Cocoa Diet Prevents Antibody Synthesis and Modifies Lymph Node Composition and Functionality in a Rat Oral Sensitization Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Animals and Diets
2.3. Sample Collection and Processing
2.4. Determination of Total Polyphenol Content
2.5. IgA and Specific Anti-OVA Antibodies
2.6. Immunofluorescence Staining and Flow Cytometry Analysis
2.7. Gene Expression from MLN Lymphocytes
2.8. Cytokine Quantification
2.9. Statistical Analysis
3. Results
3.1. Food and Water Intake, Flavonoid Absorption and Body Weight
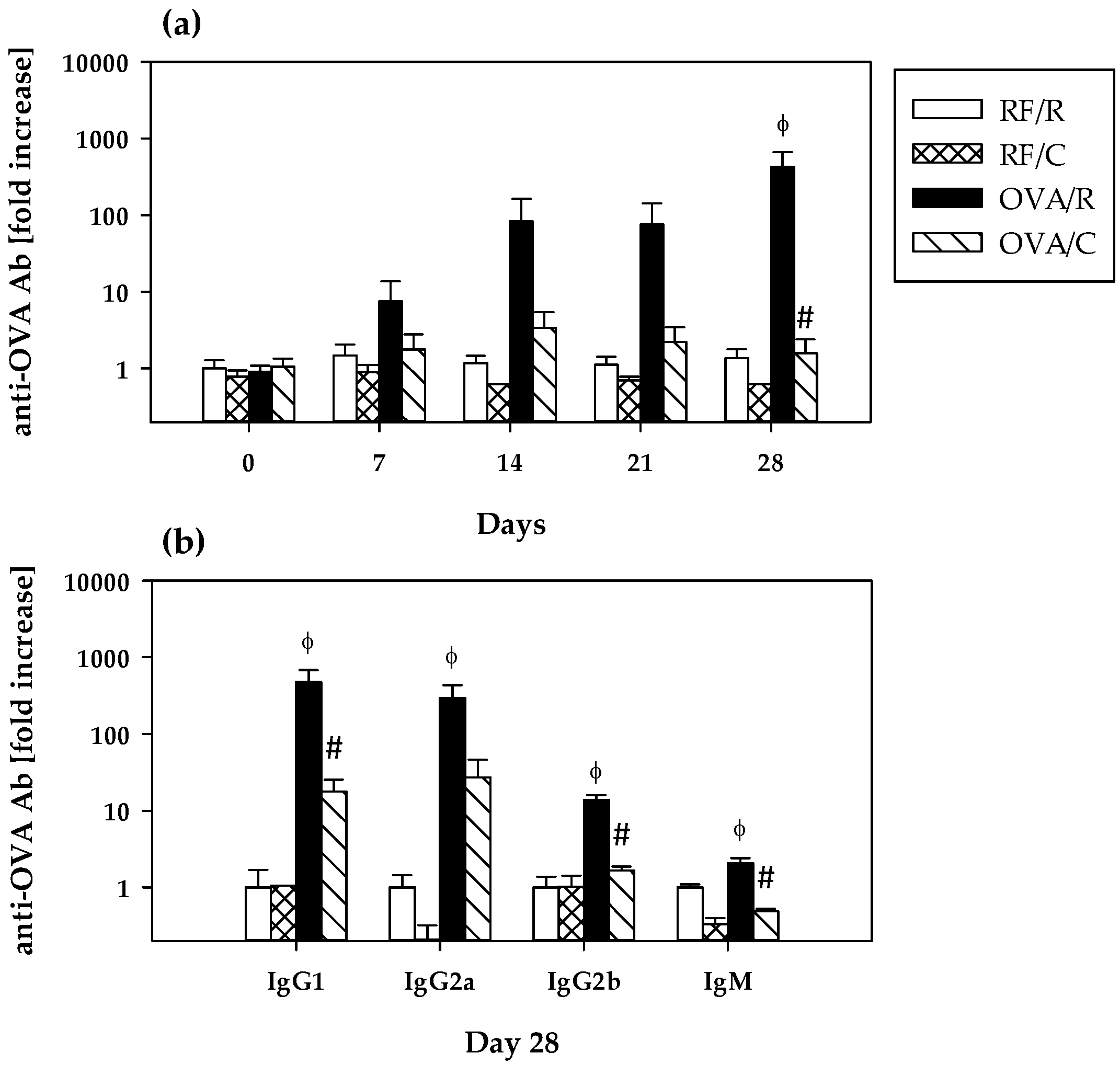
3.2. Immune Response to OVA: Serum Anti-OVA Antibodies
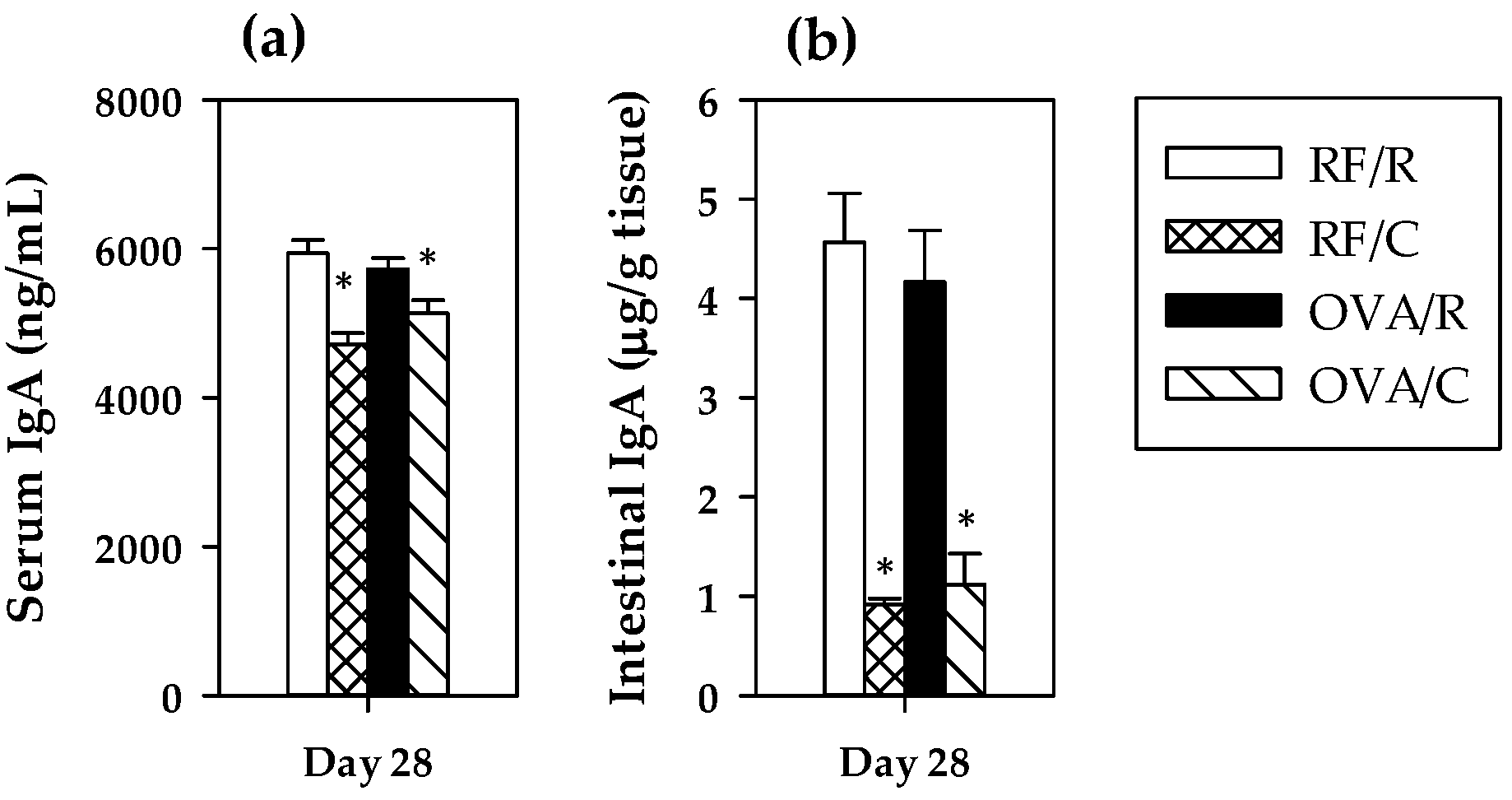
3.3. Total IgA Antibodies: Serum and Intestinal Concentrations
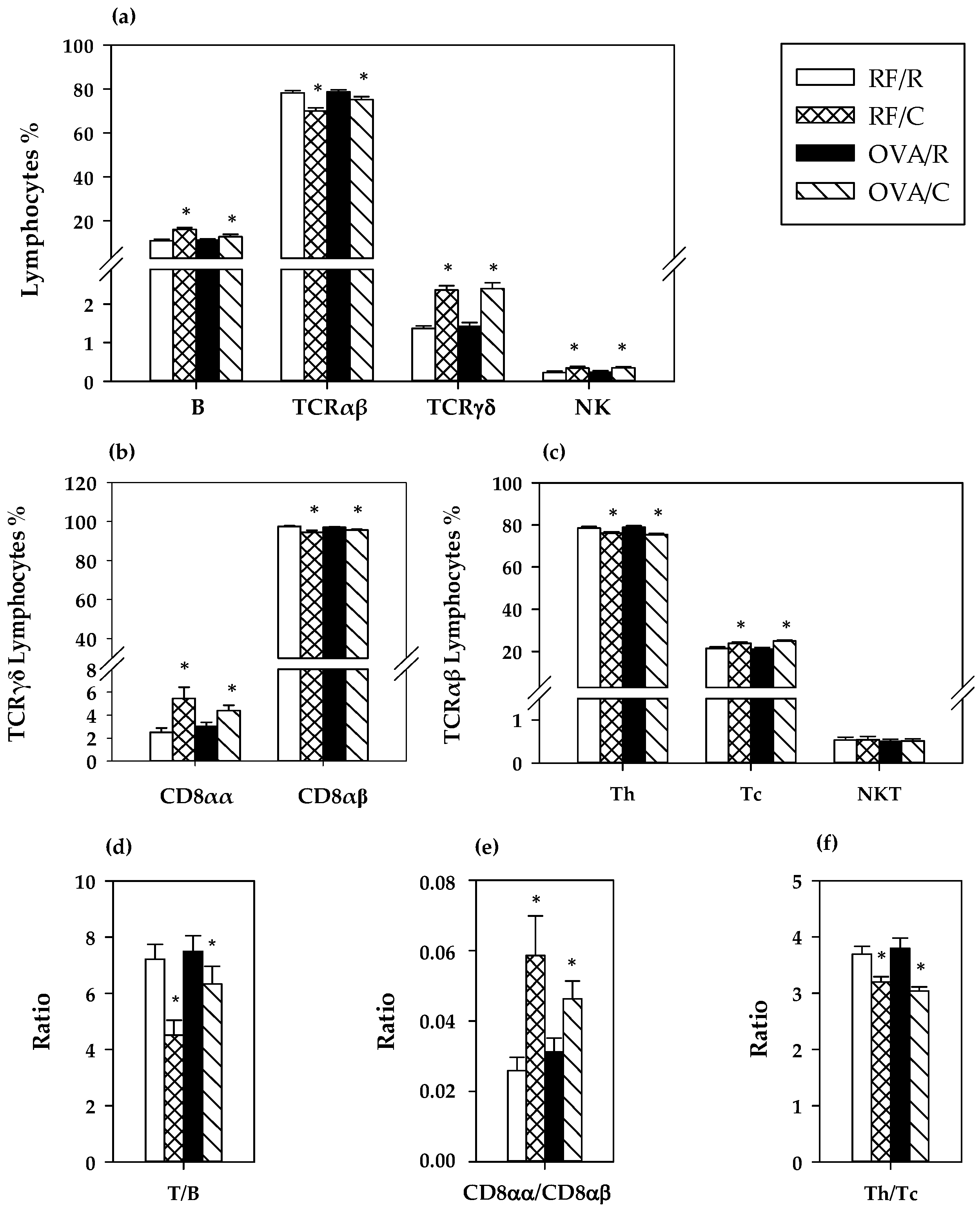
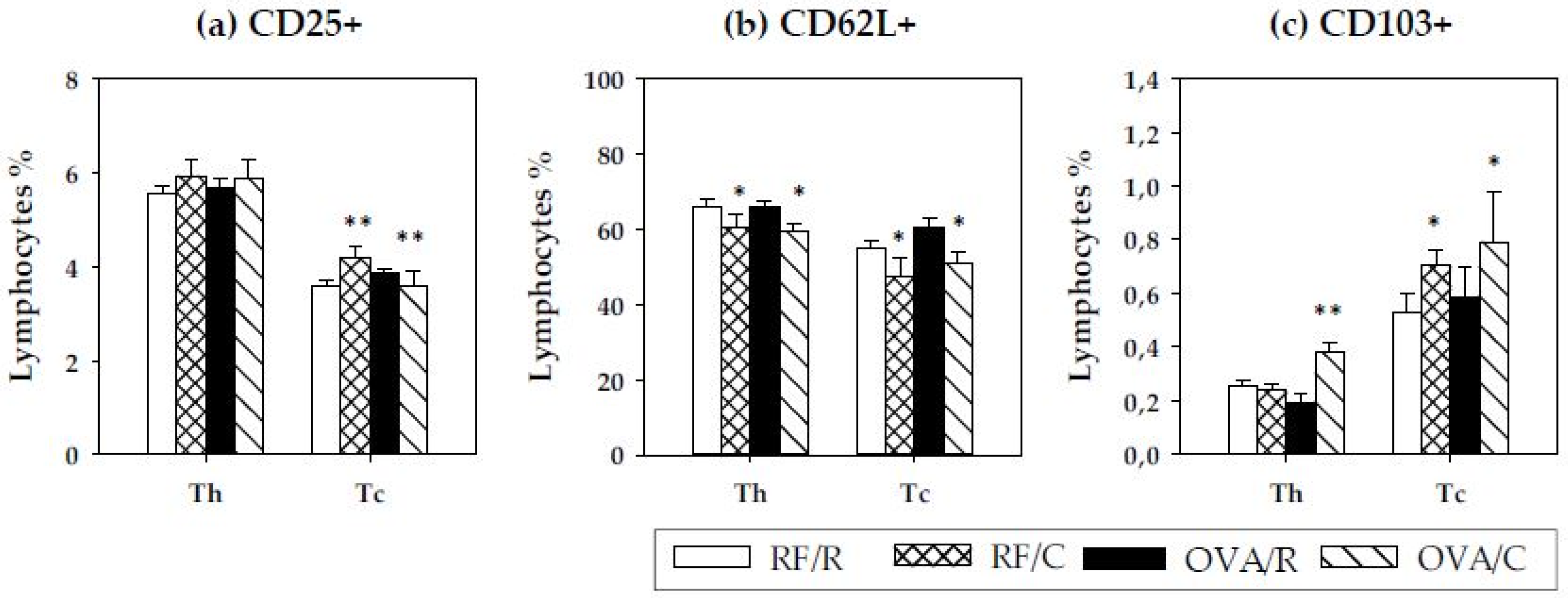
3.4. Lymphocyte Composition of MLN
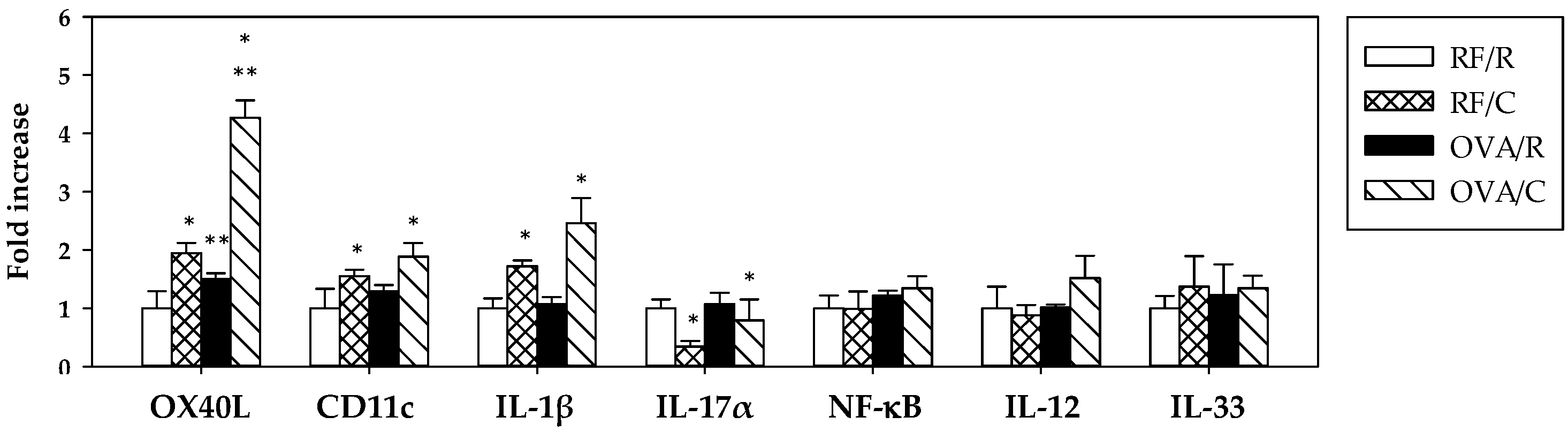
3.5. Gene Expression and Cytokine Production in MLN Cells
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| APC | allophycocyanin |
| BSA | albumin from bovine serum |
| CT | cholera toxin |
| DMEM | Dulbecco’s Modified Eagle Medium |
| ELISA | enzyme-linked immunosorbent assay |
| FBS | fetal bovine serum |
| FITC | fluorescein isothiocyanate |
| GALT | gut-associated lymphoid tissue |
| IFN | interferon |
| I | inventoried |
| Ig | immunoglobulin |
| IL | interleukin |
| MLN | mesenteric lymph nodes |
| NF | nuclear factor |
| OPD | o-phenylenediamine |
| OVA | ovalbumin |
| OVA/C | sensitized group fed cocoa diet |
| OVA/R | sensitized group |
| PBS | phosphate-buffered saline |
| PCR | polymerase chain reaction |
| PE | phycoerythrin |
| PerCP | peridininchlorophylla protein |
| RF/C | reference group fed cocoa diet |
| RF/R | reference group |
| Tc | T cytotoxic |
| TCR | T cell receptor |
| Th | T helper cells |
| TNF | tumor necrosis factor |
References
- Crown, P.L.; Hurst, W.J. Evidence of cacao use in the Prehispanic American Southwest. Proc. Natl. Acad. Sci. USA 2009, 106, 2110–2113. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and chocolate in human health and disease. Antioxid. Redox Signal. 2011, 15, 2779–2811. [Google Scholar] [CrossRef] [PubMed]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Cocoa polyphenols and their potential benefits for human health. Oxid. Med. Cell. Longev. 2012, 2012, 906252. [Google Scholar] [CrossRef] [PubMed]
- Bordiga, M.; Locatelli, M.; Travaglia, F.; Co, J.D.; Mazza, G. Evaluation of the effect of processing on cocoa polyphenols: Antiradical activity, anthocyanins and procyanidins profiling from raw beans to chocolate. Int. J. Food Sci. Technol. 2015, 50, 840–848. [Google Scholar] [CrossRef]
- Cooper, K.A.; Donovan, J.L.; Waterhouse, A.L.; Williamson, G. Cocoa and health: A decade of research. Br. J. Nutr. 2008, 99, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Latif, R. Chocolate/cocoa and human health: A review. Neth. J. Med. 2013, 71, 63–68. [Google Scholar] [PubMed]
- Stahl, L.; Miller, K.B.; Apgar, J.; Sweigart, D.S.; Stuart, D.A.; McHale, N.; Ou, B.; Kondo, M.; Hurst, W.J. Preservation of cocoa antioxidant activity, total polyphenols, flavan-3-ols, and procyanidin content in foods prepared with cocoa powder. J. Food Sci. 2009, 74, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Castell, M.; Perez-Cano, F.; Abril-Gil, M.; Franch, A. Flavonoids on Allergy. Curr. Pharm. Des. 2014, 20, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Hirano, T.; Higa, S.; Arimitsu, J.; Maruta, M.; Kuwahara, Y.; Ohkawara, T.; Hagihara, K.; Yamadori, T.; Shima, Y.; et al. Flavonoids and related compounds as anti-allergic substances. Allergol. Int. 2007, 56, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Pérez-Cano, F.J.; Ramírez-Santana, C.; Castellote, C.; Izquierdo-Pulido, M.; Permanyer, J.; Franch, A.; Castell, M. Spleen lymphocyte function modulated by a cocoa-enriched diet. Clin. Exp. Immunol. 2007, 149, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Pérez-Cano, F.J.; Ramos-Romero, S.; Pérez-Berezo, T.; Castellote, C.; Permanyer, J.; Franch, A.; Izquierdo-Pulido, M.; Castell, M. Intestinal immune system of young rats influenced by cocoa-enriched diet. J. Nutr. Biochem. 2008, 19, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Berezo, T.; Ramiro-Puig, E.; Pérez-Cano, F.J.; Castellote, C.; Permanyer, J.; Franch, A.; Castell, M. Influence of a cocoa-enriched diet on specific immune response in ovalbumin-sensitized rats. Mol. Nutr. Food Res. 2009, 53, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Abril-Gil, M.; Massot-Cladera, M.; Pérez-Cano, F.J.; Castellote, C.; Franch, A.; Castell, M. A diet enriched with cocoa prevents IgE synthesis in a rat allergy model. Pharmacol. Res. 2012, 65, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Sabra, A.; Bellanti, J.A.; Rais, J.M.; Castro, H.J.; Mendez de Inocencio, J.; Sabra, S. IgE and non-IgE food allergy. Ann. Allergy Asthma. Immunol. 2003, 90, 71–76. [Google Scholar] [CrossRef]
- Moriyama, T. Diversity of Food Allergy. J. Nutr. Sci. Vitaminol. 2015, 61, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Vighi, G.; Marcucci, F.; Sensi, L.; Di Cara, G.; Frati, F. Allergy and the gastrointestinal system. Clin. Exp. Immunol. 2008, 153, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, A.J.; Smith, K. Mesenteric lymph nodes at the center of immune anatomy. J. Exp. Med. 2006, 203, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Pawankar, R. Allergic diseases and asthma: A global public health concern and a call to action. World Allergy Organ. J. 2014, 7, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Sampson, H.A. Food allergy. J. Allergy Clin. Immunol. 2010, 125, S116–S125. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Abril-Gil, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Induction of an oral sensitization model in rats. Clin. Immunol. Endocr. Metab. Drugs 2014, 1, 89–101. [Google Scholar] [CrossRef]
- Berin, M.C.; Mayer, L. Immunophysiology of experimental food allergy. Mucosal Immunol. 2009, 2, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Flach, C.-F.; Lange, S.; Jennische, E.; Lönnroth, I.; Holmgren, J. Cholera toxin induces a transient depletion of CD8+ intraepithelial lymphocytes in the rat small intestine as detected by microarray and immunohistochemistry. Infect. Immun. 2005, 73, 5595–5602. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-J.; Kim, H.-A.; Seo, K.H.; Lee, H.-K.; Kang, B.Y.; Im, S.-Y. Cholera toxin breakdowns oral tolerance via activation of canonical NF-κB. Cell. Immunol. 2013, 285, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Nowak-Węgrzyn, A.; Sampson, H.A. Future therapies for food allergies. J. Allergy Clin. Immunol. 2011, 127, 558–573. [Google Scholar] [CrossRef] [PubMed]
- Syed, A.; Kohli, A.; Nadeau, K.C. Food allergy diagnosis and therapy: Where are we now? Immunotherapy 2013, 5, 931–944. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Franch, A.; Castellote, C.; Castell, M.; Pérez-Cano, F.J. Cocoa flavonoid-enriched diet modulates systemic and intestinal immunoglobulin synthesis in adult Lewis rats. Nutrients 2013, 5, 3272–3286. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Berezo, T.; Franch, A.; Ramos-Romero, S.; Castellote, C.; Pérez-Cano, F.J.; Castell, M. Cocoa-enriched diets modulate intestinal and systemic humoral immune response in young adult rats. Mol. Nutr. Food Res. 2011, 55, S56–S66. [Google Scholar] [CrossRef] [PubMed]
- Abril-Gil, M.; Garcia-Just, A.; Pérez-Cano, F.J.; Franch, À.; Castell, M. Effect of a cocoa-enriched diet on immune response and anaphylaxis in a food allergy model in Brown Norway rats. J. Nutr. Biochem. 2016, 27, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Abril-Gil, M.; Garcia-Just, A.; Pérez-Cano, F.J.; Franch, À.; Castell, M. Development and characterization of an effective food allergy model in Brown Norway rats. PLoS ONE 2015, 10, e0125314. [Google Scholar] [CrossRef] [PubMed]
- Bridle, B.W.; Wilkie, B.N.; Jevnikar, A.M.; Mallard, B.A. Deviation of xenogeneic immune response and bystander suppression in rats fed porcine blood mononuclear cells. Transpl. Immunol. 2007, 17, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Serebrisky, D.; Lee, S.Y.; Huang, C.K.; Bardina, L.; Schofield, B.H.; Stanley, J.S.; Burks, A.W.; Bannon, G.A.; Sampson, H.A. A murine model of peanut anaphylaxis: T- and B-cell responses to a major peanut allergen mimic human responses. J. Allergy Clin. Immunol. 2000, 106, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-Y.; Oh, S.; Lee, K.; Jang, Y.-J.; Sohn, M.-H.; Lee, K.-E.; Kim, K.-E. Murine model of buckwheat allergy by intragastric sensitization with fresh buckwheat flour extract. J. Korean Med. Sci. 2005, 20, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Foss, N.; Duranti, M.; Magni, C.; Frøkiaer, H. Assessment of lupin allergenicity in the cholera toxin model: Induction of IgE response depends on the intrinsic properties of the conglutins and matrix effects. Int. Arch. Allergy Immunol. 2006, 141, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Oh-oka, K.; Nakamura, Y.; Ishimaru, K.; Matsuoka, S.; Okumura, K.; Ogawa, H.; Hisamoto, M.; Okuda, T.; Nakao, A. Dietary resveratrol prevents the development of food allergy in mice. PLoS ONE 2012, 7, e44338. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Abril-Gil, M.; Torres, S.; Franch, À.; Castell, M.; Pérez-Cano, F.J. Impact of cocoa polyphenol extracts on the immune system and microbiota in two strains of young rats. Br. J. Nutr. 2014, 112, 1944–1954. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, M.C.; Sallusto, F.; Marinaro, M.; Vendetti, S.; Riccomi, A.; De Magistris, M.T. Effects of the adjuvant cholera toxin on dendritic cells: Stimulatory and inhibitory signals that result in the amplification of immune responses. Int. J. Med. Microbiol. 2002, 291, 571–575. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Berezo, T.; Franch, A.; Castellote, C.; Castell, M.; Pérez-Cano, F.J. Mechanisms involved in down-regulation of intestinal IgA in rats by high cocoa intake. J. Nutr. Biochem. 2012, 23, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Fujihashi, K.; Kato, H.; van Ginkel, F.W.; Koga, T.; Boyaka, P.N.; Jackson, R.J.; Kato, R.; Hagiwara, Y.; Etani, Y.; Goma, I.; et al. A revisit of mucosal IgA immunity and oral tolerance. Acta Odontol. Scand. 2001, 59, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, Y. Mucosal immunity and the onset of allergic disease. Allergol. Int. 2013, 62, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Pabst, O.; Mowat, A.M. Oral tolerance to food protein. Mucosal Immunol. 2012, 5, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Berezo, T.; Ramírez-Santana, C.; Franch, A.; Ramos-Romero, S.; Castellote, C.; Pérez-Cano, F.J.; Castell, M. Effects of a cocoa diet on an intestinal inflammation model in rats. Exp. Biol. Med. (Maywood) 2012, 237, 1181–1188. [Google Scholar] [CrossRef] [PubMed]
- Guy-Grand, D.; Cerf-Bensussan, N.; Malissen, B.; Malassis-Seris, M.; Briottet, C.; Vassalli, P. Two gut intraepithelial CD8+ lymphocyte populations with different T cell receptors: A role for the gut epithelium in T cell differentiation. J. Exp. Med. 1991, 173, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Bol-Schoenmakers, M.; Marcondes Rezende, M.; Bleumink, R.; Boon, L.; Man, S.; Hassing, I.; Fiechter, D.; Pieters, R.H.; Smit, J.J. Regulation by intestinal γδ T cells during establishment of food allergic sensitization in mice. Allergy 2011, 66, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Fujihashi, K.; Dohi, T.; Kweon, M.-N.; McGhee, J.R.; Koga, T.; Cooper, M.D.; Tonegawa, S.; Kiyono, H. γδT cells regulate mucosally induced tolerance in a dose-dependent fashion. Int. Immunol. 1999, 11, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Untersmayr, E.; Jensen-Jarolim, E. Mechanisms of type I food allergy. Pharmacol. Ther. 2006, 112, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Locke, N.R.; Stankovic, S.; Funda, D.P.; Harrison, L.C. TCRγδ intraepithelial lymphocytes are required for self-tolerance. J. Immunol. 2006, 176, 6553–6559. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Getahun, A.; Heiser, R.A.; Detanico, T.O.; Aviszus, K.; Kirchenbaum, G.A.; Casper, T.L.; Huang, C.; Aydintug, M.K.; Carding, S.R.; et al. γδ T cells shape preimmune peripheral B cell populations. J. Immunol. 2016, 196, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Deniz, G.; Erten, G.; Kücüksezer, U.C.; Kocacik, D.; Karagiannidis, C.; Aktas, E.; Akdis, C.A.; Akdis, M. Regulatory NK cells suppress antigen-specific T cell responses. J. Immunol. 2008, 180, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Romero, S.; Pérez-Cano, F.J.; Castellote, C.; Castell, M.; Franch, À. Effect of cocoa-enriched diets on lymphocytes involved in adjuvant arthritis in rats. Br. J. Nutr. 2012, 107, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Raffler, N.A.; Rivera-Nieves, J.; Ley, K. L-selectin in inflammation, infection and immunity. Drug Discov. Today Ther. Strateg. 2005, 2, 213–220. [Google Scholar] [CrossRef]
- Agace, W.W.; Higgins, J.M.; Sadasivan, B.; Brenner, M.B.; Parker, C.M. T-lymphocyte-epithelial-cell interactions: Integrin αE(CD103)β7, LEEP-CAM and chemokines. Curr. Opin. Cell Biol. 2000, 12, 563–568. [Google Scholar] [CrossRef]
- Sathaliyawala, T.; Kubota, M.; Yudanin, N.; Turner, D.; Camp, P.; Thome, J.J.C.; Bickham, K.L.; Lerner, H.; Goldstein, M.; Sykes, M.; et al. Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity 2013, 38, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Nandakumar, S.; Miller, C.W.; Kumaraguru, U. T regulatory cells: An overview and intervention techniques to modulate allergy outcome. Clin. Mol. Allergy 2009, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, A.B.; Berin, M.C. Gastrointestinal dendritic cells promote Th2 skewing via OX40L. J. Immunol. 2008, 180, 4441–4450. [Google Scholar] [CrossRef] [PubMed]
- Jember, A.G.-H.; Zuberi, R.; Liu, F.T.; Croft, M. Development of allergic inflammation in a murine model of asthma is dependent on the costimulatory receptor OX40. J. Exp. Med. 2001, 193, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Tang, Y.; Hu, X.; Wang, Q.; Lei, W.; Zhou, L.; Huang, J. Regulation of Th1/Th2 balance through OX40/OX40L signalling by glycyrrhizic acid in a murine model of asthma. Respirology 2015, 21, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Zubairi, S.; Sanos, S.L.; Hill, S.; Kaye, P.M. Immunotherapy with OX40L-Fc or anti-CTLA-4 enhances local tissue responses and killing of Leishmania donovani. Eur. J. Immunol. 2004, 34, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Ramiro, E.; Franch, À.; Castellote, C.; Pérez-Cano, F.; Permanyer, J.; Izquierdo-Pulido, M.; Castell, M. Flavonoids from Theobroma cacao down-regulate inflammatory mediators. J. Agric. Food Chem. 2005, 53, 8506–8511. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.C.; He, J.; Wu, C.Y.; Kelsall, B.L. Cholera toxin suppresses interleukin (IL)-12 production and IL-12 receptor beta1 and beta2 chain expression. J. Exp. Med. 1999, 189, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Saluja, R.; Khan, M.; Church, M.K.; Maurer, M. The role of IL-33 and mast cells in allergy and inflammation. Clin. Transl. Allergy 2015, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, H.; Koya, T.; Kagamu, H.; Kimura, Y.; Sakamoto, H.; Yamabayashi, C.; Furukawa, T.; Sakagami, T.; Miyabayashi, T.; Hasegawa, T.; et al. IL-17 eliminates therapeutic effects of oral tolerance in murine airway allergic inflammation. Clin. Exp. Allergy 2012, 42, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Demont, A.; Actis-Goretta, L.; Holvoet, S.; Lévêques, A.; Lepage, M.; Nutten, S.; Mercenier, A. Identification of epicatechin as one of the key bioactive constituents of polyphenol-enriched extracts that demonstrate an anti-allergic effect in a murine model of food allergy. Br. J. Nutr. 2014, 112, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Laouini, D.; Alenius, H.; Bryce, P.; Oettgen, H.; Tsitsikov, E.; Geha, R.S. IL-10 is critical for Th2 responses in a murine model of allergic dermatitis. J. Clin. Investig. 2003, 112, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Veenbergen, S.; Samsom, J.N. Maintenance of small intestinal and colonic tolerance by IL-10-producing regulatory T cell subsets. Curr. Opin. Immunol. 2012, 24, 269–276. [Google Scholar] [CrossRef] [PubMed]
| Group | Oral Administration Days 0, 2, 4, 7, 9, 11, 14, 16, 18, and 21 | Diet |
|---|---|---|
| RF/R (n = 9) | Vehicle (1 mL/rat) | AIN-93M |
| RF/C (n = 9) | Vehicle (1 mL/rat) | 10% cocoa |
| OVA/R (n = 9) | 50 mg OVA + 30 µg CT/rat | AIN-93M |
| OVA/C (n = 9) | 50 mg OVA + 30 µg CT/rat | 10% cocoa |
| Components | Reference Diet | 10% Cocoa Diet | |
|---|---|---|---|
| AIN-93M (g/kg Diet) | Basal Mix (g/kg Diet) | Cocoa Powder (g/kg Diet) | |
| Carbohydrates | 721.9 | 692.5 | 16.8 |
| Proteins | 140.8 | 118.2 | 23.1 |
| Lipids | 38.7 | 27 | 11.5 |
| Fiber | 50 | 24.5 | 35.6 |
| Micronutrients | 48.6 | 37.8 | 6.3 |
| Flavonoids 1 | 0 | 0 | 4.02 |
| Theobromine | 0 | 0 | 2.5 |
| Total | 1000 | 1000 | |
| Day | Variable | Group 1 | |||
|---|---|---|---|---|---|
| RF/R | RF/C | OVA/R | OVA/C | ||
| 0 | Body weight | 59.9 ± 4.27 | 59.78 ± 4.58 | 60.86 ± 4.24 | 58.62 ± 4.16 |
| 0–7 | Food intake | 10.38 ± 2.35 | 13.46 ± 1.12 | 9.72 ± 2.50 | 13.43 ± 0.82 |
| 0–7 | Water intake | 12.06 ± 0.17 | 23.85 ± 4.03 | 12.06 ± 1.81 | 23.04 ± 3.04 |
| 7 | Body weight | 92.7 ± 6.05 | 82.28 ± 5.30 * | 94.00 ± 5.88 | 80.90 ± 4.80 * |
| 7–14 | Food intake | 11.65 ± 0.97 | 13.05 ± 0.51 | 11.72 ± 1.04 | 12.77 ± 0.33 |
| 7–14 | Water intake | 9.68 ± 0.90 | 21.08 ± 3.11 | 10.68 ± 0.65 | 22.64 ± 2.10 |
| 14 | Body weight | 129.04 ± 5.96 | 107.82 ± 5.84 * | 129.03 ± 6.06 | 109.35 ± 4.53 * |
| 14–21 | Food intake | 9.63 ± 1.03 | 11.12 ± 0.95 | 9.07 ± 0.92 | 11.24 ± 0.12 |
| 14–21 | Water intake | 9.12 ± 0.26 | 17.26 ± 0.09 | 9.13 ± 0.38 | 21.39 ± 0.99 |
| 21 | Body weight | 154.29 ± 3.72 | 133.80 ± 5.24 * | 152.73 ± 5.67 | 135.55 ± 4.63 * |
| 21–28 | Food intake | 6.79 ± 1.86 | 9.74 ± 0.51 | 6.82 ± 1.99 | 8.8 ± 1.36 |
| 21–28 | Water intake | 9.11 ± 0.10 | 14.99 ± 0.53 | 10.24 ± 0.96 | 25.45 ± 4.04 |
| 28 | Body weight | 174.13 ± 3.23 | 153.32 ± 5.67 * | 171.61 ± 4.35 | 150.53 ± 2.57 * |
| Sample | Cytokine | Group 1 | |||
|---|---|---|---|---|---|
| RF/R | RF/C | OVA/R | OVA/C | ||
| MLN | IFN-γ | 1.000 ± 0.110 | 1.484 ± 0.661 | 2.158 ± 0.843 | 1.521 ± 0.323 |
| MLN | IL-4 | 1.000 ± 0.080 | 0.807 ± 0.113 | 0.895 ± 0.083 | 0.768 ± 0.110 |
| MLN | TNF-α | 1.000 ± 0.065 | 0.860 ± 0.040 | 1.033 ± 0.087 | 0.901 ± 0.089 |
| MLN | IL-10 | 1.000 ± 0.042 | 1.106 ± 0.133 | 1.584 ± 0.302 | 1.241 ± 0.033 |
| gut lavage | IFN-γ | N.D. | N.D. | N.D. | N.D. |
| gut lavage | IL-4 | N.D. | N.D. | N.D. | N.D. |
| gut lavage | TNF-α | N.D. | N.D. | N.D. | N.D. |
| gut lavage | IL-10 | 1.000 ± 0.586 | 17.512 ± 3.783 # | 25.177 ± 3.151 ** | 17.060 ± 3.739 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camps-Bossacoma, M.; Abril-Gil, M.; Saldaña-Ruiz, S.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Cocoa Diet Prevents Antibody Synthesis and Modifies Lymph Node Composition and Functionality in a Rat Oral Sensitization Model. Nutrients 2016, 8, 242. https://doi.org/10.3390/nu8040242
Camps-Bossacoma M, Abril-Gil M, Saldaña-Ruiz S, Franch À, Pérez-Cano FJ, Castell M. Cocoa Diet Prevents Antibody Synthesis and Modifies Lymph Node Composition and Functionality in a Rat Oral Sensitization Model. Nutrients. 2016; 8(4):242. https://doi.org/10.3390/nu8040242
Chicago/Turabian StyleCamps-Bossacoma, Mariona, Mar Abril-Gil, Sandra Saldaña-Ruiz, Àngels Franch, Francisco J. Pérez-Cano, and Margarida Castell. 2016. "Cocoa Diet Prevents Antibody Synthesis and Modifies Lymph Node Composition and Functionality in a Rat Oral Sensitization Model" Nutrients 8, no. 4: 242. https://doi.org/10.3390/nu8040242
APA StyleCamps-Bossacoma, M., Abril-Gil, M., Saldaña-Ruiz, S., Franch, À., Pérez-Cano, F. J., & Castell, M. (2016). Cocoa Diet Prevents Antibody Synthesis and Modifies Lymph Node Composition and Functionality in a Rat Oral Sensitization Model. Nutrients, 8(4), 242. https://doi.org/10.3390/nu8040242








