Diet Quality and Adequacy of Nutrients in Preschool Children: Should Rice Fortified with Micronutrients Be Included in School Meals?
Abstract
:1. Introduction
2. Experimental Section
2.1. Raw Material
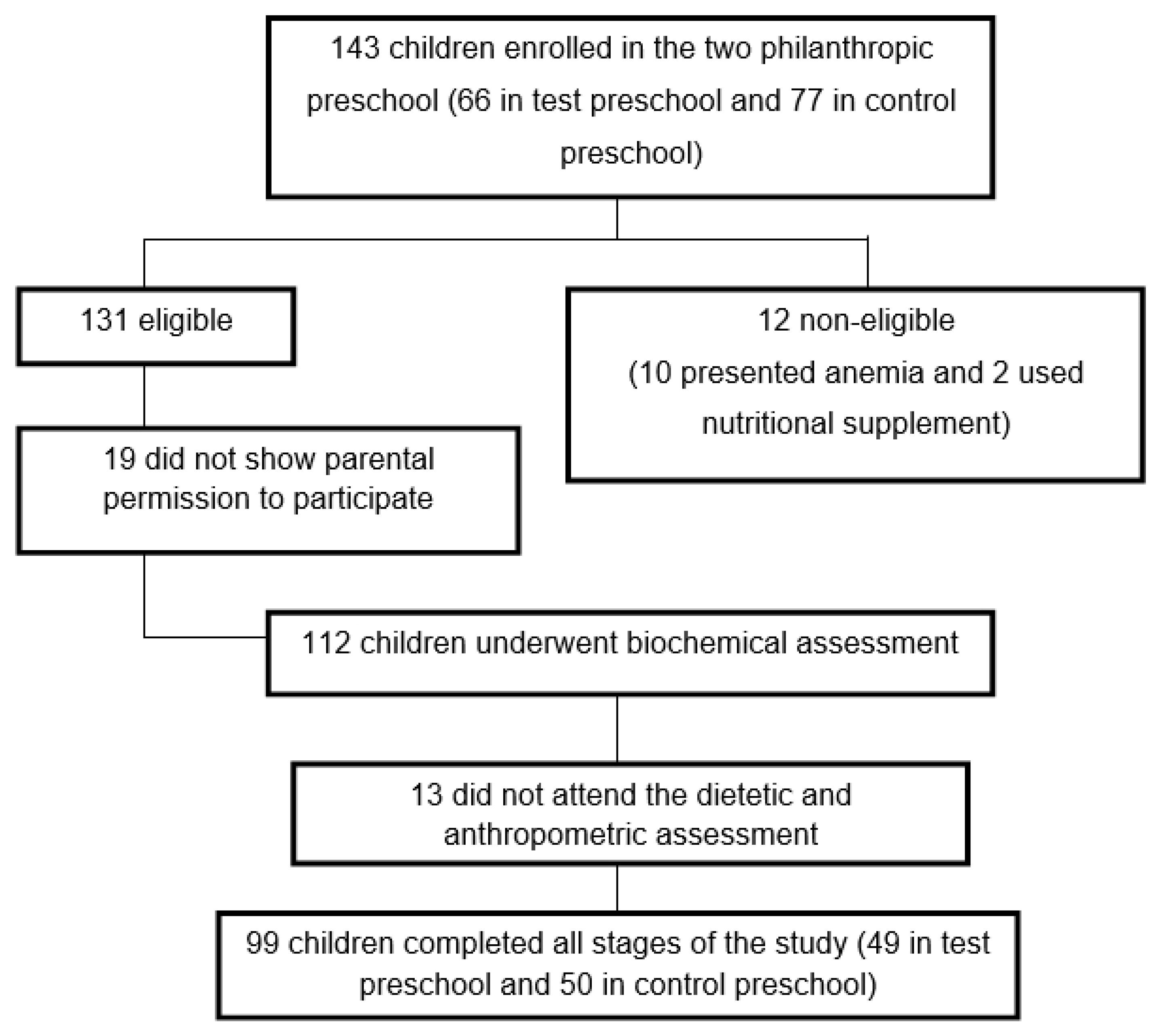
2.2. Sample
2.3. Experimental Design
2.4. Laboratory Tests
2.5. Dietary Assessment
2.6. Determination of the Healthy Eating Index (HEI)
2.7. Statistical Analysis
2.8. Ethical Aspects
3. Results
3.1. Sample Characterization
3.2. Laboratory Tests
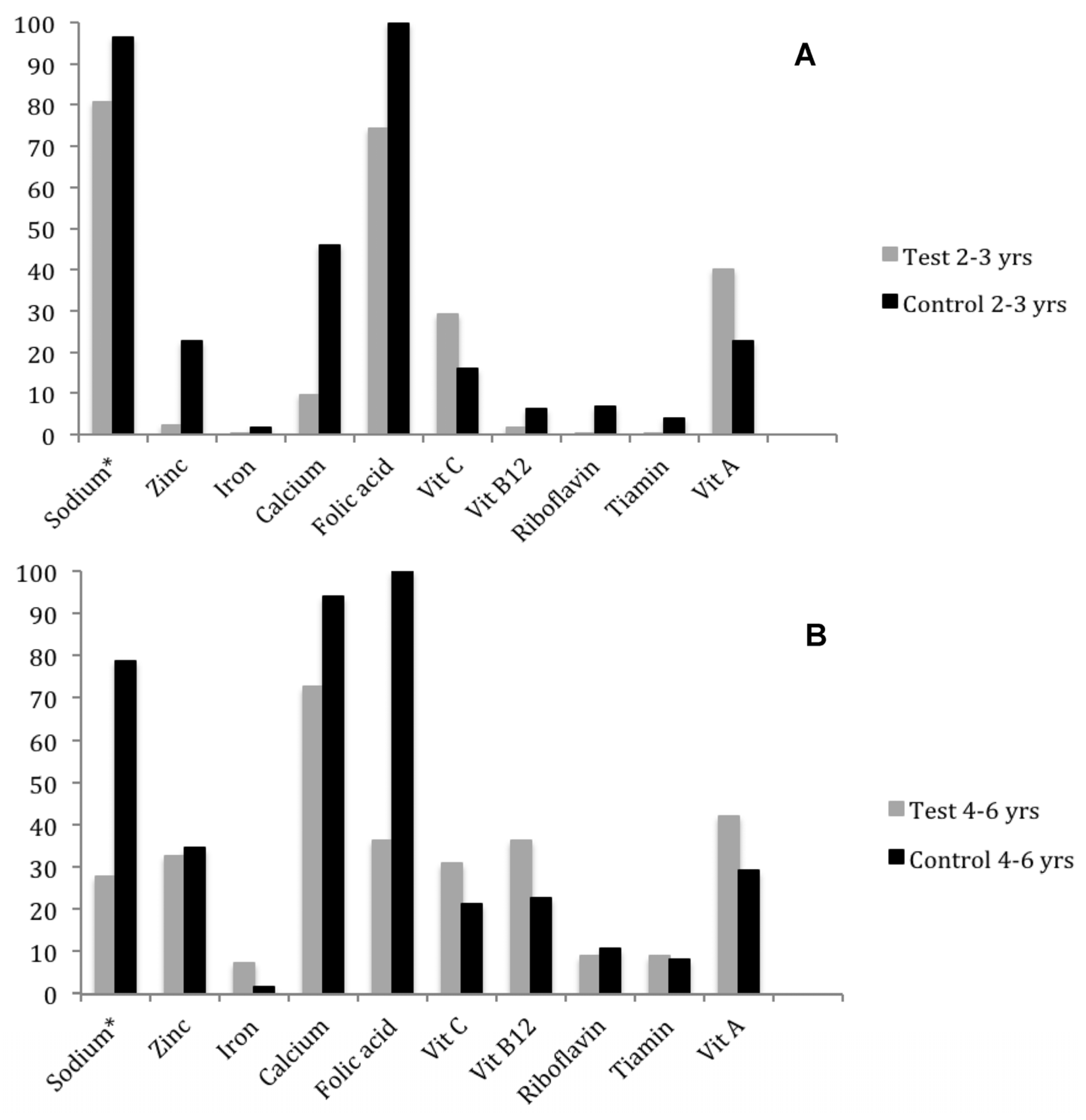
3.3. Consumption and Prevalence of Inadequate Nutrient
3.4. Comparison of Iron, Zinc, Vitamin and Folic Acid Intake with the Individual Consumption Target
3.5. Diet Quality Assessment of Preschool Children
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AI | Adequate Intake |
| DRI | Dietary Recommended Intake |
| EAR | Estimated Average Requirement |
| EER | Estimated Energy Requirement |
| SoFAAS | Calories from Saturated Fat, Added Sugar and Alcohol |
| HEI | Healthy Eating Index |
| RDA | Recommended Dietary Allowances |
| SD | Standard Deviation |
| UL | Upper Level |
| UR® | Ultra Rice® |
References
- Cagliari, M.P.P.; Paiva, A.A.; Queiroz, D.; Araujo, E.S. Food consumption, anthropometry and morbidity in preschool children from public day care centers of Campina Grande, Paraíba. Nutrire 2009, 34, 29–43. [Google Scholar]
- Santos, N.P.P. Avaliação dos HábitosAlimentares de Crianças Entre os 3 e os 7 anos de Idade do ExternatoLisbonense; Trabalho de Investigação, Faculdade de Ciências da Nutrição e Alimentação, Universidade do Porto: Porto, Portugal, 2010. [Google Scholar]
- Filha, E.O.S.; Araújo, J.S.; Barbosa, J.S.; Gaujac, D.P.; Santos, C.F.S.; Silva, D.G. Consumo dos gruposalimentaresemcriançasusuárias da redepública de saúde do município de Aracaju, Sergipe. Rev. Paul. Pediatr. 2012, 30, 529–536. [Google Scholar] [CrossRef]
- Von Grebmer, K.; Saltzman, A.; Birol, E.; Wiesmann, D.; Prasai, N.; Yin, S.; Yohannes, Y.; Menon, P. Global Hunger Index: The Challenge of Hidden Hunger; International Food Policy Research Institute (IFPRI): Bonn, Germany; Washinghton, DC, USA; Dublin, Republic of Ireland, 2014; pp. 1–54. [Google Scholar]
- Ruel-Bergeron, J.C.; Stevens, G.A.; Sugimoto, J.D.; Ross, F.F.; Ezzati, M.; Black, R.E.; Kraemer, K. Global update and trends of hidden hunger, 1995–2011: The hidden hunger index. PLoS ONE 2015, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Menezes, M.C.; Horta, P.M.; Santos, L.C.; LOPES, A.C.S. Avaliação do consumoalimentar e de nutrientes no contexto da atençãoprimária à saúde. Ceres 2011, 6, 175–190. [Google Scholar]
- Previdelli, A.N.; Andrade, S.C.; Pires, M.M.; Ferreira, S.B.G.; Fisberg, R.M.; Marchioni, D.M. Índice de Qualidade da DietaRevisado para populaçãobrasileira. Rev. Saude Publica 2010, 45, 794–798. [Google Scholar] [CrossRef]
- Triches, R.M. Promoção do consumoalimentarsustentável no contexto da alimentação escolar. Trab. Educ. Saúde 2015, 13, 757–771. [Google Scholar] [CrossRef]
- Vellozo, E.P.; Fisberg, M. A contribuição dos alimentosfortificadosnaprevenção da anemia ferropriva. Rev. Bras. Hematol. Hemoter. 2010, 32, 140–147. [Google Scholar] [CrossRef]
- Chakravarty, I. Food-based strategies to control vitamin A deficiency. Food Nutr. Bull. 2000, 21, 135–143. [Google Scholar] [CrossRef]
- Zancul, M.S. Fortificação de alimentos com ferro e vitamina A. Medicina 2004, 37, 45–50. [Google Scholar]
- Della Lucia, C.M.; Novaes, J.F.; Sant’Ana, H.M.P. Osdesafios da fortificação de alimentos. Bol. Cent. Pesqui. Process. Aliment. 2012, 30, 197–208. [Google Scholar]
- Lee, J.; Hamer, M.L.; Eitenmiller, R.R. Stability of retinylpalmitate during cooking and storage in rice fortified with ultra rice fortification technology. J. Food Sci. 2000, 65, 915–919. [Google Scholar] [CrossRef]
- Institute of Medicine (IOM). Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; The National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- Mera, M.; Thompson, H.; Prasad, C. How to calculate sample size for an experiment: A case-based description. Nutr. Neurosci. 1998, 1, 87–91. [Google Scholar]
- Institute of Medicine (IOM). Dietary References Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids; the National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Institute of Medicine (IOM). Dietary References Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium and Zinc; the National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Institute of Medicine (IOM). Dietary References Intakes for Calcium and Vitamin D; the National Academy Press: Washington, DC, USA, 2010. [Google Scholar]
- Guenther, P.M.; Reedy, J.; Krebs-Smith, S.M.; Reeve, B.B.; Basiotis, P.P. Development and Evaluation of the Healthy Eating Index-2005: Technical Report; Center for Nutrition Policy and Promotion: Alexandria, VA, USA; U.S. Department of Agriculture: Washington, DC, USA, 2007.
- Brasil. GuiaAlimentar Para a PopulaçãoBrasileira: PromovendoaAlimentaçãoSaudável; Ministério da Saúde: Brasília, Brasil, 2008.
- De Carvalho, M.C.; Baracat, E.C.E.; Sgarbieri, V.C. Anemia ferropriva e anemia de doençacrônica: Distúrbios do metabolismo de ferro. Segur. Alim. Nutr. 2006, 13, 54–63. [Google Scholar]
- Gleerup, A.; Rossander-Hulthén, L.; Gramatkovski, E.; Hallberg, L. Iron absorption from the whole diet: Comparison of the effect of two different distributions of daily calcium intake. Am. J. Clin. Nutr. 1995, 61, 97–104. [Google Scholar] [PubMed]
- Osório, M.M. Fatores determinantes da anemia em crianças. J. Pediatr. 2002, 78, 269–278. [Google Scholar] [CrossRef]
- Lynch, S.R. Interaction of iron with other nutrients. Nutr. Rev. 1997, 55, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.M.P.; Pereira, M.Z. Efeito da fortificação com ácidofóliconaredução dos defeitos do tubo neural. Cad. Saude Publica 2007, 23, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.; Tomar, S.K. Folate: A functional food constituent. J. Food Sci. 2009, 74, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Vieira, M.N.C.M.; Japur, C.C.; Resende, C.M.M.; Monteiro, J.P. Valores de referência de ingestão de nutrientes para avaliação e planejamento de dietas de crianças de um a oitoanos. Medicina 2008, 41, 67–76. [Google Scholar]
- Silva, A.P.; Vitolo, M.R.; Zara, L.F.; Castro, C.F. Effects of zinc supplementation on 1- to 5-year old children. J. Pediatr. 2006, 82, 227–231. [Google Scholar] [CrossRef]
- Dias, D.M.; Moreira, M.E.C.; Gomes, M.J.C.; Toledo, R.C.L.; Nutti, M.R.; Sant’Ana, H.M.P.; Martino, H.S.D. Rice and bean targets for biofortification combined wth high carotenoid content crops regulate transcriptional mechanisms increasing iron bioavailability. Nutrients 2015, 7, 9683–9696. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, K.B.F.; Costa, N.M.B.; Alfenas, R.C.G.; De Paula, S.O.; Minim, V.P.R.; Bressan, J. Estresseoxidativo: Conceito, implicações e fatoresmodulatórios. Rev. Nutr. 2010, 23, 629–643. [Google Scholar] [CrossRef]
- Lemos, S.B.; Liberali, R.; Coutinho, V.F.; Assumpção, C.O. Biodisponibilidade de ferro e a anemia ferroprivanainfância. Ens. Ciênc. Ciênc. Biol. Agrár. Saúde 2012, 16, 213–228. [Google Scholar]
- Program for Appropriate Technology in Health. An Invisible Bounty: PATH’s Manufactured “Grain” Adds Nutrients to Rice. Available online: http://www.path.org/projects/ultra_rice.php (accessed on 27 February 2013).
- Hiza, H.A.B.; Casavale, K.O.; Guenther, P.M.; Davis, C.A. Diet quality of Americans differs by age, sex, race/ethnicity, income, and education level. J. Acad. Nutr. Diet. 2013, 113, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Kourlaba, G.; Kondaki, K.; Grammatikaki, E.; Birbilis, M.; Oikonomou, E.; Roma-Giannikou, E. Diet quality of preschoolers in Greece based on the healthy eating index: The GENESIS study. J. Am. Diet. Assoc. 2009, 109, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Philippi, S.T.; Latterza, A.R.; Cruz, A.T.R.; Ribeiro, L.C. Pirâmidealimentaradaptada: Guia para escolha dos alimentos. Rev. Nutr. 1999, 12, 65–80. [Google Scholar] [CrossRef]
- Costa, F.P.; Machado, S.H. O consumo de sal e alimentosricosemsódiopodeinfluenciarnapressão arterial das crianças? Cienc. Saude Coletiva 2010, 15, 1383–1389. [Google Scholar] [CrossRef]
- Brasil. GuiaAlimentar Para a PopulaçãoBrasileira; Ministério da Saúde: Brasília, Brasil, 2014.
- Oliveira, C.L.; Fisberg, M. Obesidadenainfância e adolescência—Uma verdadeiraepidemia. Arq. Bras. Endocrinol. Metab. 2003, 47, 107–108. [Google Scholar] [CrossRef]
- Brião, M.S.; Geiger, J.M.; Silva, D.O.; Borges, L.R. Avaliação do consumo de açúcar dos frequentadores e colaboradores da Unidade de Alimentação e Nutrição de umaInstituiçãoFilantrópica de Pelotas/RS. In Proceedings of the XIX Congresso de IniciaçãoCientífica e da II MostraCientífica, Pelotas, Brasil, 11 November 2010.
| Component | Characteristic | Proposal HEI, 2005 * | Recommendation of the Brazilian Food Guide ** (in 2000 Kcal) | Recommendation of the Brazilian Food Guide *** (in 1000 kcal) |
|---|---|---|---|---|
| 1. Total fruit | Includes whole fruits and juice | Recommendation of fruits’ servings/1000 kcal | 3 servings | 1.5 serving |
| 2. Whole fruits | Excludes juice | Half of fruits’ recommendation/1000 kcal | 1.5 serving | 0.75 serving |
| 3. Total vegetable | Includes all vegetables. Legumes are part of this group only if the meat group’s recommendation is reached. | Recommendation of vegetables’ servings/1000 kcal | 3 servings | 1.5 serving |
| 4. Dark green and orange vegetables and legumes | Includes only dark green and orange vegetables. Legumes are part of this group only if the meat group’s recommendation is reached. | Daily sum of My Pyramid’s recommendations for the subgroups of dark green and orange vegetables/1000 kcal. Approximately 1/3 of the recommended servings for total vegetables/1000 kcal | 1.5 serving | 0.75 serving |
| 5. Total grains | Includes all foods from the grain group. | Recommendation of grain servings/1000 kcal | 6 servings | 3 servings |
| 6. Whole grains | Includes only whole grains and derivatives. | Half of the grain recommendation/1000 kcal | 3 servings | 1.5 serving |
| 7. Dairy products | Includes milk and dairy products and soy based products. | Recommendation of milk and dairy product servings/1000 kcal | 4 servings | 2 servings |
| 8. Meat, eggs and legumes | Includes meat, eggs and legumes. | Recommendation of meat servings/1000 kcal | 2 servings | 1 serving |
| 9. Oil | Includes vegetable oil and unhydrogenated vegetable oil, fish oil, nuts and seeds. | Recommendation of oil servings/1000 kcal | 1 serving | 0.5 serving |
| 10. Saturated fat | Corresponds to the percentage of total calories from the saturated fat. | Minimum limit (7%), intermediate (10%) and maximum (15%), according to the national recommendations. | Minimum limit (7%), intermediate (10%) and maximum (15%) | - |
| 11. Sodium | Corresponds to the ingestion of the mineral sodium in milligrams. | Estimative considering the sodium recommendation and the medium ingestion of energy. | Minimum limit (700 mg/1000 kcal), intermediate (1100 mg/1000 kcal) and maximum/1000 kcal) | - |
| 12. Calories from solid fats, sugar and alcohol (SoFAAS) | Corresponds to the calories from solid fat (saturated and trans in milk and dairy products, meat, butter, lard, chocolate); sugar (used in recipes and added) and alcoholic beverages. | Minimum limit corresponds to the percentile 86 (50%) and maximum limits for the 10 thpercentile (20%) | - | Minimum limits corresponds to the percentile 86 (10%) and maximum limits to the percentile 16 (35%) of the population |
| Components | Minimum Score (0) | Intermediate Score (8) | Maximum Score (5 *, 10 ** or 20 ***) | ||
|---|---|---|---|---|---|
| Total fruit 1 | 0 |  | 1.5 serving/1000 kcal * | ||
| Whole fruit 1 | 0 |  | 0.75 serving/1000 kcal * | ||
| Total vegetable 1 | 0 |  | 1.5 serving/1000 kcal * | ||
| Dark green and orange vegetables and legumes 1 | 0 |  | 0.5 serving/1000 kcal * | ||
| Total grains 1 | 0 |  | 3 servings/1000 kcal * | ||
| Whole grains 1 | 0 |  | 1.5 servings/1000 kcal * | ||
| Dairy products | 0 |  | 1.5 serving/1000 kcal ** | ||
| Meat, eggs and legumes | 0 |  | 1 serving/1000 kcal ** | ||
| Oil 1 | 0 |  | 0.5 serving/1000 kcal ** | ||
| Saturated fat 1 | ≥15% of TEV |  | 10% do VET |  | 7% of TEV ** |
| Sodium 1 | ≥2 g/1000 kcal |  | 1.1 g/1000 kcal |  | ≤0.7 g/1000 kcal ** |
| SoFAAS2 | ≥35% of TEV |  | ≤10% of TEV *** | ||
| Variables | Test Preschool | Control Preschool | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Before Intervention | After Intervention | Difference | p a | Before Intervention | After Intervention | Difference | p a | pBaseline b | |
| Erythrocytes (millions/mm3) | 4.90 ± 0.32 (4.92) | 4.85 ± 0.36 (4.87) | −0.04 ± 0.27 (−0.04) | 0.305 | 4.86 ± 0.42 (4.95) | 4.90 ± 0.34 (4.92) | 0.04 ± 0.29 (0.00) | 0.367 | 0.670 |
| Hemoglobin (g/dL) | 12.34 ± 0.80 (12.10) | 12.54 ± 0.88 (12.30) | 0.19 ± 0.64 (0.00) | 0.111 | 12.40 ± 0.87 (12.30) | 12.57 ± 0.95 (12.50) | 0.18 ± 0.64 (0.10) | 0.079 | 0.781 |
| Hematocrit (%) | 39.27 ± 2.27 (39.05) | 39.14 ± 2.82 (38.60) | −0.13 ± 2.16 (−0.20) | 0.696 | 38.87 ± 2.68 (38.40) | 39.17 ± 2.62 (38.75) | 0.29 ± 2.21 (0.25) | 0.334 | 0.464 |
| MCV (fL) | 80.33 ± 4.29 (80.94) | 80.66 ± 3.95 (81.54) | 0.32 ±1.24 (0.27) | 0.097 | 80.20 ± 4.81 (80.56) | 80.05 ± 4.71 (79.54) | −0.16 ± 2.16 (0.30) | 0.628 | 0.769 |
| MCH (pg) | 25.22 ± 1.53 (25.59) | 25.74 ± 1.53 (26.08) | 0.52 ±0.63 (0.60) | <0.001 | 25.59 ± 1.95 (25.90) | 25.61 ± 1.86 (25.53) | 0.02 ± 1.02 (0.16) | 0.223 | 0.314 |
| MCHC (%) | 31. 37 ± 0.56 (31.40) | 31.87 ± 0.66 (31.85) | 0.50 ± 0.73 (0.45) | <0.001 | 31.89 ± 0.78 (31.99) | 31.97 ± 0.68 (32.20) | 0.08 ± 0.77 (0.22) | 0.079 | 0.866 |
| Folic acid (ng/mL) | 17.62 ± 3.08 (19.50) | 19.72 ± 5.08 (21.00) | 2.10 ± 5.99 (3.20) | 0.003 | 17.38 ± 5.28 (17.35) | 17.58 ± 4.23 (17.35) | 0.20 ± 5.11 (0.00) | 0.796 | 0.962 |
| Thiamine (µg/L) | 72.12 ± 25.85 (66.67) | 194.76 ± 93.33 (145.84) | 122.64 ± 98.62 (85.11) | <0.001 | 58.29 ± 16.51 (53.69) | 101.19 ± 61.23 (95.19) | 42.89 ± 33.50 (37.19) | <0.001 | 0.012 |
| Ferritin (ng/mL) | 32.80 ± 15.89 (30.65) | 34.17 ± 16.10 (28.45) | 1.82 ± 14.50 (2.75) | 0.082 | 12.38 ± 0.84 (12.30) | 36.45 ± 15.34 (34.50) | 24.07 ± 15.53 (21.70) | <0.001 | <0.001 |
| Serum zinc (µg/dL) | 80.97 ± 11.88 (81.40) | 110.21 ± 22.76 (108.80) | 29.25 ± 23.31 (26.30) | <0.001 | 95.86 ± 21.90 (95.40) | 92.13 ± 15.92 (92.10) | −3.73 ± 27.76 (−7.20) | 0.378 | 0.070 |
| CRP (mg/dL) | 0.26 ± 0.59 (0.01) | 0.12 ± 0.32 (0.02) | −0.15 ± 0.44 (0.00) | 0.128 | 0.36 ± 1.06 (0.03) | 0.34 ± 0.57 (0.10) | −0.02 ± 1.15 (0.01) | 0.226 | 0.516 |
| Variables | 2 to 3 Years Old | 4 to 6 Years Old | ||||
|---|---|---|---|---|---|---|
| Test Preschool | Control Preschool | p | Test Preschool | Control Preschool | p | |
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |||
| Med (Min-Max) | Med (Min-Max) | Med (Min-Max) | Med (Min-Max) | |||
| Vitamin A | 276.82 ± 621.66 | 304.93 ± 125.47 | 0.090 | 435.41 ± 893.06 | 392.43 ± 207.05 | 0.040 |
| (RE) * | 206.12 (40.21–1300.38) | 271.72 (94.55–561.74) | 248.04 (38.74–4994.90) | 367.61 (117.70–947.14) | ||
| Thiamine | 1.28 ± 0.33 | 0.96 ± 0.32 | 0.003 | 2.70 ± 1.60 | 1.19 ± 0.49 | <0.001 |
| (mg) ** | 1.28 (0.69–2.14) | 0.91 (0.31–1.75) | 2.18 (0.98–7.53) | 1.04 (0.75–2.71) | ||
| Riboflavin | 1.49 ± 0.40 | 1.09 ± 0.46 | 0.004 | 1.42 ± 0.69 | 1.13 ± 0.51 | 0.052 |
| (mg) ** | 1.48 (0.53–2.32) | 1.04 (0.28–2.00) | 1.36 (0.44–3.51) | 1.02 (0.44–2.50) | ||
| Vitamin B12 | 2.75 ± 0.97 | 2.39 ± 1.10 | 0.244 | 4.33 ± 9.75 | 2.18 ± 1.61 | 0.063 |
| (µg) ** | 2.87 (0.63–5.20) | 2.21 (0.89–5.20) | 2.25 (1.14–54.67) | 1.94 (0.28–8.28) | ||
| Vitamin C | 48.64 ± 66.36 | 64.79 ± 50.63 | 0.006 | 36.34 ± 28.23 | 99.66 ± 94.73 | <0.001 |
| (mg) * | 24.09 (11.08–322.76) | 50.81 (21.54–221.74) | 29.66 (6.66–128.62) | 74.79 (16.86–456.33) | ||
| Folic acid | 99.68 ± 31.05 | 51.78 ± 22.99 | <0.001 | 218.19 ± 170.54 | 54.34 ± 24.61 | <0.001 |
| (µg) * | 92.46 (57.68–171.58) | 43.87 (21.54–96.57) | 160.79 (53.16–790.18) | 44.91 (24.22–125.75) | ||
| Calcium | 827.62 ± 255.93 | 520.35 ± 250.55 | <0.001 | 668.73 ± 220.91 | 495.70 ± 195.45 | 0.004 |
| (mg) ** | 802.89 (494.62–1392.31) | 518.78 (123.50–993.42) | 601.91 (321.96–1306.57) | 443.50 (190.08–908.68) | ||
| Iron | 8.94 ± 1.65 | 7.55 ± 2.18 | 0.021 | 10.34 ± 4.04 | 8.78 ± 2.19 | <0.001 |
| (mg) ** | 9.11 (5.52–11.81) | 7.53 (3.61–12.20) | 11.90 (5.92–13.52) | 9.05 (4.60–12.50) | ||
| Zinc | 5.31 ± 1.39 | 6.98 ± 5.93 | 0.207 | 10.83 ± 14.53 | 9.22 ± 12.39 | 0.063 |
| (mg) * | 5.02 (3.23–8.30) | 5.67 (3.27–33.70) | 7.11 (3.13–83.60) | 5.86 (3.97–65.30) | ||
| Sodium | 1934.95 ± 313.51 | 2551.54 ± 709.51 | 0.001 | 1750.76 ± 695.22 | 2948.56 ± 1083.02 | <0.001 |
| (mg) * | 1951.70 (1320.60–2596.60) | 2539.55 (1432.00–3934.30) | 1581.35 (797.60–3986.50) | 2636.05 (1647.20–5682.10) | ||
| Variables | Test preschool | Control Preschool | p | ||||
|---|---|---|---|---|---|---|---|
| Mean ± SD | % Score Zero | % Maximum Score | Mean ± SD | % ScoreZero | % Maximum Score | ||
| Med (Min-Max) | Med (Min-Max) | ||||||
| Total fruit * | 0.9 ± 1.6 0.0 (0.0–5.0) | 66.7 | 6.3 | 1.9 ± 2.0 1.8 (0–5.0) | 45.1 | 13.7 | 0.011 |
| Whole fruit * | 0.8 ± 1.7 0.0 (0.0–5.0) | 77.1 | 10.4 | 1.3 ± 1.9 0.0 (0.0–5.0) | 62.7 | 9.8 | 0.155 |
| Total vegetable * | 2.8 ± 1.9 2.8 (0.1–5.0) | - | 25.0 | 3.5 ± 1.5 3.9 (0.0- 5.0) | 2.0 | 33.3 | 0.055 |
| Dark green and orange vegetables and legumes | 2.3 ± 1.8 2.0 (0.0–5.0) | 12.5 | 22.9 | 3.5 ± 1.7 4.4 (0.0–5.0) | 2.0 | 49.0 | <0.001 |
| Total grains * | 4.0 ± 0.9 4.1 (2.2–5.0) | - | 20.8 | 3.8 ± 0.9 3.9 (2.0–5.0) | - | 14.6 | 0.235 |
| Whole grains * | 0,0 ± 0,0 0.0 (0.0–0.0) | 100 | - | 0,0 ± 0,0 0.0 (0.0–0.0) | 100 | - | 1.000 |
| Dairy products * | 9.1 ± 1.7 10.0 (3.7–10.0) | - | 68.8 | 7.8 ± 2.8 9.3(1.9–10.0) | - | 43.1 | 0.006 |
| Meat, eggs and legumes * | 9.1 ± 1.8 10.0 (1.3–10.0) | - | 68.8 | 9.5 ± 1.1 10.0 (5.0–10.0) | - | 68.6 | 0.795 |
| Oil * | 6.2 ± 3.4 6.2 (0.2–10.0) | - | 33.3 | 9.3 ± 1.3 10.0 (5.0–10.0) | - | 70.6 | <0.001 |
| Saturated fat * | 6.4 ± 3.8 7.6 (0.0–10.0) | 16.7 | 25.0 | 7.1 ± 3.8 9.1 (0.0–10.0) | 11.8 | 35.3 | 0.255 |
| Sodium * | 5.3 ± 3.2 5.3 (0.0–10.0) | 6.3 | 2.1 | 1.3 ± 1.8 0.3 (0.0–6.3) | 45.1 | - | <0.001 |
| SoFAAS ** | 10.2 ± 5.1 10.3 (0.0–20.0) | 4.2 | 4.2 | 8.4 ± 5.2 8.2 (0.0–20.0) | 11.8 | 2.0 | 0.087 |
| Total score * | 57.2 ± 12.6 59.6 (34.0–77.2) | - | - | 57.3 ± 10.7 59.5 (34.6–75.0) | - | - | 0.936 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Lucia, C.M.; Rodrigues, K.C.C.; Rodrigues, V.C.C.; Santos, L.L.M.; Cardoso, L.M.; Martino, H.S.D.; Franceschini, S.C.C.; Pinheiro-Sant’Ana, H.M. Diet Quality and Adequacy of Nutrients in Preschool Children: Should Rice Fortified with Micronutrients Be Included in School Meals? Nutrients 2016, 8, 296. https://doi.org/10.3390/nu8050296
Della Lucia CM, Rodrigues KCC, Rodrigues VCC, Santos LLM, Cardoso LM, Martino HSD, Franceschini SCC, Pinheiro-Sant’Ana HM. Diet Quality and Adequacy of Nutrients in Preschool Children: Should Rice Fortified with Micronutrients Be Included in School Meals? Nutrients. 2016; 8(5):296. https://doi.org/10.3390/nu8050296
Chicago/Turabian StyleDella Lucia, Ceres M., Kellen Cristina C. Rodrigues, Vivian Cristina C. Rodrigues, Laura Luiza M. Santos, Leandro M. Cardoso, Hércia S. D. Martino, Sylvia C. C. Franceschini, and Helena Maria Pinheiro-Sant’Ana. 2016. "Diet Quality and Adequacy of Nutrients in Preschool Children: Should Rice Fortified with Micronutrients Be Included in School Meals?" Nutrients 8, no. 5: 296. https://doi.org/10.3390/nu8050296
APA StyleDella Lucia, C. M., Rodrigues, K. C. C., Rodrigues, V. C. C., Santos, L. L. M., Cardoso, L. M., Martino, H. S. D., Franceschini, S. C. C., & Pinheiro-Sant’Ana, H. M. (2016). Diet Quality and Adequacy of Nutrients in Preschool Children: Should Rice Fortified with Micronutrients Be Included in School Meals? Nutrients, 8(5), 296. https://doi.org/10.3390/nu8050296




