × 3 days Food records, Compliance: empty packets |
| E-selectin | I: o C: o | |||||||||
| C:_Flavanol-poor cocoa drink, 240 mL/day | n.d. | ICAM-1 | I: o C: o | |||||||
| VCAM-1 | I: ↓ C: o ΔI > ΔC | |||||||||
| Grassi et al., 2008 [61] | 19 | Untreated stage I hypertension, impaired glucose tolerance, NS Age: 45 ± 1 BMI: 26.5 ± 0.3 CRP: n.d. | I: Flavanol-rich chocolate (Cuorenero, Sugar Company), 100 g/day | I: 111 | 15 | Crossover | Serum | CRP a | I: o C: o | No flavonoid-rich food, 1 week cocoa-free run-in and washout, Food records daily |
| C: White chocolate (Milka, Kraft Foods), 100 g/day | C: 0 | |||||||||
| Muniyappa et al., 2008 [62] | 20 | Stage 1 hypertension, overweight/obesity, NS, antihypertensive medication (n = 6) discontinued Age: 51 ± 2 BMI: 33.2 ± 1.4 CRP: n.d. | I: Flavanol-rich cocoa drink with cocoa powder (CocoaPro, Mars), water, 300 mL/day | I: 174 | 14 | Placebo-controlled, double-blind, crossover | Serum or plasma | E-selectin | I: o C: o | 1 week Run-in and 1 week washout with low-flavanol diet, Compliance: cocoa powder or placebo packets |
| ICAM-1 | I: o C: o | |||||||||
| VCAM-1 | I: o C: o | |||||||||
| C: Flavanol-poor placebo drink, with water, matched for energy, macronutrients, similar in color, taste and packaging, 300 mL/day | C: 2 | MCP-1 | I: o C: o | |||||||
| IL-6 | I: o C: o | |||||||||
| TNF-α | I: o C: o |
| Study (Reference) | n | Participants | Intervention | EC (mg) | IP (d) | Study Design | Sample | Parameter | Results | Annotations |
|---|---|---|---|---|---|---|---|---|---|---|
| Balzer et al., 2008 [63] | 41 | Type 2 diabetes, stably-treated, NS Age: 64 ± 1 BMI: 31.6 ± 0.8 CRP: 4.9 ± 1.1 | I: Flavanol-rich cocoa drink, with CocoaPro cocoa powder (Mars), 750 mL/day | I: 203 | 30 | Double-blind, parallel group | Plasma | CRP | I: o C: o | No dietary restrictions, dietary intake not determined, compliance: empty cocoa sachets, epicatechin in plasma |
| C: Flavanol-poor cocoa drink with CocoaPro cocoa powder (Mars), similar in taste, matched for energy and macro-, micronutrient composition, 750 mL/day | C: 17 | |||||||||
| Grassi et al., 2008 [61] | 19 | Impaired glucose tolerance, untreated stage I hypertension, NS Age: 45 ± 1 BMI: 26.5 ± 0.3 CRP: n.d. | I: Flavanol-rich chocolate (Cuorenero, Sugar Company), 100 g/day | I: 111 | 15 | Crossover | Serum | CRP a | I: o C: o | No flavonoid-rich food, 1 week cocoa-free run-in and washout, food records daily |
| C: White chocolate (Milka, Kraft Foods), 100 g/day | C: 0 | |||||||||
| Monagas et al., 2009 [64] | 42 | Diabetes or ≥3 cardiovascular disease risk factors (smoking, hypertension, hypercholesterolemia, obesity, family history of premature coronary heart disease) Age: 70 ± 2 BMI: 27.6 ± 0.8 CRP: 0.5 ± 0.3 | I: Cocoa drink, prepared with cocoa powder (Nutrexpa) and skim milk, 500 mL/day | I: 46 | 28 | Crossover | Serum or plasma | CRP a | I: o C: o | Flavonoid-rich food limited, 3 × 3 days food records, ompliance: patient reports and epicatechin metabolites in plasma and urine, 2 weeks run-in, no washout-period |
| C: Skim milk, 500 mL/day | C: 0 | P-selectin | I: ↓ C: o I < C (d28) | |||||||
| E-selectin | I: o C: o | |||||||||
| ICAM-1 | I: ↓ C: o I < C (d28) | |||||||||
| VCAM-1 | I: o C: o | |||||||||
| MCP-1 | I: o C: o | |||||||||
| IL-6 | I: o C: o | |||||||||
| Monocytes | LFA-1 | I: o C: o | ||||||||
| Mac-1 | I: o C: o | |||||||||
| VLA-4 | I: ↓ C: o I < C (d28) | |||||||||
| SLex | I: o C: o | |||||||||
| CD36 | I: ↓ C: o I < C (d28) | |||||||||
| CD40 | I: ↓ C: o I < C (d28) | |||||||||
| T cells | LFA-1 | I: o C: o | ||||||||
| VLA-4 | I: o C: o | |||||||||
| SLex | I: o C: o | |||||||||
| CD40 | I: o C: o | |||||||||
| Mellor et al., 2010 [65] | 12 | Type 2 diabetes, no steroids, no changes in chronic medication Age: 68 (median); 42–71 (range) BMI: n.d. CRP: 2.8 ± 0.5 | I: Polyphenol-rich chocolate (Nestlé), 45 g/day | I: 17 | 56 | Double-blind, crossover | Serum or plasma | CRP a | I: o C: o | 4 weeks washout, compliance: empty wrappers: 93.8%, no changes in diet and lifestyle, dietary recalls |
| C: Polyphenol-free chocolate (Nestlé), 45 g/day, matched for energy and macronutrients | C: <2 | |||||||||
| Stote et al., 2012 [66] | 19 | Impaired or normal glucose tolerance, obese, NS Age: 46 ± 1 BMI: 36.8 ± 0.2 CRP: n.d. | I: Cocoa drink with 28 g cocoa (different flavanol content, Mars), water, 300 mL/day | I1: 184 I2: 72 I3: 34 | 5 | Single-blind, crossover | Serum or plasma (0, 0.5, 1, 1.5, 2.0 h after an oral 75-g-glucose load) | CRP a | ↓ by dose | 10 days washout, no dietary changes |
| ICAM-1 | o by dose | |||||||||
| IL-6 | ↓ by dose | |||||||||
| C: Flavanol-poor drink with water (Mars), 300 mL/day, matched for energy, macro-, micronutrients | C: 4 | |||||||||
| Parsaeyan et al., 2014 [67] | 100 | Type 2 diabetes Age: 54 ± 1 BMI: 28 ± 0.5 CRP: n.d. | I:_Cocoa drink (10 g cocoa, 10 g milk powder, 250 mL water), 2 drinks/day | n.d. | 42 | Parallel group | Serum | CRPa | I: ↓ C: o | Dietary records, no restrictions |
| IL-6 | I: ↓ C: o | |||||||||
| TNF-α | I: ↓ C: o | |||||||||
| C: Cocoa-free drink (10 g milk powder, 250 mL water), 2 drinks/day | n.d. | |||||||||
| Rostami et al., 2015 [68] | 60 | Type 2 diabetes, blood pressure ≥140/90 mm Hg, stable medication, NS Age: 60 ± 1 BMI: 29.8 ± 0.6 CRP: unclear | I: Dark chocolate (Farmand), 25 g/day | n.d. | 56 | Placebo-controlled, double-blind, parallel group | Serum | CRP a | I: ↓ C: o; No effect by ANCOVA adjusted for age, sex, energy intake | No further chocolate, 3 days food records |
| C: White chocolate (Farmand), 25 g/day, isocaloric, same color and shape, identically wrapped | n.d. |
| Study (Reference) | n | Participants | Intervention | EC (mg) | IP (d) | Study Design | Sample | Parameter | Results | Annotations |
|---|---|---|---|---|---|---|---|---|---|---|
| Farouque et al., 2006 [69] | 40 | Coronary artery disease, 55% with hypertension, 95% with hypercholesterolemia, mostly medically treated Age: 61 ± 6 BMI: 27.5 ± 2.4 CRP: 1.2 (median) | I: Flavanol-rich chocolate (48 g/day; Mars) + one cocoa beverage per day (Mars) | I: 107 | 42 | Placebo-controlled, double-blind, crossover | Plasma | CRP a | I: o C: o | No dietary restrictions, dietary intake not determined, compliance: wrapper amount and patient reports |
| P-selectin | I: o C: o | |||||||||
| C: Isocaloric placebos | C: 5 | E-selectin | I: o C: o | |||||||
| ICAM-1 | I: o C: o | |||||||||
| VCAM-1 | I: o C: o I > C (d0) I = C (d42) | |||||||||
| Heiss et al., 2010 [70] | 16 | Coronary artery disease, medically treated, NS Age: 64 ± 3 BMI: 27.8 ± 1.8 CRP: 1.8 ± 0.4 | I: Flavanol-rich cocoa drink with CocoaPro cocoa powder (Mars), 2 drinks/day, prepared with skim milk or water | I: 59 | 30 | Double-blind, crossover | Plasma | CRP a | I: o C: o | 1 week washout |
| Peripheral blood mononuclear cells | Chemotaxis | I: o C: o | ||||||||
| C: Flavanol-poor cocoa drink, 2 drinks/day, with skim milk or water, matched for energy, macro-, micronutrients; similar in taste and package | C: 1 | |||||||||
| Flammer et al., 2012 [48] | 20 | Congestive heart failure, NS Age: 59 ± 3 BMI: 25.8 ± 1.0 CRP: 2.9 ± 0.7 | I: Flavanol-rich dark chocolate (Nestlé Noir Intense, Nestlé), 40 g/day | I: 36 | 28 | Double-blind, parallel group | Plasma | CRP a | I: o C: o | 24 h before flavonoid-low diet |
| C: Cocoa-liquor-free chocolate (Nestlé), 28.4 g/day, matched for fat and sugar content, identically wrapped | C: 0 | |||||||||
| Horn et al., 2014 [72] | 16 | Coronary artery disease, NS, medically treated, 38% diabetes, 88% hypertension, 96% hyperlipidemia, 63% prior smoking Age: 64 ± 1 BMI: 28.8 ± 0.5 CRP: 1.8 ± 0.1 | I: Flavanol-rich cocoa drink with cocoa powder (Mars), 2 drinks/day | I: 118 | 30 | Double-blind, crossover | Plasma | EMP (CD41+) | I: o C: o | 1 week washout |
| C: Flavanol-poor cocoa drink with cocoa powder (Mars), 2 drinks/day, matched for macro- and micronutrients, energy, and methylxanthines | C: 2 | EMP (CD144+) | I: ↓ C: o I < C (d30) | |||||||
| EMP (CD31+/41-) | I: ↓ C: o I < C (d30) |
| Study (Reference) | Allocation Concealment | Masking of Participants | Masking of Researchers | Dropouts Clearly Reported | Industry Funding | Cocoa Products as Gift from Industry | Compliance Measured | Crossover Design | Carry-Over Effects Possible a | Dietary Intake Documented | Dietary Restrictions |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Bolus consumption | |||||||||||
| Schramm et al., 2001 [43] | Y | Y | Y | Y | N | Y | - | Y | N | - | Y |
| Heptinstall et al., 2006 [44] | ? | Y | Y | - | N | Y | - | Y | N | - | Y |
| Flammer et al., 2007 [45] | Y | Y | Y | - | N | Y | - | N | - | - | Y |
| Davison et al., 2012 [47] | Y | N | Y | - | Y | Y | - | Y | N | Y | Y |
| Flammer et al., 2012 [48] | Y | Y | Y | Y | Y | Y | - | N | - | - | Y |
| Mellor et al., 2013 [49] | Y | Y | Y | - | Y | Y | - | Y | N | - | Y |
| Vázquez-Agell et al., 2013 [46] | ? | N | N | Y | N | Y | - | Y | N | - | Y |
| Loffredo et al., 2014 | Y | N | Y | - | N | N | - | Y | N | - | N |
| Basu et al., 2015 [50] | ? | Y | Y | - | N | N | - | Y | N | - | Y |
| Regular consumption—healthy | |||||||||||
| Grassi et al., 2005 [51] | ? | N | N | Y | N | ? | N | Y | N | Y | Y |
| Kurlandsky and Stote, 2006 [ 52] | Y | N | N | Y | N | Y | N | N | - | Y | Y |
| Crews et al., 2008 [53] | Y | Y | Y | Y | Y | Y | Y | N | - | N | Y |
| Njike et al., 2011 [54] | Y | Y | Y | Y | Y | Y | N | Y | N | Y | Y |
| Tzounis et al., 2011 [55] | Y | Y | Y | Y | N | Y | Y | Y | N | Y | Y |
| Ibero-Baraibar et al., 2014 [56] | ? | Y | Y | N | N | N | Y | N | - | Y | Y |
| Sarriá et al., 2014 [57] | N | N | N | Y | Y | Y | Y | Y | Y | Y | Y |
| West et al., 2014 [58] | ? | Y | Y | Y | Y | Y | N | Y | N | N | Y |
| McFarlin et al., 2015 [59] | ? | Y | Y | N | Y | Y | N | Y | N | N | Y |
| Regular consumption—pre-/hypertension | |||||||||||
| Grassi et al., 2005 [51] | ? | N | N | Y | N | ? | N | Y | N | Y | Y |
| Wang-Polagruto et al., 2006 [60] | ? | Y | Y | Y | Y | Y | Y | N | - | Y | Y |
| Grassi et al., 2008 [61] | ? | N | Y | Y | N | Y | N | Y | N | Y | Y |
| Muniyappa et al., 2008 [62] | Y | Y | Y | Y | N | Y | Y | Y | N | N | Y |
| Regular consumption—diabetes, impaired glucose tolerance | |||||||||||
| Balzer et al., 2008 [63] | ? | Y | Y | Y | N | Y | Y | N | - | N | N |
| Grassi et al., 2008 [61] | ? | N | Y | Y | N | Y | N | Y | N | Y | Y |
| Monagas et al., 2009 [64] | ? | N | Y | Y | N | Y | Y | Y | Y | Y | Y |
| Mellor et al., 2010 [65] | Y | Y | Y | Y | N | Y | Y | Y | N | Y | N |
| Stote et al., 2012 [66] | ? | N | Y | Y | N | Y | N | Y | N | N | Y |
| Parsaeyan et al., 2014 [67] | N | N | N | - | N | N | N | N | - | Y | N |
| Rostami et al., 2015 [68] | Y | N | Y | Y | N | Y | Y | N | - | Y | N |
| Regular consumption—coronary heart disease | |||||||||||
| Farouque et al., 2006 [69] | Y | Y | Y | N | Y | Y | Y | Y | Y | N | N |
| Heiss et al., 2010 [70] | ? | Y | Y | N | Y | Y | N | Y | N | N | N |
| Flammer et al., 2012 [48] | Y | Y | Y | Y | Y | Y | Y | N | - | N | Y |
| Horn et al., 2014 [72] | Y | Y | Y | - | Y | Y | N | Y | N | N | N |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellinger, S.; Stehle, P. Impact of Cocoa Consumption on Inflammation Processes—A Critical Review of Randomized Controlled Trials. Nutrients 2016, 8, 321. https://doi.org/10.3390/nu8060321
Ellinger S, Stehle P. Impact of Cocoa Consumption on Inflammation Processes—A Critical Review of Randomized Controlled Trials. Nutrients. 2016; 8(6):321. https://doi.org/10.3390/nu8060321
Chicago/Turabian StyleEllinger, Sabine, and Peter Stehle. 2016. "Impact of Cocoa Consumption on Inflammation Processes—A Critical Review of Randomized Controlled Trials" Nutrients 8, no. 6: 321. https://doi.org/10.3390/nu8060321
APA StyleEllinger, S., & Stehle, P. (2016). Impact of Cocoa Consumption on Inflammation Processes—A Critical Review of Randomized Controlled Trials. Nutrients, 8(6), 321. https://doi.org/10.3390/nu8060321






