1. Introduction
Despite the well-documented health and economic consequences of obesity [
1,
2,
3,
4], incidence rates have been steadily increasing since 1980 [
5]. This trend is generally attributed to a change in our dietary environment (e.g., [
6,
7,
8,
9]). Weight gain occurs when energy intake exceeds energy expenditure [
10]. In response, individuals are advised to eat less and/or to exercise more. Despite the apparent simplicity of this advice, most attempts to reduce body weight are either unsuccessful or weight that has been lost is regained over time [
11,
12].
An alternative approach is to change the environment in which people live [
13]. According to “nudge theory,” small targeted interventions (“nudges”) to change the environment (“choice architecture”) can have modest but cumulative effects on food intake and body weight [
14]. A nudge should fulfil the following criteria: (i) it should target automatic processes; (ii) it should have a reliable effect on behaviour; (iii) it should not affect freedom of choice; and (iv) it should not significantly change economic incentives [
14]. To date, studies evaluating nudge theory have explored the effects, inter alia, of reducing the availability of food [
15] and of changing the position of a food on a menu [
16]. In addition, reducing the portion size [
17] and the energy density [
18] of food can reduce both energy intake and body weight. A potential concern with the latter approach is that the pleasantness of a reformulated low energy-dense food can decrease over time (the “missing calorie effect”), which might undermine its efficacy as a weight-management product [
19,
20]. Here, we explored the extent to which it is possible to increase post-meal fullness and reduce meal size, simply by capitalizing on natural variation in the oral processing of everyday meals.
“Oral processing” refers to the pattern of behaviours associated with eating a food (e.g., bites, chews, swallows, and rate of eating). The degree to which foods require oral processing may be one aspect of our “obesogenic” environment that has changed in recent years [
21]. Energy-dense foods are now available that can be consumed with relatively little oral processing [
22]. In both humans [
23,
24,
25] and rats [
26], food texture (
i.e., viscosity, hardness, and chewiness) moderates the satiety response to food. For this reason, some researchers have suggested that liquid calories (
i.e., energy-containing drinks) deliver weaker satiation than equicaloric solid foods [
27,
28,
29,
30]. This is because oral exposure is limited [
31]. Consistent with this interpretation, the effects of food viscosity on energy intake can be abolished when eating rate is controlled [
23,
32] and eating at a slower rate reduces meal size [
33,
34]. These acute effects are consistent with evidence that a faster eating rate is associated with a higher body mass index (BMI) [
35,
36,
37,
38]. Moreover, training obese adolescents to moderate their rate of eating produces a clinically significant and sustained reduction in body weight [
39].
To date, studies have tended to explore effects of eating rate by instructing participants to consume a single food faster or slower, or by modifying the texture of food and assessing the effects of these manipulations on food intake and self-reported hunger [
22,
33,
34]. Based on a recent meta-analysis [
33], these types of experimental manipulations tend to achieve a 60% difference in eating rate (g/min; range: 17% to 143%). However, it remains to be determined whether the same variation in eating rate is observed across a range of unmodified everyday foods.
Previously, only one study has explored the association between eating rate and satiation across a broad range of commonly consumed products [
40]. In this study, participants consumed taster portions (50 g) of a wide range of foods and beverages. Participants consumed these small portions (solid foods were pre-cut) and were instructed to eat without pausing between bites. Each participant was then offered
ad libitum access to the same sample. Intake correlated positively with rate of consumption. However, drinks were included, together with a wide variety of foods (e.g., snacks, main meals, and confectionery). This raises questions about the extent to which this relationship would be observed across a range of otherwise similar meals that would normally be consumed in the same context. One other study has explored associations between oral processing behaviours and expected satiation [
41]. Again, participants consumed small taster portions (50 g). Foods that were eaten at a slower rate, with smaller bite sizes, and with more orosensory exposure were expected to confer greater satiation. These findings are encouraging because they suggest that foods might be modified to increase satiation and satiety by promoting oral processing. However, to test this proposition further it is essential to demonstrate these relationships in full portions of everyday main meals, eaten on separate occasions. For the first time, this study quantified variation in oral processing across a range of unmodified main meals and explored the relationship between this variation and measures of satiation and satiety.
In Study 1, our primary objective was to establish the extent to which measures of expected satiation, satiation, and satiety are associated with differences in the oral processing of commonly consumed main meals. Female participants attended the laboratory over 20 lunchtime sessions. In each session, they consumed a 400-kcal portion of a different commercially-available pre-packaged main meal. Prior to consumption, expected satiation was assessed. During each meal, oral processing behaviours were characterised using video-recordings of the mouth and real-time measures of plate weight. Hunger and fullness ratings were elicited pre- and post-consumption. In Study 2, we sought to; (i) demonstrate that differences in oral processing (observed in Study 1) are sufficient to influence food intake and; (ii) establish that the effects of oral processing can be observed when both males and females are included in the sample and across different test foods that are matched for their portion size (g). We selected two meals (same energy density) from Study 1 that had very different effects on oral processing but were equally liked. On separate days, participants consumed a 400-kcal portion of either the “fast” or “slow” meal, followed by an ad libitum meal.
3. Interim Discussion
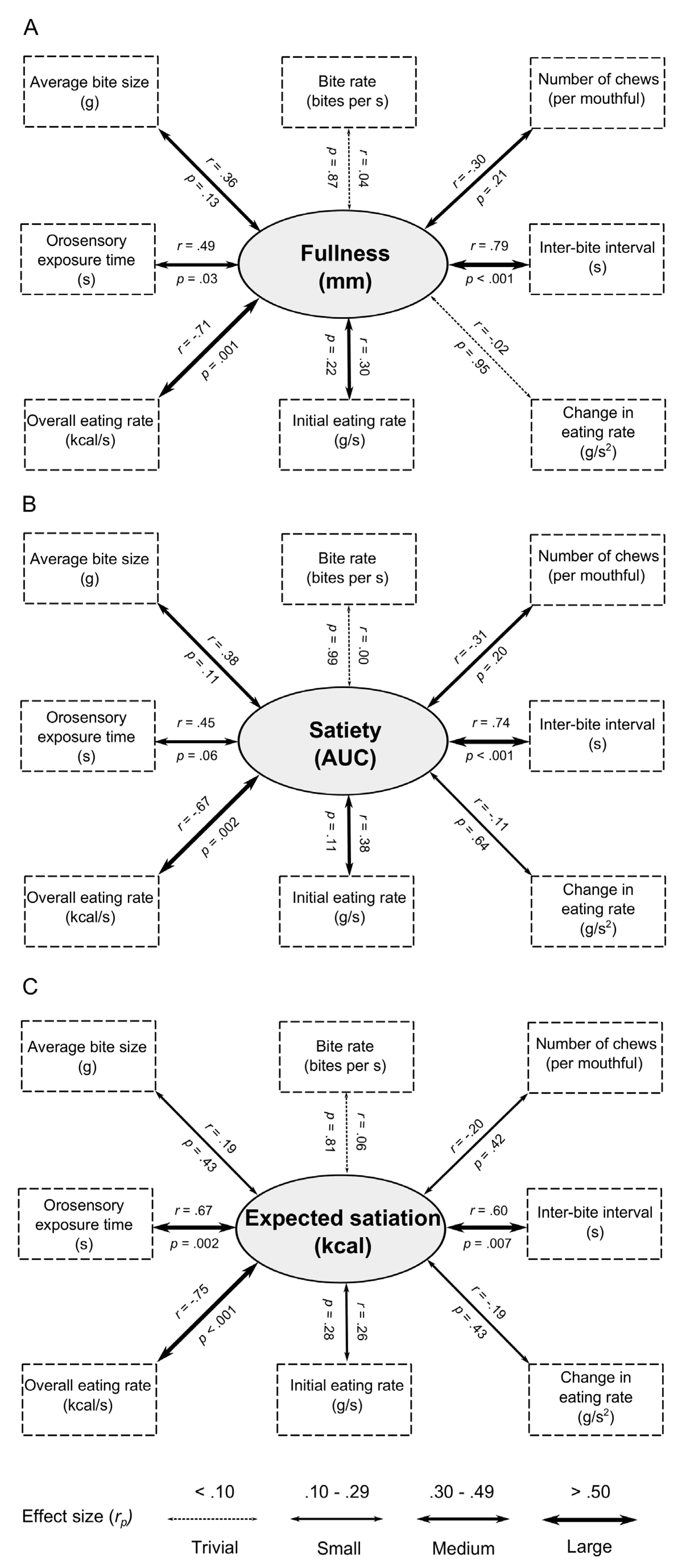
In Study 1 we sought to quantify variation in the oral processing of pre-packaged meals and to determine whether this is associated with differences in post-meal fullness and expected satiation. In the first instance, we wanted to establish whether there might be meaningful differences in oral processing across a single product category—commercially available pre-packaged meals. Our analysis revealed large differences in every measure of oral processing. Forde
et al. [
41] found that sample portions (50 g) of savoury meal components had an orosensory exposure time that ranged between 28.0 s for canned tomatoes to 349.0 s for tortilla chips (a 12 fold difference). In our study, orosensory exposure varied from 169.0 s for a 285 g portion of sausage and mashed potato to 426.9 s for a 316 g portion of vegetable biryani (a 2.5 fold difference). Nevertheless, we still observed strong relationships between our measures of oral processing. Consistent with related observations [
31], we found a very strong negative relationship between eating rate (kcal/s) and orosensory exposure time (
r = −0.92), and between eating rate and inter-bite interval (
r = −0.65).
We also observed large variation in the other dependent measures. Respectively, liking, expected satiation, post-meal fullness, and actual satiety varied across our test foods by 61%, 93%, 33%, and 32%. This is consistent with previous observations that equal-energetic servings of different foods differ greatly in their expected [
72] and actual [
42] satiation and satiety.
We hypothesised that our measures of oral processing would correlate with measures of satiation and satiety. Our across-food analysis revealed strong relationships between satiation (fullness) and more orosensory exposure time, longer pauses between bites, and a slower eating rate. These measures of oral processing also predicted satiety (fullness post-meal over three hours). Together, these findings are consistent with studies that have manipulated oral processing by modifying food texture or by explicitly instructing participants to eat faster or slower than normal [
21,
24,
25,
33,
34,
60]. However, this is the first study to demonstrate that these relationships can be observed in the variation of oral processing associated with a range of unmodified main meals.
Here, we have taken a correspondence between oral processing, post-meal fullness and satiety to indicate evidence for a causal effect of oral processing on satiation and satiety. However, we acknowledge that alternative explanations exist. One possibility is that differences in food volume mediate these relationships (i.e., items that are larger require more oral processing and also promote greater fullness). To address this concern, in our subgroup analyses, we selected two subsamples of the 20 foods that were closely matched for their energy density. Despite the fact that we reduced our power from 20 to 5 foods, a significant relationship was still observed between orosensory exposure time and satiety (AUC response). Although, we note that this was only evident in our low energy-density subsample. In our assessment of post-meal fullness, correlations failed to reach significance. Therefore, based on the current data, we are unable to dismiss an explanation based on a possible mediating effect of food volume. Nevertheless, in both subsamples, the effect sizes were generally unmodified, suggesting a lack of statistical power (caused by a reduction in sample size) rather than a change in trend. For example, and importantly, in both the high and low energy-density sub-groups, moderate to large effect size negative correlations were still observed between eating rate, satiation and satiety. Explanations based on the small differences in food volume cannot account for this pattern of results.
In our across-food analysis of the relationships between oral processing behaviours and expected satiation, we also observed the same significant associations with orosensory exposure time, inter-bite interval and eating rate. These data suggest that the effects of oral processing on satiation and satiety might be learned and anticipated before a meal begins. This relationship with expected satiation is important, because expected satiation plays a central role in the control of meal size and food intake [
48,
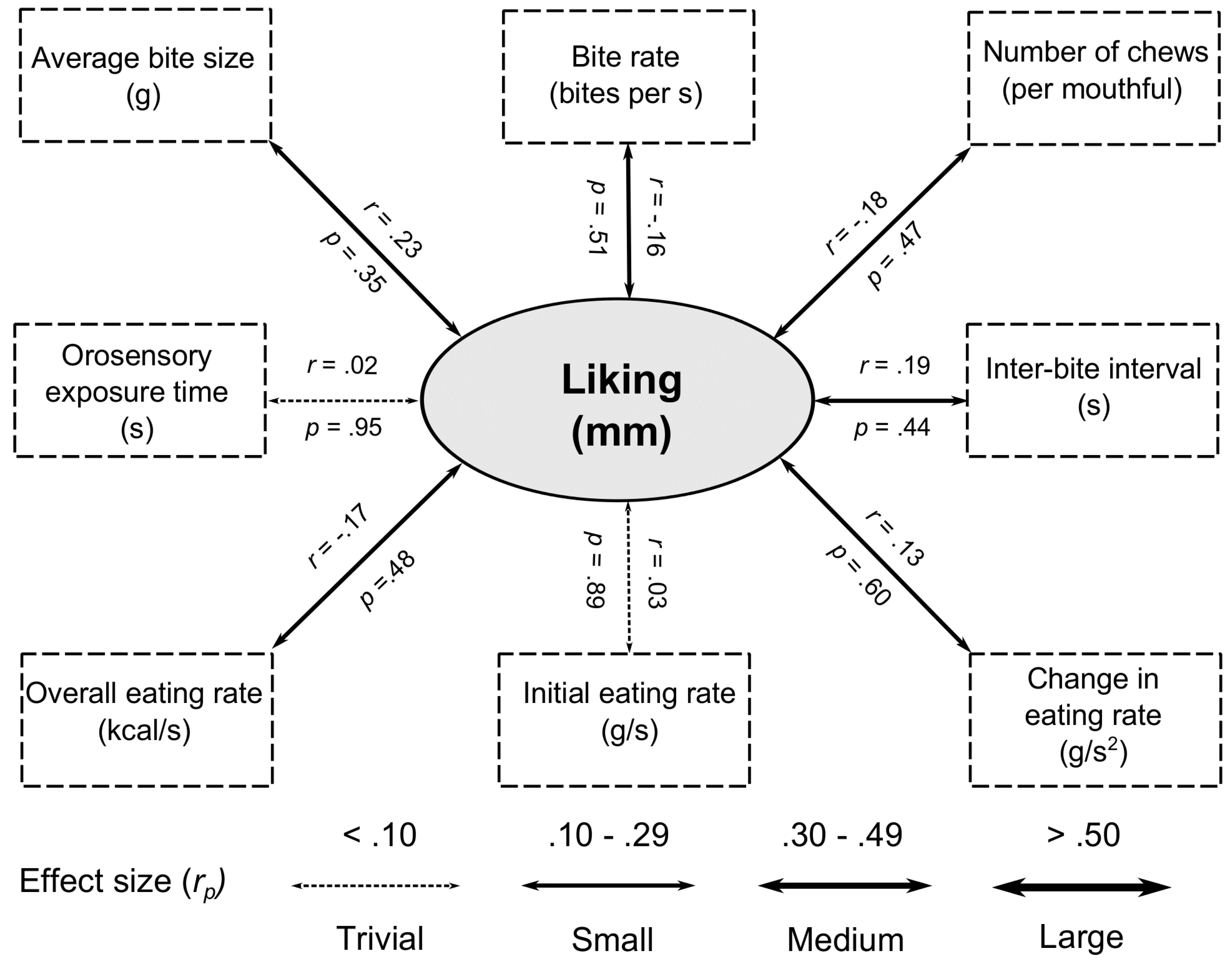
73]. By contrast, we failed to find significant relationships between liking and any oral processing characteristic. All of the associated effect sizes were small or trivial, suggesting that variations in liking of everyday meals is not sufficient to impact oral processing.
We also observed that foods that were expected to deliver greater satiation before eating were associated with greater post-meal satiation and satiety. These associations are consistent with previous research which has manipulated expected satiety for a fruit smoothie and shown that this has a marked effect on post-meal hunger and fullness [
67,
74].
Together, the results suggest that foods that are eaten slower, with longer pauses between bites, and more orosensory exposure, deliver more satiation/satiety and that these relationships are expressed in beliefs associated with the expected satiation of foods. However, three outstanding questions remain. First, we have established relationships between oral processing and subjective ratings of hunger and fullness (fullness composite score). Although these measures may reflect a proclivity to engage in eating they may not predict the amount of food that will be eaten [
73,
75,
76]. It remains to be determined whether the variation in oral processing observed here has a meaningful effect on subsequent food intake.
Second, as noted earlier, our subgroup analysis involved a comparison of foods that were closely matched for their weight. Our strategy reflects an attempt to control for the effect of differences in food volume (weight) to show independent effects of oral processing. Even after controlling for food weight, several relationships between oral processing, expected satiation and actual satiety remained and the effects sizes of the relationships were generally unmodified. However, as noted earlier, our subgroup analysis also suffered from reduced statistical power. To address this concern, the effects of oral processing should be explored in foods that are matched for their energy density. Finally, in Study 1, we tested only females. It remains to be determined whether relationships with oral processing are also observed in males.
In response to these issues we conducted a second study. Specifically, we had two objectives. First, we explored whether the differences in oral processing that were observed in Study 1 can influence food intake at a subsequent ad libitum meal. Second, we sought to establish whether the effects of oral processing can be observed when both males and females are included in the sample and across different test foods that are otherwise matched for their portion size (g) and energy density.
5. General Discussion
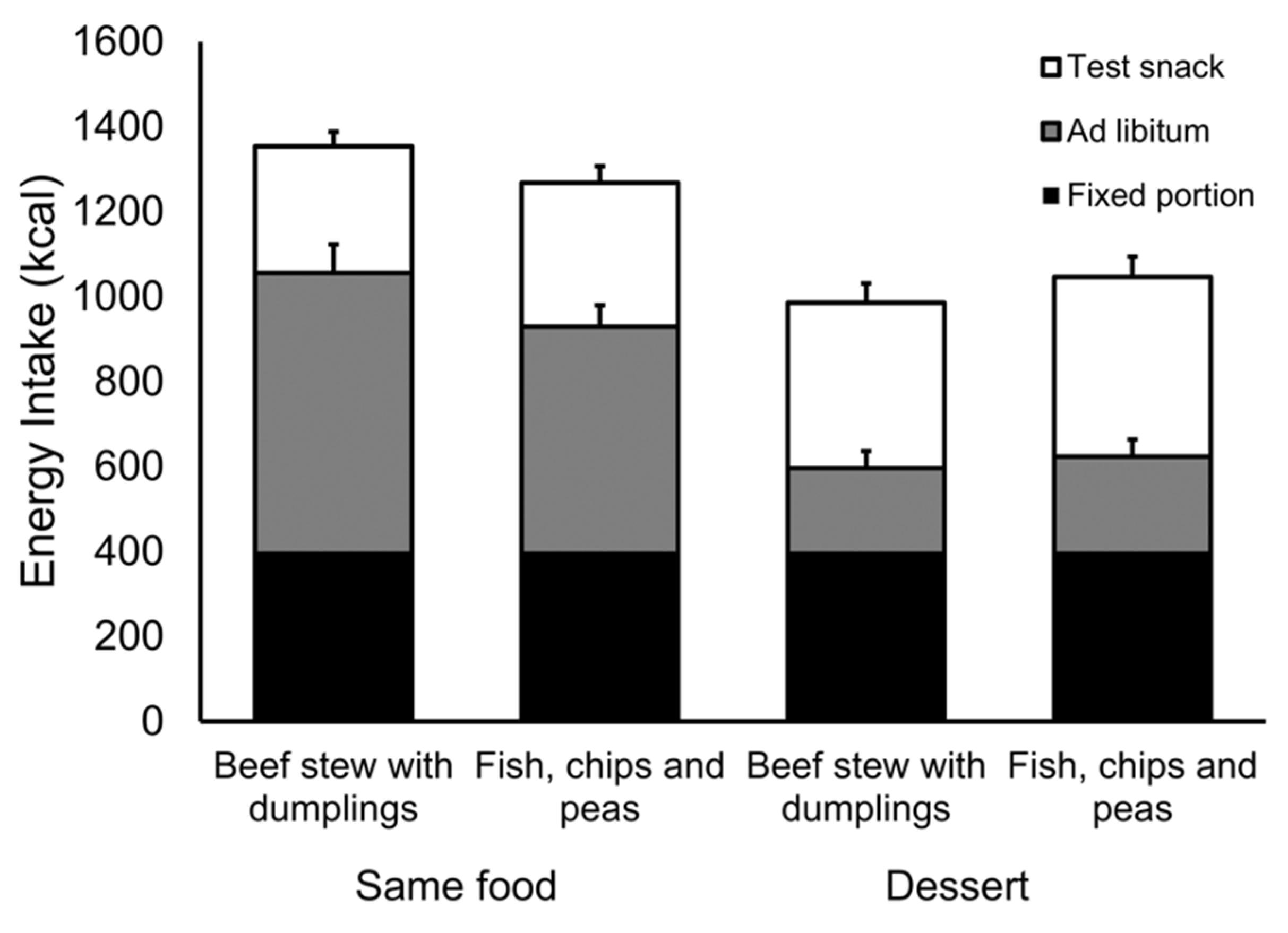
Study 1 demonstrated that across a range of unmodified hot meals, variation in oral processing is associated with differences in expected satiation and with fullness and satiety after the meals have been consumed. In Study 2 we compared two meals, one that is consumed faster than the other. They were otherwise matched for their liking, energy density, and portion size. Nevertheless, the slow meal produced greater fullness and it reduced subsequent food intake relative to the fast meal. We found little evidence that participants compensated for this difference in intake at a subsequent snack meal.
Our selection of foods in Study 2 was based on the assumption that they would promote different oral processing characteristics. From the outset we had a concern about “regression to the mean”. Specifically, that differences in oral processing observed in our sample in Study 1 might otherwise be attributed to chance and that these differences would not be replicated in Study 2. It was gratifying to see very similar effects preserved across studies and across pairs of ad libitum meal types (same or dessert). On this basis we are reasonably confident that differences in the oral processing of our test meals are reliable.
In Study 2 the effect of oral processing was only evident in the intake of the ad libitum meal when it was the same food as the fixed portion. Intake of the dessert was relatively small and we suspect that a dislike for this food masked the same effects of oral processing on intake. Consistent with this proposition, irrespective of fixed-portion meal type (fast or slow), participants reported relatively less fullness after consuming the dessert and consumed more at the ad libitum snack meal, one hour later. Alternatively, it may be relevant that participants were exposed to the slow and fast foods to a greater extent in the same conditions than in the dessert conditions. Thus, there was more opportunity for the differential effects of oral processing to be expressed. This cumulative effect (fixed portion + ad libitum meal) might explain why we saw significant differences in the same conditions but not in the dessert conditions.
Estimates suggest that a reduction of 100 kcal per day would be sufficient to prevent weight gain in 90% of the population [
77]. Although we note that the exact value of this kilocalorie reduction per day will vary considerably between individuals [
78]. Our observed effect of oral processing on intake was modest (6.5% across the entire 150 min test session—86 kcal). Nevertheless, it would be sufficient to achieve this target. Broadly, this approach is consistent with the idea of a dietary nudge—the prospect that a small reduction in energy intake (between 8% and 16% per day) can be implemented by stealth and can have a considerable impact on health [
15,
16]. In this respect, we note that the effects were observed when both male and female participants were included in the sample and that both of the foods in Study 2 were equally liked. Moreover, they were rated as having the same textural qualities (hardness, chewiness
etc.), which suggests that awareness of these differences is not a prerequisite for a change in behaviour. Recent research has demonstrated that it might be possible to introduce subtle modifications to recipes or formulations to promote particular aspects of oral processing (e.g., orosensory exposure) [
24,
25]. Again, this might be achieved covertly and it contrasts a strategy based on reductions in energy density. At a population level, this might represent a viable target for food manufacturers to help to nudge their consumers to reduce energy intake. Further research is needed to establish that modifications to foods that promote particular aspects of oral processing have a reliable effect on behaviour. This work might consider the role of meal variety, flavour intensity, flavour complexity, and unit size. In addition, future research should consider the extent to which people adapt to manipulations in oral processing. In particular, our studies raise questions about compensation over longer periods, over a day or more. Based on previous observations [
17] we suspect this is unlikely. However, this remains to be determined.
Our primary concern was to investigate whether, and irrespective of individual differences in oral processing, specific oral processing variables are reliably associated with expected satiation, post-meal fullness, and satiety, across a range of foods. Nevertheless, we recognise that our measures of oral processing could equally be applied to the study of individual differences. Specifically, an alternative approach might be to assess a larger number of participants on a limited number of foods and to explore which oral processing behaviours are the best predictors of expected satiation, post-meal fullness, and satiety, within each food separately. It would also be interesting to explore whether particular individuals are more responsive to the effects of oral processing. This type of analysis was beyond the scope of our study but represents an interesting avenue for future research.
In this work we are not making strong claims about a causal role for oral processing in satiation and satiety. Our observations are primarily correlational in nature. Nevertheless, in this context, and as we note earlier, there is very strong independent evidence implicating oral processing as a causal determinant of satiation and satiety [
21,
79,
80]. For recent reviews see Robinson
et al. [
33] and Hogenkamp and Sciöth [
34].
Finally, this work raises questions about the underlying mechanism that supports these effects of oral processing. Historically, researchers have tended to refer to a general concept of “oral metering”—the mouth monitoring the arrival of nutrients [
81]. However, the precise cognitive and physiological event(s) that trigger this metering remain unclear and this merits continued investigation. More generally, de Graaf [
22] has argued that the taste system updates the brain about the inflow of nutrients via cephalic phase responses (CPRs). CPRs are physiological responses that prepare the body for the arrival of nutrients. In future, it would be interesting to look at naturally occurring relationships between CPRs and specific oral processing behaviours. In particular, this index of CPRs might take the form of a thermic response to the food [
82]. An alternative or additional possibility is that oral processing affects memory for recent eating [
83,
84] by promoting better encoding of a food that is eaten at a slower rate [
85]. These ideas are currently being explored in ongoing work in our group.